Abstract
Brain sections from control cynomolgus monkeys (Macaca fascicularis) used in toxicology studies were evaluated retrospectively in order to better understand spontaneous background changes in this species. Hematoxylin and eosin–stained slides from 76 animals (38 males and 38 females) of 9 studies were examined. Eleven animals (9 males and 2 females) were each observed to have 1 to 3 findings within the brain sections examined, for a total of 19 findings. No findings were noted in the spinal cord. The most common finding was focal to multifocal perivascular infiltration of mononuclear cells, affecting the parenchyma, the meninges, or the choroid plexus. Additionally, focal gliosis was observed in 6 animals and a single focus of hemosiderin deposition (coincident with focal gliosis and mononuclear cell infiltrate) was noted in 1 animal. Most of the glial foci were composed of cells consistent with microglial cells, with or without admixed lymphocytes. All findings were of slight or minimal severity, lacked an apparent cause, and were considered incidental and of negligible biologic significance. An awareness of the spontaneous incidence of these background findings may facilitate the discernment of toxicologically relevant effects when these findings are observed.
The cynomolgus monkey is a non-rodent species frequently used in toxicity studies for safety assessment of pharmaceuticals, particularly biologic entities, for which there may be no relevant alternative species. Interpretation of histopathology from studies with nonhuman primates can be confounded by considerable variability among animals (in terms of age, weight, sexual maturity, and geographic source) as well as by the group size limitations due to animal conservation and expense. Historical control data are valuable for establishing the incidences of spontaneous background findings among animals within a given testing facility; however, variations among pathologists in nomenclature and recording thresholds for individual findings can hinder the validity and utility of these databases, particularly for subtle changes and/or uncommon findings.
Retrospective analyses of spontaneous findings in cynomolgus monkeys have been reported (Chamanza et al. 2010; Shimoi et al. 1998) providing valuable information to toxicologic pathologists. In this retrospective assessment of brain sections from control cynomolgus monkeys, an individual veterinary pathologist (experienced in neuropathology) reviewed sections from 9 studies (comprising 76 animals) conducted within 1 facility, ensuring uniform assessment of subtle or infrequent changes.
Materials and Methods
Nine toxicologic studies (identified as A through I, Table 1), conducted at Charles River Laboratories, Reno, Nevada, which used purpose-bred and experimentally naive cynomolgus monkeys of Chinese origin, were identified for retrospective histopathologic evaluation of brain from control animals. The 76 control monkeys, comprising 38 females and 38 males, ages 28.6 to 99.7 months (mean 45.5 months) at study end, received oral vehicle (one study) or parenteral vehicle formulations of variable composition with volumes ranging from 2.0 to 10.0 ml/kg administered by intravenous bolus or infusion injection and/or subcutaneous injection, weekly or biweekly for the duration of the given study’s dosing phase (ranging from 14 days to 9 months). Animals were assigned identifying sequential numbers (males odd and females even) for the purpose of this review. For each animal, glass slides of formalin-fixed, paraffin-embedded, hematoxylin and eosin (H&E)-stained sections of brain were examined. All slides of brain (335 total; ranging from 2 to 15 slides per animal) were examined by one of the authors (M.B.) and overall incidences of specific observations were determined. Sections of spinal cord were also examined from each animal. The emphasis for review was cellular infiltrates and glial cell accumulations in the central nervous system tissue, but all microscopic changes interpreted to represent antemortem morphologic differences from normal (i.e., not artifactual changes associated with tissue collection, fixation, and/or processing) were recorded.
Summary of Animals Reviewed
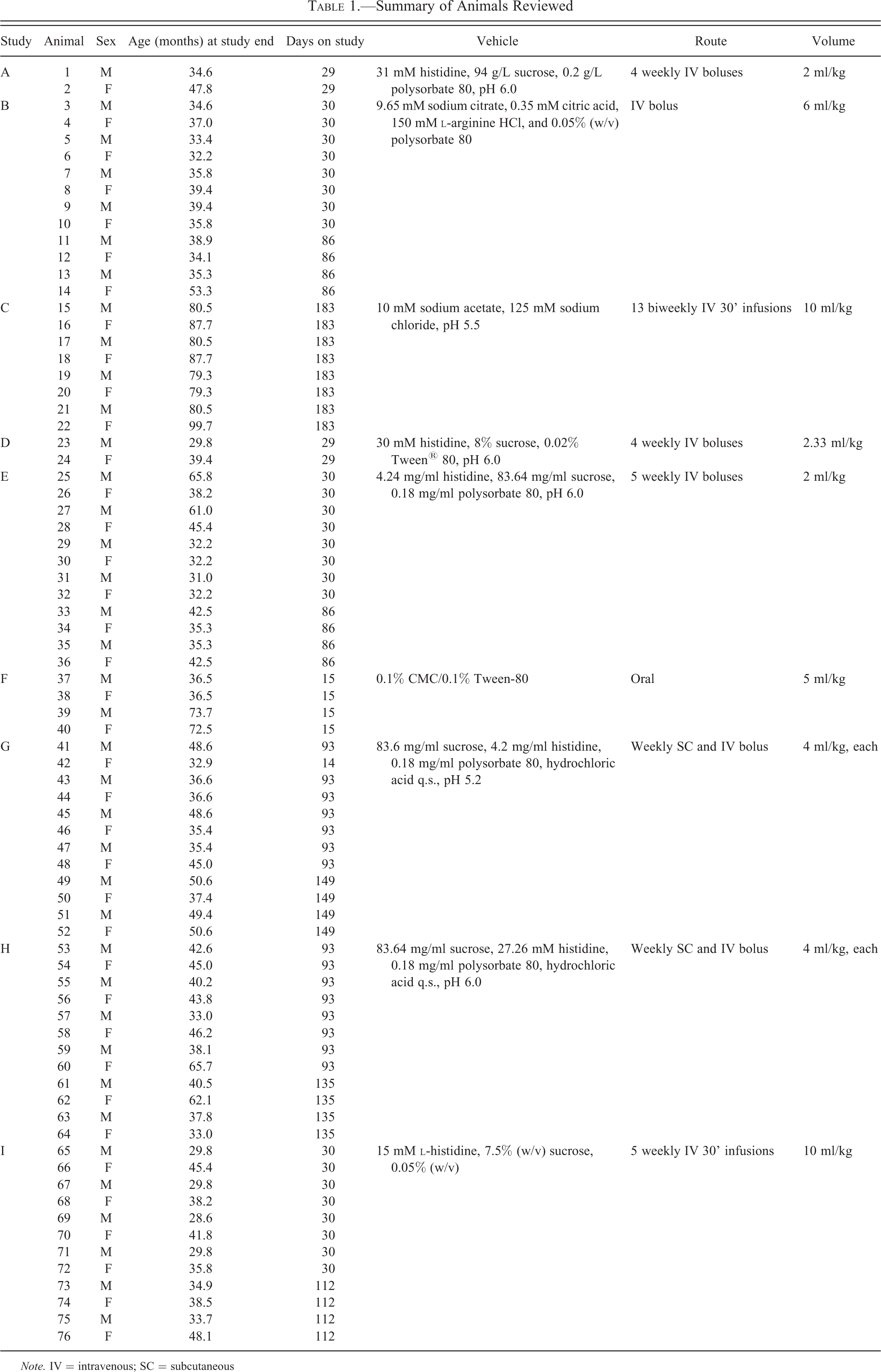
Note. IV = intravenous; SC = subcutaneous
Results
Of the 76 animals evaluated, 11 (9 males and 2 females) had one or more slight to minimal focal changes involving the brain parenchyma, meninges, or choroid plexus (Table 2). No histopathologic findings were noted in spinal cord. The 11 animals having microscopic brain changes (representing 14.5% of the animals reviewed) had been maintained on the corresponding studies for 29 days to 6 months (mean 82 days).
Summary of Findings
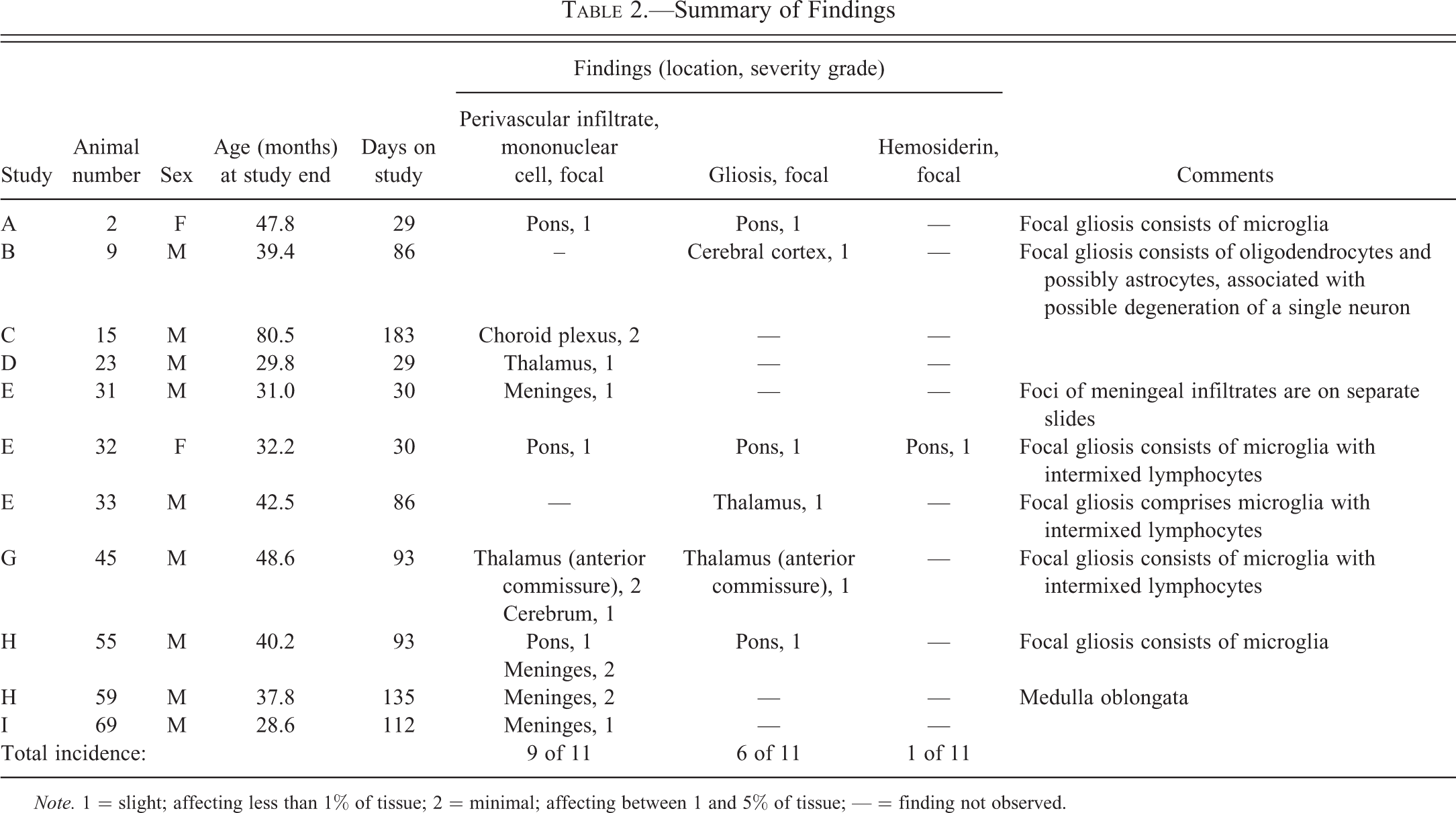
Note. 1 = slight; affecting less than 1% of tissue; 2 = minimal; affecting between 1 and 5% of tissue; — = finding not observed.
Perivascular mononuclear cell infiltrates (Figures 1 through 3) were the most commonly observed change within the monkeys examined, affecting 10 animals (13% incidence). Six foci of parenchymal perivascular mononuclear cell infiltrate (Figure 1) were noted among 5 animals (2 females and 3 males; 6.6% incidence). Three foci were noted within the pons, 2 within the thalamus, and 1 within the cerebrum. Five additional foci of mononuclear infiltrates were noted within the meninges (Figure 2) of 4 animals (all males) and 1 focus was present within choroid plexus of 1 male (Figure 3).
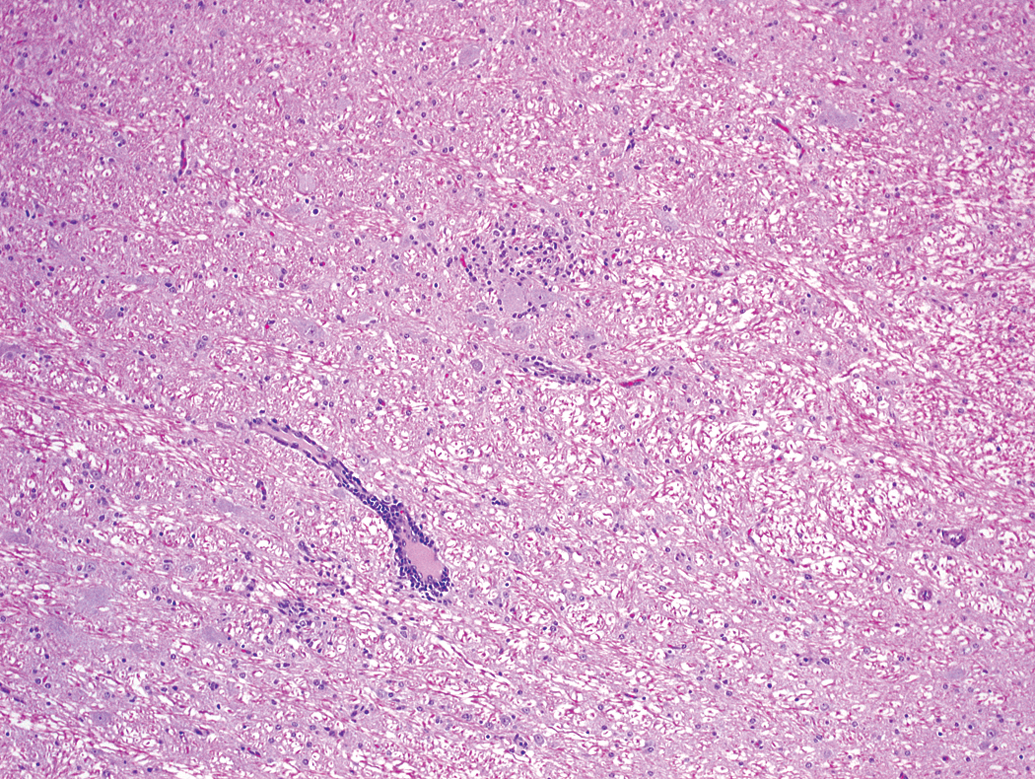
Animal A2. A perivascular mononuclear cell infiltrate (lower left) is adjacent to focal gliosis (center).
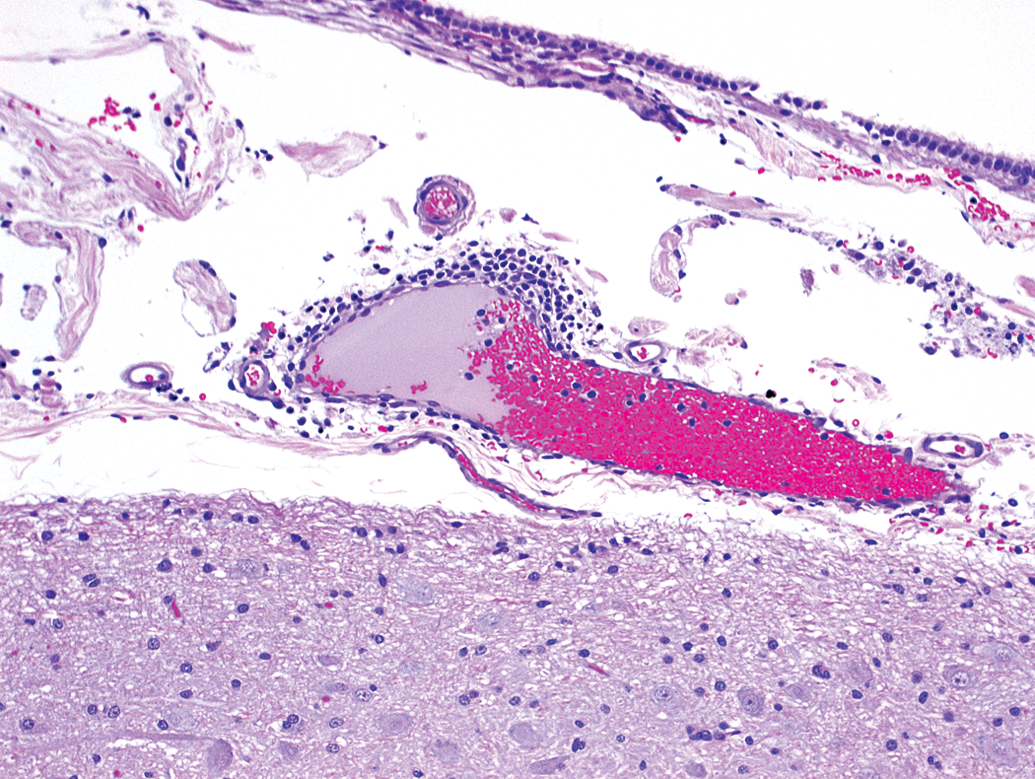
Animal E31. Meningeal perivascular infiltrates of mononuclear cells.
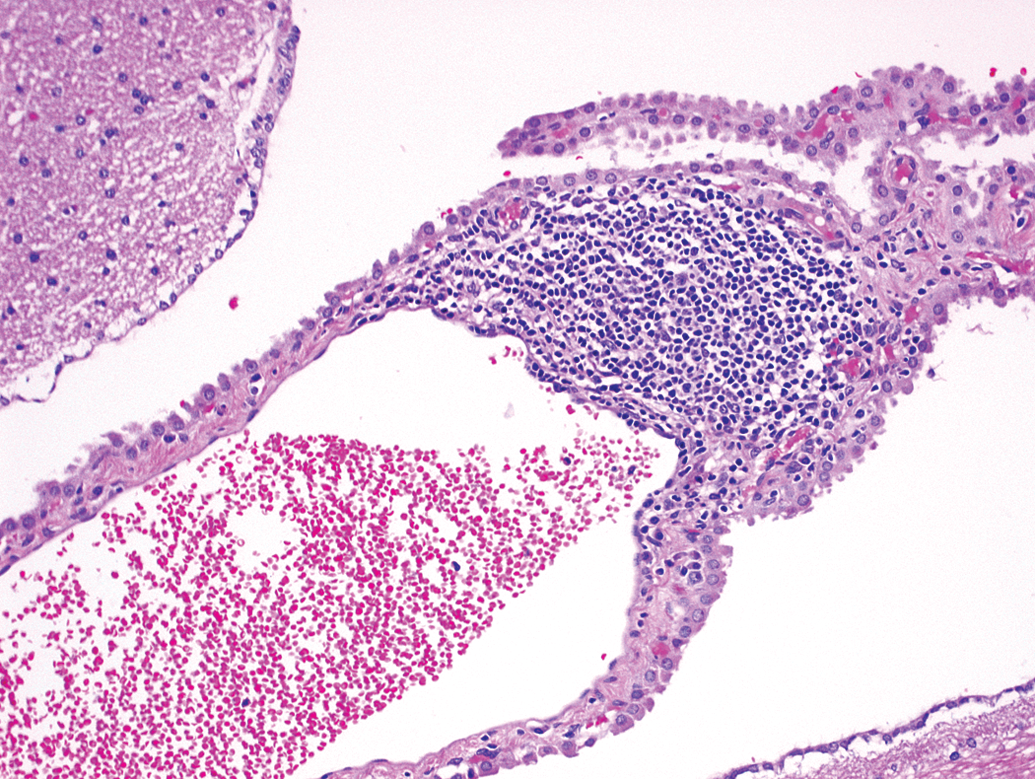
Animal C15. Mononuclear cell infiltrate is associated with a vessel within the choroid plexus.
Focal gliosis (prominence of glial cells due to hypertrophy and/or locally increased numbers) was observed in each of the 6 animals (4 males and 2 females; 8% incidence), occurring within the pons (3 foci), cerebral cortex (1 focus), or thalamus (2 foci). Most foci of gliosis (5 of 6; 6.6% overall incidence) were characterized by the presence of cells morphologically consistent with microglia, with or without admixed lymphocytes (Figures 4 and 5). Microglial cells were identified as having fusiform to bean-shaped, dense (dark blue) nuclei without discernible nucleoli. One focus (within the cerebral cortex) was composed of oligodendrocytes (with few astrocytes) loosely clustered around a possibly degenerate neuron (Figure 6). Oligodendrocytes were distinguished as ovoid cells, typically smaller than astrocytes, having clear cytoplasm and central round to ovoid dark nuclei, with a rim of marginated chromatin and one or more nucleoli. All instances of focal gliosis were graded slight (lowest severity grade used by the reviewing pathologist).
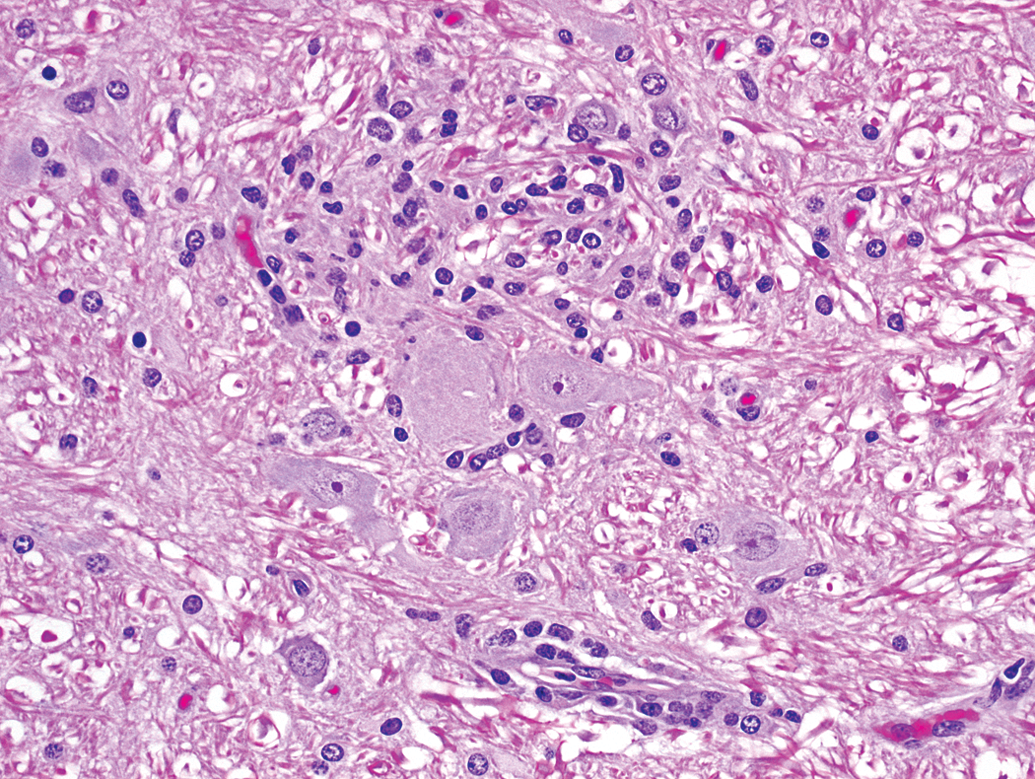
Animal A2. Higher magnification of the focal gliosis in Figure 1 shows a cluster of cells consistent with microglia adjacent to neurons.
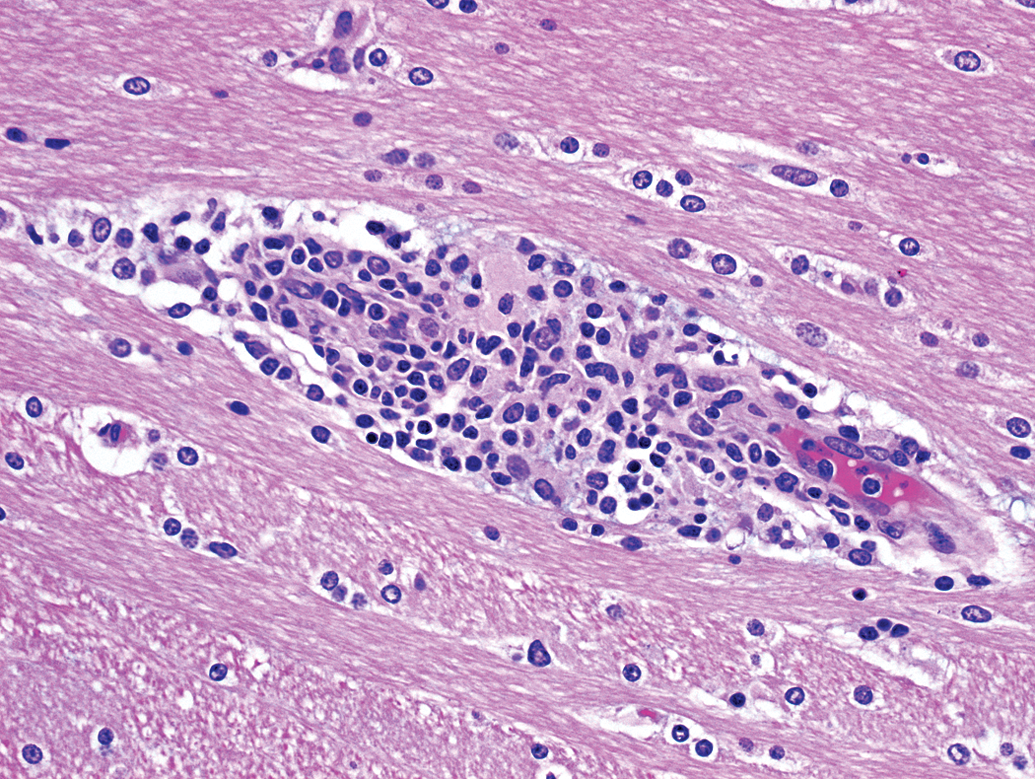
Animal G45. Focal gliosis composed of microglia intermixed with lymphocytes.
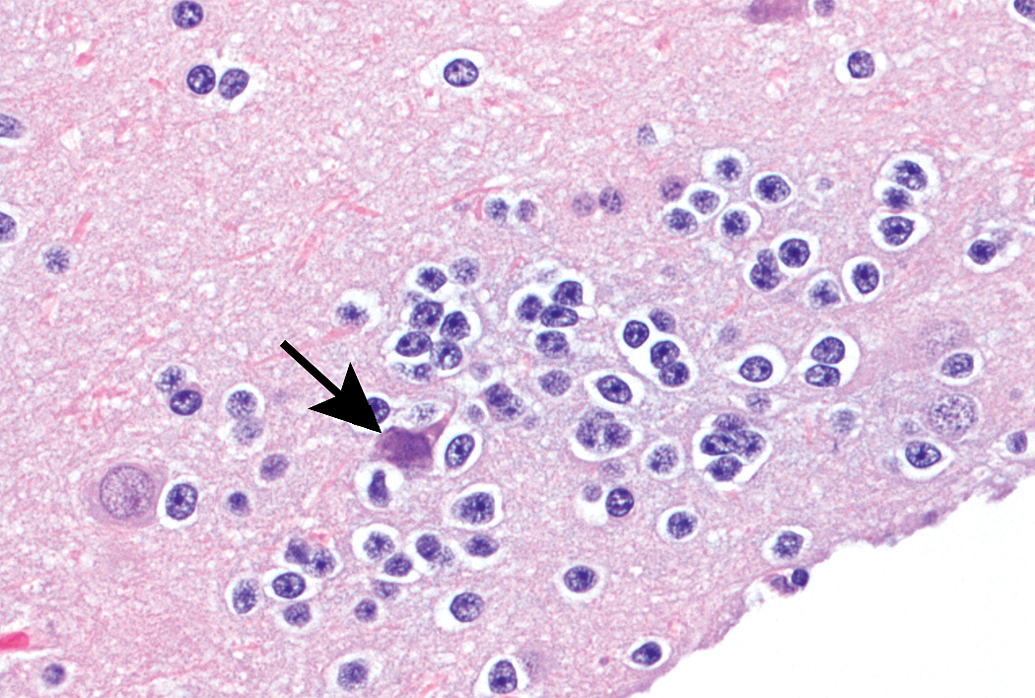
Animal B9. Focal gliosis consists of oligodendrocytes associated with a possibly degenerate neuron.
One animal (female E32) had a single focus of pigment consistent with hemosiderin (a product of hemoglobin degradation). The pigment was associated with focal gliosis and perivascular mononuclear cell infiltrate in the pons.
Discussion
Background infiltrative findings within the brains of control cynomolgus monkeys are not common, as noted in a previous review (Chamanza et al. 2010) and further substantiated in this report. Minor microscopic findings having no functional in-life correlates occur with low incidence among control animals and must be distinguished from a toxicologic effect when present among animals administered a test compound. The spontaneous background infiltrative changes observed in this review consisted of minimal focal cellular responses to apparently incidental changes within central nervous tissues. In all cases within this review, a single anatomic site in each animal was affected by slight (grade 1) to minimal (grade 2) change, sometimes characterized by 2 or 3 concurrent findings. Identification of the components of cellular foci and infiltrates in the animals reviewed was limited to morphologic characteristics discernible on H&E-stained sections. Definitive designation of cell types would require further characterization using immunohistochemical techniques or other staining procedures, efforts that were not feasible with these study specimens.
Changes were noted with slightly greater frequency among males versus females; however, the small numbers of affected animals in this review preclude definitive conclusions regarding the gender predilections of these observations. The mean, distribution, and range of ages among animals having microscopic change were similar to those of the entire test group. Incidences among individual studies had no apparent relation to vehicle administration. The probability of identifying microscopic change was unavoidably increased among animals having more slides examined; however, 1 affected animal had representation of only 2 brain slides.
Mononuclear cell infiltrates are commonly observed within various tissues (most notably liver, kidneys, and heart) of control cynomolgus monkeys used in toxicity studies (Chamanza et al. 2010; Shimoi et al. 1998). Similar mononuclear cell infiltrates were noted, although less frequently, within brain sections from control animals of these reported studies, as were observed among animals in the current review. The perivascular accumulations of lymphocytes and macrophages may represent immune responses originating within the Virchow–Robin space, continuous with the subarachnoid space and outside of the blood-brain barrier (Summers, Cummings, and de Lahunta 1995). When there is a breach of the blood-brain barrier (normally maintained by specialized endothelial cells interacting with other local cell types including astrocytes) infiltrates may extend into the neural parenchyma and a response from glial cells, most notably microglia, may occur. Such a process may explain the appearances of focal gliosis in 3 of the animals described herein, in which foci consisted of a mixture of lymphocytes and microglial cells. Four of the 5 animals (A2, E32, G45, and H55) having focal gliosis with microglial cells also had perivascular mononuclear cell infiltrates within the same section. A focus of pigment consistent with hemosiderin was observed in 1 animal (E32). This change was coincident with focal gliosis as well as perivascular mononuclear cell infiltrate, suggesting a prior discontinuity in the blood-brain barrier with extravasation of erythrocytes and subsequent degradation of hemoglobin.
The possibility of residual inflammation from prior microbial or viral infection cannot be excluded as the cause of the lymphocytic accumulations detected among the animals examined; however, the focal and limited distribution of the infiltrates as well as the history of close monitoring and careful husbandry of these purpose-bred animals makes that circumstance less probable.
Unlike astrocytes and oligodendrocytes (so-called macroglia), which are of neuroectodermal origin, microglia are of mesodermal origin, arising from progenitor cells that migrate prenatally to the central nervous system (Chan, Kohsaka, and Rezaie 2007). Although microglia are not distinguishable from peripheral macrophages by surface markers (Graeber and Streit 2010; Imai et al. 1996), they are functionally different from perivascular macrophages within the central nervous system. In addition to their more familiar phagocytic role, microglia fulfill the functions of homeostatic surveillance and maintenance. So-called resting microglia are dispersed within the parenchyma, having numerous motile, radiating and branching, attenuated processes (generally not evident in H&E preparations) monitoring changes in the local environment (Nimmerjahn, Kirchhoff, and Helmchen 2005). In response to a variety of signals (including ischemia, cytokine release, loss of blood-brain barrier integrity, change in synaptic functional status, and change in ion homeostasis), microglia rapidly become activated, changing gene expression profiles and morphology (reflected as hypertrophy and decreased size and complexity of processes leading to an amoeboid appearance), enabling a variety of functions including proliferation, neuronal support, antigen presentation, chemotaxis, cytotoxic capability, and growth factor release (Kettenman et al. 2011; Streit 2000). The rat facial nerve axotomy model (Graeber et al. 1988) has demonstrated the immediate response of microglia in support of source-affected motor neuronal cell bodies within the brainstem, with microglial involvement preceding any astrocytic role by days. This more rapid response (as compared to astrocytes) by microglial cells is also evident in primary toxic neuropathies, including the neurotoxicant 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP; Breckenridge et al. 2013). In some (but not all) cases, activated microglia develop phagocytic capabilities. Phagocytic activity by microglial cells, however, was not evident in the foci observed in this review. Numbers of microglial cells have been shown to increase with age in cynomolgus monkey brains (Kodama et al. 2010) and can persist following traumatic injury (Nagamoto-Combs et al. 2007). Increased numbers of activated microglial cells have been associated with extracranial conditions including hypercholesterolemia in rabbits and heart disease in people (Streit and Sparks 1997).
Local proliferation of oligodendrocytes, as observed focally in animal B9, may be seen in association with inflammatory demyelination (Summers, Cummings, and de Lahunta 1995) and can be a component of satellitosis. The isolated focus of loosely aggregated, unremarkable oligodendrocytes observed in this review was not consistent with an extensive demyelinating process or neoplastic change, but likely represents a minor, focal perturbation or a normal glial rest.
Footnotes
Author Contribution
The studies referenced in this article were sponsored by AbbVie Inc. or Biogen Idec. AbbVie Inc. and Biogen Idec contributed to the design, analysis, and interpretation of data, writing, reviewing, and approving the publication. Charles River Laboratories, Nevada, was contracted by AbbVie and Biogen Idec to conduct the nonclinical studies that generated all of the histology slides subsequently examined by Mark Butt of Tox Path Specialists, LLC, to provide the data in this publication. All coauthors were involved in the discussion, reviewing, and approving of the publication.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Surekha Akella, George Foley, and Katharine Whitney are employees of AbbVie. Suezanne Parker is a current employee and Wendell Davis is a former employee of Biogen Idec.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
