Abstract
Sphingosine-1-phosphate (S1P) lyase is considered as a drug target in autoimmune diseases based on the protective effect of reducing activity of the enzyme in animal models of inflammation. Since S1P lyase deficiency in mice causes a severe, lethal phenotype, it was of interest to investigate any pathological alterations associated with only partially reduced activity of S1P lyase as may be encountered upon pharmacological inhibition. Both genetic reduction of S1P lyase activity in mice and inhibition of S1P lyase with a low-molecular-weight compound in rats consistently resulted in podocyte-based kidney toxicity, which is the most severe finding. In addition, skin irritation and platelet activation were observed in both instances. The similarity of the findings in both the genetic model and the pharmacological study supports the value of analyzing inducible partially target-deficient mice for safety assessment. If the findings described in rodents translate to humans, target-related toxicity, particularly podocyte dysfunction, may limit chronic systemic treatment of autoimmune diseases with S1P lyase inhibitors. Furthermore, partial deficiency or inhibition of S1P lyase appears to provide an in vivo rodent model to enable studies on the mechanism of podocyte dysfunction.
Introduction
Sphingosine-1-phosphate (S1P) lyase is an intracellular microsomal enzyme that catalyzes the irreversible degradation of S1P in a retroaldol reaction dependent on the cofactor pyridoxal 5′phosphate (for review, see Serra and Saba 2010). S1P lyase is expressed at various degrees in many mammalian tissues (Genter et al. 2003; Serra and Saba 2010) except for platelets (Yatomi et al. 1997) and erythrocytes (Ito et al. 2007).
Deficiency or partial deficiency of S1P lyase in vivo causes a rise in S1P levels in tissues and blood (Bagdanoff et al. 2010; Billich et al. 2013; Vogel et al. 2009). Inhibition of S1P lyase leads to a disruption of the S1P gradient between lymphoid tissues and blood, which normally feature low and high concentrations, respectively (Schwab et al. 2005). Since the normal S1P gradient is required for the egress of T cells from the lymphoid tissues, disruption of the gradient by (partial) S1P lyase deficiency or inhibition leads to reduced T-cell egress into the blood (Bagdanoff et al. 2010; Billich et al. 2013; Schwab et al. 2005; Vogel et al. 2009; Yu et al. 2010). Also in healthy subjects, inhibition of S1P lyase causes a decrease in peripheral T-cell numbers (Bagdanoff et al. 2010). Partial deficiency or inhibition of S1P lyase has been shown to confer protection in models of inflammation such as delayed-type hypersensitivity (DTH), experimental autoimmune encephalomyelitis (EAE), and collagen-induced arthritis, supporting the concept that inhibition of S1P lyase may be a therapeutic option in the treatment of autoimmune diseases (Bagdanoff et al. 2010; Billich et al. 2013; Weiler et al. 2014).
Fully S1P lyase–deficient mice do not survive beyond the first weeks of life (Schmahl et al. 2007; Vogel et al. 2009), having a median life span of 29 days (Vogel et al. 2009). Loss of S1P lyase is associated with defects in lung (accumulation of proteinaceous exudates in alveoli, which impairs gas exchange), heart (increased interstitial cellularity and vacuolation in myocardium), urinary tract (widespread ballooning vacuolation, degeneration, and apoptosis of urothelial cells), bone (osteopetrosis and enlarged dysfunctional osteoclasts), kidney (glomerular changes), and vasculature (hemorrhage and microaneurysm) as well as causing myeloid hyperplasia (Schmahl et al. 2007; Vogel et al. 2009). Human knock-in mice harboring one allele or two alleles of human S1P lyase express less than 10% and 20% of normal S1P lyase activity, respectively. This partial restoration of S1P lyase is sufficient to fully protect both humanized mouse lines from the lesions in lung, heart, urinary tract, and bone that develop in fully S1P lyase–deficient mice, whereas lymphodepletion still occurs (Vogel et al. 2009).
The aim of this study was to comprehensively characterize pathological alterations associated with partial deficiency or inhibition of S1P lyase in rodents and to support drug safety assessment for S1P lyase inhibitors. Inducible partially S1P lyase–deficient mice as well as rats treated with a low-molecular-weight S1P lyase inhibitor in the context of EAE were analyzed.
Materials and Methods
Animals
Tamoxifen-inducible S1P lyase–deficient (SPLFlox/Flox Cre+) and corresponding control mice (SPLFlox/Flox Cre−, Cre+, and Cre−) were generated as described in Billich et al. (2013). Female Dark Agouti (DA) rats were purchased from Janvier (France). Mice and rats were housed for different periods of time in individually ventilated cages equipped with a suitable rodent house and nesting material, receiving a standardized chow and water ad libitum. Housing and studies were performed in conformity with the Swiss Animal Welfare Law ensuring humane treatment and care of the laboratory animals.
Mouse Sheep Red Blood Cell (SRBC)-induced DTH Reaction
Some analyses were performed with inducible S1P lyase–deficient (and corresponding control) mice, in which an SRBC-induced DTH reaction was induced 8 weeks after tamoxifen treatment, as described in Billich et al. (2013).
Rat EAE Model and Treatment With Low-molecular-weight S1P Lyase Inhibitor
Female DA rats were immunized with 75 µg myelin oligodendrocyte glycoprotein (amino acids 1-129) emulsified in incomplete Freund's adjuvant supplemented with 2 mg/ml heat-inactivated Mycobacterium tuberculosis at the base of the tail. An S1P lyase inhibitor, (R)-6-(4-(4-benzyl-7-chlorophthalazin-1-yl)-2-methylpiperazin-1-yl)nicotino nitrile (compound 1 in the study by Loetscher et al. 2013), was dosed as a solution in Capmul (Abitec) twice daily at 2 and 10 mg/kg, starting 2 days before EAE induction.
Sampling
At indicated time points, urine was sampled from mice or rats for determination of urine protein–creatinine ratio. Under terminal anesthesia with 5% isofluran, blood was withdrawn for clinical chemistry (serum from blood without anticoagulant) as well as hematology and investigations on platelets (EDTA blood). At the end of the mouse and rat studies, organs/tissues were sampled for histopathology, electron microscopy, and determination of tissue S1P concentrations, as indicated.
Urine Protein–Creatinine Ratio
Urine creatinine (uCREA) and total protein (uPROT) concentrations were determined with a Cobas 6000® analyzer (Roche Diagnostics, Switzerland). For each sample, the urine protein–creatinine ratio was calculated as follows: uPROT divided by uCREA.
Clinical Chemistry
Aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, and creatine kinase activities, total bilirubin, triglyceride, cholesterol, glucose, total protein, album in, globulin, urea, creatinine, calcium, phosphorus, chloride, sodium, potassium, and magnesium concentrations were determined using a Cobas 6000 analyzer (Roche Diagnostics).
Hematology
Hematology analysis was performed using an ADVIA 120® automatic hematological analyzer (Siemens Healthcare Diagnostics, Germany).
Platelet Investigations
Platelet surface–associated P-selectin (CD62P) was determined using an Alexa488-conjugated anti-CD62P antibody detected with an Life Science Research (LSR) II flow cytometer (Becton Dickinson, Allschwil, Switzerland). The evaluation of soluble P (sP)-selectin was carried out from frozen EDTA plasma samples at a 1/50 dilution using an ELISA kit (Quantikine® mouse sP-selectin; R&D Systems, UK). Optical density values were measured using a VERSAmax microplate reader (Molecular Devices, Sunnyvale, CA).
Histopathology
In the context of an SRBC-induced DTH study with partially S1P lyase–deficient (SPLFlox/Flox Cre+) and corresponding SPLFlox/Flox Cre− control mice, animals were sacrificed 8 weeks after tamoxifen treatment (Billich et al. 2013). Lungs, heart, urinary bladder, sternum, femur and knee joints, kidneys, aorta, mesentery, skin, liver, gallbladder, stomach, small and large intestine, pancreas, adrenal glands, brain, eyes, Harderian glands, salivary glands, mandibular and mesenteric lymph nodes, spleen, and thymus were fixed in neutral phosphate-buffered formalin, embedded in paraffin wax, sectioned, and stained with hematoxylin and eosin (H&E). Additionally, kidney sections were stained with periodic acid–Schiff's reagent (PAS). The tissue sections were examined by light microscopy. In other studies, partially S1P lyase–deficient (SPLFlox/Flox Cre+) and corresponding SPLFlox/Flox Cre−, Cre+, and Cre− mice were housed for 7 weeks and up to 14 to 15 mo. Histopathological analysis of H&E-stained sections was performed on a number of tissues, including lungs, heart, sternum, kidneys, skin, liver, gallbladder, stomach, small and large intestine, adrenal glands, brain, ovaries, uterus, vagina, testes, mandibular and mesenteric lymph nodes, spleen, and thymus. Histopathological demonstration of progression of glomerulopathy was based on Trichrome- and PAS-stained sections of kidney from partially S1P lyase–deficient (SPLFlox/Flox Cre+) mice 7 mo after tamoxifen treatment. In the context of a 2-week EAE study, in which DA rats were treated twice daily with either 2 or 10 mg/kg of a low-molecular-weight S1P lyase inhibitor or corresponding vehicle or water for 16 days starting 2 days before EAE induction (Weiler et al. 2014), sections of lungs, kidneys, skin, brain, and thymus were stained with H&E (lungs, skin, brain, and thymus) or PAS (kidneys) and histopathologically analyzed.
Electron Microscopy
Samples for electron microscopy were taken at necropsy from partially S1P lyase–deficient (SPLFlox/Flox Cre+) and corresponding SPLFlox/Flox Cre− control mice 7 weeks after tamoxifen treatment. Kidney samples were fixed with 3% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.4, for about 1 hr at 4°C and with 1% OsO4 in 0.1 M cacodylate buffer, pH 7.4, for 1 hr at 4°C. After fixation, the tissues were dehydrated in graded acetone solutions and embedded in Epon. Semi-thin sections were stained with toluidine blue. Ultrathin sections were counterstained with uranyl acetate and lead citrate and examined by transmission electron microscopy.
Determination of S1P and Ceramide Concentrations
S1P and ceramide in kidney samples were determined as described in Billich et al. (2013). S1P and ceramide in skin and ceramide in blood samples were determined according to the method followed by Merrill et al. (2005) with slight modification using an SL-6460 liquid chromatography-electrospray ionization-tandem mass spectrometer (triple Quad MS) from Agilent Technologies AG (Basel, Switzerland). LC separation was carried out on Agilent 1200. The mouse skin samples were homogenized by crushing in liquid N2. The liquid/liquid extraction was performed in an eluent mixture of 80% B (5 mM ammonium acetate in methanol containing 1% acetic acid and 0.25% tetrahydrofuran) and 20% A (5 mM ammonium acetate in methanol/H2O [1:1] containing 1% acetic acid and 0.25% tetrahydrofurane). For separation, a 2.1 × 100 mm, 1.8 µm Zorbax SB-C18 column was operated at 50°C. The flow rate was 700 μl/min using a gradient of 0–0.1 min 1% B isocratic, in 1.9 min to 100% B, and stays at 100% B for 6 min. Total analysis time was 12.5 min.
Statistical Analysis
A normality test was performed to assure that the data were normally distributed. The equal variance test was used to check the assumption that the sample was drawn from populations with the same variance. If both normality and equal variance tests passed and in the case of significant results of a one-way analysis of variance, multiple comparisons were performed using the Dunnett’s test. In the case of comparison of only 2 groups, a t-test was done. If the normality and/or equal variance test failed, a suitable transformation (log, square root) was applied, and if the normality and/or equal variance test still failed, the nonparametric Kruskal–Wallis test was used. *P < .05 was considered statistically significant.
Results
Pathological Alterations Associated with Partial S1P Lyase Deficiency in Mice
Partially S1P lyase–deficient (SPLFlox/Flox Cre+) and corresponding SPLFlox/Flox Cre− control mice have been studied in a model of SRBC-induced DTH 8 weeks after tamoxifen treatment (Billich et al. 2013). Histopathological examination of tissues from the same animals revealed alterations in kidneys and skin as relevant microscopic findings associated with partial S1P lyase deficiency (Supplementary Figure 1). In the kidneys, minimal increase in mesangial matrix associated with obliteration of some capillary lumen was found. Subsequent PAS staining of kidney sections did not suggest deposition of immune complexes to explain the observed glomerulopathy. Skin findings included minimal to mild epidermal hyperplasia (acanthosis) with orthokeratotic hyperkeratosis associated with a discrete increase in dermal inflammatory cells.
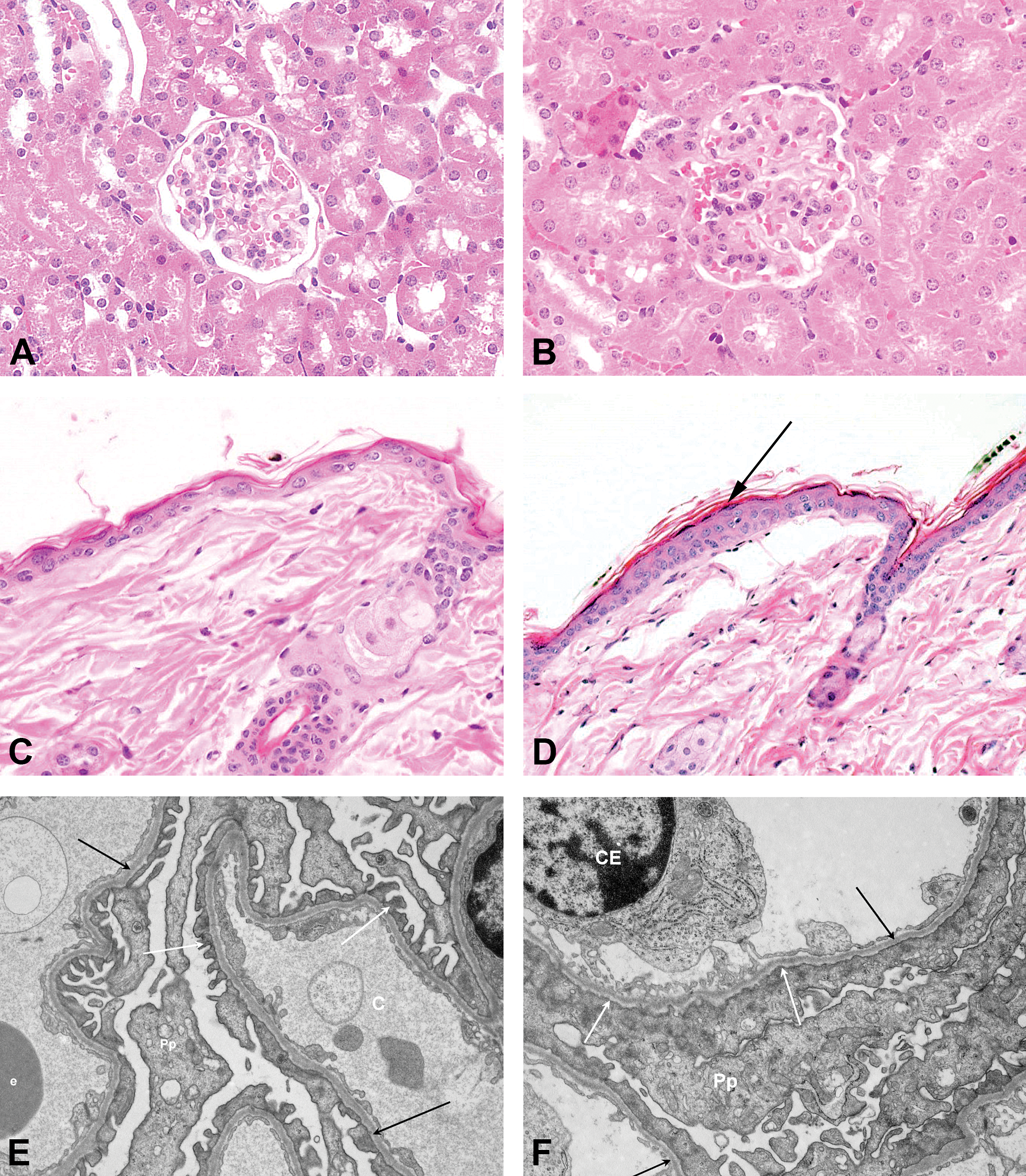
Pathological alterations in kidneys and skin of partially S1P lyase–deficient and corresponding control mice 7 weeks after tamoxifen treatment. (A) H&E-stained kidney and (C) H&E-stained skin sections from SPLFlox/Flox Cre− control mice with no abnormalities. (E) Electron microscopic image of kidney from SPLFlox/Flox Cre− control mice with normal podocyte foot processes (white arrows) and basement membranes (black arrows). (B) H&E-stained kidney section from partially S1P lyase–deficient (SPLFlox/Flox Cre+) mice showing increase in mesangial matrix with obliteration of some capillary lumina. (D) H&E-stained skin section from partially S1P lyase–deficient (SPLFlox/Flox Cre+) mice showing acanthosis/orthokeratotic hyperkeratosis (black arrow). (F) Electron microscopic image of kidney from S1P lyase–deficient (SPLFlox/Flox Cre+) mice showing effacement of podocyte foot processes (white arrows) and normal basement membranes (black arrows). Pp = cytoplasm of podocyte; CE = capillary endothelial cell nucleus; C = capillary lumen; e = erythrocyte.
Similar findings in kidneys and skin were observed in partially S1P lyase–deficient (SPLFlox/Flox Cre+) mice when compared to corresponding SPLFlox/Flox Cre−, Cre+, and Cre− control mice 7 weeks after tamoxifen treatment without induction of the DTH reaction (Figure 1). However, dermal inflammatory cells were not found in the skin of SPLFlox/Flox Cre+ mice without SRBC-induced DTH. Electron microscopic analysis of kidneys revealed effacement of podocyte foot processes of the partially S1P lyase–deficient mice only (interpretation based on the examination of few glomeruli of 2 SPLFlox/Flox Cre− control and 4 partially S1P lyase–deficient SPLFlox/Flox Cre+ mice, where the foot process effacement was diffuse). The glomerular alterations were associated with a decreased serum total protein concentration (−12% [males] and −26% [females] compared with control), related with decreases in both, albumin (−7% [males] and −28% [females]) and globulin (−24% [males] and −18% [females]) concentrations, an increase in the urine protein–creatinine ratio (6.8-fold vs. control in female mice only), and increased serum concentrations of cholesterol (+43% in female mice only) and triglycerides (+65% in female mice only).
Histological findings in lymphoid organs of SPLFlox/Flox Cre+ mice included medullary enlargement in the thymus, decrease or absence of germinal center development, and lymphoid depletion (primarily T-cell areas) in lymph nodes (not shown).
Platelet investigations were only done with samples from male mice (Table 1). Partial S1P lyase deficiency was associated with 1.4-fold increased platelet surface expression of P-selectin associated with 1.2-fold increased plasma concentrations of sP-selectin.
Effects of Partial S1P Lyase Deficiency on Mouse Platelet Membrane P-selectin (CD62p) Expression, Plasma sP-selectin, and Collagen-induced WBPA.

Values are expressed as mean ± SD. n = number of animals; MFI = mean fluorescence intensity; AUC = area under the curve; WBPA = whole blood platelet aggregation; s-P = soluble P; S1P = sphingosine-1-phosphate.
*p < .05 vs. SPLFlox/Flox Cre−.
S1P concentrations were markedly elevated in kidneys (SPLFlox/Flox Cre−, average 0.1 pmol/mg; SPLFlox/Flox Cre+, average 10.3 pmol/mg; Billich et al. 2013) and skin (Figure 2A) and moderately in blood (Billich et al. 2013), whereas C16 ceramide concentrations remained unchanged in kidneys (Billich et al. 2013), were decreased (alongside with other ceramides) in skin (Figure 2A), and increased in blood (the determination of C16 ceramide concentration in blood was done in the context of the SRBC-induced DTH study; Figure 2B), if S1P lyase was partially absent. Altogether these results suggest that partial S1P lyase deficiency causes, in addition to its immunological effects (see also the study by Billich et al. 2013), protein-losing glomerulopathy due to podocyte dysfunction, skin irritation, and platelet activation.
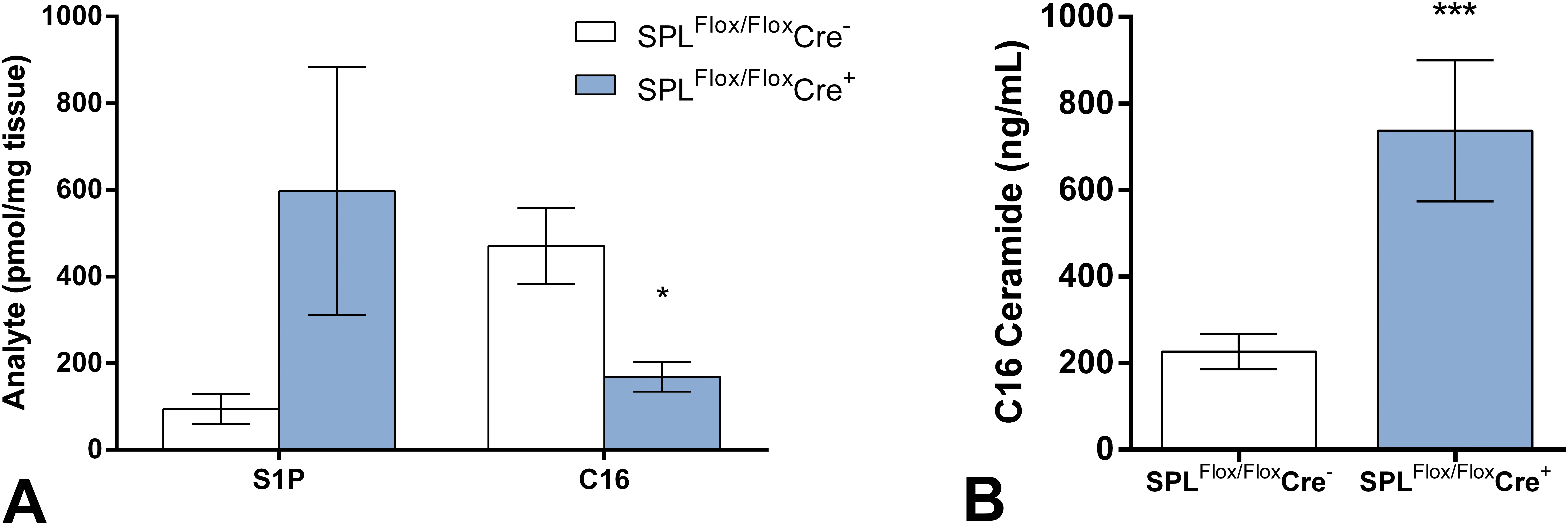
Concentrations of selected sphingolipids in skin and blood of partially S1P lyase–deficient mice. (A) S1P and C16 ceramide concentrations in the skin of partially S1P lyase–deficient (SPLFlox/Flox Cre+) and corresponding control (SPLFlox/Flox Cre−) mice 7 weeks after tamoxifen treatment. *p < .05 vs. SPLFlox/Flox Cre−. (B) C16 ceramide concentrations in the blood of partially S1P lyase–deficient (SPLFlox/Flox Cre+) and corresponding control (SPLFlox/Flox Cre−) mice in the context of an SRBC-induced DTH study 8 weeks after tamoxifen treatment. ***p < .005 vs. SPLFlox/Flox Cre−. S1P = sphingosine-1-phosphate; SRBC = sheep red blood cell; DTH = delayed-type hypersensitivity.
SPLFlox/Flox Cre+ and corresponding SPLFlox/Flox Cre−, Cre+, and Cre− mice were housed for up to 14 to 15 mo. Mortality in the group of SPLFlox/Flox Cre+ mice was substantially higher than that in the other groups, indicating a significant long-term impact of partial S1P lyase deficiency. At study termination, only 14% of SPLFlox/Flox Cre+ mice, compared to 79%, 98%, and 95% for the corresponding SPLFlox/Flox Cre−, Cre+, and Cre− mice, respectively, were available for examination. Histopathological analysis of kidneys revealed progression of glomerulopathy with severe and chronic glomerular lesions resembling focal and segmental glomerulosclerosis, tubular degeneration, and interstitial fibrosis and inflammation associated with partial deficiency of S1P lyase (Figure 3A and B) accompanied by chronically elevated urine protein–creatinine ratios (Figure 3C). The kidney lesions were considered to be the main underlying cause of premature death of the animals.
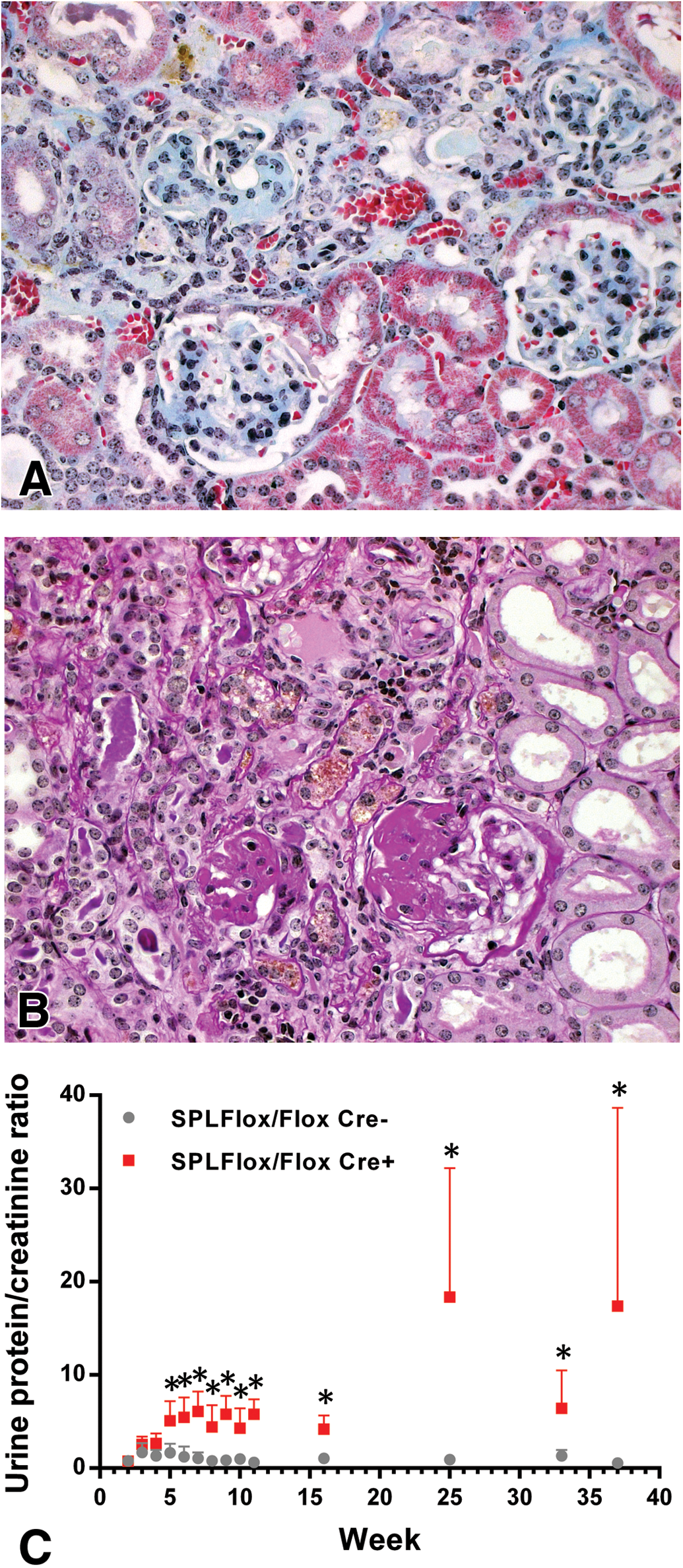
Progression of glomerulopathy in partially S1P lyase–deficient mice. (A) Trichrome and (B) PAS-stained section of kidney from partially S1P lyase–deficient (SPLFlox/Flox Cre+) mice 7 mo after tamoxifen treatment, with severe chronic glomerular lesions similar to focal and segmental sclerosis accompanied by tubular degeneration and interstitial fibrosis with focal inflammatory infiltrate. (C) Time course of urine protein–creatinine ratio from partially S1P lyase–Figure 3.—(Continued) deficient (SPLFlox/Flox Cre+) and corresponding control (SPLFlox/Flox Cre−) mice. *p < .05 vs. SPLFlox/Flox Cre−. S1P = sphingosine-1-phosphate; PAS = periodic acid–Schiff's reagent.
Pathological Alterations Induced by a Low-molecular-weight S1P Lyase Inhibitor in Rats in the Context of EAE
In order to test the effect of a low-molecular-weight inhibitor of S1P lyase, an EAE study was performed with the inhibitor 1, a reversible active-site-directed inhibitor of S1P lyase, featuring IC50 = 214 nM in a biochemical assay (Loetscher et al. 2013). DA rats were treated twice daily with either 2 or 10 mg/kg inhibitor 1 for 16 days, starting 2 days before EAE induction (Weiler et al. 2014). Relevant microscopic findings associated with the S1P lyase inhibitor at 10 (but not 2) mg/kg twice a day (bid) included alterations in kidneys, skin, and lungs. In kidneys, hypertrophic podocytes and protein droplets, with no changes in basement membrane, glomerular capillaries, or mesangial matrix were found (Figure 4). This was associated with an increase in the urine protein–creatinine ratio (Figure 4C), a decrease in serum albumin (−27% at 10 mg/kg bid) and total protein concentrations (−9% at 10 mg/kg bid), and an increase in serum urea (+37% at 10 mg/kg bid), creatinine (+33% at 10 mg/kg bid), and inorganic phosphorus (+18% at 10 mg/kg bid) concentrations. A very strong increase in S1P and a moderate increase in C16 ceramide concentrations in kidneys were also noted (Figure 4D).
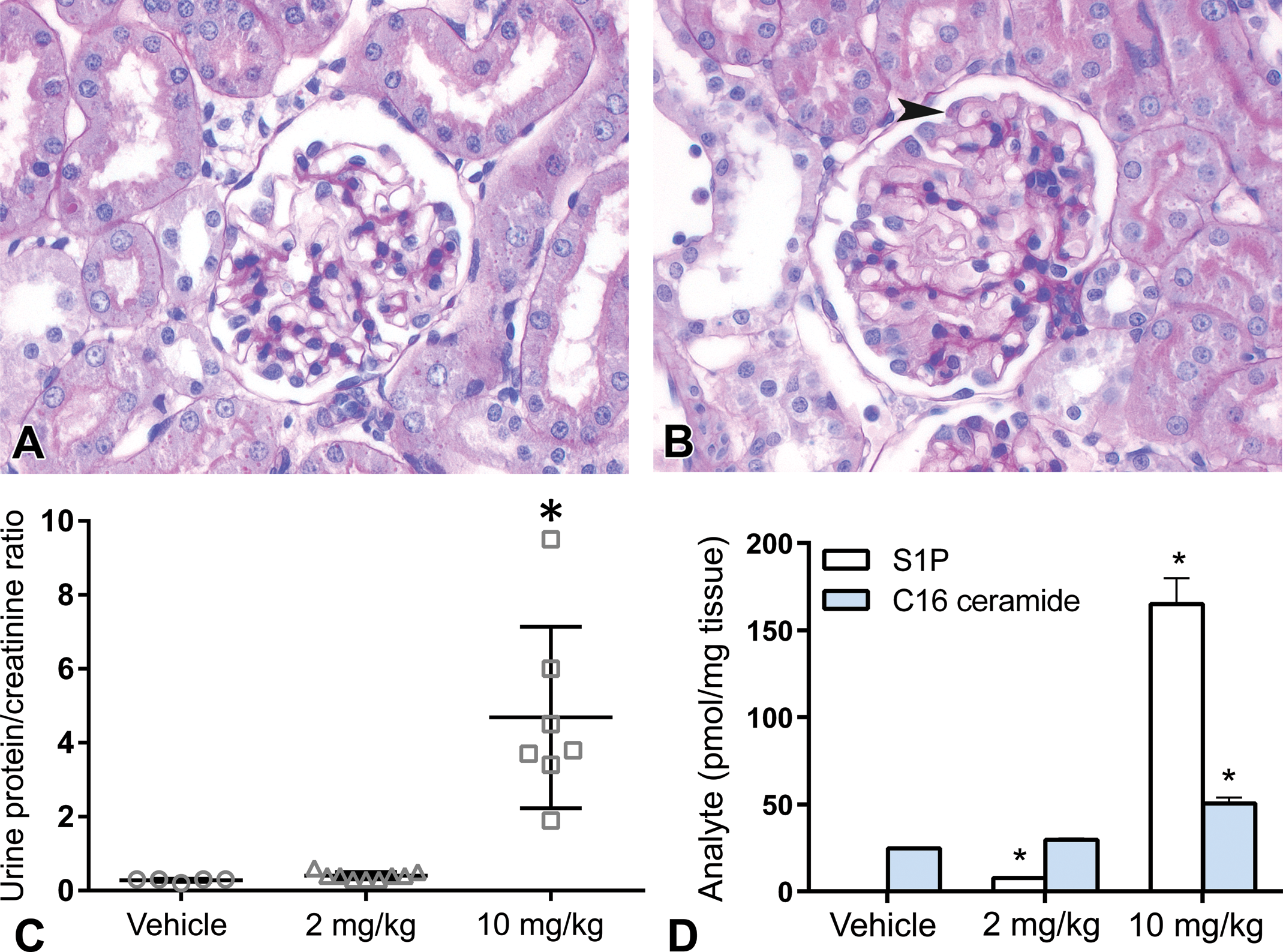
Pathological alterations in kidneys of DA rats treated with S1P lyase inhibitor for 16 days in the context of EAE. (A) PAS-stained section of kidney from vehicle-treated DA rats showing normal morphology. (B) PAS-stained section of kidney from DA rats treated with 10 mg/kg bid S1P lyase inhibitor showing hypertrophic podocytes containing protein droplets (arrow head). (C) Urine protein–creatinine ratio in DA rats treated with 2 or 10 mg/kg bid S1P lyase inhibitor or vehicle. *p < .05 vs. vehicle control. (D) S1P and C16 ceramide concentrations in the kidney of DA rats treated with 2 or 10 mg/kg bid S1P lyase inhibitor or vehicle. *p < .05 vs. vehicle control. DA = Dark Agouti; EAE = experimental autoimmune encephalomyelitis; S1P = sphingosine-1-phosphate; PAS = periodic acid–Schiff's reagent; bid = twice a day.
Throughout the skin (Figure 5), mixed cell inflammation (dermatitis) was found. As redness was a marked macroscopic in-life finding in this study, ear skin was examined in detail. In addition to the dermatitis, minimal interstitial edema accompanied by slight epidermal acanthosis, with or without orthokeratosis/scab and ulceration/cellular crusts, was found.
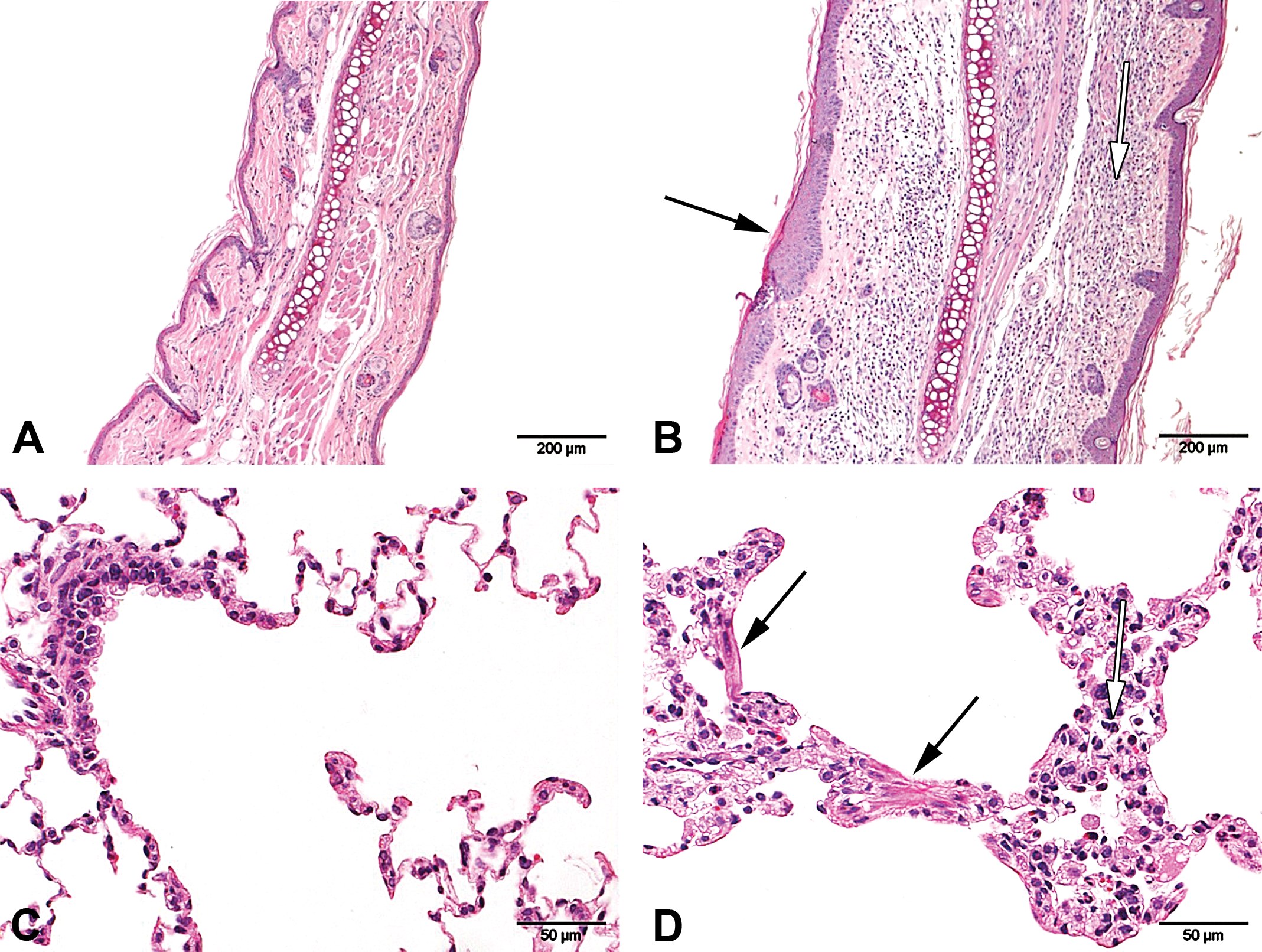
Pathological alterations in skin and lungs of DA rats treated with S1P lyase inhibitor for 16 days in the context of EAE. (A) H&E-stained skin (ear) and (C) H&E-stained lung sections from vehicle-treated DA rats with no abnormalities. (B) H&E-stained skin (ear) section from rats treated with 10 mg/kg bid S1P lyase inhibitor showing acanthosis (black arrow) and inflammation (white arrow). (D) H&E-stained lung section from rats treated with 10 mg/kg bid S1P lyase inhibitor showing smooth muscle hypertrophy/hyperplasia (black arrows) and mixed inflammatory cell infiltrate (white arrow). DA = Dark Agouti; EAE = experimental autoimmune encephalomyelitis; S1P = sphingosine-1-phosphate; bid = twice a day.
In lungs (Figure 5), minimal smooth muscle hypertrophy/hyperplasia of terminal bronchioles and alveolar ducts, mixed inflammatory cell infiltrates in alveolar septae, peribronchiolar/perivascular inflammation associated with edema and a minimal increase in the number of alveolar macrophages were identified. However, some of the findings may represent an exaggeration of underlying multifocal granulomatous inflammation of alveolar septae in the lungs, which was also observed in the control groups and thus may be linked to EAE.
It should be mentioned that thymi were markedly depleted/atrophic upon treatment of rats in the EAE model with 10 mg/kg bid of the S1P lyase inhibitor. Thymus atrophy is commonly seen as a consequence of general toxicity and considered to be caused by stress. It remains therefore unknown if and to what extent the thymus changes might have been caused by inhibition of S1P lyase.
Inhibition of S1P lyase also appeared to cause a decrease in RBC count, hemoglobin, hematocrit, and platelet count as well as an increase in P-selectin expression on platelets (4.2-fold at 10 mg/kg bid; Table 2).
Effects of S1P Lyase Inhibitor on EAE Rat Platelet Membrane P-selectin (CD62p) Expression.

Values are expressed as mean ± SD. n, number of animals; MFI, mean fluorescence intensity; S1P = sphingosine-1-phosphate; bid = twice a day.
Finally, an increase in serum alanine and aspartate aminotransferase and alkaline phosphatase activities associated with a decrease in serum total bilirubin concentration were observed as a consequence of pharmacological inhibition of S1P lyase in the rat EAE model, suggesting an additional effect in the liver. However, liver had not been sampled for histological examination.
Altogether these results showed that treatment of rats with the S1P lyase inhibitor (4-benzyl-phthalazin-1-yl)-2-methyl-piperazin-1-yl]-nicotinonitrile caused, besides protection from EAE and immunological alterations (see also the study by Weiler et al. 2014), protein-losing glomerulopathy due to podocyte dysfunction, skin irritation, and platelet activation and may also be associated with pathological alterations in other tissues such as lung, liver, thymus, and the RBC system.
Discussion
S1P lyase has been identified as an interesting target for the treatment of autoimmune diseases (Bagdanoff et al. 2010; Billich et al. 2013; Serra and Saba 2010; Weiler et al. 2014) and more recently also for muscle disorders (Saba and de la Garza-Rodea 2013). Therefore, it is of considerable interest to explore toxicological liabilities of this target, given the fact that its inhibition inherently gives rise to an increase in the bioactive lipid S1P in tissues. Mice deficient in S1P lyase succumb early in life to multiorgan failure (Schmahl et al. 2007; Vogel et al. 2009) likely caused by excessive amounts of S1P accumulated in all tissues and associated with a major derailment of innate immune functions and lipid metabolism (Allende et al. 2011; Bektas et al. 2010). Obviously, this apparent target-mediated toxicity would prevent the use of S1P lyase inhibitors in case this toxicity would also occur in humans and under conditions where the enzyme would only be partially inhibited, as will likely be the case with pharmaceutical inhibition.
Therefore, we conducted a comprehensive characterization of pathological alterations associated with partial deficiency or inhibition of S1P lyase in rodents. As major hazards, we identified severe kidney toxicity as well as skin irritation and platelet activation. Additional effects on lung, liver, and the RBC system, besides the expected immunological alterations, could not be excluded.
Reduced S1P lyase activity was consistently associated with glomerular proteinuria, most likely resulting from podocyte dysfunction due to reduced S1P lyase activity. The initial involvement of podocytes was demonstrated by electron microscopically characterized podocyte foot process effacement, resulting in proteinuria and associated clinical chemistry findings. S1P lyase activity in kidneys of partially S1P lyase–deficient mice was reduced by >90%, resulting in chronic intrarenal S1P concentrations of ∼10 pmol/mg (instead of 0.1 pmol/mg normally; Billich et al. 2013). C16 ceramide concentrations were not significantly changed in kidneys (Billich et al. 2013). Intrarenal S1P concentrations in rats treated with the S1P lyase inhibitor in the context of EAE for 16 days were moderately increased at 2 mg/kg bid (average 7.7 pmol/mg) and markedly increased at 10 mg/kg bid (average 165 pmol/mg). At 10 (but not 2) mg/kg bid C16 ceramide concentrations were also doubled (average 50.6 pmol/mg instead of 24.7 pmol/mg normally). However, despite this correlation, it is unclear whether these sphingolipids are causally involved in podocyte dysfunction. In an immortalized mouse podocyte cell line, messenger RNA expression of the S1P receptors S1P1, S1P2, S1P3, and S1P4 (but not S1P5) was detected, suggesting a role of S1P in podocytes (Awad et al. 2011). Furthermore, podocyte biology has been shown to be influenced by S1P-signaling pathways in rats in vivo (Awad et al. 2011) and in human podocytes in vitro (Ren et al. 2009). Hence, increased S1P concentrations in glomeruli may explain the observed podocyte dysfunction associated with partial deficiency or inhibition of S1P lyase.
The second consistent nonimmunological finding associated with reduced S1P lyase activity was skin epidermal acanthosis and hyperkeratosis with dermal inflammatory cell infiltrates. The alterations were more severe in rats having received 10 mg/kg bid of the S1P lyase inhibitor for 16 days in the EAE model than in partially S1P lyase–deficient mice. Although skin concentrations of S1P or ceramides were not determined in the S1P lyase inhibitor–treated rats, based on the differences in kidney S1P concentrations, it is assumed that the reduction in S1P lyase activity is generally more severe in the rats treated with 10 mg/kg bid of the S1P lyase inhibitor than in the partially S1P lyase–deficient mice. In the skin of partially S1P lyase–deficient mice, S1P concentrations increased and ceramide concentrations decreased, and it may be the balance between these sphingolipids that is impacting skin physiology in the case of reduced S1P lyase activity. Ceramides play an important role in maintaining skin barrier function (for review, see Wartewig and Neubert 2007), and interference with skin barrier integrity, for example, by reducing skin ceramide concentrations, may result in epidermal acanthosis and associated skin alterations. The concomitant increase in S1P in the case of reduced S1P lyase activity may keep these pathological alterations to a certain extent under control, as S1P has been described to inhibit epidermal acanthosis, to control nonallergic skin inflammation in mouse models (Hong et al. 2008; Schaper 2012), to inhibit keratinocyte proliferation, and to enhance keratinocyte differentiation in vitro (Kim et al. 2004; Vogler et al. 2003).
The third consistent nonimmunological finding associated with reduced S1P lyase activity was platelet activation characterized by increased expression of P-selectin on platelets. Additional investigations in partially S1P lyase–deficient mice further revealed increased concentrations of sP-selectin and mildly (statistically not significant) enhanced collagen-stimulated maximal platelet aggregation. Reduced S1P lyase activity was associated with moderately increased concentrations of S1P in blood (Billich et al. 2013). Additional investigations in partially S1P lyase–deficient mice further revealed increased concentrations of C16 ceramide in blood (in the context of an SRBC-induced DTH study). S1P via activation of the S1P2 receptor has been associated with human platelet activation and aggregation at sub-micromolar concentrations (Randriamboavonjy et al. 2009), whereas a link between naturally occurring ceramides and platelet activation or aggregation has not been established so far. Therefore, the increased S1P concentration in blood may play a role in platelet activation associated with reduced S1P lyase activity. Since platelet activation represents a key step in the formation of thrombi, and since S1P has been implicated in coagulation (for review, see Obinata and Hla 2012), reduced S1P lyase activity may facilitate or aggravate thrombosis. The impact of reduced S1P lyase activity on atherosclerosis, for which a role of platelet activation is also discussed (Lievens and von Hundelshausen 2011), remains unclear as multiple S1P receptors positively and negatively regulate atherosclerosis (Okamoto et al. 2011).
For a better understanding of the significance of other alterations observed in partially S1P lyase–deficient mice or in rats treated with an S1P lyase inhibitor in the EAE model, additional studies are required. For example, the mechanisms underlying the observed decrease in RBCs or the release of serum transaminases and alkaline phosphatase in rats treated with S1P lyase inhibitor in the EAE model remain unclear.
Conclusion
The results of this study demonstrate that inhibition of S1P lyase causes, besides immunological alterations and its protective effect in EAE (Billich et al. 2013; Weiler et al. 2014), podocyte-based protein-losing glomerulopathy, skin epidermal acanthosis, and hyperkeratosis with dermal inflammatory cell infiltrates, and platelet activation, and may also be associated with pathological alterations in other tissues such as lung, liver, and the RBC system. The similarity of findings between partially S1P lyase–deficient mice and rats treated with S1P lyase inhibitors in the context of EAE strongly supports the value of analyzing genetically modified partially target-deficient mice for target safety assessment. If the findings described in rodents translate to humans, particularly if human podocytes are similarly sensitive to reduced S1P lyase activity as rodent podocytes, target-related glomerular toxicity may limit chronic systemic treatment of autoimmune diseases with S1P lyase inhibitors. Furthermore, partial deficiency or inhibition of S1P lyase appears to provide an in vivo rodent model enabling studies on the mechanism of podocyte dysfunction and warrant further research on the role of sphingolipids in podocyte biology.
Footnotes
Acknowledgments
We thank the following Novartis colleagues for their excellent support: C. Debon, R. Schaffner, P. Scheubel, and S. Schoepfer (technical and experimental support); and D. Brees, P. Heining, and M. Kammüller (scientific support).
Author Contribution
Authors contributed to conception or design (JS, AW, AB, PM, UJ), data acquisition, analysis, or interpretation (JS, AG, DL, AW, AP, ES, SC, CB, PM, UJ); drafting the manuscript (JS, AW, UJ); and critically revising the manuscript (DL, AW, AP, SC, CB, FP, AB, PM, UJ); and gave final approval (JS, DL, AW, AS, AP, CB, AB, PM, UJ). All authors agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
