Abstract
This article describes acute toxicity data in cynomolgus monkeys following oral treatment with vildagliptin, a dipeptidyl peptidase-4 inhibitor. Acute toxicity symptoms in cynomolgus monkeys include edema formation of the extremities, tails, and face associated with skeletal muscle necrosis, and elevations of lactate dehydrogenase, creatine kinase, alanine transaminase, and aspartate aminotransferase activities in the serum; hypothermia; hypotension; tachycardia; moribundity; and death in a few isolated instances. In surviving animals, symptoms were reversible even if treatment was continued. Cynomolgus monkeys from Mauritius appear more sensitive than monkeys of Asian origin. The underlying mechanism(s) of these symptoms in cynomolgus monkeys is currently not well understood, although a vascular mechanism including initial vasoconstriction and subsequent vascular leakage in distal extremities may play a role. The monkey data are reviewed and discussed in the context of other preclinical and clinical data, and it is concluded that acute toxicity following vildagliptin treatment is a monkey-specific phenomenon without relevance for humans.
Keywords
Dipeptidyl peptidase (DPP)-4 belongs to a family of postproline serine peptidases that includes DPP-8, DPP-9, fibroblast activation protein, and attractin (Sedo and Malik 2001; Chen, Kelly, and Ghersi 2003). Inhibitors of DPP-4 represent a novel class of drugs for the treatment of type 2 diabetes. The desired pharmacologic action of DPP-4 inhibitors results in the decrease in metabolism of incretin hormones, for example, glucagon-like peptide-1 and glucose-dependent insulinotropic peptide. Vildagliptin is a novel, orally bioavailable DPP-4 inhibitor (Villhauer et al. 2003). In a previous article, we described skin lesions in Asian cynomolgus monkeys that developed after 3 weeks of treatment with vildagliptin (Hoffmann et al. 2014). Evidence was provided that skin effects in monkeys are of vascular origin and that effects on the neuropeptide Y system, in combination with increased peripheral sympathetic tone, play an important pathomechanistic role in the pathogenesis of cutaneous toxicity in monkeys.
Based on the feedback from the Food and Drug Administration, Novartis was advised that DPP-4 inhibitors, at high doses, could induce acute responses in monkeys that include peripheral edema and elevations of lactate dehydrogenase (LDH) and creatine kinase (CK) activity in the serum. The present article describes an acute toxicity syndrome in cynomolgus monkeys following oral treatment with vildagliptin. Cynomolgus monkeys from Mauritius appear more sensitive than monkeys of Asian origin. Possible mechanisms of the manifestation of the acute toxicity syndrome were investigated including the role of impurities and degradation products. This article describes the results of 6 separate studies performed in cynomolgus monkeys (Macaca fascicularis) of Asian or Mauritian origin (see Table 1).
Studies with Vildagliptin in Cynomolgus Monkeys.

Note. CK = creatine kinase; LDH = lactate dehydrogenase; ALT = alanine transaminase; AST = aspartate aminotransferase; NOAEL = no observable adverse effect level.
Materials and Methods
Animals
The Asian and Mauritian monkeys were obtained from Covance (Denver, PA, or Alice, TX), Worldwide Primate (Miami, FL), or Rhenos/Pacific Forest Resources (Chestnut Hill, MA, via Buckshire Corporation via New Iberia Research Center, LA). The captive bred animals were between 2 and 7 years of age and weighed between 1.8 and 5.6 kg at the initiation of the studies. Animals were singly housed in primate cages throughout the duration of the studies (room temperature 68–76°F, 30–70% relative humidity, and 12-hr light/12-hr dark cycle). Certified Primate Diet #5048 (PMI Feeds, Inc., Richmond, IN) was supplemented with fresh fruit daily. The food ration (12 biscuits for males and 10 biscuits for females) and water from automatic dispensers of the animal drinking supply were available ad libitum. Experiments were conducted in accordance with Novartis animal care regulations and governmental guiding principles in the use of animals.
Treatment with vildagliptin
Vildagliptin was administered daily for the indicated periods of time via oral gavage as a solution in purified water (United States Pharmacopeia) to male and female cynomolgus monkeys (Table 1). Different batches of vildagliptin were investigated to test whether impurities/degradation products cause acute toxicity. An ultrapure batch of vildagliptin was prepared with intensive parts per million search for impurities. “Stressed” vildagliptin material was also used, that is, vildagliptin was dissolved in water and maintained at room temperature for 5 days to force formation of degradation products.
Cardiovascular Effects
Animals have previously been surgically implanted with telemetry transmitter (DSI D70-PCTP, St. Paul, MN). The telemetry transmitter body was implanted into the abdominal cavity wall in order to monitor core body temperature. To monitor arterial blood pressure, a pressure catheter was inserted into the femoral artery, and the tip of the catheter was further advanced to the abdominal aorta just below the renal arteries. The electrocardiogram (ECG) electrodes (lead II) were tunneled subcutaneously to the left (approximately 10th or 11th intercostal space) and right region (at or about the area of the thoracic inlet) of the thoracic cavity and sutured in place using the appropriate suture material. The telemetry parameters including blood pressure, heart rate, lead II ECG, and core body temperature were recorded continuously from approximately 1.5 hr prior to dosing (baseline) to 24-hr post dose. The hemodynamic parameters, ECG intervals (P duration, PR, R-R, QRS, and QT intervals), and body temperature were measured and reported as 15-min average values through 24-hr post dose.
Clinical Pathology Parameters
Approximately 1.5 ml blood were collected from the femoral vein via needle and syringe and transferred to a serum collection tube. After centrifugation (3,000 rpm for 10 min), approximately 200 μl of serum were transferred into a cryogen tube and stored frozen at approximately −20°C or below. The serum was used for the analyses of all clinical chemistry parameters. When necessary, serum was stored refrigerated up to 3 days at 2 to 8°C until analysis (e.g., CK isoenzymes). Automated methodologies for clinical chemistry were employed (Hitachi 917; Software version: # 02-49; Boehringer Mannheim/Hitachi 917 Operator’s Manual). Reagents were Roche Diagnostics kits (Roche Diagnostics; Indianapolis, IN).
Troponin I and Troponin T tests were performed at Ani Lytics, Inc. (Gaithersburg, MD), using an electrochemiluminescent immunoassay (BioVeris, Inc., Gaithersburg, MD)
Pathology
Complete necropsies were performed recording macroscopic abnormalities for all protocol tissues. Animals were anesthetized with an intramuscular injection of a mixture of ketamine HCL plus xylazine HCL and euthanized by induction of deep anesthesia with a sodium pentobarbital based intravenous solution followed by exsanguination. Representative samples of the protocol tissues listed (66 tissues) were collected in studies 1, 2, 3, and 5. Fixation and storage of specimens were in 10% neutral-buffered formalin, except for the initial fixation of testes and epididymides (Bouin’s) and eyes (3% Sorensen’s buffered glutaraldehyde). All tissue sections processed were examined and assessed with all observations being recorded in the raw data. All assessments were peer reviewed by another pathologist.
Toxicokinetics
Measurement of vildagliptin concentrations in plasma was performed using liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS) as described recently (Hoffmann et al. 2014). In brief, sample extraction was performed by solid phase extraction of the plasma samples on 10-mg Oasis 96-well extraction plates. The extracted samples were eluted from reverse phase columns using isocratic elution conditions. The detection was performed on API4000 mass spectrometers under positive mode. The transition monitored was from m/z 304 → 154. The lower limit of quantification was 0.1 ng/ml.
Results
Acute Toxicity Following Vildagliptin Administration Inhibition (Studies 1 and 2 in Table 1)
In study 1, cynomolgus monkeys from the Asian mainland were used. A high dose of 160 mg/kg vildagliptin was found to cause a rapid onset (after single dose) of edema and reddening of the distal extremities, tail and face, a decrease in body temperature, inappetence, and moribundity. The acute edema was accompanied by a decrease in serum protein concentrations and marked and transient increases in the activities of LDH, ALT, AST, and CK isoenzymes in serum. Extreme elevations of serum CK activity were observed in some animals (exceeding 40,000 U/L). Skeletal muscle damage was the cause of the elevations in serum CK activity in affected animals, supported by the preponderance of CK3 (MM isoenzyme) activity. There was no cardiac contribution to elevated CK levels, as CK2 activity was minimal and cardiac Troponin I and T concentrations were normal. A decrease in red blood cell count was observed in the 2 animals that were most severely affected and euthanized on days 3 and 5. No effects on serum electrolyte levels were seen.
The 2 most severely affected animals (1 male and 1 female) received veterinary intervention (including fluid therapy and pain medication) without improving their condition. The male monkey was treated intravenously with the antihistamine diphenhydramine, which did not lessen the clinical symptoms. The 2 most severely affected animals were euthanized for humane reasons during the 2-week dosing period (1 after a single dose and 1 after 3 doses). In the euthanized animals, skeletal muscle necrosis in the forearm, hand, and feet and intramuscular hemorrhage were seen histologically together with nonspecific inflammation and infiltration of mononuclear, lymphoid/neutrophilic cells. Skeletal muscles from the psoas and diaphragm were not affected, and no cardiac lesions were observed light microscopically.
Vildagliptin-induced acute effects were transient even with continued treatment. In surviving animals at necropsy after 2 weeks’ treatment, swelling/edema was no longer present and elevated enzyme activities had returned to normal. There were also no histological changes in skeletal muscles of the surviving animals.
It should be noted that in study 1 and in following studies reporting acute toxicity symptoms, there was a considerable interanimal variability in the acute response to vildagliptin treatment. In study 1, one animal had no adverse reactions at the dose of 160 mg/kg during a 2-week treatment period, while other (5 of 6) animals treated with the same dose had acute adverse effects of varying severity following 1 or more doses, and 2 animals had to be euthanized due to severe moribundity (Table 2). Table 2 also compares acute toxicity symptoms in individual animals with vildagliptin plasma exposures on day 2. There was no correlation between occurrence/severity of acute toxicity symptoms and plasma exposure. Indeed, the animal with the highest vildagliptin plasma concentration did not show acute toxicity symptoms at all during the 2-week treatment period. Of relevance, exposure at 160 mg/kg in study 1 was not higher than in the previous 13-week study at the same dose that did not produce acute toxicity (Hoffmann et al. 2014).
Acute Toxicity Symptoms (Edema Formation) in Individual Animals of Study 1 Compared with Vildagliptin Plasma Concentration after 160 mg/kg.
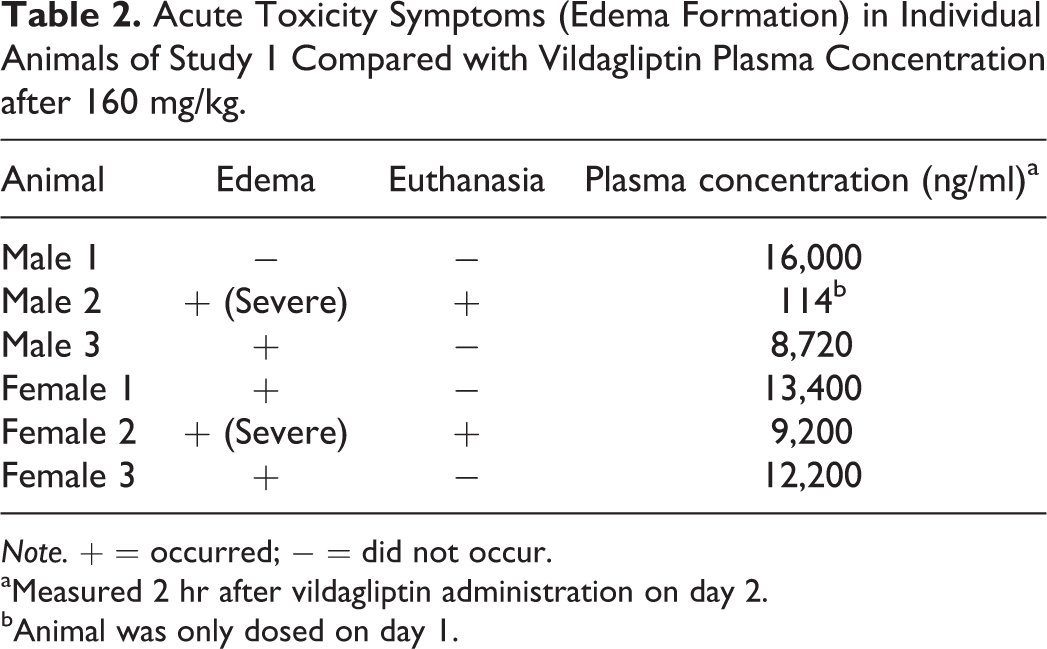
Note. + = occurred; − = did not occur.
aMeasured 2 hr after vildagliptin administration on day 2.
bAnimal was only dosed on day 1.
Since acute toxicity following vildagliptin administration had not been observed in previous cynomolgus monkey studies (Hoffmann et al. 2014), a study was initiated to see whether the acute toxicity syndrome could be reproduced. Study 2 was performed in another laboratory (Charles River Laboratories Preclinical Services, Senneville, Quebec, Canada) with planned doses of 20, 40, and 80 mg/kg to identify the no observable adverse effect level (NOAEL). A staggered dose administration design was used, that is, on day 1, only 1 male and 1 female monkey were treated with the low dose of 20 mg/kg followed by another male and female monkey of the low-dose group on study days 2 to 5. Due to the toxicity observed on day 1, the planned doses of 40 and 80 mg/kg were not given. Administration of vildagliptin at the low-dose level of 20 mg/kg resulted in adverse clinical signs such as swelling of the extremities and increases in CK, LDH, ALT, and AST activities. Consequently, an NOAEL was not determined in this study. No microscopic skeletal muscle changes were observed at the end of the 5-day treatment period. Since this study was performed in monkeys from Mauritius, the question arises whether monkeys from Mauritius are more sensitive compared to Asian monkeys.
Role of Batch Impurities and Degradation Products (Studies 3–5 in Table 1)
The purpose of study 3 was to compare the effects of 2 batches of vildagliptin that were produced using different synthetic routes and consequently exhibiting different impurity profiles. Batch 1 was the batch in studies 1 and 2, and batch 2 was synthesized using a different synthetic route. The initial intent of the study was to administer the 2 vildagliptin batches at doses of 80 mg/kg for 5 days to Mauritian monkeys (n = 1/sex/dose). Due to mortality and other severe adverse signs, the study design was amended to a single dose. Administration of a single dose of 80 mg/kg (batch 1) resulted in the death of the female monkey within 8-hr post dose. Skeletal myofiber degenerations in hypothenar muscles with accompanying acute hemorrhages were observed microscopically.
Clinical signs of swollen limbs and/or muzzle, cold to the touch, and/or decreased locomotor activity were noted on one or more days in all 3 surviving animals administered a single 80 mg/kg dose of vildagliptin (batches 1 and 2). In the surviving animal treated with batch 1 and the female treated with batch 2, changes in clinical pathology parameters were seen on days 2 to 5. They reflected the presence of muscle damage, characterized by increased serum activity of CK, with lesser contributions from AST, ALT, and LDH.
Study 4 was conducted in an attempt to determine whether a degradation product(s) may be responsible for the acute toxicity noted in cynomolgus monkeys. Two fractions (early and late fraction of the vildagliptin peak separated by chromatography) of stressed vildagliptin (forced formation of degradation products by maintaining vildagliptin in solution at room temperature for 5 days) were administered to Asian monkeys at doses of 160 mg/kg for 2 days (n = 2/sex/group). No mortality was observed. No adverse clinical signs were noted in animals administered the early fraction or the males administered the late fraction. One female administered the late fraction had swollen extremities on days 2 to 6 after treatment and a moderate increase in the serum activities of muscle related enzymes (CK, ALT, AST, and LDH). Vildagliptin plasma exposure in study 4 was comparable to exposures in previous experiments at 160 mg/kg (data not shown).
In study 5, the potential role of batch impurities in the manifestation of acute toxicity symptoms was further investigated. An ultrapure batch was prepared, and doses of 20 or 40 mg/kg were planned to be administered for 5 days (n = 3/sex/group). The initial purpose of this study was to determine the NOAEL for clinical signs of edema in Mauritian monkeys. However, due to the adverse effects noted on day 1, no further dosing occurred and the animals were evaluated for 15 additional days. Following a single dose of 20 mg/kg vildagliptin ultrapure, 1 male animal presented with slight edema of the extremities on day 2. Following a single dose of 40 mg/kg of ultrapure vildagliptin, 1 male was found dead, and 1 male was euthanized in moribund condition within 10-hr post dose. Prior to death/moribund euthanization, these animals had swollen extremities and muzzles, decreased motor activity, were cold to the touch, and/or became recumbent. The remaining male administered 40 mg/kg had swollen extremities on day 1 but was asymptomatic by day 2. The 3 females administered the 40-mg/kg dose of vildagliptin ultrapure were less severely affected, with 1/3 presenting with swollen extremities on day 1. The remaining 2 females administered 40 mg/kg had transiently swollen extremities on days 1 to 4.
Changes indicative of moderate to severe skeletal muscle damage (increased CK activity with varying contributions by LDH, AST, and ALT) were seen in both dose groups. However, the 2 high-dose males that died or were euthanized on study day 1 did neither show histological signs of skeletal muscle damage nor increased skeletal muscle enzyme activities, suggesting that these animals succumbed prior to sufficient muscle membrane injury to allow enzyme leakage. In this study, all animals that developed peripheral edema had minimal to mild decreases in serum albumin concentrations. Decreases in serum albumin are most likely attributable to leakage from the vasculature associated with increased vessel permeability, although proteinuria cannot be excluded, since urinalysis was not performed in this study. Based on the results of this study, an NOAEL was not established.
Summarizing the results from studies 3, 4, and 5, it has been concluded that there was no apparent association between the impurity/degradation profiles of different vildagliptin batches and the observed acute toxicities in monkeys. Vildagliptin itself with a negligible amount of impurities can also cause acute toxicity.
Acute Cardiovascular Effects (Study 6 in Table 1)
A telemetry study (study 6) was performed to investigate acute cardiovascular effects after administration of a high vildagliptin dose. Four male Asian monkeys were treated with single doses of 160 mg/kg of stressed vildagliptin. In 1 animal, acute toxicity symptoms (edema formation and recumbency) started to develop 2.5 hr after vildagliptin administration. The clinical signs were preceded by a transient slight increase in blood pressure and a transient reduction of core body temperature (Figure 1). Observed cardiac effects prior to edema formation were a transient occurrence of premature ventricular complexes and tall T waves (ECG not shown in Figure 1). Effects on cardiovascular parameters and body temperature reversed transiently. But 4.5 hr after vildagliptin administration, body temperature started to decrease dramatically associated with some fluctuations and eventually a sharp drop of blood pressure and an increase in heart rate. Of note, an increased heart rate was observed during a period (∼3 to 6-hr post dose) when blood pressure was augmented, that is, the increase in heart rate was not a reflex tachycardia due to hypotension. No cardiac arrhythmias were observed during this period. The animal was euthanized 7 hr after dosing. Cardiac contractility was not determined in this study.
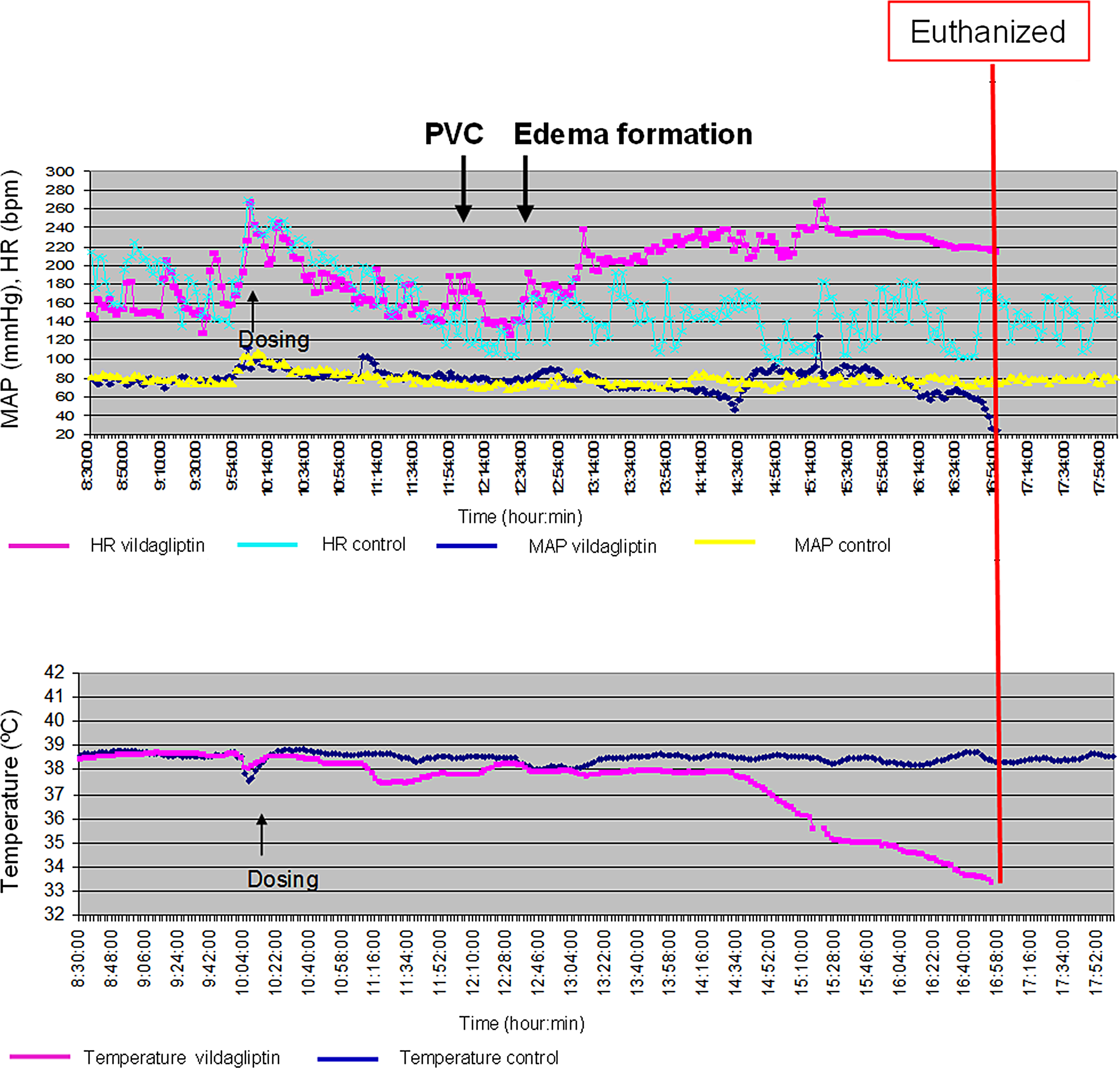
Effects of vildagliptin treatment on cardiovascular parameters in a cynomolgus monkey treated with 160 mg/kg. HR = heart rate; MAP = mean arterial pressure; PVC = premature ventricular complexes.
Discussion
Acute Toxicity Syndrome
Symptoms of acute toxicity in cynomolgus monkeys following vildagliptin treatment include moribundity; edema formation in distal extremities and muzzles associated with skeletal muscle necrosis, and elevations of LDH, CK, ALT, and AST activities in serum; hypothermia; hypotension; tachycardia; and death in some instances. The time course of the occurrence of these symptoms is shown in Figure 2. The symptoms are reversible in surviving animals even if treatment is continued.
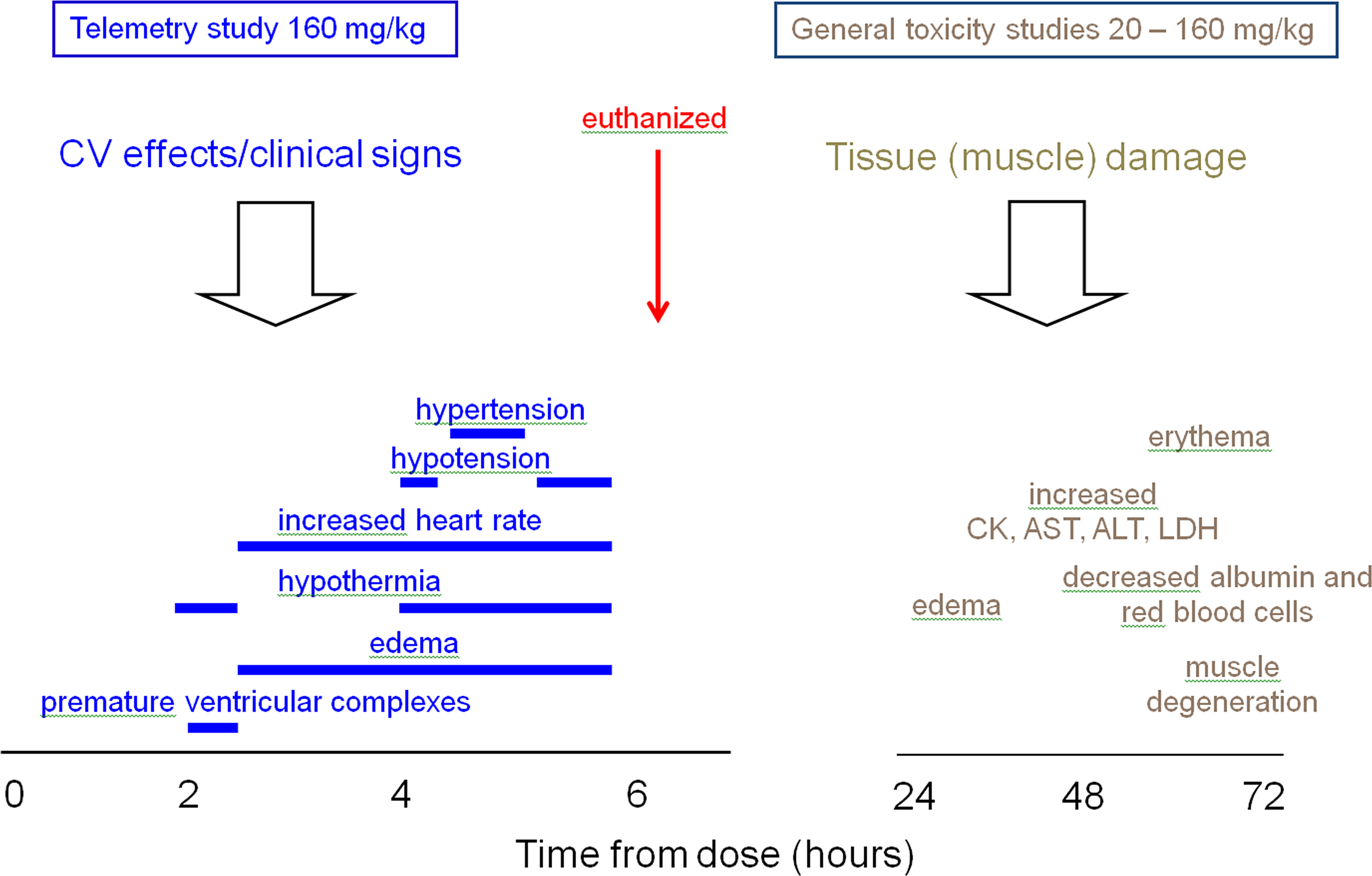
Time course of transient acute toxicity symptoms in monkeys.
It is currently not clear why this acute toxicity syndrome was not observed in previous cynomolgus monkey studies with vildagliptin (Hoffmann et al. 2014) using similar doses that produced comparable vildagliptin plasma exposures. It should be noted, however, that in the present studies, there was a considerable interanimal variability in the occurrence and severity of acute toxicity symptoms. In previous studies, only cynomolgus monkeys of Asian origin were used, and the high dose of 160 mg/kg was only used once in a toxicity study.
We initially postulated the hypothesis that degradation products or impurities may be involved in the manifestation of acute toxicity. Consequently, batches with different degradation products and impurities were investigated as well as an ultrapure vildagliptin batch. Taken together, these experiments suggest that there is no apparent association between the degradation/impurity profiles of the batches and observed acute toxicities in monkeys. Therefore, it is concluded that acute toxicity symptoms in monkeys are caused by vildagliptin itself.
Mechanistic Considerations
The etiology of the acute toxicity symptoms in cynomolgus monkeys remains uncertain. Necropsy and histological evaluations did not provide clear mechanistic insight. Consistent across studies, a transient elevation of serum enzyme activities (CK, LDH, ALT, and AST) was observed during the development of the acute toxicity. This enzyme activity increase is caused by damaged skeletal muscle cells, whereas cardiomyocytes were not affected as indicated by the CK isozyme pattern and normal cardiac Troponin I and T levels in animals showing acute toxicity. Moreover, there were no cardiac lesions observed microscopically.
We considered the hypothesis that massive skeletal muscle damage may have released intracellular potassium causing hyperkalemia which in turn would impair the electrophysiological function of excitable cells in the cardiovascular and central nervous systems. However, serum potassium concentrations were not increased in severely affected animals (study 1) and life-threatening cardiac arrhythmias were not seen in a telemetered animal that died (study 6). It is also important to note that 2 Mauritian monkeys treated with 40 mg/kg vildagliptin showed edema formation of the extremities and died or were sacrificed in moribund condition without increased skeletal muscle enzymes (study 5), suggesting that these animals succumbed prior to severe skeletal muscle membrane injury and that edema formation in the extremities is not due to massive skeletal muscle damage.
There is no evidence that vildagliptin is myotoxic. It is therefore plausible to assume that skeletal muscle damage is caused by an acute initial vasoconstrictory mechanism. This means impaired vasomotor or, more general, disturbed vascular function would be the common underlying mechanism for skeletal muscle lesions described in the present article and previously reported skin lesions in distal extremities (Hoffmann et al. 2014). Vasogenic edema is characterized by an extravasation of fluid due to increased permeability of capillary endothelial cells to macromolecular serum proteins, for example, albumin. Normally, the entry of plasma protein–containing fluid into the extracellular space is limited by tight endothelial cell junctions, but in the presence of massive injury, there is increased permeability of capillary endothelial cells to fluid, large molecules, and potentially even corpuscular elements such as red blood cells. This could explain the observed decreases in serum albumin concentrations and intramuscular hemorrhage in monkeys with acute toxicity syndrome (study 1).
A further pathomechanistic aspect to consider is the compartment syndrome. Because the connective tissue that defines the muscular compartment does not stretch, a small increase in the volume due to swelling of the muscles within the compartment can cause the pressure to rise greatly and further reduce blood supply to the myocytes and augment damage. The compartment syndrome is frequently seen after ischemia and reperfusion. It is possible that an ischemia reperfusion situation may occur in the skeletal muscles after vildagliptin treatment due to a transient vasoconstriction.
Another hypothesis is the possible role of vasoactive peptides (substance P and neuropeptide Y) that could produce plasma exudation from the vascular space leading to edema formation. Substance P and neuropeptide Y are DPP-4 substrates. Evidence has been provided that vildagliptin affects the neuropeptide Y system (Hoffmann et al. 2014; Walker et al. 1991).
Few data are currently available to understand the possible relevance of immune-mediated mechanisms and potential effects on the complement system. It is well known that the DPP-4/CD26 protein plays an important role in immune response, and endogenous substrates for DPP-4/CD26 are involved in immunomodulation (Matteucci and Giampietro 2009). Available data that are indicative of an immune process in the manifestation of the acute toxicity syndrome include edema formation with nonspecific inflammation and infiltration of mononuclear, lymphoid/neutrophilic cells, and high interanimal variability of findings.
A possible role of DPP-8 or DPP-9 inhibition in the manifestation of the acute toxicity syndrome is currently not evident. However, data show that plasma concentrations of vildagliptin at doses that produce acute toxicity in Mauritian and Asian monkeys exceed the Ki values for DPP-8 and DPP-9 inhibition (Hoffmann et al. 2014).
In naive Mauritian macaques, a higher incidence of myocardial degeneration, subendocardial hemorrhage, and arterial medial degeneration/hemorrhage were observed compared with Asian monkeys (Indonesian source animals; Vidal et al. 2010). At present, it appears difficult to conclude whether these results are related to the findings in the present article.
Integrated Assessment—Relevance of Monkey Findings for Humans
An NOAEL for acute toxicity is currently not established in Mauritian monkeys. The lowest observable adverse effect level values in Mauritian monkeys are 20 mg/kg for edema formation and 40 mg/kg for acute death. In Asian monkeys, swelling and acute death were observed at 160 mg/kg. No acute toxicity was seen in Asian monkeys at 80 mg/kg (Hoffmann et al. 2014). Multiples of exposures that produce acute toxicity symptoms in both strains of cynomolgus monkeys compared with human therapeutic exposure at 50 mg twice a day are summarized in Table 3. These values suggest that Mauritian monkeys are more sensitive than Asian monkeys.
Exposure Multiples at LOAEL for Acute Toxicity in Asian and Mauritian Cynomolgus Monkeys Compared with Exposure in Humans at the Therapeutic Dose of 50 mg Twice a Day.a
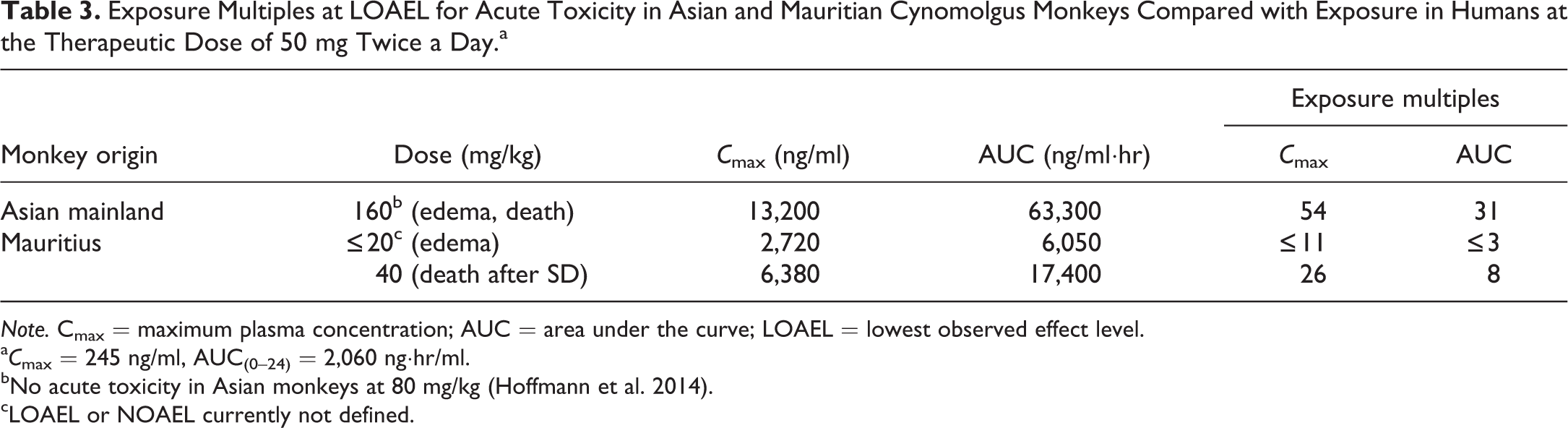
Note. Cmax = maximum plasma concentration; AUC = area under the curve; LOAEL = lowest observed effect level.
a C max = 245 ng/ml, AUC(0–24) = 2,060 ng·hr/ml.
bNo acute toxicity in Asian monkeys at 80 mg/kg (Hoffmann et al. 2014).
cLOAEL or NOAEL currently not defined.
It is important to note that the severe acute toxicity following vildagliptin treatment has been observed in cynomolgus monkeys only. In other animal species (rats, mice, and dogs), high doses of vildagliptin did not result in acute toxicity. Thus, the observed acute vildagliptin-induced toxicity in monkeys is likely species-specific.
A comprehensive review of the clinical trial and postmarketing data has been performed focusing on events that could possibly be related to the acute symptoms observed in cynomolgus monkeys, including events of edema, muscle-related events, CK elevations, and death. These clinical and postmarketing data reveal no evidence of acute symptoms similar to those observed in cynomolgus monkeys.
Conclusions
Acute toxicity occurs with high variability in cynomolgus monkeys but not in other species including humans. Cynomolgus monkeys from Mauritius appear more sensitive than monkeys of Asian origin. Acute toxicity is caused by vildagliptin itself and not by impurities or degradation products. The etiology of the acute toxicity symptoms in cynomolgus monkeys is not well understood, although a vascular mechanism including initial vasoconstriction and subsequent vascular leakage in distal extremities may play a role. Acute toxicity following vildagliptin treatment appears to be a monkey-specific effect without relevance for humans.
Footnotes
Authors Contribution
Authors contributed to conception or design (MK, DG, ES, DL, MH, PB, SB, PH); data acquisition, analysis, or interpretation (LM, MK, DG, ES, DL, MH, PB, SB, PH); drafting the manuscript (PH); and critically revising the manuscript (LM, MK, DG, ES, DL, MH, PB, SB). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
