Abstract
To characterize the hepatic lesions in Fischer 344 (F344) rats afflicted with large granular lymphocyte (LGL) leukemia, the livers of rats with LGL leukemia at various stages were examined histopathologically and immunohistochemically. The morphologic features in the livers of rats afflicted with LGL leukemia were diffuse, uniform-sized, granular, or micronodular lesions consisting of hepatocytes showing centrilobular atrophy and perilobular hypertrophy (CAPH) without fibrosis. With progression in the stage of the LGL leukemia, the severity of the CAPH of hepatocytes increased resulting in fatty change and/or single-cell necrosis, along with compensatory hyperplasia of the hepatocytes, finally resulting in lesions similar to those seen in nodular regenerative hyperplasia (NRH) in the human liver. The CAPH of hepatocytes was a nonspecific tissue adaptation against ischemia or hypoxemia and/or imbalance in blood supply due to disturbance in the portal circulation and hemolytic anemia induced by the leukemia cells. In addition, direct and/or indirect hepatocellular injuries by leukemia cells were considered to be necessary for the formation of human NRH-like lesions. Morphogenetic investigation of the livers of rats afflicted with LGL leukemia may be helpful to clarify the pathogenesis of NRH in the human liver.
Introduction
Large granular lymphocyte (LGL) leukemia is extremely common in Fischer 344 (F344) and Wistar/Furth rats but rare in other strains (Tarone, Chu, and Ward 1981). Among F344 rats aged more than 20 months, LGL leukemia is the main cause of death (Stromberg and Vogtsberger 1983). The incidence of LGL leukemia shows a marginal, but statistical, increase due to some chemical treatment (Thomas et al. 2007). Therefore, risk assessment studies for carcinogenicity using F344 rats are viewed with some suspicion by the scientific community and regulatory agencies in the United States because of the unknown mechanism of tumorigenesis and variability of the historical control data on its incidence (Thomas et al. 2007).
The primary lesion of LGL leukemia originates in the spleen (Stromberg and Vogtsberger 1983) and not in the bone marrow, even though this neoplastic disease is referred to as a leukemic disorder. Splenomegaly is the most common characteristic macroscopic finding in rats afflicted with LGL leukemia (Stromberg and Vogtsberger 1983). Histopathologically, LGL leukemia cells accumulate in the marginal zone of the spleen at an early stage and the affected spleen is also often congested simultaneously (Losco and Ward 1984). The diagnosis of LGL leukemia is difficult in the early stage because of the indistinguishable morphology of the cells from the normal cell components and the small number of leukemia cells. However, the presence of leukemia cells in the hepatic sinusoids is very helpful in the diagnosis of LGL leukemia in the early stage (Dunnick et al. 1989).
Nodular regenerative hyperplasia (NRH) of the liver is a benign condition occurring at a high incidence in humans characterized by the presence of nodular hepatocellular lesions, and the incidence is not related to the gender (Armed Forces Institute of Pathology [AFIP] 2001). Histologically, the nodules are distributed diffusely throughout the liver and consist of hepatocytes showing centrilobular atrophy and periportal hypertrophy (CAPH) without fibrosis (AFIP 2001). These same histopathological findings are also observed in the livers of rats with LGL leukemia. Human NRH is known to be associated with a variety of conditions such as rheumatic diseases, myeloproliferative disorders, and chronic venous congestion (AFIP 2001). However, the pathogenesis of this hepatic disorder remains unclear despite its high incidence, and many years passed since the lesions were first recognized. Heterogeneity in the hepatic blood supply, arising from focal obliteration of the portal vein branches and compensatory augmentation of the arterial blood supply, has been suspected as being involved in the pathogenesis (Wanless 2002). Wanless insisted that NRH is a secondary and nonspecific tissue adaptation to heterogeneous distribution of the blood flow and does not represent a specific entity (Wanless et al. 1980; Wanless 1990). In fact, Weinbren and Washington established an experimental rat model of diffuse nodular hyperplasia in the liver, similar to NRH, by performing portacaval anastomosis (Weinbren and Washington 1976).
LGL leukemia cells are known to preferentially infiltrate the liver (Stefanski, Elwell, and Stromberg 1990), and the livers of rats with LGL leukemia show a micronodular or granular appearance macroscopically (Eustis et al. 1990) as well as enlargement in the later stages (Stromberg and Vogtsberger 1983). Despite the disease being well known, the detailed histopathologic characteristics of the hepatic changes have not been described in the literature, and the morphogeneses of the hepatic enlargement and nodularity are still unclear. There is only one textbook in which LGL leukemia is recognized as an underlying cause of NRH in the F344 rat liver (Eustis et al. 1990). This type of hepatocellular hyperplasia has never reported in detail and it is different entity from nonregenerative and regenerative hepatocellular hyperplasia (Thoolen et al. 2010), in that it has a diffuse distribution.
CAPH of hepatocytes is a common histopathological finding in both human NRH and NRH-like lesions in rats with LGL leukemia, and the histopathological features suggest that hypoxia plays a role in its pathogenesis. Therefore, we conducted histopathological examination of the extrahepatic portal vein as well as intrahepatic portal veins to certify the possible cause of the intrahepatic circulatory disturbance in NRH-like lesions. In addition, to investigate the role of hypoxemia in the pathogenesis of the NRH-like lesions in F344 rats, we also examined the livers of rats with severe extramedullary hematopoiesis (EMH) in the spleen secondary to persistent anemia. We describe the histopathological characteristics of the hepatic lesions in rats afflicted with LGL leukemia, which are similar to the lesions in human NRH, and tried to clarify the pathogenesis of the NRH-like lesions in the rat liver modified by the stage of progression of the LGL leukemia. In addition, the morphological and biological changes of the leukemia cells with progression of the disease were also investigated.
Materials and Methods
Tissue samples were obtained from both male and female F344/Du rats (Charles River Laboratories Japan, Atsugi, Japan) used in 6 of the 2-year carcinogenicity studies (studies A, B, C, D, E, and F) performed in our laboratory. Animals were mainly nontreatment control animal. In case of treated animals, their histological features were certified to be comparable to those of control animals. Moreover, animals with moderate to marked chronic progressive nephropathy or vascular abnormalities were not used for examination, even control animals.
The present study consisted of 3 segments (segment 1 from study A, segment 2 from studies B and C, and segment 3 from studies D, E, and F), and details of the examinations in each segment are shown in Table 1. In segments 1 and 2, LGL leukemia in the study materials was diagnosed according to the presence of histologic characteristics such as congestion and lymphoid depletion in the spleen and the presence of leukemic cells in the hepatic sinusoids in addition to cytological characteristics such as azurophilic cytoplasmic granules and erythrophagocytosis of leukemic cells on blood smears, although there are several cases unable to differentiate LGL leukemia from T-cell lymphoma without verification for the LGL marker, OX-8 (Hayashi, Nonoyama, and Miyajima 1989). Also, LGL leukemia in segment 3 materials was diagnosed based on the macroscopic necropsy findings, such as hepatosplenomegaly and the rough surface of the liver.
Summary of study design.

Note: D = dead; M = moribund sacrificed; S = schedule sacrificed; LGLL = large granular lymphocyte leukemia.
aStaging by Dunnick et al. (1989).
bFull organs/tissues in the carcinogenicity study.
The staging of the LGL leukemia was according to the following criteria. LGL leukemia was divided into 3 stages according to the severity (Dunnick et al. 1989). Stage 1 (early stage): The spleen was almost intact in size or slightly enlarged with infiltration by small numbers of neoplastic LGL lymphocytes, with or without slight congestion in the red pulp; very few neoplastic cells were found in the liver sinusoids and no identifiable neoplastic cells were found in other organs. Stage 2 (intermediate stage): The spleen was moderately enlarged with moderate to large numbers of neoplastic cells in the red pulp; the splenic architecture, including germinal centers and periarterial lymphoid sheaths, remained intact; minimal to moderate involvement of the liver; aggregates/masses of neoplastic cells generally limited to the blood vessels of the spleen and liver, but neoplastic cells could sometimes be detected in other organs. Stage 3 (late stage): The disease process is advanced with multiple organ involvement; the liver was moderately to markedly enlarged and nodular; variable degenerative changes of the hepatic parenchyma associated with the accumulation of neoplastic cells; accumulation of neoplastic cells in other organs, including lungs, lymph nodes, kidneys, brain, and adrenal glands.
Archival 10% neutral buffered formalin-fixed, paraffin-embedded organs and tissues from 3 carcinogenicity studies (studies A, B, and C) at Biosafety Research Center (BSRC) were used. The liver tissues were trimmed from all lobes, that is, the left, medial, right, and caudate lobes. The extrahepatic portal veins used in segment 3 were sampled at the time of necropsy of each animal from 3 carcinogenicity studies (studies D, E, and F) processed routinely and embedded in paraffin. All sections were cut at 5 μm from paraffin-embedded blocks and stained with hematoxylin and eosin (HE).
The procedures for handling and care of the animals used in the present study were approved by the Animal Care Committee of BSRC.
Segment 1: Histopathologic Examination of the Liver and Correlations among the Parameters
We examined the histological characteristics of the hepatic lesions of rats with various stages of LGL leukemia and the effects exerted by the leukemia cells on the liver with progression of the disease. In addition, measurement of spleen size was done to know the relationships between spleen size and NRH development or stage of LGL leukemia and the mitosis of hepatocytes were counted to certify whether the NRH-like lesions in F344 rats are proliferative lesions or not.
The spleen enlargement was graded by the length of long axis as follows: none no enlargement; minimal, less than 49 mm; slight, 50–59 mm; moderate, 60–69 mm; and marked, more than 70 mm. Moreover, the number of mitotic hepatocytes in 50 high-power fields (HPFs) of the left lobe from these cases was also counted to evaluate the proliferative activities of the hepatocytes in NRH-like lesions. The liver tissues showing no remarkable changes from 10 each of schedule-sacrificed males and females in study A were used as controls for the number of mitotic figures of the hepatocytes.
The mitosis of hepatocytes was graded by the number of mitosis per 50 HPFs in the liver tissue as follows: no increase, less than 3; minimal, 4 to 10; slight, 11 to 30; moderate, 31 to 50; and marked, more than 51. The livers, other full organs, and tissues obtained in the 1 carcinogenicity study (study A) from a total of 48 male and female rats with various stages of LGL leukemia were examined histopathologically, and the correlations among the spleen size, stage of LGL leukemia, development of NRH-like lesions, and the morphology and biological behaviors of the leukemia cells were investigated. The correlation between the stage of LGL leukemia, spleen size, and development of NRH-like lesion was analyzed statistically by 1-sided Cochran–Armitage trend test for tendency of both parameters and 1-sided Fisher’s exact test for difference in incidence of each parameter.
Segment 1: CAPH of Hepatocytes in the Livers of Rats without LGL Leukemia
CAPH of hepatocytes, which is a characteristic histopathologic finding in the livers of rats with LGL leukemia, is also observed in the livers of rats without LGL leukemia. Therefore, full organs/tissues in carcinogenicity studies from 9 nonleukemia rats showing CAPH of the hepatocytes from study A were examined histopathologically and the results were used to clarify the pathogenesis of the NRH-like lesions in the LGL leukemia rat liver.
Segment 2: Histochemistry and Immunohistochemistry
The livers from the rats with stages 2 and 3 of LGL leukemia were examined histochemically and immunohistochemically to clarify the differences and similarities between NRH in the human liver and the NRH-like lesions in the livers of the rats afflicted with LGL leukemia.
Representative liver sections from 20 rats afflicted with stage 2 or stage 3 LGL leukemia from 2 carcinogenicity studies (studies B and C) were stained by special histochemical methods, that is, the Berlin blue reaction for iron, Schmorl reaction for lipofuscin, Watanabe’s silver impregnation for reticular fibers, elastica van Gieson stain for elastic fibers, and Azan–Mallory stain for collagen fibers. The spleen and adrenal gland tissues from aged F344 rats were used as positive controls for histochemistry of iron and lipofuscin, respectively. Also, using the liver section of aged F344 rats it was certified that reticular, collagen, and elastic fibers were stained appropriately.
Immunohistochemical staining of representative sections by the labeled streptavidin–biotin method (LSAB kit; Dako A/S, Glustrup, Denmark) was also performed using several antibodies such as antihuman α-smooth muscle actin (α-SMA; Dako A/S), anti-porcine vimentin (Dako A/S), antihuman factor VIII–related antigen (FVIII-RAg; Dako A/S), anti-rat macrophage (clone: anti-CD8α antibody; Serotec Ltd., Oxford, England), and anti-rat proliferating cell nuclear antigen (PCNA; Dako A/S). Pretreatments were performed by trypsin digestion for FVIII-RAg, pepsin digestion for ED-1, and microwave oven heating for PCNA. Immunoreaction with the primary antibodies was carried out for 12 hr at 4°C. Thereafter, the following procedures were carried out according to the manufacturer’s instruction. Internal tissues in the liver were used as positive controls, that is, vascular endothelial cells except for sinusoids for FVIII-RAg, vascular muscle cells for α-SMA, and Kupffer cells for vimentin and macrophages. For PCNA immunostaining, spleen from 10-week-old rat was used as a positive control, and the appropriateness of immunostaining was certified by specific, high positivity in lymphocytes of germinal centers. Also, the livers from 2 age-matched, nonleukemia rats were used as controls. Sections minus primary antibodies were used as the negative controls.
For precise evaluation of the proliferative activities of the hepatocytes, the number of mitotic hepatocytes was compared with the PCNA labeling index (LI). PCNA LI was calculated based on the number of PCNA-positive hepatocytes per 1,000 hepatocytes in the left lobe that was counted manually.
Segment 3: Histopathologic Examination of the Extrahepatic Portal Veins
The extrahepatic portal veins from the rats with advanced LGL leukemia were examined histopathologically to confirm whether the imbalance in the intrahepatic blood flow due to the circulatory disturbance in the portal vein possibly played an important role in the pathogenesis of the NRH-like lesions in the rats with LGL leukemia as in NRH in the human liver (Wanless et al. 1980; Wanless 1990).
Representative transverse sections of the extrahepatic portal veins that did not show any macroscopic lesions were examined histologically and the materials were obtained from 30 rats that were diagnosed with advanced LGL leukemia by macroscopic examination based on gross findings such as hepatosplenomegaly and a rough surface of the liver. The cross sections were made at their central part of the whole of the extrahepatic portal vein sampled.
Results
The typical and most common macroscopic findings in the rats afflicted with late stage of LGL leukemia include marked enlargement of the spleen and liver along with yellowish discoloration of the abdominal and subcutaneous connective tissues due to jaundice (Figure 1). In addition, the surface of the enlarged liver often showed a granular appearance caused by the diffusely distributed uniform-sized micronodules (Figure 1).
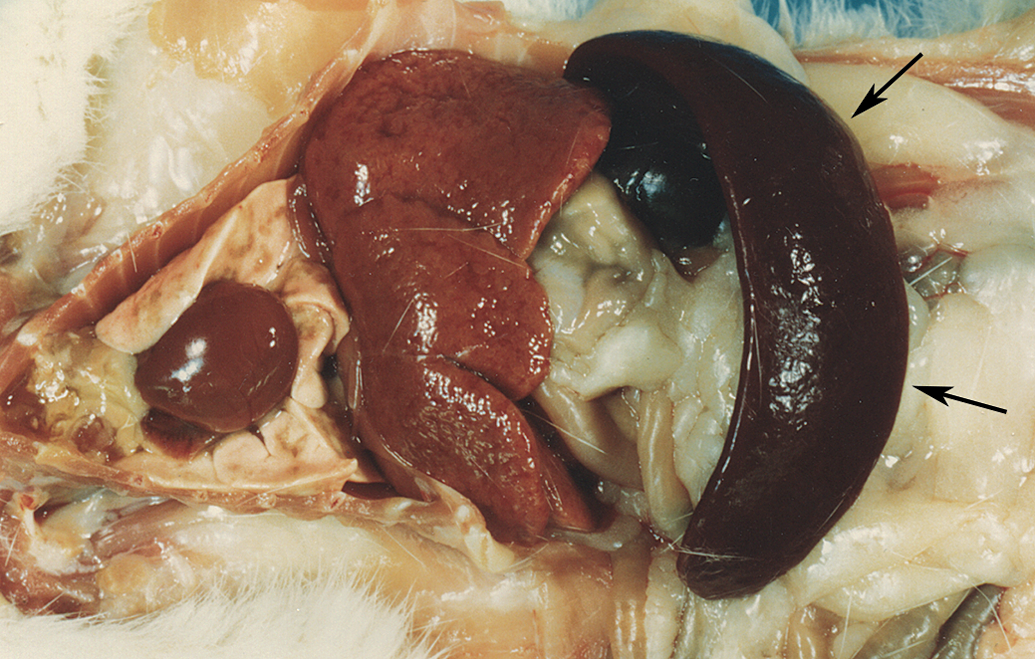
Typical gross appearances of the internal organs of a rat afflicted with LGL leukemia at necropsy. Hepatosplenomegaly is evident. The spleen shows a dark red congested appearance (arrows). The liver shows granular or micronodular surface. LGL = large granular lymphocyte.
Segment 1: Histopathology of the Liver of the Rats Afflicted with LGL leukemia at Various Stages
The spleen size, development of NRH-like lesion, number of mitosis of hepatocytes, and the major histopathologic findings of the liver of rats with LGL leukemia at various stages are presented in Tables 2 and 3. The number of LGL leukemia rats at stages 1, 2, and 3 was 6 (2 males and 4 females), 19 (4 males and 15 females), and 23 (12 males and 11 females), respectively. All rats at stage 1 were schedule sacrificed and their spleen showed no or minimal enlargement (less than 45 mm in length at long axis). At stage 2, there were 13 schedule-sacrificed rats and 6 dead or moribund-sacrificed rats of age 93 weeks or more, and 6 animals did not show splenic enlargement. At stage 3, there were 4 schedule-sacrificed rats and 19 dead or moribund-sacrificed rats of age 66 weeks or more and spleen enlargement was observed in all rats. In the age-matched control rats without LGL leukemia, no remarkable histopathologic changes were observed in the liver and the number of mitotic figures of hepatocytes/50 HPFs ranged from 0 to 3 in all the rats.
Summary of spleen size, development of nodular regenerative hyperplasia, and number of mitosis of hepatocytes in the liver of rats bearing LGL leukemia at various stages.

Note: HPFs = high-power fields; LGLL = large granular lymphocyte leukemia; NRH = nodular regenerative hyperplasia.
aStaging by Dunnick et al. (1989).
b± = ≤49; + = 50–59; ++ = 60–69; +++ = ≥70.
c− = not present; + = present.
d− = ≤3; ± = 4–11; + = 10–30; ++ = 31–50; +++ = ≥51.
Positive tendency by Cochran–Armitage trend test when compared between less than 49 mm group and more than 50 mm group: ### p < .001.
Significantly different from the stage 1 + NRH present group by Fisher’s test (1 sided): ***p < .001.
Summary of histopathologic findings in the liver of rats bearing LGL leukemia at various stages.
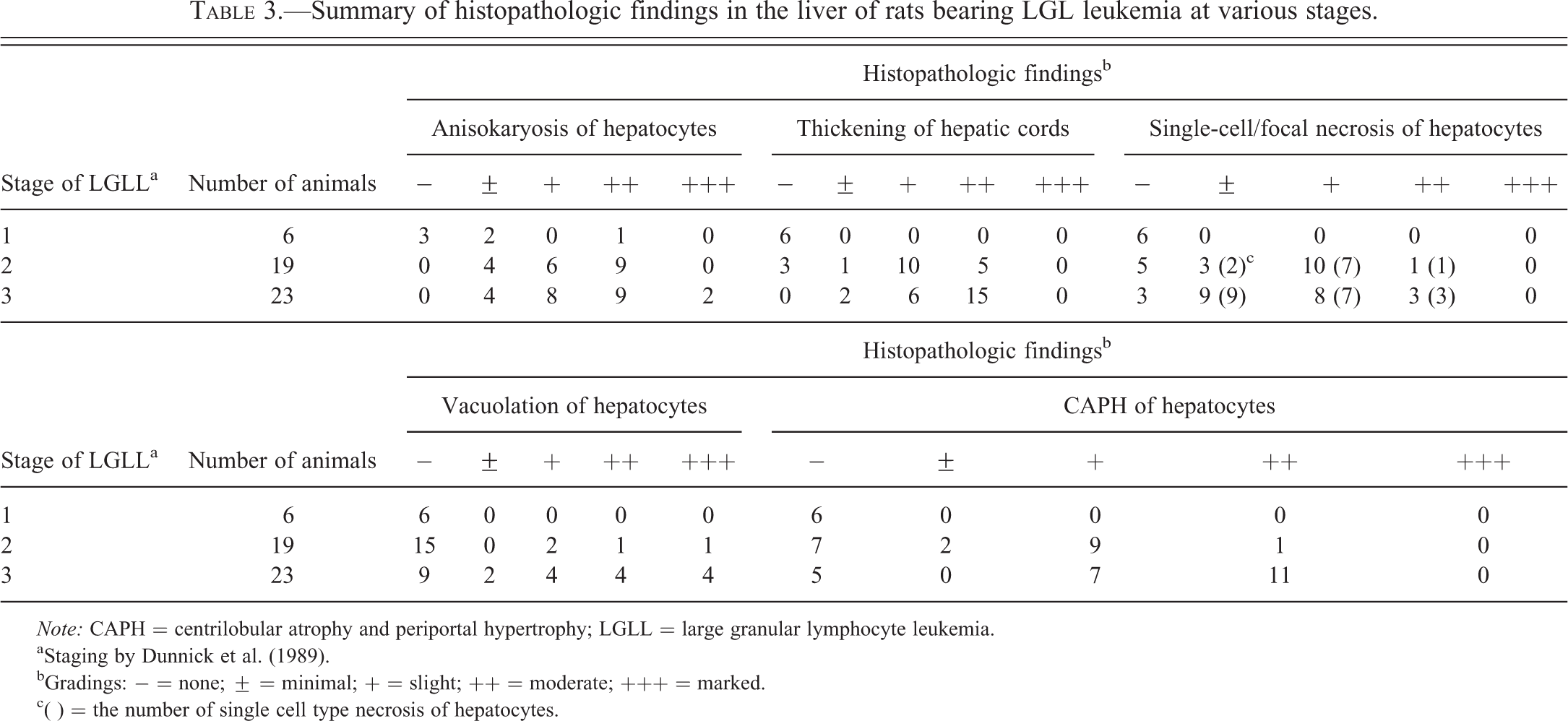
Note: CAPH = centrilobular atrophy and periportal hypertrophy; LGLL = large granular lymphocyte leukemia.
aStaging by Dunnick et al. (1989).
bGradings: − = none; ± = minimal; + = slight; ++ = moderate; +++ = marked.
c( ) = the number of single cell type necrosis of hepatocytes.
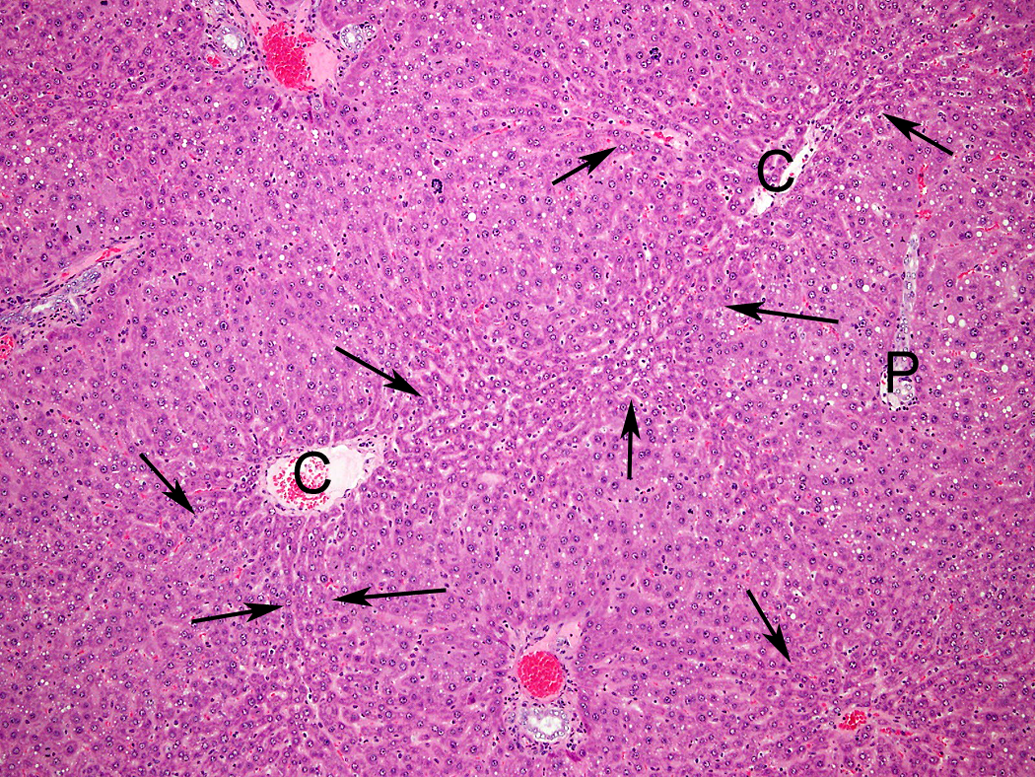
In the animals with stage 1 LGL leukemia, the histopathologic findings in the liver were mainly limited to the appearance of LGL leukemia cells in the sinusoids, increased number of mitoses in hepatocytes, and/or anisokaryosis. In the animals with stage 2 LGL leukemia, the CAPH of hepatocytes without fibrosis (Figure 2) was observed in the liver of 12 of the 19 cases. Translobular atrophic cords consisting of periacinar (centrilobular) hepatocytes were also seen, resulting in reconstruction of hepatic lobules accompanied by hypertrophic periportal hepatocytes. These reconstructed lobules corresponded to the macroscopic micronodular appearance, that is, the so-called nodules in human NRH. In addition, proliferative and hypertrophic changes in the hepatocytes as demonstrated by anisokaryosis, increased number of mitoses in hepatocytes (Figure 3A), and/or thickening of the hepatic cords were consistently observed in all or almost cases, which are accompanied by single-cell and/or focal necrosis (14 cases) and/or vacuolation in the centrilobular hepatocytes (4 cases). Vascular changes such as focal thickening of the walls of the portal veins (Figure 3C and 3B: age-matched, nonleukemia control rat liver) and infiltration of the subendothelial intimal tissue of the portal veins by leukemic cells (Figure 3D) were observed in some cases. In the animals with stage 3 LGL leukemia, several changes constituting the hepatic lesions those with stage 2 disease, such as CAPH, anisokaryosis of hepatocytes, and thickening of the hepatic cords were much more pronounced. Anisokaryosis and thickening of the hepatic cords were observed in all cases examined (23 cases). Moreover, single-cell necrosis of hepatocytes (19 cases; Figure 3E) and hepatocytes engulfing apoptotic bodies in the centrilobular area were frequently observed in the absence of any inflammatory changes. Mitotic figures in hepatocytes (Figure 3F) were increased in number along with marked thickening of the hepatic cords (Figure 3G) and the nodular appearance of the liver became more conspicuous in the advanced stage of the disease. However, even in cases with advanced disease, the nodules were not fused and individual nodules were uniform in size and distributed diffusely. In the portal area, leukemic cell emboli in the lymphatic vessels (Figure 3H), diffuse thickening of the portal veins (Figure 3I), or thrombi in the portal vein (Figure 3J) were infrequently detected in cases with advanced disease. Most of the injured portal veins were located in the middle and left lobes, while the hepatic arteries were completely intact in almost all cases. The CAPH of hepatocytes without fibrosis was detected in the liver in 18 out of 23 cases. Brown pigment-containing Kupffer cells were evident in the livers of rats with moderately thickened hepatic cords and the number of rats with such cells in the liver increased as the disease stage progressed (stage 2: 1 rat; stage 3: 9 rats).
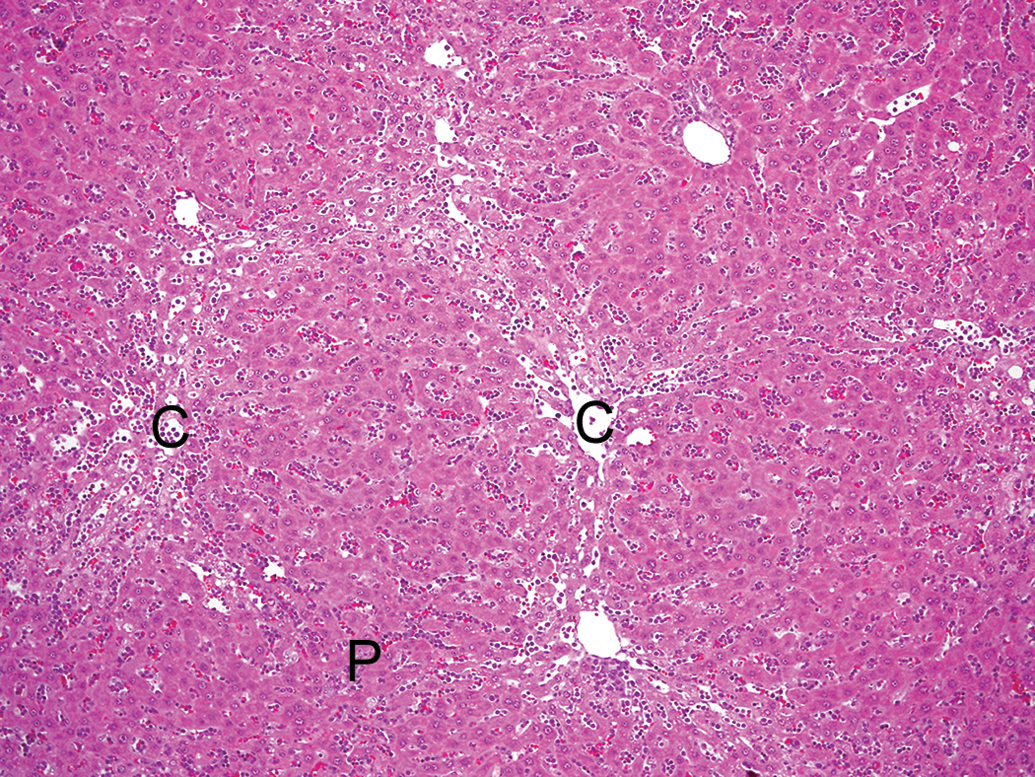
Typical histological appearance of an NRH-like lesion in the liver. Micronodules are distributed diffusely throughout the lobe, and no fibrosis is seen. The nodules consist of hepatocytes showing centrilobular atrophy and periportal hypertrophy. C = central veins; P = portal areas, HE stain, ×100. NRH = nodular regenerative hyperplasia.
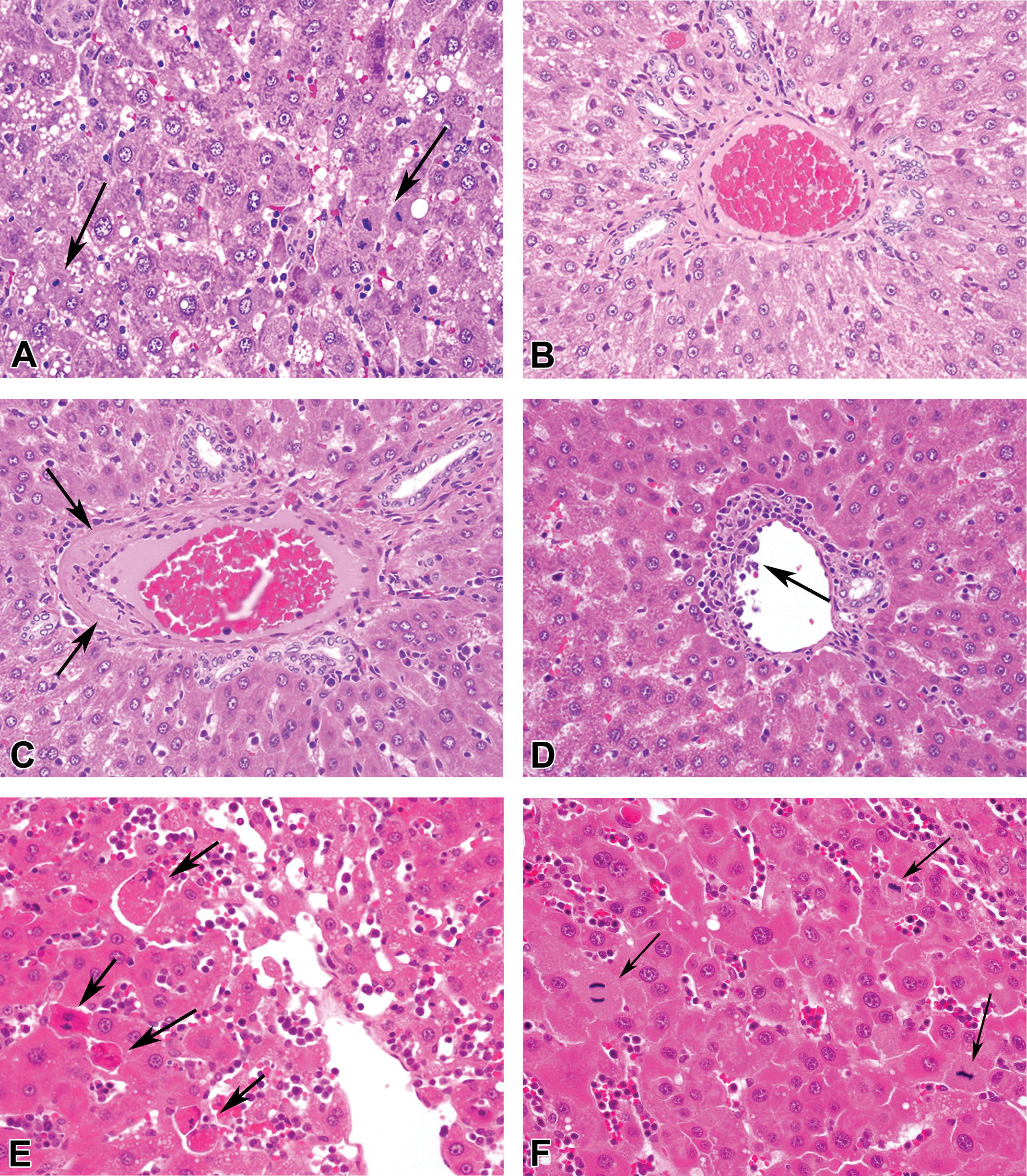
Histopathological findings in the liver. (A) Anisokaryosis and mitoses (arrows) in hepatocytes, ×400. (B) Age-matched, nonleukemia rat for comparison. No thickening of the portal vein, ×400. (C) Focal thickening of the wall of the portal vein (arrows), ×400. (D) Leukemic cell infiltration into the subendothelium of the portal vein, ×400. (E) Single-cell necrosis of centrilobular hepatocytes (arrows), ×400. (F) Mitosis in hepatocytes (arrows), ×400. (G) Thickening of the hepatic cords, ×200. (H) Leukemic cell embolus in a lymphatic vessel (arrows), ×200. (I) Diffuse thickening of the wall of the portal vein (arrows), ×200. (J) Thrombus formation in the portal vein (arrows), ×200. HE stain. Liver from (A) age-matched, nonleukemia rat; (B–D) rats with LGL leukemia, stage 2; (E–J) rats with LGL leukemia, stage 3. LGL = large granular lymphocyte.
While all of the leukemia cells in stage 1 of the disease showed typical cytological features such as round, oval, or reniform nuclei with irregular nuclear membranes and scant cytoplasm, the cytological features in stage 3 cases were variable, ranging from typical to pleomorphic. The number of mitotic figures in the hepatocytes was correlated with the severity of the anisokaryosis, hypertrophy of the periportal hepatocytes, and/or thickening of the hepatic cords.
Segment 1: Correlation between Histopathology and Each Parameter
Between spleen size and the stage of LGL leukemia. The results are shown in Table 2. The correlation between the spleen size and the stage of LGL leukemia was statistically significant (p < .001) by Cochran–Armitage trend test. Moreover, using Fisher’s exact test it was reported that the number of rats with stages 2 and 3 LGL leukemia with a spleen size of more than 50 mm was significantly different from the rats with stage 1 LGL leukemia, wherein based on the spleen size, there were 2 groups, that is, those with a spleen size of less than 49 mm and those with more than 50 mm. In 2 cases showing no correlation, the spleen was less than 49 mm in size despite the disease stage being classified into stage 3, and marked pigment deposition in the Kupffer cells was seen in both cases.
Between NRH-like lesion development and the stage of LGL leukemia. The results are shown in Table 2. The correlation between the development of NRH-like lesions and the stage of the LGL leukemia was statistically significant (p < .001) by Cochran–Armitage trend test. The comparison of incidence of NRH-like lesion between stage 1 and stage 2 or 3 showed significant difference by Fisher’s exact test. The NRH-like lesions developed at stage 2 LGL leukemia, and there were no cases with these lesions among the animals with stage 1 LGL leukemia. Conversely, 5 cases with stage 3 LGL leukemia failed to show NRH-like lesions, but diffuse sinusoidal dilatation due to severe leukemia cell infiltration or unknown cause was observed in these cases.
Between spleen size and NRH-like lesion development. The results are shown in Table 4. The correlation between spleen size and the development of the NRH-like lesions was statistically significant (p < .001) by Cochran–Armitage trend test. The comparison of incidence of NRH-like lesion between spleen size shows a positive relationship and the results were statistically significant by Fisher’s exact test, wherein based on the spleen size, there were 4 groups, that is, group with a spleen size of less than 49 mm, group with 50–59 mm, group with 60–69 mm, and group with more than 70 mm. The spleen became larger and the liver began to show noticeable NRH-like lesion as the stage of the LGL leukemia advanced.
The correlation between NRH-like lesion development and spleen size.

Note: NRH = nodular regenerative hyperplasia; − = not present; + = present.
Positive tendency by Cochran–Armitage trend test: ### p < .001.
Significantly different from group less than 49 mm by Fisher’s test (1 sided): *p < .05, ***p < .001.
Segment 1: CAPH of Hepatocytes in the Livers of Rats without LGL Leukemia
CAPH of hepatocytes was also observed in the livers of 9 rats without LGL leukemia (Figure 4). All of the 9 rats showed severe EMH in the spleen with large tumors (30–100 mm in length at long axis) or abdominal mesothelioma, and most of the animals were moribund-sacrificed or died. The degree of nodularity in the liver of the rats with EMH in the spleen was milder than that in rats afflicted with LGL leukemia. Single-cell necrosis of the centrilobular hepatocytes was not prominent in these rats, although the focal necrosis of hepatocytes was comparable to that seen in the rats afflicted with LGL leukemia. In addition, the hepatocytes showed lower proliferative activities compared to the livers of rats afflicted with LGL leukemia. No anisokaryosis or increased mitoses in hepatocytes was noted in the livers of 8 rats. On the other hand, centrilobular and/or multifocal necrosis of hepatocytes was observed in 4 rats. The number of mitotic figures in the hepatocytes of 8 rats was comparable to that of the control animals. The cause of anisokaryosis and of increased mitoses in the hepatocytes of each rat remains unknown.
Centrilobular atrophy (arrows) and periportal hypertrophy of hepatocytes in the liver of a rat with severe splenic extramedullary hematopoiesis. C = central vein; P = portal area; HE stain. ×100.
Segment 2: Histochemistry and Immunohistochemistry
Hemosiderin-laden macrophages accumulated in areas of atrophy and necrosis bridging neighboring central veins (Figure 5A) and were also observed in the sinusoids. Almost all pigment granules deposited in the liver, especially in the Kupffer cells and monocytes, were positive for Berlin blue and were identified as hemosiderin. Small amounts of lipofuscin granules were also detected in the hepatocytes by Schmorl reaction. Reticular fibers formed a dense network in areas where the centrilobular hepatocytes had collapsed and been lost from, however, there was no increase in collagen fibers, elastic fibers, or reticular fibers in any region of the liver, even in the advanced cases (Figure 5B).
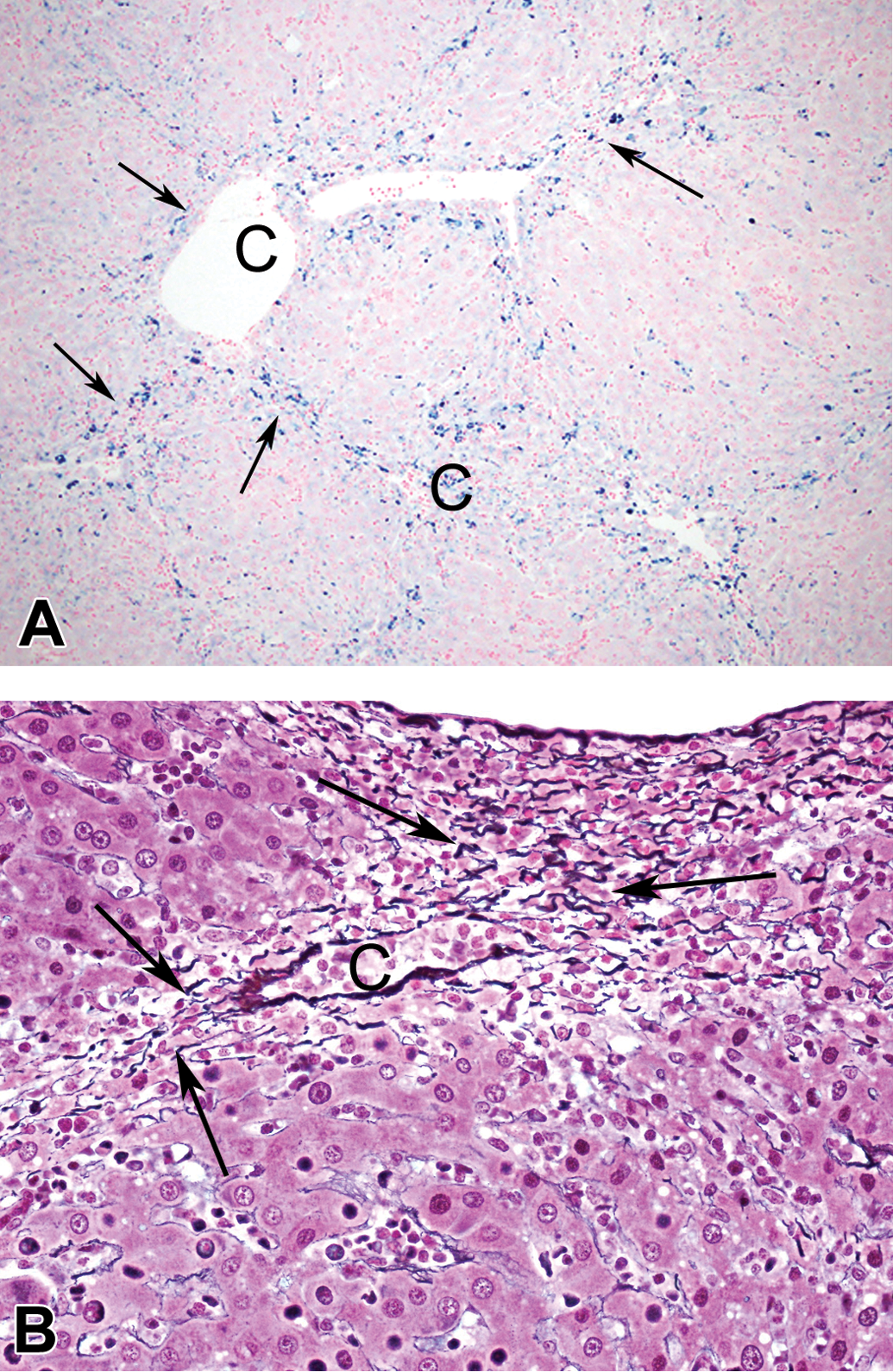
(A) Accumulation of hemosiderin-laden macrophages in areas bridging neighboring central veins (arrows). C = central vein, Berlin blue reaction, ×100. (B) Aggregation of reticulin fibers around the central vein (arrows), with collapse of the centrilobular hepatocytes. C = central vein. Watanabe’s silver impregnation, ×200.
The results of immunohistochemistry were summarized in Table 5. α-SMA was expressed in the areas where the reticular fibers formed a dense network (Figure 6A) or in the sinusoids of the nodules (Figure 6B). Similarly, FVIII-RAg was demonstrated in some sinusoids within the nodules (Figure 6C). However, both the expressions of α-SMA and FVIII-RAg in the sinusoids were limited in some of the advanced cases. ED-1-positive cells were increased in number in areas bridging the neighboring central veins but were rare within the nodules in some advanced cases (Figure 6D), although ED-1-positive cells were regularly detected in the hepatic sinusoids in the cases with relatively early stages of the disease. Vimentin was expressed in the leukemia cells, sinusoidal lining cells, monocytes and lymphocytes within the sinusoids, and proliferating bile duct epithelial cells but not in the hepatocytes. PCNA-positive hepatocytes were more frequent in the livers of cases showing anisokaryosis, increased mitoses in hepatocytes, and thickening of the hepatic cords than in those of cases without these findings and controls.
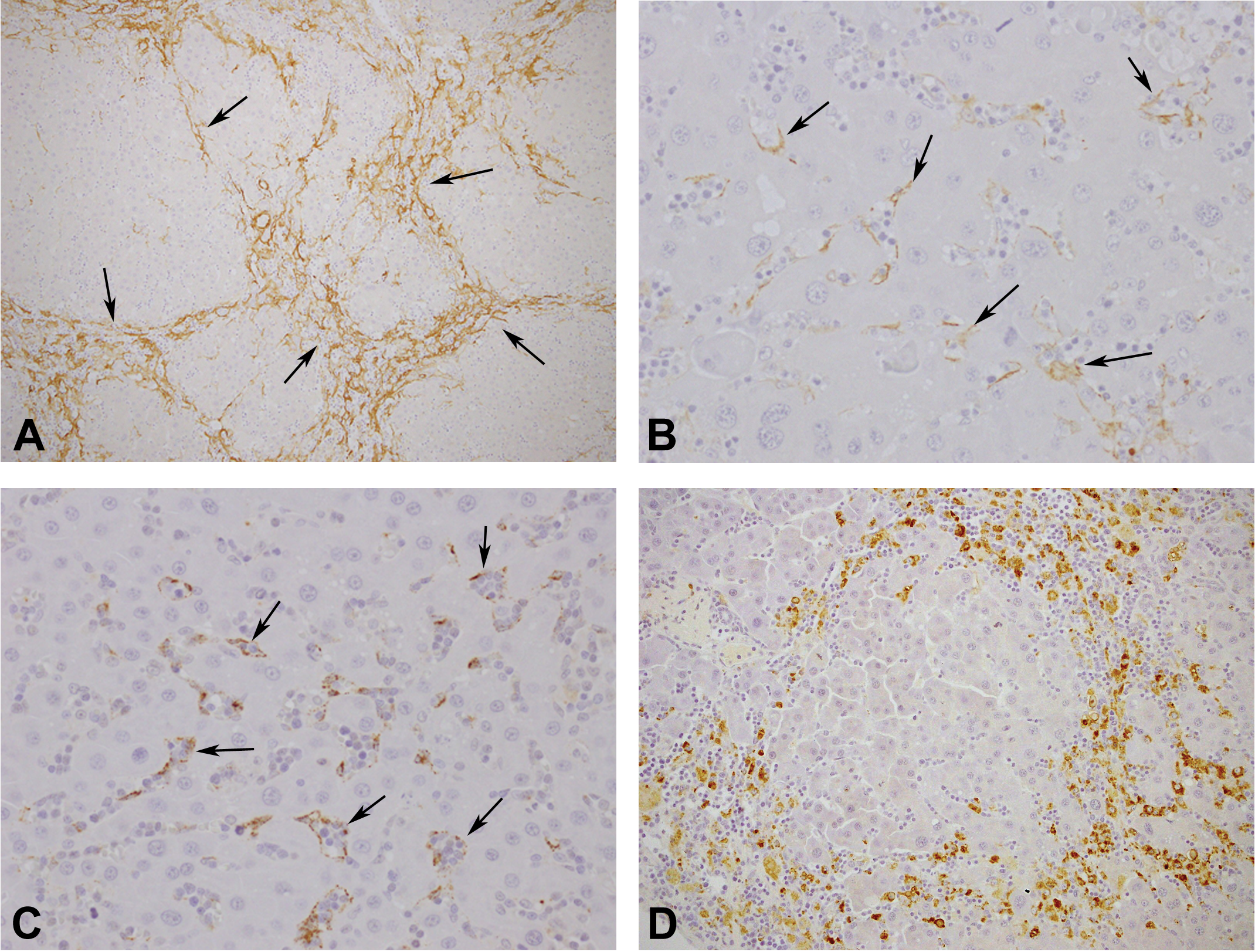
Immunohistochemical staining of the liver of rats with stage 3 LGL leukemia. (A) α-SMA expression is enhanced in the atrophic area bridging neighboring central veins (arrows), ×200. (B) α-SMA expression is enhanced along the sinusoids within the nodule (arrows), ×400. (C) FVIII-RAg expression is observed in the sinusoidal endothelial cells within the nodule (arrows), ×400. (D) ED-1-positive cells are not present within the nodule, ×200. LGL = large granular lymphocyte; α-SMA = α-smooth muscle actin; FVIII-RAg = factor VIII-related antigen.
Summary of immunohistochemical examination results in advanced cases of nodular regenerative hyperplasia in LGL leukemia rat liver.

Note: α-SMA = α-smooth muscle actin; ED-1 = anti-CD68 antibody; FVIII-RAg = factor VIII-related antigen.
aNo expression in normal liver.
bKupffer cells express in normal liver.
cGradings: − = none, + = slight, +++ = marked.
PCNA-LIs showed a positive relationship to the number of mitotic figures in the hepatocytes, consistent with a previous report (Sarli et al. 1995). Therefore, it was determined that the mitotic index of hepatocytes was an appropriate indicator of proliferative activity.
Segment 3: Histopathological Examination on the Extrahepatic Portal Veins
Of the 30 cases examined, thrombus formation and subendothelial infiltration of the extrahepatic portal vein by leukemic cells were observed only in 1 animal each and the reminder did not show any abnormal changes.
Discussion
The main hepatic changes in the rats afflicted with stage 2 or stage 3 LGL leukemia include NRH-like lesions composed of CAPH of hepatocytes and thickening of the hepatic cords with diffuse distribution. In addition, vascular abnormalities were observed in the intrahepatic portal veins but not intrahepatic arteries and extrahepatic portal vein. As the stage of LGL leukemia progresses, the nodularity of the liver became evident and vacuolation of hepatocytes composed of the nodules was appeared limited to centrilobular area. In the liver nodules of advanced cases of LGL leukemia, α-SMA and FVIII-RAg were expressed in sinusoidal lining cells and ED-1-positive macrophages/monocytes decreased in number in sinusoids.
NRH in the liver of humans is also characterized histologically by CAPH of hepatocytes and thickening of hepatic cords, but vascular abnormalities have been reported to be present in the hepatic arteries (Wanless 1990) and/or portal vein (Wanless 2002). The distribution of the lesion is basically diffuse (Scheuer and Lefkowitch 1994), but normal parenchyma intervenes the nodules in some cases (AFIP 2001). Moreover, hepatocytes composed of the nodules tend to be larger than normal hepatocytes and all of the hepatocytes within the nodules may become glycogen storage or fat as the nodules grow (AFIP 2001). Human NRH is primarily diagnosed based on the irregularity of the hepatic cords, hypertrophy of the hepatocytes, and the presence of binucleated hepatocytes, with mitoses in hepatocytes being observed only occasionally (Steiner 1959). Also, in earlier reports, human NRH seemed to be commonly associated with passive congestion in cases of severe heart failure and tuberculosis (Steiner 1959). However, the NRH-like lesions in the liver of the LGL leukemia rats are considered to be obvious proliferative changes because of the high proliferative activity of the hepatocytes as demonstrated in this study. Mitoses in the hepatocytes were more evident in stage 2 than in stage 3 of the disease. That is, it would appear that CAPH of hepatocytes is preceded by proliferative changes in the hepatocytes of the LGL leukemia rat liver.
The CAPH of hepatocytes and the vacuolation and single-cell necrosis of hepatocytes appear to be attributed to ischemia of the hepatic parenchyma. These ischemic changes in hepatocytes may result from the hypoxemia or anemia secondary to the hemolysis induced by the leukemia cells (Stromberg et al. 1983), although the other data such as clinical pathology data for anemia or other tests showing vascular perturbation were lacking. The imbalance in blood supply in the liver has been suspected to be involved in the pathogenesis of NRH in humans (Wanless 2002) and the cause needs to be clarified. In fact, hemodynamic change induced by experimental portacaval anastomosis resulted in diffuse nodular hyperplasia in the rat liver (Weinbren and Washington 1976). NRH in the human liver is often accompanied by portal hypertension (Scheuer and Lefkowitch 1994; AFIP 2001). The most prominent histological finding in F344 rats bearing LGL leukemia is splenic congestion. In general, most of human splenic congestion may result from portal hypertension. Therefore, the imbalance in blood supply, anemia, or portal hypertension in the pathogenesis of NRH-like lesion in F344 rats were discussed in addition to the analysis of the significance of histopathlogical changes observed.
Judging from the diffuse distribution of the uniform-sized lesions in the liver, we suspected that the cause of the NRH-like lesions existed in the vessels near the hilus of the liver. NRH, focal nodular hyperplasia, and partial nodular transformation in human liver are benign nodular lesions showing common histological changes and their cause is considered to be due to vascular abnormalities (AFIP 2001). A rat model of selenium-induced hepatic lesions has been reported as an animal model of human NRH (Bioulac-Sage et al. 1992). In this animal model, the medial lobe is atrophic instead of a hypertrophied caudate lobe (Bioulac-Sage et al. 1992). In the rat liver, ligation of the portal vein branches supplying the left and median lobes of the rat liver is followed by marked atrophy of these lobes but not coagulative necrosis (Kerr 1971). Also, the blood draining from spleen tends to flow to the left side of the liver (Malarkey et al. 2005). Based on these observations, it is likely that the true cause of the NRH-like lesions is circulatory disturbance in the portal venous system and not in the hepatic arterial system. In fact, the lesions in the intrahepatic vessels of LGL leukemia rats were limited to the portal venous system. The present study also demonstrated thickening of the vascular wall of the intrahepatic portal veins but not of the extrahepatic portal veins. On the other hand, vascular lesions in human NRH are also noted in the arteries of the liver (Wanless 1990).
The most prominent histological characteristic of LGL leukemia is splenic congestion (Losco and Ward 1984) accompanied by anemia (Stromberg, Kociba, et al. 1990). In the early stage of the disease, the splenic congestion seems to be caused by the accumulation of erythrocytes and the fluid derived from the destroyed erythrocytes (Stromberg, Kociba, et al. 1990). Hemolysis induces splenic congestion in humans (Petroianu 2007) and rodents (Hejtmancik et al. 2002), but the degree of splenic enlargement due to hemolysis is mild (up to 2–6 times the normal weight of the spleen; Hejtmancik et al. 2002; Poulet et al. 2010) as compared with that observed in the LGL leukemia rats (up to 25 times the normal weight at the maximum; Stromberg 1990). In LGL leukemia, splenic congestion is evident and consistent throughout the course of the disease. Splenomegaly in LGL leukemia may be related to portal hypertension as well as neoplastic proliferation of the leukemia cells, judging from the evident congestion in the spleen. In humans, lymphoproliferative and myeloproliferative disorders have been known to be associated with portal hypertension (Shaldon and Sherlock 1962; Dubois et al. 1993) and hepatic and portal vein thrombosis (Boughton 1991). The high incidence of NRH-like lesions in the LGL leukemia rat liver suggests that splenic organotropy of LGL leukemia (Stromberg and Vogtsberger 1983) might play an important role in this site-specific occurrence of lesions in the portal venous system.
In human case, increased blood flow in the spleen due to hematogenic tumors seems to lead to an increase in portal blood flow, resulting in portal hypertension (Dubois et al. 1993). Forward flow theory of portal hypertension, paying attention to the increased splanchnic blood flow maintaining portal hypertension, supported this phenomenon (Vorobioff, Bredfeldt, and Groszmann 1983). Therefore, we considered that increased blood flow in the spleen and/or other organs due to hemolytic anemia and/or leukemia cell proliferation leads to the functional portal hypertension. Although portal hypertension induced by hyperthyroidism was also reported (Oren et al. 1999), a possibility that hyperthyroidism may play a role in the portal hypertension in LGL leukemia rats was excluded because of no case of hyperthyroidism noted in the present study. Also, sympathetic nervous activity suggested a role in contraction of the portal vein as indicated in treatment with clonidine, an α2-agonist, known to be useful in the treatment of portal hypertension in case of liver cirrhosis (Roulot et al. 1989). However, no sympathetic nervous activity–related changes were observed in LGL leukemia rats. The possibility that vascular injuries including thrombus formation in the portal vein may develop at early stage of LGL leukemia in rats cannot be denied, referring to the human leukemia cases of portal thrombosis (Boughton 1991). In a rat model of NRH, portal hypertension developed before the formation of the nodules in the liver (Bioulac-Sage et al. 1992). Canine model of NRH also shows that NRH is often associated with portal hypertension resulting from the obliteration of small portal veins (McEntee et al. 1998). Therefore, the portal hypertension may have an important role in the pathogenesis of NRH-like lesion, although there are negative data on the relationship with portal hypertension in human NRH (Wanless 1990).
Also, nodularity, single-cell necrosis of the centrilobular hepatocytes, and proliferative activity of the hepatocytes were all increased in the livers of the LGL leukemia rats, although ischemia was a common pathogenetic factor in both the LGL leukemia rats and the nonleukemia anemic rats. The facts that the CAPH of hepatocytes also developed in the livers of animals with severe EMH in the spleen indicated hypoxemia has a role in its pathogenesis, but NRH-like lesion did not develop in these cases. That is, hypoxemia alone cannot induce NRH-like lesion. Therefore, it is possible that gradual necrosis of the hepatocytes that triggers regeneration of the hepatocytes and repeated cycles of necrosis and regeneration of hepatocytes may result in nodule formation in LGL leukemia rat liver as in the case of the regenerative nodules observed in human liver cirrhosis.
In livers of rats with advanced LGL leukemia, α-SMA expression in the sinusoids of the noninjured areas seems to be induced either by direct injury of the leukemia cells or by mechanical hemostatic pressure as seen in human cases (Schmitt-Gräff et al. 1991). Also, the finding of hemosiderin deposition indicates the presence of vascular injury. In an in vitro study, endothelial cell stimulation by fluid shear stress induced smooth muscle cell migration (Ono et al. 1991). In an animal model of NRH, endothelial cell injury was limited to the sinusoids, without affecting the hepatic veins, hepatic arteries, or portal veins, and sinusoidal endothelial cell injury may lead to portal hypertension and result in the development of NRH (Bioulac-Sage et al. 1992).
Hepatocellular carcinoma (HCC) is nourished only by arterial blood as demonstrated by the positive tumor staining in transarterial angiography (Breedis and Young 1954) and the existence of no or only a few portal areas on histopathological examination. In addition, histopathological examination of small HCC tumors in humans indicated that sinusoidal endothelial cells in the HCC begin to express FVIII-RAg as the arterial blood supply to the HCC increases (Kin et al. 1994). Similarly, in an animal model of hepatocarcinogenesis, sequential observation of the sinusoids within the tumor nodule demonstrated that the endothelial cells began to express FVIII-RAg and the number of Kupffer cells decreased as the arterial blood supply increased (Yamamoto et al. 1996; Shoji et al. 1994). FVIII-RAg expression in the sinusoids indicates sinusoidal capillarization (Schaffner and Popper 1963), which has also been observed in selenium-induced hepatic lesions (Dubuisson et al. 1995). Therefore, FVIII-RAg expression and the lack of Kupffer cells in the hepatic sinusoids in the present study may also suggest that the main route of blood supply to the liver in cases with advanced LGL leukemia was the hepatic artery. These findings may also be indirect indicators of imbalance in blood flow or abnormal intrahepatic circulation.
Although LGL leukemia cells had a relatively uniform appearance, pleomorphic tumor cells were detected in some cases with the intermediate or late stage of disease. In vitro study revealed the heterogeneity and cytotoxic activity of LGL leukemia cells (Ward and Reynolds 1983). A study involving transplantation of LGL leukemia cells into syngeneic rats demonstrated that increased passage of leukemia cells was associated with a decreased longevity of the rats (Stromberg, Grants, et al. 1990; Stromberg et al. 1985). The relationship between the morphology and biological behavior of the LGL leukemia cells could not be clearly characterized in the present study because there were cases with the intermediate stage of the disease that showed evidence of endothelial cell injury and cases with the late stage of the disease in which the typical morphology was still maintained. It appears that the biological malignancy of LGL leukemia cells increases as the cells proliferate within the body. Although hepatic injury associated with hematological malignancies is rare in humans (Ohtani et al. 2008; Anderson et al. 2001), the present study demonstrated that LGL leukemia cells caused liver injury as a secondary effect via inducing vascular endothelial cell injury and/or hemolytic anemia. It has been reported that in the rat model of selenium-induced hepatic lesions, sinusoidal endothelial cell injury may play a role in the pathogenesis. As for the NRH-like lesions in the LGL leukemia rat liver, hemolysis may have a more important role in the pathogenesis than in endothelial cell injury because no evidence of endothelial cell injury was observed in either of the 2 cases in which NRH-like lesions developed despite the small spleen size (less than 49 mm).
The present study findings suggest that hemolytic anemia, imbalance in blood flow, and endothelial cell injuries in the liver play important roles in the pathogenesis of the NRH-like lesions in the livers of LGL leukemia rats. Various conditions have been implicated in the development of NRH in the livers of humans; however, the precise cause is still unknown. The occurrence of NRH-like lesions in F344 rats was limited exclusively to rats with LGL leukemia, although the pathophysiological conditions varied in severity depending on the stage of progression of the disease. Investigation of the LGL leukemia rat liver may be helpful to clarify the pathogenesis of NRH in the human liver. To investigate the presence or absence of the portal hypertension in LGL leukemia rats may lead to a clue of the pathogenesis.
Footnotes
Acknowledgment
We wish to thank to Dr. Katsumi Kobayashi (National Institute of Health Sciences, Japan) for statistical analysis.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
