Abstract
Glucagon-like peptide-1 (GLP-1) receptor agonist therapy has been implicated as a possible risk factor for acute pancreatitis in patients with type 2 diabetes. Dulaglutide is a long-acting GLP-1 receptor agonist in development for treatment of type 2 diabetes. The effects of dulaglutide were evaluated in male Zucker diabetic fatty (ZDF) rats to examine whether dulaglutide may induce or modulate pancreatitis. Rats were randomized to dose groups receiving twice-weekly subcutaneously administered dulaglutide 0.5, 1.5, and 5.0 mg/kg/dose (corresponding human plasma exposures following twice-weekly dosing are 3-, 8-, and 30-fold, respectively) for 13 weeks or to vehicle control. Following termination, serially trimmed sections of pancreases were stained with hematoxylin and eosin or co-stained with an epithelial marker and a marker of either proliferation or apoptosis. Efficacious reductions in glucose and hemoglobin A1c occurred at all dulaglutide doses. Lipase activity was unaffected, and there were modest increases in total and pancreatic amylase activities at all doses without individual microscopic inflammatory correlates. Microscopic dulaglutide-related pancreatic changes included increased interlobular ductal epithelium without ductal cell proliferation (≥0.5 mg/kg), increased acinar atrophy with/without inflammation (≥1.5 mg/kg), and increased incidence/severity of neutrophilic acinar pancreatic inflammation (5.0 mg/kg). In summary, dulaglutide treatment was associated with mild alterations in ductal epithelium and modest exacerbation of spontaneous lesions of the exocrine pancreas typically found in the ZDF rat model.
Keywords
Introduction
Glucagon-like peptide-1 (GLP-1) receptor agonists and dipeptidylpeptidase-4 (DPP-4) inhibitors are incretin-based therapies used to treat type 2 diabetes (Grigoropoulou et al. 2013). GLP-1 is a proglucagon-derived peptide that improves glucose homeostasis by enhancing glucose-dependent insulin secretion, inhibiting inappropriately high levels of postprandial glucagon secretion, delaying gastric emptying, and reducing food intake (Baggio and Drucker 2007; Holst, Vilsboll, and Deacon 2009). GLP-1 has a short half-life due to its rapid degradation by DPP-4 (Drucker 2006). Several drugs are currently commercially available that affect the GLP-1 pathway to control hyperglycemia and include the GLP-1 receptor agonists exenatide (AstraZeneca), liraglutide (Novo Nordisk), albiglutide (GlaxoSmithKline), and lixisenatide (Sanofi) and the DPP-4 inhibitors sitagliptin (Merck), vildagliptin (Novartis), alogliptin (Takeda), linagliptin (Boehringer Ingelheim and Eli Lilly), and saxagliptin (Bristol-Myers Squibb and AstraZeneca). Prior studies in humans suggest that GLP-1 receptor agonist–based therapies may be associated with acute pancreatitis (Denker and Dimarco 2006; Tripathy et al. 2008; Iyer et al. 2012; Cure, Pileggi, and Alejandro 2008; Singh et al. 2013; Ayoub et al. 2010; Ahmad and Swann 2008) and pancreatitis-related hospitalization (Singh et al. 2013). However, this has not been confirmed, as the number of clinical cases is low and study results have been inconsistent (Denker and Dimarco 2006; Macconell et al. 2012; Tripathy et al. 2008; Iyer et al. 2012; Dore et al. 2011; Dore, Seeger, and Chan 2009; Garg, Chen, and Pendergrass 2010; Cure, Pileggi, and Alejandro 2008; Singh et al. 2013; Ahmad and Swann 2008; Ayoub et al. 2010). Moreover, type 2 diabetes itself is a risk factor for developing pancreatitis (Noel et al. 2009; Girman et al. 2010; Butler et al. 2010).
Preclinical studies have given conflicting results regarding whether GLP-1 receptor agonist–related pancreatic changes are associated with pancreatitis (Koehler et al. 2009; Tatarkiewicz et al. 2010; Nyborg et al. 2012; Vrang et al. 2012; Tatarkiewicz et al. 2013; Gier et al. 2012; Nachnani et al. 2010; Matveyenko et al. 2009). Several preclinical studies in various rodent models for diabetes or pancreatitis have attributed ductal metaplasia, exocrine pancreas degeneration, and pancreatic duct abnormalities to administration of GLP-1 receptor agonist–based drugs (Gier et al. 2012; Vrang et al. 2012; Nachnani et al. 2010; Matveyenko et al. 2009). In one study, pancreatic acinar inflammation, pyknosis, and higher lipase activity were seen in Sprague-Dawley rats chronically administered exenatide (Nachnani et al. 2010). Another study reported exenatide-induced pancreatic duct cell expansion with mucinous metaplasia containing cells resembling low-grade pancreatic intraepithelial neoplasia (PanIN) in Sprague-Dawley rats (Gier et al. 2012). In contrast, exenatide administration to diabetic obese mice or rats given a high-fat diet modulated pancreatitis-associated gene expression but did not exacerbate chemically induced pancreatitis (Koehler et al. 2009; Tatarkiewicz et al. 2010). Liraglutide administered to mice, rats, and nonhuman primates did not induce macroscopic or microscopic pancreatitis when dosed up to 2 years and up to levels 60 times higher than in humans (Nyborg et al. 2012).
The U.S. Food and Drug Administration (FDA) has requested that sponsors of marketed and in development incretin-based drugs conduct 3-month pancreatic toxicity studies in a rodent model of diabetes (Egan et al. 2014). Three-month exposure to exenatide or liraglutide in Zucker diabetic fatty (ZDF) rats did not affect pancreatic exocrine structure or function, had no effect on pancreatic ductal cell proliferation (Vrang et al. 2012; Tatarkiewicz et al. 2013), and exenatide did not alter apoptosis in pancreatic ducts (Tatarkiewicz et al. 2013).
Dulaglutide (LY2189265) is a long-acting GLP-1 receptor agonist in development as a once-weekly subcutaneous injection for the treatment of type 2 diabetes. Compared to native GLP-1, dulaglutide is less susceptible to degradation by DPP-4 and the increased molecular weight slows absorption and minimizes renal clearance, resulting in a prolonged half-life in humans of approximately 5 days. Additionally, dulaglutide exhibits GLP-1-mediated effects, including potentiation of glucose-dependent insulin secretion, inhibition of glucagon secretion, delay of gastric emptying, and weight loss. This study investigated the potential effects of dulaglutide on pancreatic exocrine biomarkers, histology, and histomorphometry in male ZDF rats when administered as a twice-weekly subcutaneous injection for 13 weeks. The ZDF rat strain was selected as the experimental model since male rats, with managed diet, typically develop adult-onset type 2 diabetes and related complications by 12 weeks of age.
Materials and Methods
Animals
All studies were performed in accordance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare. Male ZDF rats (Charles River Laboratories, Kingston, NY) were housed individually in cages in a controlled environment (19°–25°C, humidity 30–70%, and 12 hr of light and dark). The care and use of the animals on the study were conducted in accordance with the guidelines of the U.S. National Research Council and the Canadian Council on Animal Care (CCAC). Any animal euthanized on the study (for humane reason or per study schedule) underwent exsanguination from the abdominal aorta following isoflurane anesthesia. Animals had access to water and were fed standard certified pelleted commercial laboratory diet (PMI Certified Rodent 5008 irradiated: PMI Nutrition International Inc., Richmond, IN).
Study Design
Eight-week-old rats were randomized using a computer-based system to dose groups receiving twice-weekly dulaglutide 0.5, 1.5, or 5.0 mg/kg/dose (corresponding human plasma exposures following twice-weekly dosing are 3-, 8-, and 30-fold, respectively; Food and Drug Administration 2014) or to vehicle control of a similar volume. Rats were subsequently subdivided into main study and toxicokinetic groups. The main study group was euthanized at the end of the treatment period and used for gross pathology, histopathology, immunohistochemistry, and clinical chemistry assessments. The toxicokinetic group had blood drawn throughout the study to evaluate toxicokinetic and clinical chemistry parameters. The main study group included 20 rats per dose group and 27 in the control group, and the toxicokinetic group consisted of 12 rats in each of the dosed groups and 16 in the control group. Additional animals were added to the control group to ensure adequate numbers of animals at study termination in the event of increased mortality due to complications (e.g., urinary tract or preputial yeast infections) of uncontrolled hyperglycemia.
Test rats in both the main and toxicokinetic groups were administered dulaglutide subcutaneously in 10 mM citrate buffer (pH 6.5), 4.6% mannitol (w/v), and 0.02% (w/v) polysorbate 80 in sterile water for injection. Control rats received the same solution without dulaglutide. All injections were given in a 0.5 ml/kg volume. Dosing commenced on consecutive days with approximately equal numbers of rats from each group being dosed on each day and continued twice weekly for 13 weeks for a total of 26 doses. The actual dose was based on the most recent body weight of each rat.
Body Weight, Food Consumption, and Clinical Chemistry Assessments
Individual body weights were measured at randomization and twice weekly, beginning the week prior to the start of dosing. Additionally, main study rats were weighed (fasted) before scheduled necropsy. Food consumption was measured twice weekly for each rat in the main study group starting the week prior to initiation of dosing. Blood (fasted) was collected for evaluation of clinical chemistry, hemoglobin A1c (HbA1c), and insulin levels from all toxicokinetic rats once pretreatment and on Days 29, 60, and 88 at 24 hr postdose and from all main study rats at termination. Clinical chemistry assessments included lipase, glucose, creatinine, and total and pancreatic amylase. Pancreatic amylase alone was analyzed from main study rats at Week 92 (termination). All measurements were performed on a Modular Analytics® biochemistry analyzer (Roche, Basel, Switzerland).
Toxicokinetic Evaluation
Blood samples were collected from a subset of toxicokinetic rats (3/group) via the jugular vein on Days 1, 43, and 88 to assess dulaglutide exposure. On Days 1 and 88, blood was drawn predose and at 4, 12, 24, 48, and 96 hr postdose and on Day 43 predose and 24 hr postdose. Blood samples from the main study rats were collected at necropsy.
Plasma samples were analyzed for dulaglutide content using a validated enzyme-linked immunosorbent assay (ELISA) method. Toxicokinetic parameters were calculated for peak concentration (Cmax), time to Cmax (Tmax), and area under the mean concentration time curve (AUC). The standard error (SE) of the AUC by dose group, day (as appropriate), and arithmetic means for the plasma concentration data was generated using WinNonlin version a5.2.1 (Pharsight, St. Louis, MO).
Gross Pathology and Histopathology
On completion of the treatment period (Day 92), all main study rats were euthanized after an overnight fast, and a complete gross examination of the carcass was conducted. The pancreas from each rat was collected and trimmed in the following manner. The bile duct was located and transected from the liver leaving it attached to the pancreas. The mesentery and greater omentum containing the pancreas were dissected from the spleen, stomach, duodenum, and colon, leaving a small, approximately 1-cm section of duodenum open and attached to the pancreas. For fixation, this dissected tissue was then attached to plastic, which was labeled to maintain the orientation of the pancreas edges with respect to the duodenum and spleen.
For trimming and evaluation, the portions of the pancreas were identified as follows. The head of the pancreas was contained within the mesoduodenum of the duodenual loop and the mesocolon portions of the dorsal mesentery. The body was contained within the mesoduodenum, mesocolon, and greater omentum. The structure was composed of the wedge-shaped portion extending from the narrow base adjacent to the transverse colon to the greater curvature of the stomach proximally and distally to include the duodenal papilla. The body contained the common duct, which was the primary focus of this study. The tail region containing the splenic duct was contained primarily within the gastrosplenic ligament of the greater omentum, extending from the hilum of the spleen to the region of the dorsal mesentery (Eustis, Boorman, and Hayashi 1990; Richards et al. 1964; Evans and Christensen 1979).
After collection, the pancreas was weighed, fixed in 10% neutral-buffered formalin, trimmed in cross section in its entirety, embedded in paraffin, and cross sectioned. Two sets of step sections (each set approximately 100 µm apart; ≥6 sections/rat) were collected to ensure inclusion of the main pancreatic interlobular ducts, which included the common pancreatic duct and associated pancreatic duct glands. One slide was stained with hematoxylin and eosin (H&E) and the other used for immunohistochemistry analysis (see below). Lesions were evaluated using standard severity grade levels of 1 to 5 (1 = minimal; 2 = slight; 3 = moderate; 4 = marked; and 5 = severe) and entered into a data collection system (PathData version 6.2b5 Pathology Data Systems Ltd., Rotkreuz, Switzerland; Shackelford et al. 2002).
Immunohistochemistry
Pancreatic sections were stained with antibodies for the epithelial marker cytokeratin-19 (from LifeSpan BioSciences, Seattle, WA) and co-stained using antibodies against Ki-67 (clone SP6; Thermo Scientific, Fremont, CA) to assess cell proliferation or co-stained with the terminal deoxynucleotidyl transferase (TdT) dUTP nick-end labeling (TUNEL) assay (Apoptag Peroxidase Apoptosis detection kit; EMD Millipore, Billerica, MA) to evaluate apoptosis. For morphometric analysis of TUNEL and cytokeratin staining, slides were chosen, which contained the common pancreatic duct as determined by histologic evaluation of H&E and cytokeratin-19/Ki-67 stained slides. A minimum of 1,000 cytokeratin-19-positive duct cells per animal were counted, and the total number of duct cells positive for cytokeratin-19 that were either Ki-67 positive or TUNEL positive was quantitated, and the percentages of proliferation and apoptosis, respectively, were calculated.
Statistical Analysis
For each pharmacokinetic, biochemical, survival, clinical observation, and body weight parameter of interest, group variances were compared using Levene’s test at the .05 significance level. When the differences between group variances were not found to be significant, a parametric one-way analysis of variance (ANOVA) was performed. When significant differences among the means were indicated by the ANOVA (p ≤ .05), then Dunnett’s “t” test was used to perform the group mean comparisons between the reference group and each dose group. Whenever Levene’s test indicated heterogeneous group variances (p ≤ .05), the nonparametric Kruskal–Wallis test was used to compare all considered groups. When the Kruskal–Wallis test was significant (p ≤ .05), then the significance of the differences between the reference group and each test group was assessed using Dunn’s test. Findings are significant with a p ≤ .05.
Results
Dulaglutide Pharmacokinetics
Tmax was observed 12 hr postdose on Day 1 and 24 hr postdose on Day 88 for all 3 dosing regimens (Table 1). Systemic exposure to dulaglutide, as defined by Cmax and AUC0–96hr, increased with increasing dose levels on Days 1 and 88, and the dose was proportional between 0.5 and 5.0 mg/kg dulaglutide doses (Table 1). No accumulation was observed following repeated dulaglutide administration for 13 weeks. Dulaglutide exposure was generally comparable between main study animals at necropsy and toxicokinetic animals at the corresponding time point (data not shown).
Dulaglutide exposure parameters.
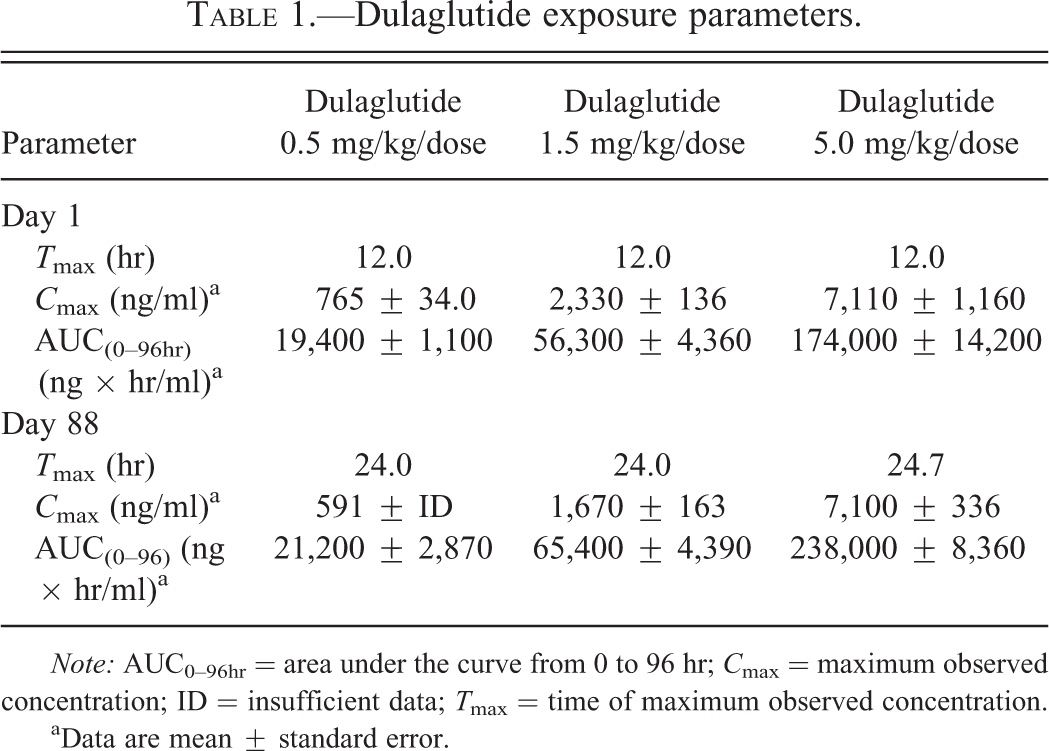
Note: AUC0–96hr = area under the curve from 0 to 96 hr; Cmax = maximum observed concentration; ID = insufficient data; Tmax = time of maximum observed concentration.
aData are mean ± standard error.
Survival, Clinical Observations, and Body Weight
Two rats were found dead during the study: a control rat on Day 49 and a rat from the dulaglutide 5.0 mg/kg group on Day 75. Both rats had gross renal pelvis dilation, and the dulaglutide 5.0-mg/kg animal had enlarged kidneys. It is likely that renal changes contributed to these animals’ mortality since ZDF rats often develop extensive renal lesions (Vora et al. 1996).
No dulaglutide-related clinical signs were observed during the study. Body weight and body weight gain in control rats were consistent with the ZDF diabetic disease model and consisted of increases in body weight gain at 12 weeks of age (4 weeks following dosing in this study), which remained stable for the remainder of the study (Table 2; Shiota and Printz 2012). A dose-related decrease in body weight in dulaglutide-administered rats was noted at Week 4, and a dose-related increase in body weight was observed from Week 4 to Week 12 compared with controls. Control rats increased their food consumption throughout the study while dulaglutide-treated rats decreased consumption, with rats given 0.5, 1.5, and 5.0 mg/kg/dose consuming 5%, 14%, and 12% less food, respectively, compared with controls.
Mean summary of body weight up to 12 weeks.
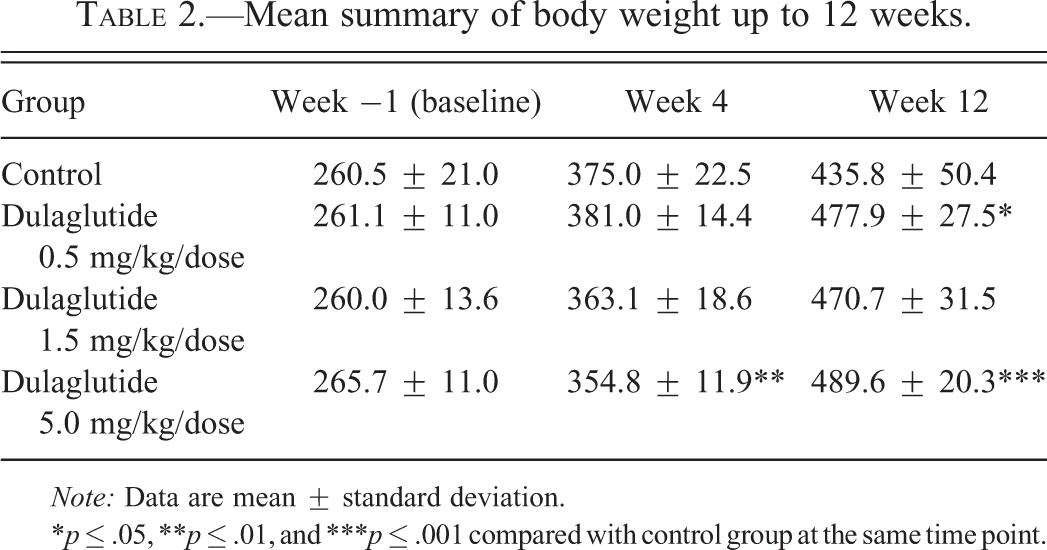
Note: Data are mean ± standard deviation.
*p ≤ .05, **p ≤ .01, and ***p ≤ .001 compared with control group at the same time point.
Clinical Biochemistry
Mean values of HbA1c, glucose, and lipase (Figure 1A, B, and E) increased in the control toxicokinetic group between pretreatment and at Days 30, 61, and 89. Since ZDF rats can suffer from ascending urinary tract infections, which may affect renal function and may then affect serum amylase levels, we also measured creatinine values, which were 0.26, 0.42, 0.46, and 0.32 mg/dL on Days −10, 30, 61, and 89, respectively, in the control group. Insulin also increased by 85% in toxicokinetic control rats between pretreatment and Day 30 and remained stable thereafter (Figure 1C).
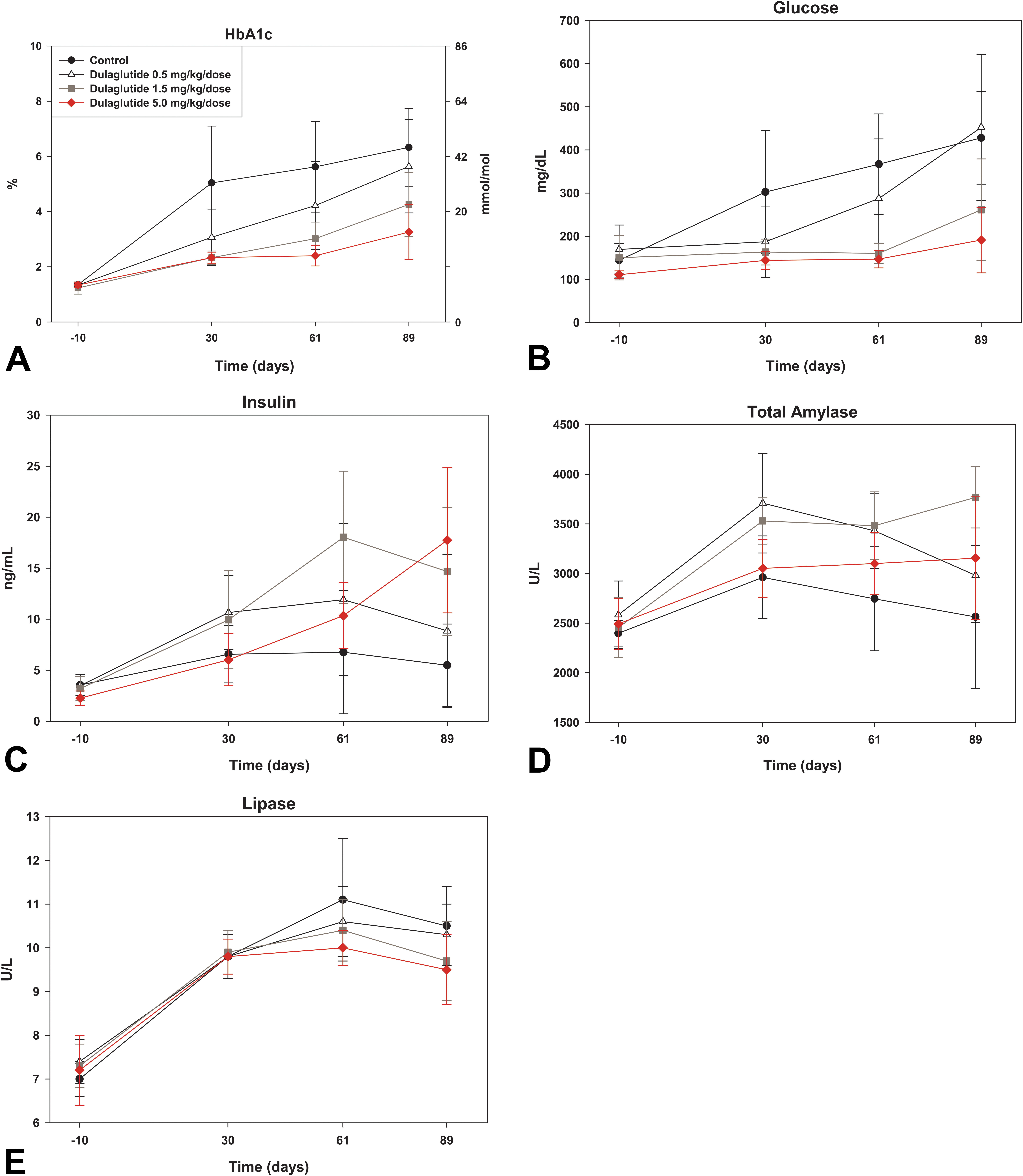
Summary of (A) HbA1c, (B) glucose, (C) insulin levels, (D) total amylase, and (E) lipase activities in toxicokinetic rats administered (black circles) no dulaglutide (control) or administered (black triangles) dulaglutide 0.5 mg/kg/dose, (gray squares) dulaglutide 1.5 mg/kg/dose, or (red diamonds) dulaglutide 5.0 mg/kg/dose. HbA1c = hemoglobin A1c.
Dulaglutide administration resulted in dose-related decreases in plasma levels of HbA1c and glucose compared with the controls (Figure 1A and B). Dose-related decreases in glucose concentrations (−38% to −60% of control group mean) correlated with decreases in HbA1c (−33% to −57% of control group mean) in all groups. Mild to moderate increases in insulin concentration (51–223% of control group mean) were observed with the 3 dulaglutide doses by Day 89 (Figure 1C). A similar pattern of changes in glucose, HbA1c, and insulin was observed in rats in the main study group at study termination (Day 92; data not shown).
All 3 dose levels of dulaglutide increased total (Figure 1D) and pancreatic amylase activities (Figure 1D). Total and pancreatic amylase values were taken at Day 92 (termination) in the dosed animals only, with mean group values of 2,266, 2,542, 2,757, and 3,006 U/L for control, 0.5, 1.5, and 5 mg/kg, respectively. Creatinine blood levels (data not shown) by Day 89 in dosed rats were compared with control rats. Increases in total amylase activity (13–47% of control group mean) were not dose related. There were no concurrent changes in dosed rats compared with control rats in lipase activity over this time period (Figure 1E). Similarly, in main study rats, increases in amylase and pancreatic amylase, but not lipase, were observed in groups administered dulaglutide at 1.5 and 5.0 mg/kg (data not shown).
Morphology and Histomorphology
There were no pancreatic macroscopic findings or weight changes related to dulaglutide administration. Generally, microscopic changes were not pronounced and were present in both control and dosed rats. All 3 doses had a non-dose-dependent increase in the incidence and a slight increase in severity of increased ductal epithelium in the main pancreatic interlobular ducts (Table 3). The term “increased ductal epithelium” was selected to describe a generalized increase in the overall ductal epithelium in the absence of cytologic changes (e.g., cellular atypia and increased mitoses). This observation was assigned semi-quantitative severity grades as follows. In minimally affected animals (n = 13), including 1 control rat, the increased ductal epithelium formed low tufts of cuboidal epithelium lining the main duct lumen. In rats with a slight severity score (n = 11), the lining of epithelium was often folded, and in the animals judged to be moderately affected (n = 2; one each from the dulaglutide 1.5 and 5.0 mg/kg dose groups), papillae extended into the pancreatic duct lumen (Figure 2B). There were differences in angles during sectioning that occasionally produced the appearance of lack of epithelial contact with the basement membrane, but the cells examined in the ductular cross sections in this study were normal in appearance, cuboidal to columnar, without loss of orientation to the basement membrane or piling. The region of the pancreas affected for the majority of rats was in the body and, in a smaller number, in the head. Pancreatic duct glands in the areas of the affected duct lining epithelium were normal in quantity and morphology (Figure 2B).
Summary of pancreas histopathological findings.
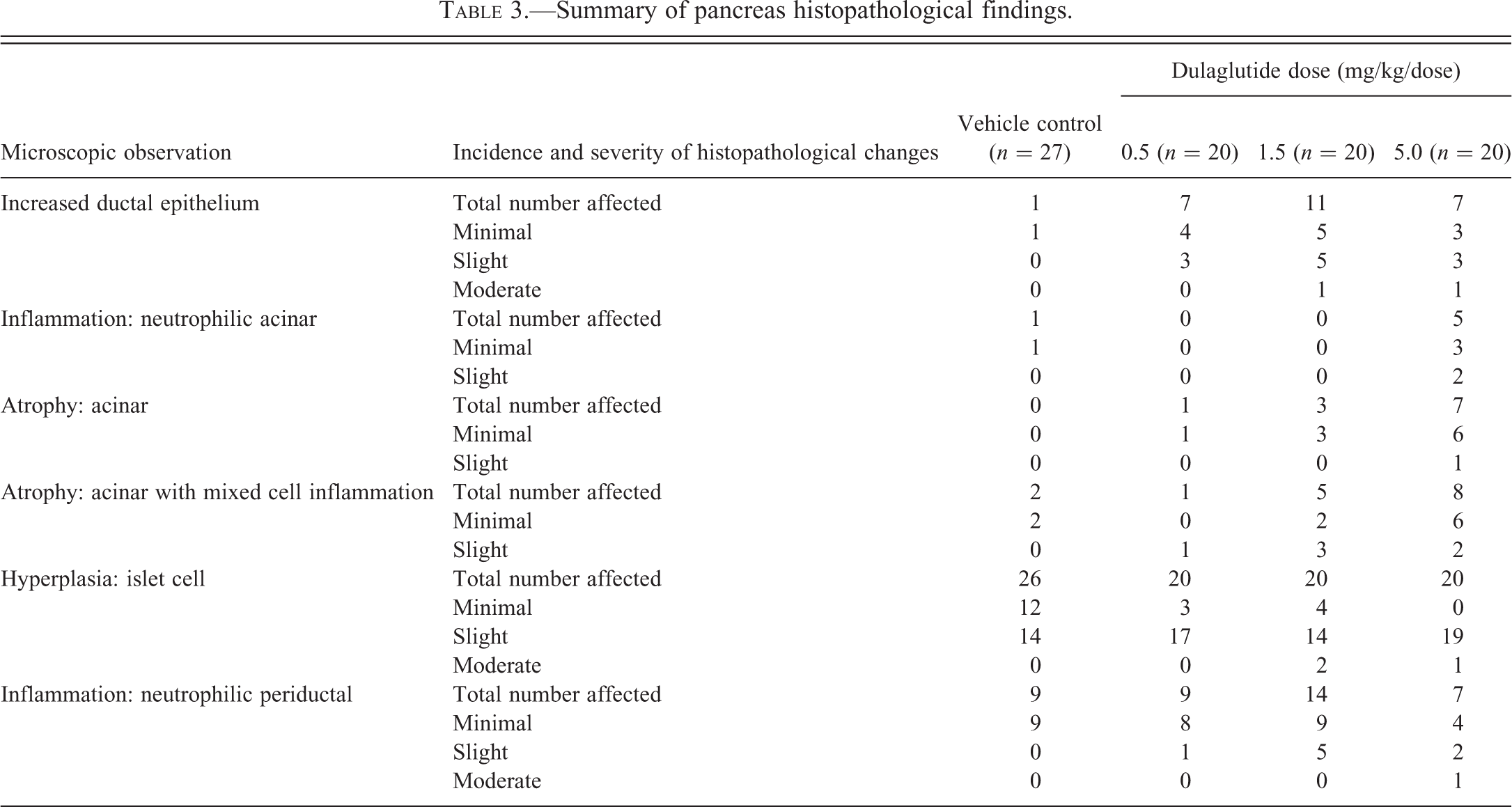
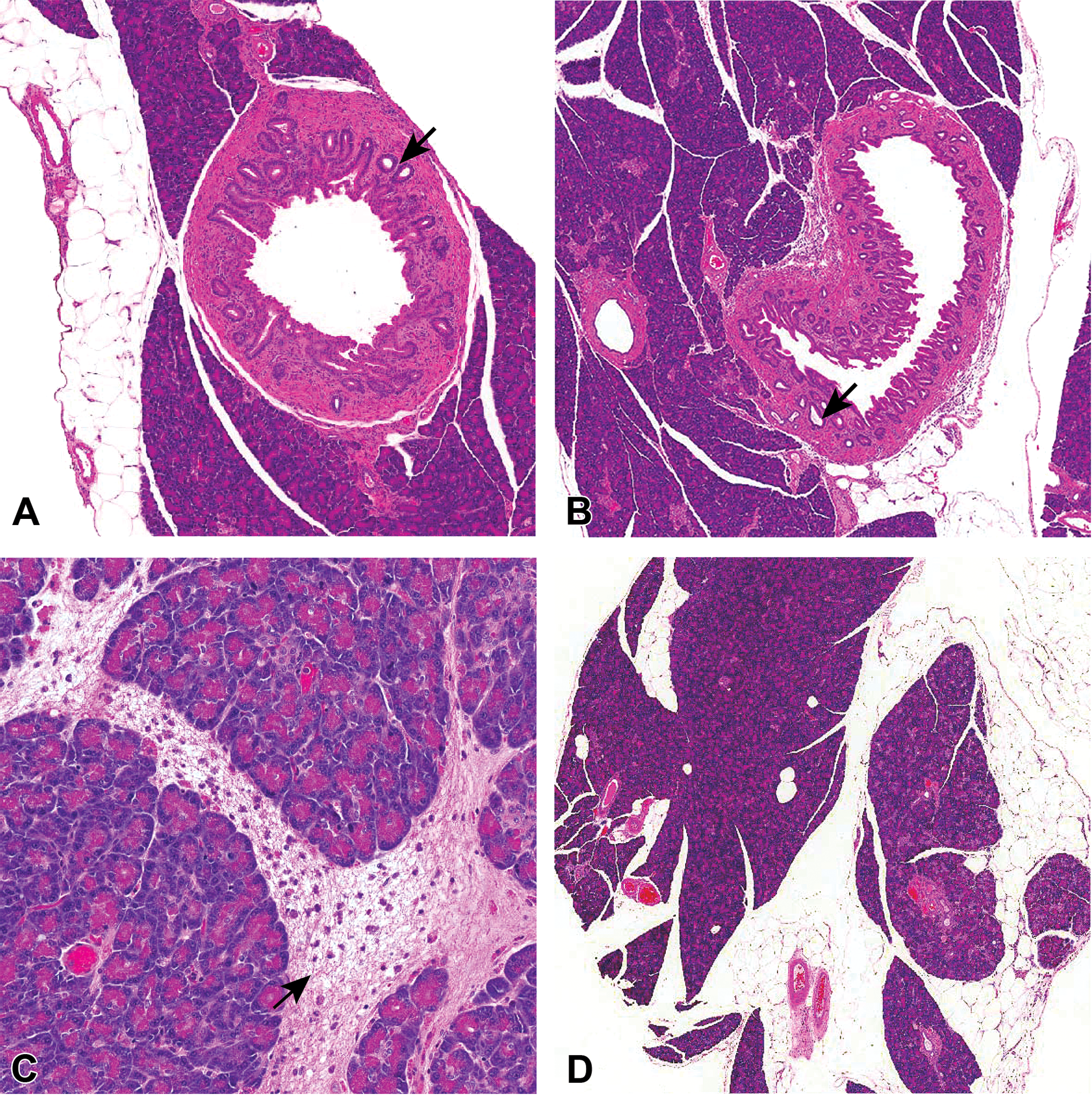
Pancreatic histomorphology from control and dulaglutide-treated rats. (A) Control rats with normal duct morphology with minimal increased duct epithelium (magnification 4×). Arrow indicates pancreatic duct glands. (B) Increased luminal epithelium, slight, is apparent in the pancreas of dulaglutide (1.5 mg/kg)-treated rats, with papillary projections of the epithelial cells lining the duct protruding into the lumen (magnification 4×). The pancreatic duct glands are normal in both control and dulaglutide (1.5 mg/kg)-treated rats. Small black arrow indicates the lumen of the duct gland. (C) Neutrophilic acinar inflammation, minimal locally extensive, characterized by increased space between lobules (edema) containing the inflammatory cells. Inflammatory cells also extend onto the serosal surface and into the interstitium (magnification 10×). Large white arrow indicates edema with fibrin and neutrophilic infiltrates. (D) The incidence and degree of atrophy, minimal, of the exocrine pancreas were similar between control and dulaglutide-treated rats. Lobe on the right shows atrophy of the exocrine pancreas. There are decreased acini, increased ductules, and more prominent interstitium. Lobe on the left has normal exocrine tissue (magnification 2×).
In the dulaglutide 5.0 mg/kg/dose group, there was an increased incidence and severity (minimal to slight) of neutrophilic inflammation in the acinar tissue compared with the control group (5 animals vs. 1 animal, respectively; Table 3), which, in some rats, involved the serosa and extended into the immediately adjacent mesentery (Figure 2C). No rats administered dulaglutide at 0.5 or 1.5 mg/kg showed neutrophilic inflammation.
There was an increased incidence and severity of atrophy of the acinar epithelium in all dulaglutide dose groups compared with the control group. In both control and dulaglutide-treated rats, the atrophy was accompanied by mixed cell inflammation (consisting of mononuclear cells and neutrophils) that was distinct from the neutrophilic inflammation of the acinar tissue. Lobular atrophy was usually focal in distribution and more frequently found in the pancreatic tail region. The incidence of islet cell hyperplasia and neutrophilic periductal inflammation was comparable between control and dosed groups, and these morphologic findings are common in the ZDF model (Chadwick et al. 2013). It is noted that the rats administered 1.5 and 5.0 mg/kg of dulaglutide appeared to have a slightly greater severity score for both islet cell hyperplasia and neutrophilic periductal inflammation.
Histomorphometry
The incidence of epithelial cell proliferation and apoptosis in the duct epithelium, as assessed by Ki-67 and TUNEL labeling, respectively, was low and similar between the control and dulaglutide-treated rats. The range of Ki-67-positive cells was 0–1.26% in controls and 0–4.24% in rats administered dulaglutide 5.0 mg/kg. In rats with increased duct epithelium (see above), there was no consistency between the area with increased duct epithelium and an increased level of Ki-67 staining. The range of TUNEL/cytokeratin-19-positive cells was 0–0.77% in controls and 0–0.50% in rats administered dulaglutide 5.0 mg/kg.
Discussion
Scientific literature and the media have suggested a potential association between the use of GLP-1 receptor agonists and pancreatitis (Denker and Dimarco 2006; Tripathy et al. 2008; Iyer et al. 2012; Cure, Pileggi, and Alejandro 2008; Singh et al. 2013; Ayoub et al. 2010; Ahmad and Swann 2008); however, extensive reviews of both preclinical animal and clinical human data by the FDA and European Medicines Agency (EMA) find these assertions to be inconsistent with their reviewed data (Egan et al. 2014).
This current study investigated the effects of dulaglutide on pancreatic enzymes and exocrine morphology in an animal model of type 2 diabetes. The study used doses that provided systemic exposures that were 3-, 8-, and 30-times higher relative to the maximum human AUC. Dulaglutide resulted in efficacious reductions in the plasma levels of glucose and HbA1c, which reflect dulaglutide pharmacological activity. Lipase activity was unaffected, and there were mild to modest increases in total and pancreatic amylase activity at all doses without individual microscopic inflammatory correlates. There were no pancreatic weight changes or macroscopic pancreatic findings related to dulaglutide administration. Microscopic dulaglutide-related changes for all 3 dulaglutide doses included increased incidence or severity of interlobular ductal epithelium without alterations in ductal cell proliferation or apoptosis. Dulaglutide doses ≥1.5 mg/kg increased acinar atrophy with or without inflammation, and the 5.0 mg/kg dose of dulaglutide increased the incidence and severity of neutrophilic acinar pancreatic inflammation. However, this inflammation was present in only 25% of the highest dosed rats, was often focal with minimal to slight severity scores, and was not associated with concurrent dulaglutide-associated changes in the plasma levels of circulating enzymatic biomarkers. A relationship between acinar atrophy and mixed cell inflammation with neutrophilic acinar inflammation was not apparent. Islet cell hyperplasia is typically found in ZDF rats, with adjacent exocrine tissue having associated inflammatory changes and atrophy (Li et al. 2006). Hence, the inflammation and atrophy in some acinar foci could be the result of proximity to hyperplastic islet cells and not a direct effect of dulaglutide on acinar tissue. This is consistent with the lack of a strong dose–response relationship between dulaglutide and inflammation.
The increased ductal epithelium occurred in both controls and dulaglutide-treated rats, although these changes were more common and of greater severity with dulaglutide administration. The increase in ductal epithelium was not dose responsive and was not associated with significant changes in cell proliferation or apoptosis. This is in agreement with other studies that found no induction of cell duct proliferation in the pancreas of ZDF rats administered liraglutide or exenatide (Vrang et al. 2012; Tatarkiewicz et al. 2013), and with the observation that there was no effect on apoptosis in the pancreases of exenatide-treated ZDF rats (Tatarkiewicz et al. 2013). In contrast, a study by Gier et al. (2012), which investigated the effect of 12-weeks of exenatide on pancreatic duct glands in Sprague-Dawley rats, found increased pancreatic duct glands with mucinous metaplasia and cytological atypia resembling low-grade pancreatic intraepithelial neoplasia. Gier et al. found most changes in the head of the pancreas, and we noted changes also in the body and tail. The difference in findings between our study and that of Gier et al. may reflect experimental design. Our study used the ZDF rat model of type 2 diabetes, while Gier et al. used normoglycemic Sprague-Dawley rats. The studies further differed in how the pancreatic histologic sections were generated and morphometric techniques performed. We chose to examine the main pancreatic duct and associated pancreatic duct glands via numerous serial cross sections rather than longitudinal sections as evaluated by Gier et al. (2012). Cross sections allowed examination of large numbers of sections cut at similar angles, minimizing variability resulting from trimming and sectioning, which influences the presentation of duct epithelium and the pancreatic duct glands. The preparation of histologic sections in the current study ensured a consistent, thorough, and efficient examination of the pancreas for a large number of rats in preclinical safety assessment, with evaluation of the main pancreatic interlobular ducts ensuring inclusion of the common pancreatic duct and associated glands.
The mechanism of the increases in ductal epithelium was not apparent, and determination if this was a primary, receptor-mediated effect of dulaglutide or secondary to alterations in nutritional or metabolic status was not possible. The observation that increases in ductal epithelium were not due to changes in cell proliferation or apoptosis may reflect functional change(s), such as decreased secretory content, rather than pathology. Dulaglutide, consistent with other GLP-1 receptor agonists, caused decreases in food consumption, and reductions in food consumption are known to influence secretory activity (Keller and Layer 2005; Smith, Sunter, and Case 1982). Alterations in nutritional status can cause structural alterations in the acinar and ductal system in the rat, monkey, and human, such as ductal hyperplasia with epithelial stratification (Sandhyamani, Vijayakumari, and Balaraman Nair 1999), increased intraluminal material (Geldof et al. 1992), and cystic dilation (Wesson, Sparaco, and Smith 2008). Alternatively, the ductal alterations in dulaglutide-treated rats could result from a transient effect on cell proliferation or apoptosis, which was not detected in our analysis (which assessed a single time point) or be due to altered cell differentiation. The scope of the current study design did not allow for a definitive assessment of the mechanism of this effect, and further studies would be needed to both characterize the morphologic changes over time and further evaluate the mechanism of significance of the ductal alterations. Lipase activity is considered more diagnostic of pancreatitis than amylase with >3-fold the upper limit of lipase activity being indicative of pancreatitis (Frossard, Steer, and Pastor 2008). In our study, elevations in lipase and amylase activity were not consistent with pancreatitis. Similar to other studies that evaluated the effect of exenatide (Tatarkiewicz et al. 2013, 2010; Vrang et al. 2012; Gier et al. 2012) or liraglutide (Vrang et al. 2012) in rat models of diabetes, dulaglutide did not affect pancreatic weight or increase lipase activity in male ZDF rats. Although dulaglutide was associated with a slight increase in amylase activity (13–47% of control group mean), there was not a 1:1 relationship on an individual animal basis between total amylase measurements and the concurrence of inflammation (neutrophilic or mixed cell) in the control or dulaglutide-treated groups. The actual incidence of inflammation was <50% for mixed cell inflammation and <33% for acinar neutrophilic inflammation. Similarly, no correlation was found between the magnitude of change and the presence of acinar atrophy in the control or dulaglutide-treated groups. Others studies also did not find an association between administration of liraglutide (Vrang et al. 2012) or exenatide (Tatarkiewicz et al. 2013) and differences in acinar cell atrophy and inflammation between control and treated rats. Like dulaglutide, other GLP-1 receptor agonists slightly increase amylase activity in ZDF rats. Exenatide increased total amylase by about 30% and pancreatic amylase by approximately 25% (Tatarkiewicz et al. 2013; Vrang et al. 2012), and liraglutide increased pancreatic amylase activity by 18% (Vrang et al. 2012). In contrast, exenatide significantly increased lipase activity and did not alter amylase activity in Sprague-Dawley rats (Nachnani et al. 2010). Currently, it is not known if GLP-1 receptor expression in acinar cells in rats is similar to that in humans and macaques. In humans and macaques, the GLP-1 receptor is predominately present in the β-cells and also to a lesser extent in acinar cells, however, it is unclear if the receptor is expressed in acinar cells in rats (Gier et al. 2012; Xu et al. 1999; Zhou et al. 1999; Wild et al. 2006; Gotthardt et al. 2002; Pyke et al. 2014). Hence, the effects of GLP-1 agonists on amylase activity in rats may be indirect, perhaps acting through paracrine communication between β-cells and acinar cells (Tatarkiewicz et al. 2013).
The absence of a strong relationship between the incidence and severity of pancreatitis and diabetes in rodent models has been noted previously (Chadwick et al. 2013). The Chadwick study characterized the incidence of spontaneous pancreatic lesions in 3 rat strains frequently used in pharmaceutical toxicology studies: Sprague-Dawley, ZDF, and HIP rats (transgenic rats carrying the human islet amyloid polypeptide gene that is used as a model for type 2 diabetes; Matveyenko et al. 2009; Chadwick et al. 2013). No animals in the Chadwick study were treated with GLP-1-based therapies. The study found there was no relationship of plasma glucose markers (fructosamine and HbA1c) with the incidence of microscopic changes observed in the pancreas across the different rat models, and there was also no correlation of amylase or lipase levels with exocrine pancreas microscopic findings. Spontaneous pancreatic inflammation was seen in 40% (57/144) of animals across strains. Hence, histological changes ascribed to GLP-1-based therapies are commonly detected in the different rat models in the absence of drug administration. The results of Chadwick et al. point out the challenges of extrapolating cause–effect relationships in nondiabetic and diabetic rat models to human safety.
In conclusion, previous studies that evaluated the effect of GLP-1 receptor agonists in rats have produced inconsistent results in regard to drug-related changes in the pancreas. In this study, administration of dulaglutide to ZDF rats twice weekly for 13 weeks at systemic exposures up to 30 times greater than human treatment levels was associated with mild alterations in ductal epithelium and modest increases in spontaneous lesions of the pancreas that typically occur in this model. The current study differs from previous reports in the extensive evaluation of serial cross sections, with the consistent presentation of the main pancreatic duct with pancreatic duct glands and acinar tissue from all regions of the pancreas. These data build upon the evidence in other animal models that GLP-1 receptor agonists do not cause major histopathological changes in the exocrine pancreas. However, it is essential that ongoing clinical and pharmacoepidemiological studies continue to monitor the use of GLP-1 receptor agonist–based therapies in humans.
Footnotes
Acknowledgments
The authors would like to thank the team leaders and technical staff of the Necropsy, Histology and Histomorphometry Departments in Pathology (Charles River Laboratories-Montreal) for their attention to detail in method development and excellent execution of special procedures. We would also like to thank Tamara Ball, MD, of inVentiv Health Clinical for editorial support.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AU, RAB, JLB, JS, and JAM are employees of Eli Lilly and Company. JM, FP-B, and AP are employees of Charles River Laboratories, which has a contractual relationship with Eli Lilly and Company.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Eli Lilly and Company.
Authors’ Contribution
Authors contributed to conception or design (AP, AU, RB, JM, JB, JV); data acquisition, analysis, or interpretation (AP, AU, RB, JM, JB, JS, FP, JAM, JV); drafting the manuscript (AU, RB, JM, JB, JV); and critically revising the manuscript (AU, RB, JM, JB, JS, FP, JAM, JV). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
