Abstract
There is increased interest to use minipigs in ocular toxicology studies due to their anatomical similarities with human eyes and as a substitute for nonhuman primates. This requires adaptation of enhanced optical coherence tomography (OCT) techniques and of ocular relevant immunohistochemistry (IHC) or in situ hybridization (ISH) markers to porcine eyes. In this study, OCT and OCT angiography (AngioOCT) were performed on adult Göttingen minipigs. To increase structural information on retinal and choroidal vasculature, OCT data were speckle denoized and choroidal blood vessels were segmented with threshold filtering. In addition, we established a set of IHC and ISH markers on Davidson’s fixed paraffin-embedded minipig eyes: neurofilament-160, neuronal nuclei, calretinin, protein kinase C-α, vimentin, glial fibrillary acidic protein, glutamine synthetase, ionized calcium-binding adaptor molecule-1, rhodopsin, synaptophysin, postsynaptic density protein-95, retinal pigment epithelium (RPE)-specific protein-65, von Willebrand factor, α-smooth muscle actin, desmin, and Ki-67, thus enabling visualization of retinal neuronal and glial cells, photoreceptors, synapses, RPE, blood vessels, myocytes, macrophages, or cell proliferation. Using ISH, transcripts of vascular endothelial growth factor A, angiopoietin-2, and endothelial tyrosine kinase were visualized. This article describes for the first time in minipig eyes speckle noise–free OCT, AngioOCT, and a set of IHC/ISH markers on Davidson’s fixed paraffin-embedded tissues and helps to establish the minipig for ocular toxicology and pharmacology studies.
Keywords
Introduction
The principle of replacement, reduction, and refinement (3Rs) needs to be considered when selecting approaches for regulatory testing of human and veterinary medicinal products (Directive 2010/63/EU and revising Directive 86/609/EEC). In an attempt to reduce the use of nonhuman primates, we increasingly consider Göttingen minipigs for regulatory toxicology studies, including investigation of ocular drug delivery routes. In addition, the minipig is an interesting species in ocular research translatable to human disease conditions, such as glaucoma (Dan et al. 2008; Galdos et al. 2012; Ruiz-Ederra et al. 2005), retinitis pigmentosa (Ross et al. 2012), proliferative vitreoretinopathy (Umazume et al. 2012), choroidal neovascularization (CNV; Kiilgaard et al. 2005; Lassota et al. 2008), retinal degeneration (Huang et al. 2000) or detachment (Iandiev et al. 2006), in transplantation studies (Klassen et al. 2008), and as a model for selective retinal capillary closure in diabetic retinopathy (Foulds et al. 2010).
Due to the close anatomical and functional similarity of porcine and human eyes, advanced human ophthalmologic diagnostics can be applied (McLellan and Rasmussen 2012; Rosolen et al. 2012). There is increased interest to include optical coherence tomography (OCT) in ocular toxicology studies for safety profiling as it may be used as in vivo biomarker for longitudinal assessment and complement histopathology (McLellan and Rasmussen 2012; Rosolen et al. 2012). This noninvasive technique obtains live subsurface images of ocular structures at near microscopic resolution, which are highly translatable to clinics. Although OCT imaging has continuously provided more detailed information on the retinal zones in human medicine, deeper posterior ocular layers such as the choroid are still difficult to visualize due to the lack of penetration or signal loss through adsorption or scattering. In clinics, investigation of the choroid is important for the diagnosis and management of age-related macular degeneration, central serous chorioretinopathy (Okamoto, Matsuura, and Ogata 2015), tumors (Francis et al. 2015), diabetes (Rayess et al. 2015), glaucoma (Blumberg et al. 2015; Jia, Wei, et al. 2014; Mrejen and Spaide 2013), or multiple sclerosis (Wang et al. 2014). With the introduction of the enhanced depth imaging (EDI) technique (Wong, Koizumi, and Lai 2011), the choroid could be imaged with improved detail using spectral domain OCTs (SDOCT; Spaide, Koizumi, and Pozzoni 2008; Spaide 2009). Major advantages of the advanced swept source OCT (SSOCT) are higher speed and deeper tissue penetration, which allow for improved analysis and reconstruction of the choroid (Esmaeelpour et al. 2014). However, speckle noise–obscuring visualization of choroidal microvasculature remains a major drawback of OCT imaging. Recently, OCT angiography (AngioOCT) was introduced as novel dye-less method enabling three-dimensional (3D) visualization of the retinal and choroidal vascularization with depth-resolved information (Jia, Bailey, et al. 2014; Jia et al. 2015).
In pigs, ocular fluorescent dye angiography (Galdos et al. 2012) and high-quality SDOCT have been established (McLellan and Rasmussen 2012; Rosolen et al. 2012). In addition, there is increasing knowledge about the correlation of in vivo OCT scan results with histopathology of the porcine eye (Fatehee et al. 2011; Gloesmann et al. 2003; Huang et al. 2000; Rosolen et al. 2012). However, to the best of our knowledge, speckle noise–free OCT techniques including SSOCT and AngioOCT have not yet been described for pigs.
In toxicology studies, characterization of ocular changes depends on ophthalmologic examination and histopathology supported by special techniques including immunohistochemistry (IHC) or in situ hybridization (ISH). For pigs, a panel of cell type–specific antibodies has been established in paraformaldehyde-fixed cryosectioned eyes (Guduric-Fuchs et al. 2009; Johansson, Eftekhari, and Warfvinge 2010). However, in toxicology studies, the generally used fixatives for preserving eye morphology are modified or conventional Davidson’s fixative (DF), which provide improved tissue preservation (Latendresse et al. 2002). In order to examine eyes, histopathologically and simultaneously evaluate immunohistochemical markers in toxicity studies, a panel of markers was established for rat eyes fixed in DF (McKay et al. 2009). However, in the current literature, this information is not available for porcine eyes. Also, ISH is rarely performed on DF-fixed tissues (Hasson et al. 1997) and, to our knowledge, has not been described for DF-fixed porcine eyes. With increasing need for novel ocular therapies targeting vascular endothelial growth factor (VEGF) or other VEGF-related modulators of angiogenesis (Penn et al. 2008), availability of ISH methods for the detection of these targets in DF-fixed minipig eyes may also become important.
The purpose of our work was 2-fold: (1) to demonstrate for the first time speckle noise–free OCT and applicability of dye-free AngioOCT for improved in vivo visualization of retinal and choroidal vasculature in minipigs which can be routinely applied in preclinical studies and (2) to show applicability of a set of tissue-based IHC or ISH markers for neuronal and glial cells, photoreceptors, synapses, retinal pigment epithelium (RPE), macrophages, and vasculature and cell proliferation on DF-fixed paraffin-embedded minipig eyes while preserving good morphology for detailed histopathologic examination of eyes in toxicology studies.
Materials and Methods
Animals
Two male and 4 female adult Göttingen minipigs (Ellegaard Göttingen Minipigs A/S, Denmark) were used in this study. Animals were 15 to 33 months old, with body weights ranging between 14 and 24 kg. Minipigs were housed in pairs in double-spaced indoor kennels in an air-conditioned animal room (20°C ± 2°C) with a 12-hr light/dark cycle environment and background music coordinated with light hours. They were provided with standard minipig diet and tap water ad libitum. All animals were observed at least during the feeding period to ensure they were in good health. Ophthalmologic examinations were performed at intervals of 3 to 4 weeks and up to 4 times per animal. All procedures were in accordance with the respective Swiss regulations and according to the animal permissions granted by the Institutional Animal Care and Use Committee of the Cantonal Veterinary Office and the statement of the Association for Research in Vision and Ophthalmology for the Use of Animals in Ophthalmic and Vision Research. The Roche Innovation Center Basel is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
OCT
OCT scanning of the anterior and posterior ocular segments was performed in a dimly lit room. Following sedation by intramuscular (IM) injection (1 ml/15 kg body weight) of a hydrochloride powder mixture of 250 mg tiletamine and 250 mg zolazepam (Zoletil 100®; Virbac Laboratories, France) dissolved in 6.25 ml xylazine (20 mg/ml), 1.25 ml ketamine (100 mg/ml), and 2.5 ml butorphanol (10 mg/ml), animals were placed on a custom-made adjustable table in a prone position, covered with a heated warm air blanket, and monitored to maintain core body temperature. Heart rate and peripheral oxygen saturation (PulsOxy, SurgiVet V3402; Smiths Medical PM Inc., WI) were monitored and maintained at normal levels throughout the imaging sessions. Lactated Ringer’s solution was infused via an intravenous (IV) catheter (10 ml/kg/hr). Eye lids were fixed with an eye lid spreader, and pupils were dilated with 1 to 2 applications of 5% tropicamide eye drops (Mydriaticum Stulln®; Pharma Stulln GmbH, Stulln, Germany). Corneal wetting was performed by frequent application of an eye moistening spray (Dynawell 3®; Schalcon Spa, Rome, Italy).
OCT imaging was performed using 2 different laser systems: (a) SDOCT with an EDI mode (SDOCT, Spectralis HRA OCT®; Heidelberg Engineering, Germany) and (b) advanced 1050 nm SSOCT (DRI OCT-1 Atlantis® and DRI OCT Triton®; Topcon, Japan). SDOCT volumes were measured in an area of 8.6 mm × 5.7 mm × 1.9 mm, 261 multiline scan type sections, and 10 frame automatic real-time sampling in EDI mode. The SSOCT cube was imaged with a 3D scan pattern, 6.0 mm × 6.0 mm area, 256 cross scans were taken with a scan density of 512 × 128 (128 horizontal scan lines comprising 512 A-scans). Proper positioning of the eye was achieved by moving the head to get a rectangular scan axis with centering on the optic disc. Scan beam was focused manually or automatically for the SDOCT or SSOCT system, respectively.
OCT data were speckle denoized to increase structural information according to the method described (Gyger et al. 2014), and choroidal blood vessels were segmented with threshold filtering. In addition, in conjunction with the regular OCT scanning, AngioOCT was performed using a 1050 nm SSOCT and an angiography prototype software (DRI OCT Triton; Research Tool SS-OCT Angio Beta; Topcon, Japan). The 3D scan pattern was 3.0 mm × 3.0 mm, 256 B-scans.
Tissue Processing for Histopathology
Two weeks after the last OCT scanning, 2 minipigs were returned to stock. Four minipigs were deeply sedated by IM injection (1 ml/15 kg body weight) of a hydrochloride powder mixture of 250 mg tiletamine and 250 mg zolazepam (Zoletil 100), dissolved in 6.25 ml xylazine (20 mg/ml), 1.25 ml ketamine (100 mg/ml), and 2.5 ml butorphanol (10 mg/ml). Eyes were collected immediately after exsanguination of the animals following sacrifice by IV injection of an overdose of approximately 120 mg pentobarbital/kg body weight. Each globe was fixed by immersion in 200 ml of commercially available Davidson’s solution (Product No. A3200; AppliChem, Darmstadt, Germany) for 48 hr (without postfixation in 10% neutral-buffered formalin or 70% ethanol), sectioned in a vertical midsagittal plane of section through the pupil and including the optic disc, embedded in paraffin, and processed by routine histologic methods. Morphology of the eyes of all minipigs was diagnosed as normal by light microscopic evaluation of hematoxylin–eosin stained sections. As presence of macrophages was expected to be low or absent in untreated minipig eyes, a paraffin block from a DF-fixed pig eye diagnosed with vitreal and mild retinal perivascular macrophage infiltration, obtained from a different study, was used to establish an IHC marker for macrophages in porcine eyes.
IHC
Table 1 lists the panel of primary antibodies used for demonstration of RPE, macrophages, and vasculature and cell proliferation in DF-fixed paraffin-embedded minipig eyes, and Table 2 depicts the primary antibodies used for detection of retinal neuronal and glial cells, photoreceptors, and synapses. In addition, antibody source, host, secondary antibodies, and our optimized dilutions, incubation times, and recommended methods for pretreatment are detailed. In brief, paraffin-embedded eyes were sectioned at 3 to 4 μm, mounted onto standard glass slides, dried overnight at 40°C, deparaffinized, subjected to the antibody-specific pretreatments and immunostained in a Ventana Discovery XT® immunostainer (Ventana Medical Systems, Inc., Tucson, AZ). Ventana antibody diluent (Cat. 251-018) was used for dilution of the primary and secondary antibodies. After incubation with the primary antibody, the secondary antibody directed against the host species of the primary antibody was applied. Slides were stained with the Ventana Red Map TM® kit (Cat. 760-123) using the Streptavidin–biotin alkaline phosphatase detection system (red stain) and with the Ventana OmniMap anti-Rabbit HRP® detection kit (Cat. 760-4311) combined with the Purple® kit (Cat. 760-229, purple stain, only performed for α-smooth muscle actin [αSMA]), respectively, and were counterstained with hematoxylin. For negative controls, the primary antibody was replaced by the corresponding immunoglobulin isotype. Positive controls included sections from formalin-fixed minipig brain and liver to demonstrate localization of the ionized calcium-binding adaptor molecule-1 (Iba1) antigen to microglial cells and macrophages including Kupffer cells, respectively, brain to demonstrate localization of synaptophysin antigen to neurons, ileum to demonstrate localization of the Ki-67 antigen to proliferating crypt epithelium, and the minipig retina itself as internal control based on published data on retinal distribution of the majority of markers detected by immunofluorescence (Guduric-Fuchs et al. 2009; Johansson, Eftekhari, and Warfvinge 2010). In order to better visualize a positive staining reaction in the RPE, a modified melanin bleaching technique was employed (Kivelä 1995) prior to the IHC for RPE-specific protein-65 (RPE65). After manual deparaffinizing, slides were placed in phosphate-buffered saline (PBS) containing 10% H2O2 at room temperature overnight, transferred into PBS, and processed for IHC as detailed above.
Antibodies Used for Immunolabeling of Proliferation, Vasculature, and Retinal Pigment Epithelium (RPE) in Davidson’s Fixed Paraffin-embedded Minipig Eyes.
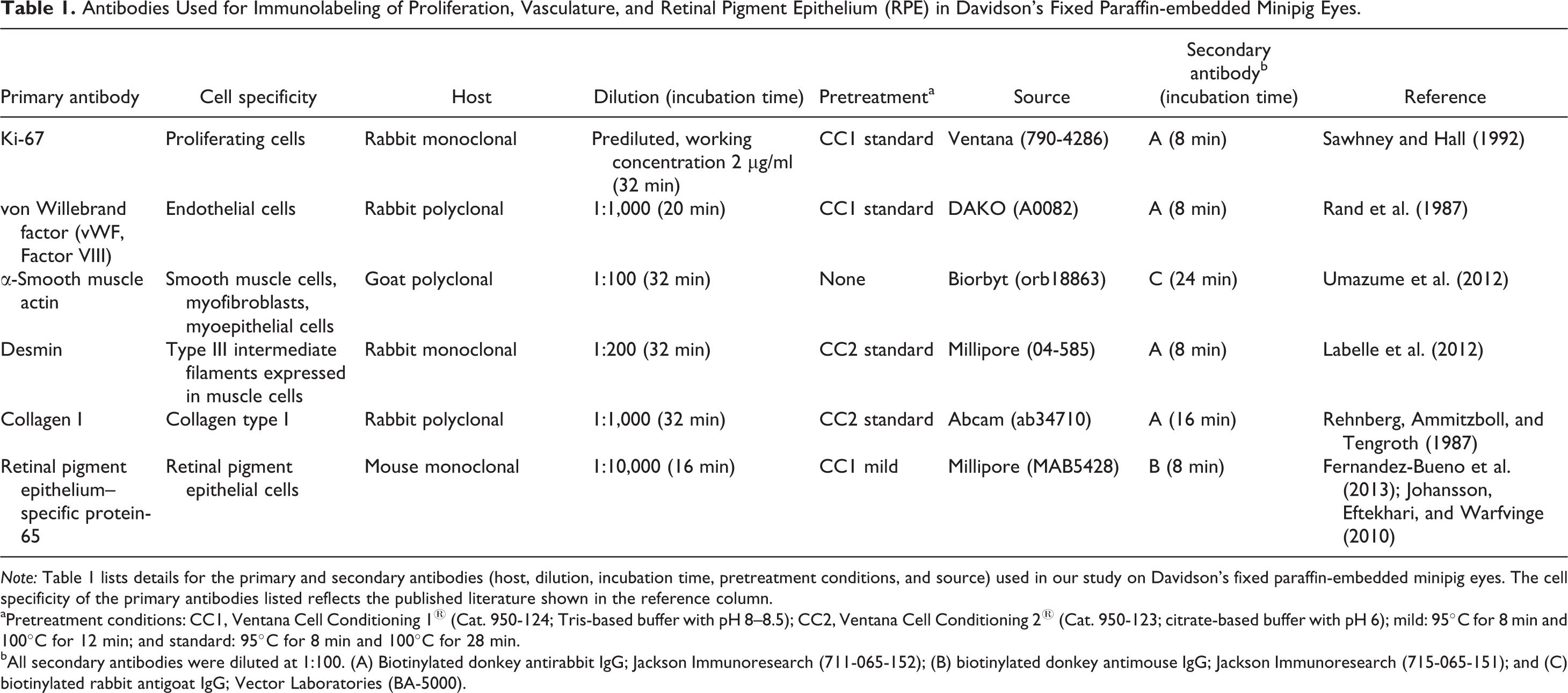
Note: Table 1 lists details for the primary and secondary antibodies (host, dilution, incubation time, pretreatment conditions, and source) used in our study on Davidson’s fixed paraffin-embedded minipig eyes. The cell specificity of the primary antibodies listed reflects the published literature shown in the reference column.
aPretreatment conditions: CC1, Ventana Cell Conditioning 1® (Cat. 950-124; Tris-based buffer with pH 8–8.5); CC2, Ventana Cell Conditioning 2® (Cat. 950-123; citrate-based buffer with pH 6); mild: 95°C for 8 min and 100°C for 12 min; and standard: 95°C for 8 min and 100°C for 28 min.
bAll secondary antibodies were diluted at 1:100. (A) Biotinylated donkey antirabbit IgG; Jackson Immunoresearch (711-065-152); (B) biotinylated donkey antimouse IgG; Jackson Immunoresearch (715-065-151); and (C) biotinylated rabbit antigoat IgG; Vector Laboratories (BA-5000).
Antibodies Used for Immunolabeling of the Neuroretina in Davidson’s fixed Paraffin-embedded Minipig Eyes.
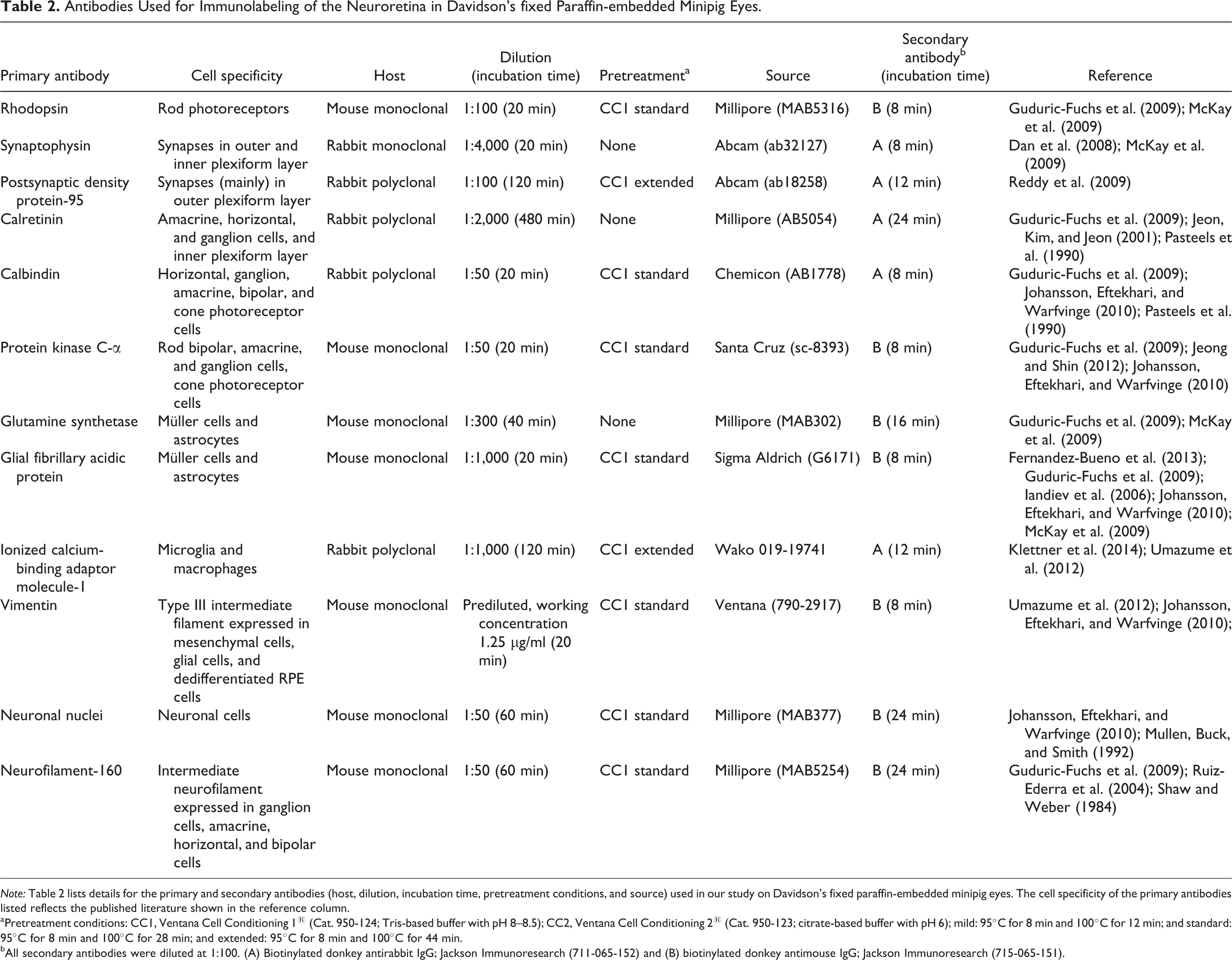
Note: Table 2 lists details for the primary and secondary antibodies (host, dilution, incubation time, pretreatment conditions, and source) used in our study on Davidson’s fixed paraffin-embedded minipig eyes. The cell specificity of the primary antibodies listed reflects the published literature shown in the reference column.
aPretreatment conditions: CC1, Ventana Cell Conditioning 1® (Cat. 950-124; Tris-based buffer with pH 8–8.5); CC2, Ventana Cell Conditioning 2® (Cat. 950-123; citrate-based buffer with pH 6); mild: 95°C for 8 min and 100°C for 12 min; and standard: 95°C for 8 min and 100°C for 28 min; and extended: 95°C for 8 min and 100°C for 44 min.
bAll secondary antibodies were diluted at 1:100. (A) Biotinylated donkey antirabbit IgG; Jackson Immunoresearch (711-065-152) and (B) biotinylated donkey antimouse IgG; Jackson Immunoresearch (715-065-151).
Slides were examined by light microscopy, and the location of red (Red Map kit) or purple (Purple kit) was indicated as membranous, cytoplasmic, or nuclear. For markers of retinal structures and cells, specific positivity was described according to the respective retinal cell layers, that is, RPE, inner and outer segments of the photoreceptor layer (PRL), outer nuclear layer (ONL) with nuclei of cones and rods, outer plexiform layer (OPL) with synapse of cone pedicles and rod spherules upon various bipolar cell and horizontal cell types, inner nuclear layer (INL) with nuclei of bipolar, Müller, amacrine and horizontal cells, inner plexiform layer (IPL) with synapses of bipolar cells upon amacrine cells and dendrites of ganglion cells, the ganglion cell layer (GCL) with ganglion cells and displaced amacrine cells, and the nerve fiber layer (NFL) including astrocytes.
ISH
VEGFA, angiopoietin-2 (ANGPT2), and tyrosine kinase, endothelial (TEK) messenger ribonucleic acid (mRNA) were visualized in minipig retina samples obtained from DF-fixed paraffin-embedded eyes, using RNAscopeVS® (Advanced Cell Diagnostics, Inc., Hayward, CA), an RNA ISH method that permits signal amplification and background suppression (Wang et al. 2012). Eye tissue sections (3–4 μm thick) were deparaffinized and treated with protease according to the manufacturer’s instructions. VEGFA-, ANGPT2,- or TEK-specific mRNA target probe sets for Sus scrofa were provided by the manufacturer (Gene ID 397157, targeted bps 4 through 422 of the VEGFA complementary deoxyribonucleic acid [cDNA] sequence, Gene ID 396730, targeted bps 441 through 1,391 of the ANGPT2 cDNA sequence, and Gene ID 396729, targeted bps 501 through 3,122 of the TEK cDNA sequence). Following signal amplification, slides were stained with Fast Red and counterstained with hematoxylin. All steps of this procedure were carried out using a Ventana Discovery Ultra® (Ventana Medical System, Tucson, AZ) immunostainer. Probes to the bacterial gene bacterial dihydrodipicolinate reductase and the endogenous ubiquitin C mRNA were used as negative and positive controls, respectively. Specific staining signals were identified by light microscopy as red, punctate dots in the cytoplasm.
Results
OCT Imaging of the Chorioretinal Vasculature and Optic Disc
In minipigs, color fundus photographs could be readily obtained with the integrated multimodal Triton® color fundus imaging system (Figure 1A). The vertically oval optic nerve head and its central pale depression (optic cup), white disc margin with darkly pigmented inferior rim, medium-sized retinal blood vessels descending into the cup, and a few larger retinal blood vessels were outlined. Choroidal blood vessels were located around the optic disc in a circumlinear pattern. AngioOCT imaging (Figure 1B) revealed a superficial, dense, filigrane capillary network between the larger retinal blood vessels which had been poorly visible by color fundus photography. These superficial blood vessels appeared to be directed radially toward the optic disc, following the direction of the retinal nerve fibers. Smaller branching blood vessels emerged at an angle of 90° or, more frequently, at an acute angle. The deep retinal vascular complex consisted of a honeycomb-like bed of capillaries (Figure 1C). The choriocapillaris appeared as fairly uniform structure, with alternating bands caused by underlying choroidal blood vessels. The optic disc margin was undulated. Several depressions in the optic nerve head represented superficial openings in the lamina cribrosa (Figure 1D).
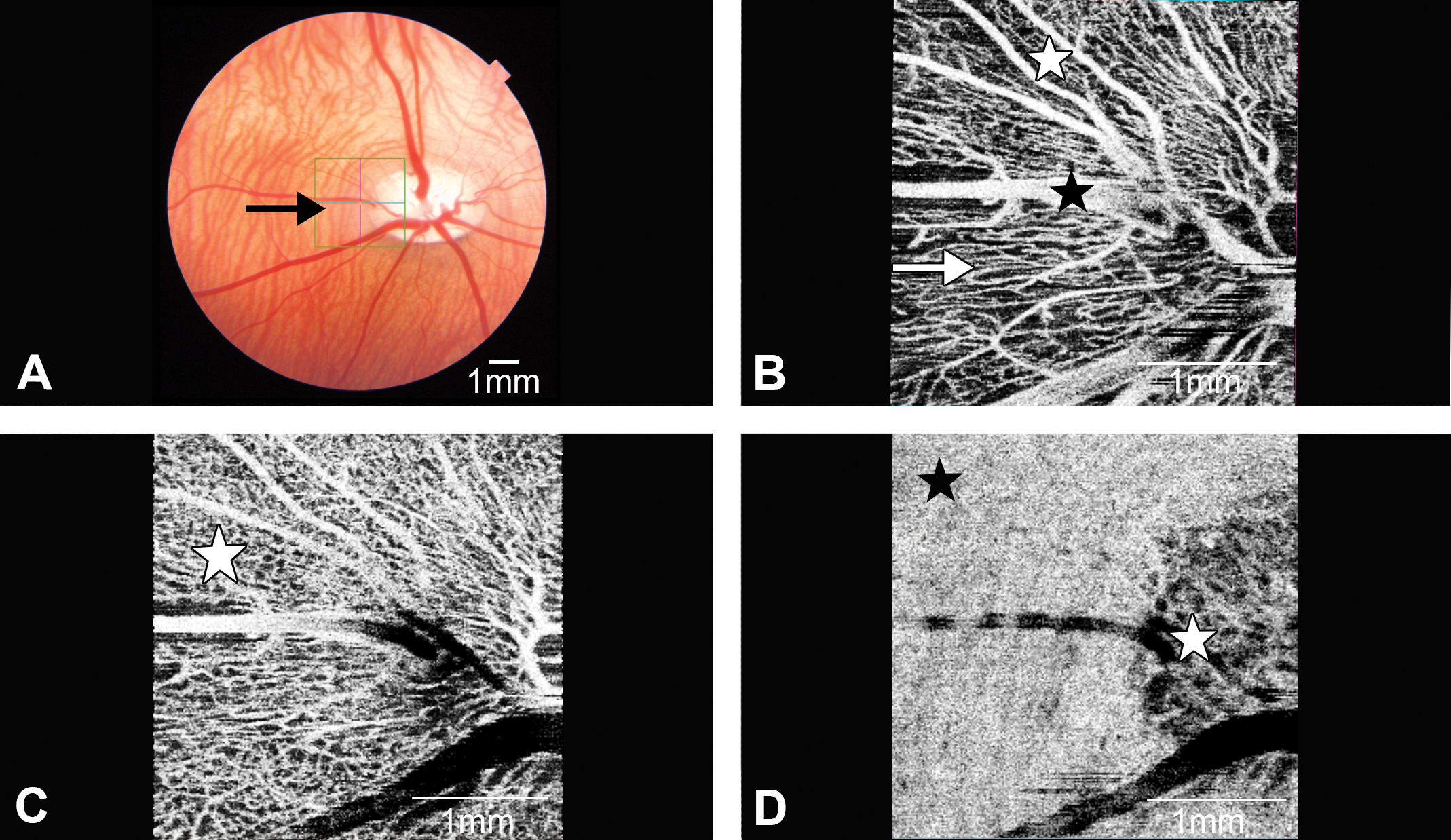
Swept source optical coherence tomography (OCT)-angiography (AngioOCT) of minipig retina and optic disc. (A) Color fundus photography documenting retinal and choroidal blood vessels and optic nerve head. Highlighted box (arrow) indicates area of swept source OCT imaging. (B) Advanced computerized imaging of superficial large (black asterisk) and medium-sized (white asterisk) retinal blood vessels using AngioOCT without dye injection. Blood vessels located superficially in retinal nerve fiber layer (arrow) are directed toward optic nerve head. Between blood vessels, only few blood vessel branches are found. (C) AngioOCT imaging of vascular meshwork of deep retinal vascular complex. In this metabolism-rich zone, abundant vascular interconnectivities are arranged in numerous loops (asterisk). (D) Choriocapillaris (black asterisk) and superficial pores of lamina cribrosa (white asterisk) of optic disc are visualized.
Cross-sectional OCT imaging of the minipig posterior ocular segment revealed clearly demarcated retinal zones adjacent to the outer vitreal cortex which consisted of multiple dense, hyperreflective lines parallel to the retinal surface (Figure 2A). Superficial larger retinal blood vessels protruded into the vitreal cortex and demonstrated a typical posterior signal loss. Choroidal blood vessels appeared as hyporeflective holes. Speckle noise removal was achieved without major distortion of the normal retinal structure (Figure 2B). It resulted in a gain of structure contour and better demarcation between tissue and fluid-filled cavities, thus facilitating extraction of choroidal blood vessels. In 3D density OCT images, retinal and choroidal blood vessels were located on top of each other (Figure 2C). Choroidal blood vessel extraction via postprocessing threshold filtering (Figure 2D) did not reveal any choroidal vascular loops that could be clearly separated from each other. In addition, the connection between the blood vessel fragments appeared to be partially interrupted, especially in areas with superimposed vessels and weak OCT signals. Presence of speckle noise had a different distribution in EDI-SDOCT (Figure 2E) and SSOCT (Figure 2F) images, respectively. EDI-SDOCT imaging resulted in better resolution due to superimposition (oversampling) of 20 B-scans into 1 image (Figure 2E). In contrast to EDI-SDOCT, SSOCT imaging with single B-scan acquisition was more blurred due to diffuse speckle noise distribution (Figure 2F).
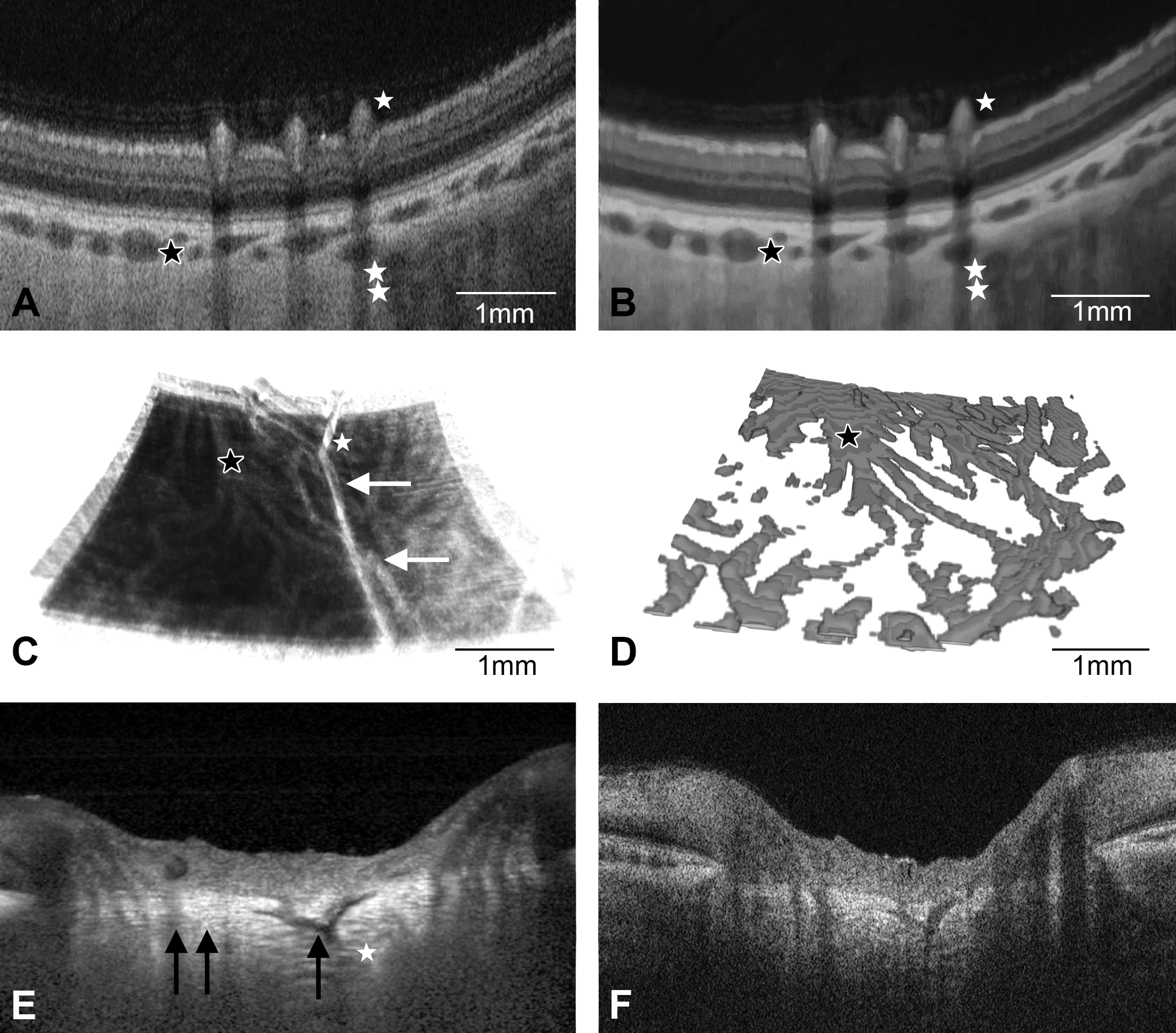
Optical coherence tomography (OCT) of minipig eye with inner and outer retinal vascular complex. (A) Conventional cross-sectional swept source optical coherence tomography (SSOCT) of minipig retina. Three superficial medium-sized retinal blood vessels protrude into the dense outer vitreal cortex and are associated with vertical linear “shadows” (single white asterisk). Choroidal blood vessels are shown as hyporeflective black cavities (black asterisk), and deep scleral blood vessels are visible (double white asterisk). Note diffuse blur due to of speckle noise. (B) Speckle noise–freed SSOCT of the same area depicts clearly outlined retinal blood vessels (single white asterisk). Choroidal (black asterisk) and deep scleral blood vessels (double white asterisk) are better visible after speckle noise removal. (C) Three-dimensional (3D) OCT of retinal (white asterisk) and choroidal (black asterisk) blood vessels. Larger retinal blood vessels appear as straight lines (white arrows). (D) 3D representation of extracted choroidal blood vessels (black asterisk) from speckle noise–freed 3D-OCT data. (E) Enhanced depth imaging (EDI) mode of conventional spectral domain optical coherence tomography (SDOCT) cross scan in optic disc with central blood vessel (single black arrow) and hyperreflective band of lamina cribrosa (double black arrow). Note that the averaged EDI-SDOCT enhances visualization of the posterior part of the optic disc (white asterisk) but also produces more noise. (F) Conventional SSOCT cross scan without averaging of the same optic disc demonstrating a more uniform speckle noise distribution.
IHC of Davidson’s Fixed Paraffin-embedded Minipig Eyes
Tables 1 and 2 list the panel of primary antibodies, which we successfully established for immunohistochemical demonstration of RPE, macrophages, vasculature and cell proliferation, and retinal neuronal and glial cells, photoreceptors, and synapses, respectively, in DF-fixed paraffin-embedded minipig eyes. In addition, antibody source, host, secondary antibodies, and our optimized dilutions, incubation times, and recommended methods for pretreatment are summarized. For all antibodies stated, we were able to demonstrate the specific staining of the cell type or structure they were described to identify, referenced in the literature listed in Tables 1 and 2.
Using an antibody to Ki-67 to demonstrate cell proliferation, strong specific nuclear immunostaining was detected in individual corneal basal epithelial cells undergoing chromatin condensation during cell proliferation (Figure 3A). Likewise, Ki-67 positivity was present in the proliferating crypt epithelium of the minipig ileum used as positive control.
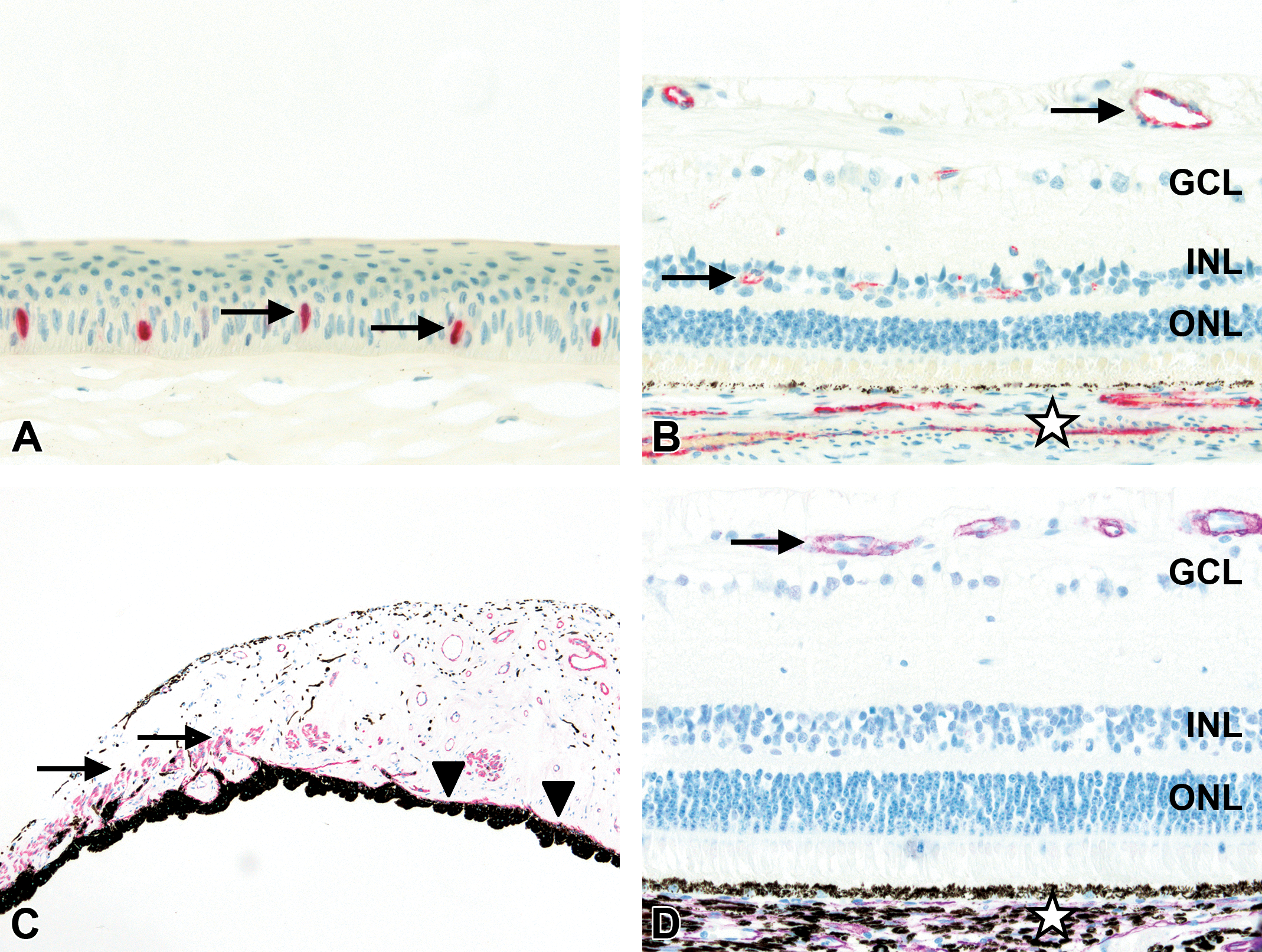
Immunohistochemistry of minipig eye. (A) Nuclear labeling for Ki-67 of individual basal epithelial cells in cornea (arrows; 40× original objective). (B) Labeling for von Willebrand factor of vascular endothelial cells of inner structures of neuroretina (arrows) and choroid (asterisk; 40× original objective). (C) Labeling for α-smooth muscle actin (αSMA) of iris sphincter (arrows) and dilator muscle (arrowheads; 10× original objective). (D) Labeling for αSMA of vascular smooth muscular vessel wall of inner structures of neuroretina (arrow) and choroid (asterisk; 40× original objective). For orientation, ganglion cell layer (GCL), inner nuclear layer (INL), and outer nuclear layer (ONL).
Using an antibody to von Willebrand factor (vWF, Factor VIII), strong specific immunolabeling was detected in endothelial cells of capillaries, small and larger blood vessels of all ocular compartments including the choroid and inner structures of the neuroretina (Figure 3B) but not in the lens or cornea.
α-SMA, a marker for smooth muscle and myofibroblasts, was localized in the smooth muscle wall of blood vessels of all ocular compartments including the choroid and inner structures of the neuroretina (Figure 3D), but not in the lens or cornea. In addition, αSMA was expressed in the ciliary muscle and in the iris sphincter and dilator muscles (Figure 3C).
Using antidesmin, type III intermediate filaments were detected in the muscle wall of large blood vessels in the episclera, retrobulbar orbital fat and vascular circle of the optic nerve, in the ciliary muscle, and in the iris sphincter muscle. However, there was no desmin positivity in the iris dilator muscle. Using anticollagen type I, specific positive staining was detected in the sclera, in the stroma of the cornea, iris and ciliary body, and in the lamina cribrosa and epi- and perineurium of the optic nerve.
RPE65 was detected by an anti-RPE65 antibody within the cytoplasm of the single layer of RPE cells containing pigment granules, located between the outer segments of the photoreceptors of the retina and choriocapillaris (Figure 5A). Likewise, this antibody worked well on DF-fixed paraffin-embedded minipig ocular tissue following bleaching to eliminate the melanin content within the RPE (Figure 5B).
Rod outer segments of the retinal photoreceptors were intensely labeled with an antirhodopsin antibody, whereas inner segments displayed a much weaker staining (Figure 5C). Antibodies against the intermediate filaments glial fibrillary acidic protein (GFAP) and vimentin, and against glutamine synthetase, were used as glial-specific markers in the retina (Figure 4). Astrocytes and Müller cells are variants of retinal macroglia. Both contribute to the formation of the glia limitans of the retina (inner limiting membrane [ILM]), blood vessels, and the glial sheaths of neurons (Hollander et al. 1991). Specific cytoplasmic expression of GFAP was detected in the NFL including the ILM, along the GCL, and around blood vessels in the neuroretina. This was consistent with a specific staining of astrocytes with their cell bodies and processes in the NFL, and Müller cell processes. In addition, there was specific GFAP staining of fine linear processes extending from the ILM to the INL, corresponding to Müller cell processes and cell bodies (Figure 4A). Using an antibody to glutamine synthetase, specific cytoplasmic staining was detected in a fine linear pattern, reaching from the ILM and NFL through the IPL to the INL. This was consistent with a specific staining of astrocytes in the NFL and of Müller cell processes and cell bodies in the IPL and INL, respectively (Figure 4B). Vimentin immunolabeling was present in the NFL, with fine processes through the IPL to the INL, and around blood vessels in the neuroretina and choroid (Figure 4C).
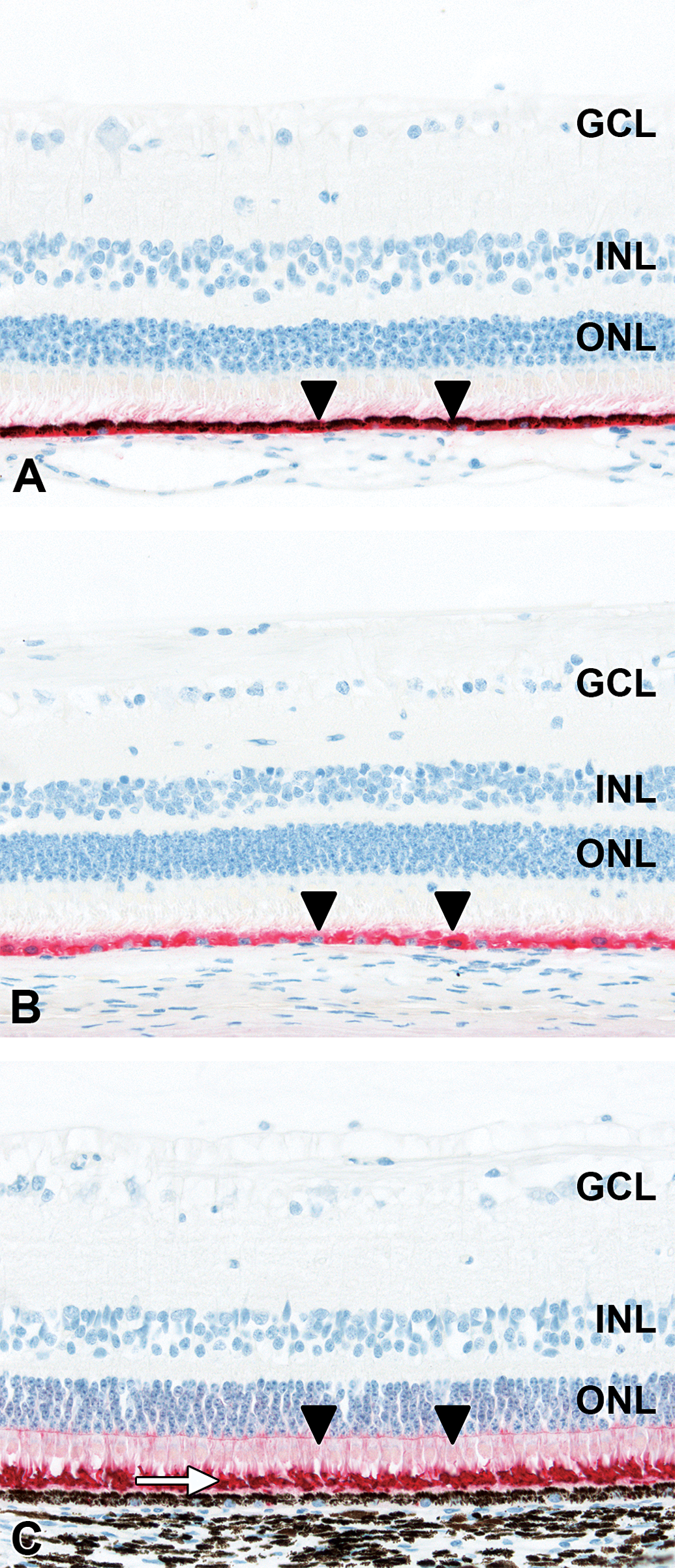
Immunohistochemistry of minipig retinal glia. (A) Labeling for glial fibrillary acidic protein in nerve fiber layer (NFL), ganglion cell layer (GCL) around blood vessels (asterisk), and of linear processes extending to the inner nuclear layer (INL; arrow). (B) Labeling for glutamine synthetase in a fine linear pattern (arrows) reaching from NFL to INL. (C) Labeling for vimentin from NFL with fine processes to INL (arrows) and around blood vessels in neuroretina and choroid (arrowheads). For orientation, GCL, INL, and outer nuclear layer (ONL). 40× original objective.
Immunohistochemistry of minipig neuroretina. (A) Labeling for retinal pigment epithelium-65 (RPE65) of RPE (arrowheads). (B) Labeling for RPE65 of RPE (arrowheads) on a slide bleached before immunohistochemistry. (C) Labeling for rhodopsin more intense in rod outer segments (arrow) than in inner segments in photoreceptor layer (arrowheads). For orientation, ganglion cell layer (GCL), inner nuclear layer (INL), and outer nuclear layer (ONL). 40× original objective.
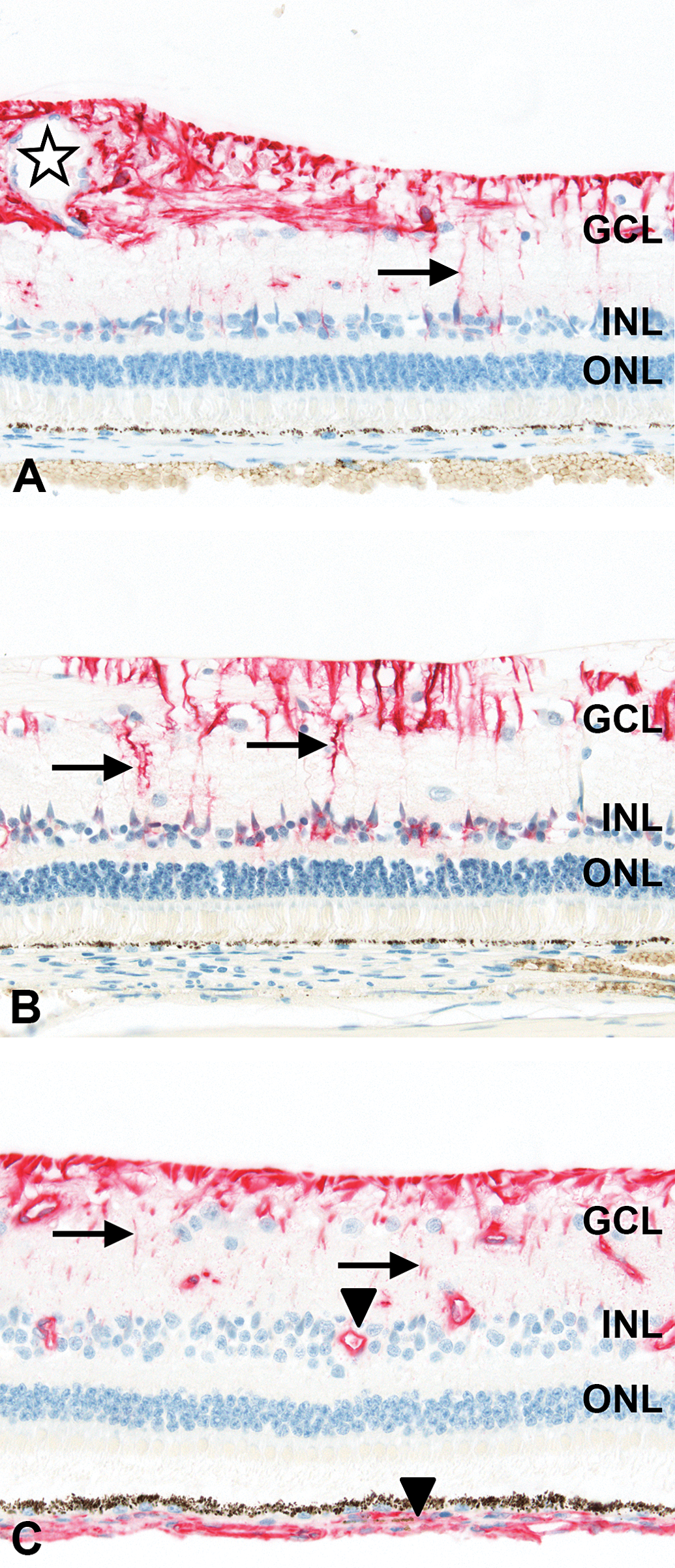
Using an antibody to Iba1, microglial cells were detected in the inner zones of the retina, predominantly in the INL with extending processes into the GCL, INL, and OPL (Figure 6A). As Iba1 is described to be also expressed on macrophages (Umazume et al. 2012), we used this antibody on a paraffin block from a DF-fixed pig eye diagnosed with vitreal and retinal perivascular macrophage infiltration and were able to demonstrate a specific positive cytoplasmic reaction in macrophages and activated microglial cells (Figure 6B). Likewise, Iba1 antigen was shown to localize to microglial cells or macrophages including Kupffer cells in the minipig brain or liver, respectively, used as positive controls.
Immunohistochemistry of minipig neuroretina. (A) Labeling for ionized calcium-binding adaptor molecule-1 (Iba1) of microglial cells with extending processes (arrow). (B) Labeling for Iba1 of vitreal macrophages (arrowheads). (C) Labeling for synaptophysin of retinal synapses in outer plexiform layer (OPL; arrow) and inner plexiform layer (IPL; asterisk) as well as of processes in outer nuclear layer (ONL) and inner nuclear layer (INL) and of the photoreceptor inner segments. (D) Labeling for postsynaptic density protein-95 of synapses in OPL (arrow). (E) Labeling for neurofilament-160 of ganglion cells (arrow) and their axonal processes in nerve fiber layer. In addition, staining of OPL and IPL consistent with the processes of horizontal and bipolar and amacrine cells, respectively. (F) Labeling for neuronal nuclei of ganglion cells (strong nuclear and less intensive cytoplasmic staining; arrow) and of amacrine cells in INL (arrowheads). For orientation, ganglion cell layer (GCL), INL, and ONL. 40× original objective.
Synapses in the retinal OPL and IPL were demonstrated using antibodies against synaptophysin (Figure 6C) and postsynaptic density protein-95 (PSD95; Figure 6D). Immunolabeling with antisynaptophysin as a marker for synapse integrity in the retina revealed strong specific staining of the photoreceptor terminals in the OPL and of the terminals of the bipolar cells in the IPL. In addition, there was weaker staining of processes in the ONL and INL and of the photoreceptor inner segments. Likewise, synaptophysin positivity was present in neurons in the brain used as positive control. Labeling with anti-PSD95 was prominent and intense in the retinal OPL only.
Retinal ganglion, amacrine, horizontal, and bipolar cells were identified using antibodies against neurofilament-160 (NF160) or neuronal nuclei (NeuN; Figure 6), and calretinin, calbindin, or protein kinase C-α (PKCα; Figure 7).
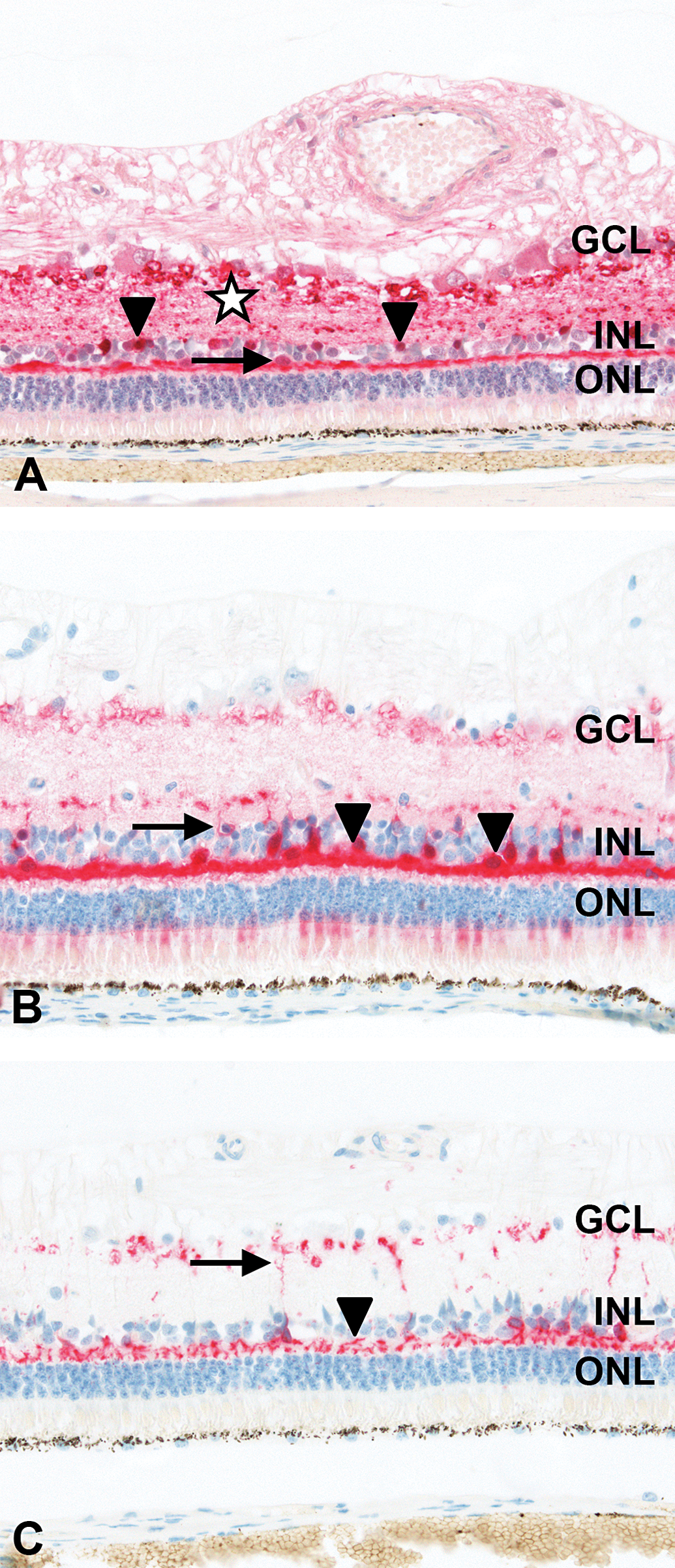
Immunolabeling of minipig neuroretina. (A) Labeling for calretinin of amacrine cells at inner rim of inner nuclear layer (INL; arrowheads) and their processes in inner plexiform layer (IPL; asterisk). In addition, labeling of horizontal cells (arrow) at outer rim of INL, and in the outer plexiform layer (OPL) and of ganglion cells. (B) Labeling for calbindin of horizontal cells at outer rim of INL and their processes in OPL (arrowheads). In addition, labeling of cell bodies in INL and processes in IPL consistent with bipolar cells (arrow). (C) Labeling for protein kinase C-α of synaptic connections of bipolar cells in OPL (arrowhead) and IPL (arrow). For orientation, ganglion cell layer (GCL), INL, and outer nuclear layer (ONL). 40× original objective.
Using an antibody to intermediate NF160, intense-specific immunolabeling was detected in the cytoplasm of ganglion cells in the GCL and their axonal processes in the NFL and extending to the optic nerve. There was strong specific staining in the retinal OPL, consistent with processes of horizontal and bipolar cells, and at the inner margin of the INL adjacent to the IPL, corresponding to the processes of amacrine cells (Figure 6E).
Applying a neuron-specific antibody against NeuN, specific strong labeling was present in the nuclei but also less intensely in the cytoplasm of ganglion cells in the GCL. Stained cells in the GCL had different sizes and thus likely included displaced amacrine cells. NeuN also weakly stained the soma of amacrine cells at the inner margin of the INL. In addition, there was weak NeuN staining in the cytoplasm of RPE cells (Figure 6F).
Using an antibody against calretinin, amacrine cell bodies located in a tier-like fashion at the inner margin of the INL and displaced amacrine cell bodies in the GCL were strongly and specifically positive. In addition, the IPL was diffusely positive, consistent with immunolabeling of amacrine cell processes. Calretinin was also expressed in individual cell bodies of horizontal cells at the outer side of the INL and in the OPL. Weak positivity for calretinin was present in ganglion cells (Figure 7A).
Using an anticalbindin antibody, horizontal cells showed strong specific cytoplasmic and nuclear staining of their cell bodies in the outer rim of the INL and axonal processes in the OPL. Specific expression of calbindin in cell bodies was also present within the INL, consistent with soma of bipolar cells. In addition, there was weaker staining of cell bodies at the inner rim of the INL (amacrine cells), of axonal processes in the IPL (bipolar cells), of individual ganglion cells in the GCL, and of sparse inner photoreceptor segments suggestive of cones (Figure 7B).
Using an antibody against PKCα, intense-specific staining was detected at the level of synaptic connections of bipolar cells in the OPL and IPL. Strong PKCα-expression was also present along their axonal processes in the IPL and in the cytoplasm of their cell bodies in the INL. In addition, small cell bodies in the GCL stained positive for PKCα, suggesting involvement of displaced amacrine cells (Figure 7C).
ISH of Davidson’s Fixed Paraffin-embedded Minipig Eye Samples
Using an mRNA-ISH method, we were able to detect VEGFA, ANGPT2, and TEK mRNA in well-preserved sections obtained from DF-fixed paraffin-embedded minipig eyes.
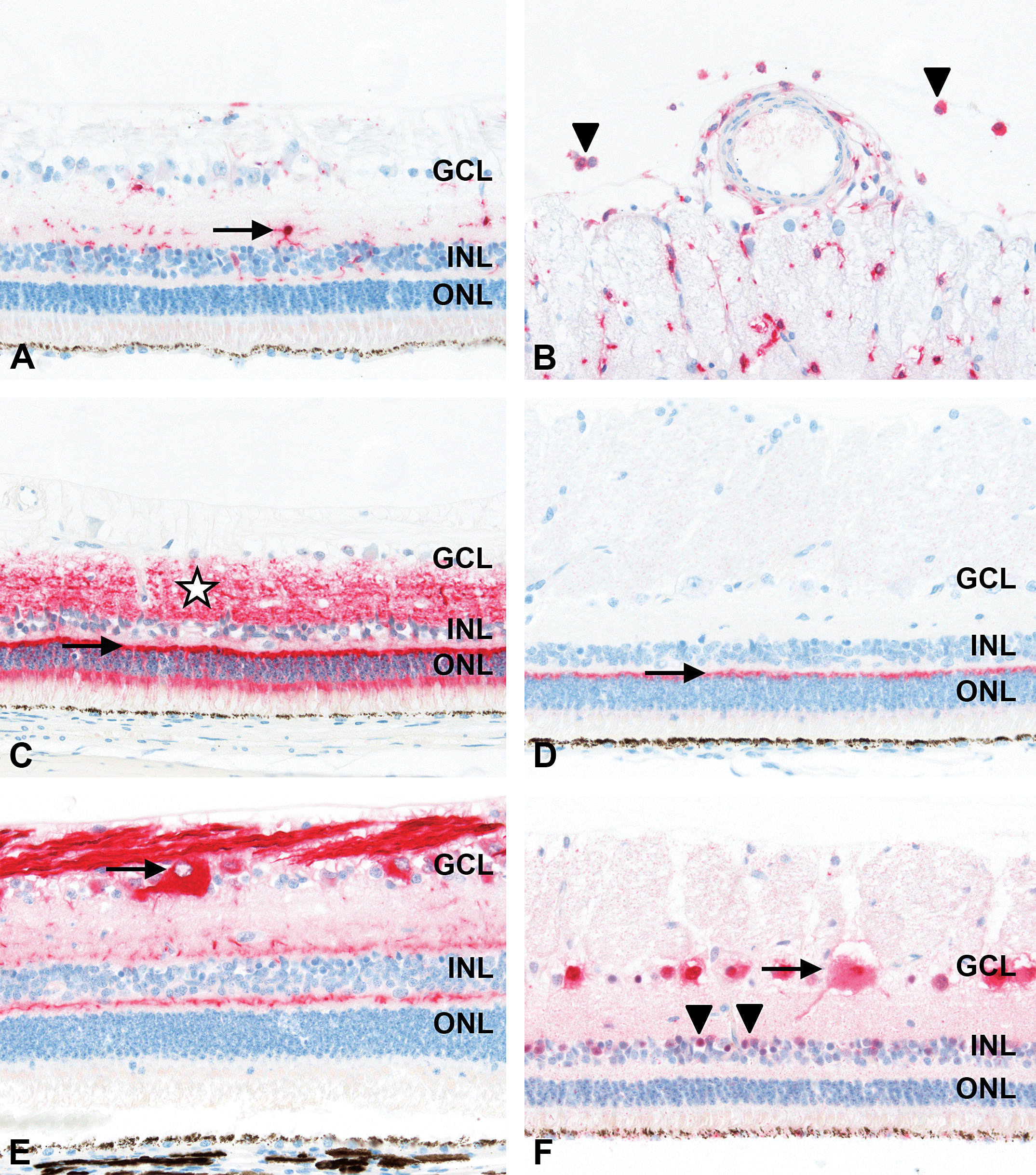
In the retina, VEGFA mRNA was found to localize predominantly in the cytoplasm of ganglion cells, in the INL (consistent with Müller cell bodies), and within RPE cells, which was obscured by the high melanin content in this structure. In addition, scarce positivity was present in the ONL (Figure 8A). VEGFA transcripts were also detected in axons of the optic nerve, in the lens epithelium including the equatorial region, and minimally in the conjunctival epithelium, corneal epithelium, stroma, and endothelium.
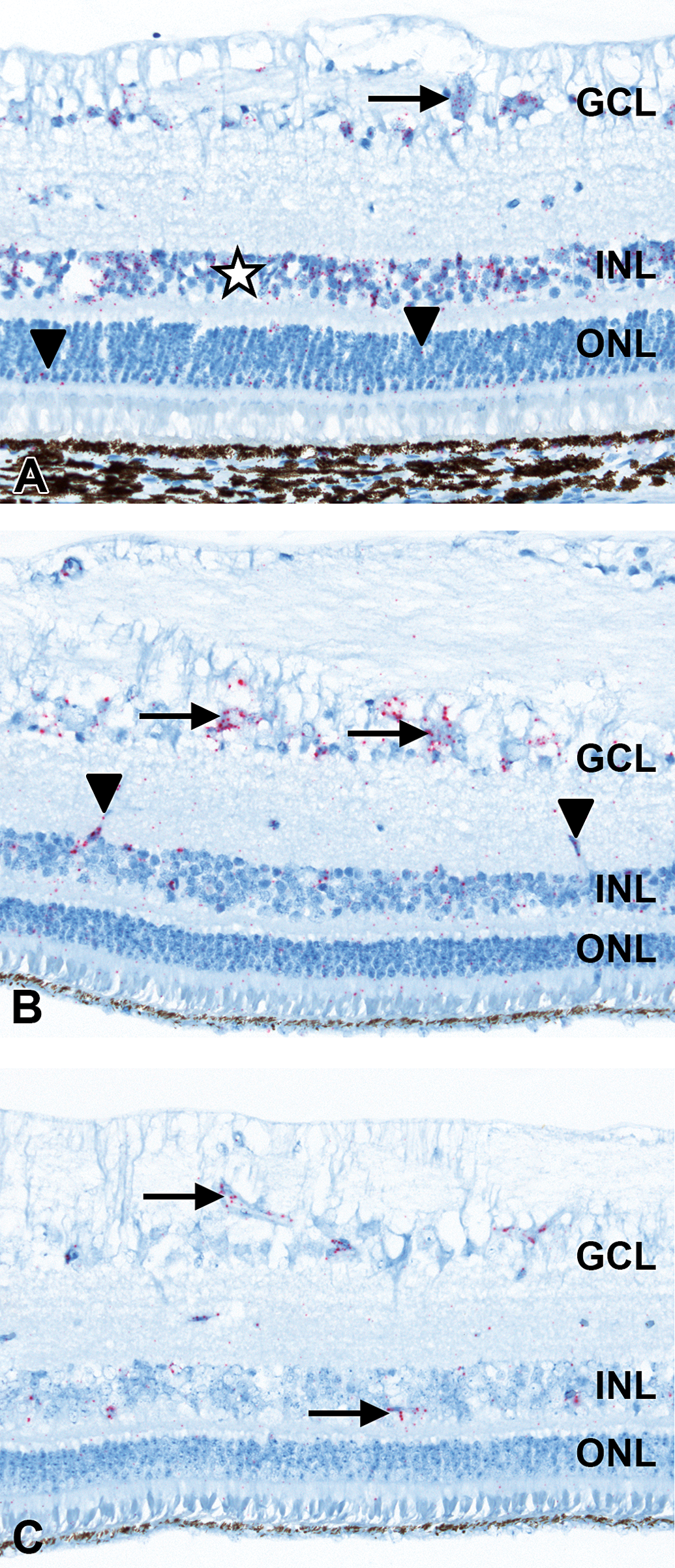
In situ hybridization of minipig neuroretina. (A) Labeling for vascular endothelial growth factor A messenger ribonucleic acid (mRNA) in ganglion cells (arrow) and inner nuclear layer (INL; asterisk). In addition, scarce positivity in outer nuclear layer (ONL; arrowheads). Labeling in RPE cells obscured by melanin content. (B) Labeling for ANGPT2 mRNA in ganglion cells (arrows), endothelium of retinal blood vessels (arrowheads), INL, ONL, and photoreceptor layer. (C) Labeling for tyrosine kinase, endothelial mRNA in endothelial cells of retinal capillaries and blood vessels (arrows). For orientation, ganglion cell layer (GCL), INL, and ONL. 40× original objective.
ANGPT2 mRNA was found to localize predominantly in the cytoplasm of retinal ganglion cells, endothelium of retinal blood vessels, in the INL, and less in the ONL and PRL (Figure 8B). In addition, ANGPT2 mRNA was present within endothelial cells of capillaries and larger blood vessels in all other ocular compartments.
TEK mRNA was detected by ISH in endothelial cells of retinal capillaries and blood vessels (Figure 8C) and of all other ocular compartments, except for the lens and cornea.
Discussion
As reported previously for pigs (Gekeler et al. 2007; McLellan and Rasmussen 2012; Rosolen et al. 2012), commercially available OCT instruments routinely used for humans could be well applied to anesthetized minipigs in our study. Due to the similarity in size of the minipig and human eye, no technical modifications were necessary when using SDOCT or SSOCT imaging techniques. With both techniques, there was good definition of retinal layers to allow for qualitative assessment. However, as described by others (McLellan and Rasmussen 2012; Rosolen et al. 2012), scans must be carefully examined for errors before tabulating quantitative data. As expected for conventional SDOCT and SSOCT imaging, visualization of the choroidal microvasculature in the minipig eye was obscured by speckle noise (Cheng et al. 2014). Here, we demonstrated for the first time in minipigs, speckle noise removal which resulted in a gain of structure contour and better demarcation between tissue and fluid-filled cavities, that is, blood vessels. To the best of our knowledge, this is also the first report of dye-free angiography of retinal and choroidal blood vessels in the minipig eye, using AngioOCT and speckle noise–free blood vessel extraction, respectively. It was highly encouraging to demonstrate the ability of the OCT systems to distinguish the different vascular levels in the posterior ocular segment of minipigs. First, as in humans (Jia, Bailey, et al. 2014; Jia et al. 2015), AngioOCT enabled 3D imaging of the retinal capillary network around the optic nerve head and of the deep microvascular network in minipigs. Image resolution appeared good enough to allow for measurements including an option for quantification of blood vessel volumes. Second, speckle noise–removal enabled extraction of choroidal blood vessels via postprocessing threshold filtering. Interestingly, individual choroidal vascular loops could not be clearly separated from each other. It is therefore tempting to speculate that minipigs might have a single choroidal vascular efferent system. As noninvasive imaging alternatives, speckle noise–free blood vessel extraction and AngioOCT of retinal and choroidal blood vessels may be well applied in preclinical safety studies in minipigs and in the clinical setting. It could provide valuable information on blood vessel growth, detect neovascularization or choroidal lesions, and allow for longitudinal assessment of ocular vascular diseases. In addition, better in vivo imaging of ocular structures in general might further improve correlation with histopathology (Schafer and Render 2013).
However, one drawback of the present OCT imaging in minipigs was that the described blood vessel extraction method is highly dependent on image quality and proper tissue penetration. Due to technical limitations, the region of interest was restricted to a relatively small area in the minipig eye. At this time, a defined and generally accepted level for a threshold for extraction of blood vessels is not available for the minipig. This will have to be implemented and validated in subsequent studies.
Another goal of our work was to establish a panel of tissue-based IHC and ISH markers on routinely DF-fixed paraffin-embedded minipig eyes that could be readily applied in toxicology studies. This knowledge does not currently exist in the public domain. Eyes are usually fixed intact to preserve tissue integrity. However, especially in ocular toxicology studies, it might be necessary to perform a number of postmortem investigations that may require different tissue preservation methods on a limited number of eyes. Therefore, our aim was to produce H&E stained ocular sections of good quality for standard histopathology and, at the same time, to perform IHC or ISH on sections obtained from the same specimen. We chose commercially available antibodies according to the published literature or information in the manufacturer’s datasheet. Selection criteria were proven applicability of the markers to either porcine ocular or extraocular tissue, irrespective of the fixation method, or to DF-fixed ocular tissue from rats. To the best of our knowledge, we demonstrate for the first time a panel of tissue-based IHC or ISH markers for neuronal and glial cells, photoreceptors, synapses, RPE, macrophages, and vasculature and cell proliferation in DF-fixed paraffin-embedded eyes in minipigs while preserving good morphology. The following IHC markers were successfully established on DF-fixed porcine eyes and revealed the specific staining of cells or structures they were described to identify according to the published data: NF160 for ganglion cells and their axonal processes, horizontal, bipolar, and amacrine cells (Guduric-Fuchs et al. 2009); NeuN for ganglion and amacrine cells (Johansson, Eftekhari, and Warfvinge 2010); calretinin for amacrine, horizontal, and ganglion cells (Guduric-Fuchs et al. 2009); calbindin for horizontal and bipolar cells (Guduric-Fuchs et al. 2009); PKCα for synaptic connections of bipolar cells (Guduric-Fuchs et al. 2009); GFAP for glial cells, especially Müller cells and astrocytes (Fernandez-Bueno et al. 2013); glutamine synthetase for glial cells, especially Müller cells and astrocytes (Guduric-Fuchs et al. 2009; McKay et al. 2009); vimentin for type III intermediate filament expressed in glial cells (Umazume et al. 2012); Iba1 for microglial cells and macrophages (Umazume et al. 2012); rhodopsin for photoreceptors (Guduric-Fuchs et al. 2009; McKay et al. 2009); synaptophysin for synapses (McKay et al. 2009); PSD95 for synapses, mainly in the OPL (Reddy et al. 2009); RPE65 for RPE (Fernandez-Bueno et al. 2013); vWF for blood vessel endothelia (Rand et al. 1987); αSMA for smooth muscle cells and myofibroblasts in blood vessels (Umazume et al. 2012); desmin for type III intermediate filaments expressed in muscle cells (Labelle et al. 2012); and Ki-67 for cell proliferation (Sawhney and Hall 1992).
Although our staining results to a large extent reflected the published data, there were a few differences. Consistent with the literature (Johansson, Eftekhari, and Warfvinge 2010), we observed strong NeuN expression in the nuclei of retinal ganglion cells, weaker expression in amacrine cells, and no immunoreactivity in glial cells. However, we identified a weak NeuN staining in the cytoplasm of RPE cells which we considered as most likely unspecific.
Consistent with observations in paraformaldehyde-fixed cryosectioned pig eyes (Guduric-Fuchs et al. 2009), we observed rhodopsin immunolabeling on DF-fixed minipig eyes that was in line with the observation that rhodopsin as visual pigment is located in rod outer segment discs. In contrast, although also using DF, specific strong staining of both inner and outer photoreceptor segments and of the ONL was reported for rat eyes (McKay et al. 2009). The authors mentioned that differing staining results for rhodopsin might be related to a varying distribution pattern in the retina, depending on the clone of antibody used.
It was extremely challenging to identify a reliable IHC marker for macrophages in pigs, in general. Unfortunately, only a few antibodies reactive with porcine lymphocytes, monocytes, and macrophages are available (Hwang et al. 2015). Antihuman lysozyme was reported to have positive results in paraformaldehyde-fixed paraffin-embedded pig eyes with surgically induced CNV (Lassota et al. 2008). We tested a number of antibodies mentioned in the literature for pigs and other species, including lysozyme, on formalin- and DF-fixed minipig eyes and on formalin-fixed minipig colon. However, except for anti-Iba1, none of these markers could be established successfully (unpublished data). Macrophages are not readily present in normal eyes. Therefore, we used a paraffin block from a DF-fixed pig eye diagnosed with vitreal and retinal perivascular macrophage infiltration. As microglial cells are part of the mononuclear phagocyte system in the retina and Iba1 is expressed by macrophages and microglia (Umazume et al. 2012), anti-Iba1 revealed a specific strong staining for vitreal macrophages, in addition to resting and activated microglia, in DF-fixed eyes from minipigs in our study. However, as anti-Iba1 equally stains macrophages and microglial cells, this marker appears to be of limited diagnostic utility in distinguishing these 2 cell types in the inflamed retina. In light of the increasing need for novel ocular therapies targeting VEGF or other VEGF-related modulators of angiogenesis (Penn et al. 2008), and the possible use of minipigs as animal model for CNV (Kiilgaard et al. 2005; Lassota et al. 2008), there is a demand for assessment of VEGF-target knockdown in ocular efficacy or toxicology studies. We were not aware of established ISH methods for the detection of these targets in DF-fixed porcine eyes, thus we established selected mRNA-ISH methods to detect VEGFA, ANGPT2, and TEK mRNA in DF-fixed paraffin-embedded minipig eyes of the present study. Location of these transcripts in the minipig retina was largely consistent with the literature, for VEGFA in ganglion cells, Müller cell bodies within the INL, and within RPE cells, which was partly obscured by melanin; for ANGPT2 mainly in ganglion cells, blood vessel endothelium, and INL; for TEK in blood vessel endothelium, which was consistent with the respective specific expressions stated in the literature (Smith 2007).
One limitation of the minipig eye for the applicability of IHC and ISH methods is its pigmentation. Melanin in the RPE hampers qualitative and quantitative assessment, especially when using 3,3′-diaminobenzidine (DAB) as a chromogen, in the RPE and choroidea, for example, for anti-RPE65 IHC or VEGFA mRNA, respectively. We therefore consistently applied red or purple chromogens and refrained from using DAB. In addition, we have started to adapt our protocols to include a modified melanin bleaching technique (Kivelä 1995) prior to IHC, which was already successfully established for anti-RPE65 on DF-fixed minipig eyes. The power of this bleaching method for application prior to ISH is pending further evaluation. Generally, DF provided good preservation of minipig eyes for a panel of IHC and ISH stainings and for detailed histopathologic examination. Still, tissue sections displayed some artifactural retinal detachment as described for DF-fixed eyes of rabbits (Latendresse et al. 2002). These artifacts, however, might be prevented by postfixation of eyes in 10% neutral-buffered formalin or 70% ethanol, respectively.
In conclusion, we were able to demonstrate speckle noise–free OCT imaging and dye-free AngioOCT enabling improved visualization of retinal and choroidal vasculature. Our work on different tissue-based IHC or ISH markers contributes to the available knowledge on IHC and ISH markers applicable for use in DF-fixed ocular tissue from minipigs in the research community. This is of high interest for application in systemic and ocular toxicology studies and investigative porcine animal models in ocular research. With our investigations, we have set the scene for a longitudinal assessment and monitoring of choroidal and retinal lesions via OCT as well as a proper evaluation of histopathologic lesions in the minipig. The knowledge gained from a combination of OCT imaging, standard histopathology, and IHC/ISH investigations will help to refine the minipig as a translational model for choroidal and retinal findings in humans.
Footnotes
Acknowledgments
The authors would like to thank Christelle Zundel, Rachel Neff, Virginie Ott, Petra Staeuble, and Gabrielle Branellec for their excellent technical support for IHC and ISH and Michael Siegrist and Martin Freiermuth for skilled animal handling during OCT imaging.
Author Contribution
Authors contributed to conception or design (EA, PM, BA, JF, TS, and BJ); data acquisition, analysis, or interpretation (EA, PM, BA, JF, CG, PH, MS, and BJ); and drafting the manuscript (EA, PM, and BJ). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
