Abstract
In this study, it is aimed to determine the histopathological and haematological effects of apigenin, phloretin and myricetin on Wistar immature female rats using Tier 2 of the uterotrophic assay. The female rats were divided into 17 groups with 6 rats in each group. There was a negative control group and positive control dose groups that contained 0.07 µg/kg/day, 0.7 µg/kg/day and 7 µg/kg/day of ethinyl estradiol (EE), 0.7 µg/kg/day 17α-ethinyl estradiol + 1 mg/kg/day tamoxifen and genistein. The other dose groups contain 1 mg/kg/day, 10 mg/kg/day and 100 mg/kg/day of apigenin, myricetin and phloretin. All chemicals had been given to Wistar immature female rats with oral gavage for three consecutive days. At the end of the study, blood samples were analysed for haematological parameters. Tissue samples that were taken from the liver, kidney, spleen and thyroid were histopathologically and histomorphometrically examined. There were no significant differences between oil control and other dose groups for glomerular histomorphometry. However, there were siginificant differences for thyroid histomorphometry. Especially, 10 and 100 mg/kg/day of phloretin dose groups had a siginificant increase in colloid surface area in thyroid compared with the 1 mg/kg/day of phloretin and oil control groups. Significant histopathological changes (congestion, degeneration, fibrosis and mononuclear cell infiltration) were noted in the tissue specimens obtained from the treatment groups compared with the control group. According to the results of the haematological analysis of the groups, especially the values of erythrocytes and haematocrit were increased significantly in most of the dose groups according to the oil control group.
Introduction
It is commonly recognized that some plant-derived non-nutrients called ‘phytochemicals’ may provide important health advantages if added to the diet. 1 These phytochemicals are a large class of phytoestrogens, with ‘oestrogen-like’ biological properties. Phytoestrogens are non-steroidal substances present in all plants but present in relatively high concentrations in leguminous plants. 2 Phytoestrogens are categorized into groups according to their chemical structures. The most oestrogenic activity is found in flavones, flavonols, flavanones, lignans, chalcones and isoflavones. The most common of them are isoflavones and lignans which are found in fruits, vegetables and whole grains. 3 Biological oestrogen-like activities of phytoestrogens derive from their structural similarity to human oestrogen. Having weak oestrogen activity, phytoestrogens have been associated to have many potential health benefits including effect on cancer, vascular disease, osteoporosis and menopausal symptoms.
The most studied phytoestrogenic compounds are isoflavones and soy, such as genistein, formononetin, biochanin A and daidzein. 4 Phytoestrogens were first identified when the lambs that were grazing red clovers, which contain high rates of phytoestrogen, developed infertility with proven dangerous effects. 5 Apigenin, phloretin and myricetin are found in different plants with different concentrations and they can be connected to the oestrogen receptor. 6,7 The main part of phytoestrogens is conjugated to sugar residues and for their absorption, hydrolysis of the glycosides is necessary. 8
From a clinical perspective, phytoestrogens may be used most conveniently as first-line treatment for women who have vasomotor symptoms during early natural postmenopause. This is in accordance with the recommendations of the North American Menopause Society. 9 There are a lot of studies on the protective effects of the phytoestrogens, especially on the hormone-dependent cancers. It was observed that incidence of hormone-depending cancers is lower in some countries where the intake of phytoestrogens is higher. 10 Genistein shows anticancerogenic effects as antiangiogenesis and apoptosis in both in vivo and in vitro studies. There is a study on the effect of genistein on breast cancer cells and it shows that this molecule leads to partial blockage of oestrogen receptors, but its inhibitory activity on cancer cells is very complicated. 11 It was reported that there may be a decrease in endogenous steroid hormone levels because of phytoestrogens. 12,13 Phytoestrogens are known with similar to the molecular structure of oestrogen and linking to the oestrogen receptor from in vitro studies and it is also reported that they interact with the oestrogen receptor in living organisms and create a hormonal response. 6,14 Phytoestrogens are classified according to their chemical structure and estrogenic effects and they were studied on rodent species such as rat and mouse.
Phytoestrogens also have antioxidant effects 13 and they function as antioxidants and protect the cells against the oxidant-induced DNA damage. 15 It was observed that genistein increased antioxidant enzyme activities in rats. 16 Antioxidant effects of phytoestrogens are regarded to have significant beneficial effects because of the exposure to many agents that cause oxidative stress in the body. The flavonol subgroup is the most important group of flavonoids, and this group has antioxidant activity and strong physiological activity. 17 Miricetin belongs to the flavonol subclass and is found in some fruits, tea, wine, vegetables and medicinal plants. 18
Phytoestrogens have also been reported to affect steroid biosynthesis by regulating aromatase and 5α reductase activity in vitro. 19 In vitro experiments that were using benign breast cell lines, skeletal muscle cell lines and rodent models have found that phytoestrogens show a proliferation-enhancing effect. 20,21 Although these studies show that phytoestrogens can lead to cell proliferation, other in vitro and in vivo studies have showed a concentration-dependent anti-proliferative effect of isoflavones. 22
In vivo studies in rodents also show that low-dose dietary isoflavones put an end to tamoxifen-associated mammary tumour development. 23 Some studies indicated that increased intake of phytoestrogens develops markers of bone formation and resorption in postmenopausal women, 24–26 whilst in other cases this effect is absent. 27–29 After an intravenous injection of daidzein to rats (40 mg/kg/body weight (b.w.)), daidzein was found in high concentrations in the plasma, liver, lung and kidney and in lower concentrations in the spleen, heart and skeletal muscle. 30 Tissue dispersion of isoflavones was determined in two lactating ewes after being fed with red clover silage for 1 month. 31 However, data are limited.
The obtained experimental data from animal studies are highly suggestive of beneficial effects on health, but it has also been reported that when they are consumed in high amounts in humans and animals, they can have potentially harmful effects. 32
The uterotrophic assay is a short-term screening test, and it depends on 3-day application of chemicals. It contains two tiers. ‘Tier 1’ is used to identify chemicals that have the potential to interact with the oestrogen, androgen or thyroid hormone. The other one, ‘tier 2’, is necessary for identifying any adverse endocrine-related effects caused by the chemicals and finding a relationship between the doses and the endocrine effect. In this study, we used tier 2 screening, and it is aimed to determine the histopathologic and haematological effects of apigenin, phloretin and myricetin with three different doses in vivo. Also having to do with the application of these three phytoestrogens in immature female rats, it is aimed to determine the dose or doses that have histopathologic effects with tier 2 of the uterotrophic assay.
Materials and methods
Chemicals
17α-Ethinyl estradiol (98%), tamoxifen (99%), genistein (98%), myricetin (96%), phloretin (99%) and apigenin (95%) were purchased from Sigma-Aldrich (St Louis, Missouri, USA). For haematological analyses, the kit which was compatible with the haematology analyzer (MS9-5, Melet Schloesing Laboratories, France) for veterinary purposes was used.
Animals
In this study, female Wistar albino (Rattus norvegicus) rats were used, and they were purchased from the Experimental Animals Production Center, Hacettepe University, Ankara, Turkey. All experimental procedures and animals used were endorsed by the Approval of Ethics Committee of Hacettepe University. All animals were housed in polycarbonate cages with stainless steel covers in an air-conditioned room, and the experimental period was 3 days because of the procedure of tier 2 of the uterotrophic assay and the animals were provided with 12-h light/12 h dark cycle with a temperature of 22.4 ± 1.6°C and a relative humidity of 47.2 ± 1. The animals were provided with fooddevoid of any phytoestrogens and tap water ad libitum throughout the experimental period. The diet contained uncooked chicken egg, pasta, table salt, bone soup, corn oil and wood shaving.
Doses and administration of chemicals
The female offsprings were weaned off on day 18 following birth. After birth, on the 19th day (postnatal day 19 (PND 19)), the chemicals were applied to rats. The female rats were divided into 17 groups, each containing 6 rats. In the control groups, there were positive control and oil control groups. In the negative control group, animals were only given corn oil orally and in the positive control group, animals were given oral gavage administration of 0.07 µg/kg/day, 0.7 µg/kg/day and 7 µg/kg/day of 17α-ethinyl estradiol. Other groups were dosed by oral gavage with 0.7 µg/kg/day of 17α-ethinyl estradiol+ 1 mg/kg/day of tamoxifen, 1 mg/kg/day of genistein, apigenin, phloretin, myricetin, 10 mg/kg/day of genistein, apigenin, phloretin, myricetin and 100 mg/kg/day of genistein, apgenin, phloretin and myricetin (Table 1). We used α-ethynyl estradiol and tamoxifen as a positive control because they were recommended by Organisation for Economic Co-Operation and Development (OECD) according to Office of Prevention, Pesticides and Toxic Substances (OPPTS 890.1600): uterotrophic assay. They were dissolved in corn oil. Doses were given by gavage at the same time for 3 days. At the end of the study, the animals were killed under ether anaesthesia followed by capitation.
The animal groups and doses.
EE: ethinyl estradiol.
Histopathological analysis
The thyroid gland, liver, spleen and kidney tissues were removed and fixed in 10% formol. Later, they were transferred to 70% alcohol. The tissues were embedded in 55°C paraffin, cut at 4 µm thickness, stained with Harris haematoxylin and eosin and then examined under a Leica light microscope (Germany) for histopathological evaluation and photographed with imagine program Pixera Pro 150ES (Pixera Corporation, Santa Clara, California, USA).
Haematological analysis
To determine the haematological values, blood samples were taken from the hearts of female rats using sterile syringe into containers with ethylenediaminetetraacetic acid as an anticoagulant. Erythrocyte (RBC) count, haemoglobin (Hb) concentration, mean cell volume (MCV), haematocrit, mean cell Hb (MCH), mean cell Hb concentration, and leucocyte (WBC) count were measured using a haematology analyzer (MS9-5).
Histomorphometric measurement
On the photographs of the groups of thyroid gland, using Bab Bs200prop program, histomorphometric analysis was carried out with measurements of follicular cell height, follicular size and colloid area. 33
For thyroid gland, measured traits were:
Thyroid follicle surface area: A1, A2, … , A300,
Colloid surface area: B1, B2, …, B300,
Epithelium surface area: (A1–B1), (A2–B2), …, (A300–B300).
In kidney tissue, histomorphometric measurement of glomeruli was carried out in all groups. At least a hundred glomeruli for each group were selected; the shortest and the longest diameters for each glomerulus were measured using the Bs200prop program in an Olympus BX51 system light microscope (Japan). The glomerular volume was calculated using the following formula:
Statistical analysis
Statistical analyses were performed by using an SPSS 7.0 program. All values were examined by means of univariate analysis of variance, using a one-way factorial design. Dunnett Hsu’s test was used to detect differences among groups. Data are presented as mean ± standard deviation or n (%), where applicable. Incidences of histopathological findings were analyzed by Fisher’s exact test. The value of p < 0.05 was considered statistically significant.
Results
Histopathological analysis
The incidence of exposure-related histopathologic lesions of female rats in the control and treatment groups is given in Table 2. The oil control group showed regular morphology in all tissues. Histopathological examination of the liver, kidney and thyroid revealed significantly increased frequency of congestion when compared with the control group. Tubular degeneration was increased in EE + tamoxifen group compared with the oil control group. The 100 mg/kg/day of genistein dose group showed statistically significant increase in the frequency of congestion and degeneration in liver compared with the control group, 1 mg/kg/day and 10 mg/kg/day of genistein dose group (Figure 1). Also, in kidney tissue, there was significant increase in tubular degeneration and congestion for 100 mg/kg/day dose group of genistein (Figure 2). Again, in 100 mg/kg/day of genistein, in thyroid, there were more damaged follicles compared to the other groups of genistein. Groups given 100 mg/kg/day of apigenin and phloretin dose had significant increase in liver and kidney congestion. Groups given 100 mg/kg/day dose of apigenin, myricetin and phloretin had more damaged thyroid follicles when compared with the control group (Figure 3). Congestion in the kidney tissue in 10 mg/kg/day treatment group of phloretin was observed to be more than that in the kidney tissue in 10 mg/kg/day treatment group of apigenin. The group treated with 1 mg/kg/day of myricetin dose had damaged thyroid follicles compared with the groups treated with 1 mg/kg/day of apigenin, phloretin and genistein dose. Increased megakaryocytes were observed in all 100 mg/kg/day application dose groups in spleen, and it is found to be statistically significant when compared with the control group (Figure 4). Congestion was found to be statistically significant in thyroid tissue in 100 mg/kg/day of myricetin. Further, degeneration in all tissues of all treatment groups was increased compared with the control group.
Incidence of exposure-related histopathologic lesions of immature female rats in the control and treatment groups.

aNumber of rats with the lesion/number of rats examined.
b p < 0.05 (Fisher’s exact test): significantly different from the control group.

Photomicrographs showing liver tissues of control and treatment groups. (a) Oil control group (stained with contrast to H&E, ×200); (b) 100 mg/kg/day genistein showing congestion (black arrow, ×200); (c) 100 mg/kg/day apigenin showing congestion (black arrow) and degeneration (asterisk, ×400); (d) 100 mg/kg/day phloretin showing degeneration (asterisk, ×400); (e) 0.7 μg/kg + 1 mg/kg EE + tamoxifen showing mononuclear cell infiltration (black arrow, ×400); (f) 100 mg/kg/day myricetin showing degeneration (asterisk, ×400). (All of them were stained with contrast to H&E). H&E: haematoxylin and eosin. EE: ethinyl estradiol.
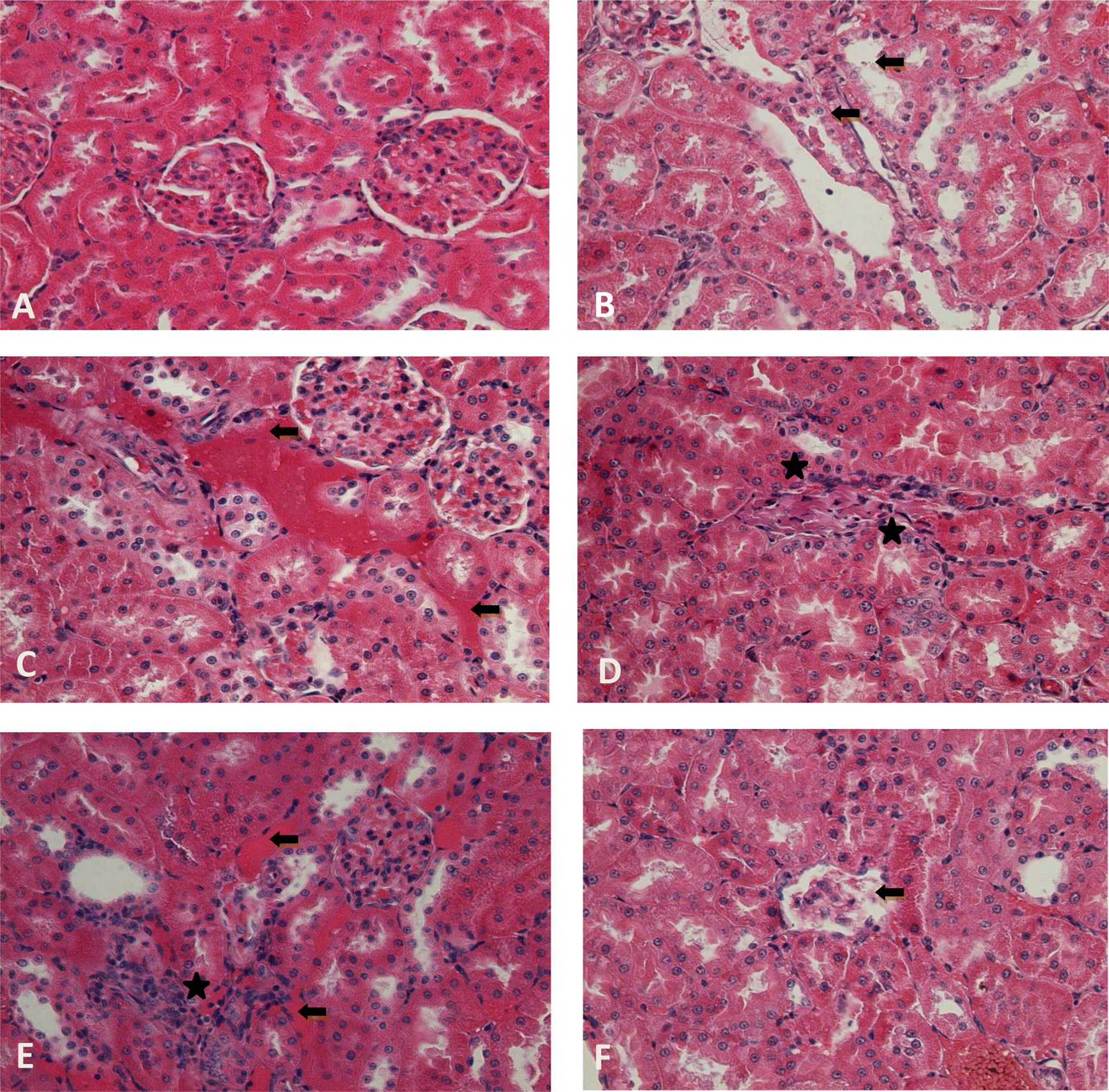
Photomicrographs showing kidney tissues of control and treatment groups. (a) Oil control group; (b) 0.7 μg/kg + 1 mg/kg EE + tamoxifen showing tubular degeneration (black arrow, ×400); (c) 100 mg/kg/day genistein showing congestion (black arrow, ×400); (d) 100 mg/kg/day apigenin showing mononuclear cell infiltration (asterisk, ×400); (e) 100 mg/kg/day phloretin showing congestion (black arrow) and mononuclear cell infiltration (asterisk, ×400); (f) 100 mg/kg/day myricetin showing degeneration (black arrow, ×400). (All of them were stained with contrast to H&E.) H&E: haematoxylin and eosin; EE: ethinyl estradiol.

Photomicrographs showing thyroid tissues of control and treatment groups. (a) Oil control group (×200); (b) 7 μg/kg EE showing colloidal degeneration (bracket) and damaged follicles (asterisk, ×400); (c) is 100 mg/kg/day genistein showing congestion (black arrow) and damaged follicles (asterisk, ×200); (d) 100 mg/kg/day apigenin showing congestion (black arrow) and colloidal degeneration (bracket, ×200); (e) 1 mg/kg/day phloretin showing damaged follicles (asterisk) and congestion (black arrow, ×200); and (f) 100 mg/kg/day myricetin showing congestion (black arrow) and damaged follicles (asterisk, ×200). (All of them were stained with contrast to H&E.) H&E: haematoxylin and eosin.
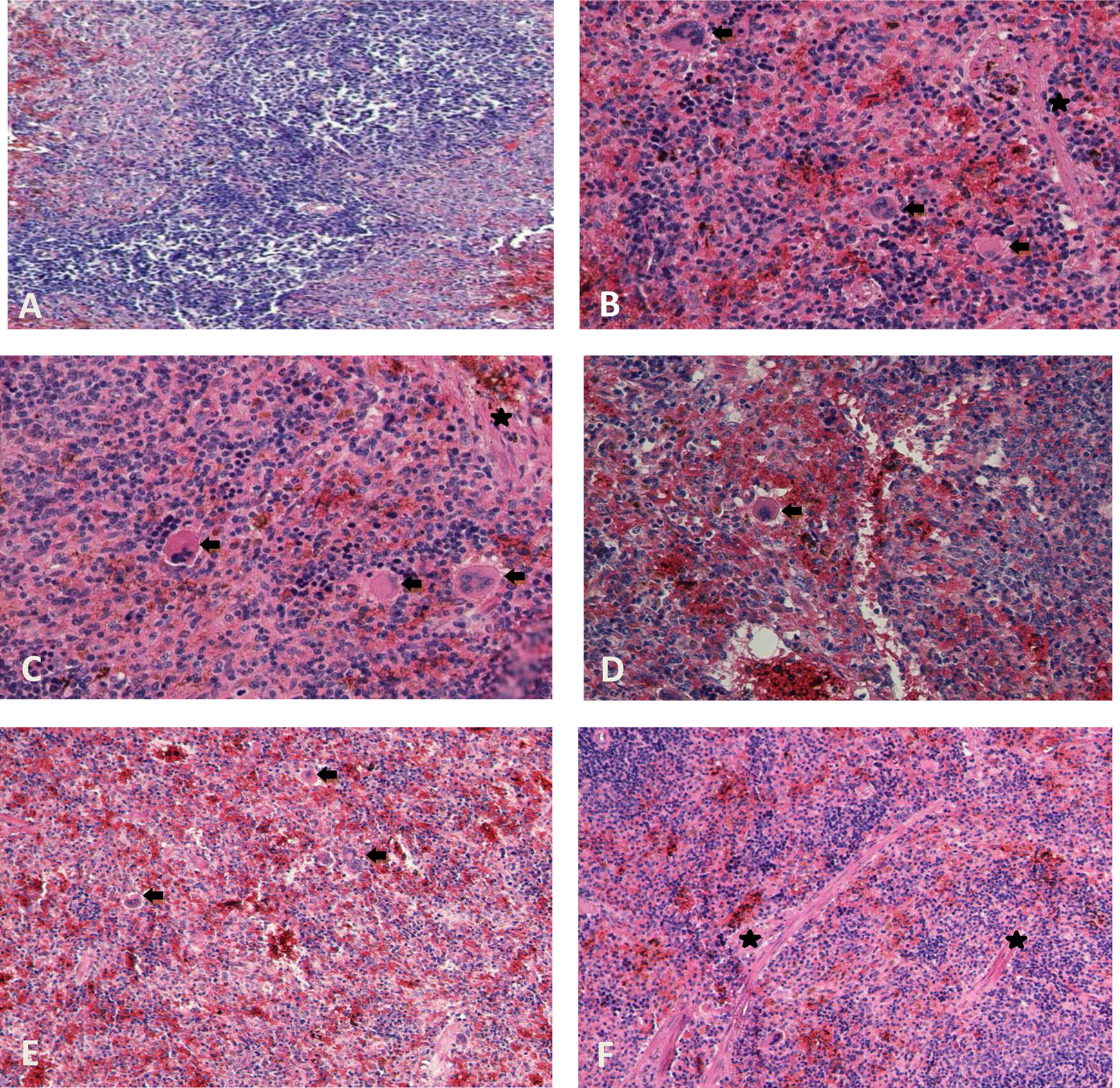
Photomicrographs showing spleen tissues of control and treatment groups. (a) Oil control group (×400); (b) 0.7 μg/kg + 1 mg/kg EE + tamoxifen showing increased number of megakaryocytes (black arrow) and fibrosis (asterisk, ×400); (c) 100 mg/kg/day genistein showing megakaryocytes (black arrow) and fibrosis (asterisk, ×400); (d) 100 mg/kg/day apigenin showing megakaryocytes (black arrow, ×400); (e) 100 mg/kg/day phloretin showing megakaryocytes (black arrow, ×200); (f) 100 mg/kg/day myricetin showing fibrosis (asterisk, ×200). (All of them were stained with contrast to H&E.) H&E: haematoxylin and eosin.
Haematological analysis
The results of the haematological analysis are presented in Table 3. According to the results of the analysis of the groups, especially the values of erythrocytes and haematocrit were increased significantly in most of the dose groups according to the oil control group. MCV, MCH and Pct (Platelet crit) were decreased in many of the groups compared with the oil control group. Especially, in 7 µg/kg/day of EE and 100 mg/kg/day of genistein dose groups, there were significant differences in the blood values. However, myricetin dose groups had almost the same values with oil control group and did not show any significant differences.
Heamatological analyses of oil control, positive control and other dose groups.

MCH: mean cell haemoglobin; Hb: haemoglobin; MCV: mean corpuscular volume; MCHC: mean corpuscular haemoglobin concentration.
a p < 0.05 significantly different from control group; n = 6.
Histomorphometric measurement of thyroid and kidney tissue
The results of histomorphometric analysis of kidney and thyroid are given in Tables 3 and 4. According to the results of the analysis of groups, 0.07 µg/kg/day of EE and 1 mg/kg/day of phloretin dose groups had significant difference statistically compared with the oil control group in follicle surface area. Then, 0.7 µg/kg/day + 1 mg/kg/day of EE + tamoxifen dose group were decreased in colloid surface area compared with the control group. Also, 10 and 100 mg/kg/day of phloretin dose groups had a siginificant increase in colloid surface area in thyroid compared with the 1 mg/kg/day of phloretin and oil control group. Furthermore, there was an increase in colloid surface area in 1 mg/kg/day and 100 mg/kg/day of myricetin dose groups. According to epithelium surface area for all groups, there was significant decrease in 10 mg/kg/day of genistein, 1 mg/kg/day of phloretin, 1 mg/kg/day and 100 mg/kg/day of myricetin dose groups compared with the oil control group.
Mean values of the thyroid morphometric characteristics according to dose groups.a

EE: ethinyl estradiol.
aData are mean ± SE.
b p < 0.05 significantly different from control group.
Glomerular histomorphometry revealed no significant differences between the control and treatment groups with respect to the analysed glomerular parameters (Table 5).
Histomorphometric measurement of glomeruli in the treatment and control groups.a

EE: ethinyl estradiol.
aData are mean ± SE.
Discussion
The current study was conducted in order to investigate the haematological and histopathological effects of some phytoestrogens in the liver, kidney, spleen and thyroid of the female rats.
Structurally, genistein and daidzein are polyphenolic compounds, similar to estradiol-17 and bind with a weaker potency to both types of estrogen receptors. Despite a lot of beneficial effects of soy isoflavones, epidemiological and experimental data also have been showing an adverse effect on human health, especially on reproductive and thyroid axis. The association between high soy isoflavones intake and goitrogenesis, also protective effect of sufficient iodine intake, was reported both in humans 36 and in different animal models. So, besides the direct beneficial effect of soybean phytoestrogens on bone tissue, isoflavones may also act indirectly, through endocrine disruption and interference with hypothalamic–pituitary–thyroid axis. Genistein and daidzein prevent the activity of thyroid peroxidase, the key enzyme in the synthesis of thyroid hormones. 37
There were statistically significant increase in 100 mg/kg/day of genistein dose group in thyroid about the damaged follicles compared with the oil control and low doses of genistein treatment groups. Also, in the histomorphometry of thyroid, there was an increase in the colloid surface area in 100 mg/kg/day of genistein. These damaged follicles may be caused due to this increase in histomorphometry. We saw the same thing in phloretin and myricetin high-dose groups. Moreover, these dose groups also had statistically significant damaged follicles. The isoflavones genistein and daidzein mediate most of their biological effects through the modulation receptors of stress–response pathways or oestrogen-dependent pathways rather than through their well-known antioxidant 38 or tyrosine kinase inhibitory properties. 39 Also there were statistically significant increased megakaryocytes in the spleen in 100 mg/kg/day of three phytoestrogens, but the incidence of fibrosis wasn’t statistically significant. This increase was compatible with the heamatalogical analysis. In liver, 10 and 100 mg/kg/day of apigenin, phloretin and myricetin dose gorups, congestion and degeneration were increased but mononuclear cell infiltration didn’t show significant changes statistically.
The affinity of phytoestrogens for oestrogen receptors results in effects on a large number of oestrogen-regulated systems, including the cardiovascular, metabolic, skeletal, reproductive and central nervous systems. In this study, we saw a lot of histopathological changes in the examined tissues. Especially in all high doses of phytoestrogens, congestion and degeneration had significant increase. In the histomorphometry of kidney, there were increases and decreases about all parameters compared with the oil control group but they were statistically significant. In spite of the statistically significant tubular degeneration of kidney in dose groups, the histomorphometry wasn’t significant. Most studies have evaluated the effects of soy-derived compounds on adiposity. For example, Long-Evans rats or ovariectomized ddY mice fed with a soy-rich diet had lost weight and had less adipose deposition than those fed on a soy-free diet. 40,41 According to our study, in thyroid tissue, increase in the frequency of fatty tissue deposition wasn’t significant. Potential mechanisms by which dietary soy may improve glucose metabolism have been recently proposed. Dietary soy increases insulin sensitivity by increasing glucose uptake in skeletal muscles. 42 This improved insulin responsiveness in mice exposed to dietary soy may take place by an improvement of the PI3K-Akt signaling by the dietary soy-activated 5′adenosine monophosphate-activated protein kinase. 42 In vivo studies on animal models have shown that low-dose dietary isoflavones block the action of tamoxifen in mammary tumour prevention 23 and genistein has been shown to reduce the growth inhibitory effects of aromatase inhibitors on breast cancer cells. 43 Intake of a high concentration of flavonoids with food (e.g. mirisetin and kuersetin) prevents proteolysis in the intestine and causes reduction in glucose intake, food intake and mineral absorption. 44 Branham et al., 14 determined the uterine oestrogen receptor binding affinities of phytoestrogens and mycoestrogens in rats. According to the analysis, genistein had the highest oestrogen receptor-binding affinity and then phloretin and the last was apigenin. Myricetin had the lowest binding affinity than the others. In a study by Barlas et al., 45 uterine lengths were increased in EE 0.7 mg/kg/day and 7 mg/kg/day dose groups, EE + tamoxifen and 100 mg/kg/day genistein and myricetin. Compounds having oestrogen activity not only caused thickening of the uterine but also an increase the length of the uterine, so the volume of the uterine also increased. The dose groups that have caused an increase in uterine wet weight also caused an increase in the height of luminal epithelial cells. There was no increase in the uterine weight in 1 mg/kg/day myricetin dose but a significant increase in luminal epithelial cell height was determined.
In this study, to investigate the effects of phytoestrogens in blood, haematological analysis was performed. Especially, the basic parameters such as leucocytes, erythrocytes, Hb and platelets were recorded and statistically analysed. In particular cases, such as infection-determining tests, monocytes, lymphocytes and the total value of leucocyte were important 46 , so in this study the dose groups were evaluated with the oil control group in terms of whether or not any difference. The groups given 7 µg/kg/day EE and 100 mg/kg/day genistein dose groups had a decrease in the percentage of lymphocytes according to the oil control but an increase in the percentage of monocytes have been identified. 100 mg/kg/day of Phloretin dose group had a significant increase in the percentage of monocytes in the blood. To diagnose diseases such as anaemia, erythrocytes and Hb, the percentage of haematocrit have great importance. In this study, Hb values had a similar level with the oil control group but erythrocyte and haematocrit values increased according to the oil control. These increases were seen in all the dose groups of apigenin, phloretin and genistein but myricetin dose groups had no difference with the oil control group. Haematological parameters evaluated in the analysis showed that these phytoestrogens and EE caused changes in the parameters of blood. Haematologic values of the rats in all myricetin dose groups had no significant differences from the oil control group.
The lack of compatible results across all species is surprising, but it depends on differences of soy foods/isoflavone supplements ingested, route of administration, time of exposure and phytoestrogen metabolism, so they may well result from a combination of these factors. There are many explanations for the inconsistent results from these studies. Although experiments of phytoestrogen interventions are countless, most experiments include only small numbers of subjects, are short in time and are poor in quality. Interventions investigated vary in type (food additives, dietary manipulation and packaged supplements) as well as in concentration and balance of active ingredients. Absorption of phytoestrogens is also different between individuals and affected by other factors such as antibiotic use. 47
When phytoestrogens are taken into the body by natural ways, they do not reach the toxic levels, but when used as dietary supplements, they will be in high doses in the body, which affect the endogenous oestrogen levels or activities, thereby changing the shape or level of the responses. So, limiting the marketing of supplements and public awareness is required. Phytoestrogens and their products are natural and herbal but their overconsumption will make adverse effects on tissue and organs.
In conclusion, there were many histopathologic effects of apigenin, phloretin and myricetin in kidney, spleen, thyroid and liver according to the amount of doses compared with the oil control group. Heamatological analyses of apigenin, phloretin and myricetin were statistically significant especially in 100 mg/kg/day dose groups. Also, histomorphometric analyses supported these findings. Finally, we concluded that there were histopathologic and haematological effects of these three phytoestrogens but further long-term experiments are required to establish the magnitude and significance of the effects of phytoestrogens, especially the effects of concentrated forms of phytoestrogen extracts, their toxicity and drug interactions, before they can be safely used as health supplements.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research was supported by Health Sciences Research Group of TUBITAK (Project number is 111S013).
