Abstract
Background
Salidroside (SAL) is an anti-inflammatory, antioxidant, anticancer, neuroprotective, and renal protective active ingredient extracted from the Chinese herb. Rhodiola Rosea. However, the role of SAL in kidney injury has not yet been elucidated. The study investigates SAL’s protective effect and mechanism in lipopolysaccharide (LPS)-induced kidney injury.
Methods
Male C57BL/6 wild-type mice (6–8 weeks old) were intraperitoneally injected with 10 mg/kg LPS for 24 h and SAL (50 mg/kg) 2 h before the LPS injection. Biochemical and TUNNEL staining assay analyses were carried out to assess kidney injury. The Elisa assay analyzed the mRNA expression of NGAL and KIM-1. RT-qPCR and Western blotting measured the mRNA and protein expression of HO-1, NQO1, Beclin1, P62, SIRT1, Nrf2, and PNCA, respectively.
Results
Our study found that mice co-treated with SAL had significantly reduced blood urea nitrogen (BUN), serum creatinine (Scr), neutrophil gelatinase-associated lipocalin (NGAL), and kidney injury molecule-1 (KIM-1) levels in serum of LPS-induced mice. SAL cotreatment potentially decreased the apoptosis rate of kidney tissue and podocytes induced by LPS. SAL significantly reduced the content of malondialdehyde (MDA) and increased superoxide dismutase (SOD) in LPS-treated mice. Autophagy-related proteins Beclin-1 increased but decreased P62 protein expression by cotreatment of SAL in LPS-injected mice. SAL enhanced the Sirtuin 1 (SIRT1) and nuclear factor erythroid 2-related factor 2 (Nrf2) protein expression in LPS-induced kidney tissues.
Conclusion
Our results speculate that SAL protects against LPS-induced kidney injury through activation of the SIRT1/Nrf2 pathway.
Introduction
Acute kidney injury (AKI), a condition of the systemic inflammatory response, is clinically characterized by critical tubular cell damage and sudden renal failure. 1 Recently reported that AKI is linked to systemic and intrarenal inflammation, resulting in reduced urine concentration and kidney damage. 2 Approximately half of the patients with AKI in bacterium-infected intensive care units (ICU) are thought to have severe sepsis or septic shock, which are now widely acknowledged as the principal causes of AKI in these patients.3,4 Due to high incidence rates, significant morbidity, and fatality rates associated with sepsis-induced AKI, this condition is becoming acknowledged as a major worldwide healthcare issue. 5 Unfortunately, despite considerable medical and therapeutic advancements, there is currently no effective way to prevent this fatal disease. 6 Therefore, it is essential to investigate new and potent therapeutic approaches to prevent this devastating disease.
Lipopolysaccharide (LPS), an endotoxin produced by bacteria that causes severe immunological and inflammatory reactions in animals, is reported to have a role in the pathophysiology of sepsis-induced AKI. 7 As a result, one of the most extensively utilized animal models to elucidate the processes underlying sepsis-induced AKI is LPS-induced AKI. 8 A growing evidence indicates that AKI is caused directly by the inflammatory response inherent to sepsis. 9 It has been shown that Sirtuin 1 (SIRT1), an NAD + -dependent class III histone deacetylase, is crucial for controlling cellular oxidative stress response, metabolism, and inflammatory disorders such as kidney disease by inhibiting the production of pro-inflammatory cytokines. 10 Previous study reported that activating SIRT1 might reduce the inflammatory response brought on by sepsis and protect the acute kidney injury. 11 A recent study showed that system xc− (a cystine-glutamate antiporter that is sodium-independent, chloride-dependent, and encoded by the SLC7A11 gene) activity and expression are mediated via the Nrf2 signal channel. 12 Moreover, researchers have shown that the SIRT1/Nrf2 pathway is associated with antioxidative stress.13,14 However, regulating the SIRT1/Nrf2 pathway could be a potential target to prevent LPS-induced kidney injury.
In traditional Chinese Medicine, Rhodiola is an herb found in western, northern, and subarctic parts of Asia. It is a member of the Rosaceae Crassulaceae family. 15 Salidroside (SAL), a tyrosol glycoside extracted from Rhodiola, is regarded as one of the active ingredients in Rhodiola for its antidepressant and anti-anxiety benefits. 16 Its molecular formula is C14H20O7, and its molecular weight is 300.3. 17 Different studies have shown that SAL has various pharmacological properties, including anti-inflammatory, anti-aging, anti-fatigue, and anti-oxidation properties.18,19 It is anticipated that SAL will emerge as a novel medicine for adjuvant cancer treatment due to its therapeutic properties in malignant tumors, including lung cancer, colorectal cancer, and kidney cancer. 20 However, the protective effect and mechanism of salidroside (SAL) on LPS-induced kidney injury is unclear. Therefore, this research aims to investigate and provide precise information about the therapeutic effects of SAL regarding LPS-induced kidney injury and to explore the specific molecular mechanism.
Materials and methods
Animal experiments
Male C57BL/6J mice (6–8 weeks old, 20–25 g in weight) were bought from Shanghai SLAC Laboratory Animal center. A total of 32 mice were randomly divided into four groups, with eight mice in each group. LPS (10 mg/kg, L2630, Sigma) was injected intraperitoneally into the mice, and the control mice were injected with 0.9% saline. For the LPS + SAL and SAL groups, the mice were pretreated with salidroside (SAL, 50 mg/kg, intraperitoneal injection, HY-N0109, MedChemExpress) 2 h before LPS administration. After 24 h, the mice were anesthetized by 1% pentobarbital sodium intraperitoneal injection and then sacrificed humanely, and then the blood and kidneys were harvested. The Ethics Committees approved (No. SSPH-009) the experimental animal procedures of our hospital.
Cell Culture
MPC5 mouse renal podocyte line was purchased from Tongpai Biotechnology Co. Ltd. Cells were cultured in an incubator (SANYO, Japan) under standard conditions (370C, 5% CO2); in RPMI-1640 (Gibco) medium containing 10% FBS (Gibco), 1% penicillin and streptomycin and recombinant murine IFN-γ (10 IU/mL). To induce differentiation, podocytes were maintained at 37°C without IFN-γ for 10–14 days. Four groupings of cells were treated for 24 h: control, LPS, LPS + SAL, and SAL. The control group cells received no intervention; cells in the LPS group were treated with LPS stimulation (5 μg/mL), cells in the LPS + SAL group were treated with LPS and SAL (100 μM), and cells in the SAL group were treated with SAL alone.
Biochemical analysis
Serum was collected to measure BUN (C013-2-1, Nanjing Jiancheng Bioengineering Institute, Nanjing, China) and Scr (C011-2-1, Nanjing Jiancheng) by colorimetric. The kidney tissue was collected and homogenized (10%, w/v), and the lysate was collected to measure MDA (S0131S, Beyotime, Shanghai, China) content, SOD (S0109, Beyotime), and CAT (S0051, Beyotime) activities using commercially available kits.
ELISA
In serum samples, the concentrations of NGAL (MLCN20, R&D Systems) and KIM-1 (MKM100, R&D Systems) were measured using ELISA kits. The 450 nm absorbance was measured in a microplate reader, and the cytokine concentrations were calculated using the corresponding standard curve.
TUNEL staining
The TUNEL apoptosis test kit (C1086, Beyotime, Shanghai, China) was utilized to identify apoptosis in isolated cells. Cells were cultured and treated with 4% paraformaldehyde in 6-well plates. Cells were washed with PBS, permeabilized with 0.3% Triton X-100, and then treated with 50 μL of TUNEL reaction fluid for an hour at 37°C. Then, the stained with DAPI cell nuclei were observed using an inverted microscope (IX51, Olympus, Japan).
Detection of ROS
Dihydroethidium (DHE; Beyotime Institute of Biotechnology; cat. no. S0063) staining was used to identify cellular ROS formation. Intracellular superoxide anions dehydrogenate DHE to produce ethidium, which enters the living cells and attaches to RNA or DNA to produce red fluorescence. The red fluorescence becomes more intense as intracellular superoxide production increases. In this study, isolated cells were initially seeded into 24-well plates at a density of 1 × 10
5
cells/well. The cells were subsequently treated with 100 nM selenium (SE) and 200 m
RT-qPCR
Using TRizol (Invitrogen, USA), total RNA was isolated from mouse kidney tissues. The reverse transcription technique created complementary DNA (cDNA) from total RNA. The mRNA was amplified by Real-Time Quantitative PCR (RT-qPCR) using SYBR Green reagent (TaKaRa, Japan) in an ABI Prism 7700 Real-Time PCR apparatus (Applied Biosystems, USA). The list of following primer sequences was used: HO-1 (forward: 5′-AAG CCG AGA ATG CTG AGT TCA-3′; reverse: 5′-GCC GTG TAG ATA TGG TAC AAG GA-3′); NQO1 (forward: 5′-AGG ATG GGA GGT ACT CGA ATC-3′; reverse: 5′-TGC TAG AGA TGA CTC GGA AGG-3′); GAPDH (forward: 5′-TCA ACA GCA ACT CCC ACT CTT CCA-3′; reverse: 5′-ACC CTG TTG CTG TAG CCG TAT TCA-3′). The qPCR results were examined using the 2-ΔΔCq technique. 21
Western blotting
RIPA buffer was used to extract the protein, and the lysates were centrifuged to produce the supernatants, which could then be used with a BCA protein assay kit to calculate the protein concentrations. Afterward, the protein was transferred to a PVDF membrane after the protein samples (50 μg) were loaded for electrophoresis on a 10% SDS-PAGE (Millipore, Bedford, MA). The 5% skim milk was used to the blocked membrane and dissolved in a TBS solution. The membrane incubated overnight at 4°C against Beclin1 (1:500, sc-48,341, mouse monoclonal, Santa Cruz), P62 (1:500, ab56416, mouse monoclonal, Abcam), SIRT1 (1:400, sc-74,465, mouse monoclonal, Santa Cruz), Nrf2 (1:500, ab92946, rabbit polyclonal, Abcam), GAPDH (1:1000, ab8245, mouse monoclonal, Abcam), and PNCA (1:1000, ab18197, rabbit polyclonal, Abcam). The immunological reactivity of these target proteins was identified using secondary antibodies that were HRP-conjugated (1:2000). The protein band was observed using ECL (Thermo, Waltham, MA, USA), and band density was measured using ImageJ software (Bio-Rad, Hercules, CA, USA).
Statistical analysis
The data is presented as mean ± SD and analyzed by SPSS20.0 statistical software. One-way ANOVA was applied to analyze the difference between three or more groups, followed by Student-Newman-Keuls tests. p < 0.05 was considered a significant difference.
Results
SAL improves kidney function in LPS-induced mice
To explore the protective effect of SAL on LPS-induced kidney injury, we assessed the renal function changes of mice. We evaluated the levels of blood urea nitrogen (BUN) serum creatinine (Scr), neutrophil gelatinase-associated lipocalin (NGAL), and kidney injury molecule-1 (KIM-1) in the serum of mice at 24 h after LPS injection. LPS administration significantly increased the levels of BUN, Scr, NGAL, and KIM-1, which were significantly attenuated by SAL cotreatment (Figures 1(a)–(d)). HE staining showed that precise and complete kidney tissues were lost in the LPS group, with ruptured glomeruli and inflammatory cell infiltration. After cotreatment with SAL, the pathological kidney damage was partially recovered (Figure 1(e)). SAL alleviates LPS-induced acute kidney injury. (a) Serum creatinine and (b) BUN levels at 12 h after LPS intraperitoneal injection in each group of mice. (c) Effect of SAL on the serum level of NGAL; (d) Effect of SAL on the serum level of KIM-1. (e) Representative HE staining images of kidney tissues. Data are presented as mean ± SD (8 mice per group). Compared to the control group, ***p < 0.001; compared to LPS group, ###p < 0.001.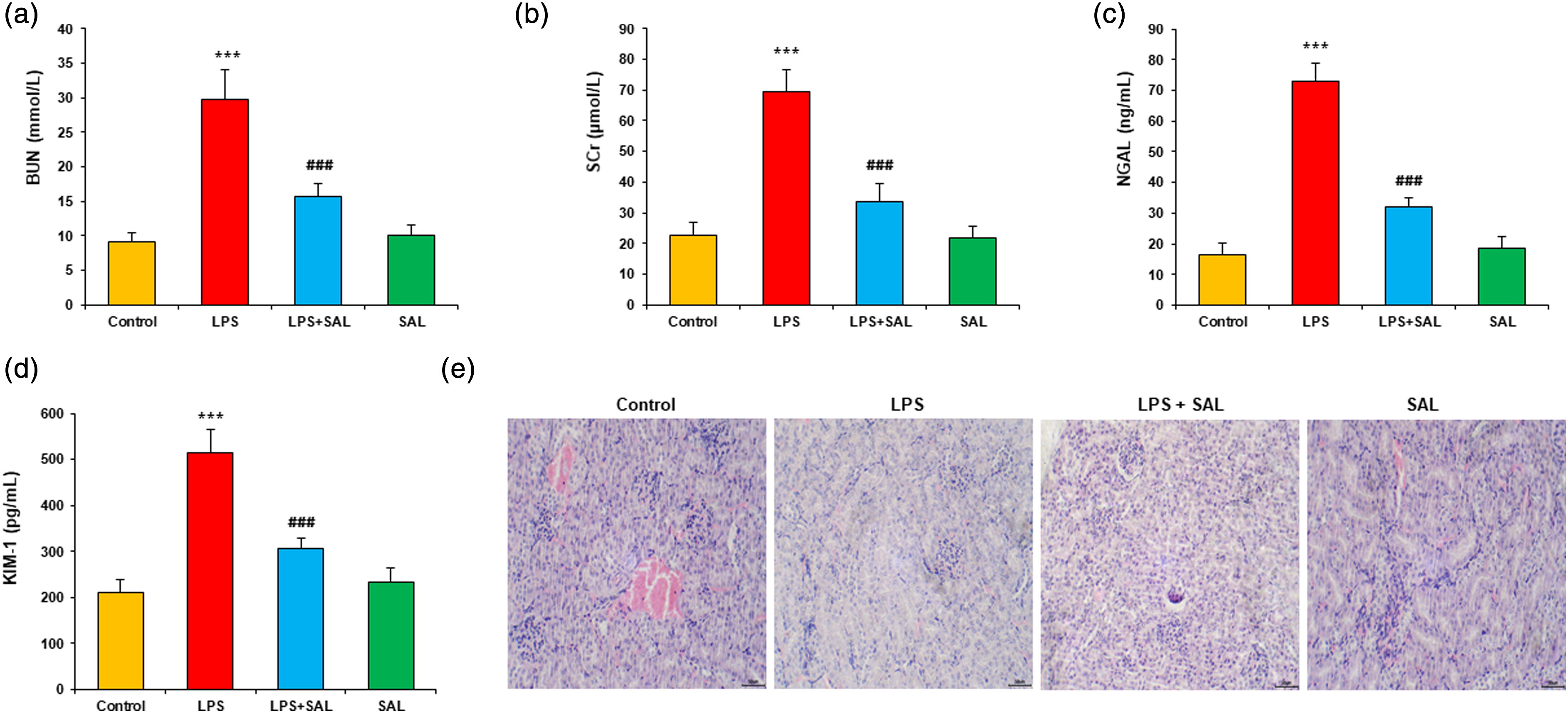
SAL inhibited LPS-induced apoptosis in kidney tissues and podocytes
The kidney tissue of mice was stained with TUNEL to evaluate apoptosis. Compared to the control group, the LPS group showed more green fluorescence (TUNEL+) in renal tissue, which was markedly reversed after SAL cotreatment (Figure 2(a)). Quantification analysis showed that SAL significantly reduced TUNEL + cells in LPS-treated mice but showed no change when used alone (Figure 2(b)). Podocytes were cultured and treated with LPS and SAL for 24 h. Annexin V/PI double staining was used to analyze the apoptosis of cultured podocytes. Compared with the control group, high LPS treatment significantly increased the apoptosis rate of podocytes. SAL cotreatment significantly reduced the apoptosis rate of podocytes induced by LPS (Figures 2(c),(d)). SAL inhibits LPS-stimulated apoptosis in kidney tissue and podocytes. (a) Representative images of TUNEL staining for each group. (b) The number of TUNEL-positive cells. (c) Flow cytometry was used for the assessment of cell apoptosis. (d) Quantification of apoptotic rate. Data are presented as mean ± SD (8 mice per group), and the cells experiment was performed in triplicates. Compared to the control group, ***p < 0.001; compared to LPS group, ##p < 0.01, ###p < 0.001.
SAL reduced ROS production in kidney tissue of LPS-induced mice
DHE staining was applied to kidney tissue and showed LPS significantly enhanced red fluorescence intensity. SAL cotreatment significantly attenuated the LPS-induced increase in DHE fluorescence intensity (Figures 3(a),(b)). Moreover, the oxidative stress proteins in renal homogenate were analyzed. SAL cotreatment significantly attenuated the LPS-induced increase in malondialdehyde (MDA) content and LPS-induced decrease in superoxide dismutase (SOD) (Figures 3(c),(d)). SAL inhibits LPS-induced ROS production and oxidative stress. (a) Representative images of DHE staining in kidney tissue (red fluorescence). (b) Quantification of DHE fluorescence intensity. The extent of oxidative stress was evaluated by measuring (c) MDA content and (d) SOD activity in kidney tissue lysates (normalized to protein). Data are presented as mean ± SD (8 mice per group). Compared to the control group, ***p < 0.001; compared to LPS group, ###p < 0.001.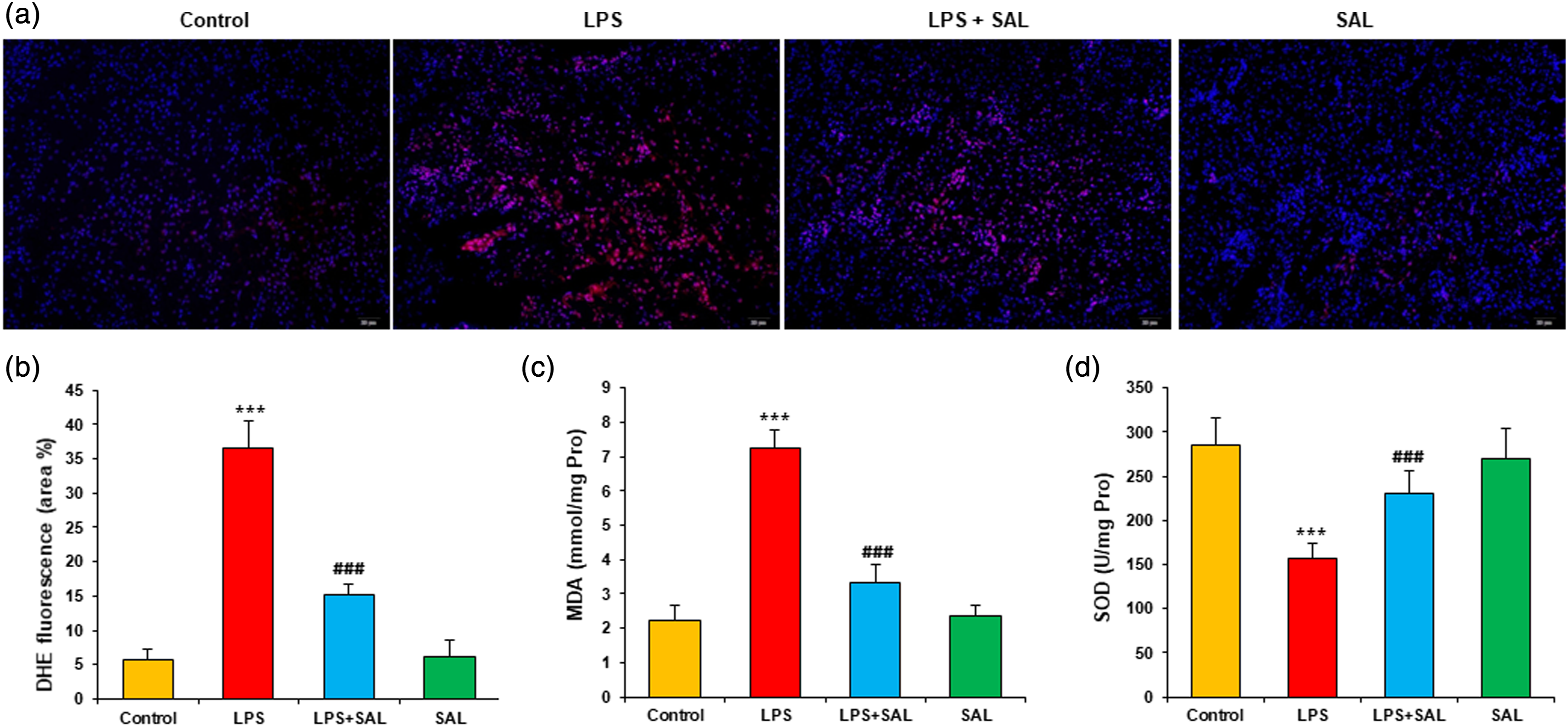
SAL promotes autophagy of podocyte stimulated by LPS
Podocytes were cultured and treated with LPS and SAL for 24 h. The degree of autophagy was evaluated by staining cells with LC3-II and DAPI. The FITC-positive green spots mainly show autophagosomes, while DAPI-positive blue spots represent nuclei. In the control group, podocytes showed autophagy to a certain extent, and LPS treatment inhibited podocyte autophagy, indicating a weakening of green fluorescence intensity (LC3-II spots). SAL cotreatment significantly enhanced autophagy and green fluorescence intensity (Figure 4(a)). Quantitative analysis showed that SAL significantly increased the percentage of LC3-II spotted cells (Figure 4(b)). Then Western blot was used to analyze the expression of autophagy-related proteins Beclin-1, and P62 (Figure 4(c)). LPS decreased Beclin1 protein level and increased P62 protein level in podocytes. SAL cotreatment markedly increased Beclin1 protein and decreased P62 protein expression (Figures 4(d),(e)). SAL promotes the autophagy of podocytes with LPS. (a) The cells were stained with LC3-II and DAPI. The cytoplasm of autophagy cells showed green fluorescence by LC3-II staining. (b) Each group’s proportion of autophagy cells was analyzed quantitatively (cells with LC3-II dots). (c) Representative gel blots depicting levels of autophagy-associated protein. Protein blots were quantified for (d) LC3-II and (e) p62. Cells experiment was performed in triplicates. Compared to the control group, ***p < 0.001; compared to LPS group, ###p < 0.001.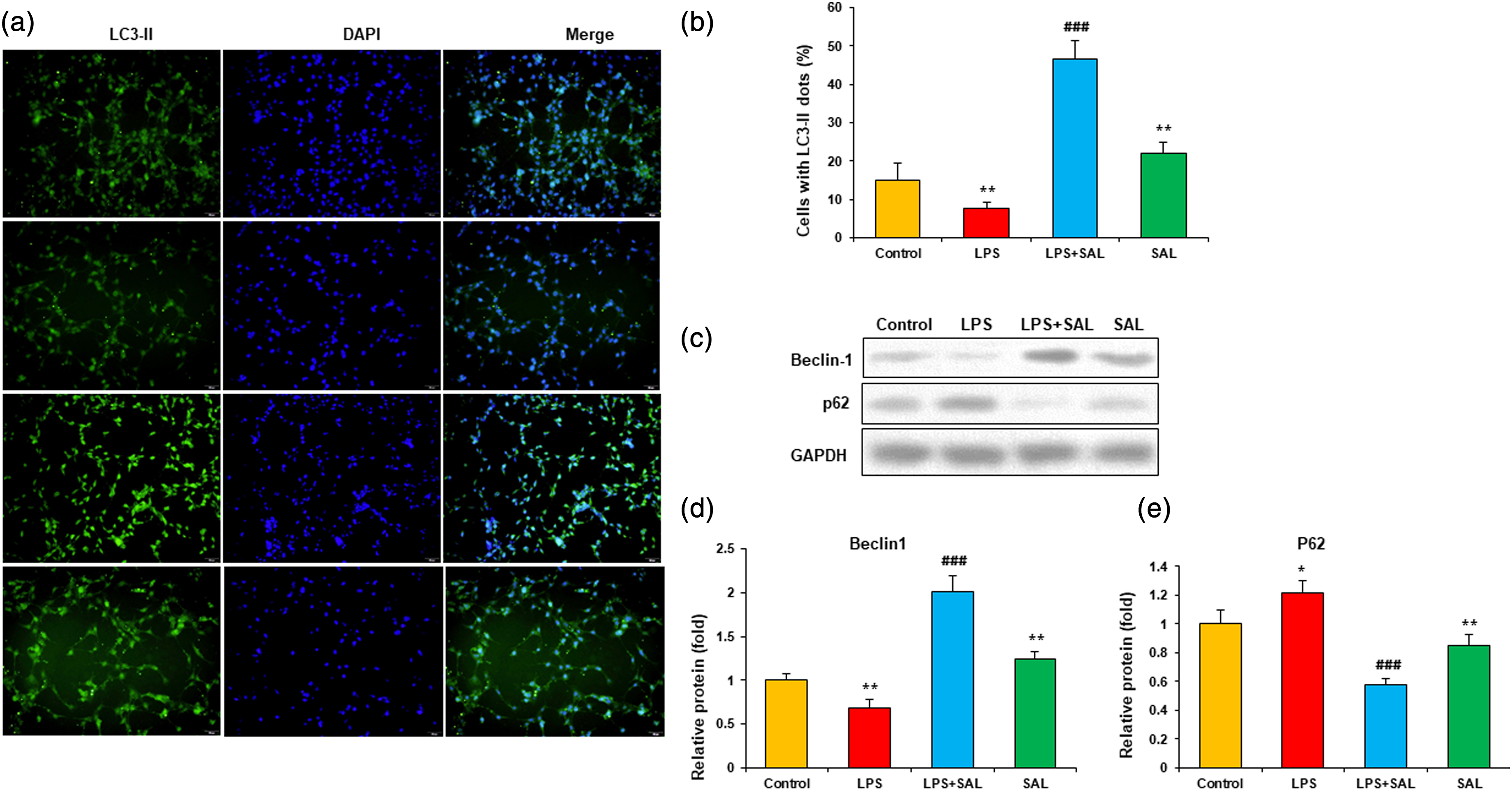
SAL activated SIRT1/Nrf2 pathway in LPS-induced kidney injury
Western blot was performed to analyze the protein expression of SIRT1, and Nrf2 in the kidney tissue of mice (Figure 5(a)). LPS decreased SIRT1 expression in kidney tissue, and SAL further increased SIRT1 expression with or without LPS (normalized to GAPDH protein) (Figure 5(b)). In addition, SAL also significantly enhanced nuclear Nrf2 protein expression in LPS-induced kidney tissues (normalized to PCNA protein) (Figure 5(c)). RT-qPCR was applied to analyze the expression of two Nrf2 downstream genes. SAL significantly increased the mRNA expression of HO-1 and NQO1 in kidney tissues of mice with LPS injection (Figures 5(d),(e)). In mice with LPS injection, SAL also increased HO-1 and NQO1 mRNA expression, but to a much lesser extent. SAL activates SIRT1 and Nrf2 pathways. (a) Representative gel blots of SIRT1 and Nrf2. These protein blots were quantified for (b) SIRT1 (normalized to GAPDH) and (c) nuclear Nrf2 (normalized to total PCNA). (d,e) The mRNA expression of Nrf2 downstream gene HO-1 and NQO1 (normalized to GAPDH). Data are presented as mean ± SD (8 mice per group). Compared to the control group, ***p < 0.001; compared to LPS group, ###p < 0.001.
Discussion
There is growing evidence that SAL has a wide range of pharmacological effects. Specifically, SAL exhibits antioxidant, anti-inflammatory, anticancer, neuroprotective, and renal protective properties both in vitro and in vivo. Our study reveals the protective effect of SAL and its potential mechanisms on LPS-induced kidney injury. The major findings are as follows: (1) SAL ameliorated pathological damage of the kidney both in vitro and in vivo. (2) SAL potentially attenuated apoptosis in kidney tissues and podocytes in LPS-induced mice. (3) SAL inhibited ROS production in kidney tissue and enhanced the autophagy of podocytes. (4) SAL significantly protected LPS-induced kidney injury via activation of the SIRT1/Nrf2 pathway.
AKI is a well-known clinical entity with a fast deterioration in renal function that contributes to multiorgan failure in severely sick patients. AKI is associated with several comorbidities. 22 The kidney functions of all groups were examined by assessing the BUN and Scr contents and observing kidney histopathological sections stained with H&E in mice with LPS-induced AKI. Severe renal pathological damage, including glomerular atrophy, tubular cell vacuolation, tubular dilation and distortion, tubular cell necrosis, and nuclear loss, occurred in the mice injected with 10 mg/kg LPS. These modifications came with an increase in the levels of Scr and BUN. However, SAL significantly inhibited these LPS-associated pathological changes, which could also decrease the Scr and BUN levels in mice with AKI. Therefore, it was initially concluded that SAL had a protective effect on the kidney function of mice with LPS-induced AKI.
A key mechanism for the occurrence and progression of AKI is apoptosis. 23 Studies revealed that inflammation and hypoxia are directly associated with increased apoptosis in kidney tissues and podocytes.24-26 We tested the apoptosis in kidney tissues and podocytes in the current research to investigate the effect of SAL on the apoptosis of the kidney. We observed that SAL significantly attenuated the apoptosis rate of kidney tissues and podocytes and inhibited the expression of the pro-apoptotic genes. Therefore, we demonstrated that SAL could potentially reduce the apoptosis level of kidney tissues and podocytes induced by LPS.
Most aerobic organisms are produced reactive oxygen species (ROS), and the concentration of ROS influences their biological function. Low concentrations of ROS are inevitable for physiological processes under normal conditions. However, excessive amounts of ROS produced during pathological conditions. 27 High levels of ROS can destroy cellular macromolecules, including proteins, lipids, and nucleic acids resulting in cellular damage. 28 Aerobic organisms have developed a robust antioxidant defense mechanism to protect against ROS-induced cellular damage. 27 In present study showed that significantly higher MDA concentration and significantly lower SOD protein expression in kidney tissue were observed in the LPS group compared with the control group (Figure 3). These findings suggest that excessive ROS during kidney injury depletes these antioxidant enzymes. In addition, SAL cotreatment SOD expression potentially increased. At the same time, MDA significantly decreased compared with the LPS group (Figure 3). These observations indicate that SAL cotreatment reduces the ROS level via upregulating the SOD protein expression.
The glomerular capillary basement membrane (GBM) and podocytes depend on autophagy to remain healthy. Studies have demonstrated that podocytes display downregulated phagocytic activity in various glomerular disorders, including diabetic nephropathy, membrane nephropathy, and focal segmental glomerulosclerosis.29-31 Moreover, the podocyte-related molecules maintain the glomerular filtration barrier. 32 The present study reveals the role of LPS-induced autophagy in the SAL cotreatment of podocytes. We found that LPS-induced podocytes showed less autophagy compared with the control group, while SAL cotreatment significantly enhanced autophagy (Figure 4). Moreover, autophagy-related Beclin1 and P62 protein expression levels were significantly increased by the cotreatment of SAL (Figure 4). These data indicated that SAL cotreatment promotes autophagy of podocytes induced by LPS.
A member of the mammalian sirtuin family, SIRT1 is a nicotinamide adenine dinucleotide (NAD+)-dependent histone deacetylase closely associated with biological processes, including cell metabolism, apoptosis, aging, inflammation, and oxidative stress. 33 Several studies have shown that SIRT1 is an essential regulator of inflammation, apoptosis, and antioxidant defense mechanisms.34-36 In addition, a recent study reported that SIRT1 might upregulate the Nrf2 expression to decrease ROS production. 14 Growing evidence suggests that Nrf2 is a crucial regulator for the function of system xc− and that severe oxidative stress significantly reduces Nrf2 and impairs homeostasis.12,14 In this study, we observed that SIRT1 and nuclear Nrf2 protein expression were upregulated in LPS-induced kidney injury via SAL cotreatment (Figure 5). Additionally, SAL cotreatment significantly increased the expression of the two Nrf2 downstream genes HO-1 and NQO1 in kidney tissues of mice with LPS injection (Figure 5). These results demonstrated that SAL inhibited LPS-induced kidney injury via activation of the SIRT1/Nrf2 pathway.
Conclusion
In our study, we showed that SAL has a potential protective effect on kidney function and pathological damage, and the specific mechanism of SAL is closely associated with its anti-apoptotic and anti-ROS production. Moreover, SAL significantly attenuated LPS-induced kidney injury by enhancing autophagy and activating the SIRT1/Nrf2 pathway (Figure 6). However, the pharmacological procedure of SAL is complicated and exerts various purposes. In addition, it is essential to extensively investigate the precise molecular mechanism of SAL in treating kidney injury at the cellular and animal levels, which provides more support for clinical trials. Model illustration on SAL-mediated protection on LPS-induced kidney injury, whose mechanisms are related to the regulation of SIRT1/Nrf2 pathways.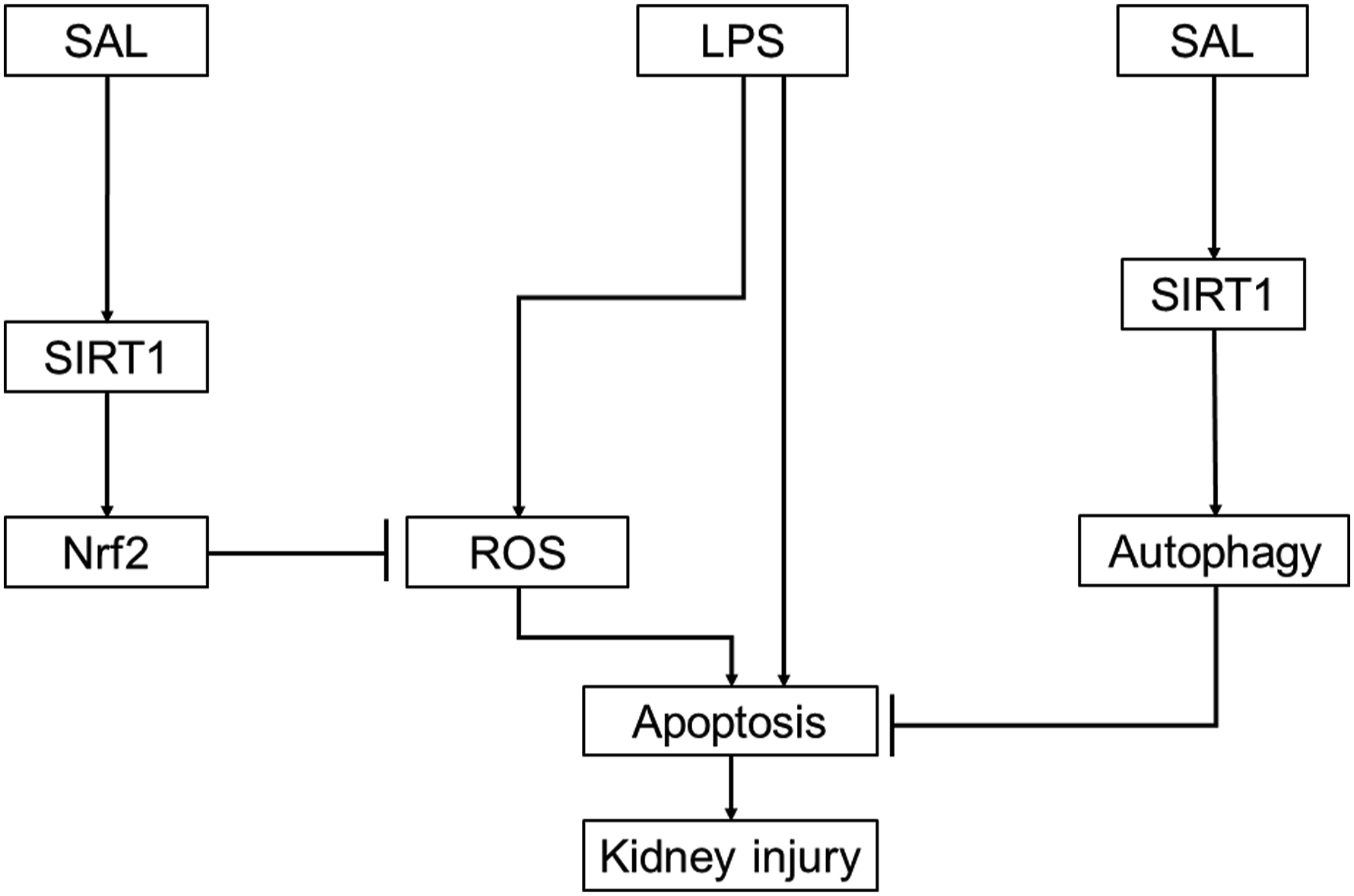
Footnotes
Authors Contributions
ZX designed the project, supervised the project, and revised the manuscript. JP and JZ have performed experiments and wrote the first draft of the manuscript. LL helped perform the experiments and collect data. TZ are analyzed the data and performed the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by (1) Shanghai Pudong New Area Health System Key Specialty Construction Project (PWZzk2022-15); (2) Shanghai Health Commission Research Project (202040143); (3) Shanghai Seventh People’s Hospital "Beidouxing" Talent Training Project (BDX2021-03).
Ethics approval and consent to participate
All experimental protocols were approved (No. SSPH-009) by the Experimental Animal Ethics Committee of the Seventh People’s Hospital of Shanghai University of Traditional Chinese Medicine. All methods were carried out in accordance with the National Institute of Health Guidelines for Care and Use of Laboratory Animals in Biomedical Research. All methods were reported in accordance with ARRIVE guidelines for the reporting of animal experiments.
Availability of data and materials
All data generated or analysed during this study are included in this article. Further enquiries can be directed to the corresponding author.
