Abstract
In vitro skin model systems are increasingly being used both in the early evaluation of therapeutic drug candidates and in confirmatory mechanistic studies. The most commonly used of these in vitro model systems are reconstituted human epidermis (RHE) models. These RHE models consist solely of epidermal keratinocytes, which comes with some limitations but also with the advantage of focusing toxicologic and pharmacologic evaluation on keratinocytes alone. RHE models can generally be implemented more quickly, easily, and reproducibly than in vivo models and can thus be used for high throughput compound screening while potentially reducing the need for animal studies. Histologic evaluation of RHE sections can be done quite easily, and the sections are very amenable to quantification via image analysis, including automated analysis. RHE model systems can provide very valuable early indications of therapeutic candidate biology, pharmacology, and toxicity; and early results have demonstrated that RHE models have been quite predictive for in vivo pharmacologic and toxicologic effects on the skin, including clinical skin toxicity.
Introduction
The 3-D in vitro human skin model systems have been increasingly used in recent years for both toxicologic applications and basic biologic/pathophysiologic research. These models have also recently gained increased acceptance for the toxicologic evaluation of topical irritation and corrosion and have also gained some initial acceptance for the evaluation of cutaneous permeability/transport.
The most commonly utilized 3-D in vitro human skin models are EpiDerm®, EpiSkin®, and SkinEthic®. All 3 are reconstituted human epidermal (RHE) models that are composed solely of normal human epidermal keratinocytes that form a 3-D reconstitution of normal human epidermis containing all viable (stratum basale, stratum spinosum, and stratum granulosum) and nonviable (stratum corneum) cell layers. All models are metabolically and mitotically active, have a lipid and ceramide profile very similar to normal human epidermis, and express normal epidermal differentiation markers such as keratin 1/10, profilaggrin, and involucrin. They are all grown in serum-free media system and are highly reproducible lot to lot.
In vitro RHE models have many advantages when compared to in vivo models, as they can be done more quickly, easily, and reproducibly. Therefore, these models can be used for high throughput compound screening while offering the additional advantage of reducing the need for animal studies. In addition, histologic evaluation of RHE sections, including quantification via image analysis, is readily done. Using automated image analysis and quantification allows for even more rapid and high throughput screening.
RHE models have been extensively evaluated for their ability to correctly predict topical skin irritation/corrosion and have all demonstrated very good accuracy and reproducibility in this regard, with a predictive ability of ≥90% (Faller et al. 2003; Hayden et al. 2003; Kandárová et al. 2005; Kandárová et al. 2006; Netzlaff et al. 2005). The ability of these models to predict skin permeability/transport and phototoxicity has been less extensively evaluated, but these models have also gained some acceptance for those uses as well (Netzlaff et al. 2005; Schäfer-Korting et al. 2008).
This review is focused on RHE models, which as mentioned earlier contain only epidermal keratinocytes. These models do have some limitations due to being composed of only 1 cell type, the epidermal keratinocyte, such as not having as well developed a barrier function as human skin (Netzlaff et al. 2005; Schäfer-Korting et al. 2008). In addition, RHE models lack dermal components such as collagen, fibroblasts, and blood vessels. They also lack other epidermal components such as melanocytes and Langerhans cells, as well as both dermal and epidermal leukocytes, such that their immunologic repertoire is limited to that which is derived from keratinocytes alone. However, the fact that RHE models consist of keratinocytes only can actually be viewed as an advantage as well: with RHE models, the evaluation is limited to epidermal keratinocytes which enables one to be able to very specifically evaluate the pharmacologic and toxicologic effects that are limited to keratinocytes without any confounding contributions from other cellular components. Even so, several different alternations have been made to the RHE model in pursuit of increasing the utility/predictive value of this model for specific purposes.
One such specific alteration that is thought to increase the predictive ability of RHE models for the evaluation of cutaneous permeability/transport, as well as enabling their use for additional assessments, such as for cutaneous wound healing, is the addition of a dermal layer to the epidermal layer of the RHE model, since all of the RHE models are composed solely of viable and nonviable cornified keratinocytes (Semlin et al. 2011). Two such 3-D in vitro models that have added dermis to the reconstituted epidermal keratinocytes are EpiDermFT® and Apligraf®. These full-thickness in vitro epidermis/dermis models have been used for wound healing studies but have yet to be fully validated for the purposes of permeability/barrier function or transport evaluation (Semlin et al. 2011). Another specific alteration is the addition of melanocytes to the epidermal keratinocytes in order to enable evaluation of cutaneous melanocyte biology as well as evaluation of hypo- and hyperpigmentation (Passeron et al. 2007). Finally, a third example of an alteration that is thought to increase the predictive value of RHE models for the evaluation of phototoxicity and cutaneous contact hypersensitization potential is the addition of Langerhans cells (Facy et al. 2004; Semlin et al. 2011). While the addition of Langerhans cells to RHE has not yet been extensively evaluated, initial results show some promise (Facy et al. 2004).
In addition to their use for the evaluation of topically applied substances, RHE models have been increasingly used for the evaluation of skin toxicity from systemically administered agents, in studies examining the systemic biologic and pharmacologic effects of a variety of cytokines and growth factors, and for investigations into the pathophysiology of cutaneous diseases such as psoriasis (Boniface et al. 2005; Boniface et al. 2007; Sa et al. 2007; Semlin et al. 2011). RHE models have proved quite useful in modeling and predicting the cutaneous effects induced in vivo, including in the clinic, by a variety of different factors; several examples follow.
In vitro Epidermal Models for the Evaluation of Skin Toxicity
RHE models have proven to be good models for the evaluation of skin toxicity. For example, the epidermal growth factor receptor (EGFR) inhibitors, such as the antibody cetuximab and the small molecule erlotinib, have a well-known clinical dose-limiting toxicity of an acneiform rash that is often linked to the demonstration of clinical efficacy (Pomerantz, Mirvish, and Geskin 2010). In the EpiDerm RHE model, both EGFR inhibitors induced a decrease in epidermal thickness with the presence of occasional necrotic keratinocytes (Figure 1A) and also induced a marked decrease in the expression of phosphoS6, an intracytoplasmic Ser/Thr protein kinase that is activated/phosphorylated in response to EGFR engagement and activation (Figure 1B).
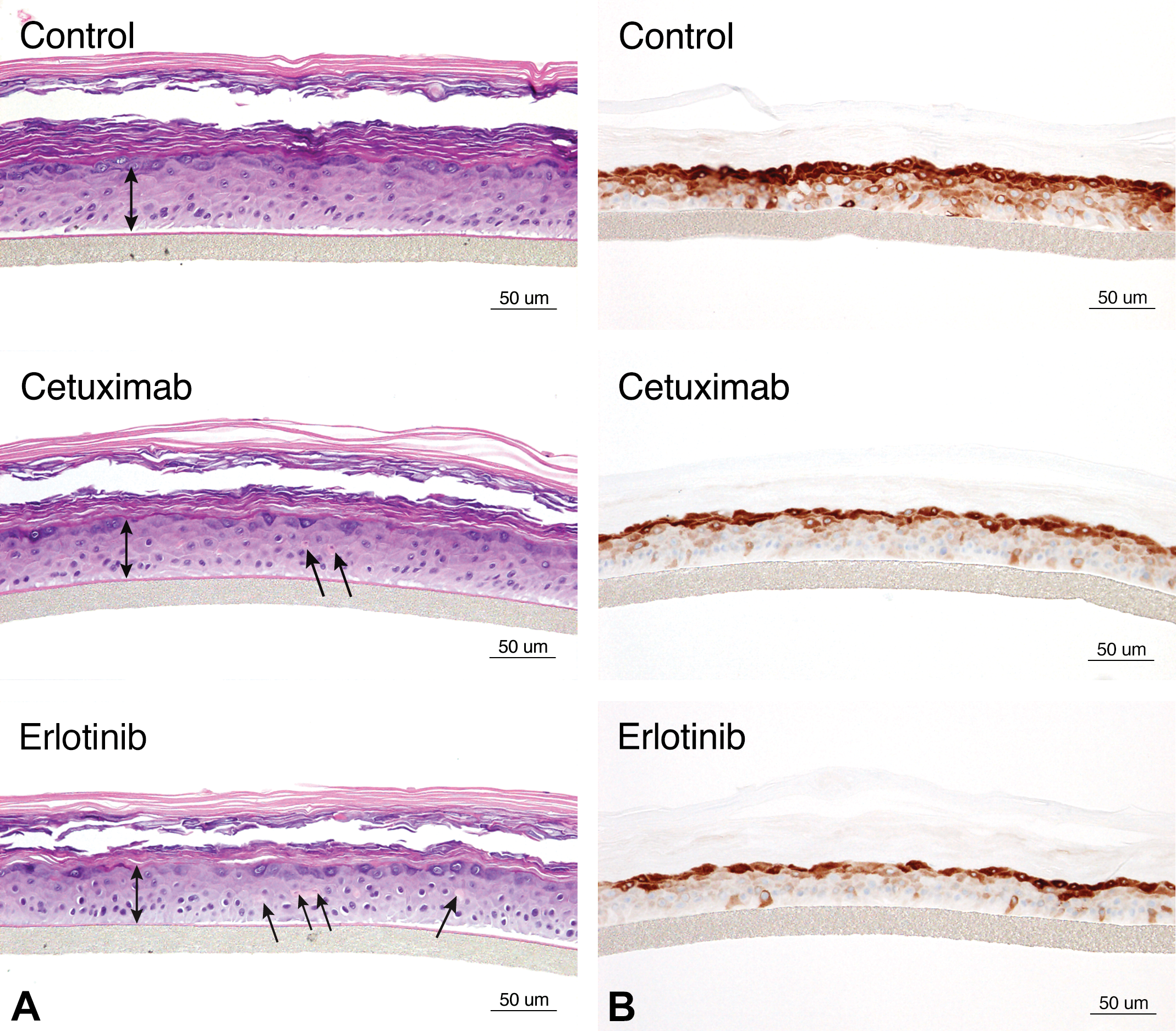
Sections of reconstituted human epidermis (EpiDerm®) stained with hematoxylin and eosin (A) and IHC stained for phosphoS6 protein (B) illustrating a reduction in the viable epidermal thickness in EpiDerm sections treated with the epidermal growth factor receptor (EGFR) inhibitors cetuximab and erlotinib (as denoted by a decreased length of the double-headed arrows compared to the untreated control). The decrease in epidermal thickness in sections treated with EGFR inhibitors is accompanied by epidermal keratinocyte single cell necrosis (arrows) as well as decreased staining for phosphoS6 (B), an intracytoplasmic effector protein within the EGFR signaling cascade. Figure reproduced with permission from Diegel, Danilenko, and Wojcinski (2013).
RHE models have also successfully mirrored/predicted the skin toxicities induced clinically by a number of small molecule kinase inhibitors, including inhibitors of MEK and B-Raf (LoRusso et al. 2010; Mandalà, Massi, and De Giorgi 2013). A specific, very interesting example of skin toxicity predicted by RHE is demonstrated by the specific inhibition of the V600E mutation of B-Raf. At low concentrations mimicking pharmacologic levels intended to inhibit the B-Raf V600E mutant pathway in malignant melanoma, inhibition of B-Raf V600E induces the paradoxical effect of epidermal hyperplasia in RHE (Figure 2). This effect is also seen in vivo in both animal models and clinically in humans, with malignant melanoma patients demonstrating beneficial effects on their malignancy but frequently developing hyperplastic epidermal lesions including keratoacanthomas and squamous cell carcinomas (Macdonald et al. 2015). This paradoxical effect is due to the stimulation of the MEK/ERK pathway in cells with wildtype B-Raf, such as in epidermal keratinocytes, while in cells with the V600E mutant B-Raf, such as malignant melanoma cells, the ERK/MEK pathway is inhibited (Hatzivassiliou et al. 2010). This stimulatory effect in the ERK/MEK pathway is illustrated in Figure 3 by increased ERK expression by IHC in B-Raf treated RHE. Since MEK inhibitors themselves induce clinical skin toxicity (LoRusso et al. 2010) and induce epidermal thinning effects in the RHE, as illustrated in Figure 4, it was hypothesized that combining an MEK inhibitor with a V600E mutant B-Raf inhibitor might both increase anti-melanoma efficacy while reducing epidermal toxicity. This hypothesis has indeed proven to be true, with patients treated with a combination of B-Raf and MEK inhibitors showing a lower clinical frequency of cutaneous tumors than patients treated with the mutant B-Raf inhibitor as a single agent (Larkin et al. 2014).
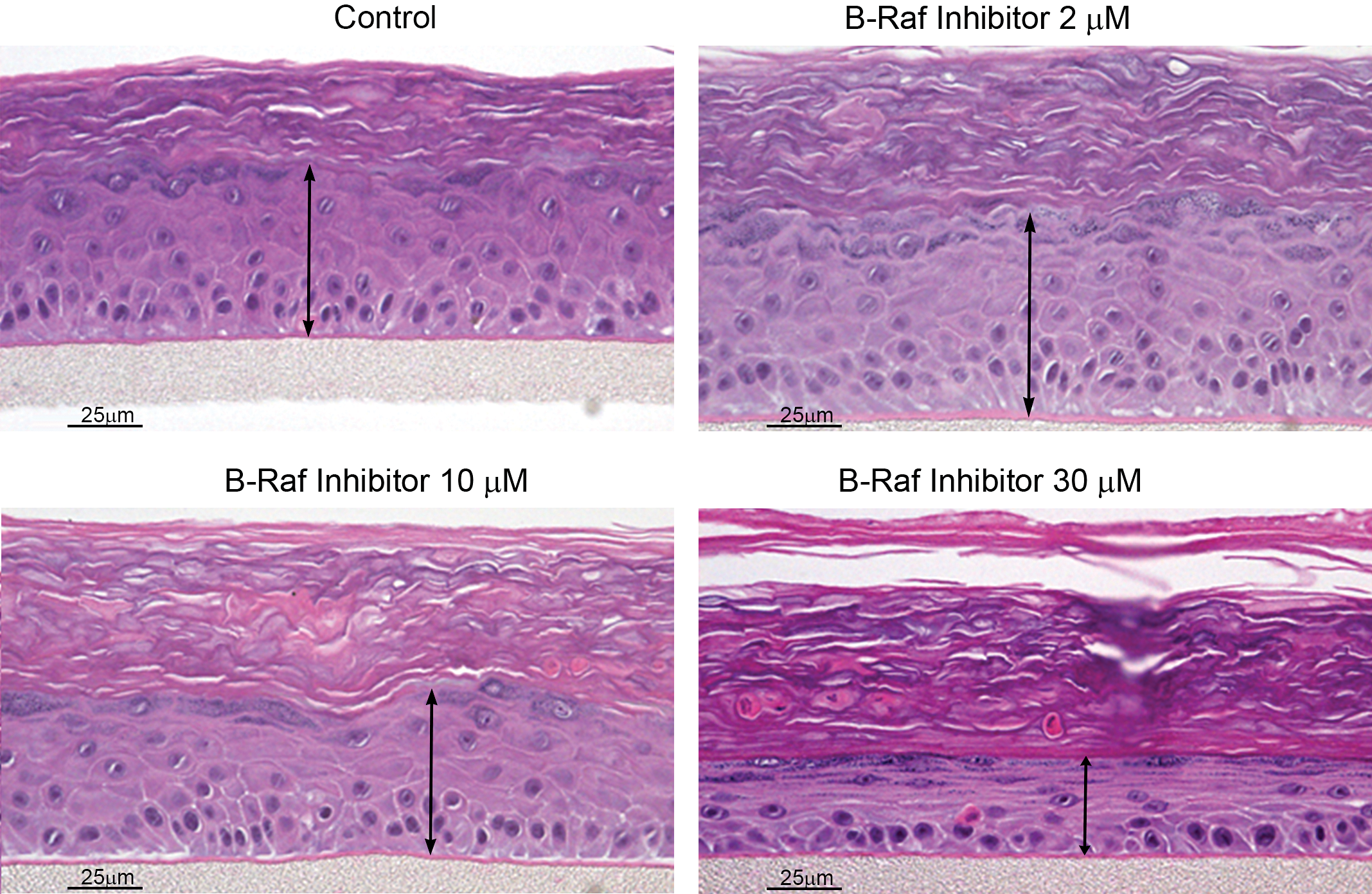
Effects of B-Raf inhibition in reconstituted human epidermis (RHE; EpiDerm®). A small molecule inhibitor of V600E mutant B-Raf induces differential concentration-dependent effects in the RHE model, with epidermal hyperplasia in RHE sections treated with a low concentration of 2-μM mimicking pharmacologic exposures but epidermal thinning in RHE sections treated at the higher concentrations of 10 and particularly at 30 μM.
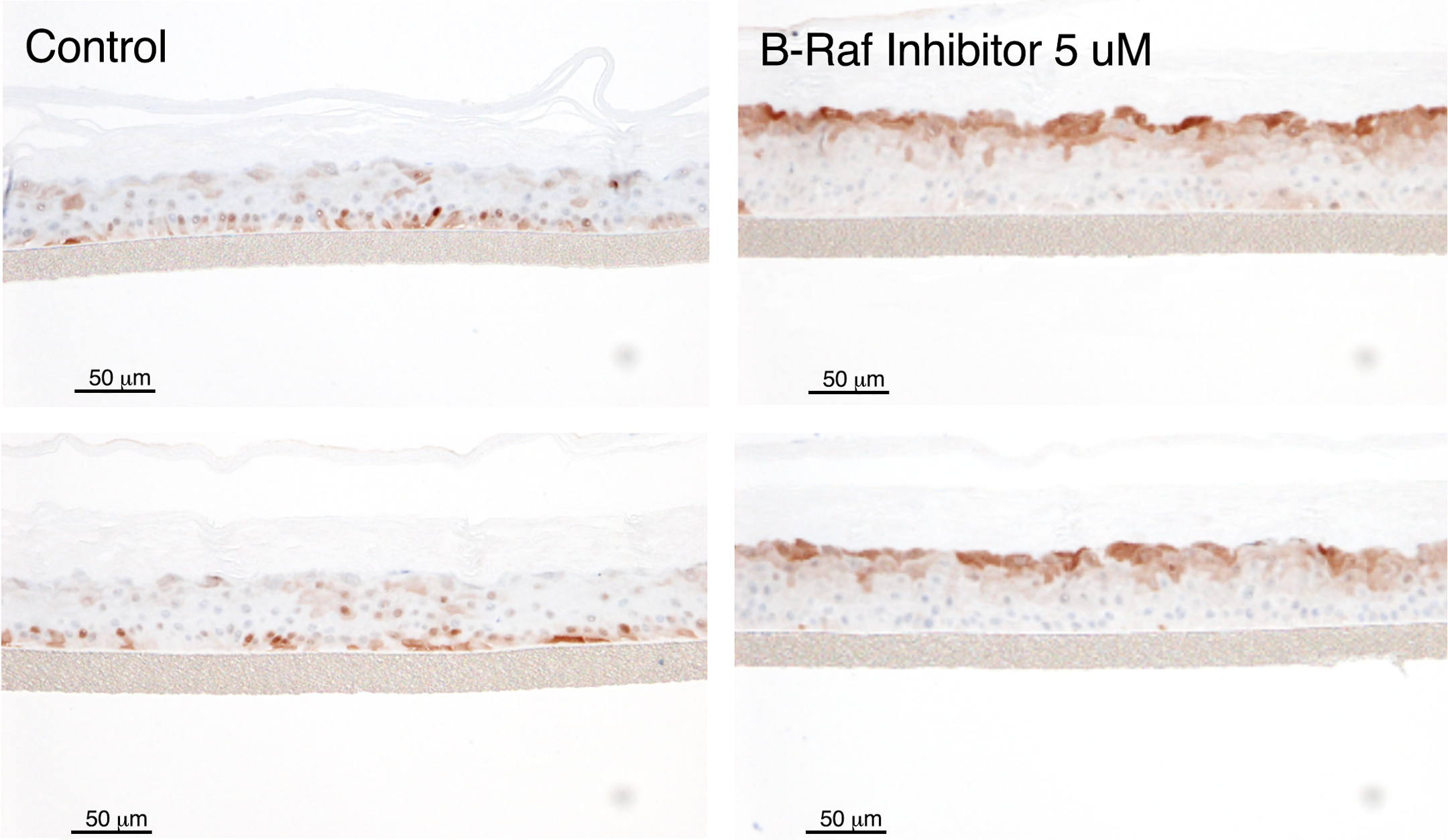
Effects of B-Raf inhibition on extracellular-signal-regulated kinase (ERK) expression in RHE (EpiDerm®). A small molecule inhibitor of V600E mutant B-Raf induces increased ERK expression in epidermal keratinocytes as detected by immunohistochemistry(IHC) staining in the RHE model, indicating activation of the mitogen-activated protein kinase kinase MAPK/ERK pathway activation.
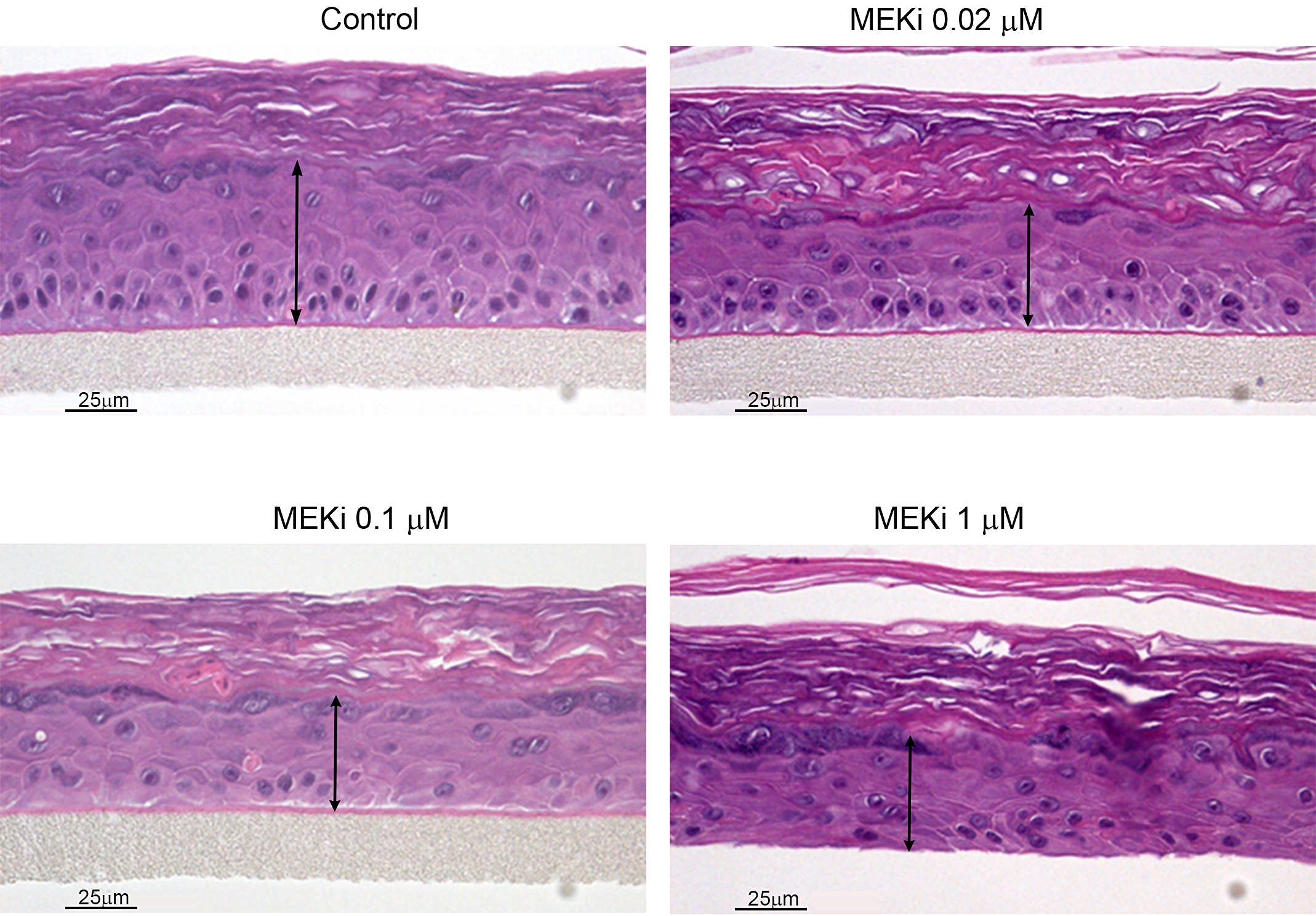
Effects of MEK inhibition in RHE (EpiDerm®). A small molecule inhibitor of MAPK/ERK kinase (MEK) induces a dose-dependent decrease in epidermal thickness in the reconstituted human epidermis model.
In vitro Epidermal Models for the Evaluation of Biology and Pharmacology
The RHE model has also been proven to be very valuable for investigations into the pathophysiologic/biologic and pharmacologic effects of cytokines and growth factors on epidermal keratinocytes. As an example, the interleukin-10 (IL-10) cytokine family members IL-19, IL-20, IL-22, and IL-24 were all found to induce morphologic, biochemical, and molecular changes consistent with some of the features found in psoriatic epidermis (Sa et al. 2007). IL-22, in particular, induced many of the same morphologic, biochemical, and molecular features in the EpiDerm RHE model that occur in the epidermis from psoriatic plaques (Boniface et al. 2005; Sa et al. 2007; Zheng et al. 2007; Figure 5). Some of the morphologic and differentiation features that IL-22 induced in RHE, such as hyperplasia and hypogranulosis (Figure 5A), upregulation of the expression of the hyperproliferative cytokeratin 6 (Figure 5B) and the antimicrobial peptide S100A7 (Figure 5C), and activation/phosphorylation of the nuclear transcription factor Stat3 (Figure 5D), are all features shared by psoriatic epidermis (Bowcock and Krueger 2005; Sano et al. 2005; Nograles et al. 2008). In notable contrast to the IL-22-induced effects on RHE, a tissue growth factor with very well-recognized effects on numerous epithelia including epidermis, namely, EGF, induced epidermal hyperplasia but did not induce any of the other effects that mimicked psoriatic epidermis in RHE (Sa et al. 2007).
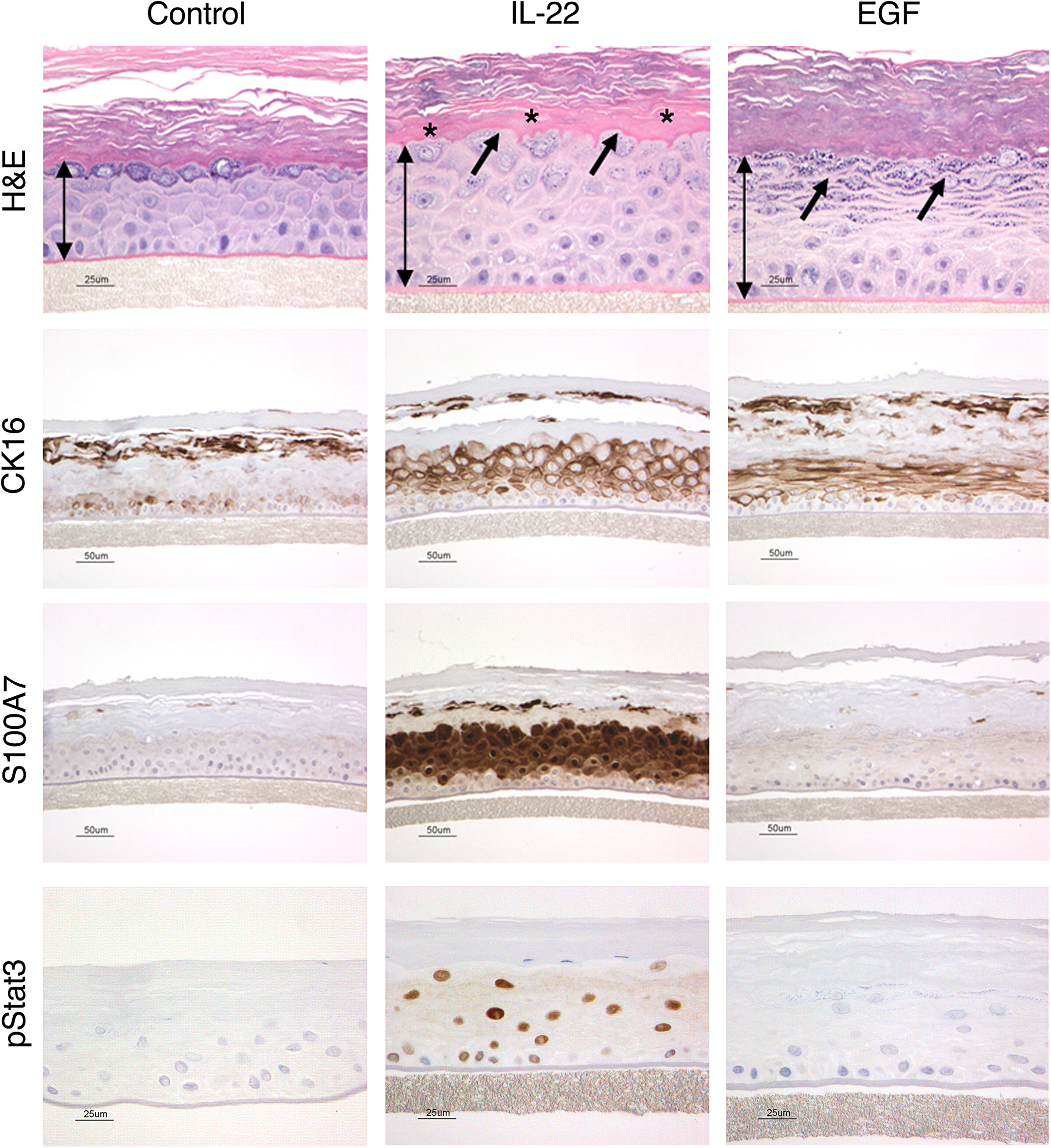
Effects of IL-22 on epidermal differentiation in RHE (EpiDerm®). H&E stained images across the top demonstrate that IL-22 induces epidermal hyperplasia, as denoted by the increased length of the double-headed arrows compared to the untreated control. In addition, IL-22 also uniquely altered normal epidermal differentiation as demonstrated by hypogranulosis, or a decrease in the granular cell layer (arrows), as well as hyalinization of the lower stratum corneum (asterisks*). In contrast, EGF also induced epidermal hyperplasia, but did not induce the other changes in epidermal keratinocyte differentiation. Similarly, images in the second row stained for cytokeratin 16, images in the third row stained for S100A7, and images in bottom row, stained for pStat3, all demonstrate that IL-22 alters epidermal differentiation and activates pSTAT3 as compared to EGF. These effects of IL-22 on reconstituted human epidermis are very similar to those seen in psoriatic epidermis. Figure reproduced from Danilenko and Wang (2012). The yin and yang of immunomodulatory biologics: assessing the delicate balance between benefit and risk, Toxicol Pathol 40, 272 -287, with permission from SAGE publications.
The ability of cytokines such as IL-22 as well as oncostatin M, a cytokine in the IL-6 family, to induce morphologic and differentiation features in keratinocytes that mimic those found in psoriatic epidermis, as well as stimulate keratinocytes to upregulate many of the same genes known to be upregulated in psoriatic skin in the absence of blood vessels and leukocytes, is remarkable and reinforces the hypothesis that epidermal keratinocytes are central to the initiation of cutaneous inflammation as well as for the initiation of the cutaneous response to injury, infection, and toxicity (Boniface et al. 2007; Sa et al. 2007). The effects induced by IL-22 in this RHE model system have been very faithfully reproduced in animal models, including in the minipig (Figure 6), where subcutanous (SC) injection of IL-22Fc induced psoriasiform epidermal hyperplasia as well as an increase in superficial dermal vascularity, likely secondary to IL-22-induced upregulation of VEGF in epidermal keratinocytes, as IL-22 has demonstrated in RHE (Sa et al. 2007). Similar epidermal and dermal effects were also seen following SC injection of IL-22Fc in humans (data not shown). Additional independent confirmation of the importance of IL-22 in the pathophysiology of psoriasis was demonstrated by significant amelioration of skin lesions following administration of an anti-IL-22 antibody in the CD45RBHi SCID (severe combined immunodeficiency) immune transfer mouse model of psoriasis (Ma et al. 2008). These findings in different animal models validated the in vivo and clinical pharmacologic relevance and predictability of this in vitro epidermal model system. Another example where the biologic effects of a growth factor in the RHE model were found to very closely mirror the effects of factor in vivo is keratinocyte growth factor(KGF), a member of the fibroblast growth factor(FGF) growth factor family (FGF-7). In the RHE model, KGF induces substantially greater epidermal hyperplasia than IL-22 or any other member of the IL-10 family (Sa et al. 2007). KGF’s hyperplastic effects in RHE closely mirrored the marked hyperplasia that KGF induced in minipig skin wounds (Danilenko et al. 1995), as well as in the buccal oral mucosa of both normal rhesus monkeys and normal healthy humans (Danilenko 1999), again confirming the value of the RHE model in modeling and predicting in vivo effects in animals as well as in humans.
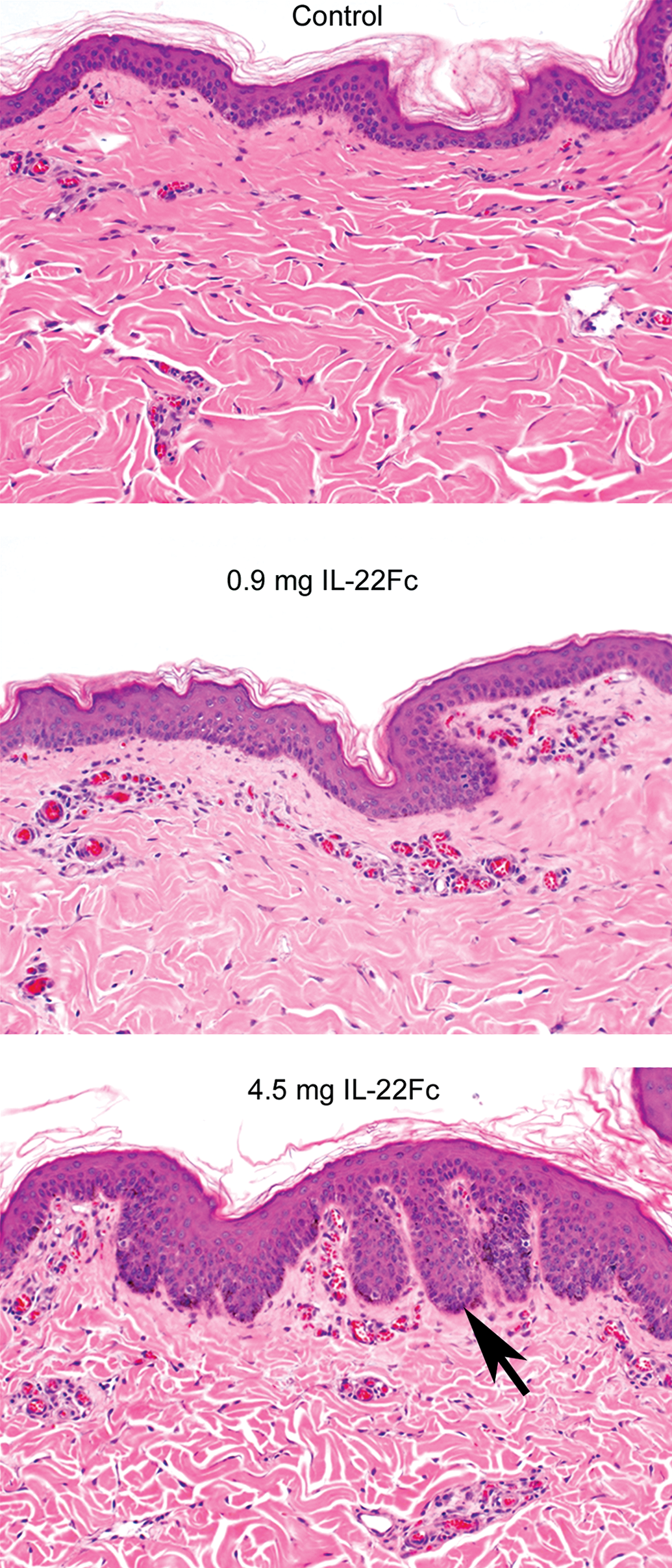
Hematoxylin and eosin stained sections of IL-22Fc subcutaneous injection sites from minipigs. IL-22Fc induces dose-dependent epidermal hyperplasia with a psoriasiform appearance as evidenced by exaggerated epidermal rete ridges (arrow) at the higher dose level of 4.5 mg as well as an increase in superficial dermal vascularity at both dose levels. These findings are very similar to those seen in RHE treated with IL-22, validating the in vivo relevance and predictability of this in vitro epidermal model system.
Plucked Human Scalp Hair Follicle Analysis
Another in vitro/ex vivo model whose use has increased recently is the ex vivo use of plucked human scalp hair follicles as surrogate pharmacodynamic biomarkers for measuring response to drug treatment, particularly antitumor agents, based on their sensitive and rapid response to drug exposure. The use of plucked hair follicles allows for the measurement of pharmacodynamic response(s) that is noninvasive and allows for repeated sampling without unduly stressing the subject, although care must be taken to ensure sample quality. Plucked hair follicles can be evaluated by immunohistochemistry or immunofluorescence to identify up- or downregulation of specific markers, such as Ki-67, EGFR, phospho-p27, phosphor-histone H3, phosphor-MAPK, or phosph-Rb (Randall and Foster 2007). Plucked follicles are also amenable to image analysis in order to quantify treatment-related changes of specific markers. For example, a trend toward downregulation of fatty acid binding protein 4(FABP4) expression in plucked human scalp hair follicles was found to be correlated with a diagnosis of schizophrenia, suggesting that the use of plucked hair follicles could be a beneficial genetic biomarker resource for brain diseases (Maekawa et al. 2015).
Conclusions
In vitro skin model systems, particularly RHE models, are increasingly being used both in the early evaluation of therapeutic drug candidates and in confirmatory mechanistic studies. The in vitro RHE models have many advantages when compared to in vivo models, as they can be done more quickly, easily, and reproducibly and thus can be used for high throughput compound screening while also reducing the need for animal studies. In addition, histologic evaluation of RHE sections, including quantification (both manual and automated) via image analysis, can be performed quite readily. RHE model systems can provide very valuable early indications of therapeutic candidate biology, pharmacology, and toxicity; and early results have demonstrated that RHE models have been quite predictive for in vivo pharmacologic and toxicologic effects in the skin, including clinical skin toxicity.
Footnotes
Acknowledgments
We would like to thank Susan Sa and Wenjun Ouyang of Genentech for their collaborative work on IL-22 in the RHE model and in particular thank Susan for her work in establishing the RHE model as a routine screening assay at Genentech. We would also like to thank Rama Pai and Carter Fields of Genentech for their collaborative work on the toxicity studies with small molecule kinase inhibitors and with the EGFR inhibitors, respectively, in the RHE model. In addition, we would like to thank Tanja Zabka of Genentech for the initial slide review and the original digital images of minipig skin used in ![]() . Finally, we would like to thank Harbeen Grewal, Mausumi Debnath, and the rest of the team at Anshin Biosolutions for their assistance in formatting the figure images.
. Finally, we would like to thank Harbeen Grewal, Mausumi Debnath, and the rest of the team at Anshin Biosolutions for their assistance in formatting the figure images.
Author Contribution
Authors contributed to conception or design (DMD); data acquisition, analysis, or interpretation (DMD, GL, DD); drafting the manuscript (DMD); and critically revising the manuscript (DMD, GL, DD). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
