Abstract
Circulating microRNAs (miRNAs) show promise as biomarkers due to their tissue-specific expression and high stability. This study was conducted to investigate whether nervous system–enriched miR-9* and hippocampus-enriched miR-384-5p could be indicators of neurotoxicity in serum. Rats were given a single administration of trimethyltin (TMT) chloride at 6, 9, or 12 mg/kg by gavage, and brain and serum were collected 1, 4, and 7 days after administration. MiR-9* and miR-384-5p levels in serum and hippocampus were analyzed by reverse transcriptase polymerase chain reaction (RT-PCR), and their neurotoxicity detection sensitivities were compared with nervous symptoms, auditory response, and histopathology. TMT caused tremor, hypersensitivity, and decreased auditory response at 12 mg/kg on day 1 and at 9 mg/kg on day 4. Histopathologically, neural cell death and glial reaction were observed in brain (mainly hippocampus) at 12 mg/kg on day 1, 4, and 7 and at 6 and 9 mg/kg on day 4 and 7. MiR-9* and miR-384-5p levels were elevated in serum at 9 and 12 mg/kg on days 4 and 7 (at 9 mg/kg on day 7, miR-9* only) but were not changed in hippocampus. These miRNAs were considered to be elevated with the evolution of neural cell death and were thus considered possible novel indicators of neurotoxicity.
Introduction
MicroRNAs (miRNAs) are a class of small noncoding RNAs (∼25 nucleotides), which play significant roles in regulating a wide variety of cellular and biological processes, including growth, development, differentiation, proliferation, cell cycle control, and cell death (Ambros 2004; Kloosterman and Plasterk 2006; Miska 2005).
Recently, the use of miRNAs in the biofluid as biomarkers has been attracting attention, as they have high stability in blood and reflect various physiological changes and diseases. Some circulating miRNAs have been successfully revealed as biomarkers for several cancers, cardiovascular disease, and liver injury (Cuk et al. 2013; Hannafon and Ding 2013; Mitchell et al. 2008; Sukata et al. 2011; Tokarz and Blasiak 2012; Wang et al. 2013; Wang et al. 2009). Thus, miRNAs are considered promising as biomarkers.
In the nervous system, miRNAs also play important roles in growth, development, differentiation, function, and pathogenesis of neurodegenerative diseases (Davis et al. 2008; Dogini et al. 2008; Edbauer et al. 2010; Haramati et al. 2010; Junn and Mouradian 2012; Wanet et al. 2012; Zhang et al. 2010). Several nervous system–enriched or nervous system-specific miRNAs have been reported (Geloso, Corvino, and Michetti 2011; Schratt et al. 2006; Sempere et al. 2004; Yokota 2009; Zhao et al. 2006). In the past several years, researchers have reported that some miRNAs in blood are possible biomarkers in humans or animals having brain tumors (Skog et al. 2008), neurodegenerative disease (Margis and Rieder 2011), or brain injury. As brain injury markers, miR-16, miR-92a, and miR-765 in plasma from traumatic brain injury patients within the first 24-hr post-injury were suggested (Redell et al. 2010), and let-7i in both serum and cerebrospinal fluid in traumatic brain injury rats at 3-hr and 24-hr post-blast wave exposure was suggested (Balakathiresan et al. 2012). In addition, the brain-specific miR-124 increased in plasma from rats 8 hr after brain injury produced by occlusion of middle cerebral artery and peaked at 24 hr (Omar et al. 2009).
Recently, it has been reported that miRNA expressions in the nervous tissues could be altered by exogenous chemicals in association with neurotoxicity (Kaur, Armugam, and Jeyaseelan 2012; Tal and Tanguay 2012; Wang et al. 2010). Neurotoxicity is a critical problem in the development and supply of chemicals and drugs, as the nervous system plays an extremely important function for life but generally has an extremely low potential for regeneration. Moreover, no useful biomarkers have been established from biofluid that reflect neurotoxicant-induced abnormalities in the nervous system. Currently, establishment of noninvasive biomarkers in the nervous system that reflect neurotoxicity is desirable. We therefore hypothesized that blood miRNAs could be novel neurotoxicity indicators.
We focused on miR-9* and miR-384-5p as potential indicators of neurotoxicity, because these miRNAs were reported as brain-enriched miRNAs. MiR-9* is expressed especially in the developing nervous system, regulating neuronal development (Krichevsky et al. 2006; Yoo et al. 2009; Zhao et al. 2006), and its anomalous expression is associated with abnormal developments of nervous system such as spina bifida aperta and anencephaly (Wei et al. 2013; Zhang et al. 2010), neurodegenerative disease (Packer et al. 2008), and brain tumors (Nass et al. 2009; Schraivogel et al. 2011). On the other hand, miR-384-5p was shown to be highly expressed in hippocampus in a profiling study of rat brain miRNAs (He et al. 2007). In addition, miR-9* and miR-384-5p levels in blood are extremely low under normal conditions, and therefore they are expected to be easily detectable as indicators if their blood levels can be elevated to reflect brain abnormality.
The present study was conducted to examine potentialities of miR-9* and miR-384-5p as blood indicators of neurotoxicity. To accomplish this, we used a trimethyltin (TMT)-induced rat neurotoxicity model. TMT is an organotin compound used in plastic stabilizers, chemostabilizers, and fungicides. It induces nervous symptoms (such as vocalization, seizures, hyperactivity, and tremor), learning and memory deficits, and hearing loss in humans and experimental animals (Brown et al. 1979; Bushnell and Evans 1985; Crofton et al. 1990; Dyer et al. 1982; Ishida et al. 1997; Perretta, Righi, and Gozzo 1993). In addition, TMT selectively damages neurons in the central nervous system, especially hippocampus and pyriform cortex, after a single exposure (Balaban, O'Callaghan, and Billingsley 1988; Bouldin et al. 1981; Geloso, Corvino, and Michetti 2011; Little et al. 2012). We investigated whether serum miR-9* and miR-384-5p levels could be altered by TMT treatment and compared their sensitivities of neurotoxicity detection with other examined neurotoxicity parameters such as nervous symptoms, auditory response, and histopathology.
Material and Methods
Reagents and Animals
TMT chloride was purchased from Kanto Chemical Co., Inc. (Tokyo, Japan). Synthetic miR-3 from Drosophila melanogaster (dme-miR-3) was obtained from Hokkaido System Science Co., Ltd. (Hokkaido, Japan). A total of 50 male, 6-week-old Crl:CD(SD) rats (SPF) were purchased from Charles River Laboratories Japan, Inc. (Shiga, Japan) and housed in suspended aluminum cages (1 rate per cage) in a room kept at 24 ± 2°C and 40 to 70% RH with a 12-hr light–dark cycle. Tap water via automatic stainless steel nozzles and CRF-1 pellet diet (Oriental Yeast Co., Ltd., Tokyo, Japan) were freely available throughout the study. The animals were observed daily and were used for the experiments after a 1-week acclimation period.
Animal Treatments
All experiments were performed in accordance with the Guide for Animal Care and Use of Sumitomo Chemical Co., Ltd. At the age of 7 weeks, 50 rats were divided into 4 groups (12 or 14 animals per group). Each group was given a single administration of TMT at 0, 6, 9, or 12 mg/kg via oral gavage (day 0). Observation periods were set for 1, 4, and 7 days after administration for each dose group (4 or 6 animals per each period). Daily observation of clinical symptoms was carried out throughout the study. Body weight was measured on days 1, 4, and 7. For examination of auditory response, animals’ responses to auditory stimuli (metal clicker) were scored on days 1, 4, and 7.
Sampling for Tissue and Blood
On days 1, 4, and 7, whole blood samples were drawn from the abdominal aorta of all animals under isoflurane anesthesia. Brains were immediately collected, weighed, and sagittally cut. The left half-brains were fixed in 4% paraformaldehyde phosphate buffer solution for histopathology. The right half-brains were regionally dissected in a prompt manner, and hippocampi were frozen in liquid nitrogen and preserved at −80°C in a freezer for miRNA assays. The whole blood samples were separated into serum and cellular fractions after leaving at room temperature for 30 min. The serum samples were preserved at −80°C in a freezer for miRNA assays.
Histopathology, Immunohistochemistry, and TUNEL Method
About 5-μm thick slices of the fixed half-brains were transversely cut at 4 levels (level 1: forebrain; level 2: center of cerebrum, including hippocampus; level 3: cerebellum and pons; level 4: medulla), embedded in paraffin, sectioned in 3-μm slices, stained with hematoxylin and eosin (HE), and examined under a light microscope. For immunohistochemistry and TUNEL method, 5-μm thick sections obtained from paraffin-embedded brains of animals sacrificed on day 4 and day 7 were used. After deparaffinization, immunohistochemistry using anti-glial fibrillary acidic protein (GFAP) antibody, anti-ionized calcium–binding adapter molecule 1 (Iba1) antibody, and anti-proliferating cell nuclear antigen (PCNA) antibody was performed on a Discovery XT automated staining instrument (Ventana Medical Systems, Inc.) in accordance with the manufacturer’s instructions. Heat-induced epitope retrieval in CC2 solution (Ventana Medical Systems, Inc.) was performed except for anti-GFAP. Inhibition of endogenous peroxidase using Inhibitor D (Ventana Medical Systems, Inc.) and blocking using Blocker (Ventana Medical Systems, Inc.) were also performed in accordance with standard procedures. The sections were incubated with anti-PCNA mouse monoclonal antibody (1:200 dilution, Dako Japan Ltd., Kyoto, Japan) for 32 min at 37°C, or anti-GFAP rabbit polyclonal antibody (1:200 dilution, Dako Japan Ltd., Kyoto, Japan), or anti-Iba1 rabbit polyclonal antibody (1:500 dilution, Wako, Osaka, Japan) for 1 hr at 37°C. Immunoreactivity was detected and visualized using Histofine Simple Stain Rat MAX-PO (MULTI; Nichirei Biosciences Inc., Tokyo, Japan) and DAB Map kit (Ventana Medical Systems, Inc.) before the sections were counterstained using hematoxylin.
Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) method was carried out using an ApopTag® Peroxidase in situ Apoptosis Detection Kit (Merck Millipore, MA) as recommended by the manufacturer. The slides were incubated with proteinase K (Dako Japan Ltd., Kyoto, Japan) for 15 min at room temperature. Visualization was performed using liquid stable 3,3-diaminobenzidine tetrahydrochloride (DAB; Nacalai Tesque, Kyoto, Japan) before the sections were counterstained using hematoxylin.
RNA Extraction
Prior to the extraction of total RNA from the serum, 0.2 fmol of synthetic miR-3 from D. melanogaster (dme-miR-3), which is represented on rat microarrays from Agilent Technologies (Santa Clara, CA,) as a negative control, was spiked in each sample as an internal standard (Sukata et al. 2011). Total RNA was isolated from serum in accordance with the protocol for PARISTM Kit (Ambion, Life Technologies Japan Ltd., Tokyo, Japan). The procedure for the extraction from the hippocampus was carried out in accordance with the protocol of mirVanaTM miRNA Isolation Kit (Ambion, Life Technologies Japan Ltd., Tokyo, Japan).
Real-time RT-PCR of Mature miRNAs
MiR-9* and miR-384-5p levels in serum in all dose groups on days 1, 4, and 7 and in hippocampus in all dose groups on day 7 were analyzed by real-time RT-PCR. TaqMan® MicroRNA Assays (Applied Biosystems, Life Technologies Japan Ltd., Tokyo, Japan), which include probes and primers, were used for all target miRNAs (miR-9* and miR-384-5p) and control miRNAs (miR-3 for serum and U6 for hippocampus). Real-time RT-PCR was performed in accordance with the protocol of TaqMan® MicroRNA Assays using 7500 Fast Real-Time PCR System (Applied Biosystems, Life Technologies Japan Ltd., Tokyo, Japan). All reactions were run in duplicate.
After completion of reaction, the threshold value was manually set above the baseline displayed on the amplification plot, and the mean cycle threshold (CT) was determined from the duplicate PCRs. Relative quantification of gene expression was evaluated by utilizing a comparative CT. The CT values for each mature miRNA reaction were subtracted from the respective CT value of the miR-3 control for serum or of U6 control for hippocampus, resulting in the ΔCT value. The control average ΔCT value was arbitrarily used as a constant that was subtracted from ΔCT values of all samples to determine ΔΔCT value. The power 2−ΔΔCT was then calculated, and fold changes compared to control group were generated.
Statistical Methods
For analysis of the data of body weight, brain weight, and miRNA expression, Bartlett’s test in StatLight (Yukms Co., Ltd., Tokyo, Japan) was applied to test the homogeneity of variance among the groups. When the groups were accepted to be homogeneous, Dunnett’s multiple comparison (parametric) procedure in the same package was used for comparison of data among groups. When the groups were heterogeneous by Bartlett’s test, Dunnett’s multiple comparison (nonparametric) in the same package was used. Statistical significance was evaluated at p < .05 and .01.
Results
General Condition, Nervous Symptoms, and Auditory Response
Effects on body weight and brain weight, nervous symptoms, and auditory response are indicated in Table 1. One animal administered 12 mg/kg was sacrificed in a moribund condition on day 4 due to decreased body weight and severe nervous symptoms, and excluded from other examinations. In the TMT-treatment groups, body weight was significantly decreased at 12 mg/kg on days 4 and 7 and slightly decreased at 9 mg/kg on days 4 and 7. Absolute brain weight was not changed in any group. Nervous symptoms such as tremor and hypersensitivity were observed in several animals at 12 mg/kg from day 2 and at 9 mg/kg from day 4. A few animals at 12 mg/kg showed decreased locomotor activity from day 2. Decreased auditory response was observed in 1 animal at 12 mg/kg on day 1, in 3 animals at 12 mg/kg on days 4 and 7, and in 1 animal at 9 mg/kg on days 4 and 7.
Effects of TMT treatment on body weights, brain weights, nervous symptoms, auditory response, and histopathology.
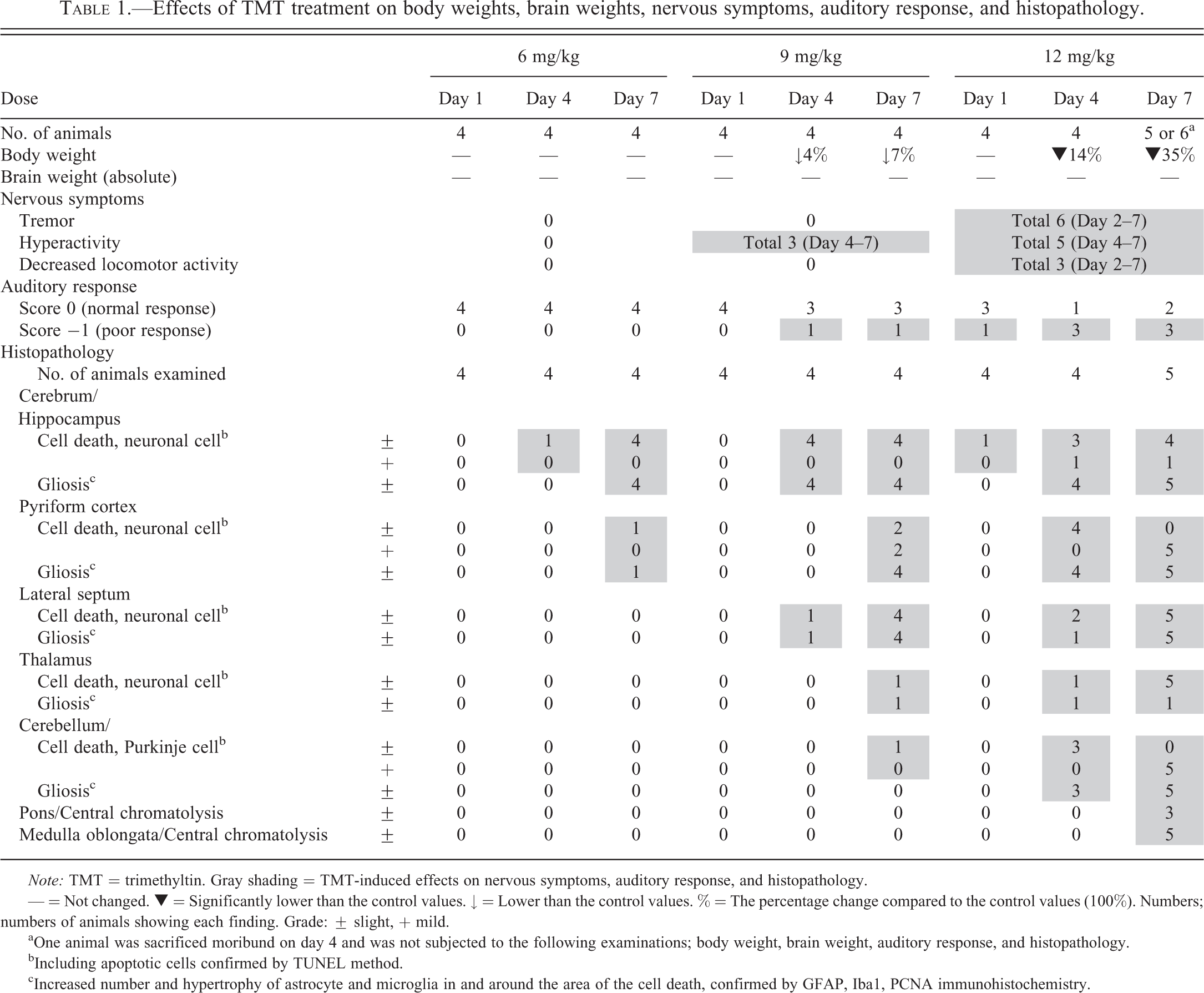
Note: TMT = trimethyltin. Gray shading = TMT-induced effects on nervous symptoms, auditory response, and histopathology.
— = Not changed. ▼ = Significantly lower than the control values. ↓ = Lower than the control values. % = The percentage change compared to the control values (100%). Numbers; numbers of animals showing each finding. Grade: ± slight, + mild.
aOne animal was sacrificed moribund on day 4 and was not subjected to the following examinations; body weight, brain weight, auditory response, and histopathology.
bIncluding apoptotic cells confirmed by TUNEL method.
cIncreased number and hypertrophy of astrocyte and microglia in and around the area of the cell death, confirmed by GFAP, Iba1, PCNA immunohistochemistry.
Histopathological Changes in the Brain
The results of histopathological examination are shown in Table 1 and in Figures 1 to 4. Neural cell death was observed in hippocampus, pyriform cortex, lateral septum, and thalamus in the cerebrum and in Purkinje cells in the cerebellum at 12 mg/kg from day 1 and at 6 and 9 mg/kg from day 4. On day 1, cell death was noted in only a few pyramidal cells in hippocampus in 1 animal at 12 mg/kg. Subsequently, the severities of this finding and affected regions were increased in a dose- and time-dependent manner. Pyramidal cells in Ammon’s horn CA1, CA3, and CA4 areas in hippocampus were the most susceptible (Figure 1). Neural cells in pyriform cortex and lateral septum were moderately susceptible and granule cells in the dentate gyrus and Purkinje cells in the cerebellum were mildly damaged. Central chromatolysis of neural cells in pons and medulla oblongata was also observed at 12 mg/kg on day 7. In TUNEL method, some of the dead neural cells showed positive reaction (Figure 2). Neural cell death was accompanied by reactions of glial cells. Immunohistochemical stains for Iba1 (Figure 3A) and GFAP (Figure 3B) revealed hypertrophy and increased number of microglias and astrocytes in and around the area of the cell death, which gradually increased from day 4. In addition, PCNA immunohistochemistry showed increased PCNA-positive proliferating cells were increased mainly in reactive glial cells in hippocampus and pyriform according to neural cell damage, and were also slightly increased in dentate gyrus granule cells, indicating neurogenesis at 12 mg/kg on days 4 and 7 (Figure 4) and at 9 mg/kg on day 7.
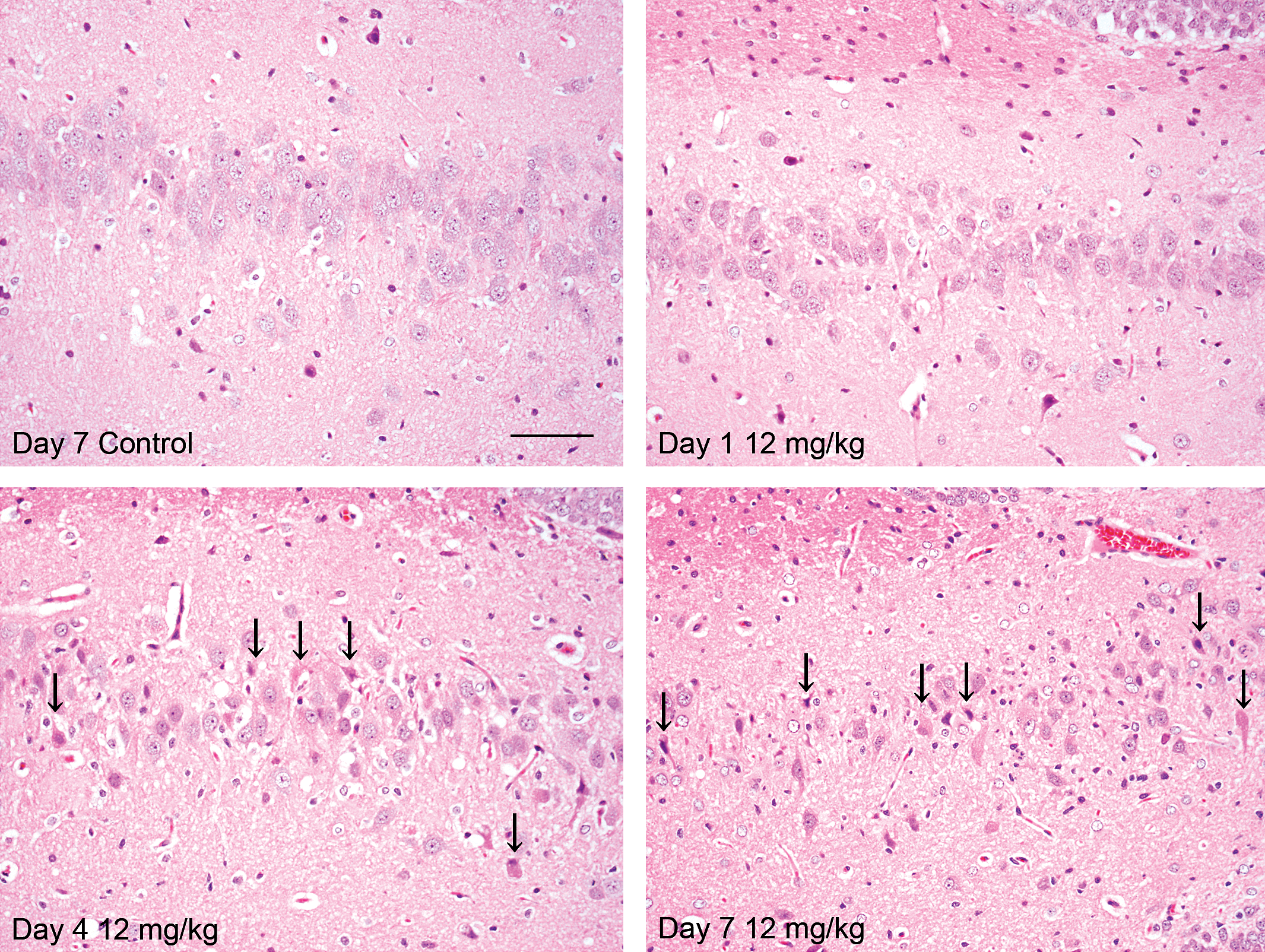
Photographs of histopathological findings. Neural cell death (arrows) and peripheral infiltration of glial cells in hippocampus (CA3) in TMT-treated animals. Hematoxylin eosin. Scale bar = 50 μm. TMT = trimethyltin.
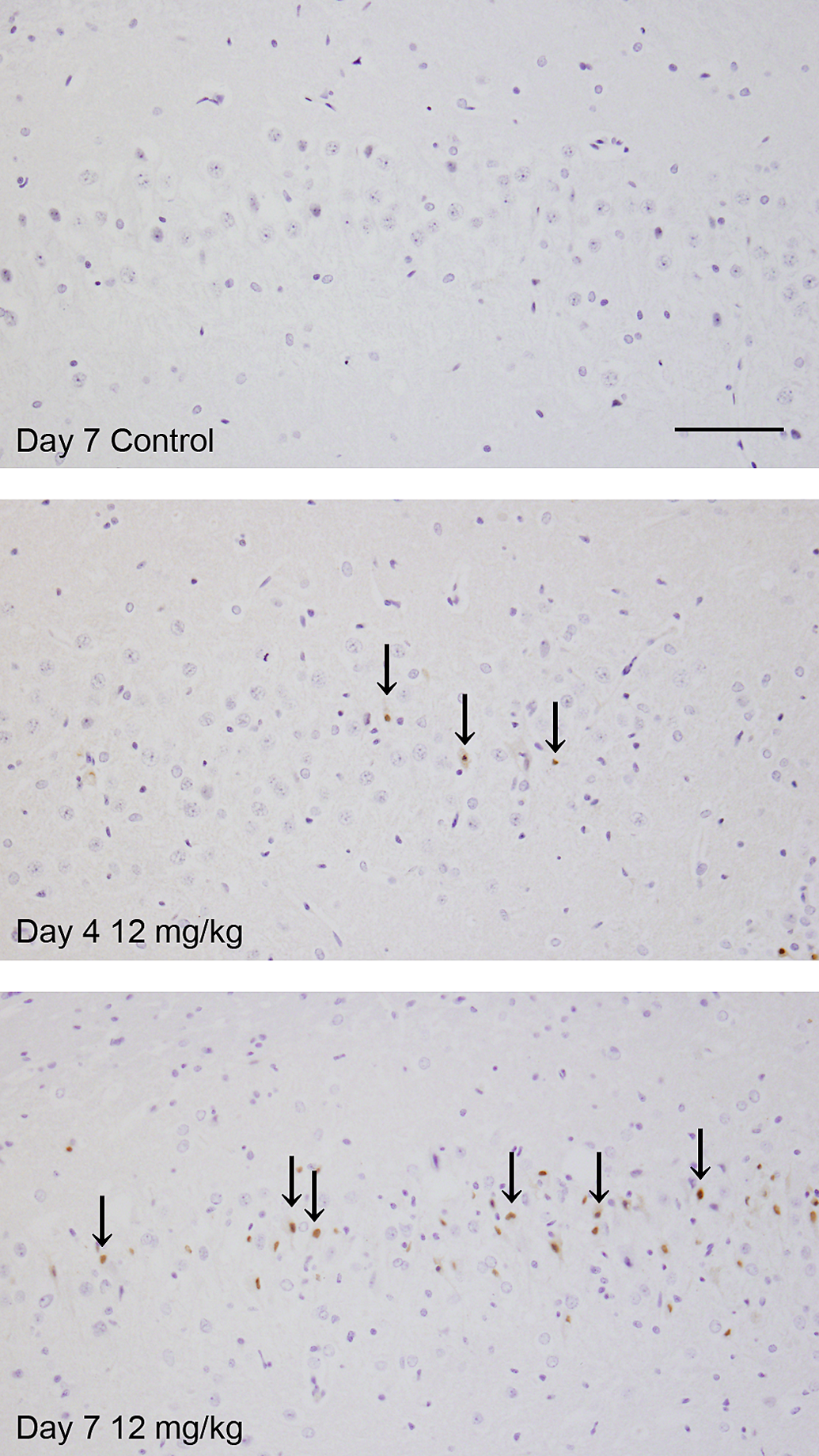
Photographs of TUNEL method. Increased TUNEL-positive cells indicating apoptosis in hippocampus (CA3) in TMT-treated animals. Scale bar = 50 μm.
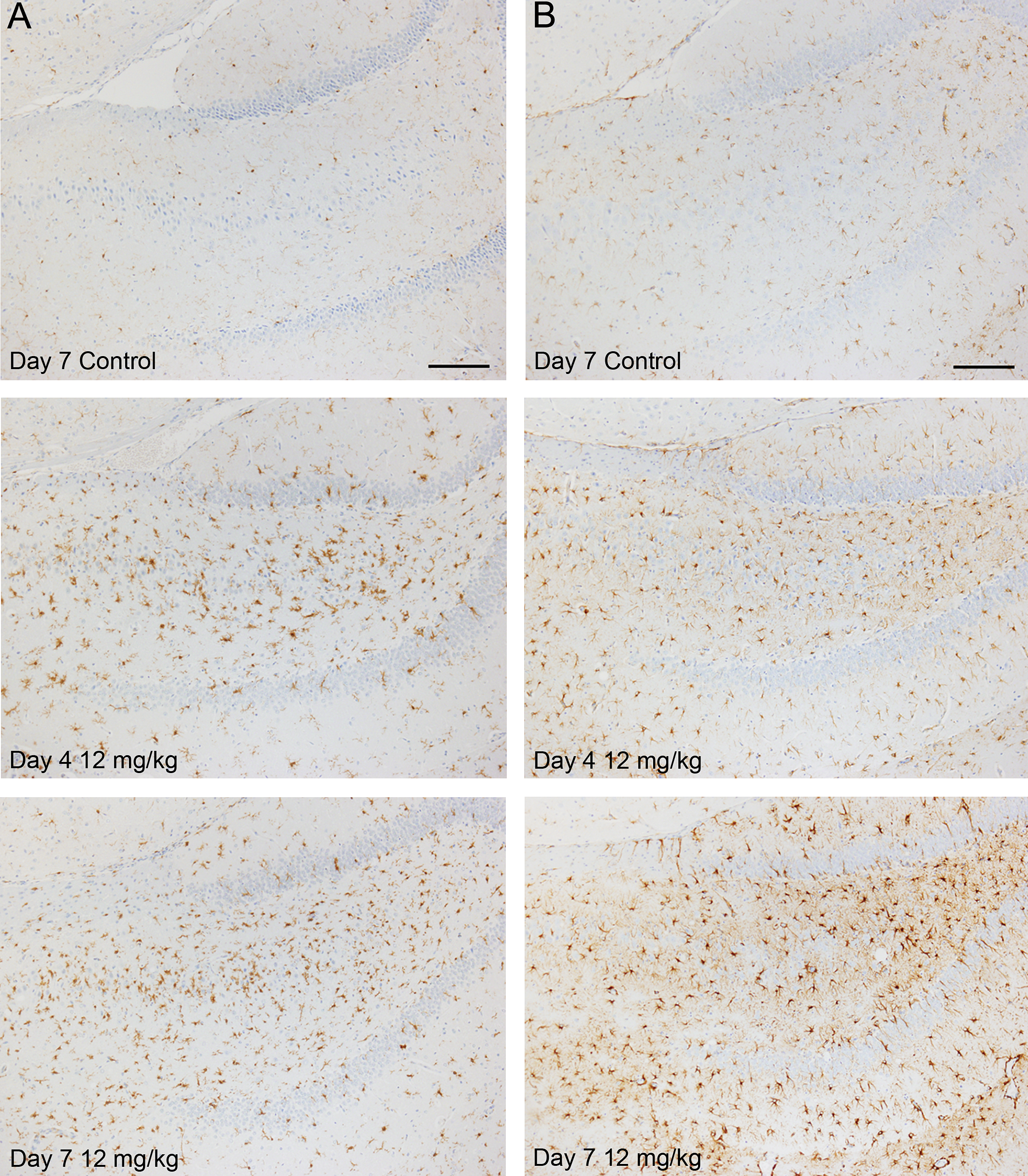
Photographs of immunohistochemical stains for Iba1 and GFAP. (A) Iba1. Increased Iba1-positive cells (microglias) in hippocampus, especially CA3 area, in TMT-treated animals. Scale bar = 100 μm. (B) GFAP. Increased GFAP-positive cells (astrocytes) in hippocampus in TMT-treated animals. Scale bar = 100 μm. GFAP = glial fibrillary acidic protein; Iba1 = ionized calcium–binding adapter molecule 1.
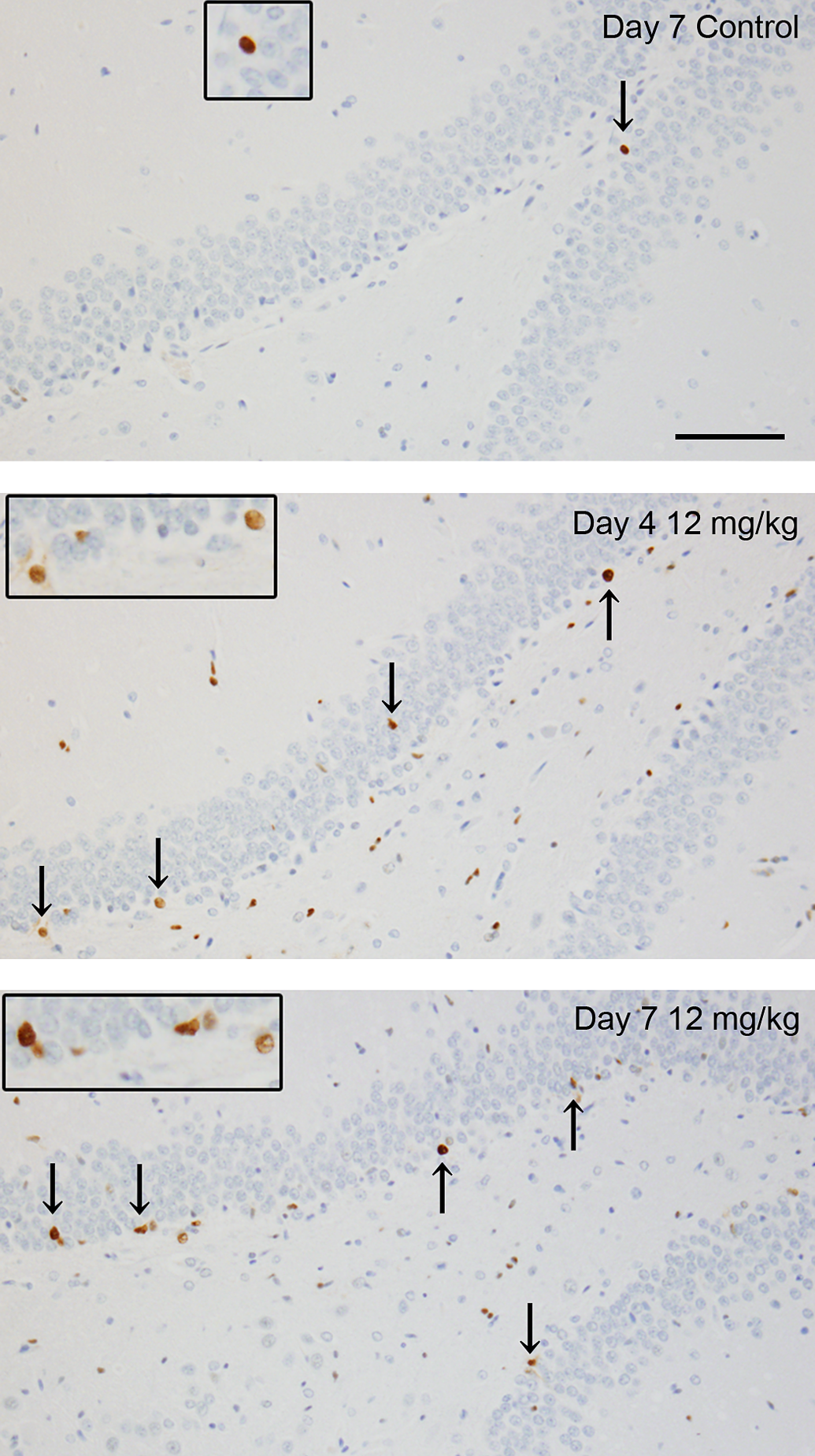
Photographs of immunohistochemical stains for PCNA. Increased PCNA-positive cells (proliferating cells) were observed mainly in glial cells in CA3 area and slightly dentate gyrus granule cells (arrows) in TMT-treated animals. PCNA. Scale bar = 50 μm. PCNA = proliferating cell nuclear antigen; TMT = trimethyltin.
MiR-9* and MiR-384-5p Levels in Serum and Hippocampus
MiRNA levels in serum are shown in Figure 5. The color-coded individual plots indicate the individual relative fold change of serum miRNA level and individual information about nervous symptoms, decreased auditory response, and histopathological changes.
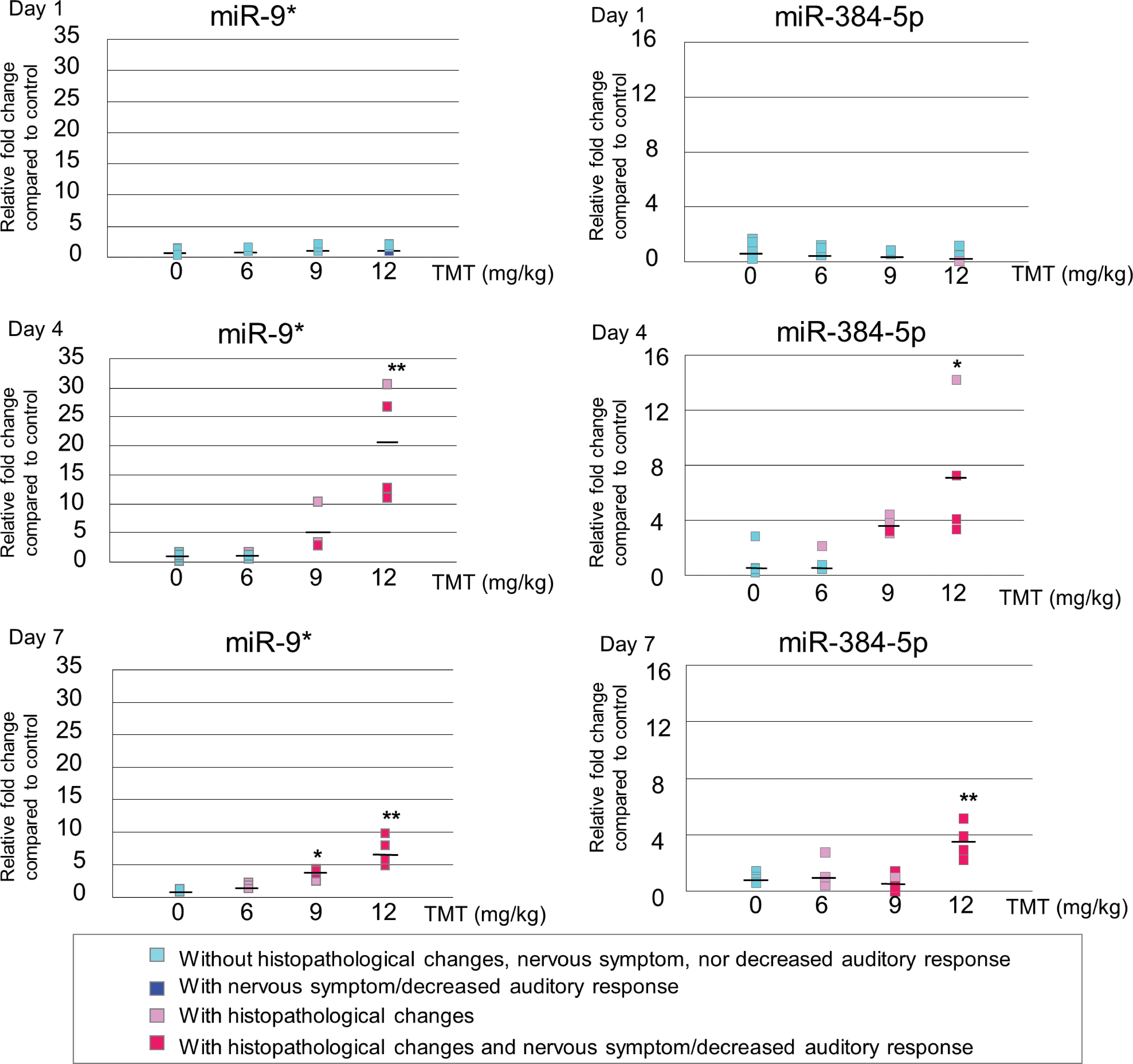
Effect of TMT treatment on miR-9* and miR-384-5p levels in serum. MiRNA levels were analyzed by RT-PCR and expressed as relative fold change compared to the control mean levels. The color-coded individual plots indicate the individual relative fold change of serum miRNA and individual information about the presence or absence of nervous symptom, decreased auditory response, and histopathological changes. Bars show mean values. Significantly different from the control at *p < .05 and **p < .01, respectively. TMT = trimethyltin; RT-PCR = reverse transcriptase polymerase chain reaction.
No change in either miRNA was observed in any group on day 1. MiR-9* level was significantly elevated at 12 mg/kg on day 4 and at 9 and 12 mg/kg on day 7, and it was slightly elevated at 9 mg/kg on day 4, without statistical significance. MiR-384-5p level was significantly elevated at 12 mg/kg on days 4 and 7, and it was slightly elevated at 9 mg/kg on day 4, without statistical significance. Mean fold changes compared to control groups of serum miR-9* were 4.9 at 9 mg/kg and 20.4 at 12 mg/kg on day 4 and 3.5 at 9 mg/kg and 7.0 at 12 mg/kg on day 7. Mean fold changes of serum miR-384-5p were 3.6 at 9 mg/kg and 7.2 at 12 mg/kg on day 4, and 3.6 at 12 mg/kg on day 7. On each observation day, the degree of elevation increased with dose; however, both serum miRNA levels were more highly elevated on day 4 than on day 7. In addition, the degree of elevation of serum miR-9* was larger than that of miR-384-5p at all measured points. From the data of individual animals, on day 4, almost all animals showing histopathological changes expressed high serum miR-9* and miR-384-5p levels. On the other hand, on day 7, both miRNAs were less elevated than on day 4. Elevation of miR-9* level was detected in severely affected animals at 9 and 12 mg/kg that showed both histopathological changes and nervous symptom/auditory response, and elevation of miR-384-5p levels was similarly detected in severely affected animals at 12 mg/kg. In contrast to the results of serum miRNAs, both miRNA expression levels in hippocampus on day 7 were unchanged (data not shown).
Discussion
In this study, we detected elevations of serum miR-9* and miR-384-5p levels caused by TMT treatment, and compared their sensitivities for detecting neurotoxicity with other examined parameters. These serum miRNAs were considered to be promising markers for detecting neurotoxicity and can be used in noninvasive ways.
On day 1, one animal administered 12 mg/kg showed a slight histopathological change in hippocampus, but no change in either miRNA level was observed in any group. Since only a few pyramidal cell deaths were observed only in 1 animal at this point, miRNA alteration was considered to be at an undetectable level. In addition, auditory response was decreased at 12 mg/kg in 1 animal. Decreased startle response was reported to be observed from 2 hr after intraperitoneal administration of TMT 4, 5, and 6 mg/kg and to persist for several weeks (Ruppert, Dean, and Reiter 1984), and high-frequency hearing loss caused by TMT is known to be associated with cochlear inner and outer hair cell and spiral ganglion cell damage (Liu and Fechter 1996; Ruppert, Dean, and Reiter 1984; Crofton et al. 1990; Young and Fechter 1986; Eastman, Young, and Fechter 1987; Fechter, Young, and Nuttall 1986). Therefore, even on day 1, decreased auditory response due to TMT-impaired auditory function may be possible. However, this parameter has low specificity for detecting neurotoxicity, since grossly decreased response to clicker sound was caused not only by impaired auditory function but also by weakened general condition. Therefore, whether this indicated neurotoxicity or not could not be decided. In addition, decreased locomotor activity observed from day 2 at 12 mg/kg was considered to be due to weakened condition of animals rather than to TMT-induced neurotoxicity.
On day 4, serum miR-9* and miR-384-5p levels were elevated at 9 and 12 mg/kg, with the increased incidence and severity of histopathological changes with dose, decreased auditory response, and nervous symptoms such as tremor and hyperactivity that were reported as major symptoms induced by TMT (Dyer et al. 1982). Focusing on the changes at 9 and 12 mg/kg, elevation of serum miRNA levels was detectable even in animals not showing nervous symptoms or decreased auditory response. This suggests that miRNA levels changed in response to TMT with greater sensitivity than did presentation of nervous symptoms. On the other hand, 1 animal at 6 mg/kg showed only histopathological change in hippocampus. As in the 1 animal administered 12 mg/kg on day 1 described previously, it was considered that the neural cell damage was too slight to result in detectable serum miRNA alteration.
On day 7, susceptibilities of both serum miRNAs were slightly lower than on day 4. Elevation of serum miR-9* expression level could be detected only in severely affected animals at 9 and 12 mg/kg that showed both histopathological changes and nervous symptom/auditory response. Elevation of serum miR-384-5p levels was also detectable only in severely affected animals, at 12 mg/kg.
From the view of the time course, both serum miRNA levels were most highly elevated on day 4 and the increases were slightly less on day 7. From this result, elevations of both miRNA levels in serum were considered to peak around day 4. Histopathologically, TMT induced neuronal cell death with glial reaction mainly in hippocampus, pyriform cortex, and lateral septum in the cerebrum with an increase in the severity and the incidence with dose and time. Focusing on histopathological changes in hippocampus, which is the most susceptible region, at 9 mg/kg and 12 mg/kg the severity and the incidence of neuronal cell death reached a plateau from day 4 and glial reaction increased from day 4 to day 7, as confirmed by Iba1 and GFAP immunohistochemistry. Recovery response to neuronal cell loss in dentate gyrus granule cells also occurred on day 4 and day 7, as confirmed by PCNA immunohistochemistry. These results were consistent with other reports. Neuronal cell death in hippocampus appeared 2 days after a single intraperitoneal administration of 8 mg/kg TMT and reached maximum 4 days after administration (Balaban, O'Callaghan, and Billingsley 1988). TMT-induced neurodegeneration in rodents enhanced neurogenesis in the dentate gyrus granule cells and CA3 regions, in which a regeneration of granule cell layer neurons has been hypothesized (Corvino et al. 2005; Corvino et al. 2013; Geloso, Corvino, and Michetti 2011; Ogita et al. 2005; Ogita et al. 2012). Considering the time course, elevation of both miRNA levels was considered to be most susceptible at the early to middle stage of damage in hippocampus. At 9 mg/kg and 12 mg/kg in hippocampus, TMT caused cell damage that reached a plateau from day 4 and elevation of serum miRNA levels occurred at that point, and glial reactions then increased even after serum miRNA levels passed the peak of elevation and dropped down to control levels. From this result, the mechanism of elevation of serum miR-9* and miR-384-5p levels was speculated to be a leakage from damaged brains.
The significance of circulating miRNAs has been attracting attention. As it has been reported that tumor-derived miRNAs can enter the circulation even when originating from an epithelial cancer type (Mitchell et al. 2008), elevated miRNA is both downregulated and upregulated in tissues. Some miRNAs may be packed into exosomes and excreted into the bloodstream and other body fluids for intercellular communication (Hannafon and Ding 2013). These extracellular circulating miRNAs are stable and resistant to degradation by nucleases (Turchinovich et al. 2011). Moreover, miRNAs leaked from necrotic or lysed cells are also known to exist in the circulation in tissue injury (Su et al. 2012; Wang et al. 2009). In traumatic brain injury cases, plasma miR-765 and miR-124 levels elevated immediately after injuries were considered to have been released from the injured brain (Omar et al. 2009; Redell et al. 2010). Apoptotic bodies are also known to come into circulation, as microvesicles (Simpson et al. 2009).
In our case, leakage from both necrotic or lysed cells and apoptotic cells was considered to contribute to elevations of serum miRNA levels. The mechanism of selective neuronal death caused by TMT is not fully understood. Both necrotic and apoptotic mechanisms have been reported; however, several in vitro and in vivo studies have indicated that TMT-induced cell death was mainly due to apoptosis, with an increase in TUNEL labeling, DNA fragmentation, and activation of caspase family members (Fiedorowicz et al. 2001; Figiel and Fiedorowicz 2002; Geloso, Corvino, and Michetti 2011; Jenkins and Barone 2004; Morita et al. 2008; Qing et al. 2013; Shintani et al. 2007). In the present study, apoptotic bodies were not clearly observed, and most dead cells on HE slides were considered as necrotic cells. However, TUNEL-positive cells among the dead cells were observed on days 4 and 7 at similar or slightly higher degree with time. Considering findings from HE slides and the fact that TUNEL method is not a specific marker for apoptosis, observed TUNEL-positive cells could actually include some amount of necrotic cells. However, it was also reported that apoptosis of pyramidal cells in hippocampus reached maximum at 5 days after gavage treatment of TMT in water to rats at a dose of 9 mg/kg (Morita et al. 2008). Therefore, in the present study, some of the cell deaths of pyramidal cells in hippocampus were apparently caused by apoptosis, and this might have contributed to the peak in elevation of serum miRNAs.
Moreover, the degree of elevation of serum miR-9* level was higher than that of miR-384-5p at all points in time. Since miR-384-5p is specifically expressed in hippocampus, but miR-9* is expressed widely in the nervous system, higher elevation of serum miR-9* level may reflect not only damage from hippocampus but also damage from other brain regions such as pyriform cortex and lateral septum.
In this study, neither miRNA expression levels in hippocampus on day 7 were changed. A question as to why expression levels of either miRNA in hippocampus did not alter in this study despite the existence of neuronal cell loss arises, but one possible reason is that alterations of these miRNAs in serum are more sensitively detectable than those in hippocampus, since the levels are very low in serum but high in brain under normal conditions. In contrast to our results, some reports suggested that miR-9* was increased or decreased in the nervous tissues in association with abnormal development of nervous system, neurodegenerative disease, and brain tumors (Nass et al. 2009; Packer et al. 2008; Schraivogel et al. 2011; Wei et al. 2013; Zhang et al. 2010). In neural tube defects (NTDs), a downregulation of miR-9* has been reported. In particular, reports on rat spina bifida aperta, a common NTD, suggested repressor element-1 silencing transcription factor (REST)-regulated miR-9*chromatin remodeling regulatory mechanism may play a role in the neuronal deficits of NTDs (Wei et al. 2013; Zhang et al. 2010). In addition, research on Huntington's disease (HD) showed that expression levels of miR-9/miR-9* were decreased in the cerebral cortex of HD as the disease progresses and that they target 2 components of REST complex, REST and corepressor element-1 silencing transcription factor (CoREST; Packer et al. 2008). Meanwhile, miR-9/9* are overexpressed in human brain primary tumors and CD133+ glioblastoma stem cells, and calmodulin-binding transcription activator 1 (CAMTA1), a novel tumor suppressor, was found as an miR-9/9* target (Nass et al. 2009; Schraivogel et al. 2011). In the present study, molecular changes relating to cellular differentiation are considered to be caused to some degree by TMT treatment in association with neurogenesis. However, no detectable alteration of miR-9* expression level in hippocampus was induced during the observation period. Therefore, as mentioned previously, the mechanism of elevated serum miR-9* level was apparently the leakage rather than the functional induction. On the other hand, miR-384-5p was shown to be highly expressed in hippocampus in a profiling study of rat brain miRNAs (He et al. 2007), and recently, decreased expressions of miR-384-5p in ischemic myocardium of rats and hypoxic rat ventricular cells have been reported. In these models, apoptosis was upregulated by miR-384-5p and was downregulated by miR-384-5p inhibitor, by targeting phosphatidylinositol-4,5-bisphosphate 3-kinase, catalytic subunit delta (PI3K p110δ; Bao et al. 2013), but little is known about the function of miR-384-5p in brain. No histopathological changes in heart were observed in this study (data not shown). If functional alteration of miR-384-5p induced by ischemia had occurred in brain by TMT treatment, its tissue level should be decreased, but it was actually not changed. Moreover, the elevation of serum miR-384-5p level was consistent with the severe hippocampus neuronal cell damage. Therefore, though elucidation of its function in brain and expression in other organs are still challenges for the future, the mechanism of elevation of its serum level was considered the leakage from damaged neuronal cells rather than the functional alteration induced by ischemia in brain or heart.
In this study, we successfully detected elevation of brain-enriched miRNAs in serum in association with neuronal damage caused by TMT treatment. So far, blood miRNAs as markers of nervous system cancer, disease, and injury have been reported by several researchers. However, no blood miRNA marker in neurotoxicity model has been reported. To our knowledge, this is the first report showing that blood miRNAs were elevated in a neurotoxicant-induced rat model. In conclusion, miR-9* and miR-384-5p were considered as possible novel indicators of neurotoxicity that can be detected by a noninvasive approach. Although further research is still needed, they are expected to be applied in evaluation of neurotoxicity in safety assessment of chemical materials and agricultural chemicals. Furthermore, these miRNAs might be also related to human neuronal lesions, and therefore they are also expected to be used in monitoring of human neuronal lesions or in evaluation of efficacy of drugs targeting nervous system disorders.
Footnotes
Acknowledgment
We wish to express our appreciation to Ms. Yamaguchi-Katsura and Ms. Maeda for the expert technical assistance they kindly provided. We also thank all members of the Environmental Health Science Laboratory of Sumitomo Chemical Co., Ltd., and all members of the Laboratory of Veterinary Pathology of Osaka Prefecture University for their invaluable support.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
