Abstract
This review focuses on the anatomy, histologic preparation, and pathologic evaluation of extraparenchymal bile and pancreatic ducts (BPDs) and their openings at the duodenal papillae in the cynomolgus macaque (Macaca fascicularis), the Beagle dog (Canis familiaris), the Wistar Hanover rat (Rattus norvegicus), and the CD1 mouse (Mus musculus). In nonclinical safety assessment, intraparenchymal BPDs (with sections of liver and pancreas, respectively) are evaluated routinely. However, detailed evaluation of the extraparenchymal BPDs or the duodenal papillae is not included. In the context of nonclinical safety assessment studies, this review describes situations in which evaluation of extraparenchymal ductal structures and duodenal papillae may be useful in characterizing test article–related changes; elucidates anatomic similarities between human, macaque, and dog and notable differences in rats and mice; and consolidates the information required for the histopathologic evaluation of these tissues.
Keywords
Introduction
Oral gavage, which simulates ingestion of a tablet or pill, is a very common route of administering therapeutics. After being absorbed in the gastrointestinal tract and metabolized by the liver, many xenobiotics (or their metabolites) are excreted in bile. A higher intrabiliary concentration of toxic compounds raises the possibility of biliary toxicity (Levine 1981). For compounds that undergo enterohepatic recirculation, toxicity of bile ducts, associated pancreatic ducts, or the openings of these ducts at the duodenal papillae could be enhanced. This may be a result of repeated exposure, chemical transformation during the process of recirculation, or anatomic/physiologic changes in ducts over time (Roberts et al. 2002; Hegyi and Petersen 2013).
Toxicity of Extraparenchymal Biliary and Pancreatic Ducts and Duodenal Papillae
In certain conditions, effects of xenobiotics may be either specific to or more severe in the distal portion of the biliary and pancreatic ductal trees, that is, the extraparenchymal ducts and their openings at the duodenal papillae, as compared to intraparenchymal ducts. The existing literature includes examples in humans and other species, although the mechanism of action is not elucidated in all cases. In humans, examples of xenobiotic effects in these extraparenchymal tissues include increased resistance of the sphincter of Oddi impeding bile flow at the major duodenal papilla with pentazocine, a narcotic analgesic (Staritz et al. 1986), and extrahepatic biliary stenosis after hepatic arterial infusion of floxuridine, an antimetabolite chemotherapeutic (Aldrighetti et al. 2001). In dogs, relaxation of the sphincter of Oddi by an antispasmodic agent (n-ethyl-3-piperidyldiphenylacetate hydrochloride) being developed to replace atropine was described as a pharmacologic effect (Chen and Beckman 1952). In rats, macroscopic enlargement and histologic mucosal hyperplasia of the common bile duct were noted in rats fed a vitamin A-deficient diet containing polybrominated biphenyl (Darjono, Stowe, and Aust 1983). Occasionally, to better capture target pathology and improve risk assessment, such changes have been described in safety assessment packages as well. One example is that of dilatation of the extraparenchymal bile ducts along with bile duct hyperplasia, cholangitis, necrosis, and pericholangitis described with amlexanox, an anti-inflammatory and antiallergic immunomodulator (Center for Drug Evaluation and Research approval package for amlexanox oral paste 1996). Figures 1 and 2 provide examples of observations in a few nonclinical studies, where macroscopic and histologic changes indicated that evaluation of the extraparenchymal biliary and pancreatic ducts (BPDs) and their openings into the duodenum was appropriate.
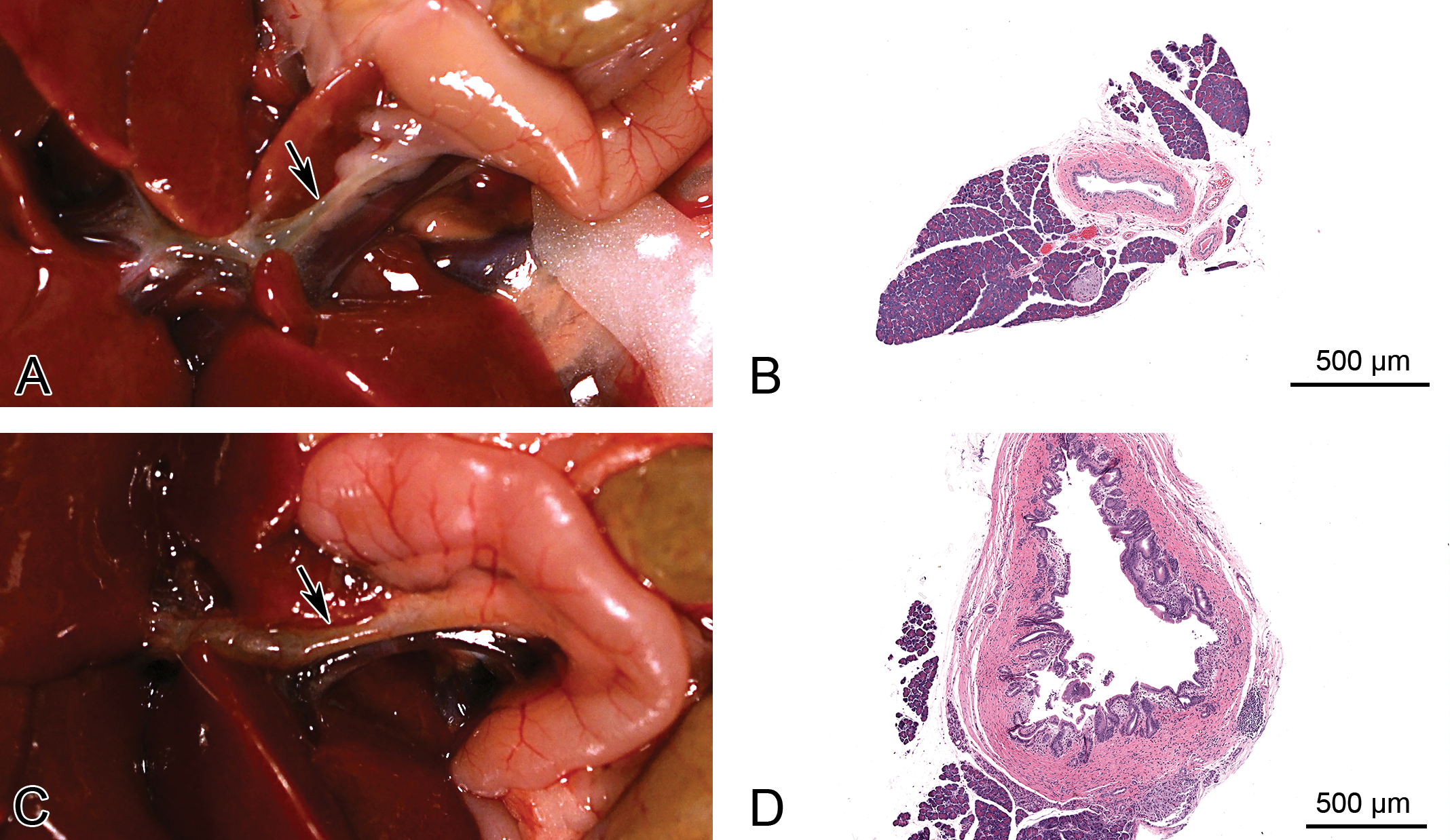
Common bile duct (arrow) in rat (a species that lacks the gall bladder). (A and B) normal duct in an untreated rat. (C and D) Dilated duct with mural thickening and leukocytic infiltrate in a rat treated with a xenobiotic. (B and D) H&E stain.
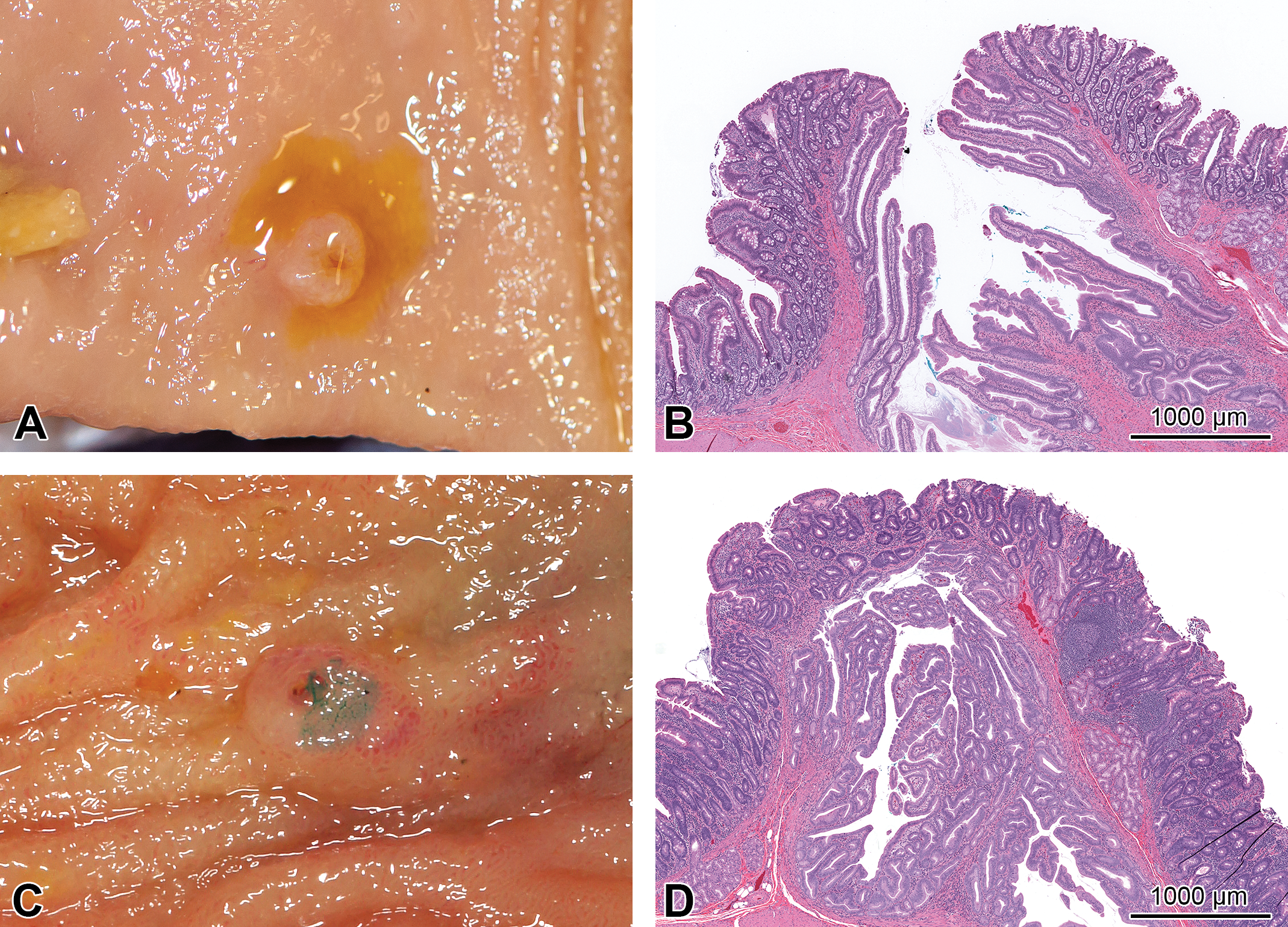
Major duodenal papilla in a macaque. (A and B) Normal papilla in an untreated macaque with background leukocytic infiltrate. (C and D) Macroscopic reddening of the major papilla (stained at necropsy with green dye to aid identification at trimming) due to congestion and expansion of the lamina propria with increased leukocytic infiltrate associated with inflammation of the duodenal papilla in a macaque treated with a xenobiotic. (B and D) H&E stain.
Depending on the severity, inflammatory changes resulting from these toxic effects may range from localized changes that may extend from the point of initiation in the duct(s) to tissues around these ducts (such as the pancreas), to rupture of the duct(s), and to subsequent peritonitis by leakage of bile. Some causes of toxicity specific to the extraparenchymal ducts include the following: 1. Anatomic features: At the level of the duodenum, the sphincter of Oddi prevents not only the release of bile and pancreatic secretion into the duodenum but also reflux of intestinal contents into the common bile duct and/or the pancreatic duct (Allescher 1989; Woods et al. 2005). Changes in neural input, hormonal input (especially cholecystokinin), nitric oxide activity, smooth muscle activity, or changes due to regional inflammation may alter the function of the sphincter (Toouli 2009). A dysfunctional sphincter of Oddi predisposes the distal extraparenchymal BPDs to pathologic changes. Near the duodenum, since pancreas surrounds the distal extraparenchymal BPDs, pancreatitis can predispose to spread of inflammation from the pancreatic lesion to the distal extraparenchymal BPDs or even cause obstruction in chronic cases. Further, the absence of valves in the common bile duct allows bile to flow in either direction. Accordingly, the distal BPDs (the two duct systems being closely associated) can themselves serve as a conduit for the spread of a toxic lesion and for secondary ascending infections with intestinally derived microbes once innate immunity provided by the sphincter of Oddi and/or the lining epithelium is breached.
In species that have a gall bladder, higher concentrations of a xenobiotic or metabolite may result in more severe changes in the gall bladder, where bile is concentrated (Greaves 2012). Rats lack a gall bladder and are known to physiologically “compensate” by enlargement of the duct system (Oldham-Ott 1997). In rats, it may be important to determine whether a change like ductal dilatation (Figure 1) is pathologic (secondary to outflow obstruction) or physiologic (compensatory) by considering accompanying clinicopathologic changes (Barber-Riley, Chan, and Tong 1967). 2. Biologic/physiologic variations: Across species, there is significant heterogeneity in biological function and physiologic interactions of lining epithelium at various levels in BPDs (Strazzabosco and Fabris 2008; Hegyi et al. 2011; Tabibian et al. 2013). Thus, cells in the extraparenchymal BPDs behave differently from cells lining the intraparenchymal ducts. The differences relate to a variety of cellular functions including but not limited to secretion, absorption, sensitivity to hormones, metabolic activity, cell signaling, and immunological activity of the lining cell. Moreover, the embryonic origins of intra- and extrahepatic ducts are different (Lemaigre 2009). The extrahepatic biliary tree develops from an outpocket of the endoderm in the embryonic foregut, located caudal to the liver. Thus, when considering toxic changes, unlike hepatoblast-derived cholangiocytes lining intrahepatic bile ducts, cholangiocytes lining the extrahepatic ducts may share similarities to duodenal enterocytes. As a corollary, pathologists should consider possible changes in the extrahepatic bile duct when a duodenal lesion is observed. 3. Chemical alterations: Alteration in the milieu of ductal/duodenal contents by enzymatic pancreatic secretion at the point where pancreatic secretion is released into the common bile duct or the duodenum (depending on the species being evaluated) or when biliary and pancreatic secretions get admixed with duodenal contents at the duodenal papillae may result in a change in the chemical reactivity of the xenobiotic or its metabolite(s) predisposing these regions to toxicity. Changes in pH, changes in concentrations of bile, which may itself be toxic, and changes in the amount of mucin being produced by lining epithelia may further impact the toxicity (Hegyi and Petersen 2013).
This article reviews available literature, provides information required for gross and histologic evaluation of the extraparenchymal BPDs and their openings into the duodenum, and also provides recommendations for safety assessment studies. Data in this article were gathered posteuthanasia at necropsy in nonclinical toxicology studies involving the use of animals. All animals were humanely treated and necropsied per standard techniques. All studies were conducted in accordance with institutional and governmental guidelines for the use and care of animals.
A literature search within the PubMed (MEDLINE) database was conducted (on and before September 23, 2014), searching for relevant literature. The following key words were used, alone or in combination, both as MESH terms and as free text words: macaque, dog, canine, mouse, rat, rodent, extraparenchymal, extrahepatic, bile duct, pancreatic duct, common bile duct, biliopancreatic, ampulla of Vater, duodenal papilla, sphincter of Oddi, toxicity, enterohepatic recirculation, anatomy, physiology, histology, embryology, Brunner’s glands, tissue list, guideline, safety assessment, biomarker, and imaging. Selected articles were screened in order to find out whether they could be valuable for this review, and if so, to determine the relevance of the articles. References cited within these articles were also screened and included when relevant.
Review of Gross Anatomy
The anatomy of extraparenchymal BPDs and their openings at the duodenal papillae is similar in human, macaque, and dog (Figures 3 and 4). It is somewhat different in rats and mice (Figure 3). Although anatomic variations exist in all species, our attempt here is to provide a brief, practical overview of the common anatomic presentations of extraparenchymal BPDs and duodenal papillae in adult animal species evaluated in safety assessment protocols and to aid the gross dissection and histologic processing of these structures. For further details, readers are encouraged to review the cited literature.
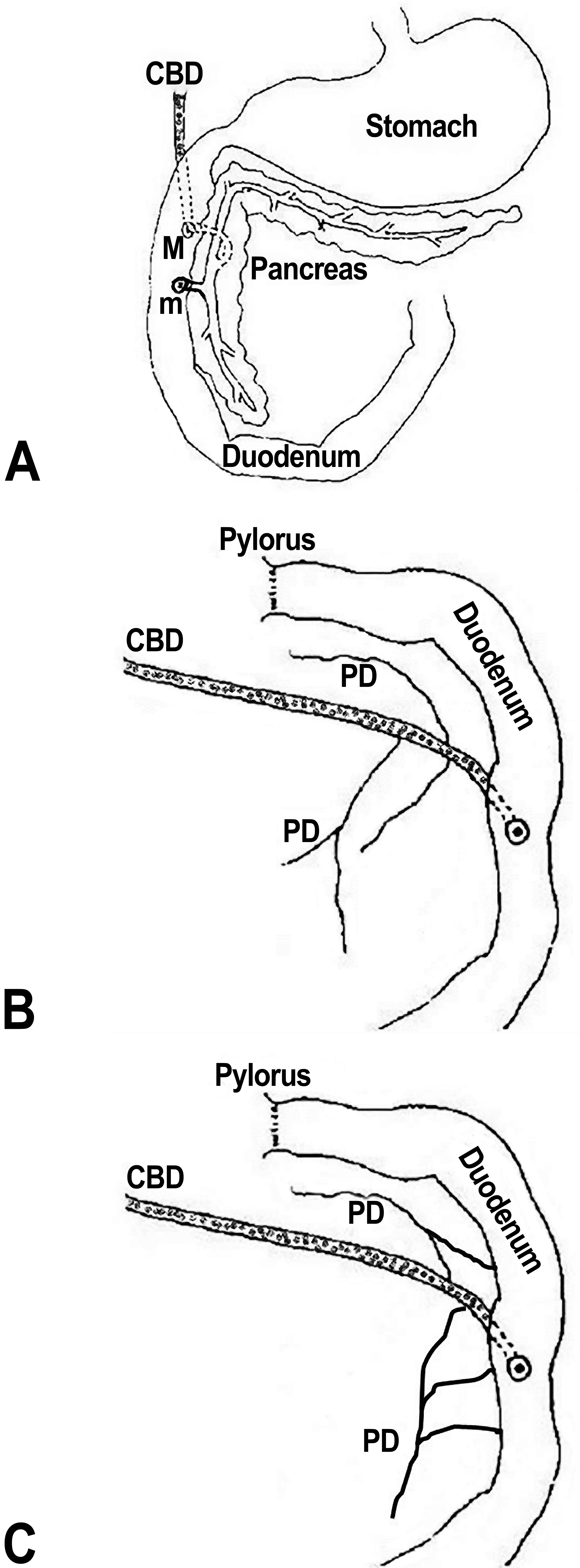
Extraparenchymal bile and pancreatic ducts. (A) Macaque and dog; (B) rat; (C) mouse (modified from Farinon et al. 1975). In rats and mice, the pancreatic ducts join the common bile duct before it enters the duodenum at the major duodenal papilla. Additionally, in mice, multiple smaller accessory pancreatic ducts release their contents directly into the duodenum, at one or more minor duodenal papilla(e). CBD = common bile duct; M = major duodenal papilla; m = minor duodenal papilla; PD = pancreatic duct.
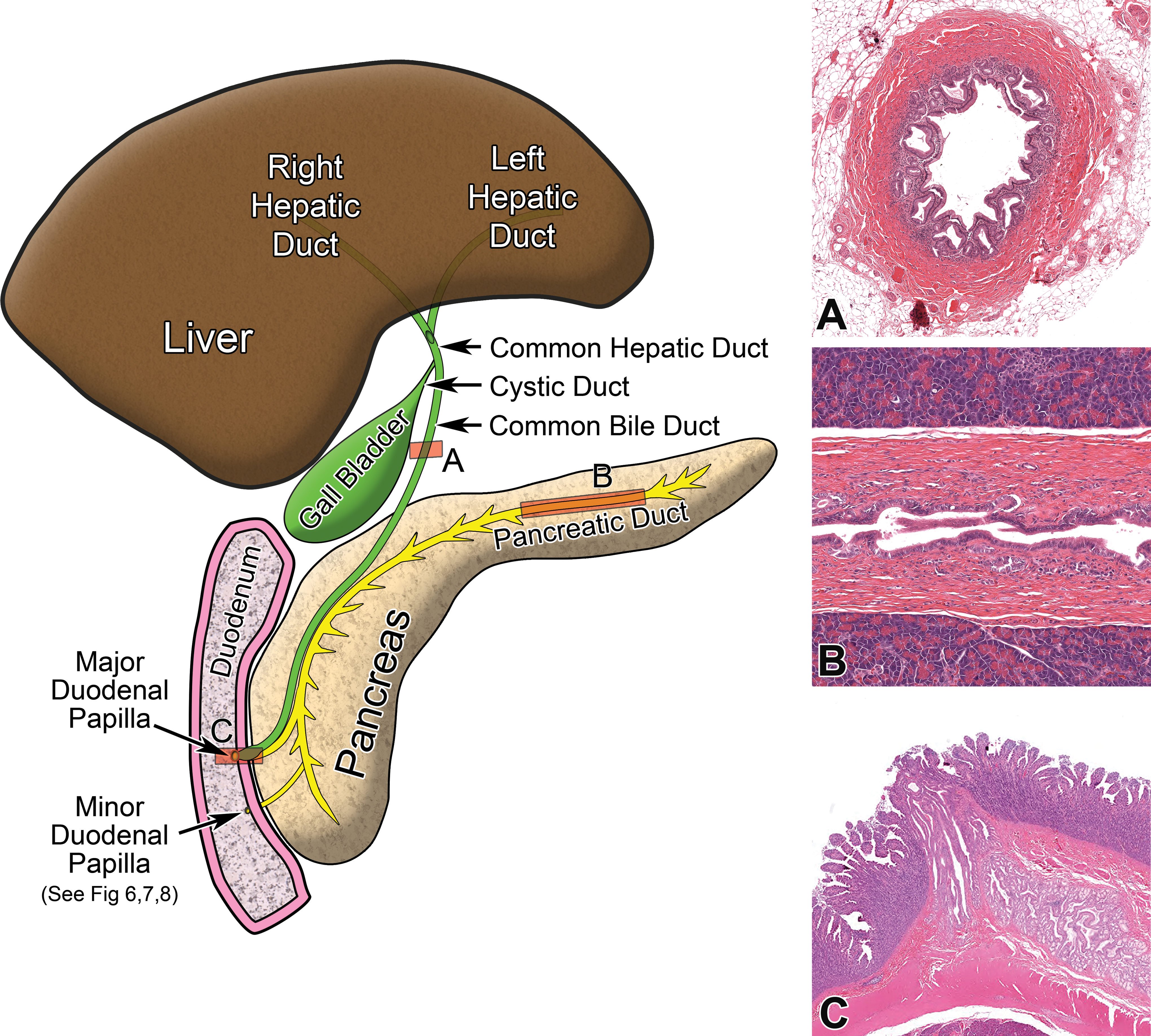
Diagram to illustrate various segments of the extraparenchymal biliary and pancreatic ducts and the duodenal papillae that can be sampled in a dog. Insets (A, B, and C) represent H&E-stained histologic sections taken from a few of these tissues.
Similar to human, in macaques and dogs, the hepatic and cystic ducts join to form the common bile duct (Figures 3A, 4, and 5), which along with the pancreatic duct releases contents into the duodenum at the major duodenal papilla. The major papilla is located approximately 4.5 to 12.5 cm distal to the pylorus in human, 1.5 to 3 cm distal to the pylorus in macaques, and 2 to 4 cm distal to the pylorus in dogs (Karski and Kamiński 1965; Farinon, Lampugnani, and Berri 1975). There is minor variation in the termination of the BPDs at the major duodenal papilla. In humans and macaques (Karski and Kamiński 1965), the major pancreatic duct and the common bile duct open into a sac-like structure called the ampulla of Vater (Figure 6A). The mixture of contents in the ampulla then gets released into the duodenal lumen through the major duodenal papilla. Dogs lack an ampulla (Farinon, Lampugnani, and Berri 1975), and the bile duct terminates on the major duodenal papilla alongside the pancreatic duct (Figure 7A).
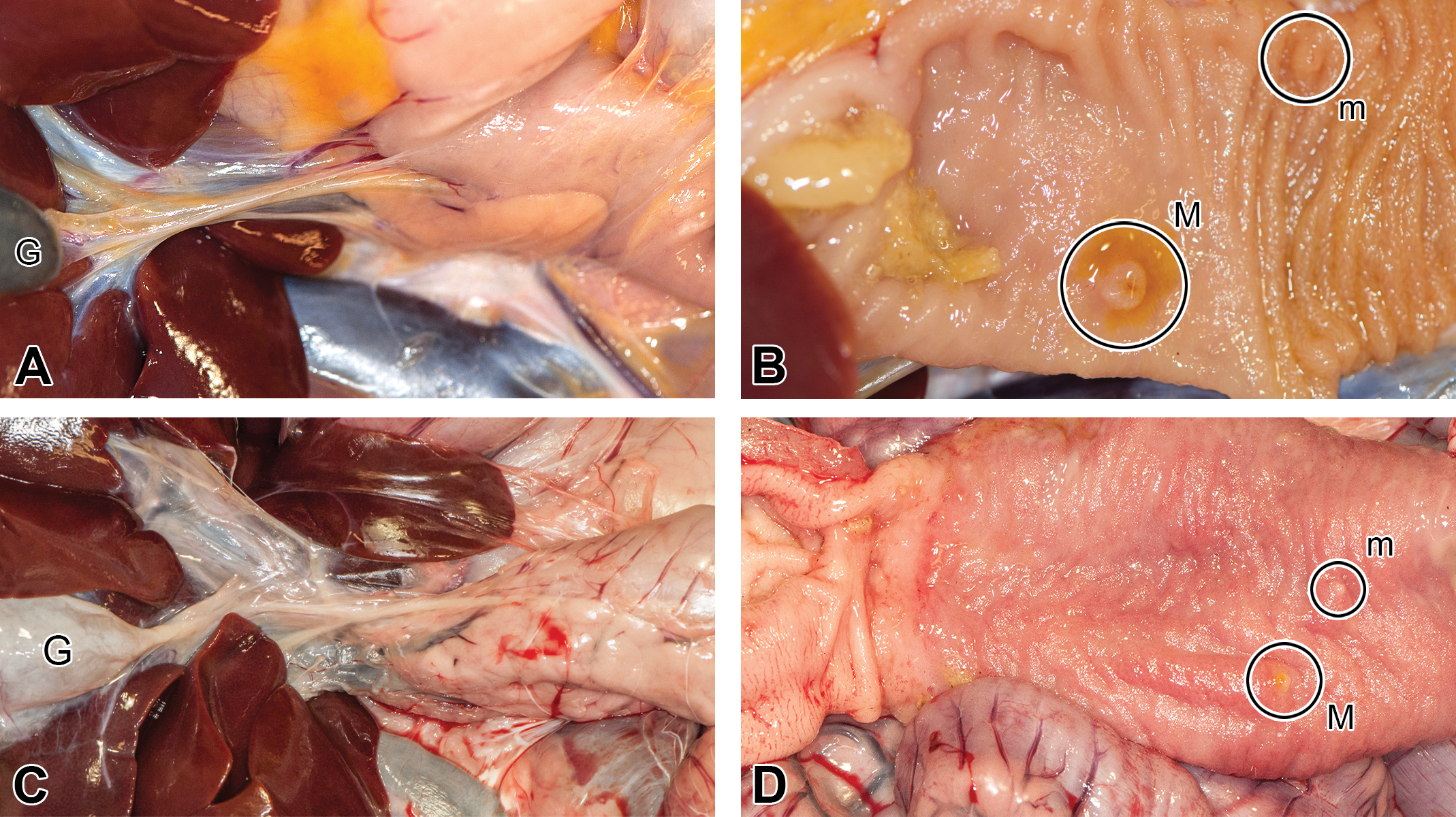
Extrahepatic bile ducts and duodenal papillae in non-rodents. (A and B) Macaque; (C and (D) dog. M = major duodenal papilla; m = minor duodenal papilla; G = gall bladder.
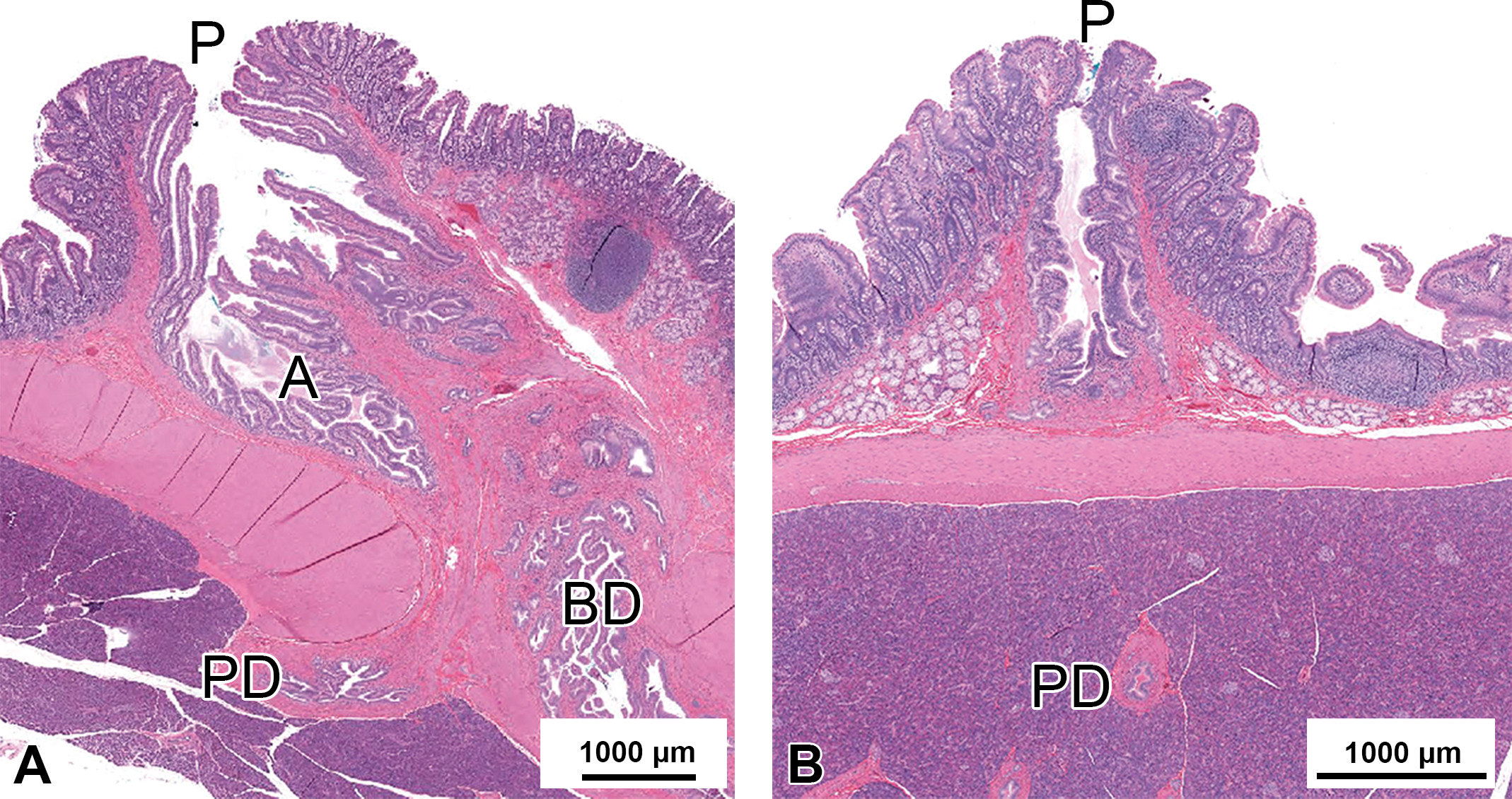
Duodenal papillae in a macaque (H&E stain). (A) Major duodenal papilla; (B) minor duodenal papilla. A = ampulla of Vater; BD = bile duct; P = papilla; PD = pancreatic duct.
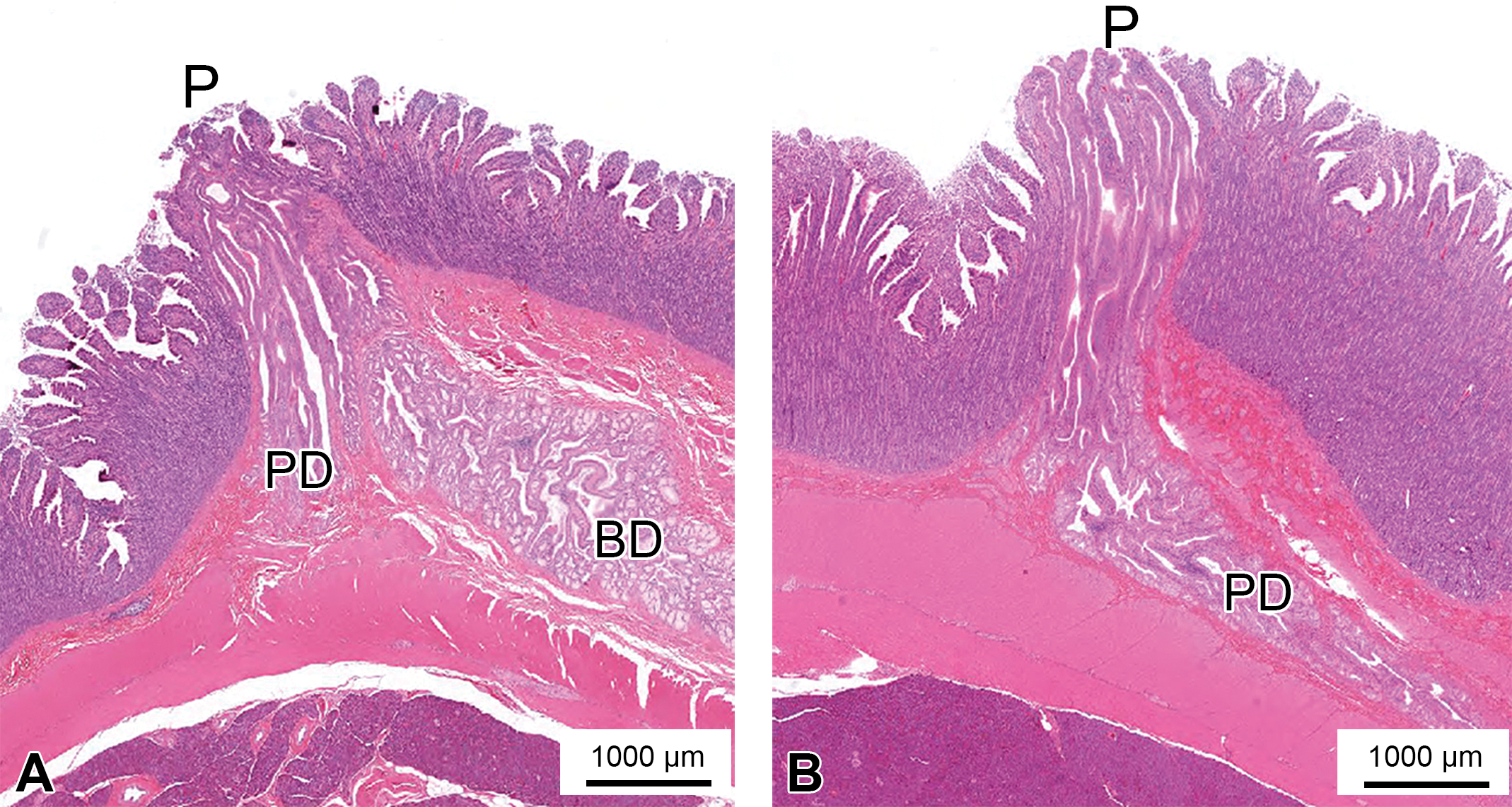
Duodenal papillae in a dog (H&E stain). (A) major duodenal papilla; (B) minor duodenal papilla. BD = bile duct; P = papilla; PD = pancreatic duct.
In humans, macaques, and dogs, the accessory pancreatic duct terminates on the minor duodenal papilla (Figure 4). In human, in a majority of the cases, the minor duodenal papilla is located 1.5 to 2.5 cm proximal to the major duodenal papilla (Karski and Kamiński 1965; Shia et al. 2014). In macaques and dogs, the minor duodenal papilla, in most cases, is located about 1 to 2 cm distal to the major duodenal papilla (Karski and Kamiński 1965; Farinon, Lampugnani, and Berri 1975).
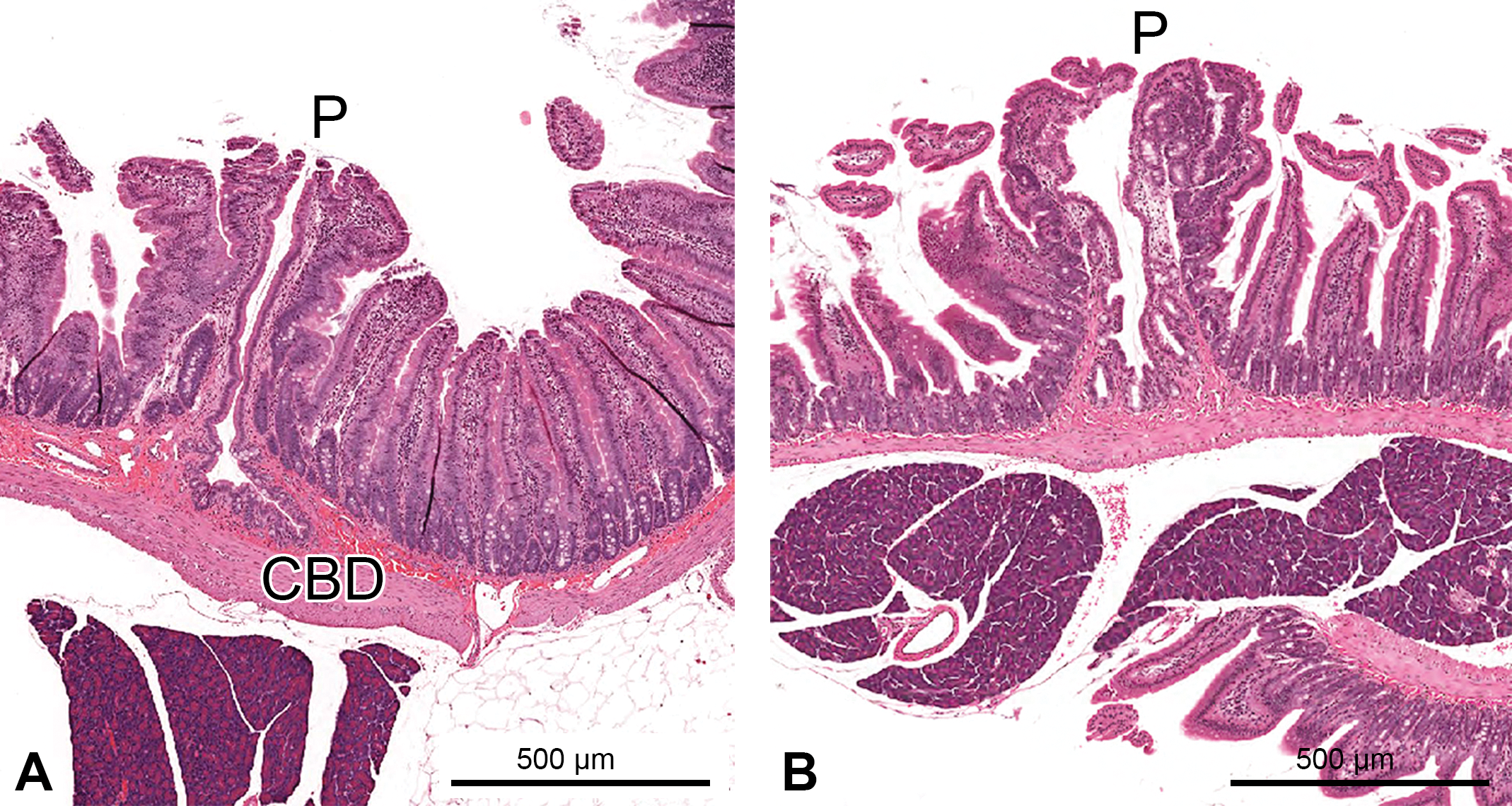
In rats, several pancreatic ducts empty into the extrahepatic common bile duct at various levels before the duct enters the duodenum at the major duodenal papilla (Farinon, Lampugnani, and Berri 1975); the minor duodenal papilla is absent in rats (Figures 3B and 8A and B). Thus, the distal segment of the extrahepatic common bile duct contains a mixture of bile and pancreatic secretions. For this reason, the term biliopancreatic duct has sometimes been used to refer to the common bile duct (Kara 2005). To maintain clarity when describing changes in pathology reports and to avoid potential confusion with effects on the pancreas due to the use of such a term, especially for readers who may not be aware of subtle interspecies anatomic differences, it is prudent to adhere to more standard nomenclature; that is, common bile duct, specifying proximal and distal segments if need be. The major papilla in the rat is usually located approximately 3 cm (range 1–4 cm) distal to the pylorus.
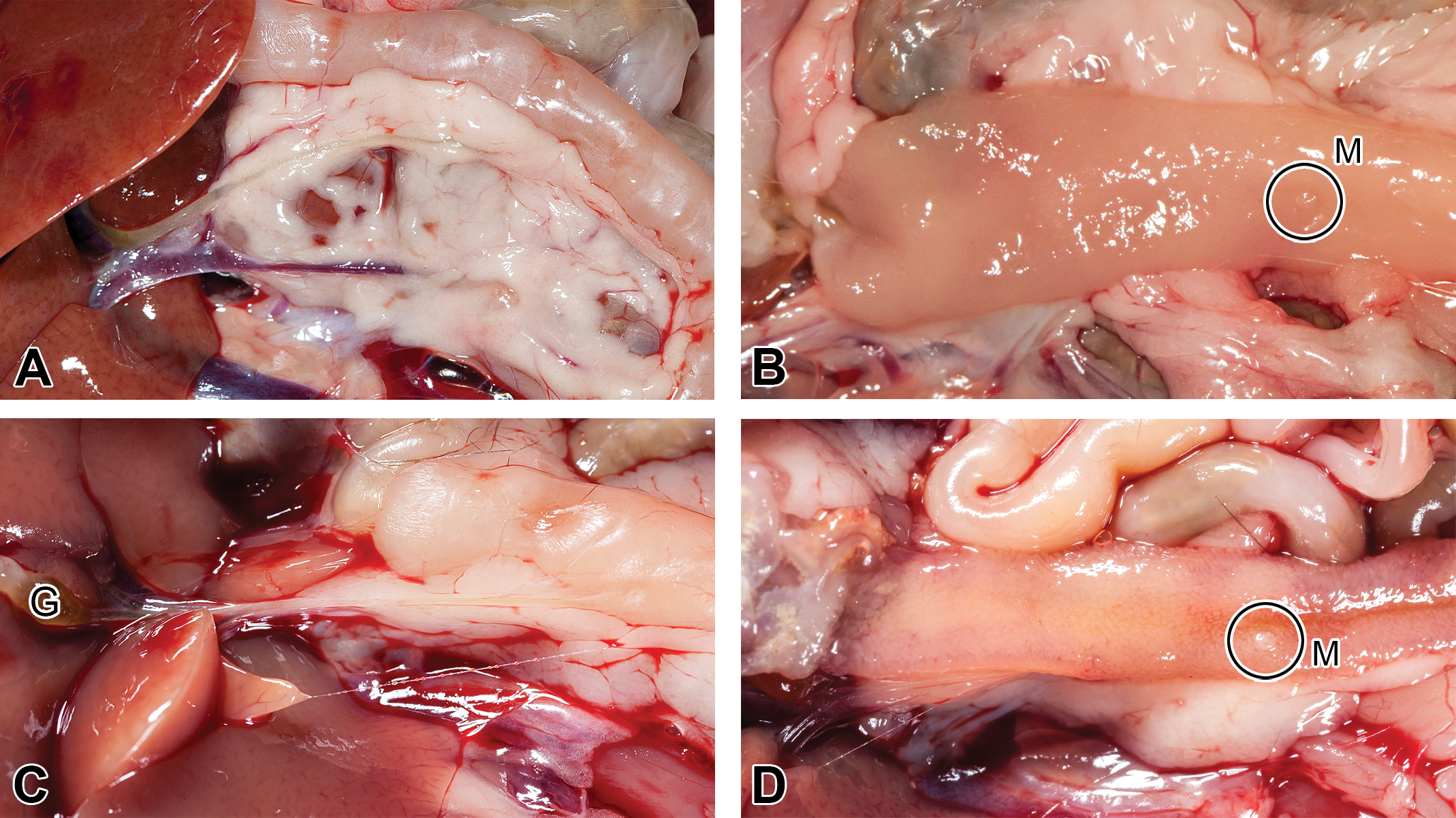
Extrahepatic bile ducts and duodenal papillae in rodents. (A and B) Rat; (C and D) mouse. M = major duodenal papilla; G = gall bladder.
In mice, the major pancreatic duct empties into the common bile duct before it enters the duodenum (Tsuji et al. 2006; Treuting, Valasek, and Dintzis 2011) at the major duodenal papilla, which is approximately 1 cm (range 0.5–1.5 cm) distal to the pylorus (Figures 3C and 8C and D). Thus, as in the case of rats, the common bile duct entering the duodenum and emptying into the major duodenal papilla contains a mixture of bile and pancreatic secretions. In addition, multiple smaller accessory pancreatic ducts that release their contents directly into the duodenum, at one or more minor duodenal papilla(e), are common in mice (Figure 3C). Hence, the anatomy is different in the 2 rodent species.
Identification of Duodenal Papillae at Necropsy
The extraparenchymal BPDs enter the duodenum at the mesenteric border. Accordingly, papillae are located on the mucosal aspect of the intestinal wall adjacent to the mesentery (Figures 5 and 8). To identify and preserve the papillae, the intestine must be opened by cutting at the antimesenteric side of the intestinal wall. Papillae are usually visualized as raised areas of mucosa projecting into the duodenal lumen. Distance from the pylorus serves as a good guide when looking for the major papilla. In rats and mice, the identification of the major papilla at necropsy can be aided by the use of a magnifying lens or dissecting microscope. After the major papilla has been identified, the minor papilla can be identified in macaques and dogs using the anatomic presentations described previously. Macroscopic identification of minor papillae may be futile in mice considering their small size and variability in location. As described previously, the minor duodenal papilla is absent in rats.
Duodenal papillae are best identified macroscopically in fresh tissue at necropsy. Postfixation and identification can be difficult in rodents due to contraction and folding of tissue during fixation. Additionally, the small size can make it quite challenging in mice. Fixation effects may be minimized by stapling the duodenal segment to a fine cardboard, with the serosal surface of the duodenum facing the cardboard. Papillae may also be marked with a tissue dye (see Figure 2C) that does not impact histologic processing or evaluation and to assist identification in fixed tissue and proper trimming.
In species that have a gall bladder, while the liver, pancreas, extraparenchymal BPDs, and the duodenum are in situ, gently compressing the gall bladder to force some bile out through the major papilla into the duodenal lumen helps identify the major papilla once the duodenal lumen has been opened and ingesta overlying the mucosa cleared. This can be challenging when the size of gall bladder is small (as in mice) or when the bladder has been emptied after feed intake.
Histologic Processing and Species Variations
There are several ways to process various segments of the extraparenchymal BPDs and the duodenal papillae to provide specimens for histologic, ultrastructural, or genomic evaluation. We outline a simplified version to capture the histologic features. For histologic preparation, after gross evaluation, formalin-fixed tissue specimens were routinely processed and paraffin embedded for preparation of H&E-stained sections.
Hepatic/Cystic/Common Bile Ducts
Segments may be processed separately (Figure 4). Transverse and longitudinal sections of the bile duct may be obtained from several locations: adjacent to the duodenum, adjacent to the liver, and from the middle of the duct. Sections from several locations may enhance the evaluation, help assess the progression of an inflammatory or degenerative process (ascending or descending), and understanding of the mechanism of a toxic event.
Pancreatic Ducts
These are often difficult to separate from the pancreatic parenchyma and hence are most efficiently processed with sections of pancreas (Figure 4) sampled from several locations for a more thorough evaluation.
Duodenal Papillae
These are processed as longitudinal sections along with adjacent duodenum and pancreas, with cut section perpendicular to the long axis of the duodenum (Figures 4, 6, 7, and 9). Bile staining of the papillary/ductal lumen with tissue dye can serve as a useful guide when trimming sections.
Major duodenal papilla in rodents (H&E stain). (A) Rat; (B) mouse. CBD = common bile duct; P = papilla.
The small size of the major duodenal papilla can make histologic processing quite challenging in smaller species, especially mice. In rats and mice, if the major papilla is grossly unidentifiable, one can increase the probability of obtaining it in histologic sections by processing several sections of the duodenum in the area where the papilla is expected to be present (duodenal segment between 2.5 and 3.5 cm distal to the pylorus in rats and between 0.5 and 1.5 cm distal to the pylorus in mice).
Of the 4 animal species discussed in this article, macaques are the only one in which one may expect to see Brunner’s glands in sections of the duodenal papillae (Figures 6, 7, and 9). In primates, Brunner’s glands typically extend past the minor duodenal papilla (Grossman 1958). In dogs, Brunner’s glands are usually not seen in sections of the duodenal papillae, since these glands are restricted to the portion of the duodenum proximal to the major duodenal papilla (Grossman 1958). In rats, Brunner’s glands are typically restricted to the first 1.5 to 2 cm of duodenum distal to the pylorus (Anand and Han 1975). In mice, Brunner’s glands are typically restricted to a 1-cm length of the pyloroduodenal junctional region (Obuoforibo and Martin 1977).
Discussion
Toxicologic effects of xenobiotics on BPDs can vary depending on the location of the ducts (intraparenchymal vs. extraparenchymal). Evaluation of changes in extraparenchymal BPDs can improve the characterization of the toxicity of certain xenobiotics. These tissues are not part of the normal tissue list for toxicology studies and must be added proactively to the prosection protocol if they are to be collected appropriately.
The tissue processing guidelines for safety assessment studies health authorities such as the United States Food and Drug Administration, the European Medicines Agency, and the Japanese Pharmaceutical and Medical Device Agency do not list the extraparenchymal BPDs; however, these guidelines clearly state that tissues known to be targets of a test article or those of its class should be evaluated in addition to the core list of tissues. The same was also described in a recent position article (Bregman et al. 2003). This approach is pragmatic. Histologic processing of extraparenchymal BPDs or the duodenal papillae is not required for all studies and should only be undertaken on an as needed basis. Subsequently, we list some recommendations for histopathologic evaluation of extraparenchymal BPDs, which may be adapted to suit the particular needs of a study or program: Evaluation of extraparenchymal biliary/pancreatic ducts and duodenal papilla(e) may be indicated when: Available literature suggests a potential for toxicity in these tissues based on the knowledge of the drug target, the compound, and/or its metabolism. Evaluations to validate such hypotheses are best undertaken in early studies where animal numbers are limited and logistics are more manageable. Biomarker profiles complement such hypotheses. For example, increased serum bilirubin concentrations and the activity of alkaline phosphatase and gamma-glutamyl transferase may indicate bile duct injury. Intrahepatic or intrapancreatic ducts are histologically identified to be targets of toxicity. Note: Lack of changes in the gall bladder may not necessarily indicate absence of changes in the distal biliary tree. Gross changes are observed in the extraparenchymal BPDs at necropsy, and these are determined to be treatment related. In areas where the common bile duct is surrounded by pancreas (adjacent to the duodenum), the surrounding pancreatic tissue must be included in sections of the bile duct to ascertain inflammatory spread and potential for pancreatic lesions. When processing the major duodenal papilla (along with portions of the ampulla of Vater in macaques) or the minor duodenal papilla, the adjacent duodenum (with Brunner’s glands in macaques) as well as the surrounding pancreas should be included in the histologic section from the duodenum (see Figures 4, 6, 7, and 9).
In macaques, the longitudinal section of the major papilla should include the underlying ampulla. Normally, the mucosal folds of the duodenal papillae and the sphincter of Oddi help avoid reflux of duodenal contents into the papillary lumen (Allescher 1989). However, this can be altered in a pathologic state. Due to its characteristic anatomic location, the ampulla may be exposed to a mix of bile and pancreatic secretions as well as duodenal contents, thereby increasing the chances of unique pathologic changes, which may help to explain why the ampullary region has a high incidence of neoplastic transformation and malignancy in humans (Lillemoe 2003). Hence, it is important to characterize treatment-related pathologic changes in this region. Considering similarities between macaques and humans (dogs lack an ampullary region in the major duodenal papilla), any data accrued from such an evaluation in macaques would be important for translation to human clinical trials.
In live animals, certain biomarkers, for example, increased serum bilirubin concentrations and the activity of alkaline phosphatase and gamma-glutamyl transferase may indicate bile duct injury. These biomarkers are generally not specific for intraparenchymal or extraparenchymal ducts, and increases are not consistently observed when toxicity was either limited to, or predominantly affected, the extraparenchymal bile ducts. Further, evaluation of biomarkers may not specify location of injury along the BPDs, may not quantify the magnitude of the morphologic change, and may not indicate whether and to what extent morphologic changes in BPDs impact adjacent tissues. For all these reasons, evaluation of changes in BPDs as well as the duodenal papillae may be important in live treated animals. As an alternative to biopsies, noninvasive imaging techniques are often utilized in human diagnostics. In addition, procedures like retrograde endoscopy are now well established for evaluating and treating disease conditions in the major duodenal papilla or the BPDs (Costamagna et al. 2008). Imaging applications may be useful to provide a translatable method to monitor toxicity in nonclinical species when extraparenchymal BPDs or duodenal papillae are affected (Nyland, Hager, and Herring 1989; Schuhmann-Giampieri et al. 1992; Hirohashi et al. 1998; Seki et al. 2000; Figueiredo et al. 2010].
In summary, awareness of anatomic differences across various species is important to characterize toxicologic pathology findings and their underlying mechanisms and also for translation to humans. We believe that the information provided in this article could be useful to the scientific community involved in nonclinical safety assessment, may enhance the quality of such evaluations, and help provide information translatable to clinical settings, thereby improving the quality of patient care.
Footnotes
Acknowledgments
We would like to thank the necropsy and histology teams at Novartis Institutes for BioMedical Research (East Hanover, NJ) and Covance Laboratories (Madison, WI) for their help and support in the gross/histologic assessment of tissues. We would also like to thank pathologists and toxicologists at Novartis for aiding some of the early work; Drs. Gary Boorman, Denzil Frost, and Pritam Sahota for their suggestions to enhance the quality of this manuscript; and Mr. Kenard Lee for his assistance with the figures.
Author Contribution
Authors contributed to conception or design (K.V., S.N., R.E., B.K., S.L., B.C., G.H., E.M., and R.J.); data acquisition, analysis, or interpretation (K.V., S.N., R.E., B.K., S.L., B.C., G.H., E.M., and R.J.); drafting the manuscript (K.V.); and critically revising the manuscript (K.V., S.N., R.E., B.K., S.L., B.C., G.H., E.M., and R.J.). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
