Abstract
Three orally administered metabotropic glutamate receptor 5 (mGluR5) negative allosteric modulators caused skin lesions consistent with delayed type-IV hypersensitivity in cynomolgus macaques in 2- and 12-week toxicity studies. Several monkeys developed macroscopic skin lesions in multiple locations after 8 to 9 days of dosing; the most prominent effects involved the genital region of males and generalized erythema occurred in both sexes. Microscopic lesions occurred in both clinically affected and unaffected areas and were characterized by lymphocytic interface inflammation, subepidermal bullae, and individual keratinocyte vacuolation/necrosis. In the 12-week study, clinical effects in 2 animals resolved with continued dosing, whereas in others the inflammatory process progressed with 1 female exhibiting systemic lymphocytic inflammation in multiple tissues. The inflammatory infiltrate consisted of CD3 and CD4 positive T lymphocytes with minimal CD68 positive macrophages and only rare CD8 positive T lymphocytes. A subset of animals given a dosing holiday was subsequently rechallenged with similar lesions developing but with a more rapid clinical onset. These skin lesions were consistent with type-IV delayed hypersensitivity with some features comparable to bullous drug eruptions in humans. A relationship between these findings and the intended mode of action for these compounds could not be ruled out, given the occurrence across different chemotypes.
Keywords
Introduction
Skin reactions such as rashes and eruptions associated with drug administration are one of the most common adverse drug reactions (ADRs) observed clinically in humans, and their incidence can be as much as 3% in all hospitalized patients (Bigby et al. 1986). ADRs can be either predictable in humans when due to the drug’s pharmacologic action (type A reactions) or unpredictable when not related to the drug’s pharmacologic action (type B reactions; Rawlins and Thompson 1991). The vast majority of the drug-induced skin reactions (DSRs) in humans tend to be type B reactions and are idiosyncratic in nature (Uetrecht and Naisbitt 2013). The underlying pathogeneses behind DSRs in humans are thought to involve immune-mediated mechanisms. Immunological variables among human populations such as T cell receptor repertoire, major histocompatibility complex (MHC) haplotype variations, and development of tolerance contribute to the idiosyncratic nature of the DSRs (Uetrecht and Naisbitt 2013).
The observation of adverse skin reactions in clinical development results in a high attrition rate estimated to be 64% according to a multi-institutional case study report from the International Life Sciences Institute (ILSI) Workshop (Olson et al. 2000). This same study report also indicated that standard preclinical animal toxicological testing was a poor predictor of clinical DSRs (Olson et al. 2000). Additionally, the human translatability of observed skin reactions in preclinical rodent or non-rodent species is poorly understood with the one exception being phototoxicity testing in guinea pigs (Olson et al. 2000). The lack of animal models that reliably predict human DSRs is a significant limitation to the understanding and risk management of such skin reactions. A rat model of Nevirapine-induced skin rash has been established and appears to show some promise (Shenton et al. 2003), although given the lack of routine use of such assays it remains uncertain how predictive they may be for use in human risk assessment.
Here we report drug-induced skin lesions in cynomolgus macaques orally administered negative allosteric modulators (NAMs) of the metabotropic glutamate receptor 5 (mGluR5), which were being developed for the treatment of levodopa-induced dyskinesia in Parkinson’s disease (PD) patients treated with levodopa (Thanvi, Lo, and Robinson 2007). Aberrant mGluR5 regulation is also implicated in several other neurologic conditions including PD, autism, fragile X syndrome, and depression (Emmitte 2013). Numerous mGlur5 NAMs are currently in various stages of clinical and preclinical development (Domin et al. 2014; Emmitte 2013; Pop et al. 2014; Williams 2012). We discuss here the observed skin findings in detail along with our efforts to characterize the pathogenesis behind these drug-induced skin lesions in cynomolgus macaques treated with orally administered mGluR5 NAMs.
Methods
Proprietary Chemicals
PF-06422913, PF-06462894, and PF-06297470 (Figure 1)
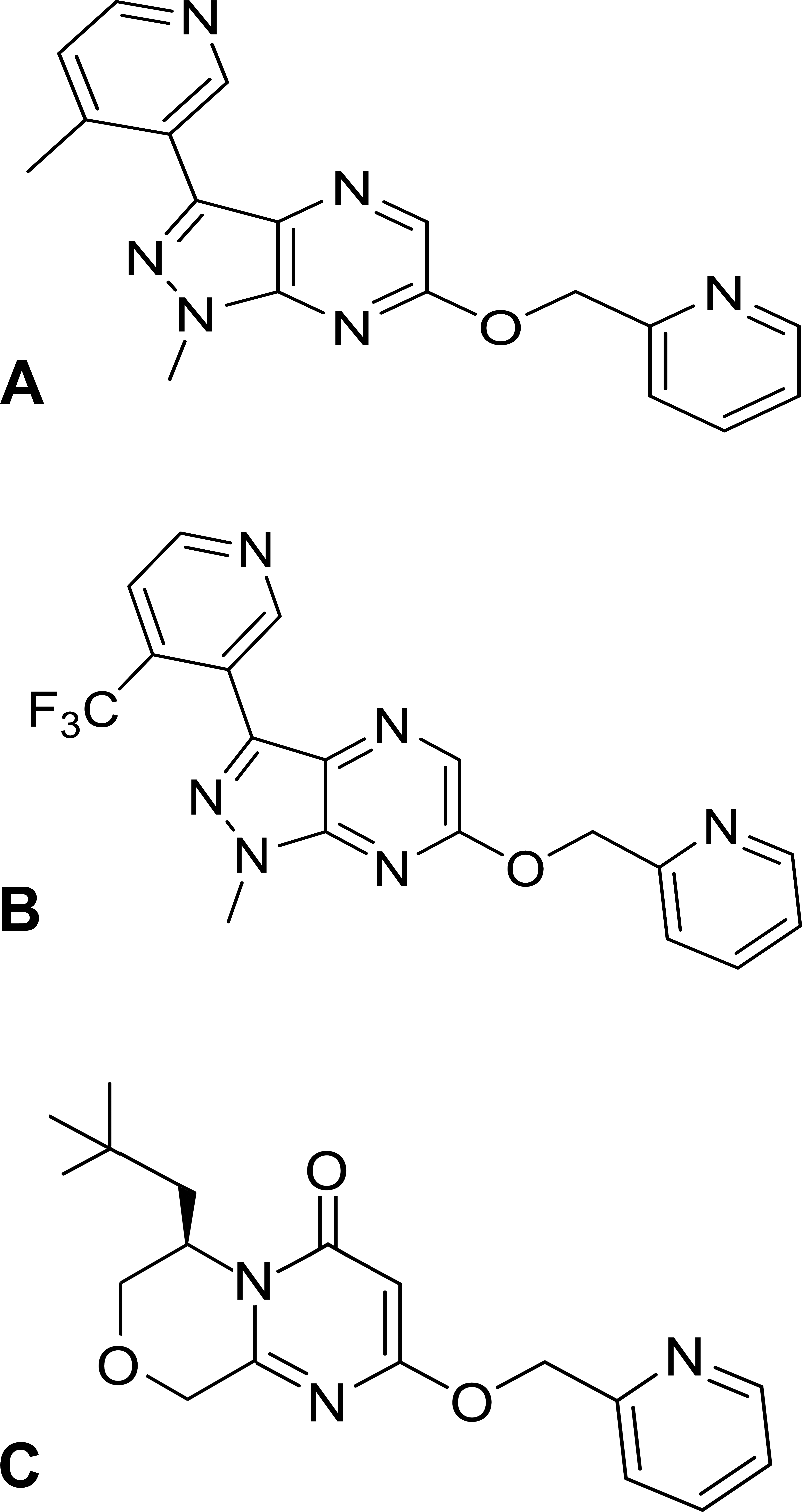
Chemical structures of three metabotropic glutamate receptor 5 (mGluR5)-negative allosteric modulators used in this study (A: PF-06297470; B: PF-06422913; and C: PF-06462894).
PF-06297470 (product no. PZ0219) and PF-06462894 (product no. PZ0231) are now commercially available at Sigma-Aldrich.
Animals and Husbandry
Male and female Mauritius origin cynomolgus monkeys (Macaca fascicularis) > 2.5 years old (males: 2.8 to 5.3 kg at dose initiation; females: 2.8 to 3.9 kg at dose initiation) were cared for in accordance with the Guide for the Care and Use of Laboratory Animals (Institute for Laboratory Animal Resources [ILAR] publication, 1996, NRC Press). Animals were housed at an indoor, Association for Assessment and Accreditation of Laboratory Animal Care International–accredited facility in species-specific housing. All experimental study protocols involving animals used in these studies and their housing conditions were reviewed and approved by Pfizer Institutional Animal Care and Use Committee (IACUC).
Certified Primate Diet (PMI Feeds 5K91; Richmond, IN) was provided daily in amounts appropriate for the age and size of the animals. Municipal drinking water, further purified by reverse osmosis, was available ad libitum to each animal via an automatic watering system. The animals were given additional supplements as a form of environmental enrichment and were given various cage-enrichment devices. Animals were maintained on a 12:12 hr light:dark cycle in rooms at 66°F to 77°F and relative humidity of 30% to 70%. All studies used naive animals. All animals were vaccinated for Measles virus and were seronegative for simian retroviruses 1 to 5, simian T lymphotrophic virus, simian immunodeficiency virus, macacine herpesvirus 1 (Herpes B virus), and Trypanasoma cruzi. Animals were also screened for Mycobacterium tuberculosis, Salmonella spp., Shigella spp., and Yersinia spp.
Study Designs
Separate 2-week oral gavage exploratory toxicity studies were conducted with PF-06422913, PF-06462894, and PF-06297470, followed by a 12-week toxicity study with PF-06297470. PF-06422913 and PF-06297470 were formulated for dosing in 0.5% (w/v) methylcellulose, while PF-06462894 was formulated in 12% sulfobutylether β-cyclodextrin. Dose volumes ranged from 5 to 10 ml/kg for PF-06422913 and PF-06297470, and 12.5 ml/kg for PF-06462894.
Two-week Exploratory Toxicity Studies
Animals received vehicle control or test article (n = 1/sex/group) once daily for 2 weeks (14 days) at the following doses: PF-06422913—15, 75, and 300 mg/kg/day. PF-06462894—50, 150, and 600 mg/kg/day. PF-06297470—5, 25, and 75 mg/kg/day.
Animals were evaluated for clinical signs (daily), body weight (weekly), food consumption (daily qualitative assessment), clinical pathology parameters (prior to the initiation of dosing and at necropsy), and plasma drug concentrations (day 1 and 13 or 14). On the day after the last dose (day 15), animals were sedated, euthanized by exsanguination, and necropsied. A selected set of tissues was collected in 10% neutral buffered formalin, Modified Davidson’s (testes and epididymis), or 3% glutaraldehyde (eye with optic nerve). Tissues were sectioned, stained with hematoxylin and eosin, and examined microscopically by a board-certified veterinary pathologist.
Twelve-week Toxicity Study
The dosing schedule for the 12-week toxicity study with PF-06297470 is included in the following table:
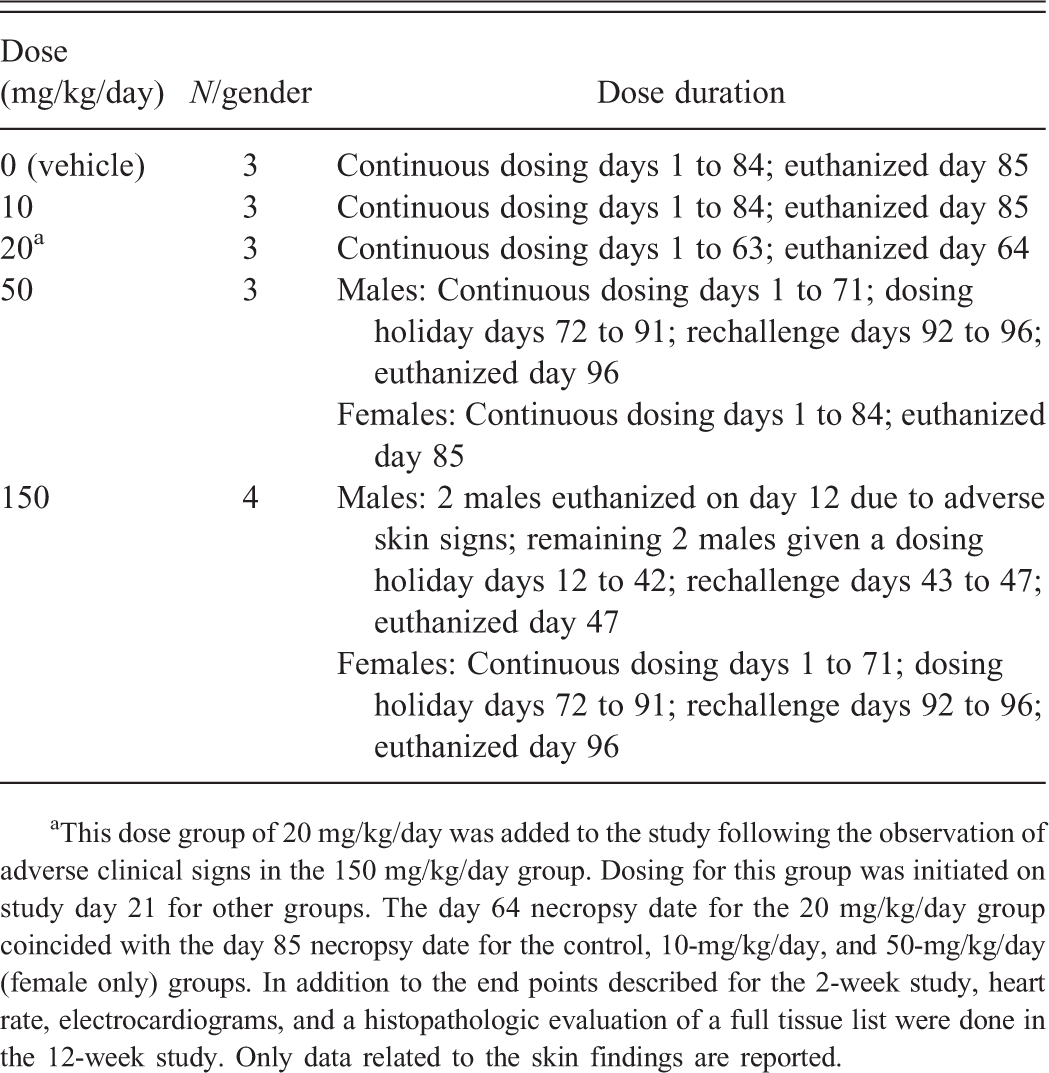
aThis dose group of 20 mg/kg/day was added to the study following the observation of adverse clinical signs in the 150 mg/kg/day group. Dosing for this group was initiated on study day 21 for other groups. The day 64 necropsy date for the 20 mg/kg/day group coincided with the day 85 necropsy date for the control, 10-mg/kg/day, and 50-mg/kg/day (female only) groups. In addition to the end points described for the 2-week study, heart rate, electrocardiograms, and a histopathologic evaluation of a full tissue list were done in the 12-week study. Only data related to the skin findings are reported.
Animals were evaluated for clinical signs (daily), body weight (weekly), food consumption (daily qualitative assessment), clinical pathology parameters (prior to the initiation of dosing, day 37, and at scheduled necropsy), heart rate and electrocardiograms (prior to the initiation of dosing and predose and 1 hr postdose during week 6), and plasma drug concentrations (day 1 and surviving animals on days 39 and 59 and select groups on day 80). For scheduled necropsies, animals were sedated on the day after the last dose, euthanized by exsanguination, and necropsied. Selected organs were weighed, and a complete set of tissues was collected in 10% neutral buffered formalin, Modified Davidson’s (testes and epididymis), or 3% glutaraldehyde (eye with optic nerve). Tissues were sectioned, stained with hematoxylin and eosin, and examined microscopically by a board-certified veterinary pathologist.
Toxicokinetics
Blood samples were collected into tubes containing K2 ethylenediaminetetraacetic acid (EDTA) at 1, 4, 7, and 24 hr following dosing on day 13 or 14 from the 2-week toxicity studies and on day 1, 39, 59, and 80 for the 12-week study. Plasma was separated and analyzed for test article content. Maximum plasma drug concentration (Cmax) and area under the plasma drug concentration–time curve (AUC) were determined for drug-treated animals using ThermoElecton Watson LIMS v. 7.2.
Immunohistochemistry (IHC) Methods
The expression patterns of CD3, CD4, CD20, CD68, and mGluR5 were evaluated by IHC on skin sections collected at necropsy from the 12-week study. Briefly, pretreatment steps for CD3, CD68, and CD8 were accomplished using the Biocare Decloaking Chamber (Biocare Medical, Concrod, CA) with a pH 6 citrate buffer (Biogenex, Fremont, CA) for CD3 and CD68 and a pH 8 EDTA buffer (Life Technologies, Grand Island, NY) for CD8. CD4 and mGluR5 were run on a Ventana XT (Ventana Medical Systems, Tucson, AZ) platform utilizing cell conditioner #1 for pretreatment. A 1/250 dilution of mouse antihuman CD68, clone KP1 (Leica Biosystems, Buffalo Grove, IL), and a 1/50 dilution of mouse antihuman CD8, clone 1A5 (Leica Biosystems) were incubated for 60 min at room temperature and detected with Dako Mouse Envision HRP+ (Dako, Carpinteria, CA) followed by Dako Liquid DAB+. CD3 staining was performed at a 1/500 dilution of rabbit antihuman CD3, clone SP7 (Thermo Scientific, Waltham, MA), incubated for 60 min at room temperature, and detected with Dako Rabbit Envision HRP+ followed by Dako Liquid DAB+. CD3, CD68, and CD68 IHC were run using the Biocare intellPATH IHC instrument. A 1/500 dilution of rabbit antihuman mGluR5, clone EPR2425Y (Epitomics, Burlingame, CA), was incubated for 60 min using the heating function of the Ventana XT followed by detection with OmniMAP Rabbit HRP (Ventana Medical Systems). CD4 staining was performed at a 1/50 dilution of mouse antihuman CD4, clone 1F6 (Leica Biosystems, Buffalo Grove, IL), incubated for 120 min at room temperature, and detected with UltraMAP Mouse AP (Ventana Medical Systems). Primate lymphoid tissues were utilized as positive controls for CD marker IHC, while brain (caudate lobe) was used for mGluR5. Matched mouse and rabbit IgG isotype controls were included with all IHC runs as negative controls. All IHC-stained slides were counterstained with hematoxylin and evaluated by light microscopy.
Results
Macroscopic (grossly apparent) and microscopic changes observed in cynomolgus monkeys orally administered with PF-06422913, PF-06462894, or PF-06297470 for 2 weeks daily are summarized in Table 1. Table 2 summarizes the toxicokinetic (TK) data on day 1 of dosing for all compounds in the 2- and 12-week studies. In general, all of the compounds exhibited an increase in exposure (Cmax and AUC(0-24)) with an increase in dose.
Macroscopic (gross) and microscopic (histopathologic) skin findings observed in cynomolgus macaques treated with PF-06422913, PF-06462894, or PF-06297470 for up to 2 weeks.
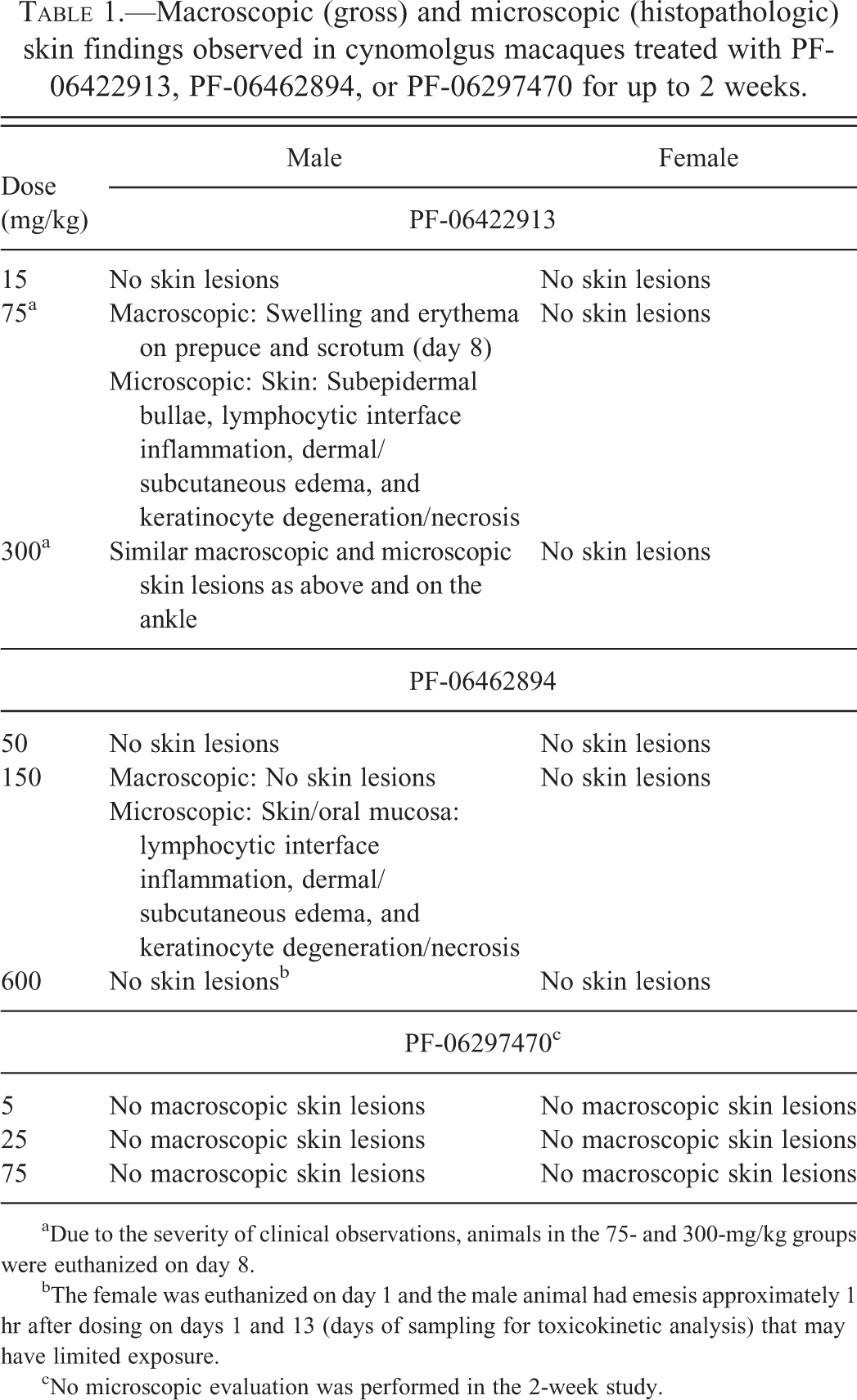
aDue to the severity of clinical observations, animals in the 75- and 300-mg/kg groups were euthanized on day 8.
bThe female was euthanized on day 1 and the male animal had emesis approximately 1 hr after dosing on days 1 and 13 (days of sampling for toxicokinetic analysis) that may have limited exposure.
cNo microscopic evaluation was performed in the 2-week study.
Mean toxicokinetic parameters following day 1 of dose administration.
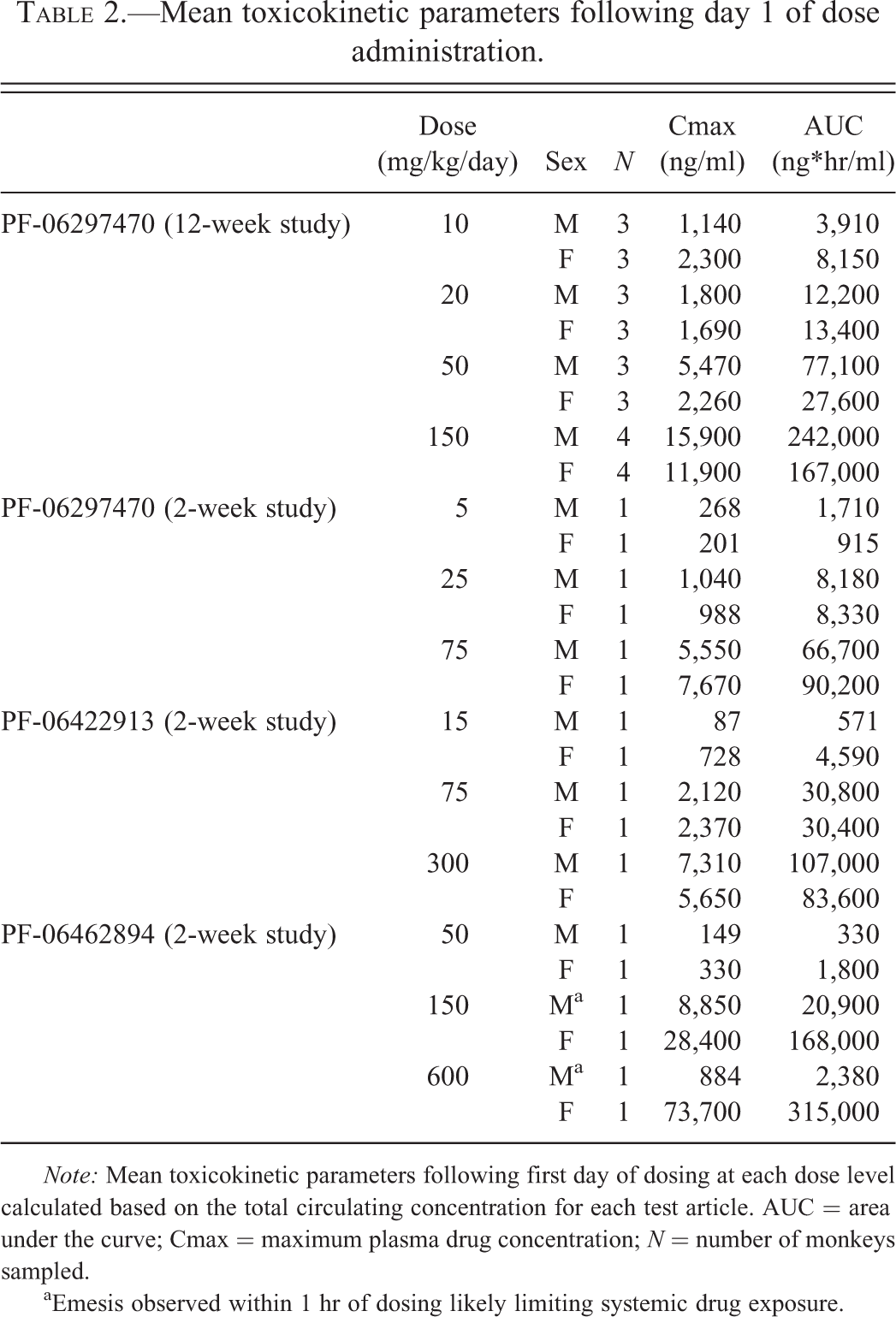
Note: Mean toxicokinetic parameters following first day of dosing at each dose level calculated based on the total circulating concentration for each test article. AUC = area under the curve; Cmax = maximum plasma drug concentration; N = number of monkeys sampled.
aEmesis observed within 1 hr of dosing likely limiting systemic drug exposure.
Macroscopic skin lesions appearing on day 8 in the 2-week studies included erythema and swelling in the preputial and scrotal areas or ankle of the 75- and 300-mg/kg males treated with PF-06422913. Due to the severity of these clinical observations, these animals were euthanized on day 8. No macroscopic skin lesions were noted in females treated with PF-06422913 for 2 weeks or in either sex treated with either PF-06462894 or PF-06297470 for 2 weeks.
In a 12-week toxicity study with PF-06297470, several macaques began to show macroscopic skin lesions on day 9 (Table 3), some of which progressed to open sores. The reason why these lesions were noted on day 9 in this study but not in the earlier 2-week study is uncertain, although this could be related to the much higher exposures achieved in the 12-week study (i.e., AUC-based therapeutic index of ∼150× in the 12-week study vs. ∼31× in the 2-week study).
Clinical observations in a 12-week study with PF-06297470 in cynomolgus macaques.
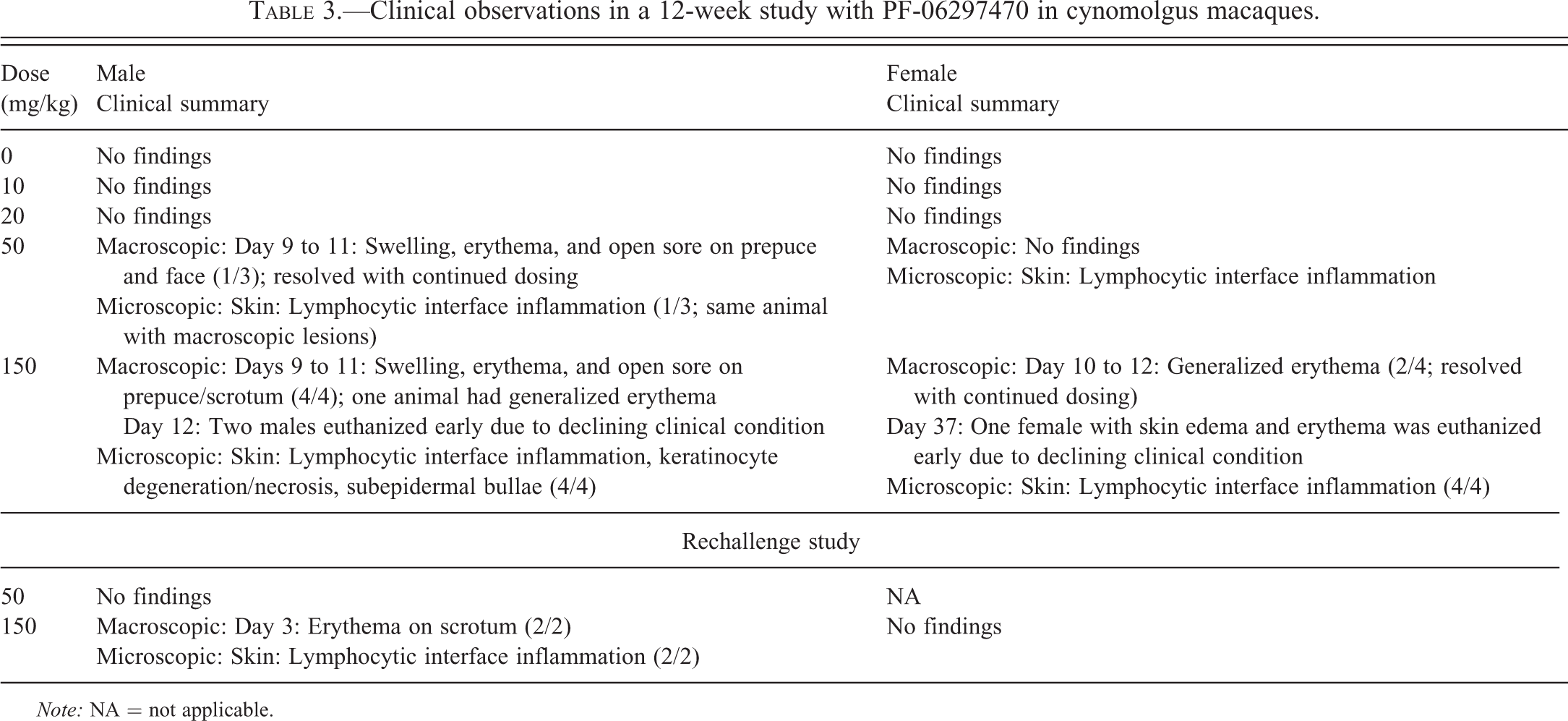
Note: NA = not applicable.
The macroscopic skin lesions observed in the mid- and high-dose males treated with PF-06422913 for 2 weeks corresponded with a spectrum of histopathologic skin lesions (Table 1). The 75- and 300-mg/kg males had multifocal areas of epidermal separation from underlying dermis (subepidermal bullae) of the scrotum and/or prepuce (Figure 2A; normal skin from control animal is shown in Figure 2B). Microscopically, these bullous spaces were filled with amphophilic fluid containing minimal inflammatory cells (predominately lymphocytes; Figure 2C). Other skin changes observed in this animal included lymphocytic inflammation at the epidermal–dermal interface (interface inflammation; Figure 3C), perivascular lymphocytic infiltrates, edematous expansion of the dermis and subcutis, and individual keratinocyte degeneration and necrosis characterized by cytoplasmic hypereosinophilia and nuclear pyknosis predominantly involving the basal layer (Figure 3A). Occasional areas of full-thickness transepidermal necrosis were also seen (Figure 3B). All these aforementioned histopathologic skin changes except for the subepidermal bullae were observed in the 150-mg/kg male treated with PF-06462894, even though there were no clinical observations or macroscopic lesions observed. This animal also had involvement of oral mucosa (interface inflammation) similar to that noted in the skin. In PF-06297470-treated animals from the 2-week study, skin sections were not collected for microscopic evaluation, as this was the initial exploratory toxicity study conducted among those referenced here and skin lesions had not yet been observed.
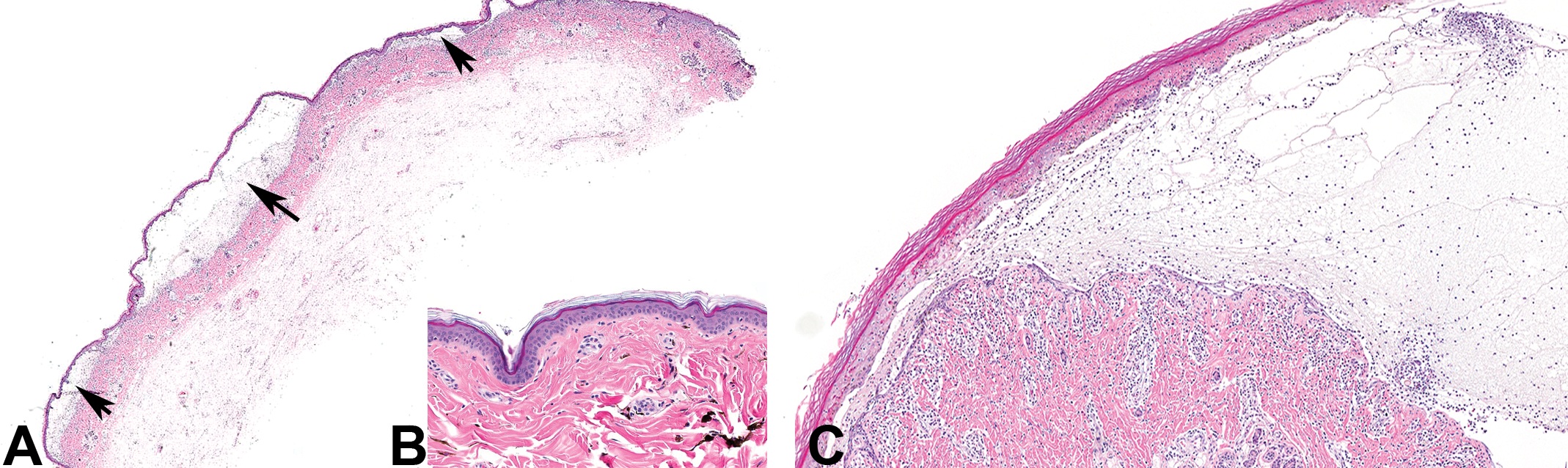
Three metabotropic glutamate receptor 5 (mGluR5) negative allosteric modulators (NAMs) caused subepidermal bullae. Photomicrographs A and C illustrate bullae seen in the skin of the monkeys treated with PF-06422913, PF-06462894, and PF-06297470. Figure A is a representative image (4×) showing multiple subepidermal bullae (thick arrows). The inset figure B illustrates skin from a control animal (10×). Figure C is a higher magnification view (10×) of a subepidermal bulla that is filled with clear fluid admixed with minimal inflammatory cells.
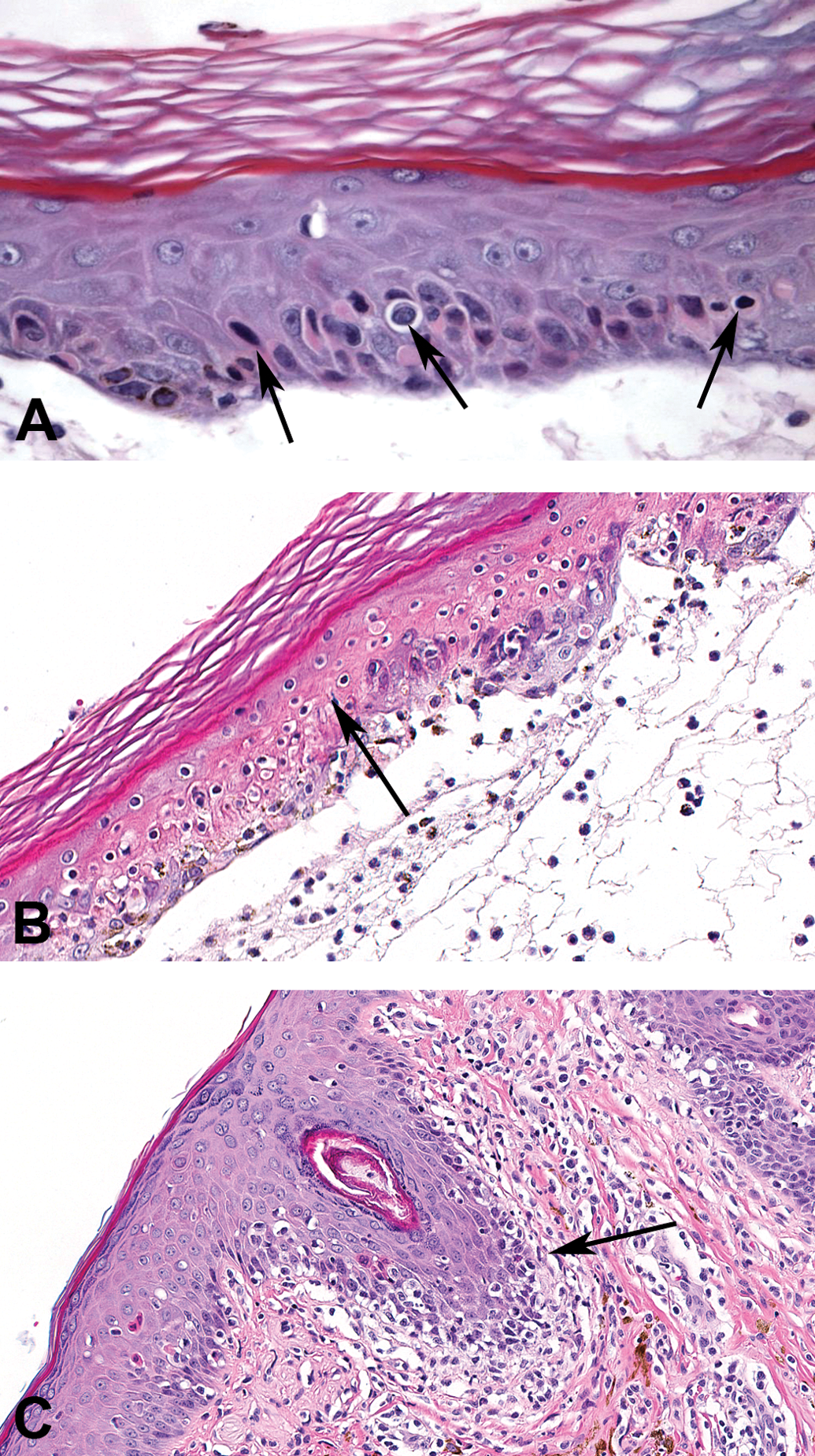
Spectrum of skin lesions seen in metabotropic glutamate receptor 5 (mGluR5) negative allosteric modulator-treated animals. Observed drug-induced lesions included individual keratinocyte degeneration and necrosis (A), full-thickness trans-epidermal necrosis (B), and lymphocytic interface dermatitis (C).
The macroscopic and microscopic characteristics of the skin lesions in the 12-week study with PF-06297470 were similar to those noted in the 2-week study with PF-06462894 and PF-06422913 (Figures 2 and 3). Table 3 presents clinical observations including skin lesion incidences noted in the 12-week study. Skin lesions were noted macroscopically and/or microscopically in preputial, scrotal, and dorsal skin areas. In addition, 1 high-dose female that displayed generalized erythema early in the study (days 10–12) but resolved with continued dosing later went on to develop a systemic inflammatory response involving T-cell-dominant lymphocytic inflammation in the liver (Figure 4A), kidneys (Figure 4B), and brain (figure not shown), resulting in the declining clinical condition and early euthanasia of this animal on day 37. Two males from this high-dose (150 mg/kg) group were also euthanized early on day 12 due to declining clinical conditions. In addition to the preputial, scrotal, and dorsal skin areas, several animals exhibited similar interface inflammation in the skin from the mammary gland region as well as in the esophagus and urinary bladder mucosa.
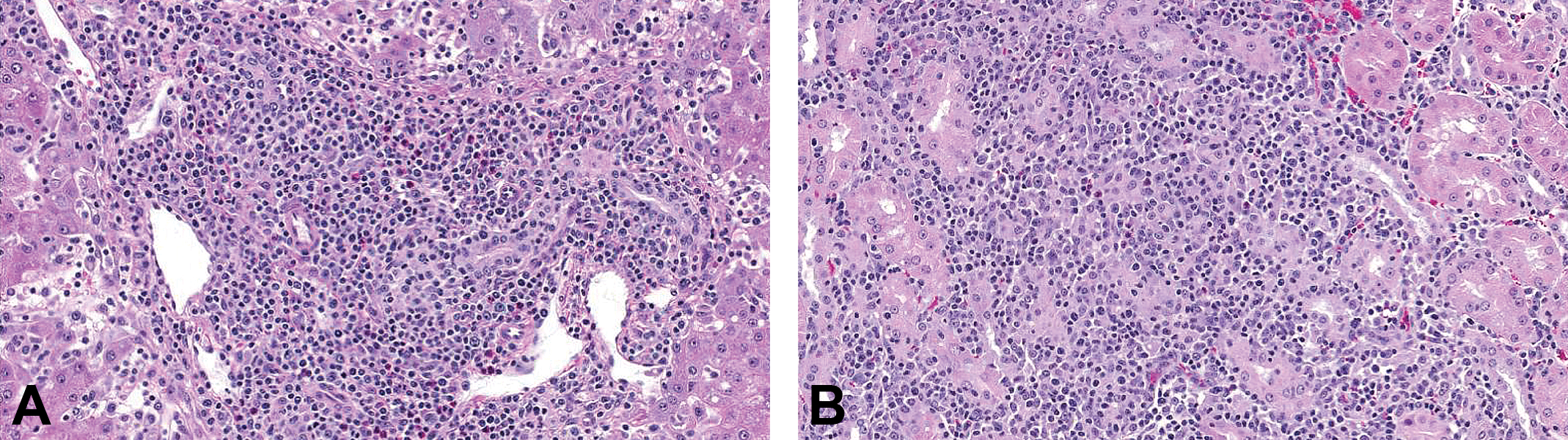
Systemic involvement seen in 1 animal treated with PF-06297470. Multifocal lymphocytic inflammation involving portal areas in liver (A) and interstitium of the renal cortex (B) was observed in 1 animal treated with PF-06297470.
Table 4 summarizes the immunohistochemical study of skin lesions characterizing the cellular phenotype of the inflammatory infiltrates in the skin of PF-06297470-treated monkeys. The majority of inflammatory cells at the epidermal–dermal interface stained positively for the pan-T lymphocyte marker CD3 and helper T lymphocyte marker CD4 (Figure 5A and B). A variable but small percentage of cells in this inflammatory process stained positively for the macrophage marker CD68, although only rare CD8-positive cytotoxic T lymphocytes were present and most of these were intermingled with more numerous CD4-positive lymphocytes in the dermis (Figure 5C and D). Additional immunostains of affected skin areas did not reveal significant numbers of CD20-positive B lymphocytes or changes in mGluR5 protein expression in the skin or inflammatory infiltrates (data not shown).
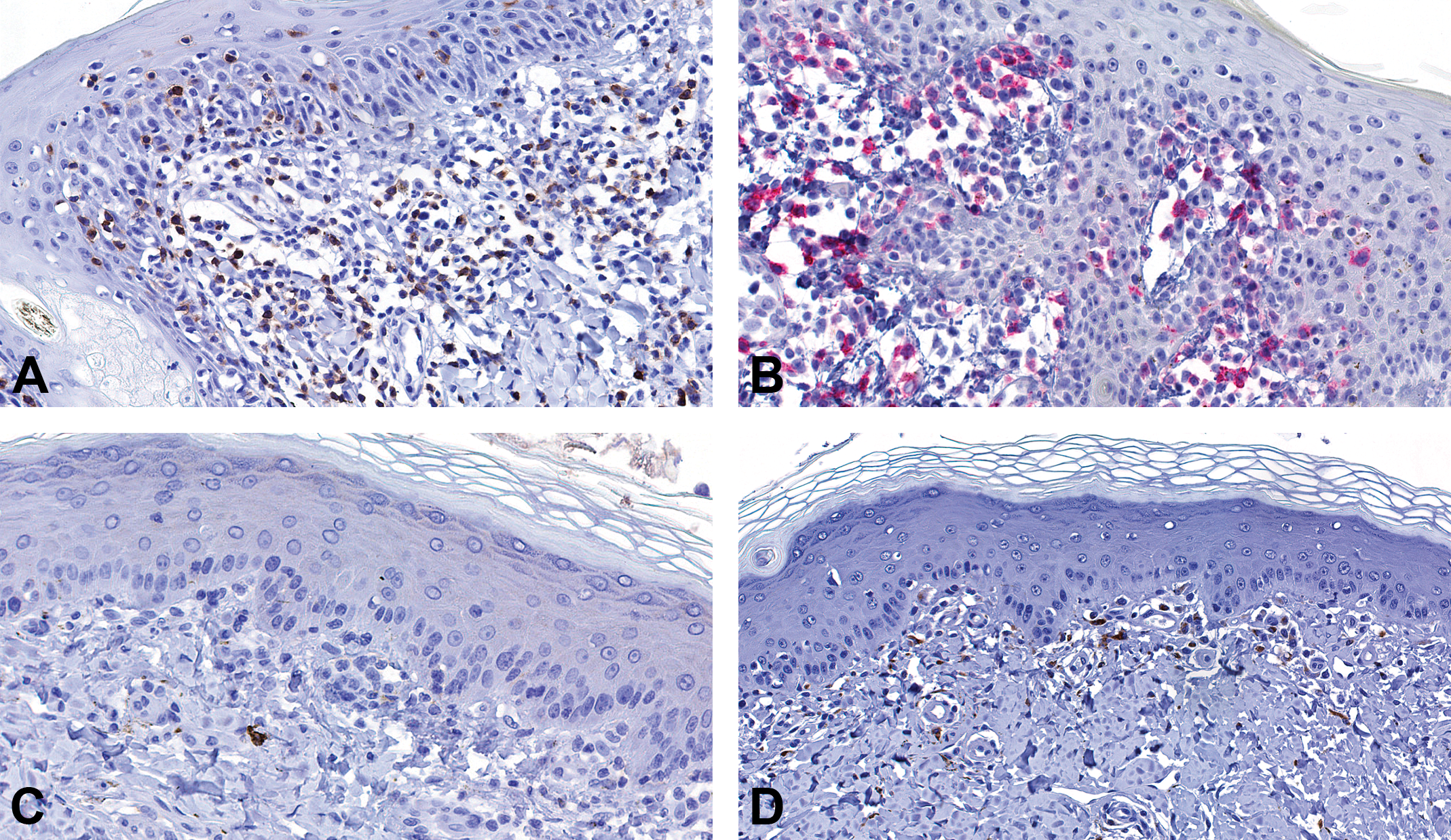
Inflammatory cells in drug-induced skin lesions are CD4 T lymphocytes. At the interface, inflammatory cells predominately stained positively for T lymphocytes markers CD3 (A) and CD4 helper T cells (B). There were very few CD8-positive cytotoxic T cells (C) and only minimal numbers of CD68-positive macrophages (D).
Immunohistochemical staining summary of skin lesions.

Note: mGluR5 = metabotropic glutamate receptor 5.
Rechallenge Study Results
In the 12-week cyno study with PF-06297470, a subset of animals (2 males from 150-mg/kg group) that displayed skin findings on days 10 to 12 were given a 4-week dosing holiday to allow for complete reversal of their clinical observations and then were rechallenged with the same daily dose of compound for 5 days. In these rechallenge animals, the time of onset for clinical observations in the skin was shorter, starting on day 3 as compared to day 9 upon initial exposure. The more rapid occurrence upon rechallenge is consistent with what would be expected with a hypersensitivity reaction. It has also been reported that rechallenge can result in a more severe response, although this was not noted in these animals. At the microscopic level, the skin findings in these rechallenge animals were of similar severity to other animals in the study (data not shown).
Discussion
DSRs are common events in human clinical trials and can potentially be lethal (Downey et al. 2012). The risk factors and mechanism are not well understood for DSRs, which makes it difficult to understand why some people react while others do not. Unfortunately, these effects are only rare if ever predicted by preclinical toxicity studies. The reason for this discrepancy is largely unknown but may be related to the mechanism being dependent on a specific immune-mediated response unique to certain individuals but generally not common in large numbers of individuals. Whether or not the lack of preclinical predictability is related to a true lack of sensitivity to this mechanism in commonly used preclinical species or perhaps a function of the relatively low animal numbers utilized in toxicity studies is unknown although both factors could be involved. Here, we report the development of drug-induced skin lesions in cynomolgus monkeys orally administered small-molecule mGluR5 NAMs.
Rodent preclinical studies with each of these mGluR5 NAMs did not demonstrate any skin findings with either 2 weeks (PF-06422913, PF-06462894, and PF-06297470) or 12 weeks (PF-06297470) of oral administration, despite rats achieving similar drug exposure to cynomolgus monkeys (data not shown). Initial attempts to establish beagle dog as the non-rodent toxicity species were abandoned due to emesis and inadequate oral exposures resulting in the use of cynomolgus macaques as the non-rodent species.
Before the relationship between these mGluR5 NAMs and skin lesions was established, a number of differential diagnoses were considered including various viral or bacterial (toxic shock syndrome) causes. However, these differentials were quickly ruled out for a number of reasons including the lack of characteristic viral effects (inclusions, syncytia, etc.), seronegativity for Herpes B virus, and history of vaccination for Measles virus. Moreover, in due time, the dose-related association between these characteristic skin findings and 3 different mGluR5 NAMs in independent studies confirmed the test article relationship. The characteristic microscopic findings (interface inflammation), time of onset, more rapid onset after rechallenge, and T lymphocyte-dominant immunophenotype were all consistent with a type-IV hypersensitivity reaction; however, these skin effects frequently displayed a clear dose relationship which was considered unusual for a mechanism typically associated with an idiosyncratic pattern in humans. Other possibilities that were considered potential factors, but not explored in great detail, included local accumulation of compound in the skin and/or local generation of a reactive drug metabolite capable of triggering a type-IV hypersensitivity inflammatory reaction. Interestingly, skin melanocytes in humans are known to express mGluR5 (Frati et al. 2000). However, an immunohistochemical investigation did not reveal an appreciable mGluR5 protein expression in lesioned skin including epidermis, melanocytes, or inflammatory infiltrates.
Taken collectively, these drug-induced skin lesions were consistent with a delayed type-IV hypersensitivity reaction. The primary factors leading to this conclusion included a time of onset that was consistent with this mechanism (8–9 days after initiation of treatment), the CD4 positive T lymphocyte-dominant inflammatory infiltrates at the dermo-epidermal or mucosal–submucosal junctions, and the quicker recurrence of erythematous skin lesions upon rechallenge (3 days). The presence of similar inflammation in other organs including liver, kidney, and brain indicated that at least in some animals this inflammatory process goes beyond the skin and becomes more of a systemic reaction.
Depending on the immunologic effector mechanism involved, type-IV hypersensitivity is further classified as type-IVa, b, c, and d, which utilize macrophages, eosinophils, T lymphocytes, and neutrophils, respectively, as effector cells that mediate cell and tissue damage (Pichler 2003). The hypersensitivity reaction observed in these monkeys was most consistent with a type-IVc reaction due to the predominant presence of T lymphocytes. The bullae formation and epidermal necrosis are also likely mediated by infiltrating CD4 positive T lymphocytes, given the limited involvement of cytotoxic CD8 cells in the interface inflammation or within the epidermis itself. It is possible that CD4 positive T lymphocytes could take up a cytotoxic phenotype mediating cell death (Marshall and Swain 2011); however, additional efforts to further characterize the phenotype of these CD4 positive T cells were not done.
A classic type-IV hypersensitivity reaction requires a sensitization phase where the initial exposure with the offending drug occurs and an elicitation phase where reexposure to the same drug occurs (Uetrecht 2007). In this study, it is unclear how sensitization occurred. It may be that a reactive drug or its metabolite binds to host protein resulting in autoimmunity against drug-modified protein as postulated by the “hapten hypothesis” or the drug may reversibly bind to MHC and T-cell receptor (TCR) like a superantigen resulting in an autoimmune-like response as proposed by the “pharmacologic interaction (pi) hypothesis” (Uetrecht 2007). We did not identify any circulating reactive metabolites for these drugs although local formation of a reactive metabolite in the skin could not be ruled out. Additionally, it is unclear why lesions in male animals were more severe than females. There were no sex-related differences in overall exposures however, metabolite profiles were done with pooled male and female samples therefore the possibility of sex-specific differences in metabolite profiles could not be ruled out.
The dose-related response for the skin lesions observed in mGluR5 NAM-treated monkeys is a key difference from the common DSRs in humans. DSRs in humans are mostly idiosyncratic in nature with no direct dose-related response although certain dose levels have been shown to be associated with a higher incidence of DSRs with certain drugs (Uetrecht and Naisbitt 2013). DSRs in humans that have similar histologic characteristics and thought to involve immune-mediated hypersensitivity include erythema multiforme (EM; minor or major), Stevens–Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), fixed drug eruption (FDE), and bullous pemphigoid (Uetrecht 2007).
EM minor (local skin involvement only) and major (with systemic involvement) are often associated with drug exposure although current evidence suggests there may also be viral causes for these lesions (Hausmann, Schnyder, and Pichler 2010). SJS and TEN are considered by many to be a continuum of the same entity being distinguished clinically by the extent of epithelial detachment and both are potentially lethal conditions in humans (Gerull, Nelle, and Schaible 2011). Despite the monkeys in these studies having comparable features at the microscopic level, the extent of their lesions did not fit with the clinical definitions for either SJS or TEN. The effector cells in EM, SJS, and TEN are thought to be CD8 positive T lymphocytes (Hausmann, Schnyder, and Pichler 2010), which is also inconsistent with the CD4 positive T lymphocyte-predominant inflammation observed in these monkeys. Whether or not this represents a species-specific difference in mechanism or perhaps is a reflection of differences in timing of when the samples were collected remains uncertain.
The development of systemic involvement in 1 animal described here shares some characteristics with DRESS syndrome in humans, but there was no evidence of circulating eosinophilia in these animals. The possibility of these skin lesions being an FDE-like reaction described in humans cannot be completely ruled out based solely on these studies, given the limited number of rechallenge attempts although the presence of systemic involvement in 1 animal suggests this is unlikely (Gerull, Nelle, and Schaible 2011). Bullous pemphigoid is a condition mediated by an IgG response against epidermal basement membrane but is also considered unlikely in these monkeys due to the lack of a demonstrable B cell component to the inflammation (Jordon, Kawana, and Fritz 1985).
The reason for the unique dose relationship for these skin findings is unclear. This may suggest a possibility that the mechanism leading to the inflammation is more of a direct compound-related effect rather than being dependent on a particular immunological reaction that could vary by individual and lead to more of the anticipated idiosyncratic-type reactions noted in humans. Alternatively, it is possible that the population of animals utilized in these toxicity studies is enriched with a particularly susceptible haplotype making what would typically appear as an idiosyncratic pattern in a genetically diverse human population appear more dose related in a less diverse population of cynomolgus macaques. These factors are currently being explored further and this possibility may be supported by the known association between certain human leukocyte antigen (HLA) haplotypes and DSRs in humans (Gerull, Nelle, and Schaible 2011) as well as the evidence of relatively limited HLA diversity in certain populations of cynomolgus macaques (Aarnink et al. 2010). Drug accumulation cannot be ruled out as a possibility for the delay in onset of lesions and the observed dose relationship; however, CD4 positive lymphocyte-predominant inflammatory response and an earlier onset of lesion upon rechallenge support immune-mediated hypersentivity as the underlying pathogenesis for these skin lesions.
Another unique finding in this series of studies was the clinical recovery of skin findings in some animals even with continued dosing. In general, these clinical effects were mild in these animals and occurred with the same time of onset (8–9 days) as the other animals, although the apparent recovery and lack of progression of these clinical effects suggests the possibility of some degree of toleration. Interestingly, this type of immune tolerance has also been shown to occur in human patients where similar resolution of skin reactions occur despite continuous administration of the drug (Uetrecht and Naisbitt 2013). Development of anergy or regulatory T cell-mediated immunosuppression as a possible tolerance mechanism was hypothesized in these animals but was not explored further. It is also notable that some of these animals eventually went on to develop more severe lesions over time including one that developed multisystemic inflammation indicating that the pathogenesis of this toxicity is complex.
Although the exact initial events leading to the development of these lesions were not determined, the possibility of this being target associated (mGluR5) could not be completely ruled out given its development across multiple compounds representing two different chemotypes.
In summary, three different orally administered small-molecule mGluR5 NAMs representing two structurally distinct chemotypes induced an immune-mediated, delayed type-IV hypersensitivity skin reaction in cynomolgus macaques in a dose-dependent manner. Although the clinical translatability of these skin lesions is unknown, the significant clinical and microscopic similarities with potentially lethal DSRs in humans prohibited the clinical evaluation of these three compounds.
Footnotes
Acknowledgments
We thank Danielle Sullivan and Dean Wilkie for the immunohistochemistry work performed in this study.
Author Contribution
Authors contributed to conception or design (JM, GC, JW, CS, CH); data acquisition, analysis, or interpretation (GP, JM, GC, JW, CS, CH); drafting the manuscript (GP); and critically revising the manuscript (GP, JM, GC, JW, CS, CH). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
