Abstract
Differences in the responses of conventional and germfree male Sprague-Dawley rats to acute injury induced by alpha-naphthylisothiocyanate (ANIT), a well-characterized biliary epithelial toxicant, were evaluated. Conventional and germfree rats were dosed once orally with 50 mg/kg of ANIT or corn oil alone and serially sacrificed daily for the next 3 days. Germfree rats treated with ANIT tended to have greater increases in virtually all liver and biliary-related analytes compared with conventional rats treated with ANIT; however, significant differences were found only in a few of these analytes including increased bile acids on day 3, total bilirubin on day 4, glutamate dehydrogenase (GLDH) on day 3, and reduced paraoxonase 1 (PON1) on days 2 and 3. Histologic differences between the conventional and germfree rats were modest, but most pronounced on day 2 (24-hr post dosing). Based on subjective scoring, biliary necrosis, neutrophilic cholangitis, and portal tract edema were more severe in germfree rats at 24 hr post dosing compared with conventional rats. Biliary epithelial replication did not differ between treated groups, however. Overall, germfree rats had a modestly greater level of biliary tract injury based on subjective histologic scoring and clinical chemistry measurements following an acute exposure to the well-characterized biliary toxin, ANIT; however, the difference between the ANIT-treated germfree and conventional groups was modest and most evident only within the first day following exposure. These findings suggest that the microbiome did not significantly affect ANIT-induced acute biliary tract injury in the conditions of this study.
Introduction
This study was designed to evaluate the influence of the gastrointestinal microbiome on the acute toxicity of alpha-naphthylisothiocyanate (ANIT), a well-characterized biliary epithelial toxicant, in conventional and germfree male Sprague-Dawley rats. The collection of intestinal bacteria, termed the microbiome, is now recognized as an important mediator of a broad variety of bodily functions, including drug metabolism, body weight, intestinal inflammation, and carcinogenesis (Meinl et al. 2009; Nicholson et al. 2012; Wilson and Nicholson 2015; Bjorkholm et al. 2009). The microbiome affects the liver in particular in several ways. Both phase 1 and phase 2 drug metabolism in the liver and enterohepatic circulation of metabolites are affected by the microbiome. For example, in liver and the gastrointestinal tract, enzymes involved in phase 2 metabolism, sulfotransferases, are moderately elevated in germfree rats of both genders and one of the hepatic epoxide hydrolasae enzymes (EPHX2) in female rats. Bacterial metabolism can produce novel metabolites of xenobiotics as well as endogenous substances such as secondary bile acids by the actions of colonic bacteria that lead to partial dehydroxylation and removal of the glycine and taurine groups on primary bile acids (Meinl et al. 2009; Lhoste et al. 2003; Overvik et al. 1990). Intestinal bacteria can biotransform a variety of xenobiotics, altering their bioavailability and toxicity (Wilson and Nicholson 2009; Swann et al. 2009). Additionally, enterohepatic circulation of some compounds can be modified by bacterial metabolism, affecting the hepatic exposure to various metabolites (Zuo et al. 2006). Intestinal bacteria are the source of endotoxin, which can trigger hepatic or systemic injury and inflammation alone, and endotoxin-driven inflammation has been shown to accentuate some types of drug-induced hepatic injury as well (Deng et al. 2009; Luyendyk et al. 2006; Zou et al. 2009). There are only a few studies in which the modulation of toxicity by germfree rodents has been evaluated and in these studies the health consequences of the germfree state were not consistent. Spontaneous liver tumors were reduced in germfree C3H/He mice compared to mice with conventional flora (Rowland 1988). However, the microbiome was likely to reduce the carcinogenicity of chromium (Upreti et al. 2004). Hydrazine-induced toxicity was more severe in germfree rats than conventional rats, although there was no evidence of hepatic injury (Swann et al. 2009). The impact of the total absence of the microbiome in toxicant-induced biliary tract injury has not been characterized previously. Consequently, we have compared the acute toxicity of a classic biliary epithelial toxin, ANIT, in germfree and conventional rats.
Methods
Animals
In all, 36 male Sprague-Dawley™ Germfree rats (SDGF; Taconic, Germantown, NY) and 30 male Sprague-Dawley™ (NTac: SD; Taconic, Germantown, NY) rats were used in this study. The rats were approximately 8 weeks of age and weighed 150 to 400 grams at the initiation of dosing. Groups of 15 conventional and 18 germfree rats received a single dose by oral gavage of either vehicle (corn oil) or 50 mg/kg ANIT on day 1 (Figure 1).
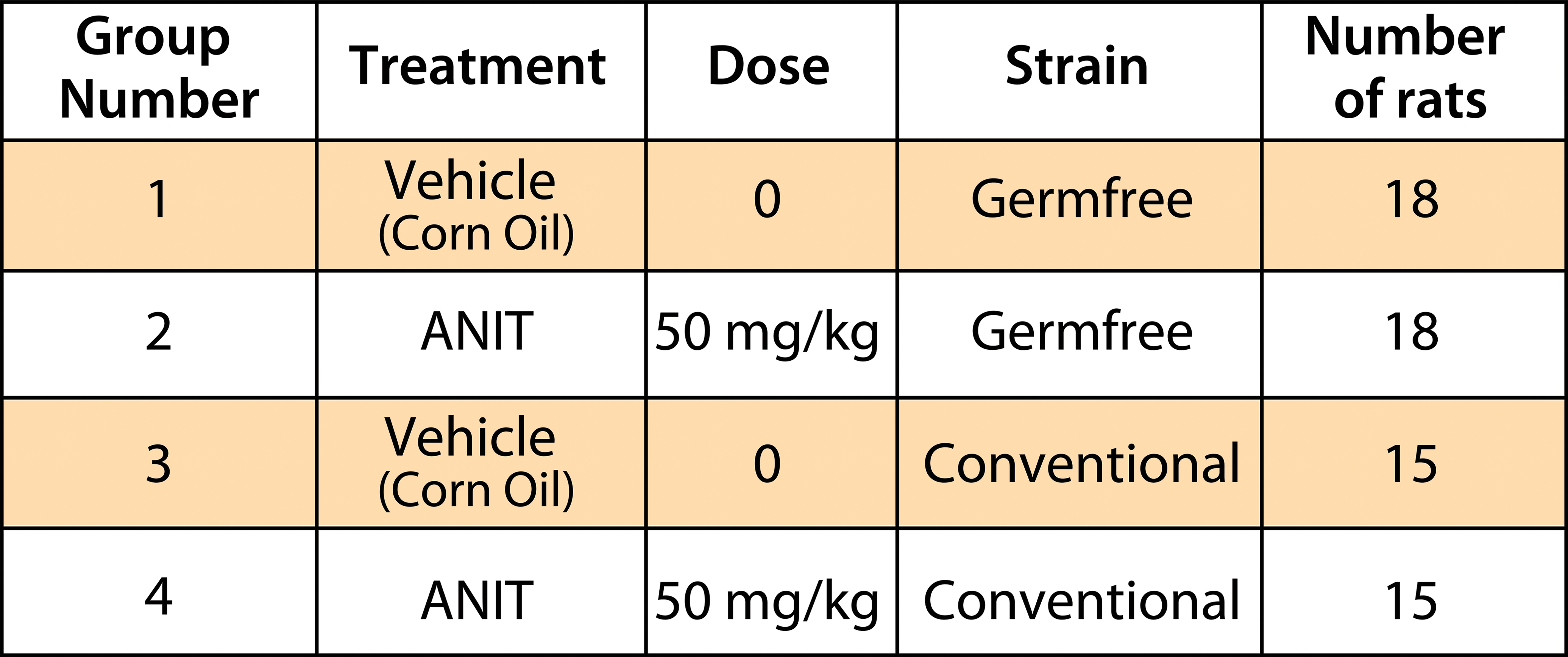
Group numbers and treatments for alpha-naphthylisothiocyanate-treated and conventional male rats.
The NTac:SD rats were group-housed at the GlaxoSmithKline (GSK) animal facility (2–3/cage) in polycarbonate solid-bottom cages with ALPHA-dri™ (Shepherd Specialty Papers, Inc., Kalamazoo, MI) in an environment with a temperature of 64–79°F, 30–70% relative humidity, and a 12-hr light/dark cycle. The SDGF rats were group-housed at the North Carolina State University (NCSU) gnotobiotic facility (3–5/cage; up to 4 cages per isolator) in polycarbonate solid-bottom cages with ALPHA-dri in animal rooms with a temperature of 64–79°F, 20–70% relative humidity, and a 12-hr light/dark cycle (the temperature and humidity inside the isolators were not monitored). Rats housed at NCSU and GSK were fed Prolab™ RMH 3500 Autoclavable Rodent Chow, made by PMI™ Nutrition International, Richmond, Indiana, ad libitum. Municipal water supply with additional treatment by reverse osmosis was available ad libitum from an automatic watering system for animals housed at GSK. Municipal water supply with additional autoclave treatment for sterilization was available ad libitum from individual water bottles for each cage for SDGF animals housed at NCSU.
Each animal at the GSK and NCSU facilities was identified by an implantable transponder identification device. The randomization program of Provantis NT Client Server Oracle was used to randomly allocate animals in the GSK facility to groups while balancing body weights across groups. Rats at the NCSU facility were allocated to groups based on shipment, such that all rats that arrived in an individual Taconic germfree shipper were placed into the same isolator, and all animals in an individual isolator were assigned to the same group.
All procedures involving animal care and use were conducted in accordance with published guidelines (National Research Council’s Guide for the Care and Use of Laboratory Animals) and the U.S. Department of Agriculture’s Animal Welfare Act and were reviewed and approved by GSK’s or NCSU’s Institutional Animal Care and Use Committee.
Clinical signs and food consumption were assessed daily. At necropsy, the body weights and liver weights were determined and the liver-to-body weight ratio was calculated. Stomach weights were also collected as a measure of potential retention of food via microbiome status or treatment-related ileus, as this could falsely elevate body weight measurements. Stomachs, with contents, were removed, separated from the esophagus and the duodenum and weighed.
Compounds
ANIT (Sigma-Aldrich, St. Louis, MO, Lot 08420DD) was formulated as a suspension in corn oil and administered to rats at a dose volume of 10 ml/kg. The vehicle and ANIT dose formulations were sterilized using a nonpyrogenic filter system with a 0.22-μm cellulose acetate, low protein binding membrane.
Tissue Collection
On days 2–4 (approximately 24, 48, 72 hr, respectively, after ANIT or vehicle administration), groups of 6 germfree and 5 conventional rats were euthanized by exsanguination after administration of isoflurane anesthesia. Animals were fasted overnight prior to euthanasia. Samples of the left lateral and the median lobe of the liver were collected and immersed in 10% neutral buffered formalin. Following fixation, liver sections were processed into paraffin blocks. Six-micron sections were stained with hematoxylin and eosin for microscopic examination.
Histopathology
Liver sections were evaluated using a subjective scale to score hepatic and biliary injury. Biliary and hepatic lesions were scored as follows: 0, no evidence of abnormality; 1, minimal; 2, mild; 3, moderate; and 4, marked manifestation of the lesion. Biliary necrosis ranged from scattered individual necrotic epithelial cells (scored as 1) to complete absence of viable biliary epithelial cells in interlobular bile ducts (scored as 4). Biliary neutrophilic infiltration varied from 1 to 3 neutrophils within or immediately adjacent to the biliary epithelium of an individual bile duct (scored as 1) to greater than 25 neutrophils within the lumen or epithelium of the bile ducts (scored as 4). Fibroblast proliferation referred to the circumferential proliferation of spindle-shaped cells (possibly myofibroblasts) around bile ducts; scoring ranged from the presence of 1 to 2 cell layers of reactive fibroblasts (scored as 1) to more than 6 layers of cells (scored as 4). Hepatic necrosis ranged from scattered individual necrotic cells (scored as 1) to confluent areas of hepatocellular necrosis that bridged between lobules (scored as 4). Biliary hyperplasia was graded using the same subjective scale lesions ranged from the presence of isolated clusters of biliary epithelial cells in residual ducts in a minority of portal tracts (scored as 1), to clusters of biliary epithelial cells in the majority of residual biliary ducts (scored as 2), to an increase in bile duct profiles, up to 4 profiles, in most portal tracts with small caliber ducts evident in the parenchyma separate from the portal tracts (scored as 3) and the presence of more than 4 profiles of bile ducts in the portal tracts with small caliber ducts evident in the parenchyma separate from the portal tracts (scored as 4). Only differences in the group mean subjective scores for each diagnosis of >0.4 units were interpreted as meaningful.
Clinical Chemistry
Whole blood was collected from the vena cava at necropsy. Serum was analyzed on an Olympus AU640e analyzer (Olympus America, Melville, NY). The following parameters were measured: alanine aminotransferase (ALT), alkaline phosphatase (ALP), aspartate aminotransferase (AST), total bilirubin (TBIL), PON1, total bile acids (BILE), GLDH, and lactate dehydrogenase (LDH).
Proliferation Assays
Immunohistochemical staining was performed on a Ventana Discovery XT (Tucson, AZ). Sections of formalin-fixed and paraffin-embedded liver were dual labeled with rabbit polyclonal antibody specific for pan cytokeratin AE1/AE3 (Abcam Inc., Cambridge, MA) to identify biliary epithelium and rabbit monoclonal antibody specific for the proliferation marker Ki-67 (Clone SP6, Lab Vision Corp., Fremont, CA) to identify proliferating biliary epithelial cells and hepatocytes. Ki-67 and pan cytokeratin-labeled slides were pretreated with ER2 (Ethylenediaminetetraacetic acid [EDTA]/TRIS pH9, Ventana Medical Systems, Tucson, AZ). Ki-67 slides were labeled with Biocare Rabbit on Rodent HRP-Polymer (Biocare Medical, Concord, CA), and developed with Leica Bond Max Refine DAB chromogen (Leica Microsystems Inc., Bannockburn, IL). Pan cytokeratin slides were labeled with Biocare Rabbit on Rodent AP (Biocare Medical, Concord, CA), and developed with Leica Bond Max AP Red chromagen (Leica Microsystems Inc., Bannockburn, IL). Sections were counterstained with 4’,6-diamidino-2-phenylindole (DAPI; Invitrogen, Carlsbad, CA) to label the nuclei.
A quantitative assessment of immunohistochemical labeling in the liver was performed using the iCyte™ Laser Scanning Cytometer (CompuCyte Corporation, Cambridge, MA). Proliferation of biliary epithelial cells was assessed by automated quantitation of Ki-67-labeled nuclei in cells that were also labeled with the AE1/AE3 pan cytokeratin antibody (indicative of biliary origin). Hepatocyte proliferation was also assessed using Ki-67 labeling; these cells had a characteristic histologic appearance and were not labeled with cytokeratin markers. The total number of DAPI-stained nuclei was counted for each liver section. This population of nuclei was further restricted through the use of gating that was applied to scatter plots of nuclei area versus circularity. In addition, laser light absorbance levels around each nucleus collected using peripheral contours was taken into consideration. Hepatocyte nuclei tended to be larger, more round and had a moderate level of scatter produced by the hepatocyte cytoplasm in contrast to nuclei of other cell types that were smaller, less round, and had little to no cytoplasm. Once gating was applied, region galleries were examined to ensure accurate segmentation of hepatocytes. Multiple test scans were conducted until the optimal placement regions and segmentation of hepatocyte nuclei was achieved. Hepatocytes that were Ki-67 positive were plotted and separated in a similar manner by factoring in the laser absorbance of the Ki-67 positive cells. These 3-3′-diaminobenzidine (DAB)-stained cells had higher absorbance levels than the Ki-67 negative, hematoxylin-stained hepatocytes that were less dense.
Within the bile ducts, Ki-67-positive and Ki-67-negative nuclei were segmented based on the amount of cytokeratin staining in the peripheral contours. Cells stained with the pan cytokeratin antibody were counted as bile duct epithelial cells. Proliferating cells were separated from nonproliferating cells based on Ki-67 DAB labeling. With this method, there was the potential for a peribiliary proliferating cell that is closely adjacent to a bile duct to be counted as a duct cell if the peripheral contour contained cytokeratin staining; however, the peripheral contours were set in close proximity to the primary event nucleus to make this a rare occurrence. Gallery images were examined of regions to confirm optimal placement of gated regions in the scatter plots. A labeling index for hepatocytes and biliary epithelium was developed using the ratios of Ki-67-stained and Ki-67-unstained biliary epithelium and hepatocytes at each time point.
Glutathione Levels
Approximately 0.5-ml whole blood was collected from each animal in groups 1 through 4 into a tube containing potassium EDTA. Immediately after collection, all samples were mixed well and placed on ice. Samples were centrifuged at approximately 2,500 g for 20 min to obtain a pellet of red blood cells (RBCs). The RBCs were washed with saline and stored frozen at or below −70°C. RBC samples from NCSU were transported on dry ice to GSK for analysis. The RBC samples were analyzed using a commercial kit (Bioxytech GSH/GSSH-412,
Statistics
Some clinical chemistry parameters were log transformed (PON1 and LDH) and were analyzed via a one-way analysis of variance (ANOVA; Littell et al. 2002; GLM Procedure of SAS/STAT 1999). For those parameters, which a transformation could not satisfy, the homogeneity of variance assumption (ALT, GLDH, AST, ALP, BILE, and TBIL) was analyzed via a one-way Welch ANOVA (GLM Procedure of SAS/STAT 1999; Littell et al. 2002). All tests were performed at the 5% level of statistical significance. The Hochberg multiplicity adjustment was applied to avoid erroneously rejecting the null hypothesis. Statistical analyses were not conducted on body weight, food consumption, glutathione levels, or organ weight data.
Results
Glutathione Levels
Group mean glutathione levels in RBCs were generally similar across time points, treatments (vehicle vs. ANIT), and strains (conventional vs. germfree), ranging from 459 to 642 μM (data not shown).
Clinical Signs, Body Weights, and Food Consumption
There were no clinical signs noted related to ANIT treatment in either the conventional or germfree rats.
ANIT-treated conventional rats euthanized on days 2 and 3 (24 and 48 hr post dose, respectively) lost up to 13.96 g of body weight beginning on day 1, whereas concurrent vehicle-treated conventional rats gained up to 15.16 g from days 1 to 3. By day 4, the ANIT-treated conventional animals began to show some recovery of body weight.
Germfree rats given vehicle did not gain as much body weight as conventional rats over the course of the study; however, the germfree rats given ANIT had slightly higher percentage of body weight loss than the conventional rats treated with ANIT.
Food consumption correlated with the body weight changes for the conventional rats, as ANIT-treatment had average food consumption decreases of 0.47-fold (compared with concurrent controls) from days 1 to 3 with no change in food consumption from days 3 to 4. Correlating decreased food consumption for ANIT-treated germfree rats of as much as 0.18-fold (compared with concurrent controls) was noted between days 1 and 4.
Organ Weights
Absolute and relative liver weights were increased on day 4 for ANIT-treated germfree rats (1.08 and 1.16-fold, respectively) and conventional rats (1.26 and 1.38-fold, respectively) compared with their respective vehicle-treated controls (Figure 2). Relative liver weights were also increased on day 3 for ANIT-treated germfree rats (1.15-fold). Absolute liver weights were similar in all groups until day 4 when the absolute liver weight of the ANIT-treated conventional rats (14.9 ± 1.2 g) exceeded that of the untreated conventional rats (11.8 ± 0.7 g)
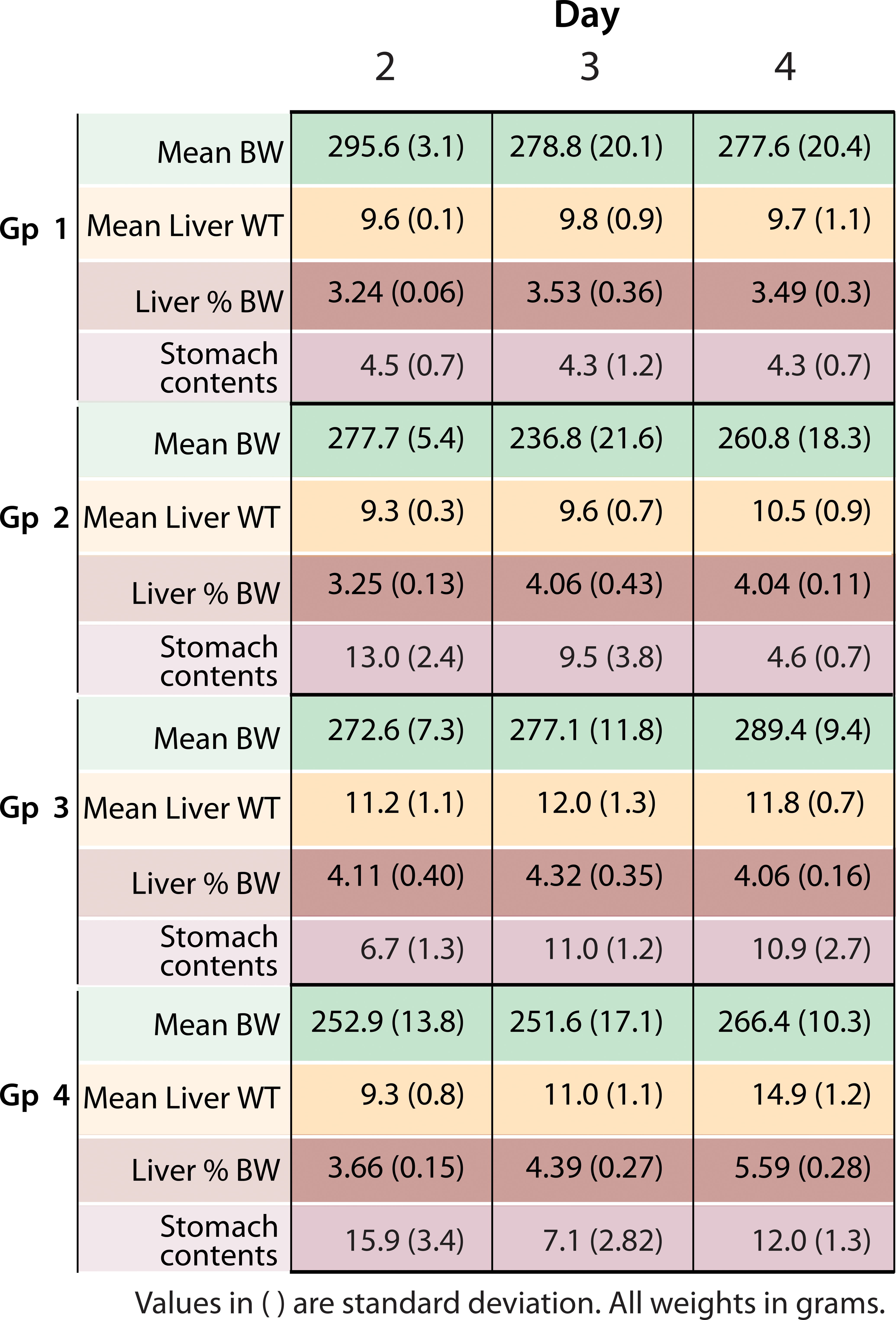
Mean body, stomach, and liver weights with liver percent body weight in alpha-naphthylisothiocyanate-treated and control male rats.
Absolute and relative stomach (with contents) weights were increased on days 2 and 3 for ANIT-treated germfree (2.2 to 3.1-fold) rats compared with their respective vehicle-treated controls. These increases were noted despite reductions in food consumption and suggest that ANIT treatment may have resulted in delayed gastric emptying.
Clinical Pathology
The germfree animals treated with ANIT were significantly different from the vehicle-treated controls for the following biliary tree and hepatocellular-related analytes: ALT, GLDH, AST, ALP, BILE, and TBIL (days 2–4); PON1 (days 2 and 4); and LDH (day 3; Figures 3 and 4).
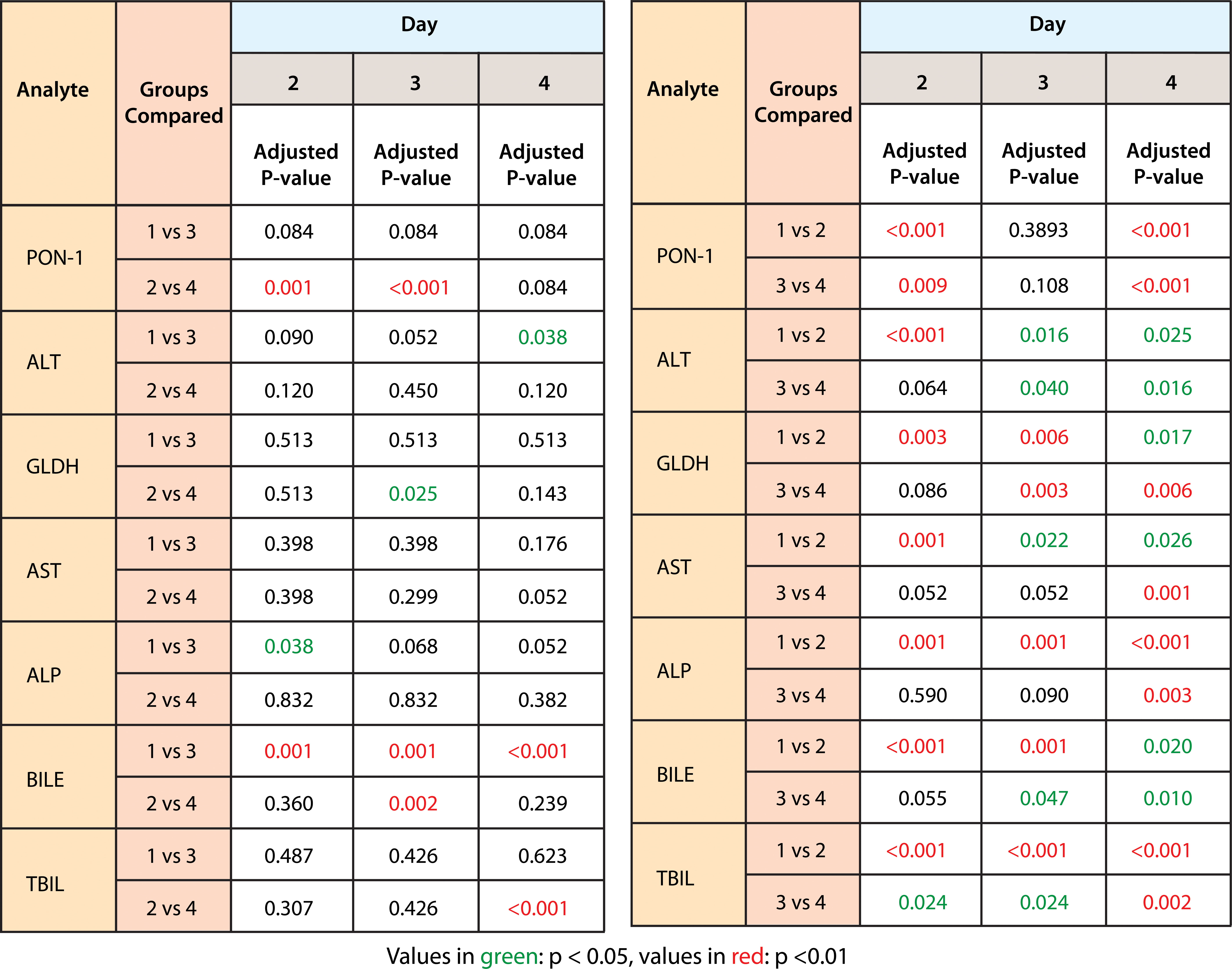
Comparison of clinical pathology analytes between germfree and conventional alpha-naphthylisothiocyanate-treated male rats and their respective controls.
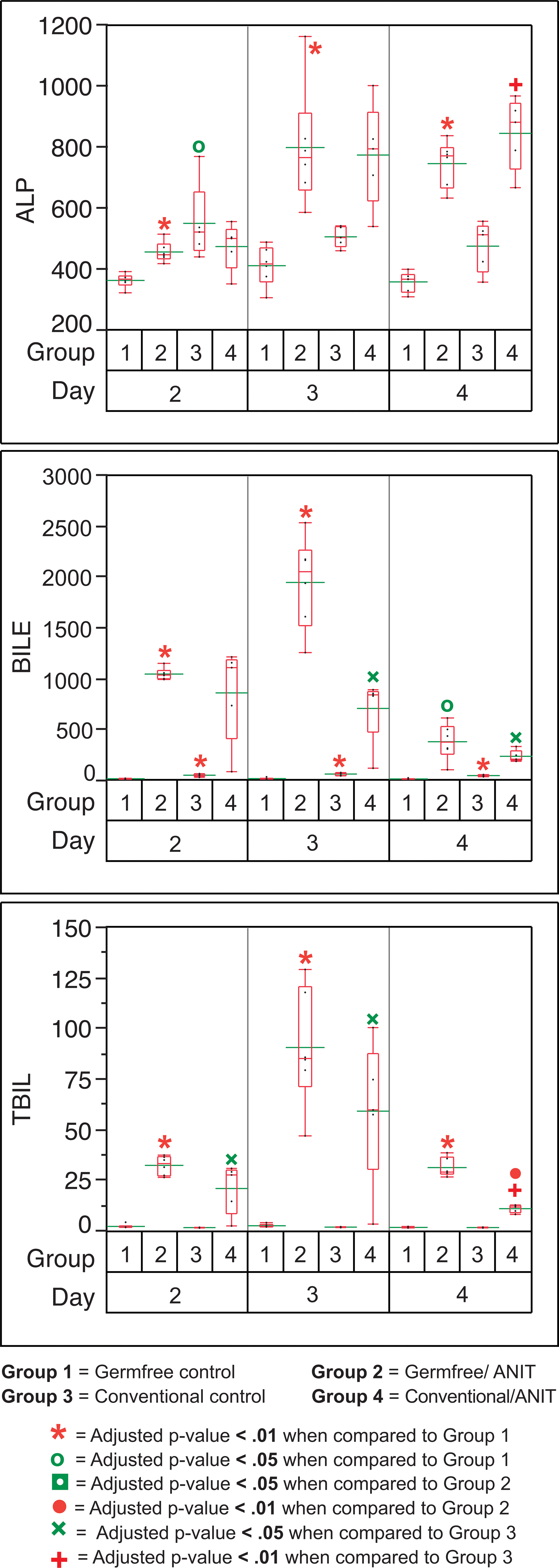
Panel A. Variability plots for biliary-related serum chemistry analytes from conventional and germfree rats treated with vehicle or alpha-naphthylisothiocyanate (ANIT). Panel B. Variability plots for selected hepatocellular-related analytes from conventional or germfree rats treated with vehicle or ANIT.
Analytes from conventional male rats treated with ANIT were significantly different from the vehicle-treated controls for LDH (day 3); PON1 (days 2 and 4); ALT, GLDH, and BILE (days 3 and 4); AST and ALP (day 4); and TBIL (days 2–4).
Germfree rats treated with ANIT tended to have greater increases in virtually all liver and biliary-related analytes compared with conventional rats treated with ANIT; however, significant differences were found only in a few of these analytes including increased BILE on day 3, T BILI on day 4, PON1 on days 2 and 3, and GLDH on day 3 (Figure 3).
Interestingly, there were some differences in the clinical chemistry results between the control germfree rats and the control conventional rats. The control conventional rats had increased ALP on day 2, ALT on day 4, and BILE on days 2 through 4 compared with the germfree control rats.
Histopathology
Administration of a single dose of 50 mg/kg ANIT orally to male conventional and germfree rats produced acute biliary duct necrosis and associated inflammation affecting interlobular and larger bile ducts that was first evident at 24 hr following administration. These lesions were persistent, being observed in ANIT-treated rats terminated approximately 72 hr after dosing, the final time point in this study (Figure 5). Reparative processes began 24 to 48 hr after the acute insult, as would be expected.
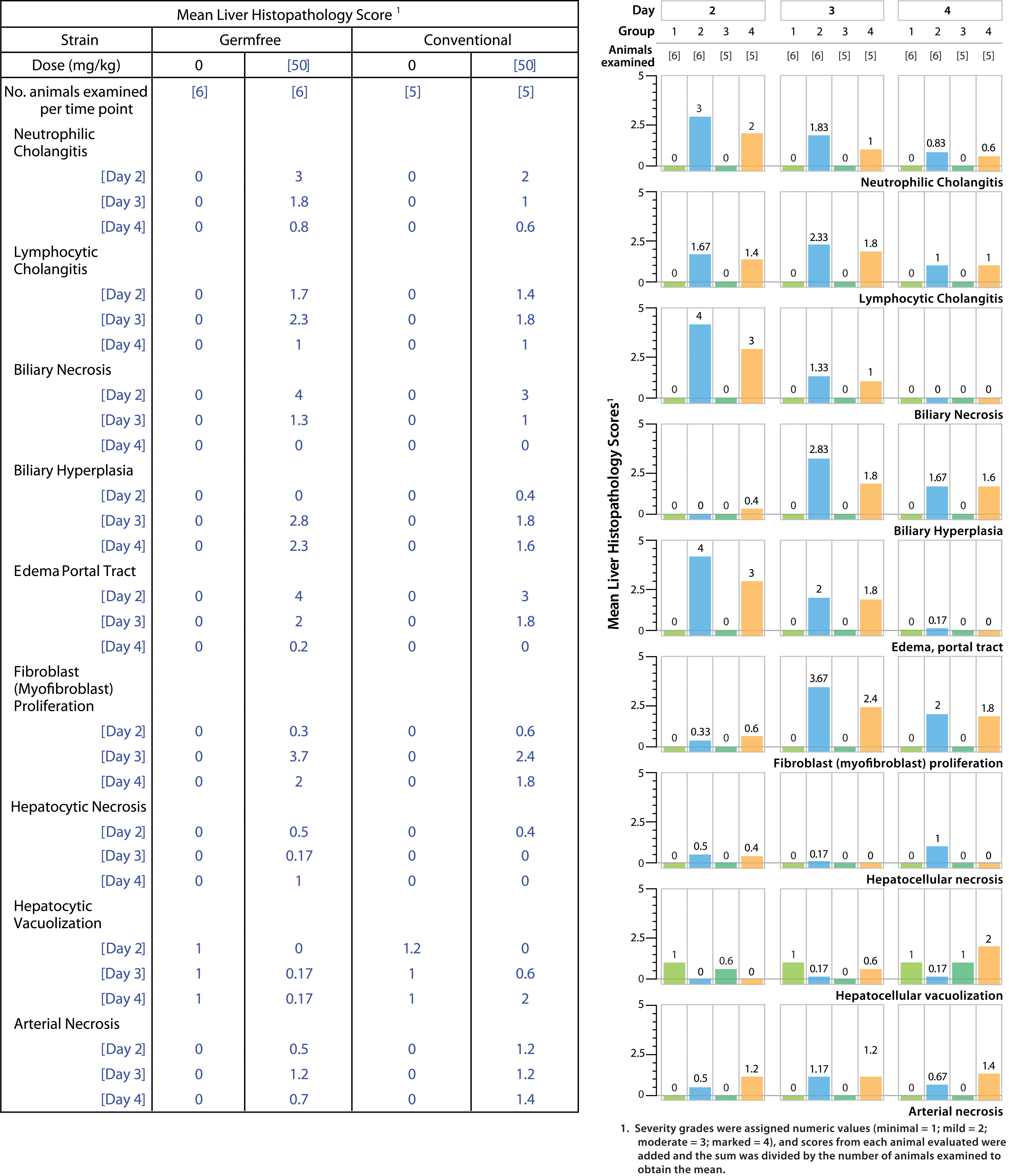
Subjective histologic scores for biliary and hepatocellular injury in alpha-naphthylisothiocyanate-treated conventional and germfree male rats.
Histologic differences between the conventional and germfree rats were modest, but most pronounced on day 2 (24 hr post dosing). Coincident with the acute biliary duct necrosis was an infiltration of neutrophils into the connective tissue of the portal tract immediately adjacent to the bile ducts, which was diagnosed as neutrophilic cholangitis. Based on subjective scoring, biliary necrosis, neutrophilic cholangitis, and portal tract edema were more severe (by one unit in each case) in germfree rats at 24 hr post dosing compared with conventional rats (Figures 5 and 6 [Panels A–C]). However, necrosis of the hepatic artery walls at this same time point was more severe in conventional rats (subjective score 1.2) than in germfree rats (subjective score 0.5). This lesion was interpreted as secondary to bile duct injury and local vessel damage rather than a primary injury of the vasculature.
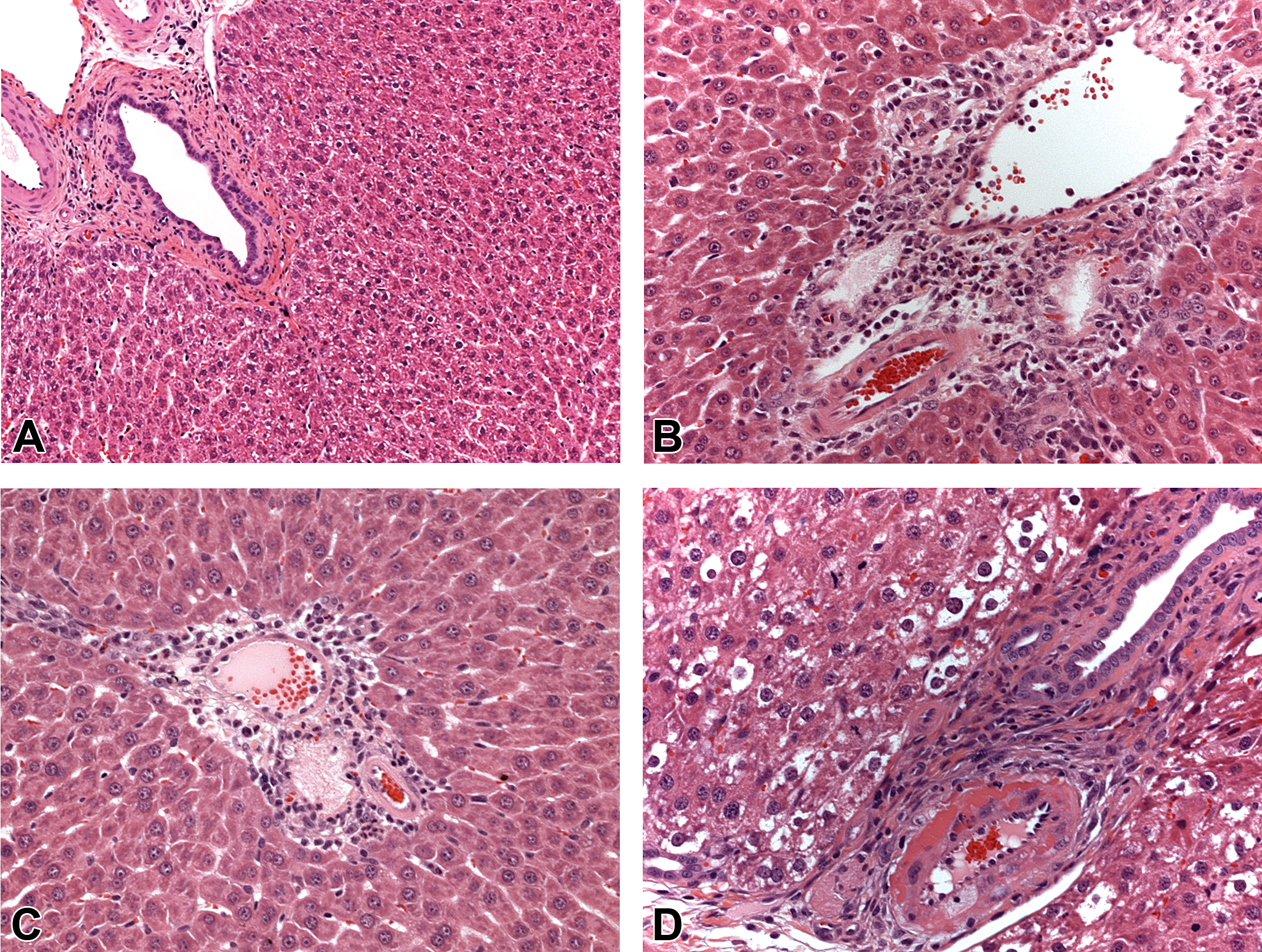
Panel A: Hematoxylin and eosin (H&E)-stained section demonstrating the appearance of a conventional germfree vehicle control rat (group 1, day 2). Panel B: Biliary epithelial necrosis, neutrophilic infiltrate, and edema in an alpha-naphthylisothiocyanate (ANIT)-treated germfree rat (group 2, day 2). Panel C: Biliary epithelial necrosis, neutrophilic infiltrate, and minimal edema in an ANIT-treated conventional rat (group 4, day 2). Panel D: Fibrinoid necrosis of the portal tract branch of the hepatic artery in an ANIT-treated conventional rat (group 4, day 4).
By day 3 (48 hr post dosing), histologic differences between germfree and conventional rats related to inflammation were less apparent. A modest increase in neutrophilic cholangitis and lymphocytic infiltrates in germfree rats was evident compared with conventional rats, but edema and biliary necrosis were similar. Differences in repair- or regeneration-related histologic changes were more evident in the germfree rats compared with the conventional rats at this time point. A circumferential ring of spindle-shaped cells (interpreted as myofibroblasts) developed around bile ducts by day 3 in both ANIT-treated groups, but was more intense in the germfree group (scores of 3.7 vs. 2.4). Hyperplasia of bile ducts was also more prominent in germfree rats compared with conventional rats on day 3 (scores of 2.8 vs. 1.8), suggesting a greater response to a more significant initial injury.
By day 4, differences between the two ANIT-treated groups were limited to the extent of biliary hyperplasia, which remained greater in the germfree group (scores 2.3 vs. 1.6), and an increase in hepatocellular necrosis (scores 1 vs. 0). Again, vascular necrosis of the hepatic artery was less evident in germfree rats than in conventional rats (scores 0.7 vs. 1.4) (Figure 6, Panel D).
Cell Proliferation
There was a prominent time-dependent increase in the biliary epithelial Ki-67 labeling index (LI) and hepatocellular Ki-67 LI in rats treated with ANIT (Figure 7) at days 3 and 4. The biliary epithelial proliferation LI was not significantly different at any of the time points when ANIT-treated germfree rats and ANIT-treated conventional rats were compared. The hepatocyte proliferation LI was greater in ANIT-treated conventional rats than ANIT-treated germfree rats, although the difference was statistically significant only at 4 days, when conventional and germfree rats had an increased group mean hepatocellular Ki-67 LI group mean of 94.9% and an LI group mean 35.5%, respectively. The immunohistochemical analyses were more sensitive for detecting hepatocellular proliferation than subjective histologic assessment, as hepatocellular mitotic increase was noted only on day 4 by light microscopy and was similar in incidence and severity for both strains. Since Ki-67 can be detected in all stages of the cell cycle, it is expected that the labeling index would exceed the mitotic figures evident by histology. Increased liver weights over time correlated with the increased cell proliferation (hepatocyte and biliary epithelium) in both germfree and conventional rats.
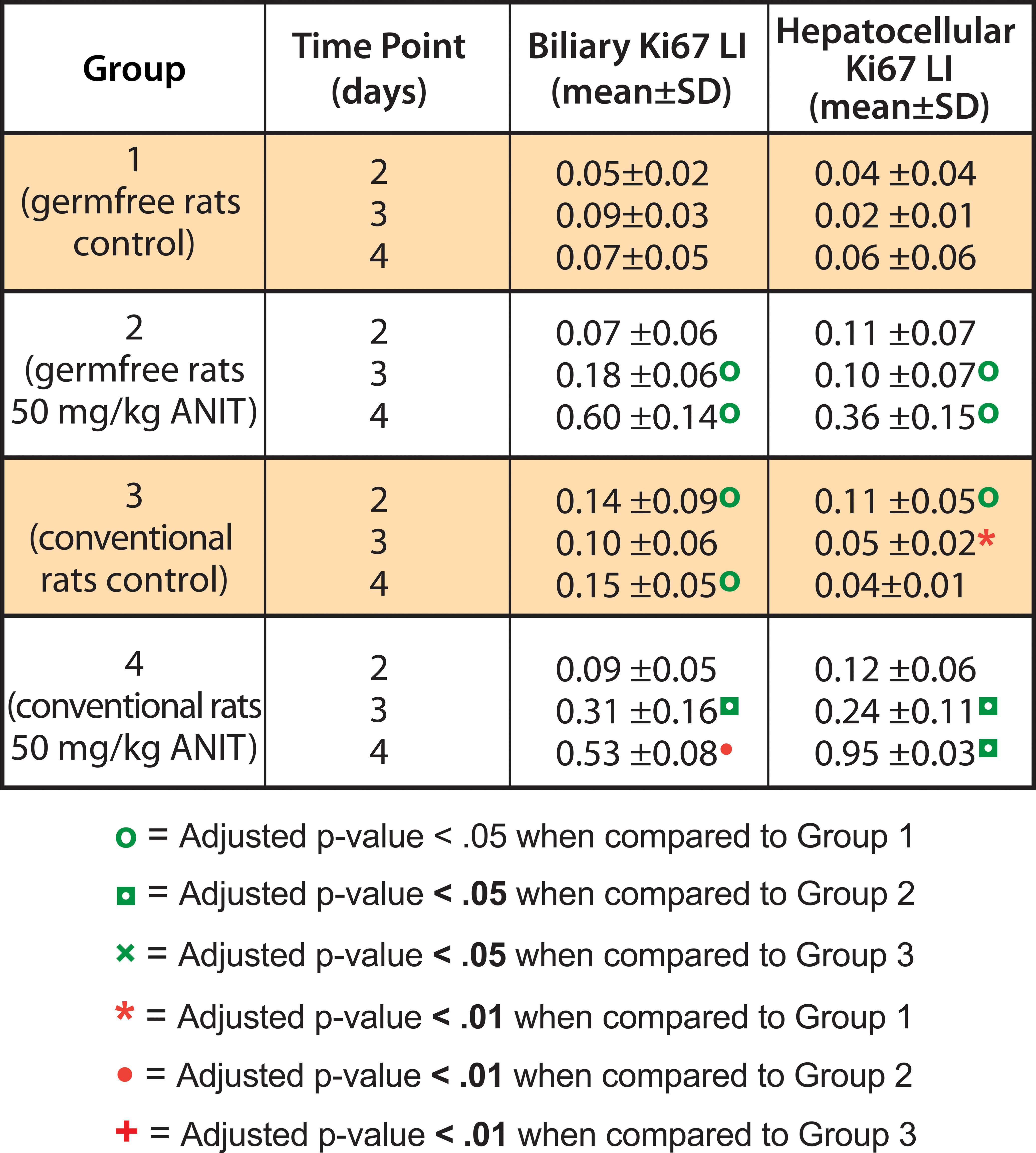
Biliary and hepatocellular Ki67 proliferation labeling indices (mean ± SD).
Discussion
The role of gut bacteria on metabolism, disease, and xenobiotic metabolism is now well recognized in a broad context (Nicholson et al. 2012). However, there have been few studies that compare the hepatic and biliary toxicity of specific compounds in germfree animals. Different effects in conventional and germfree animals would be anticipated since it is known that different populations of gut bacteria have the potential to alter the metabolism of common xenobiotics. For example, acetaminophen toxicity is likely to be increased by competition between acetaminophen and bacterially generated p-cresol for sulfation pathways (Clayton et al. 2009). Diminished o-sulphation by the liver has the potential to increase the risk of toxic injury from acetaminophen metabolites and reduce the hepatic glutathione pool. Other compounds, including ANIT, that use sulfation pathways to detoxify reactive intermediates could be similarly affected, although conversely, reduction of hepatic glutathione may inhibit ANIT toxicity (Dahm and Roth 1991). Intestinal bacteria are the source of endotoxin, which can trigger hepatic or systemic injury and inflammation. Endotoxin-driven inflammation has been shown to increase some types of drug-induced hepatocellular injury as well (Zou et al. 2009; Deng et al. 2009; Waring et al. 2006; Maddox et al. 2006). However, the absence of bacteria and associated lipopolysaccharide did not protect against ANIT-induced injury in our study. Presumably, endotoxin triggers innate immunity pathways enhancing inflammation. In two previous studies, our group has noted that the elimination of IL-10 in knockout mice and the near elimination of Kupffer cells only modestly increased biliary tract injury following acute ANIT exposure, suggesting that the biliary tree responds differently to injury than hepatocytes (Faiola et al. 2010; Cullen et al. 2013).
In this study, the histologic evidence of acute toxicity of ANIT was modestly more severe in germfree rats than that seen in conventional rats on day 2, with increased biliary necrosis, neutrophilic infiltration, and portal tract edema evident in the germfree rats, but the differences were not broadly apparent at later time points. Repair of damaged biliary ducts was accentuated in the germfree rats on day 3, evidenced by greater hyperplasia of the bile duct and proliferation of the surrounding myofibroblasts, which is associated with epithelial–mesenchymal communication during repair of biliary epithelial injury (Fabris and Strazzabosco 2011). The greater degree of repair supports the view that there was greater injury previously.
Clinical pathology evaluations supported the interpretation that germfree rats sustained greater injury to their biliary tracts than did conventional rats. The absolute levels for BILE and TBILI were increased in the ANIT-treated germfree rats compared to the ANIT-treated conventional rats at all time points, except day 2, although significant increases were limited to BILE on day 3 and TBILI on day 4.
We also observed that serum BILE levels in control conventional rats were significantly higher than in control germfree rats for all days of the study. This is in accord with earlier work showing a larger bile acid pool in conventional rats than germfree rats (Wostmann 1973). Similarly, germfree mice were noted to have lower serum bile acid levels than conventional mice, although the overall bile acid pool was greater in germfree mice (Sayin et al. 2013). Primary bile acids secreted into the intestinal lumen are metabolized into secondary bile acids by intestinal bacterial enzymes that perform deconjugation, dehydrogenation, dehydroxylation, and sulfation reactions (Midvedt and Norman 2007). Bile acid absorption is increased for hydrophobic forms and gut bacteria are known to deconjugate bile acids rendering them more hydrophobic and absorbable. Uptake from the ileum and transportation via the portal blood to the liver is increased leading to more bile acids resecreted into the bile. In germfree rats, conjugation of bile acids is limited to taurine primarily and unconjugated forms of bile acids are quite low compared to conventional rats. Hepatic extraction of unconjugated bile acids is less efficient than conjugated forms and this may explain why germfree rats with relatively lower levels of unconjugated bile acids have lower levels of bile acids in their plasma. Long identified for their role in fat digestion and hepatotoxicity, bile acids have more recently been recognized for their broad capacity for signaling in an array of organs including the liver via the farsenoid X receptor (FXR) and the plasma membrane-bound bile acid receptor TGR5 (Swann et al. 2011). The variation in bile acid forms in germfree and conventional rats leads to altered patterns of signaling evidenced by altered expression of genes involved in steroid, cholesterol, and bile acid synthesis as well as different FXR transcripts from liver, kidney, and heart (Swann et al. 2011).
Proliferation studies did not distinguish a difference in labeling index of the biliary epithelium of ANIT-treated germfree or conventional rats at any time point, although the subjective scoring of biliary hyperplasia was modestly elevated on days 3 and 4 in germfree rats compared to ANIT-treated conventional rats. The replication data indicated a robust upturn in the labeling index of hepatocytes on day 4 for both germfree and conventional rats, although the greatest increase in mitoses was seen in the conventional rats. Hepatocytes from conventional ANIT-treated rats did score higher with hepatocellular vacuolization, and there was more arteriolar necrosis in this group, but the histologic scoring of hepatocellular necrosis was higher in the germfree rats. Possibly the relatively small foci of necrosis were insufficient to drive more widespread hepatocellular proliferation. It is not clear why there was a discrepancy between the subjective scoring and the labeling index for biliary and hepatocellular features. Possibly the subjective review was not as accurate as a computer-driven scan of the entire slide. Alternatively, there may have been phenotypical changes in the biliary epithelium such as increased basophilic or pleomorphism scored as hyperplasia that occurred without a biochemical increase in Ki-67. Also, increased Ki67 labeling of hepatocytes without subsequent mitoses has been observed in rats by others (J. Foley, NIEHS, personal communication, September 12, 2015). As well, this may be due to temporal differences in the peak of hepatocyte replication between the different groups or differences in the ability of injured hepatocytes to replicate, but there is no obvious resolution to this conflict in the data.
Overall, the data support an interpretation that germfree rats have a modestly greater level of biliary tract injury following an acute exposure to ANIT, a well-characterized biliary toxin. However, the difference between the ANIT-treated germfree and conventional groups is most evident only within the first day following exposure, and after this period, the histologic and clinical pathologic differences diminish. Explanations for the acute differences in injury may be related to the gastrointestinal microbiome-related effects such as bacterial metabolism of ANIT or the presence of endotoxin or other bacterial products in the portal vasculature found only in conventional rats. It is also possible that there are differences in the rate of absorption on ANIT from the digestive tract between conventional and germfree rats that could influence blood levels during acute exposure. Greater hepatocyte necrosis, proliferation, and hepatic arteriolar necrosis may have been stimulated in the conventional rats due to their higher levels of bile acids in general and the likely differences in secondary bile acids compared to germfree rats lacking bacteria to metabolize primary bile acids. Alternatively, the oral ANIT dose given (50 mg/kg) was too high and overwhelmed the subtle differences in drug metabolism and host responses. A lower dose of ANIT could possibly produce a more discernible difference in the biliary tract response of germfree and conventional rats.
Footnotes
Author’s Note
The current affiliations for two of the authors have changed. Brenda Faiola is currently affiliated with Becton, Dickinson and Company, Corporate Preclinical Development & Toxicology, Research Triangle Park, North Carolina, USA. David H. Melich is currently affiliated with Roivant Sciences Inc., Durham, North Carolina, USA.
Author Contributions
Authors contributed to conception or design (JC, BF, HJ, RP, RM); data acquisition, analysis, or interpretation (JC, BF, DM, HJ, RP, RM); drafting the manuscript (JC, BF); and critically revising the manuscript (BF, DM, HJ, RP, RM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
