Abstract
A few reports indicated the incidence of hematolymphoid neoplasms in old CD-1 mice, but the cellular lineage of CD-1 mouse neoplasms has not been published. In this study, immunohistochemistry (IHC) was used to characterize the cellular lineage of spontaneous hematolymphoid neoplasms arising in 24 young female CD-1 mice used as health-monitoring sentinels and 32 aging female CD-1 mice used as controls in 80-week carcinogenesis studies. Lymphoblastic lymphomas of T-cell and B-cell lineage were common in mice aged 12 months or less, whereas a wide range of non-lymphoblastic B-cell lymphomas and lymphoblastic B-cell lymphomas were common in mice >12-mo-old. Renal hyaline droplets positive for lysozyme were observed in aged mice with a histiocytic-associated large B-cell lymphoma (HA-BCL) and a myeloid leukemia. Endogenous ecotropic mouse leukemia virus (MuLV) genes have been recovered from CD-1 mice, but MuLV protein expression has not been previously demonstrated. We reported for the first time the expression of a MuLV protein p30 by IHC in lymphomas and some normal tissues of both young and aging CD-1 mice. This report should help to differentiate spontaneous lymphomas and leukemias in CD-1 mice from those induced by chemicals and other methods.
Introduction
The CD-1(ICR) mouse is a multipurpose outbred research mouse. It is the most commonly outbred mouse stock that is used in aging, oncology, and toxicologic safety and efficacy testing (Maita et al. 1988; Son 2003; Baldrick and Reeve 2007; Bradley, Mukaratirwa, and Petersen-Jones 2012). There are a few reports that indicate the incidence of hematolymphoid tumors in CD-1 mice (Maita et al. 1988; Son and Gopinath 2004; Ward 2006; Baldrick and Reeve 2007; Giknis and Clifford 2010; Bradley, Mukaratirwa, and Petersen-Jones 2012). Only one of these reports (Son and Gopinath 2004) addresses the incidence of these tumors in mice less than 12 mo of age, where the other reports note the incidence in mice on 2 yearlong studies. Furthermore, only one publication has attempted to provide a morphologic classification other than the collective term lymphoma for these tumors in CD-1 mice (Bradley, Mukaratirwa, and Petersen-Jones 2012). The purpose of this study was to further characterize and classify hematolymphoid tumors with immunohistochemistry (IHC) based on cellular lineage that occurs in young and aged CD-1 mice and to determine whether a retrovirus was expressed in these tumor-burdened CD-1 mice.
Materials and Methods
Animals
Tissue samples were collected from a total of 56 female mice. Twenty-four mice were maintained in the St. Jude Children’s Research Hospital (SJCRH) sentinel program, and they either were sacrificed or died between 2008 and 2014. Sentinel mice were generally maintained until 12 to 14 mo of age and then often disposed of and not necropsied unless they were ill. Of the 218 female sentinel CD-1 mice necropsied over 6 years at SJCRH, 24 had lymphoma. Thirty-two mice were obtained from the control group of an 80-week dietary carcinogenesis study evaluated between October 2009 and April 2011 and conducted by Charles River Edinburgh (CRE). It was previously reported that 17% of males and females in the CRE 80- and 104-week studies with CD-1 mice developed hematopoietic tumors (Bradley, Mukaratirwa, and Petersen-Jones 2012). The animals were purpose bred for laboratory use; the SJCRH mice came from Charles River USA breeding facilities at Portage, Michigan, and Kingston, New York; and the CRE mice came from Charles River European suppliers (Charles River UK Ltd, Margate, Kent). All control mice incorporated into the CRE studies were obtained from groups of mice that had been sham dosed by giving untreated diet ad libitum. For compatibility reasons, only CD-1 female mice are used in the SJCRH sentinel program. Consequently, all the mice in this study from both sources were females. Male mice were not studied from the 80- or 104-wk studies, in part, because they were reported to have a lower incidence of similar morphologic types of lymphomas as did females reported by one of our coauthors (Bradley, Mukaratirwa, and Petersen-Jones 2012). The mice were housed in groups of 3 or 5 animals per cage. The temperature and humidity of the SJCRH animal rooms were automatically controlled at 20.6°C to 22.8°C and 30% to 70%, respectively, with a minimum of 15 air changes/hr. An automatic 12-hr light–dark cycle of 0600 to 1800 hr was maintained. Animals were provided with reverse osmosis purified water chlorinated at 8 ppm ad libitum and fed an ad libitum commercial rodent diet (Autoclavable Rodent Breeder Diet 5013; LabDiets, Richmond, IN). The temperature and humidity of the CRE animal rooms were automatically controlled at 19°C to 23°C and 40% to 70%, respectively, with a minimum of 15 air changes/hr. An automatic light cycle of 0700 to 1900 (12-hr cycle) was maintained. The CRE animals were provided water ad libitum and fed ad libitum medicated commercial rodent diet (Rat and Mouse Modified No. 1 Diet SQC Expanded; Special Diets Services Ltd, Essex, United Kingdom).
The SJCRH study was conducted with approval of the Institutional Animal Care and Use Committee in accordance with National Institutes of Health guidelines. The CRE studies were conducted in accordance with the UK Animals (Scientific Procedure) Act 1986, which conforms to the European Convention for the Protection of Vertebrate Animals Used for Experimental and other Scientific Purposes (Strasbourg, Council of Europe).
Histopathology
Decedent animals and live animals humanely euthanized were necropsied, and tissues from all organ systems were collected and fixed in 10% neutral-buffered formalin. All tissues were embedded in paraffin, sectioned at 4 to 5 µm, and stained with hematoxylin and eosin. The histopathology and IHC of all the lymphoid tissues and bone marrow, when available, were examined blindly by two veterinary pathologists (J.E.R. and J.M.W.), and all hematolymphoid proliferations were classified morphologically according to the recommendation of the Mouse Models of Human Cancer Consortium (Morse et al. 2002; Kogan et al. 2002). The diagnoses reported represent the consensus opinions of both pathologists along with the concurrence of the CRE veterinary pathologist (A.B.).
Additional unstained sections were processed for IHC. Tissues from all SJCRH and CRE cases were immunostained for T- and B-cell antigens and select cases for macrophage and myeloid antigens. The IHC procedures and antibodies used for the hematolymphoid antigens were as described previously (Rehg, Bush, and Ward 2012; Table 1). To detect the presence of Rauscher mouse leukemia virus (MuLV) p30, tissues from 16 SJCRH and 9 CRE mice were stained. The goat anti-p30 antibody was provided by Dr. Sandra Ruscetti, the National Cancer Institute, Frederick, Maryland (O’Brien, Simonson, and Davis 1978), and IHC p30 procedures were as described previously (Hartley et al. 2008).
Commonly used antibodies and sources for mouse hematolymphoid antigens detectable in paraffin sections.
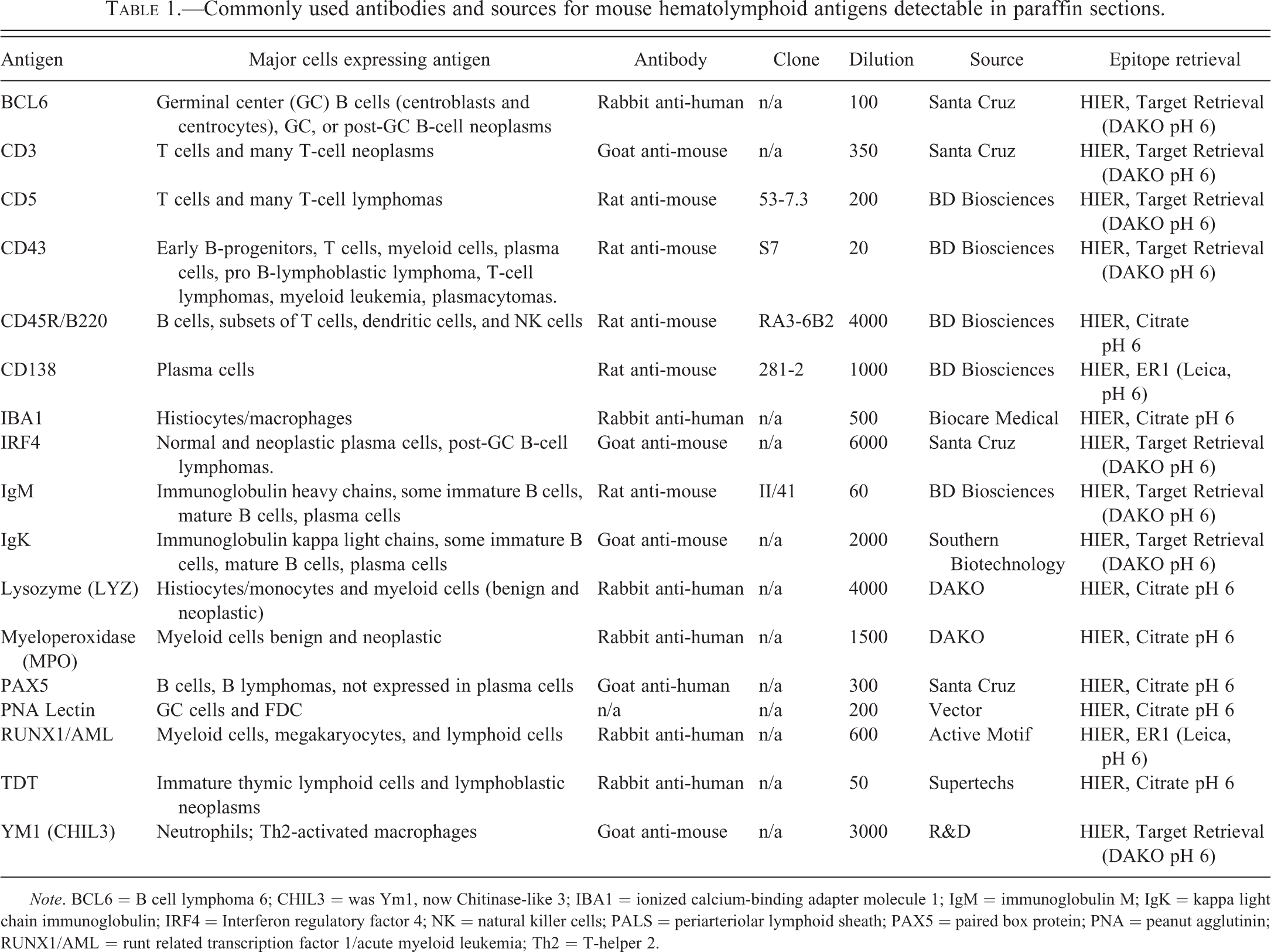
Note. BCL6 = B cell lymphoma 6; CHIL3 = was Ym1, now Chitinase-like 3; IBA1 = ionized calcium-binding adapter molecule 1; IgM = immunoglobulin M; IgK = kappa light chain immunoglobulin; IRF4 = Interferon regulatory factor 4; NK = natural killer cells; PALS = periarteriolar lymphoid sheath; PAX5 = paired box protein; PNA = peanut agglutinin; RUNX1/AML = runt related transcription factor 1/acute myeloid leukemia; Th2 = T-helper 2.
Results
Table 2 presents the spontaneous histopathological findings recorded for mice from the SJCRH sentinel program and an 80-week study at CRE. The hematolymphoid neoplasms in the SJCRH mice, except for two, occurred in mice aged 12 mo or less and were classified into 4 groups, whereas 30 of the 32 neoplasms in the CRE mice occurred in mice older than 12 mo. Some hematolymphoid neoplasms (lymphoblastic lymphoma, follicular lymphoma, and myeloid leukemia) were common to both groups, whereas other neoplasms were only observed in the CRE mice, and these mice were generally 20 mo of age or less.
Hematolymphoid neoplasms in 56 young and aging female CD-1 mice.

a The number of tumors exceeds the number of mice because 2 mice each had 2 hematolymphoid tumors.
The 32 cases of lymphoblastic lymphoma (LBL) in mice less than and greater than 12 mo of age were characterized by medium-sized blastic cells having scant pale cytoplasm, nuclei with fine stippled chromatin, and inconspicuous or small- and medium-sized nucleoli (Figures 1 and 2). The nuclei were round or irregular, and the cells formed noncohesive sheets. Tingible body macrophages were scattered among the tumor cells, giving a starry sky appearance. Morphologically, 17 cases of T-cell LBL (T-LBL) and 15 cases of B-cell LBL (B-LBL) were indistinguishable except perhaps for the involvement of thymus in T-LBL. Often, it was not possible to determine whether the thymus or mediastinal lymph nodes or both were involved. However, IHC with the use of antibodies to T-cell and B-cell antigens, the lymphoblastic lymphomas were further classified by their cellular lineage into T-LBL or B-LBL. The lymphoblastic lymphomas tend to involve the spleen, multiple lymph nodes, liver, lungs, kidneys, ovaries, and the central nervous system, but T-LBL (53%, 9 of 17) was more likely than B-LBL (7%, 1 of 15) to involve the small intestine (Table 3). Lymphoblastic lymphoma was more common (96%, 23 of 24) in mice aged 12 mo or less than in mice aged over 12 mo (33%, 10 of 30), and T-LBL was more common (67%, 16 of 24) than B-LBL (29%, 7 of 24) in the young age-group. Antigen expression among the different hematolymphoid tumors was characteristic of specific lineages and the maturation stages of the different class of tumors (Table 4). The T-LBLs were labeled with CD3 (100%, 17 of 17; Figure 3), CD5 (76%, 13 of 17), CD43 (100%, 17 of 17), and TDT (82%, 14 of 17; Figure 4). The B-LBLs labeled with, paired box protein (PAX5; 100%, 15 of 15), CD45R/B220 (40%, 6 of 15; Figure 5), immunoglobulin M (IgM; 53%, 8 of 15), CD43 (53%, 8 of 15), and TDT (13%, 2 of 15), but they did not label with kappa light chain (IgK, 0%, 0 of 15). Based on the expression of IgM, the B-LBLs consisted of 2 maturation subtypes, pro-B (47%, 7 of 15), which lack IgM immunoreactivity, and pre-B (53%, 8 of 15). The pro-B LBL labeled with CD45R/B220 (53%, 4 of 7), PAX5 (100%, 7 of 7), and CD43 (70%, 5 of 7), but they did not label with IgM (0%, 0 of 7) or IgK (0%, 0 of 7). The pre-B LBL labeled with CD45/R/B220 (25%, 2 of 8), PAX5 (100%, 8 of 8), CD43 (38%, 3 of 8), IgM (100%, 8 of 8), and IgK (0%, 0 of 8). One lymphoma of both B-LBL subtypes labeled with TDT. Unlike T-LBL where the incidence decreased with age (young 67%, 16 of 24; aging 3%, 1 of 34), the incidence of B-LBL was generally the same in young (29%, 7 of 24) and aging (24%, 8 of 34) mice.
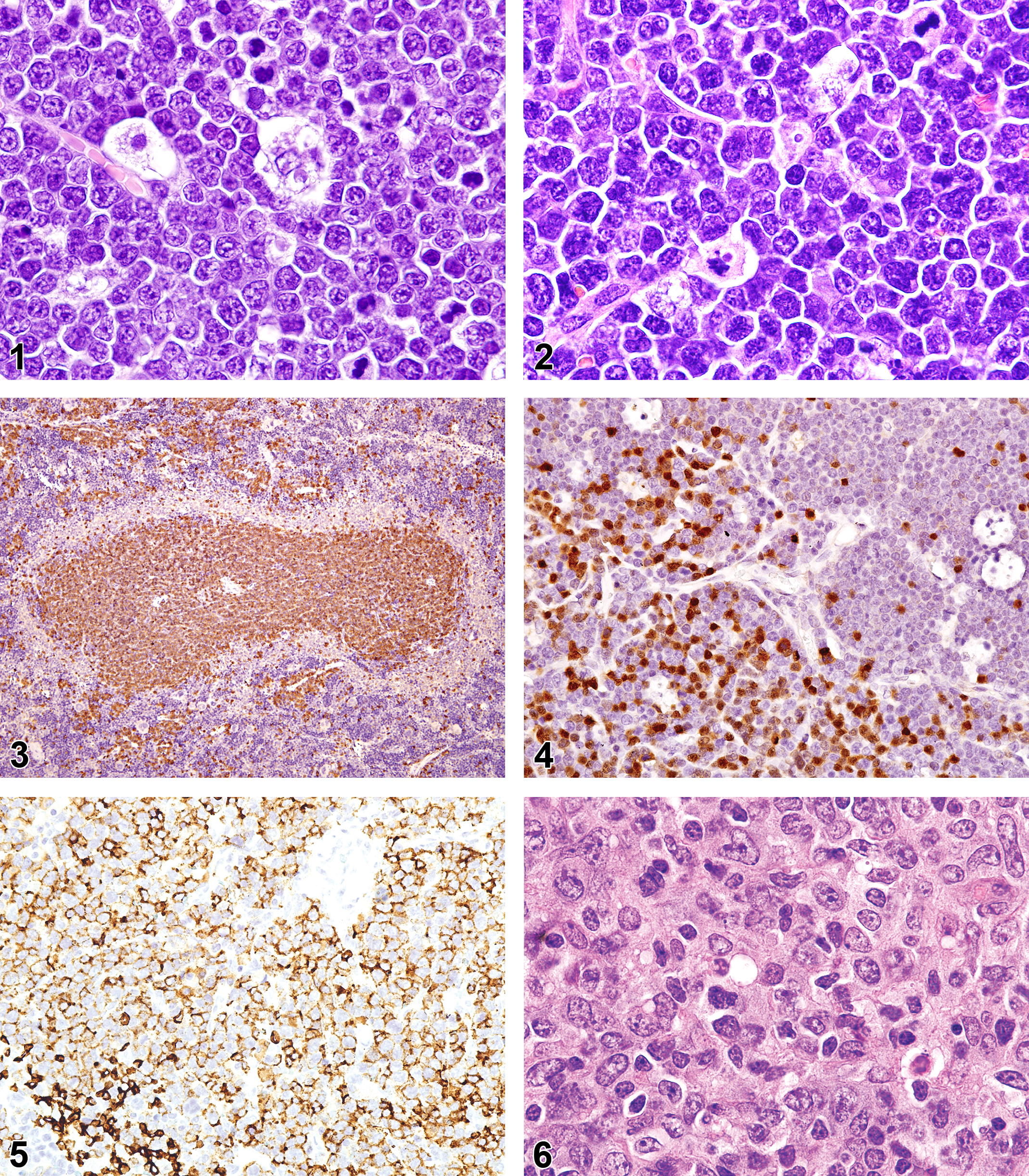
Lymphoblastic T-cell lymphoma in an 8-month-old sentinel mouse with a thymic mass. Note the uniform population of round cells with medium-sized nucleoli and starry sky macrophages. Hematoxylin and eosin.
Hematolymphoid neoplasm tissue distribution in female CD-1 mice.
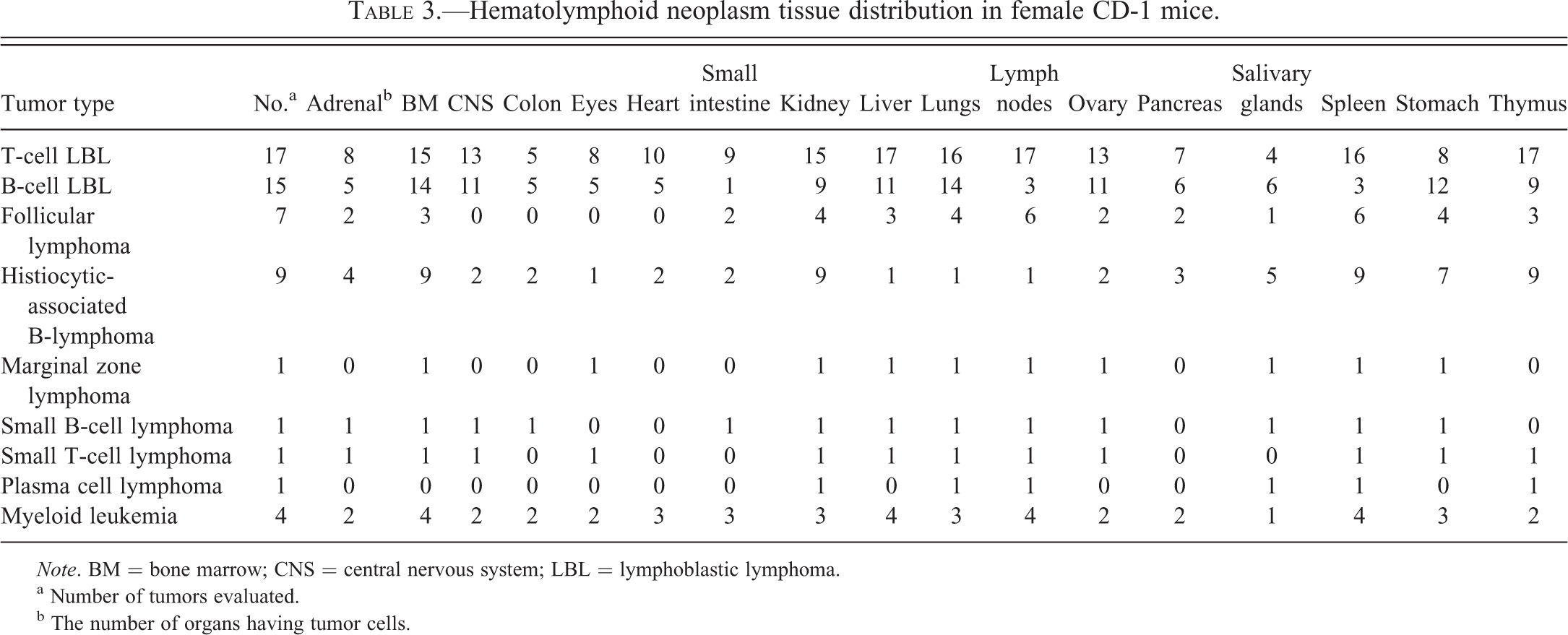
Note. BM = bone marrow; CNS = central nervous system; LBL = lymphoblastic lymphoma.
a Number of tumors evaluated.
b The number of organs having tumor cells.
Immunophenotype of hematolymphoid neoplasms in female CD-1 mice.

Note. BCL6 = B cell lymphoma 6; IBA1 = ionized calcium-binding adapter molecule 1; IgM = immunoglobulin M; IgK = kappa light chain immunoglobulin; IRF4 = Interferon regulatory factor 4; LYZ = lysozyme; MPO = myeloperoxidase; ND = not done; NK = natural killer cells; PALS = periarteriolar lymphoid sheath; PAX5 = paired box protein; PNA = peanut agglutinin; RUNX1/AML = runt related transcription factor 1/acute myeloid leukemia; Th2 = T-helper 2.
a Number of tumors evaluated.
b Number of tumors expressing antigen.
Follicular lymphoma was found in 7 mice aged more than 12 mo. The tumors involved the spleen, lymph nodes, and other tissues (Table 3). In the spleen, the neoplastic cells caused enlarged white pulp areas. The tumors were comprised of small, medium, and large cells of variable proportions with moderate cytoplasm (Figure 6). The small cells had round, irregular, or cleaved dense nuclei in the centrocytes. The medium and large tumor cells had vesicular nuclei with nucleoli juxtaposed to the nuclear membrane (centroblasts) or central/elongated prominent nucleoli (immunoblasts). Cleaved nuclei were often seen. Macrophages, plasma cells, and T lymphocytes in variable proportions were also commonly admixed among the neoplastic lymphocytes. The lymphoma cells were usually positive for PAX5 (100%, 7 of 7), CD45R/B220 (86%, 6 of 7), peanut agglutinin (86%, 6 of 7), and BCL6 (86%, 6 of 7) and less so for IgM (57%, 4 of 7) and IgK (71%, 5 of 7).
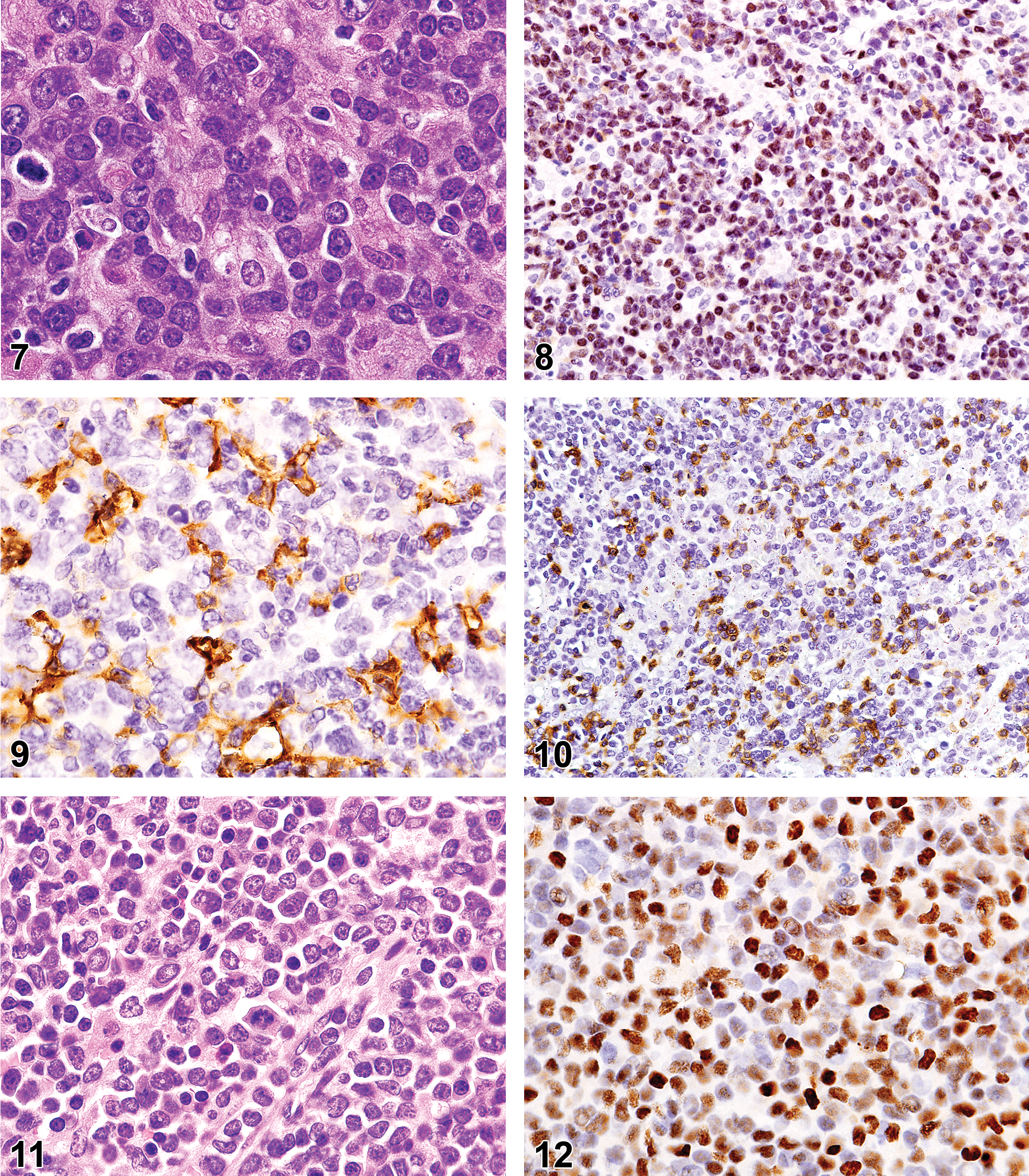
Histiocyte-associated B-cell lymphoma (HA-BCL) was diagnosed in 10 mice. The tumors were often found in several lymph nodes and spleen. Histologically, the lymphomas were composed of a mixture of immature lymphocytes with large to small nucleoli. A few cases mostly contained lymphoblasts. Interspersed along the immature lymphoid cells were histiocytic cells with abundant pale eosinophilic cytoplasm (Figure 7). The tumor lymphocytes were positive for PAX5 (100%, 9 of 9; Figure 8), CD45R/B220 (67%, 6 of 9), IgM (67%, 6 of 9), and IgK (33%, 3 of 9), while histiocytes were ionized calcium-binding adapter molecule 1 (IBA1) positive (9 of 9, 100%; Figure 9). Two (22%) of the 9 HA-BCLs had abundant infiltrating CD3+ T cells (Figure 10).
Histiocyte-associated B-cell lymphoma in a lymph node of a 21-month-old mouse. Note the pale large histiocytes with abundant eosinophilic cytoplasm among the round blastic lymphocytic tumor cells. Hematoxylin and eosin.
The marginal zone lymphoma was morphologically similar to the two small lymphocyte lymphomas, and the tumor was seen in the spleen, lymph nodes, and many other tissues (Table 3). However, the tumor cells were associated with the marginal zone of all the follicles, and they infiltrated into the red pulp bridging follicle to follicle. The tumor cells expressed PAX5 and IgM but not CD45R/B220.
Small lymphocyte lymphoma was only observed in two mice older than 18 mo of age. The tumors were characterized by small to medium lymphocytes with round condensed nuclei and a scant pale rim of cytoplasm. The tumor pattern was that of noncohesive cells with rare to a few mitotic figures. These tumors involved multiple lymphoid organs. With use of IHC and antibodies to T- and B-lymphocyte markers, the two tumors were further classified as a small cell lymphoma of T-cell lineage and a small cell lymphoma of B-cell lineage. The cells of the T-small cell lymphoma expressed CD3, CD43, and CD5, whereas the cells of the B-small cell lymphoma expressed PAX5, CD45R/B220, IgM, and IgK but not CD3.
Plasma cell lymphoma was observed in only one 21-month-old CRE mouse. The tumor involved multiple lymphoid organs and consisted of cells with abundant cytoplasm. The tumor cells had round nuclei and were arranged in noncohesive sheets. Morphologically, the cells could have been considered to be mature small lymphocytes, but with IHC the tumor cells were confirmed to be of plasma cell lineage. The tumor cells expressed the plasma cell markers CD43, CD138, and IRF4.
Myeloid leukemia developed in 4 mice aged 12 to 18 mo, with 75% occurring in CRE mice aged more than 12 mo. These neoplasms were composed of large immature myeloid cells often with ring forms (Figure 11). Tumor cells were usually positive for CD43 (100%, 4 of 4), myeloperoxidase (75%, 3 of 4), YM 1 (75%, 3 of 4), and RUNX1 (75%, 3 of 4; Figure 12), but only 1 was immunoreactive for lysozyme (25%, 1 of 4; Table 4). The tumors were often in many tissues: spleen, liver, lungs, lymph nodes, bone marrow (50%, 2 of 4), and other tissue (Table 3).
Histiocytic sarcoma was observed in 2 CRE mice aged 20 mo. The tumors consisted of infiltrates of monomorphic or pleomorphic histiocytes with abundant pink cytoplasm. The tumor cells had round, ovoid, and irregular vesicular nuclei with small nucleoli. Multinucleated giant cells were scattered among the neoplastic tumor cells. The tumors had a high mitotic index, and the tumors involved multiple organs. Although the tumor cells expressed IBA1, renal tubule hyaline droplets commonly associated with murine histiocytic sarcoma were observed only in the one HA-BCL case that had focal histiocytic sarcoma.
In SJCRH mice with lymphoma or myeloid leukemia, MuLV p30 antigen was found by IHC in tumor cells (5 of 10 cases studied; Figure 13) and in normal megakaryocytes in spleen or bone marrow (5 of 10; Figure 14). B-cell follicles in lymph node or spleen of a few mice showed p30 (Figure 15). MuLVp30 antigen was also found in salivary gland epithelium of 5 of 5 mice with lymphoma or in 1 mouse with myeloid leukemia (Figure 16), in pancreatic acinar cells of 7 of 7 mice but not in megakaryocytes of the 13-month-old normal CD1 mice, in tissues of five 4-week-old CD-1 mice nor in tissues of FVB mice, which have been reported to be free of MuLV (Teddessee-Heath et al. 2000; Morse 2007). Antigen was found in tumor cells of 3 of 9 CRE lymphoma cases and in megakaryocytes of 1 of 9 CRE mice with lymphoma.
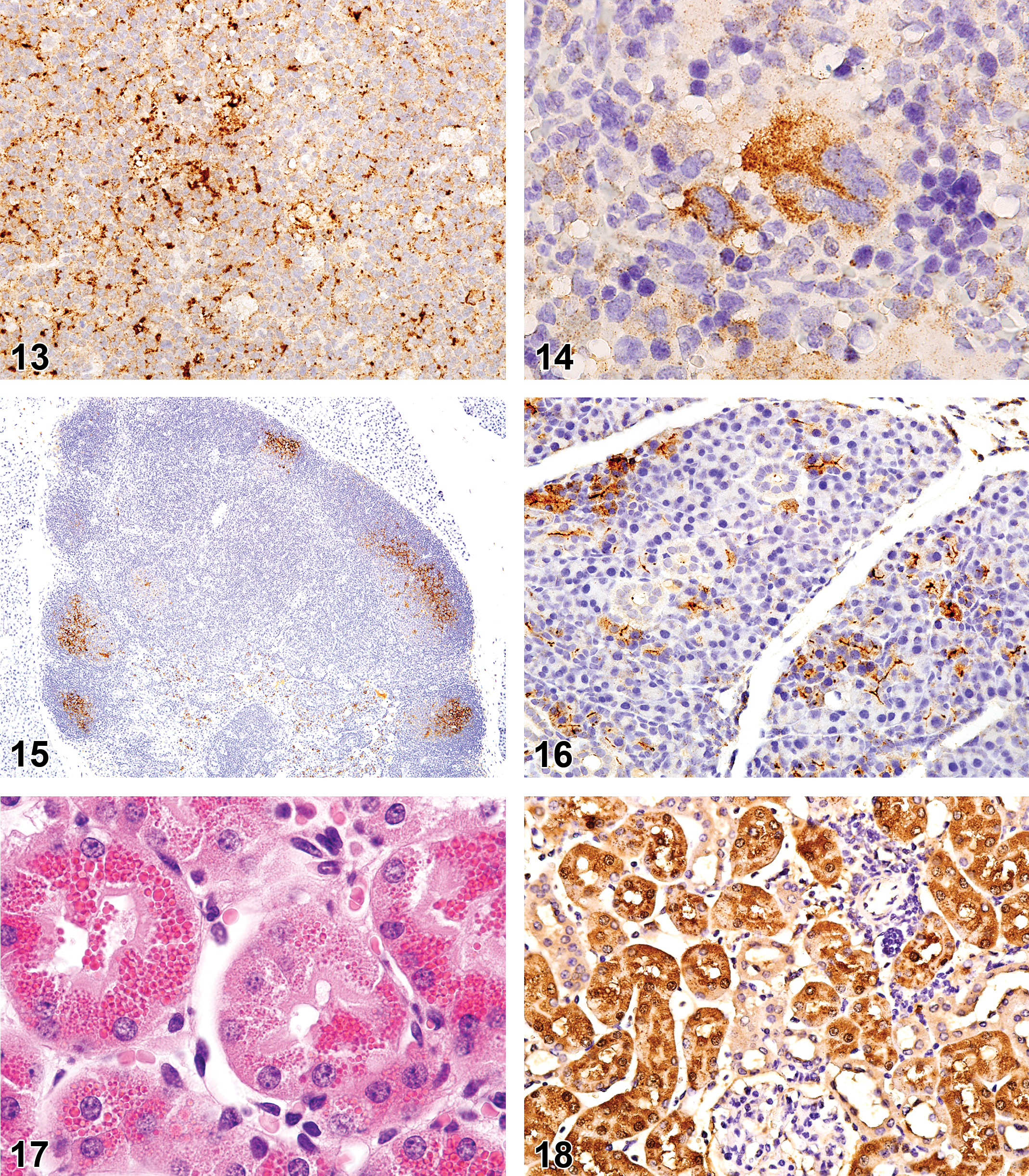
Rauscher leukemia virus (mouse leukemia virus) p30 in myeloid leukemia of a 12-month-old sentinel mouse.
Renal tubular hyaline droplets (Figure 17) were found in 6 of 9 CRE mice with HA-BCL but not in any SJCRH mice with any lymphoma or leukemia. One positive renal droplet case had both HA-BCL and focal histiocytic sarcoma. The renal droplets were positive for lysozyme in 5 of 5 mice (Figure 18). One CRE myeloid leukemia had hyaline droplets, which had strong immunoreactivity for lysozyme and a few granules with weak immunoreactivity for YM1.
Discussion
In 2004, it was reported that 17% of 101 CD-1 male and female mice less than 12 mo of age developed lymphoma (Son and Gopinath 2004). The morphologic features and cellular lineage of these lymphomas were not determined. In 2012, a report describes a range of hematolymphoid tumors of undetermined cellular lineage but with morphologic features similar to those in our report that were present in 355 control mice collected at the end of several 24-mo carcinogenicity studies (Bradley, Mukaratirwa, and Petersen-Jones 2012). Our data clearly demonstrate that with the use of IHC, the lymphoblastic lymphomas and small lymphocyte lymphomas that arise in young and old CD-1 mice consist of both T-lymphocyte and B-lymphocyte lineages. The data further demonstrate that the lymphoblastic lymphomas that occur in CD-1 mice age less than 12 mo are predominantly of T-lymphocyte lineage and that B-lymphoblastic lymphomas are equally prevalent in CD-1 mice aged less than 12 months and older than 12 months. This age and cellular distribution of lymphoblastic lymphoma in CD-1 mice is similar to that in human patients. T-cell lymphoblastic lymphoma is more prevalent than B-cell lymphoblastic lymphoma in pediatric patients (Cortelazzo et al. 2011) and has a higher rate of mediastinal tumor (Cortelazzo et al. 2011).
HA-BCL is found in variable proportions in congenic mice and recombinant inbred mice (Morse et al. 2001; Hao et al. 2010). It is uncommon in the most popular strains and stocks of mice, but our data suggest it is a common subtype of lymphoma in CD-1 mice. Other reports on lymphomas in CD-1 mice have not reported HA-BCL, but HA-BCL in these reports may be diagnosed under the collective term lymphoma or pleomorphic/follicular lymphoma (Decker et al. 2012; Bradley, Mukaratirwa, and Petersen-Jones 2012). Mechanisms for the involvement of apparent nonneoplastic histiocytes in these tumors are not known.
Infiltrating nonneoplastic T cells have been reported in mouse lymphomas and appear to be common in aging mice (Haines, Chattopadhyay, and Ward 2001). Human T-cell and histiocyte-rich, large B-cell lymphomas are considered an entity in the World Health Organization classification of human hematolymphoid tumors (De Wolf-Peeters et al. 2008). In this study, although the HA-BCLs have some similarity to the human T-cell/histiocyte-rich large B-cell lymphoma, the histiocyte and lymphocyte populations in the human T-cell/histiocyte-rich large B-cell lymphoma are far more extensive than either population in the mouse HA-BCLs.
T-cell lymphoblastic lymphomas in either the SJ or the CRE mice probably arose from thymic lymphocytes, as they often do in chemically induced thymic lymphomas or in genetically engineered mice (Ward 2006). B-cell lymphoblastic lymphomas in the study mice may have arisen from a B-cell progenitor and the small B-cell lymphoma from either a naive or an antigen experienced B cell in spleen or other lymphoid tissues. Follicular lymphoma and HA-BCL arise from germinal center B cells and diffuse large B-cell lymphomas from peripheral B cells of either germinal center or activated post-germinal center cells (Stein et al. 2008). Splenic marginal zone lymphomas in the mouse are of a B cell of an unknown stage of B-cell differentiation. The small T-cell lymphoma may have arisen from circulating T lymphocytes.
Renal hyaline droplets were first reported in rats and mice with histiocytic sarcomas (Hard and Snowden 1991) but recently were reported in CD-1 mice with lymphoma (Decker et al. 2012). The figure in the Decker report appears to be a lymphocytic lymphoma and not an HA-BCL. The CD-1 mice with lymphoma and renal hyaline droplets in the 2012 report had some evidence of Igs in tumor cells but no lysozyme in the droplets with their IHC studies being inconclusive (Decker et al. 2012). The cause of the renal droplets in mice with lymphoma that are not an HA-BCL or histiocytic sarcoma is not definitively known since they seem to be of tumor histiocyte origin. HA-BCLs show many histiocytes that are probably a source of lysozyme for renal accumulation as hyaline droplets, which others (Hard and Snowden 1991) showed contain lysozyme. Assuming the renal droplets in HA-BCL contain abundant lysozyme, we postulate, as has been postulated for histiocytic sarcoma, that the lysozyme accumulates in the renal tubules as a result of absorption of glomerular filtration fluid containing the enzyme released into the blood by the abundant histiocytes in the HA-BCL.
Renal hyaline droplets positive for lysozyme were also present in the renal tubules of 1 of the 4 myeloid leukemias. Cytoplasmic lysozyme droplets have been reported to accumulate in the renal tubules of Wistar-Furth rats injected intraperitoneally with rat myeloid leukemia (choroleukemic) cells (Klockars et al. 1974). YM1 and YM2 chitinase-like lectins have been reported to accumulate as eosinophilic droplets in renal tubules of a mouse with experimental myeloid leukemia (Marchesi et al. 2003; Marchesi et al. 2006). In the latter report, the renal tubular eosinophilic droplets coexpressed YM1 protein and lysozyme, but the myeloid tumor cells in this single case did not express lysozyme, suggesting the myeloid tumor cells were not the source of the lysozyme. In our study, in 3 of the 4 myeloid leukemias , the cells expressed YM1 (now CHIL3; Rehg, Bush, and Ward 2012; Table 1), but only the myeloid leukemia immunoreactive for lysozyme had renal tubular hyaline droplets that co-localized lysozyme and YM1, the former with strong intensity and the latter with slight positivity. To the authors’ knowledge, the present report is the first illustration of renal lysozyme-positive eosinophilic droplets associated with spontaneous mouse myeloid leukemia that reveal localization of lysozyme.
MuLV was first discovered in the 1950s (Gross 1951). Proviruses that encode infectious ecotropic and xenotropic MuLV are found in the genomes of some strains or stocks of mice but not others (Morse 2007). The MuLV genome is reported to be present in retired CD-1 breeder mice (Taddesse-Heath et al. 2000), but MuLV expression of major core proteins (p30 or p70) in the CD-1 mouse has not been previously reported. Ecotropic MuLV infection of adult mice has been shown to increase B-cell proliferation, serum IgM levels, and expression of transcripts associated with B-cell activation (Lee et al. 1995). Ecotropic MuLV has also been shown to induce a variety of lymphoma subtypes in outbred and inbred mice (Hartley et al. 2008). Our data suggest that the expression of MuLV protein in CD-1 mice should be considered in the interpretation of the use of CD-1 mice or their tissues in various toxicologic studies related to carcinogenesis, propagation of other viruses, biological responses to cell signaling, and RNA interference.
This study has characterized the subtypes and cell lineages of lymphomas that commonly develop in young and aging female CD-1 mice. We also demonstrated that renal hyaline droplets containing lysozyme may be seen in association with HA-BCLs and that the MuLV major core protein p30 is present in CD-1 lymphomas and normal tissues. The data also suggest that additional studies are warranted to determine the significance of the retroviral infection in CD-1 mice.
Footnotes
Author Contribution
All authors (JER, RR, DB, AB, JMW) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research in this study was partially supported by the National Cancer Institute of the National Institutes of Health under the Award Number P30CA021765. The work was also supported in part by the American Lebanese Syrian Associated Charities.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
