Abstract
Sampling blood for toxicokinetic (TK) evaluation in rodents is typically performed using a satellite group of animals to avoid depleting the blood volume and inducing an additional stressor in the main study animals. This practice does not allow for direct comparison of individual animal toxicity to exposure. These studies evaluated serial collection of twelve, 40-µl blood samples from each rat from either a tail clip or a saphenous vein bleed and its impact on toxicologic parameters over 4- and 14-day periods. The results show the feasibility of successfully collecting TK samples from main study animals, using either of the two techniques. Both procedures were amenable to execution by a single technician using dried blood spot sampling. Any changes observed in the primary markers of erythroid mass between the nonbled control rats and repeat sampled rats were minimal and the range of values often overlapped. This technique would improve the quality of data generated from toxicology studies by allowing a direct comparison of systemic exposure to toxicity while at the same time reducing the number of rats by obviating the need for satellite groups.
Keywords
Understanding and correlating the exposure profile (toxicokinetics [TKs]) of a drug or a new molecular entity (NME) with the associated toxicology in rodent and nonrodent preclinical species is a regulatory requirement during drug discovery and development. This information is essential for characterizing safety and understanding risks before the drug/NME is escalated to clinical testing (Lesko et al. 2000). The circulating drug/NME concentrations are measured in blood samples collected in sufficient quantities and frequency to define its exposure profiles (time vs. concentration relationship) and its TK properties (Peck et al. 1994). Collecting a full-time course (6–8 samples) of blood samples within a 24-hr period from individual rodents can be challenging due to limitations on the total volume that can be collected without impacting the animal’s health (Diehl et al. 2001). The use of satellite animals (also referred to as TK animals) for TK sampling has been the standard practice when conducting toxicology studies (Sparrow et al. 2011). The satellite TK animals can account for up to two-thirds of the total number of rodents used in toxicology studies.
Significant advancements in the sensitivity of liquid chromatography–tandem mass spectrometry instrumentation over recent years have enabled the use of small volumes of blood for analysis (Xu et al. 2007). Meanwhile, introduction of and advancements in microsampling techniques such as dried blood spot (DBS), whole blood microsampling (stored as a liquid), and microsampling of plasma have been successfully used to support drug discovery and development. For example, a single 10- to 20-µl blood sample collected using such techniques is sufficient to quantify drug/NME concentrations at a given time point postdose. The choice of bleeding technique used on a study is influenced by multiple factors including ease of collection, number of time points, volume of blood needed, and animal species.
There has been concern around performing TK blood sampling from the main study animals (also referred to as toxicology animals) mainly due to the potential impact on erythroid mass and the potential that the additional stress it may cause could confound interpretation of safety end points. These outcomes may be reflected in decreased total body weights or body weight gain, altered food consumption and activity, altered organ weights, lymphocyte depletion in thymus and spleen, altered circulating leukocyte counts, and altered reproductive functions (Everds et al. 2013). Potential challenges of distinguishing drug- versus phlebotomy-induced changes can be at least partially mitigated by using appropriate control groups that are subjected to the same procedures as those dosed with the drug or NME.
Although multiple bleeding techniques are feasible in rodents (Hem, Smith, and Solberg 1998), the use of tail bleeds (either via direct puncture of the tail vein or via clipping the distal end of the tail) seems to be the most commonly practiced technique that will enable the collection of a complete TK profile (Liu et al. 1996; Hui et al. 2007). The ability to collect 32-µl blood draws at each time point via puncturing the tail vein has been shown to have no impact on the clinical pathology parameters, compared to nonbled control rats, in a 14-day study (Powles-Glover et al. 2014). A comparison of tail clip bleeding and saphenous venipuncture has been conducted in mice, where the results showed that neither of the techniques caused stress-associated changes in leukogram; however, it was a 1-week study and consisted of only 4 serial blood collections per animal (Abatan, Welch, and Nemzek 2008). Collecting blood from the tail (as is commonly practiced for discovery TK studies) may not always be feasible, causing investigators to resort to the use of alternate bleeding techniques. For example, phlebotomy from the tail should be avoided when the drug/NME is administered intravenously via the tail vein.
The studies described here evaluated serial collection of blood samples for TK evaluations using the tail clip bleeding technique and saphenous vein phlebotomy from individual rats and its impact on toxicologic parameters over a 4-day period (6 samples of blood collected on day 1 and 6 on day 4) and a 14-day period (6 samples of blood collected on day 1 and 6 on day 14). Blood was collected as DBS samples. The study durations of 4 and 14 days were selected because these are commonly used study durations for toxicology/TK studies used during the preclinical testing of drugs/NMEs. The effect of serial phlebotomy (tail clip or saphenous vein) on parameters used in toxicity screening studies at our institution and the ease of use for each bleeding technique were evaluated.
Materials and Methods
Studies were conducted at Covance Laboratories (Greenfield, IN), which is accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International. All study designs were approved by the Eli Lilly and Company Study Design and Approval Committee and the Institutional Animal Care and Use Committee at Covance Greenfield. Female Sprague-Dawley rats were purchased from Charles River Laboratories (Portage, MI) and were group housed (2–3 per cage). The animal rooms were maintained on a 12:12-hr light–dark cycle at a room temperature that ranged from 68°F to 79°F with a relative humidity between 30% and 70%. Rats were fed Harlan Teklad Global Diets—Rodent 2014 rodent diet (Indianapolis, IN), and permitted free access to Greenfield, IN, tap water, and were allowed 1 week of acclimation prior to study start.
4-day Tail Clip Study
Rats (7–9 weeks of age) were divided into 2 groups, each with n = 10: group 1 control rats were only bled at necropsy for clinical pathology sampling; group 2 rats were bled by tail clip for TK sampling and were bled at necropsy for clinical pathology sampling. To simulate dosing in a toxicology study, all rats were given a daily oral gavage of sterile water (10 ml/kg). Each group 2 rat had 40-µl blood collected by tail clip at time 0 (day 4 only) and at 0.5, 1, 2, 4, 8, and 24 hr on days 1 and 4. Blood was collected into two 20-µl EDTA-coated capillary tubes at each timepoint and spotted on to DBS cards. Animals were weighed and food consumption was measured on days 1 and 4. Clinical observations were tabulated daily. On day 4, approximately 1 hr after collection of the terminal TK sample and immediately prior to necropsy, rats were anesthetized using isoflurane, and blood was collected from the periorbital plexus into tubes for complete blood count (K3EDTA, 1.3-ml Microtainer—Sarstedt, Newton, NC) and clinical chemistry panel (no anticoagulant, 0.6-ml Microtainer Serum Separator—Becton Dickinson, Franklin Lakes, NJ) and from the abdominal aorta for determination of coagulation panel (1.8-ml draw Na citrate [3.2%]—Becton Dickinson). After euthanasia, urine was collected by cystocentesis for measurement of total protein concentration and specific gravity. All clinical pathology data (hematology, coagulation, clinical chemistry, and urinalysis) had mean, standard deviation, and percentage change from the nonbled-only control calculated. Direct comparison of clinical pathology parameters was made between nonbled control rats and rats bled for repeated TK sampling. Alterations in clinical pathology data were interpreted by a board-certified veterinary clinical pathologist. The following tissues were collected and fixed in neutral buffered formalin, processed for light microscopic examination, stained with hematoxylin and eosin, and examined by a board-certified veterinary anatomic pathologist: adrenal glands, spleen, thymus, and bone (sternum). These tissues are often evaluated in short toxicity screening studies and may have organ weight and/or histologic alterations that occur secondary to stress in rodents (Everds et al. 2013).
14-day Tail Clip Study
A 14-day study of similar design was performed to learn if there were important differences in clinical pathology data or microscopic appearance of tissues compared to the 4-day study. Study design and protocol parameters were the same as the 4-day study described above with the following exceptions: animals were weighed and clinical observations were collected on days 1, 5, 8, 12, and 14; group 2 rats were bled by tail clip for TK sampling on study days 1 and 14; and organ weights were determined at necropsy for adrenal glands, spleen, and thymus. Each group 2 rat had 40-µl blood collected by tail clip at time 0 (day 14 only) and at 0.5, 1, 2, 4, 8, and 24 hr on days 1 and 14. Blood was collected into two 20-µl EDTA-coated capillary tubes at each timepoint and spotted on to DBS cards. Daily oral gavage, phlebotomy for clinical pathology sampling, and necropsy procedures were the same as the 4-day study.
14-day Tail Clip versus Saphenous Vein Study
This study had 3 groups of rats, each with n = 10. Group 1 control rats were bled only for clinical pathology sampling at necropsy. Group 2 rats were bled from the tail for TK sampling and at necropsy for clinical pathology sampling, and group 3 rats were bled from the saphenous vein for TK sampling and at necropsy for clinical pathology sampling. Overall study design and protocol parameters were the same as the 4- and 14-day studies described above. Approximately 40 µl of whole blood was collected from each animal in groups 2 and 3 at times 0.5, 1, 2, 4, 8, and 24 hr on days 1 and 14. Blood was collected into two 20-µl EDTA-coated capillary tubes at each timepoint and spotted on to DBS cards. Animals were weighed on days −1, 1, 4, 7, 10, and 14, and food consumption was recorded on days 1, 4, 7, 10, and 14. Collection of clinical observations, phlebotomy for clinical pathology sampling, necropsy procedures, collection of body and organ weights, and statistical approach were the same as the 14-day tail clip study.
Tail Clip Phlebotomy
A sterile scalpel was used to excise 2 to 3 mm from the tip of the tail (Hui et al. 2007). Blood (2 × 20 µl) for production of DBSs was collected with capillary tubes and spotted on filter paper. Repeated blood collections within a single day were accomplished by removing the scab from the tail with gauze and establishing blood flow.
Saphenous Vein Phlebotomy
Rats were restrained by hand so that the hind leg and tail were exposed. The skin of the thigh was grasped in a gentle but firm manner to extend one leg and occlude the saphenous vein. The fur over the saphenous vein was clipped, and a thin film of petroleum jelly was applied over the saphenous vein (Abatan, Welch, and Nemzek 2008). The use of petroleum jelly helped control the bleeding as well as reduce any skin irritation. The tip of a 23- to 25-gauge blood collection needle (Becton Dickinson) was used to puncture the saphenous vein at the point where it crosses the lateral surface of the leg. Blood (2 × 20 µl) for production of DBSs was collected with capillary tubes and spotted on DBS collection cards.
Hematologic Assessment
Well-mixed K3EDTA anticoagulated blood samples were analyzed using the ADVIA 120 Hematology System with Multispecies Software (version 3.1, Siemens Medical Solutions, Norwood, MA) and reagents from Siemens. Erythrocyte count, hemoglobin concentration, mean corpuscular volume, platelet count, total and differential leukocyte counts, and reticulocyte count were determined directly by the instrument. Hematocrit, mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration were calculated by the instrument. Freshly made blood smears were stained with Wright-Giemsa stain using the ADVIA S60 Auto Slide Stainer (Siemens Medical Solutions). The stained blood smears were used to assess blood cell morphology and confirm automated differential leukocyte counts.
Coagulation Assessment
Tubes of whole blood collected in sodium citrate anticoagulant were spun in a centrifuge to separate cells and plasma. An aliquot of citrated plasma was used to evaluate the activated partial thromboplastin time (APTT) and the prothrombin time (PT) using an STA Compact (American Bio Products, Parsippany, NJ).
Clinical Chemistry Assessment
Tubes of blood collected without anticoagulant were allowed to clot for 30 min at room temperature. Tubes were spun in a centrifuge, and serum was separated from the clotted blood. Serum was analyzed for a panel of 20 parameters using a Modular P Analyzer (Roche Diagnostics, Nutley, NJ) and Roche reagents. The activities of alanine aminotransferase, alkaline phosphatase, aspartate aminotransferase, creatine kinase, and gamma-glutamyl transferase, and the concentrations of albumin, blood urea nitrogen, calcium, creatinine, cholesterol, chloride, glucose, inorganic phosphorus, potassium, sodium, triglycerides, total bilirubin, and total protein were determined. The concentration of globulin was determined by subtracting the concentration of albumin from the concentration of total protein. The albumin/globulin ratio was calculated.
Limited Urinalysis
Aliquots of room temperature urine were placed in a handheld refractometer (Reichert Vet 360 TS Meter, Reichert Analytical Instruments—Reichert Inc., Depew, NY) for determination of urine-specific gravity. Commercially available urine test strips (Siemens Albustix, Fisher Scientific, Pittsburgh, PA) were used to determine the semiquantitative urine protein concentration.
Statistical Approach
For the 4- and 14-day tail clip studies and the 14-day tail clip versus saphenous vein study, descriptive statistics including the mean ± SD and percentage change from nonbled control rats were calculated for all clinical pathology parameters. A Student’s t-test was used to compare the clinical pathology parameters from bled repeat TK sampled rats (tail clip or saphenous vein phlebotomy) to nonbled control rats in the 4- and 14-day studies (Steel and Torrie 1980). The criterion for significance was p < .05. Tables of data also include reference intervals to assist in evaluation of the magnitude of change in hematology parameters in studies of tail clip and saphenous vein phlebotomy in repeated TK sampling studies.
Retrospective Evaluation
Additionally, a retrospective evaluation was completed using the control data from 46 historical 4-day rat studies at our institution in which tail clip bleeds were used for repeat TK sampling in toxicity screening studies. The erythrocyte count, hemoglobin concentration, hematocrit, total leukocyte count, neutrophil count, and lymphocyte count were evaluated from these studies to demonstrate the magnitude of change in cell counts in the rats, given repeat sampling for TK analysis. Comparisons were made between the nonbled control rats (n = 73) and the rats used for repeated TK sampling via tail vein bleeds (n = 61, each with six to seven 20-µl TK samplings) using a Student’s t-test (Steel and Torrie 1980; Figure 1). The criterion for significance was p < .05.
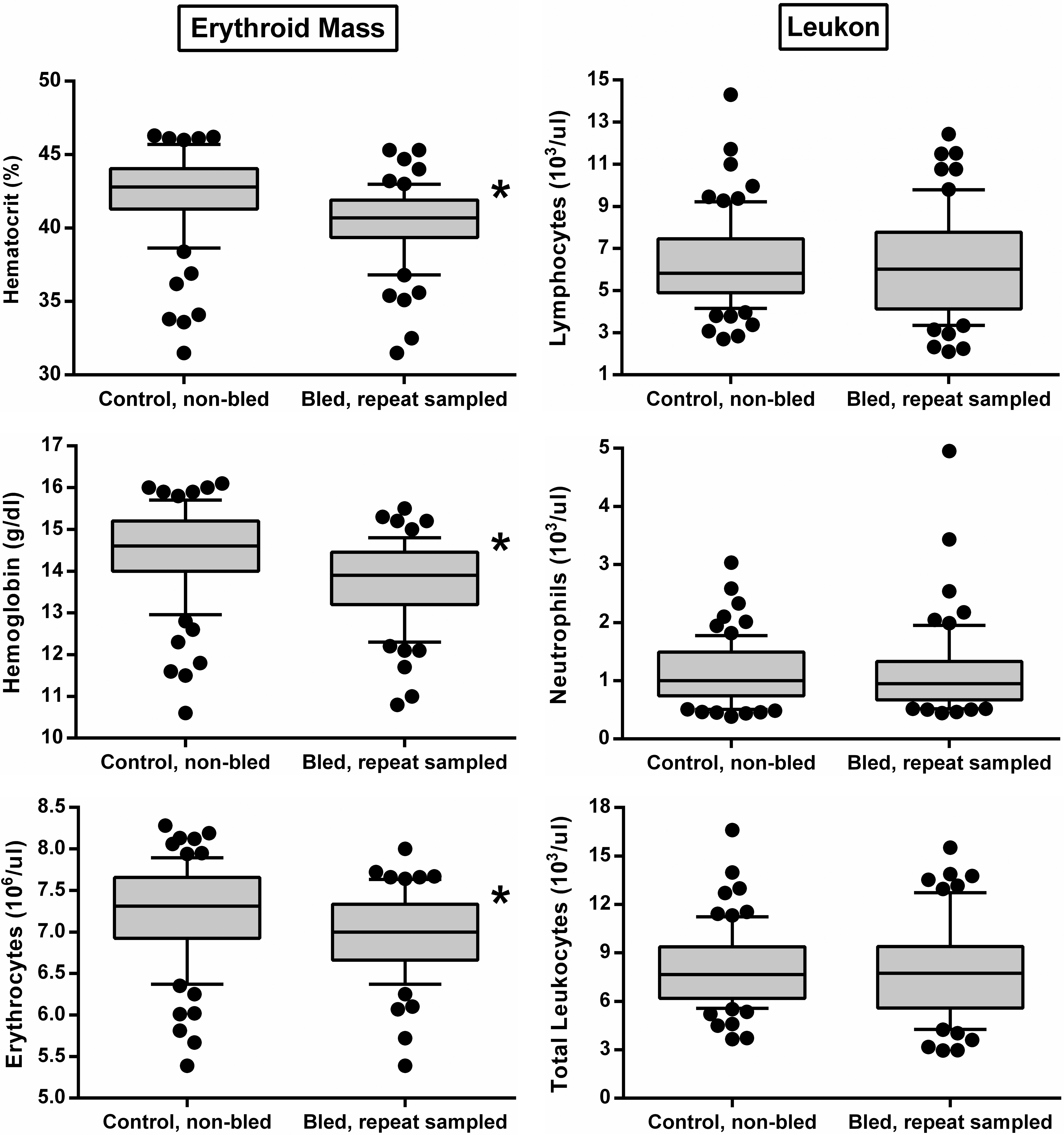
Box plots of markers of erythroid mass and the leukon from male and female rats used in forty six 4-day toxicology/toxicokinetic (TK) studies. The nonbled control rats had blood drawn from the orbital plexus for clinical pathology sampling at necropsy (N = 73 rats) and were compared to those values from rats bled for repeated TK sampling (6–7 samples of 20 µl each) and for clinical pathology sampling at necropsy (N = 61). The central lines within each box represent the median value and the outer lines (whiskers) present values from the 10th and 90th percentiles of the data. *Significantly different value, p < .05.
Results
4- and 14-day Tail Clip Studies
Rats bled by the tail clip method for collection of TK samples on study days 1 and 4 and euthanized on study day 4 had no changes in food consumption or body weight and no observations of abnormal clinical signs compared to control animals that did not have serial TK phlebotomy. Treatment-related alterations in the complete blood count were few (Table 1). Rats bled by the tail clip for TK sampling had minimal decreases (∼↓6–7%) in erythrocyte count, hematocrit, and hemoglobin concentration and minimal increases in reticulocyte counts (↑14%) compared to the nonbled control animals. There were no differences in group mean total leukocyte, differential leukocyte, or platelet counts between rats bled by tail clip for TK sampling and control rats which did not have serial TK sampling. Blood smear examination indicated minimal platelet clumping in most rats and no quantitative or morphologic alterations in leukocytes. In addition, 6 of the 10 rats bled by tail clip had slight polychromasia of erythrocytes on blood smear examination compared to 1 of the 10 nonbled control rats. The serum samples for clinical chemistry analysis and the citrated plasma samples for coagulation studies were free from lipemia and hemolysis. There were no treatment-related changes in the coagulation panel (PT and APTT), clinical chemistry panel, or limited urinalysis and no treatment-related alterations in the microscopic appearance of tissues collected.
Treatment-related changes in 4-day tail clip study.

Note. Hematology data are presented as mean ± SD. Nonbled control rats had blood drawn at necropsy for measurement of clinical pathology parameters. Bled repeat sampled rats had multiple blood draws for collection of toxicokinetic samples (tail clip) and had blood drawn at necropsy for measurement of clinical pathology parameters. N = 10/group. ↓ = decreased, ↑ = increased, th = thousands.
aSignificantly different from control.
The rats bled by repeat sampling and euthanized on day 14 had no treatment-related alterations in body weight or food consumption and no observations of abnormal clinical signs. Alterations in the complete blood count of rats bled by tail clip consisted of minimal decreases in the erythrocyte count, hemoglobin concentration, and hematocrit (↓9–11%) and slight increases in reticulocyte count (↑25%; Table 2). There were no differences in group mean total leukocyte, differential leukocyte, or platelet counts between rats bled by tail clip for TK sampling and control rats which did not have serial TK sampling. Blood smear examination indicated no differences in leukocyte or platelet morphology. Review of individual animal data in this study indicated 3 of the 10 rats bled by tail clip had minimal increases in total leukocyte counts (14,830/µl, 11,550/µl, or 15,160/µl vs. control mean 8,780/µl) due to minimal increases in neutrophil (1,600/µl, 1,100/µl, or 3,200/µl vs. control mean 1,200/µl) and/or lymphocyte counts (12,400/µl, 9,900/µl, or 11,200/µl vs. control mean 7,060/µl). Lipemia and hemolysis were not detected in the serum samples for clinical chemistry analysis and the citrated plasma samples for coagulation studies. There were no treatment-related changes in the coagulation panel (PT and APTT), clinical chemistry panel, or limited urinalysis. There were no treatment-related changes in organ weights or changes in the microscopic appearance of tissues collected. One of the rats bled by tail clip had moderate decreases in markers of erythroid mass (↓26% erythrocyte count, ↓28% hemoglobin concentration, and ↓27% hematocrit compared to the overall means for these parameters from all rats bled by tail clip). This rat also had anisocytosis and polychromasia in the blood smear.
Treatment-related changes in 14-day tail clip study.

Note. N = 10/group. Hematology data are presented as mean ± SD. Nonbled control rats had blood drawn at necropsy for measurement of clinical pathology parameters. Bled repeat sampled rats had multiple blood draws for collection of toxicokinetic samples (tail clip) and had blood drawn at necropsy for measurement of clinical pathology parameters. ↓ = decreased, ↑ = increased, th = thousands.
aSignificantly different from control.
14-day Tail Clip versus Saphenous Vein Study
All animals survived until study completion. There were no changes in clinical condition of any of the rats. Rats that had blood drawn from the tail or saphenous vein had minimal decreases in body weight (↓1–4%) and food consumption (↓3–11%) compared to nonbled control rats. Treatment-related alterations in the complete blood count were restricted to minimal decreases (∼↓4-5%) in erythrocyte count, hematocrit, and hemoglobin concentration in rats that had blood drawn from the tail or saphenous vein (Table 3). There were no differences in group mean total leukocyte, differential leukocyte, or platelet counts between rats bled by tail clip or saphenous vein for TK sampling and control rats which did not have serial TK sampling. Blood smear examination indicated minimal platelet clumping in most rats and no quantitative or morphologic alterations in leukocytes. The serum samples for clinical chemistry analysis and the citrated plasma samples for coagulation studies were free from lipemia and hemolysis. There were no treatment-related changes in the coagulation panel (PT and APTT), clinical chemistry panel, or limited urinalysis. Rats that had blood drawn from the saphenous vein had minimal to moderate hemorrhage in the subcutis of the hind leg. There were no alterations in organ weights or microscopic appearance of the tissues collected in animals that were bled by either the saphenous vein or tail clip.
Treatment-related changes in 14-day tail clip versus saphenous vein study.

Note. N = 10/group. Hematology data are presented as mean ± SD. Nonbled control rats had blood drawn at necropsy for measurement of clinical pathology parameters. Tail clip rats had multiple blood draws collected via tail clip for collection of toxicokinetic samples and had blood drawn at necropsy for measurement of clinical pathology parameters. Saphenous rats had multiple blood draws collected via saphenous vein for collection of TK samples and had blood drawn at necropsy for measurement of clinical pathology parameters. ↓ = decreased, ↑ = increased, th = thousands. There were no significant differences from control.
Discussion
In all of our studies, the decreases in erythrocyte counts, hemoglobin concentration, and hematocrit on day 4 or 14 from rats that had samples collected for repeated TK sampling were minimal in magnitude and were attributed to the sampling procedure. The decreases in primary markers of erythroid mass (erythrocyte count, hemoglobin concentration, and hematocrit) were so small in rats that had blood drawn for repeated TK sampling (either tail clip or saphenous vein) that values for these analytes often did not exceed values for reference intervals for our institution (Tables 1–3).
In the 4- and 14-day tail clip studies, some of the rats showed signs of erythroid regeneration (minimally to slightly increased reticulocyte count and erythrocyte polychromasia and/or anisocytosis) on day 4 and/or 14. There are several explanations that may account for these observations. Evidence of erythroid regeneration was expected to be mild in the 4-day study due to its short duration. In the 4-day study, rats had blood drawn for TK analysis on days 1 and 4. Signs of erythroid regeneration following an initial blood loss may vary slightly in their onset depending upon the magnitude of blood volume lost (Capel-Edwards, Wheeldon, and Mifsud 1981) but often do not manifest until posthemorrhage days 3 to 7 (Criswell et al. 2000; Brockus and Andreasen 2003). In the 4-day study, the magnitude of blood loss was minimal (6–7% decrease in primary markers of erythroid mass; Table 1), and reticulocyte counts were increased minimally (14%) compared to nonbled control rats. Necropsy followed blood collection on day 4 almost immediately (<1 hr), so no additional regenerative response would likely be detected following the second blood loss in this study.
In the 14-day tail clip study, rats had blood drawn for TK analysis on days 1 and 14. Alterations in the complete blood count of rats consisted of minimal decreases in the primary markers of erythroid mass (↓9–11%) and slight increases in reticulocyte count (↑25%) in rats bled by tail clip compared to nonbled controls (Table 2). There was slightly greater variability in the markers of erythroid mass in the groups bled by tail clip compared to the nonbled control rats. One of the rats bled by tail clip had moderate decreases in markers of erythroid mass (↓26% erythrocyte count, ↓28% hemoglobin concentration, and ↓27% hematocrit compared to the overall means for these parameters from all rats bled by tail clip), suggesting additional hemorrhage occurred from the tail after sample collection for TK analysis. Review of the blood smear from this rat indicated anisocytosis and polychromasia. The remaining 9 of the 10 rats in the group bled by tail clip in this study lacked the subjective observations of anisocytosis and polychromasia, but some had minimal increases in the reticulocyte count. Given the longer duration of this study (4 vs. 14 days), rats bled for TK analysis had more time for erythroid regeneration to occur. The greater decrease in markers of erythroid mass by the group given the tail clip in this study versus the 4-day study and the moderate decrease in markers of erythroid mass in one of the rats given the tail clip suggested that blood loss in this group was greater than that which occurred in the 4-day study. It is not unusual for some degree of erythroid regeneration to occur as late as 7 to 14 days following blood loss in rats (Criswell et al. 2000) as was shown by the polychromasia, anisocytosis, and increase in reticulocyte counts in some of the rats given the tail clip in this study. In the 14-day tail clip study, 3 of the 10 rats bled by tail clip had minimal increases in total leukocyte counts due to minimal increases in neutrophil and/or lymphocyte counts. These alterations in the leukogram were suggestive of inflammation (Latimer and Prasse 2003). Although there were no abnormal clinical signs reported by technicians at the animal facility, we hypothesized that the inflammation may have occurred at the tail clip wound which was not examined histologically in this study.
In the 14-day tail clip versus saphenous vein study, the decreases in markers of erythroid mass were of lesser magnitude and appeared similar to those that occurred in the 4-day tail clip study. Alterations in the complete blood count were restricted to minimal decreases (∼3–6%) in markers of erythroid mass in rats that had blood drawn from the tail or saphenous vein compared to nonbled controls. Changes in primary markers of erythroid mass of this magnitude are often associated with phlebotomy for repeated TK blood sampling at our institution (Table 3). There were no treatment-related alterations in total leukocyte count, neutrophil count, or lymphocyte counts in this study. The reasons for the minimal improvement in the decreases in markers of erythroid mass compared to the 14-day tail clip study and the lack of changes in the leukograms in any of the rats given the tail clip in this study were not entirely clear. We hypothesized that the technical staff improved proficiency in executing the tail clip technique and caused less blood loss and inflammation in the tail of rats during the saphenous versus tail clip study.
A retrospective analysis was conducted using hematology data from the control group in forty six 4-day rat toxicity screening studies conducted recently, using the same standardized protocol and procedure, where the animals were bled via the tail clip technique. The purpose of this analysis was to evaluate if the observations and outcomes reported in this article were consistent, when routine studies were conducted using the same technique and protocol supporting toxicology/TK studies within our organization. When compared to primary markers of erythroid mass from rats bled for clinical pathology sampling only (nonbled control rats), the erythrocyte count, hematocrit, and hemoglobin concentration in rats bled for repeated TK sampling along with clinical pathology sampling (bled, repeat sampled rats) were decreased, but the magnitude of the decrease was minimal and the range of values overlapped (Figure 1). Even though they were decreased, the primary markers of erythroid mass remained within reference intervals for our institution. Despite the small differences noted in these studies, the minimal magnitude of change in markers of erythroid mass coupled with use of concurrent vehicle controls suggested that these minimal changes would not be problematic for data interpretation in routine screening studies.
Our studies demonstrate the ability to successfully collect a complete TK profile via multiple saphenous vein bleeds or tail clip bleeds from each individual main study animal using a microsampling technique such as DBS sampling without affecting the interpretation of safety end points in the study. Both bleeding techniques caused minimal decreases in erythrocyte counts, hemoglobin concentration, and hematocrit. In our subsequent experience, tail clip bleeds can cause additional complications such as blood loss due to the cage mate rats nibbling on the scab at the site of the tail wound. In some studies, technicians have occasionally noted blood streaks inside the cage when handling animals for the next blood draw indicating continuing blood loss from the tail wound. Saphenous vein bleeds can be beneficial especially in situations where sampling from the tail may not be desirable, for example, when the drug/NME is administered intravenously via the tail vein. Additionally, the adoption of a simple microsampling technique (DBS sampling) enabled the efficient execution of both bleeding techniques described here (tail clip and saphenous bleeds), whereas the methodology described by Powles-Glover et al. (2014) required the use of two technicians.
The advancement of analytical instrumentation has enabled the use of small volumes of blood (10–20 μl) for analysis and the successful adoption of microsampling techniques such as DBS, especially in situations where sampling blood volumes are limited (i.e., rodent species). However, the adoption of DBS sampling for toxicology/TK studies has been slower than anticipated across the pharmaceutical industry. Switching from traditional plasma sampling to DBS sampling requires understanding the blood to plasma partitioning ratio, since DBS data may need to be converted to plasma equivalents and vice versa. Additionally, incorporation of DBS analysis to a drug development paradigm also requires a successful bioanalytical feasibility assessment followed by the execution of appropriate bridging studies to understand and demonstrate the correlation between the blood and plasma matrices. Other microsampling techniques such as plasma microsampling and novel blood/plasma collection devices are also being developed as alternative approaches, which could be used easily with the repeated blood sampling techniques described here, and are expected to expand the adoption of microsampling in the future.
When adopting the bleeding techniques described here for toxicology/TK studies, control group animals are also subjected to the same blood sampling schedule as the treatment groups in order to minimize hematologic effects and facilitate data interpretation between controls and treatment groups. Use of these techniques in toxicity studies is not advised when administering compounds with known effects that might prolong hemostasis and result in excessive hemorrhage. We also advise using caution in data interpretation when using these techniques for compounds which have marked known effects on erythropoiesis in longer toxicity studies.
Widespread implementation of the techniques used for repeated TK sampling described in our studies would translate to a significant reduction in the number of rodents used in typical nonclinical studies (Wickremsinhe 2015; Sparrow et al. 2011; Dainty et al. 2012) and aligns with the 3Rs—reduction, refinement, and replacement (Guhad 2005; Russell and Burch 1959), while improving the power of the safety evaluation by characterizing both compound exposure and toxicity end points in the same animals. Although this contribution was specific to TK studies conducted during the drug development process, these techniques could be widely adopted for testing xenobiotics or other compounds outside of the drug development paradigm.
Footnotes
Author Contributions
Authors contributed to conception or design (EW, MR, AP, MP, AS); data acquisition, analysis, or interpretation (EW, MR, AP, MP, AS); drafting the manuscript (EW, MP, AS); and critically revising the manuscript (EW, MR, AP, MP, AS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
