Abstract
The purpose of this article is to characterize skin lesions in cynomolgus monkeys following vildagliptin (dipeptidyl peptidase-4 inhibitor) treatment. Oral vildagliptin administration caused dose-dependent and reversible blister formation, peeling and flaking skin, erosions, ulcerations, scabs, and sores involving the extremities at ≥5 mg/kg/day and necrosis of the tail and the pinnae at ≥80 mg/kg/day after 3 weeks of treatment. At the affected sites, the media and the endothelium of dermal arterioles showed hypertrophy/hyperplasia. Skin lesion formation was prevented by elevating ambient temperature. Vildagliptin treatment also produced an increase in blood pressure and heart rate likely via increased sympathetic tone. Following treatment with vildagliptin at 80 mg/kg/day, the recovery time after lowering the temperature in the feet of monkeys and inducing cold stress was prolonged. Ex vivo investigations showed that small digital arteries from skin biopsies of vildagliptin-treated monkeys exhibited an increase in neuropeptide Y–induced vasoconstriction. This finding correlated with a specific increase in NPY and in NPY1 receptors observed in the skin of vildagliptin-treated monkeys. Present data provide evidence that skin effects in monkeys are of vascular origin and that the effects on the NPY system in combination with increased peripheral sympathetic tone play an important pathomechanistic role in the pathogenesis of cutaneous toxicity.
Introduction
Dipeptidyl peptidase (DPP)-4 belongs to a family of postproline serine peptidases that include DPP-8, DPP-9, fibroblast activation protein, and attractin (Sedo and Malik 2001; Chen, Kelly, and Ghersi 2003). Inhibitors of DPP-4 represent a novel class of drugs for the treatment of type 2 diabetes. Drugs in this class afford significant and sustained reductions in hemoglobin A1c. Thus, it is important to fully understand the benefits and potential adverse effects associated with DPP-4 inhibition.
The desired pharmacologic action of DPP-4 inhibitors results in the decrease in metabolism of incretin hormones, for example, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide. However, incretin hormones are not the only endogenous substrates for DPP-4. Indeed, there are more than 35 known endogenous peptide substrates for DPP-4 (McIntosh et al. 2005; Grouzmann, Livio, and Buclin 2009). It is therefore important to determine whether inhibition of DPP-4 will potentially modify endogenous concentrations of biologically active peptides, thereby producing additional desired or unwanted effects. By inhibiting the metabolism of the vasoactive peptides, neuropeptide Y (NPY) and substance P (SP), DDP-4 inhibition was postulated to play a role in blood pressure regulation and interaction with antihypertensive medications, for example, angiotensin-converting enzyme inhibitors (Marney et al. 2010).
Vildagliptin is a novel orally bioavailable DPP-4 inhibitor (Villhauer et al. 2003). For preclinical safety evaluation studies, the selected rodent and nonrodent species were rats and dogs, respectively. However, at the request of the Food and Drug Administration in order to address concerns of a potential DDP-4 inhibitor class effect on monkey skin, individual pharmaceutical companies developing this class of compounds were asked to conduct a 13-week oral toxicity study in monkeys.
The current article characterizes these skin lesions in cynomolgus monkeys following oral vildagliptin administration. Furthermore, in vivo and ex vivo investigational studies were performed to elucidate the underlying mechanisms of the skin lesion formation. These studies provide evidence that vildagliptin-related skin lesions in monkeys are mediated through changes in vascular tone leading to vascular remodeling in arteries and that these effects are related to an increased peripheral sympathetic activity via NPY stabilization in the associated tissue.
Materials and Methods
In Vitro Inhibition of DPP-4, DPP-8, and DPP-9 from Cynomolgus Monkeys
Cynomolgus monkey DPP-4, DPP-8, and DPP-9 enzymes were expressed recombinantly from clones derived from monkey kidney cDNA. The extracellular domain of DPP-4 was fused at the N-terminus with a CD5 secretion signal sequence. It, or full length DPP-8 or DPP-9, was fused at the C-terminus with a His6-tag, and the AviTag was generated. Recombinant protein was coexpressed with the Escherichia coli BirA enzyme (biotin ligase) in sf9 insect cells and biotinylated enzyme was affinity purified by avidin chromatography.
The fluorescence-quenched substrate H-Gly-Pro-AMC and fluorescent product AMC were purchased from Bachem (Germany). All other chemicals were obtained from Sigma-Aldrich (St. Louis, MO) at the highest purity available. Inhibitor potency and time dependence were determined kinetically in a 384-well fluorescence intensity assay format. Both enzyme and substrate were diluted in assay buffer (25 mM Tris-HCl, pH 7.4, 140 mM NaCl, 10 mM KCl, and 0.05% [w/v] CHAPS) using 384-well plates (Corning, NY). Serial dilutions of inhibitors were preincubated at room temperature with enzyme for 30 min before reactions were started with varying concentrations of substrate. The final DMSO concentration was 1%. The reaction velocity was monitored kinetically for 2 hr at 380 and 435 nm (excitation and emission, respectively) using the M5 microplate reader (Molecular Devices, Sunnyvale, CA). Potency (Ki ) was calculated using the methods of Segel (1975). Time dependence was determined by diluting an enzyme–inhibitor complex into substrate and measuring the initial and final reaction velocities. Enzyme–inhibitor half-life was determined using the methods of Copeland (2000).
Animals and Treatment with Vildagliptin
This article describes the in vivo results of 7 separate studies performed in cynomolgus monkeys (see Table 1). Vildagliptin was administered daily for the indicated periods of time via oral gavage as a solution in Purified Water (USP) to male and female cynomolgus monkeys (Maccaca fasicularis) of Asian origin. The monkeys were obtained from Covance, Denver, Pennsylvania; or Alice, Texas; World Wide Primate, Miami, Florida; or Rhenos/Pacific Forest Resources, Chestnut Hill, Massachusetts, via Buckshire Corporation via New Iberia Research Center, Louisiana. The animals were between 2 and 7 years of age and weighed between 1.8 and 5.6 kg at the initiation of the studies. Animals were singly housed in primate cages throughout the duration of the studies. Experiments were conducted in accordance with Novartis animal care regulations and governmental guiding principles in the use of animals.
Summary of in vivo studies with vildagliptin in monkeys.

Note: ECG = electrocardiogram.
Histological Evaluation
Representative skin samples (chest, digits [hand, foot], ear, tail, scrotum, and macroscopic skin lesions) were collected and fixed for a maximum of 48 hr in 10% neutral buffered formalin, in order to allow for possible immunostaining. Selected tissues were immersion-fixed in ice-cold 4% paraformaldehyde in 0.1 M phosphate-buffered saline (PBS), pH 7.2 to 7.4 for 4 hr and then transferred to 0.1 M PBS + 0.1% sodium azide (NaN3) and refrigerated for possible immunohistochemistry (IHC). Paraffin blocks of formalin-fixed tissues were cut at 6-µm thickness and stained with hematoxylin and eosin (H&E). Selected skin sections were also stained with Masson’s trichrome to distinguish muscle from collagen. Processed skin sections were evaluated microscopically and peer reviewed.
Immunohistochemistry
Immunohistochemical staining for Ki67 was performed on study slides using the fully automated instrument Discovery (Ventana Medical Systems SA, Strasbourg, France). All standard steps (deparaffination, rehydration) used reagents provided by Ventana Medical Systems SA. Nonspecific background staining was blocked using 1× casein solution in PBS (BioFX laboratories, #PBSC-0100-5x) for 30 min at room temperature and endogenous biotins were quenched using A/B blocking reagents (Ventana Medical Systems). Antibodies were applied manually using 100 μ l per slide of primary antibody previously diluted in Discovery antibody diluent (Ventana Medical Systems) at room temperature. After a short postfixation using glutaraldehyde at 0.05%, slides were incubated for 16 min with biotin-conjugated secondary antibody (Jackson Laboratories), diluted at 1/500 in Discovery antibody diluent. Detection of biotin was performed using DABMap (Ventana Medical Systems).
The proliferation rate was assessed by visual scoring of ki67 expression in distinct histological structures (e.g., excretory ducts of eccrine glands and capillaries) based on the clustering of positive cells (1–4). Inflammatory foci or histological structures adjacent to inflammatory foci were excluded.
Toxicokinetics
Measurement of vildagliptin and M20.2 (pharmacologically active metabolite) concentrations in plasma or homogenized skin samples was performed using liquid chromatography–tandem mass spectrometry. Sample extraction was performed by solid phase extraction of the plasma samples on 10-mg Oasis 96-well extraction plates. The extracted samples were eluted from reverse phase columns using isocratic elution conditions. The detection was performed on API4000 mass spectrometers under positive mode. The transition monitored was from m/z 304 → 154. The lower limit of quantification was 0.100 ng/ml. Skin samples were first homogenized after 5-fold dilution with phosphate buffer and then analyzed using the method for plasma.
Measurement of Peripheral Temperature during Cold Provocation
A cold provocation test was performed to indirectly assess the vasoconstriction of cutaneous vessels. Skin temperatures were used to indirectly evaluate blood flow in the feet of monkeys. Temperature of the plantar surface of the foot was measured under anesthesia (10 mg/kg/day ketamine plus 0.5 mg/kg/day xylazine intramuscularly [i.m.]) prior to, during, and after a cold provocation challenge by immersing of the foot in ice-cold water for 5 min. Temperature was monitored via continuous measurements at 1-sec intervals using an SST-1 skin probe connected to a PhysiTemp monitor.
Cardiovascular Effects and Urinary Norepinephrine
In the first cardiovascular study, cynomolgus monkeys (n = 4 males) received purified water (USP) once a day for 5 days via oral gavage administration. Then animals received vildagliptin treatment in an ascending dose paradigm once a day for 5 days for each dose level (0, 0.1, 0.3, 3, 10, 20 mg/kg/day). In the second cardiovascular study, ascending doses of 0, 40, 80, 160, and 240 mg/kg/day were administered once a day for 5 days for each dose level (n = 4 males). Telemetry recordings were performed with a Data Sciences International Dataquest A.R.T. 2.3 telemetry recording system from implanted animals. The telemetry parameters including blood pressure, heart rate, and lead II electrocardiogram (ECG) were recorded continuously from approximate 1.5 hr prior to dosing (baseline) to 24-hr postdose. The hemodynamic parameters and ECG intervals (P duration, PR, R-R, QRS, QT intervals) were measured and reported as approximately every 15-min average value from baseline through 24-hr postdose.
Norepinephrine was measured in urine samples (∼ 1 ml) over a 2-hr period immediately postdose to 2-hr postdose. Norepinephrine was determined by radioimmunoassay (ALPCO assay 17-RA607-150), with evaluations performed by Ani Lytics (Gaithersburg, MD).
Vascular Responses in Digital Arterioles
Digital arterioles were obtained from the subcutis of digits of monkeys that had been treated with vildagliptin (80 mg/kg/day) for 2 or 4 weeks. Digital arterioles were also obtained from an additional group of untreated monkeys to analyze the effects of vildagliptin when administered directly to the bath solution in vitro. Animals were fasted overnight (approximately 12–18 hr) prior to terminal necropsy and anesthetized with an i.m. injection of a mixture of ketamine HCL plus xylazine HCL. The third digit of the right hand was removed, transferred into a sterile 50-cc centrifuge tube filled with ice-cold sterile Dulbecco’s modified Eagle’s medium (Gibco stock # 11885084), and placed on wet ice overnight. The next day, small digital arterioles were carefully dissected from the monkey digits and perfusion experiments were performed as described (Chotani et al. 2000; Flavahan 2005). In brief, side branches were tied and the arterioles placed in cold Krebs-Ringer bicarbonate solution (in mM): 118.3 NaCl, 4.7 KCl, 1.2 MgSO4, 1.2 KH2PO4, 2.5 CaCl2, 25.0 NaHCO3, 11.1 glucose (control solution). The vessels were cannulated at both ends using glass micropipettes in a microvascular chamber (Living Systems, Burlington, VT). The arterioles were maintained at a constant transmural pressure (PTM) of 60 mmHg in the absence of flow. The chamber was superfused with control solution and maintained at 98.6 °F, pH 7.4, and gassed with 16% O2–5% CO2–balance N2. The chamber was placed on the stage of an inverted microscope (20×, Nikon TMS-F, Japan) connected to a video camera (Panasonic, CCTV camera, Japan). The vessel image was projected onto a video monitor and the internal diameter was continuously evaluated by a video dimension analyzer (Living Systems Instrumentation, Burlington VT) that was monitored using a BIOPAC (Santa Barbara, CA) data acquisition system.
Responses of digital arteries to NPY, phenylephrine, and acetylcholine were investigated. NPY and phenylephrine cause vasoconstriction via stimulation of postsynaptic NPY1 or α-adrenergic receptors on smooth muscle cells. Acetylcholine causes vasodilation via nitric oxide (NO) release from the endothelium. All responses are expressed as a percentage of the baseline diameter of the arterioles prior to the intervention. When analyzing the in vitro effect of vildagliptin on vascular function, the arterioles were incubated with 1 μM vildagliptin for 30 min prior to assessing vascular responses.
Immunofluorescence
Immunofluorescence was performed to evaluate the presence and content of various neuropeptides and receptors in the skin. Skin samples harvested were immediately fixed into cold 4% paraformaldehyde in 0.1 M PBS pH 7.2 to 7.4, for 4 hr in wet ice (approximately 39.2 °F). Samples were then transferred to excess 0.1 M PBS containing 0.01% NaN3 and kept refrigerated at least overnight. Twenty-four hours prior to cryostat sectioning, the samples were transferred into tubes containing 30% sucrose in 0.1 M PBS, pH 7.2 to 7.4 and kept refrigerated until they had sunk all the way to the bottom of the tube indicating complete penetration of the sucrose solution and optimal cryoprotection. Samples were snap frozen in chilled isopentane and stored at approximately −94 °F.
Fourteen micrometer-thick sections were cut and mounted sequentially onto 12 slides. Sectioning and mounting on corresponding slides were repeated until complete processing of the block. Slides were allowed to reach room temperature just before use, then were hydrated in PBS for 5 min, permeabilized in 0.2% triton X100/PBS for 6 min, and rinsed for 2 min in PBS. Nonspecific binding was blocked with casein (Vector SP5020 diluted 10× in double distilled H2O) for 30 min. Antibodies and protocols were optimized for the neuropeptides Substance P (SP), calcitonin gene–related peptide (CGRP), pituitary adenylate cyclase–activating polypeptide (PACAP-1), NPY, bradykinin and for the receptors of NPY1, NPY2, bradykinin 1, bradykinin 2, and neurokinin 1. For each combination, primary antibodies were mixed in PBS containing 1% bovine serum albumin (BSA) and 0.05% Tween-20. In each combination, 2 neuropeptides or neuropeptide receptors were detected simultaneously (see Table 2). In addition, antibody against smooth muscle α-actin was added to allow identification of arterial walls and the secretory portion of the eccrine glands. Incubation with the primary antibodies was performed overnight at 39.2 °F. After washing twice with PBS, the slides were incubated for 30 min with the appropriate fluorescent secondary antibodies (Molecular Probes) diluted at 1/200 in PBS containing 1% BSA and 0.05% Tween-20 (Table 3). After washing twice for 5 min with PBS, slides were counterstained for 15 to 20 min with DAPI (10 μ g/ml PBS, Molecular Probes D-1306), mounted with GelMount (Biomeda M01), and stored at 39.2 °F in the dark until image acquisition. Two negative controls were conducted either by omitting the primary antibody or using the appropriate isotype control (see Table 3). The presence or absence of a specific staining pattern was recorded for each slide/field. Then the rate of occurrence in each series was deduced and reported as a frequency per treatment group.
Antibodies used for immunofluorescence.
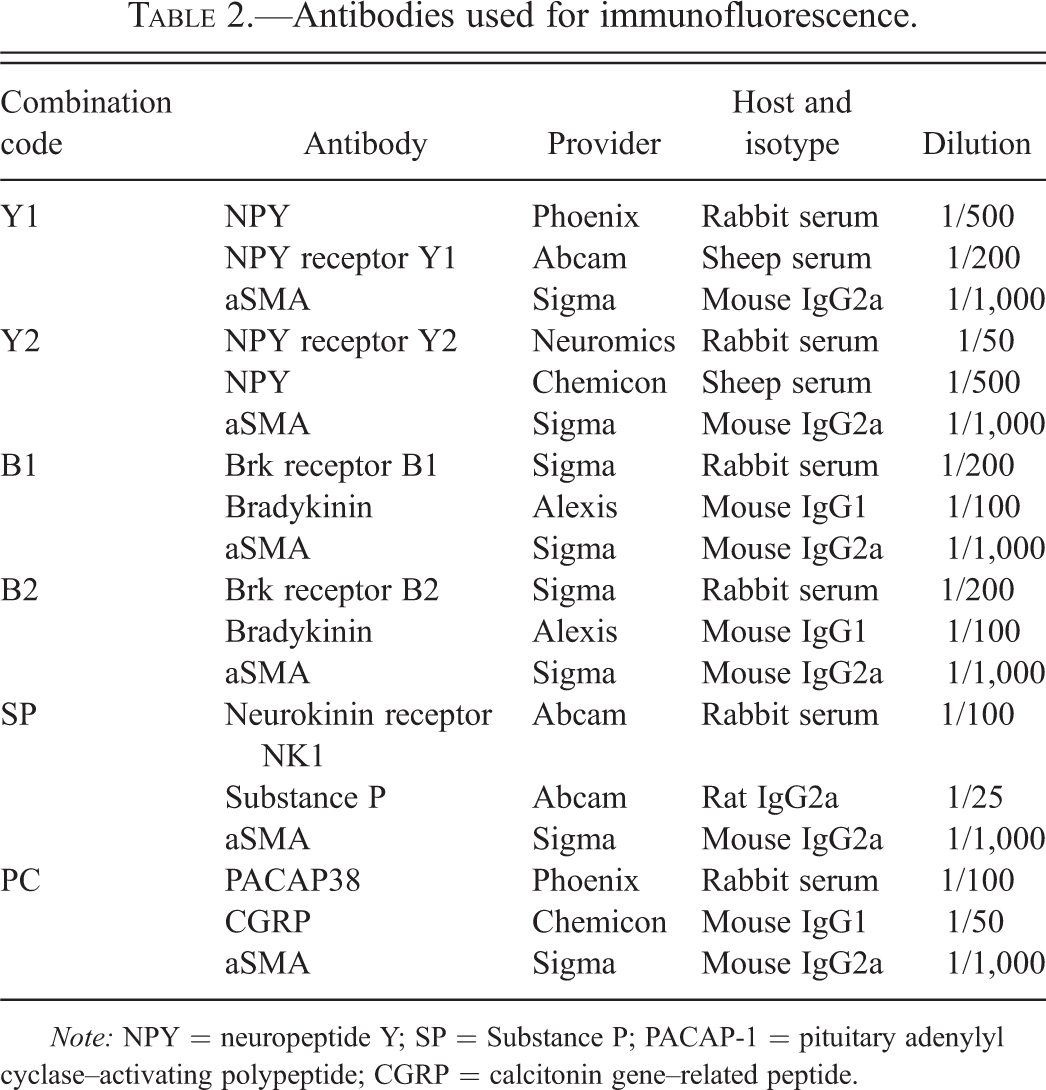
Note: NPY = neuropeptide Y; SP = Substance P; PACAP-1 = pituitary adenylyl cyclase–activating polypeptide; CGRP = calcitonin gene–related peptide.
Secondary antibodies for immunofluorescence.

Data Analysis
Data are presented as mean ± SEM. Statistical evaluation of the data from 2 means was performed by Student’s t-test. For comparison of more than 2 means, analysis of variance (ANOVA) was used: either a one-way or repeated measures ANOVA using Student–Newman–Keuls Multiple post hoc tests (GraphPad Software, San Diego, CA). A significance level of p < .05 was used. Proliferation scores for Ki67 were quantified using median scores with boxes corresponding to quartiles and bars indicating extremes of group values.
Results
In vitro Inhibition of DPP-4, DPP-8, and DPP-9 from Cynomolgus Monkeys
Vildagliptin inhibited recombinant cynomolgus monkey DPP-4 enzyme activity with a Ki of 5.0 ± 0.4 nM. The inhibition was characterized by reversible slow tight-binding kinetics with an enzyme-inhibitor half-life of 37 ± 3 min. Recombinant cynomolgus monkey DPP-8 and DPP-9 enzymes were inhibited with Ki values of 770 ± 100 nM and 68 ± 5 nM, respectively, with reversible fast-off binding kinetics with enzyme-inhibitor half-lives less than 3 min. Vildagliptin also potently inhibited endogenous DPP-4 enzyme activity from cynomolgus monkey plasma and purified kidney enzyme with 50% inhibitory concentration (IC50) values of 3.3 ± 0.4 nM and 4.1 ± 0.9 nM, respectively.
The two major metabolites of vildagliptin in cynomolgus monkey plasma are M20.2 (72 % of the administered vildagliptin dose is metabolized to M20.2 in monkeys) and M20.7 (5% of the vildagliptin dose; Figure 1). M20.7 is formed by hydrolysis of the cyano moiety to form a carboxylic acid metabolite. The other predominant metabolic pathway is O-conjugation with glucuronic acid (M20.2) leaving the pharmacophore part of the molecule unchanged. The in vitro inhibitory profiles of both of these metabolites were evaluated using recombinant cynomolgus monkey DPP enzymes. M20.2 inhibited recombinant cynomolgus monkey DPP-4 activity with a Ki value of 5.0 ± 0.6 nM, that is, its inhibitory potency is identical to the parent compound. The inhibition was characterized by reversible slow tight-binding kinetics with an enzyme-inhibitor half-life 53 ± 6 min. M20.2 was a less potent inhibitor of recombinant cynomolgus monkey DPP-8 and DPP-9, with Ki values of 5,100 ± 800 nM and 390 ± 50 nM, respectively, with reversible fast-off binding kinetics and enzyme-inhibitor half-lives of 2 min or less. The metabolite M20.7 was a very weak inhibitor of recombinant cynomolgus monkey DPP-4 activity with an IC50 value of 161 ± 21 μM, and did not inhibit recombinant cynomolgus monkey DPP-8 and DPP-9 (IC50 values >200 μM).
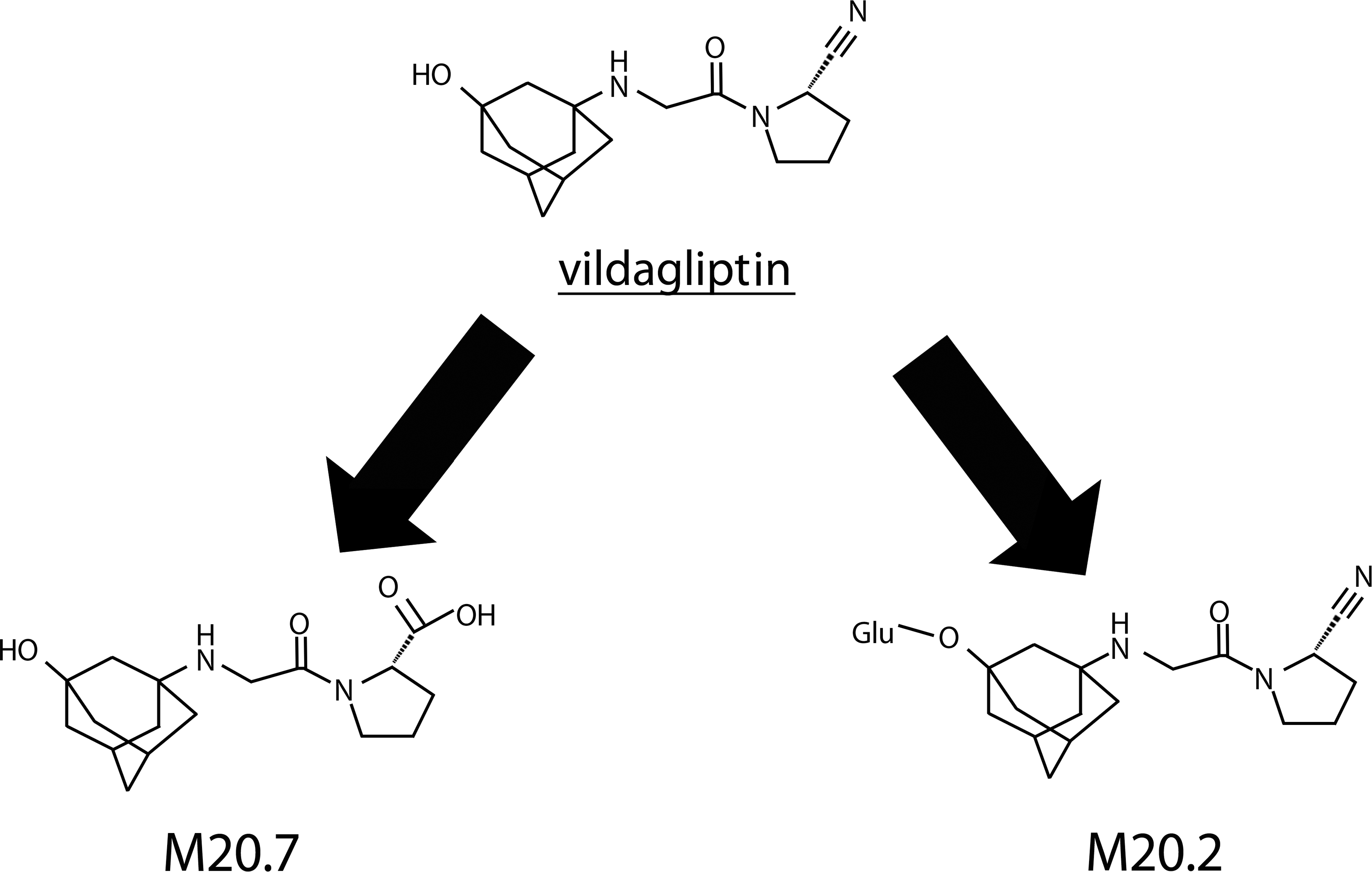
Metabolism of vildagliptin in monkeys.
Manifestation of Skin Lesions
Administration of vildagliptin to cynomolgus monkeys at oral doses of 0, 5, 20, 80, and 160 mg/kg/day (3 animals/sex/dose group) for 13 weeks (study 1) was associated with dose-dependent skin lesion formation in both sexes at all doses, although occurrence and severity of the skin effects were variable within animals of the individual dose groups. The lesions were located on the distal extremities (hands, feet, ears, and tail). Skin lesions appeared after 3 weeks of treatment and consisted of blister formation, peeling and flaking skin, erosions, ulcerations, scabs, and tail sores. Necrosis of the tail and the pinnae was observed at doses ≥80 mg/kg/day. No gender difference in frequency and severity of skin effects was observed. It should be noted that the animals did not have systemic signs of moribundity.
To further characterize the dose–effect relationship, a subsequent 13-week monkey study (study 2) was performed with lower oral vildagliptin doses. In this subsequent 13-week study at doses of 0.3, 1, 3, and 20 mg/kg/day (3 animals/sex/dose group), skin lesions were reproduced at 20 mg/kg/day and the no observable adverse effect level (NOAEL) was considered to be 3 mg/kg/day.
An additional study (study 3) was conducted to demonstrate reversibility of lesion formation and to collect skin samples for mechanistic evaluations. Monkeys were treated with oral doses of 0 or 80 mg/kg/day (6 females/dose group) vildagliptin. After 4 weeks of treatment, severe skin lesions developed in 3 animals such that vildagliptin treatment was stopped. The skin lesions on the hands and feet were clinically reversible over a course of 16 days. These results demonstrate that the cause of skin lesion formation is related to vildagliptin exposure and that the cutaneous lesions heal appropriately, demonstrating that the condition is reversible and is not due to a permanent impairment of tissue repair mechanisms.
Histological Findings
Histopathological examinations were performed in studies 1 and 2 to evaluate microscopic details of the gross cutaneous lesions. Vildagliptin-related macroscopic skin lesions occurring at the extremities correlated with microscopic findings as shown in Figure 2. Following 4 weeks of vildaglitin administration at 80 mg/kg/day (but not after 2 weeks), medial hypertrophy/hyperplasia of arterioles was seen at various levels of the dermis, particularly just below the epidermis. The endothelium also showed signs of hypertrophy and hyperplasia. Inflammatory cells were occasionally seen in the dermis and around blood vessels in both the control and treated animals, but this was not considered related to the arteriolar changes. In contrast to dermal arterioles from extremities, dermal arterioles from skin of the chest were not affected (not shown).
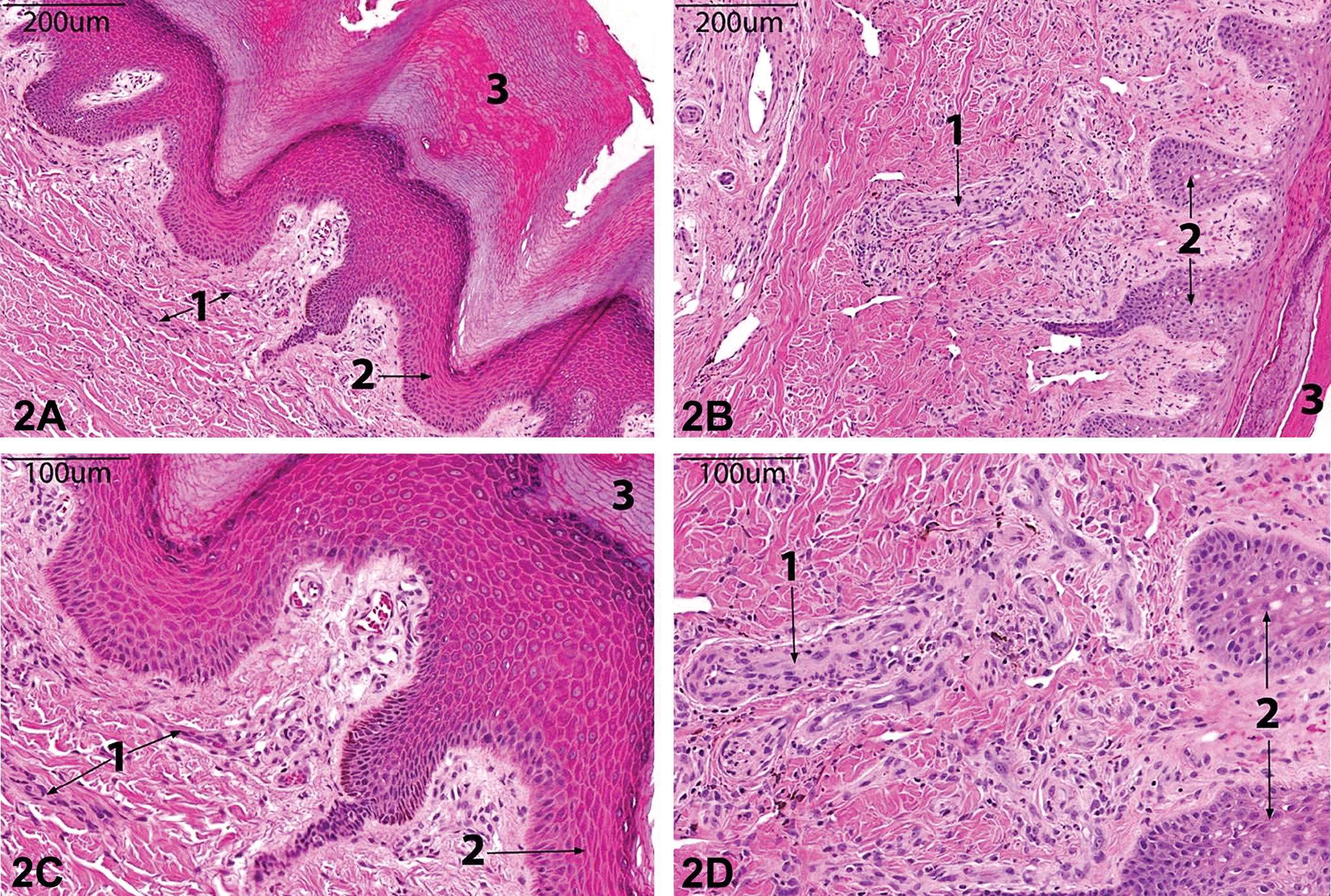
Photomicrographs of hematoxylin and eosin (H&E)-stained sections of skin from the foot. 20×: Control animal (A) and vildagliptin-treated animal (80 mg/kg/day for 4 weeks) (B); 40×: Control animal (C) and vildagliptin-treated animal (80 mg/kg/day for 4 weeks) (D). Dermal arterioles (1), epidermis (2), and keratin (3) are marked. Small- to medium-caliber arterioles (1) are thickened and arteriolar walls are much more prominent at 20× in the vildagliptin-treated animals (2B-1) compared to the controls (2A-1, 2C-1) as manifested by smooth muscle hypertrophy/hyperplasia at 40× (2D-1). An edge of epidermal erosion with inflammatory exudate mixed with keratin is visible in the vildagliptin-treated animal at 20× (2B-3).
Immunohistochemistry
The proliferation scores in epithelial cells of eccrine glands and vessels of monkey digits from study 1 animals were established by IHC for Ki67 and quantified visually by 2 independent observers. Proliferation score was high and unaffected by vildagliptin treatment in eccrine glands (Figure 3). In cutaneous vessels, the main site of Ki67 immunoreactivity was observed in the endothelial cells of the superficial dermis. Blind scoring of front digit and hind digit samples revealed treatment-related differences only in the endothelium of upper dermis vessels. Following 13 weeks of treatment with vildagliptin, the median proliferation score was 1.0 in controls compared to 2.0 and 2.5 at ≥20 mg/kg/day (Figure 4). The proliferation score returned to control values after a 4-week postdosing phase.
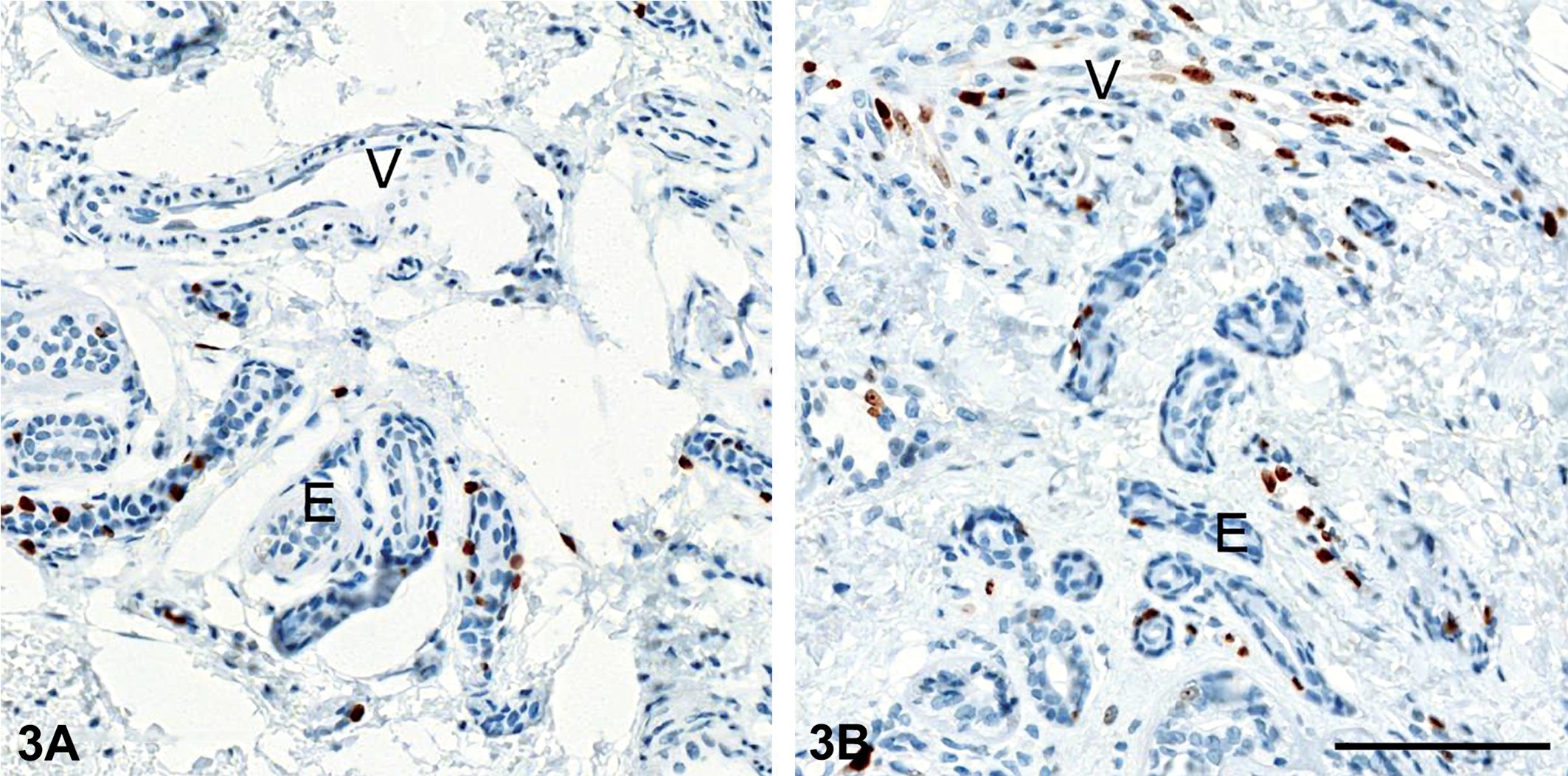
Proliferation in skin of monkeys treated with vildagliptin. Animals treated with vehicle (A) show background proliferation in epithelial cells of eccrine glands (E), whereas vessels (V) show no immunoreactivity for ki67. After vildagliptin treatment at 20 to 160 mg/kg/day for 13 weeks (B), the endothelium of small vessels in the superficial dermis showed abundant Ki67 expression (V).
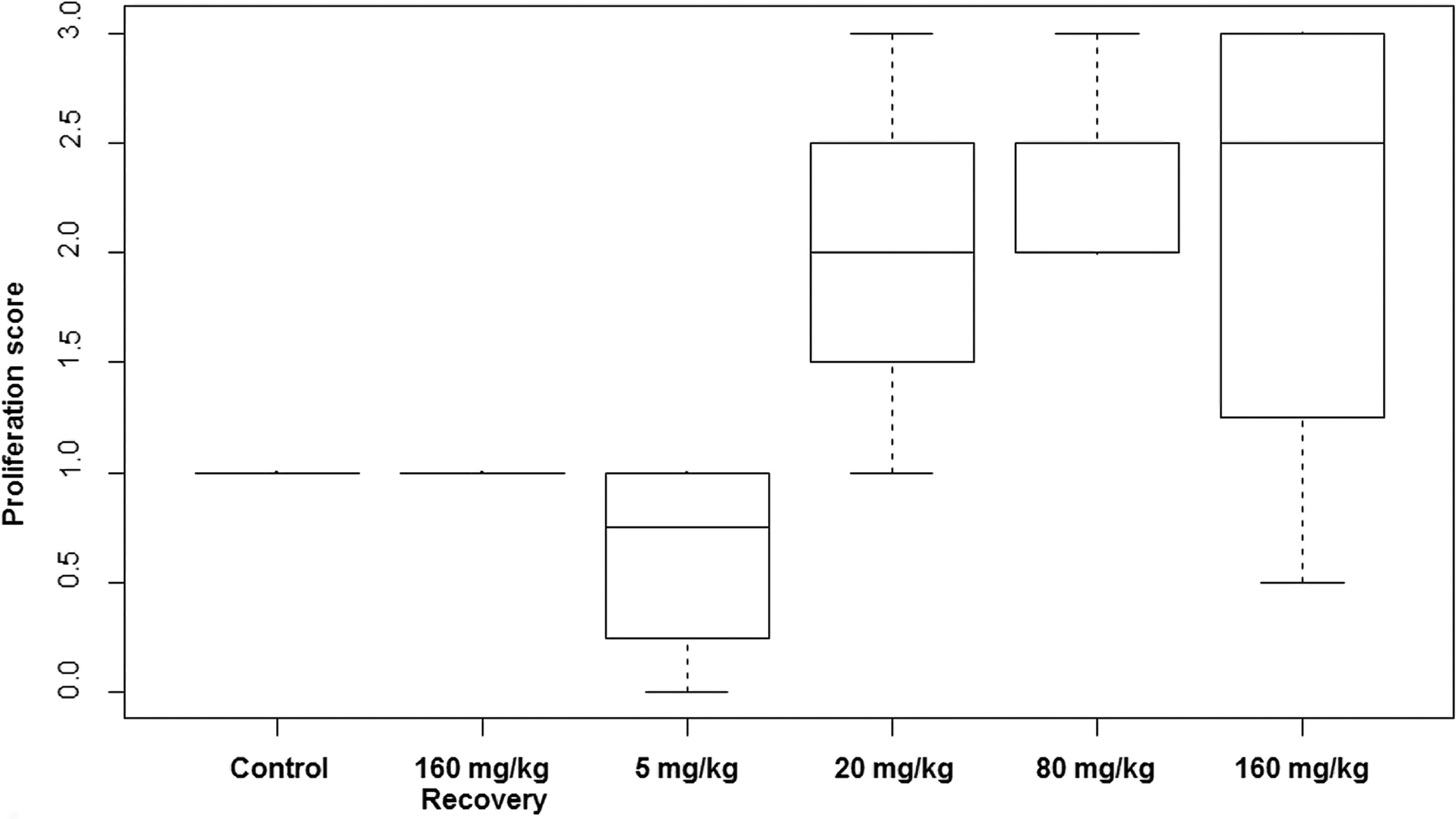
Proliferation scores in skin of monkeys treated with vildagliptin at 5 to 160 mg/kg/day for 13 weeks. Horizontal lines indicate median score, boxes correspond to quartiles, and bars indicate extremes of group values.
Effect of Increased Room Temperature
In a follow-up 13-week in vivo study (study 4), vildagliptin-associated skin lesions were investigated related to thermoregulation by elevating the animal room temperature. In this study, the ambient temperature was increased from standard room temperatures of 68 °F – 75 °F to 81 °F to 88 °F temperatures, more typical of the cynomolgus monkey’s natural climate. Monkeys were treated with doses of 0, 20, or 80 mg/kg/day (n = 4/sex/dose group). Clinically, skin lesions did not develop in vildagliptin-treated animals maintained under elevated room temperature and the severity of hypertrophy/hyperplasia of the media in dermal arterioles was minimal when compared with results from 13-week studies at standard room temperature.
Toxicokinetics
Toxicokinetic parameters after oral vildagliptin administration are summarized in Table 4. Vildagliptin was rapidly absorbed with a mean time of maximum concentration (Tmax) ranging from 1 hr (first time point of plasma sampling) to 2.5 hr postdose. Vildagliptin exposure increased proportionally with increasing dose in the tested dose range (0.3–160 mg/kg/day, studies 1 and 2). Maximum plasma concentration (Cmax) at the lowest observed skin effect level of 5 mg/kg/day was 3,000 and 2,160 nM in male and female monkeys, respectively. For the female monkeys, there was a trend toward a slightly lower exposure to vildagliptin compared to the exposure in males. Exposure to vildagliptin was similar between day 1 (data not shown) and week 11.
Mean values of toxicokinetic parameters of vildagliptin in monkey plasma after 11 weeks of treatment.

Note: Cmax = maximum concentration; AUC = area under the curve; Tmax = time of maximum concentration; M = male; F = female.
n = 3/sex.
Temperature increases in the animal room from standard room temperature of 20°C to 24°C to 27°C to 31°C (study 4) had no apparent impact on the exposure to vildagliptin when compared with results from the 13-week study at standard room temperature. Table 5 summarizes the toxicokinetic parameters in animals at elevated room temperature.
Mean values of toxicokinetic parameters of vildagliptin in monkey plasma after 11 weeks of treatment at elevated room temperature.

Note: Cmax = maximum concentration; AUC = area under the curve; Tmax = time of maximum concentration; M = male; F = female.
n = 4/sex.
Analyses of vildagliptin were also performed in skin samples from the ear where skin lesions were typically seen at lower ambient animal housing temperatures and the chest where skin lesions did not develop (study 1). Skin concentrations of vildagliptin in ear and chest skin were similar and very high. At 24-hr postdose, vildagliptin skin concentrations were ∼100× higher than plasma concentrations.
Plasma concentrations of the pharmacologically active metabolite M20.2 at 24-hr postdose were ∼6× higher than vildagliptin plasma concentrations, while the concentrations of the less lipophilic molecule M20.2 in the skin were only ∼2× the concentrations of the parent compound (study 1).
Measurement of Peripheral Temperature during Cold Provocation
Following treatment with vildagliptin at 80 mg/kg/day for 4 weeks (study 5), the recovery time after reducing the temperature in monkey feet by cold water immersion challenge was prolonged (Table 6). These data indicate that there was prolonged vasoconstriction and decreased peripheral blood flow in vildagliptin-treated animals.
Effect of cold provocation (5 min, 41°F) on temperature in feet of monkeys following 4 weeks vildagliptin treatment (80 mg/kg/day).
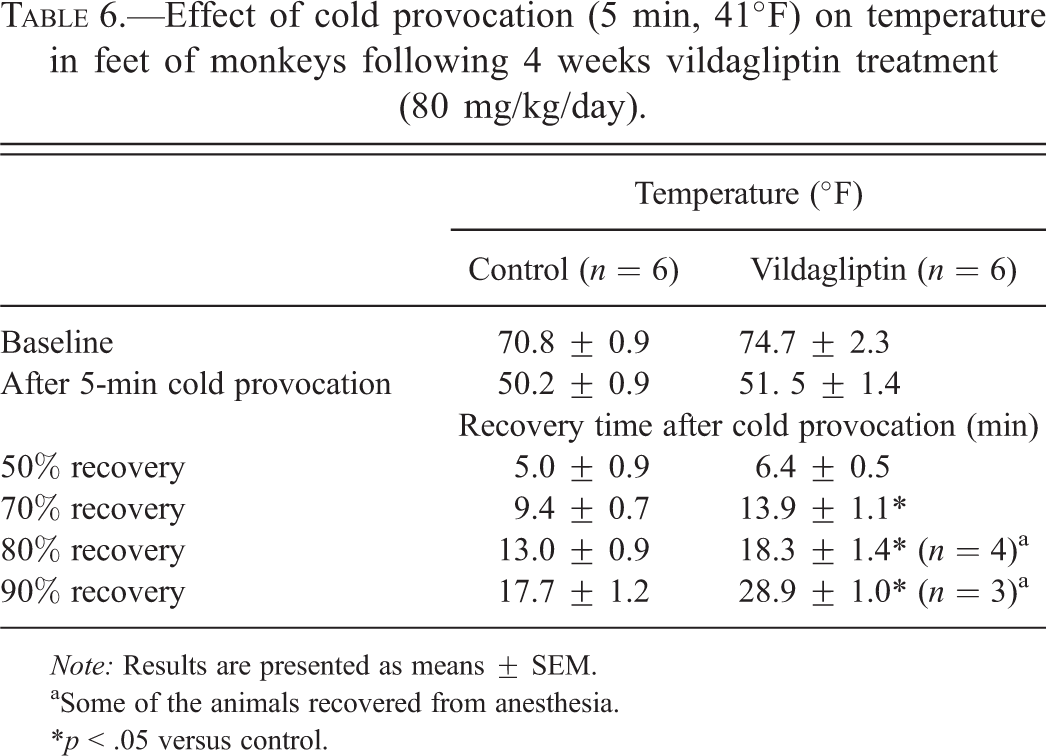
Note: Results are presented as means ± SEM.
aSome of the animals recovered from anesthesia.
*p < .05 versus control.
Cardiovascular Effects and Urinary Norepinephrine
In 2 cardiovascular telemetry studies (studies 6 and 7), the effects of vildagliptin on blood pressure, heart rate, and ECG were tested at doses of 0, 0.1, 0.3, 1, 3, 10, 20, 40, 80, 160, and 240 mg/kg/day (n = 4) to evaluate whether the impact of vildagliptin extends beyond the cutaneous vasculature. Although not reflected in mean values, a review of the individual animal data revealed a slight to moderate increase in blood pressure (5–40 mm Hg) and heart rate (5–20 beats per minute) at different time points from 30 min to 1 hr after dosing. Increases in blood pressure and heart rate were observed in all animals at doses ≥10 mg/kg/day without a clear dose-dependency. Intervals and shape of ECG waves were not affected by vildagliptin treatment, and no cardiac arrhythmias were observed.
Urinary norepinephrine concentrations were measured after 4 weeks of treatment with vildagliptin at 80 mg/kg/day (study 4). Mean concentrations of urinary norepinephrine were 3.6-fold higher in vildagliptin-treated animals compared with controls (24.4 ± 32 ng/ml vs. 6.7 ± 3.6 ng/ml, n = 6/group). Due to the low number of animals and the high interindividual variability, differences in means between control and vildagliptin-treated animals did not attain significance level (p > .05).
Vascular Responses in Digital Arterioles
To further investigate the reactivity of the cutaneous vascular system following vildagliptin treatment, monkeys were dosed for 2 or 4 weeks with vildagliptin (80 mg/kg/day) or vehicle (study 5). Small digital arteries from skin biopsies were cannulated and perfused under constant pressure. Conditions were previously optimized in the laboratory to preserve maximum vascular integrity and viability. In these preparations, NPY-induced constriction of cutaneous arteries was significantly enhanced after 4 weeks (but not 2 weeks) of vildagliptin treatment (Figure 5). In contrast, the vasoconstrictory responses to phenylephrine (0.01–1 µM) were unaffected by the 2- and 4-week vildagliptin administration (data not shown). Adding vildagliptin to the perfusion fluid of arterial preparations from naive nonvildagliptin-treated animals did not affect NPY-induced vasoconstriction (n = 6, data not shown).
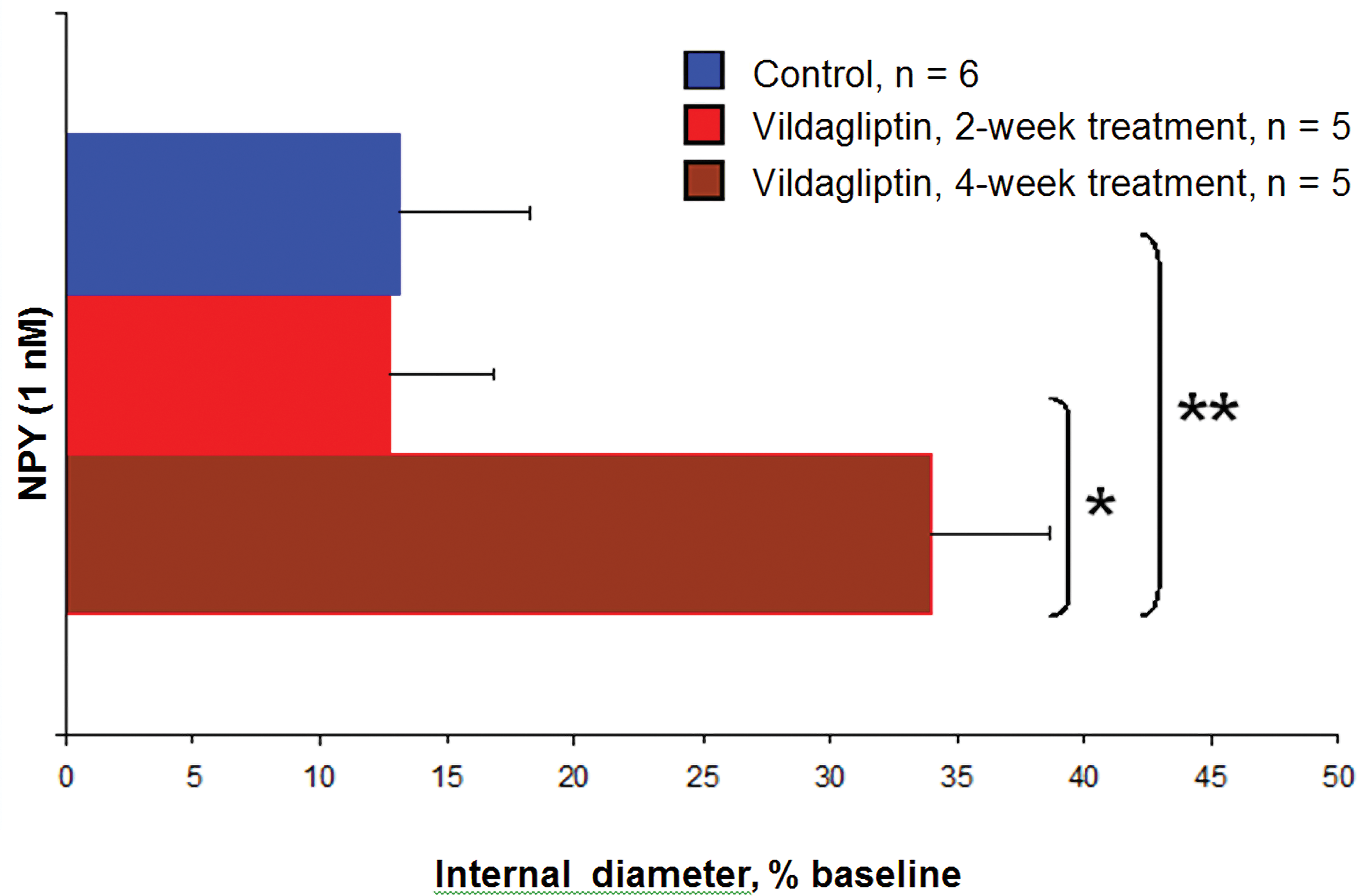
Effects of 2 or 4 weeks in vivo treatment with vildagliptin (80 mg/kg/day) on the vasoconstrictory response to neuropeptide Y (1 nM) in monkey digital arteries in vitro. Effects were evaluated during partial constriction (25–30 of baseline) to phenylephrine (0.3 μM). Results are expressed as percentage increase from predrug diameter and are presented as means ± SEM. *p < .05. **p < .01.
We also investigated the effects of 2 or 4 weeks in vivo vildagliptin treatment on acetylcholine-induced endothelium-dependent vasodilation. In these experiments, the acetylcholine effect was enhanced in arteries from digit skin biopsies after 4 weeks of vildagliptin treatment, but not after 2 weeks of treatment (Figure 6).
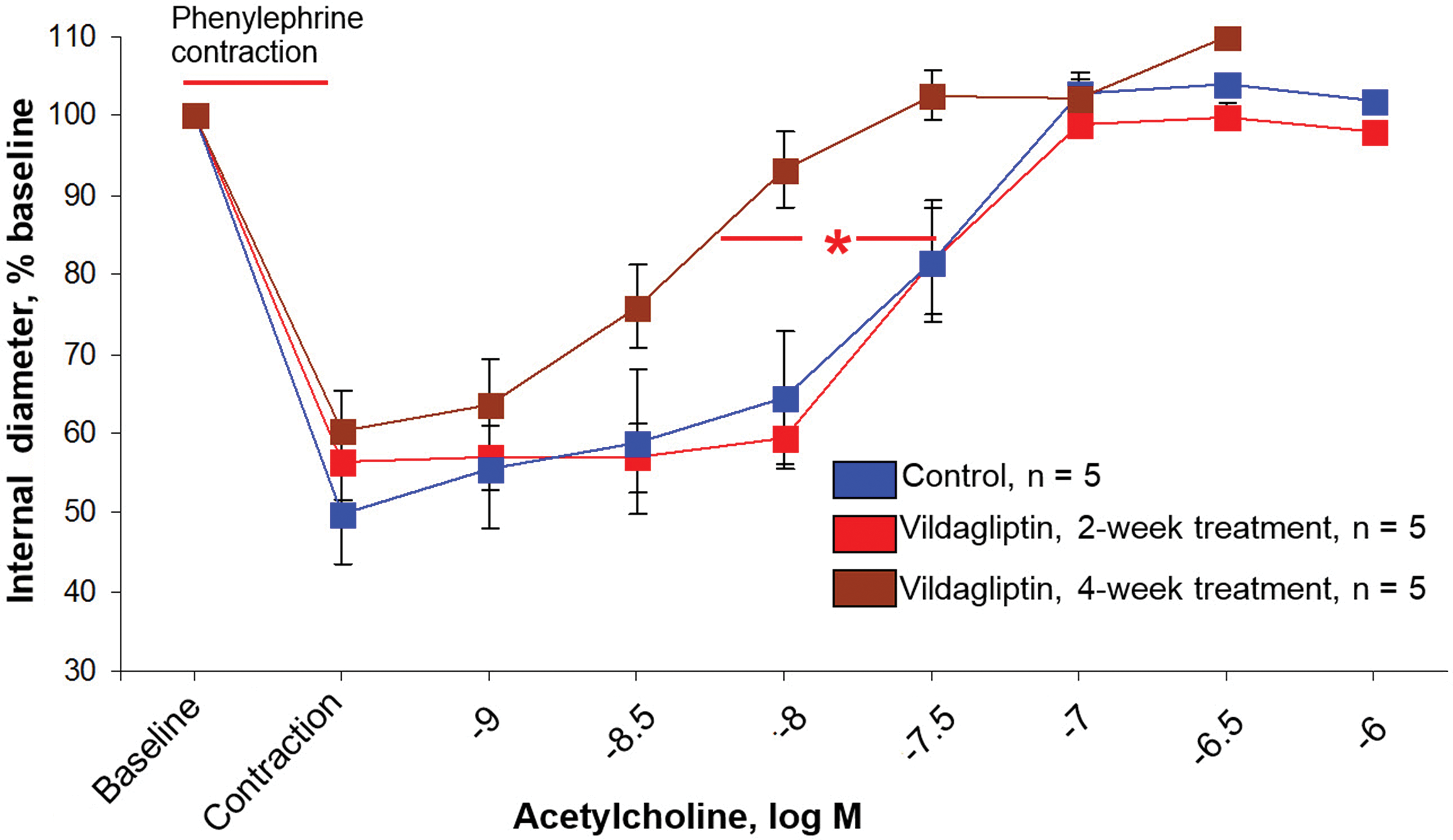
Effects of 2 or 4 weeks in vivo treatment with vildagliptin (80 mg/kg/day) on the response to acetylcholine in monkey digital arteries in vitro. Arteries were constricted with phenylephrine (0.3 μM) prior to administering acetylcholine. Results are expressed as percentage change of predrug diameter and are presented as means ± SEM. Treatment versus control at acetylcholine concentrations achieving 85% baseline diameter. *p < .05 4-week
Immunofluorescence
Tissue distributions of a number of neuropeptides and receptors (SP, CGRP, PACAP-1, NPY, bradykinin; receptors for NPY1, NPY2, bradykinin 1, bradykinin 2, and neurokinin 1) in the skin of the feet and hands (heel, digital pads of the foot and the hand, hypothenar region) of vildagliptin-treated monkeys from study 5 were evaluated. Image acquisition and analysis focused on identification of reproducible labeling patterns of neuropeptides or receptors that reflect their specific expression patterns and localization in the tissue and the frequency at which these patterns were observed in individual animal samples from the different treatment groups. Among 55 patterns recorded across the various staining combinations, only 2 patterns relating to NPY and NPY1 receptor showed a distinct difference between vildagliptin-treated animals and controls. NPY immunoreactivity was mainly found in epithelial structures, more prominently in ductal cells of eccrine glands. But in some animals, a secretory staining was also present. Interstitial labeling was less frequent and occurred rarely as diffuse cytoplasmic staining of isolated cells and more often as dots that were not obviously related to cell cytoplasm and likely corresponding to nerve endings. The major expression pattern of NPY1 receptor was an intense rim located at the luminal aspect of the epithelium of eccrine ducts. Less frequent and less intense was the diffuse staining of eccrine secretory cells. Staining of actin-containing cells such as myoepithelial or medial smooth muscle cells was even less abundant and only a few interstitial dots were found. The colocalization of NPY and NPY1 receptor was mostly found in endothelial cells, but also in the media of arteries.
In monkeys treated with vildagliptin for 4 weeks (80 mg/kg/min), NPY was more prominent around the basal layer of eccrine ducts as compared to controls (50% vs. 10%; Figure 7). NPY1 receptor rim-like distribution was more abundant among severely affected vildagliptin-treated monkeys in comparison to controls (70% vs. 40%; Figure 7).
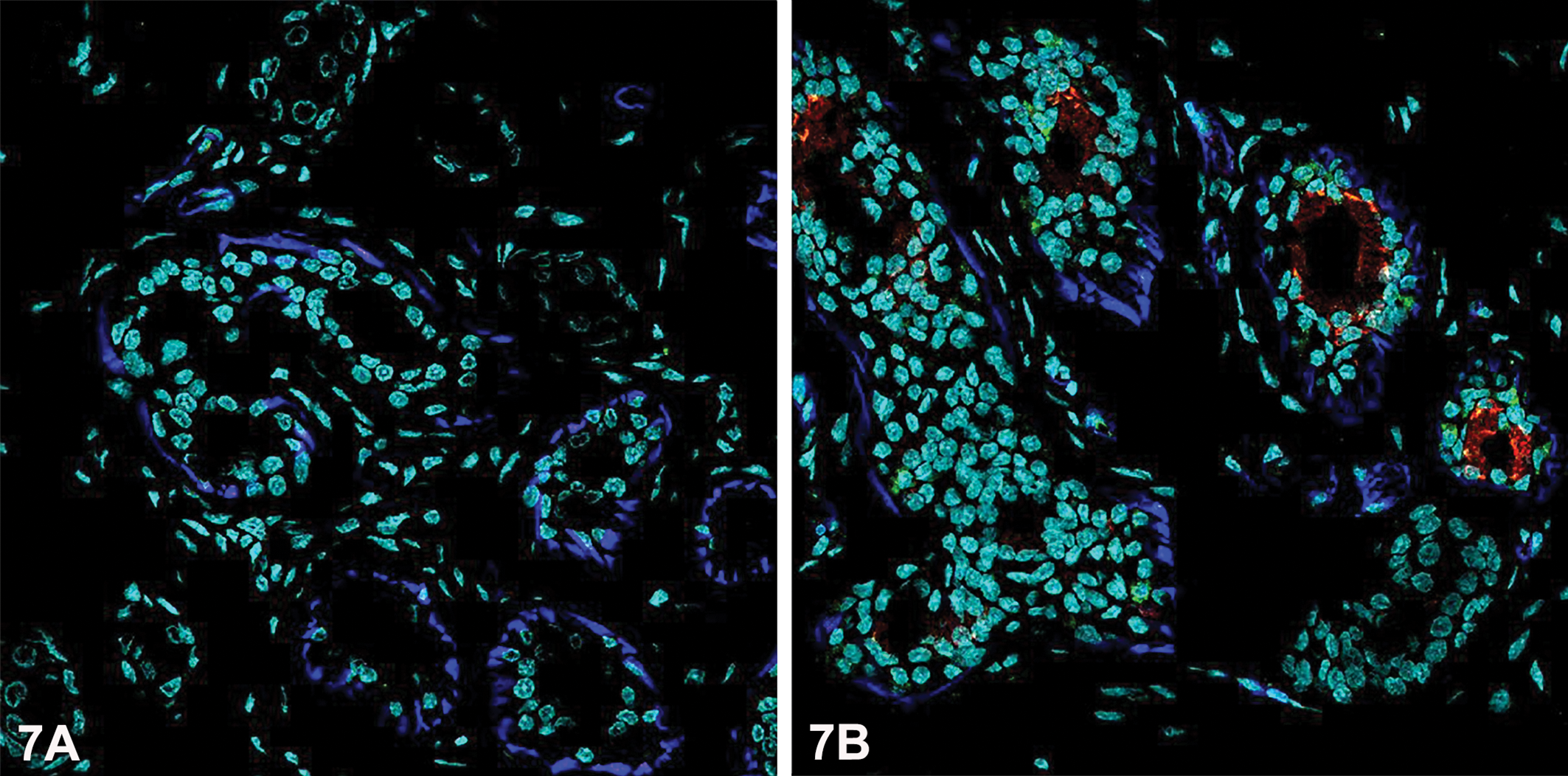
Immunofluorescence data showing NPY (green), NPY1 receptor (red), and smooth muscle actin (blue). Nuclear counterstaining is represented in a virtual channel (cyan). Eccrine glands from control animals (7A) show little or no staining. Increased expression of NPY between eccrine basal cells and NPY1 receptor in a rim at the apex of the inner epithelial cells were observed after treatment with vildagliptin at 80 mg/kg/day for 4 weeks (7B).
Discussion
Morphological Findings in the Skin of the Extremities
Vildagliptin-related skin lesions occurred on the distal extremities of monkeys at doses ≥5mg/kg/day within 3 weeks of treatment. They were dose-dependent and reversible if treatment was stopped prior to development of necrosis. Microscopically, skin lesions correlated with the time-dependent onset of medial and endothelial hypertrophy/hyperplasia of arterioles of the dermis. Although the observed vascular alterations could have theoretically been due to a direct effect of vildagliptin on arteriolar medial smooth muscle or endothelial cells, the clear and consistent distribution of lesions at the distal extremities combined with the fact that dermal arterioles at sites other than at the extremities were not affected, suggests that the primary event is pronounced vasoconstrictory activity at these sites.
The microscopic finding of endothelial hypertrophy/hyperplasia in cutaneous vessels of monkey digits correlated with an increased proliferation index in endothelial cells of the superficial dermis as observed by IHC for Ki67. Vildagliptin-treatment caused an increase in proliferation at doses comparable to where the histological findings were noted.
The in vitro inhibitory potency of the metabolite M20.2 for recombinant cynomolgus monkey DPP-4 activity is identical to the parent compound. M20.2 was a less potent inhibitor of recombinant cynomolgus monkey DPP-8 and DPP-9. Since plasma and skin concentrations of M20.2 were higher than concentrations of the parent compound, it must be assumed that M20.2 plays an important role in the manifestation of the skin lesions.
Recently, we showed in monkeys a distinct localization of DPP-4 in endothelial cells of various types of cutaneous vessels and eccrine sweat glands (Pantano et al. 2013). DDP-9 had a lower expression; DPP-8 selective antibodies were not available. The localizations of DPP-4 and DPP-9 correspond to that of cutaneous lesions observed as a result of vildagliptin exposure. Eccrine sweat glands in monkeys are particularly abundant in the affected skin regions including palms of hands, soles of feet, and on the forehead (Sato and Sato 1987; Elizondo 1988; Johnson and Elizondo 1974). Neither DPP-4 nor DPP-9 immunoreactivity was qualitatively altered by vildagliptin treatment in monkeys (data not shown). A better understanding of the physiological role of DPP-8 and DPP-9 may help explain the susceptibility of vascular beds in the distal extremities of monkeys.
Augmented Vasoconstriction in Cutaneous Arterioles of the Extremities
A vascular mechanism has previously been postulated for skin lesions in dairy calves (Coppock et al. 1989). These animals had areas of skin necrosis together with medial hypertrophy/hyperplasia of the arterioles related to the skin lesions. It was concluded that these lesions were due to chronic vasoconstriction due to food poisoning with ergot alkaloids caused by infection of the grasses by Claviceps.
In present experiments, the vasoconstriction hypothesis for the manifestation of monkey skin lesions was strongly supported by showing that skin effects were abolished or markedly reduced when the animals were housed at a higher ambient temperature closer to that found in the tropical regions from which this strain of monkeys is derived. These results suggest that lower ambient temperature increases vascular tone in these thermoregulatory areas of distal extremities and that this condition is exacerbated by vildagliptin exposure. This conclusion is also supported by the results of the cold stress experiments. The prolonged recovery time of the skin temperature following cold provocation indicates that an increased thermoregulatory vasoconstrictory response occurred in cutaneous arterioles of the extremities of vildagliptin-treated monkeys.
Increased Peripheral Sympathetic Tone
Cutaneous vasoconstriction at the extremities is a sympathetically mediated event. Present studies provided evidence that vildagliptin produces sympathetic stimulation in the monkey. In the cardiovascular telemetry study, vildagliptin produced increases in heart rate and blood pressure. The NOAEL for these hemodynamic vildagliptin effects was established at 3 mg/kg/day and was identical to the NOAEL for skin lesion manifestation. It appears reasonable to assume that the increases in blood pressure and heart rate following vildagliptin treatment are due to increased sympathetic activity. In support of this conclusion, a tendency toward an increased urinary norepinephrine concentration in monkeys dosed with 80 mg/kg/day vildagliptin was observed. In humans, Boschmann et al. (2008) showed that vildagliptin administration increased plasma norepinephrine concentrations in humans in response to meals.
The mechanism of the increased peripheral sympathetic tone following DPP-4 inhibition is not completely understood. Decreased metabolism of GLP-1 results in an increase in GLP-1 and that may be similar to a GLP-1 agonist. Previous studies in rats have shown a dose-dependent increase in blood pressure and heart rate associated with an increase in the peripheral sympathetic outflow following central and peripheral administration of a GLP-1 receptor agonist (Yamamoto et al. 2002). The authors suggest that the central GLP-1 system is a regulator of the peripheral sympathetic tone. GLP-1 is not only secreted from enteroendocrine L cells in the intestine (Creutzfeldt 2001) but is also synthesized by neurons in the caudal region of the nucleus of the solitary tract (Jin et al. 1988) and GLP-1 receptors are widespread throughout the brain (Jin et al. 1988; Larsen et al. 1997).
Vasoconstrictory actions of phenylephrine in cutaneous vessels of skin biopsies were not affected in vildagliptin-treated animals. These data support the conclusion that vildagliptin treatment did not influence postsynaptic mechanisms of the peripheral sympathetic system, that is, α-adrenoceptor activation and intracellular signaling.
Role of NPY
Another mechanism of increased peripheral sympathetic activity following DPP-4 inhibition was postulated by Walker et al. (1991) and it relates to NPY suppression of presynaptic release of norepinephrine. NPY is a 36-amino acid peptide coreleased with norepinephrine during sympathetic nerve activation. DPP-4 cleaves NPY1-36, which itself is a potent vasoconstrictor as an agonist for postsynaptic NPY1 receptors on vascular smooth muscle cells. It is postulated that inhibition of NPY1-36 degradation by DPP-4 inhibition and thus increasing NPY1-36 plays a role in the observed vasoconstriction in vildagliptin-treated animals. In sympathetic nerve endings at arterioles, the DPP-4-mediated degradation product NPY3-36 inhibits norepinephrine release via presynaptic NPY2 receptors without direct effects on the vasculature. Following DPP-4 inhibition by vildagliptin, it is conceivable that NPY3-36 formation will be reduced, which would attenuate inhibitory effects on presynaptic release of norepinephrine and which would then result in an increased norepinephrine release into the synaptic cleft. An increase in plasma norepinephrine could occur by increased spillover of norepinephrine from sympathetic synapses.
It is not evident how an elevation of NPY and NPY1 receptors in the eccrine sweat glands directly impacts the pathology observed in arterioles. Diffusion of NPY may contribute to signals detected in the surrounding arteriolar regions through less well-characterized paracrine functions. Autocrine/paracrine functions of NPY have been previously described in adrenal glands (Spinazzi et al. 2005).
Vildagliptin Effects on Ex Vivo Vascular Reactivity
Strong evidence for a pathomechanistic involvement of vasoconstriction in the manifestation of the skin lesions also stems from ex vivo analyses of vascular responses in skin biopsies from monkey digits. Following 4 weeks (but not 2 weeks) of daily vildagliptin administration, a significantly increased NPY-induced constriction of cutaneous arteries was observed, while vasoconstrictory responses to phenylephrine were unaffected by vildagliptin treatment. The latency in appearance of the augmented response to NPY is consistent with the onset of arteriolar smooth muscle hypertrophy/hyperplasia that was observed microscopically and the time of vildagliptin treatment necessary for the macroscopic manifestation of the skin lesions. It apparently takes 3 to 4 weeks of vildagliptin treatment to produce the changes in the vasculature that result in increased vasoconstrictory activity and eventually skin lesion formation. This conclusion is also consistent with the observation that addition of vildagliptin to the bathing solution did not affect NPY actions on digital arterioles from naive monkeys that were not pretreated with vildagliptin.
The selectivity of the increased vasoconstrictory effect of NPY correlates with the immunofluorescence data that revealed a selective increase in NPY and NPY1 receptors in the affected skin of vildagliptin-treated monkeys. The mechanism of the increased expression of NPY1 receptors in the skin of vildagiptin-treated monkeys is not currently understood but is consistent with earlier studies reporting that elevated NPY levels in vivo induced NPY1 receptor expression (Zukowska-Grojec et al. 1998; Hill et al. 2004; Kalra et al. 1998). The selectivity of the increased vasoconstrictory effect of NPY also suggests that the effect may be an on-target effect; NPY is the only known vasoconstrictor peptide that is a DPP-4 substrate.
Another relevant finding from the ex vivo vascular reactivity experiments was the enhancement of acetylcholine-induced endothelium-dependent vasodilation in arterioles from digital skin biopsies after 4 weeks (but not 2 weeks) of daily vildagliptin treatment. Acetylcholine activates endothelium to increase the generation of vasodilatory mediators, including NO and prostacyclin (Cohen and Vanhoutte 1995). The increased vasodilation correlates with what can be considered compensatory endothelial hypertrophy/hyperplasia in vildagliptin-treated animals. NPY may play a causal role in this process, since it has been shown that NPY stimulates endothelial cell proliferation (Steinhoff et al. 2003). Augmented dilation in response to acetylcholine after vildagliptin treatment might reflect an adaptive mechanism of enhanced activity of these mediators, which one would expect to play a protective role in the blood vessel wall.
Species Specificity of Skin Lesions
During the preclinical safety evaluation in support of the new drug application (NDA) submission, vildagliptin was given to mice, rats, and dogs for more than 4 weeks and at exposures (area under the curve [AUC], Cmax) that were comparable or higher than in the monkey studies. No skin lesions were observed in these species. Skin lesions as seen in the monkey studies were also not observed during the clinical development of vildagliptin (Ligueros-Saylan et al. 2010). Therefore, it appears that skin lesion formation following vildagliptin treatment is a species-specific phenomenon that is most likely related to the heightened sensitivity of the monkey’s cutaneous vascular system at comparatively low ambient temperatures for this species.
Limitations of This Study
Our investigations about mechanisms that seem to play a role in the manifestation of the skin lesions in monkeys were focused on the central role of DDP-4 inhibition. It is, however, not completely clear whether some non-DPP-4 mechanisms are involved. Cmax of vildagliptin and its metabolite M20.2 exceeded Ki values not only for DPP-4 but also for DPP-8 and DPP-9 in plasma and skin at doses that produced skin lesions. However, there is no data to support the conclusion that either DPP-8 or DPP-9 is involved. The physiological role of DPP-8 and DPP-9 and characterization of their respective endogenous substrates have not been well established. Previously, a causal association between inhibition of DPP-8 or DPP-9 and multiorgan histopathological changes and death in rodents has been postulated (Lankas et al. 2005). This has, however, been refuted by Burkey et al. (2008) who provided evidence that DPP-8/9 inhibition does not play a role in vildagliptin toxicity in rats.
Present experiments are limited to effects following vildagliptin treatment. It appears important to learn more about effects of other DPP-4 inhibitors on the parameters that were investigated in the present study. Of special interest would be to see whether there are differences between DPP-4 inhibitors that are known to produce skin lesions in monkeys, for example, vildagliptin and saxagliptin, and a DPP-4 inhibitor that does not cause monkey skin lesions, for example, sitagliptin.
The effect of DPP-4 inhibition by vildagliptin on NPY metabolism has not been investigated. Specifically, the reduced formation of NPY3-36 in the relevant periarteriolar compartment of vildagliptin-treated monkeys has not been measured.
Conclusion
Vildagliptin administration caused dose-dependent and reversible skin lesions involving the distal extremities of monkeys. Available data provide evidence that skin effects are of vascular origin and that effects on the NPY system together with an increased peripheral sympathetic tone play an important pathomechanistic role in the manifestation of these lesions. Since skin lesions similar to those observed in monkeys have not been observed in other species, including humans, available data support the conclusion that skin lesions following vildagliptin treatment are a monkey-specific phenomenon.
Footnotes
Acknowledgments
The authors wish to thank Diane Gunson and Steven Stefanski for their contributions as pathologists. The authors wish to thank Page Bouchard and Laura Dill Morton for their support to finalize the article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
