Abstract
A wide variety of markers are available to assess the function and pathology of the gastrointestinal (GI) tract. This review describes some of these markers with special emphasis given to markers used in dogs and cats. Small intestinal disease can be confirmed and localized by the measurement of serum concentrations of folate and cobalamin. Fecal α1-proteinase inhibitor concentration can increase in individuals with excessive GI protein loss. A wide variety of inflammatory markers are available for a variety of species that can be used to assess the inflammatory activity of various types of inflammatory cells in the GI tract, although most of these markers assess neutrophilic inflammation, such as neutrophil elastase, calprotectin, or S100A12. N-methylhistamine can serve as a marker of mast cell infiltration. Markers for lymphocytic or eosinophilic inflammation are currently under investigation. Exocrine pancreatic function can be assessed by measurement of serum concentrations of pancreatic lipase immunoreactivity (PLI) and trypsin-like immunoreactivity (TLI). Serum PLI concentration is increased in individuals with pancreatitis and has been shown to be highly specific for exocrine pancreatic function and sensitive for pancreatitis. Serum TLI concentration is severely decreased in individuals with exocrine pancreatic insufficiency.
Keywords
Introduction
There are different methodologies to assess function and/or disease of various organ systems. In general, there are morphologic and functional diagnostic tools. Morphologic diagnostic tools include such basic tools as physical examination, gross morphologic pathology, or radiography, but also more sophisticated technologies, such as computed tomography, magnetic resonance imaging, or others. The downside of these more advanced diagnostic tools to assess morphology is cost. Oftentimes the equipment is very expensive and evaluation is limited to only a few individuals per 24-hr period, making each single assessment very expensive. In contrast, novel functional tools may also involve expensive equipment, but usually biological samples can be shipped to a central location and the equipment can analyze hundreds if not thousands of samples in a 24-hr period, making each single assessment relatively affordable and allowing the investigator to evaluate multiple parameters in the same individual and at the same time. As a consequence, development of new functional diagnostic tools is rapid with a wide range of assays available for routine use.
In general, functional diagnostic tools can be divided into static and dynamic markers, with the majority of parameters being assessed being static markers. At least in part, this is likely due to the fact that static markers are usually easier to assess than dynamic markers. Both static and dynamic markers can be assessed in a variety of biological samples, such as whole blood, serum, plasma, urine, feces, cerebrospinal fluid, and many others. Dynamic markers involve administration of a stimulant, inhibiting agent, metabolite, or a pharmaceutical substance, which may be associated with adverse effects. Also, a second sample has to be collected in order to compare the post-administration result to the baseline value.
The diagnostic utility of any marker substance is determined by sensitivity, specificity, accuracy, positive predictive value, and negative predictive value (Greiner and Gardner 2000). However, it is key to recognize that these performance parameters are not fixed for a marker assay but are in fact dependant on where the cutoff value for a specific diagnosis or outcome is being placed. By moving the cutoff value up or down, the sensitivity of a marker assay may decrease, but at the same time specificity will likely increase and vice versa. This relationship of sensitivity and specificity for a marker assay for a specific outcome can be described in a receiver operating curve, and depending on the aim for the marker assay, the cutoff value for a specific diagnosis or outcome can be optimized (Gardner and Greiner 2006; Zweig and Campbell 1993).
The following is a discussion of marker assays that are commonly used to evaluate gastrointestinal (GI) function and pathology in dogs and cats. Assays for some markers maybe highly species specific, while others may not be. However, before using an assay that has been analytically validated for one species in another species, the assay should be analytically validated for that species. Also, reference intervals may differ dramatically for different species, placing many results from healthy individuals outside the working range of an assay and requiring dilution of the sample.
Marker Assays to Assess the Intestinal Tract
Serum Folic Acid Concentration
Folic acid is a water-soluble vitamin that belongs to the B-group vitamins (vitamin B9) and can be measured by automated assay systems. In general, these assays are not species specific, but the minimum sample volume may preclude using these assays in small rodents. Also, most assays have been optimized for measurement of folic assay in serum or plasma from humans and before use in a different species the assay should be analytically validated for that species.
In the diet, folic acid is mainly supplied as folate polyglutamate, which cannot be absorbed. In the proximal small intestine, folate polyglutamate is deconjugated by folate deconjugase and the resulting folate monoglutamate is absorbed by specific folate carriers that are also located in the proximal small intestine (Figure 1; Rosenberg and Godwin 1971).
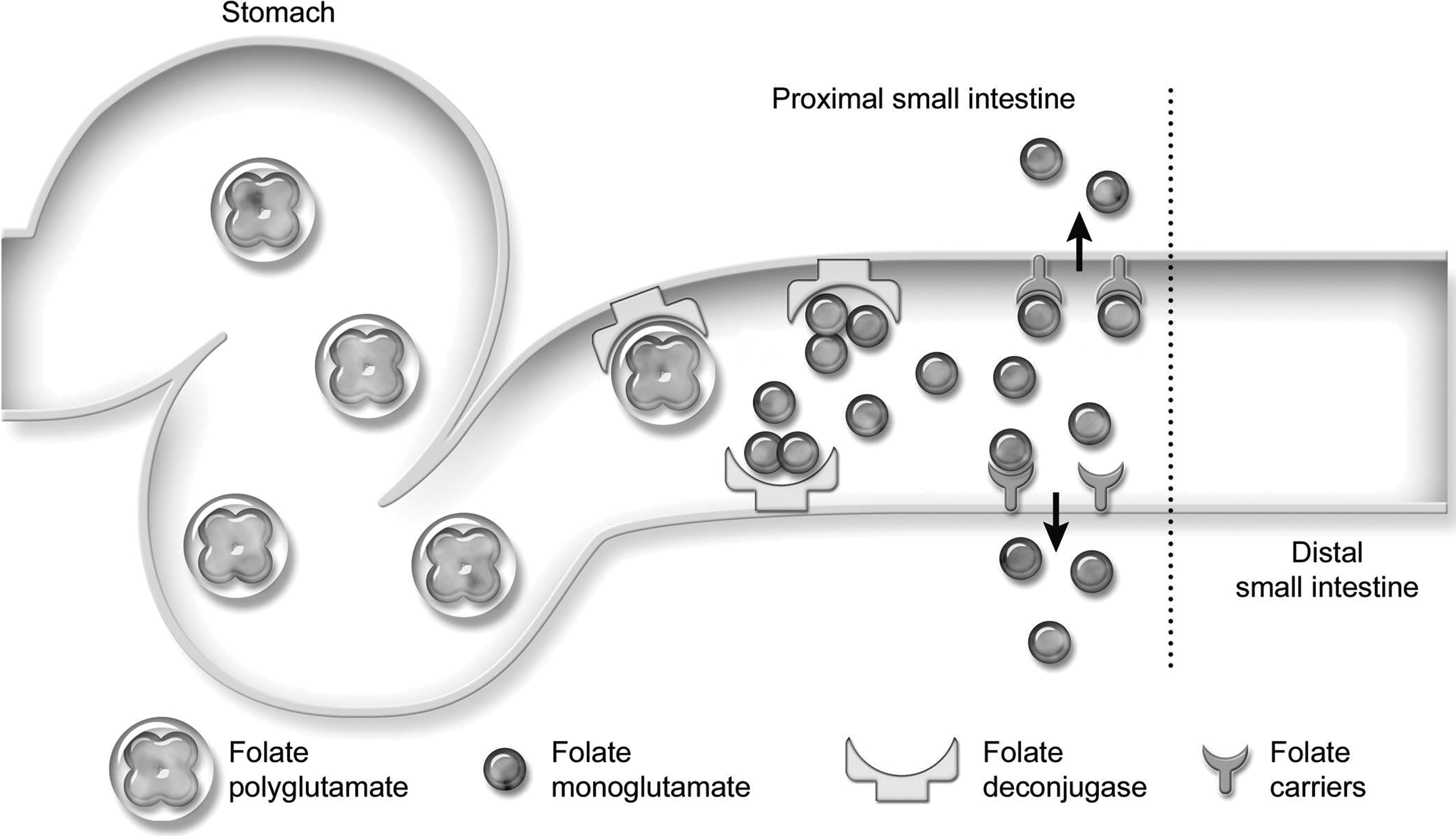
Folate absorption. Dietary folate enters the gastrointestinal tract predominantly as folate polyglutamate. Folate deconjugase, a brush border enzyme in the cranial small intestine, deconjugates folate polyglutamate to folate monoglutamate. Specific folate carriers on enterocytes in the cranial small intestine subsequently absorb folate monoglutamate. Both the deconjugase and folate carrier molecules are restricted to the cranial small intestine. Reprinted from Suchodolski and Steiner (2003) with permission from Elsevier.
In patients with proximal small intestinal disease, both folate deconjugase and folate carriers can be destroyed (Rosenberg 1975). If the disease process is severe enough either folate polyglutamate is no longer deconjugated or folate monoglutamate is no longer absorbed, leading to folate malabsorption. If malabsorption of folate persists, folate body stores are becoming depleted and serum folate concentration decreases. The same is true in patients with diffuse small intestinal disease if the proximal small intestine is involved in the disease process. Many bacterial species synthesize folic acid, and it is believed that an increased number of bacterial species (i.e., small intestinal dysbiosis) can lead to significant increases in serum folate concentrations (Suchodolski and Steiner 2003).
Serum Cobalamin Concentration
Cobalamin (vitamin B12) is also a water-soluble vitamin. While herbivores can absorb cobalamin synthesized by their own microbiota, carnivores are dependent on dietary cobalamin. However, cobalamin is plentiful in most commercial animal foods and dietary deficiencies are thus uncommon unless a carnivore is fed a vegan diet without being supplemented.
Dietary cobalamin is bound to animal-based dietary protein and cannot be absorbed in this form. In the stomach, dietary protein is digested by pepsin and HCl and cobalamin is being released. The free cobalamin is immediately bound by R-protein, which is a cobalamin transporter protein that is synthesized by the gastric mucosa (Dali-Youcef and Andrès, 2009). In the small intestine, R-protein is digested by pancreatic proteases and the free cobalamin is bound by intrinsic factor (Dali-Youcef and Andrès 2009). In dogs and cats, the vast majority of intrinsic factor is secreted by the exocrine pancreas (Fyfe 1993; Steiner 2008). This is in sharp contrast to humans, where the majority of intrinsic factor is secreted by the stomach (Dali-Youcef and Andrès 2009). Intrinsic factor/cobalamin complexes are absorbed by specific receptors in the ileum (Figure 2).
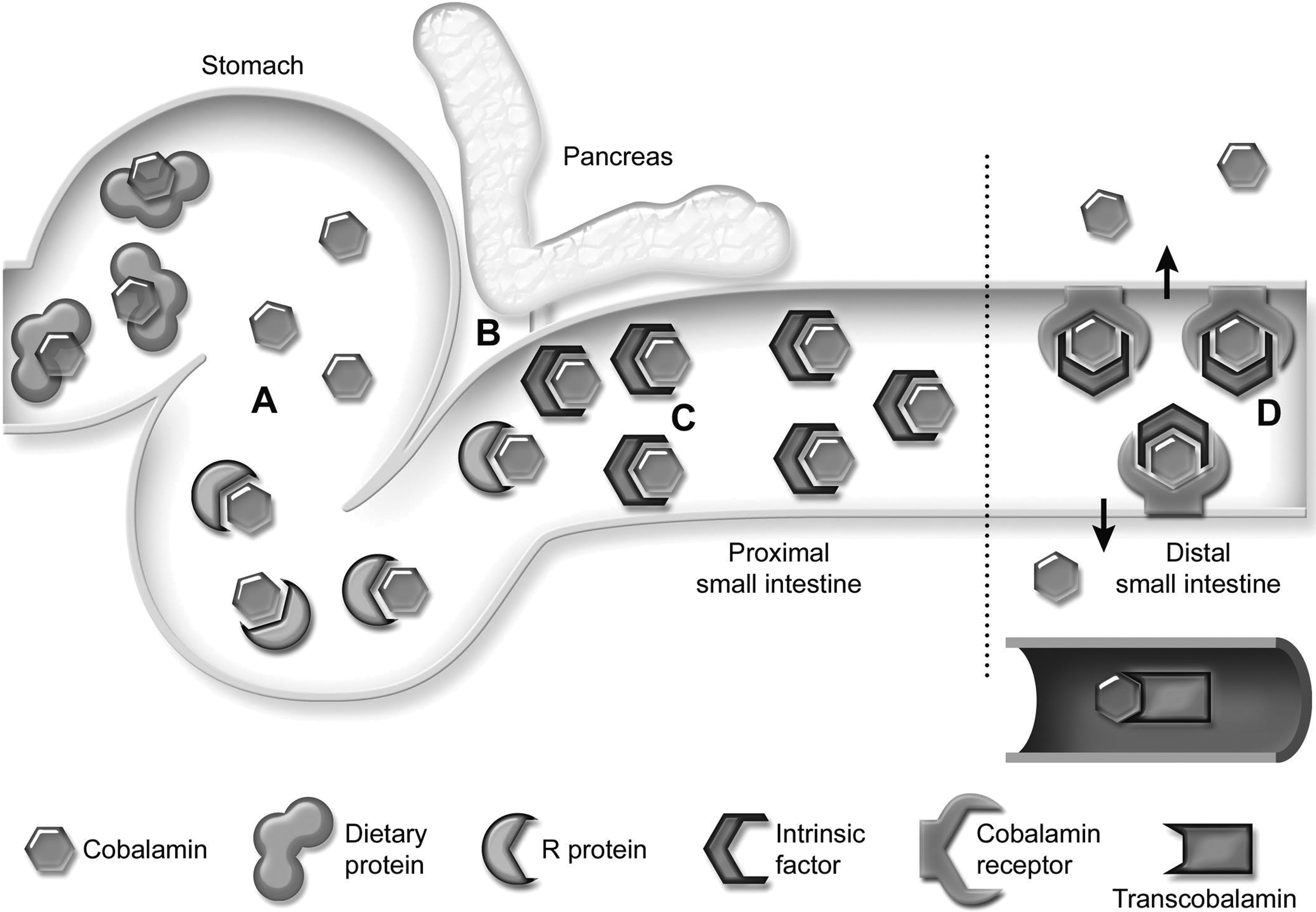
Cobalamin absorption. Dietary cobalamin is bound to dietary protein. In the stomach, pepsin and hydrochloric acid degrade the dietary protein, releasing cobalamin (A). The cobalamin is immediately bound by R-protein, which is produced by the gastric mucosa. In the duodenum, pancreatic proteinases digest the R-protein, releasing the cobalamin. Free cobalamin in the duodenum is bound by intrinsic factor (B). In humans, intrinsic factor is primarily produced by the stomach, but in dogs and cats the majority of intrinsic factor is synthesized by the exocrine pancreas. Cobalamin remains bound to intrinsic factor during passage through the cranial small intestine (C). In the distal small intestine, the cobalamin/intrinsic factor complexes are taken up by specific receptors found only on enterocytes in the ileum (D). Enterocytes then process the intrinsic factor/cobalamin complex and release cobalamin into the circulation, where a final set of binding proteins (transcobalamins) complex the vitamin and carry it to the cells. Reprinted from Suchodolski and Steiner (2003) with permission from Elsevier.
Distal small intestinal disease, if severe, will lead to destruction of cobalamin receptors in the ileum, ultimately leading to cobalamin malabsorption. However, healthy individuals store vast amounts of cobalamin, mostly in the liver, but also in the kidneys. The depletion of said body stores of cobalamin can take between weeks and even years, depending on the species, the cobalamin stores before malabsorption ensues, and the severity of the disease process (Simpson et al. 2001). Diffuse small intestinal disease can also lead to cobalamin malabsorption if the ileum is involved in the disease process. In dogs and cats, exocrine pancreatic insufficiency (EPI) also very commonly leads to cobalamin deficiency as these patients will be deficient of intrinsic factor (Batchelor et al. 2007; Thompson et al. 2009). Finally, an increased number of bacterial organisms in the small intestine (i.e., small intestinal dysbiosis) will lead to competition for the available cobalamin and may also lead to cobalamin deficiency (Suchodolski and Steiner 2003).
Serum C-reactive Protein Concentration
C-reactive protein (CRP) is an acute phase reactant that is being released by hepatocytes in response to tissue damage and/or inflammation (Krause 2001). Thus, CRP is not specific for diseases of the GI tract but can be increased in a wide variety of diseases (Burton et al. 1994; Chan, Rozanski, and Freeman 2009). However, in dogs diagnosed with inflammatory bowel disease (IBD), measurement of serum concentrations of CRP can be used to objectively assess disease activity and monitor treatment efficacy (Jergens et al. 2003). A species-specific assay for the measurement of CRP in serum samples from dogs is commercially available and has been analytically validated, but an assay for use in cats is not available (Eckersall, Conner, and Parton 1989).
Fecal α1-proteinase Inhibitor Concentration
Many GI disorders, if severe, can be associated with GI protein loss. Traditionally, the gold standard for evaluation of GI protein loss is 51Cr-albumin excretion, but this diagnostic test is labor and time intensive and is also associated with exposure of the individual and personnel to radioactivity (Frias et al. 2010).
In humans, fecal α1-proteinase inhibitor (α1PI; formerly also known as α1-antitrypsin) clearance is used to diagnose excessive protein loss in the GI tract (Bai et al. 1991; Karbach, Ewe, and Bodenstein 1983). Species-specific assays for the measurement of canine and feline but also marmoset α1-PI concentration in feces have also been developed and analytically validated (Burke, Ruaux, et al. 2012; Melgarejo, Williams, and Asem 1998). α1-PI is synthesized in the liver and inhibits a variety of different proteinases, mainly elastase and trypsin. α1-PI has a molecular mass of approximately 60,000 Da, which is similar to that of albumin. Thus, when GI disease is severe enough to be associated with GI loss of albumin, α1-PI is lost as well (Figure 3; Burke, Broussard, et al. 2012). In contrast to albumin, α1-PI is not hydrolyzed by digestive and bacterial proteinases in the GI lumen. Therefore, fecal α1-PI clearance or concentration can be used as an estimate for GI protein loss. In humans, several α1-proteinase deficiency states have been described, which thus far have not been identified in dogs or cats (Anonymus 1997). Thus, in dogs and cats it appears to be sufficient to measure fecal α1-PI concentration without determining its clearance, which dramatically improves the ease of using this test in dogs and cats versus humans.
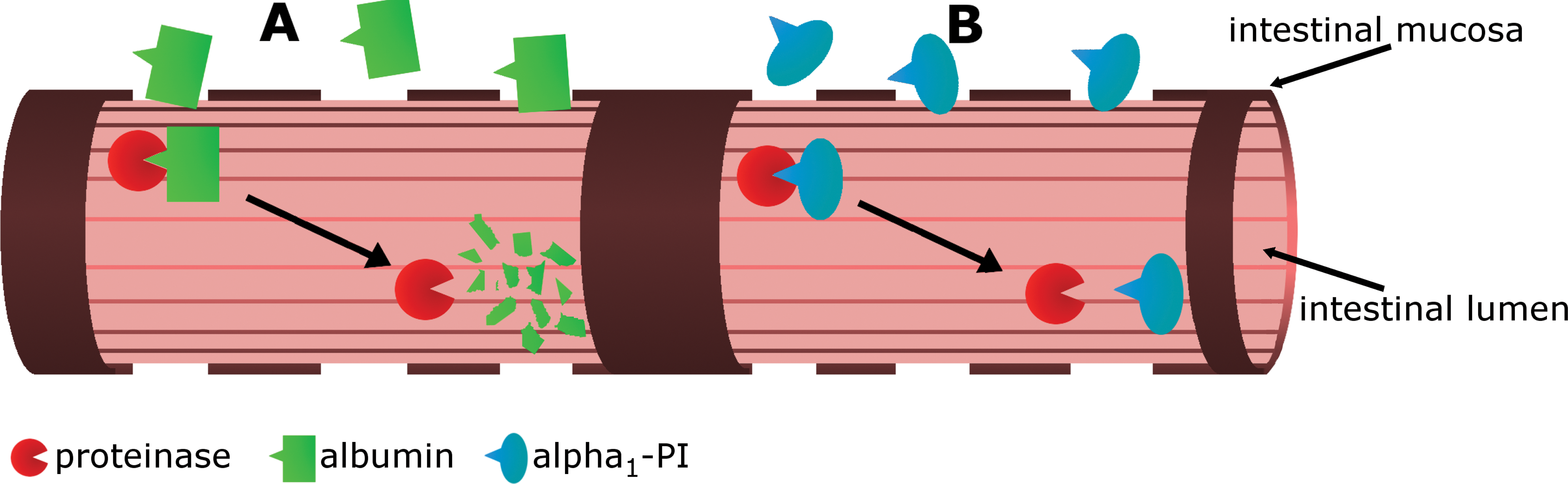
Fecal alpha1-proteinase inhibitor (α1-PI) concentration. (A) After being lost into the intestinal lumen, albumin and other plasma proteins are readily degraded by intestinal and bacterial proteinases. The resulting degradation products are no longer immunoreactive and thus cannot be detected and quantified by immunoassays. (B) α1-PI has a similar molecular size to albumin. Unlike albumin, it can resist degradation by proteinases present in the intestinal lumen. Intact α1-PI molecules can be detected in fecal samples by an immunoassay.
Clinically, fecal α1-PI concentration should be measured in dogs and cats with hypoalbuminemia that do not have clinical signs of GI disease and where an extragastrointestinal source of protein loss cannot be identified. Also, the measurement of fecal α1-PI concentration can be used to objectively monitor disease activity in dogs and cats with IBD.
Serum, Urine, or Fecal N-methylhistamine Concentrations
Mast cells may play an important role in intestinal inflammation in several species, though mast cells have not been consistently identified upon histopathological evaluation of biopsy specimens from dogs and cats with idiopathic IBD (Edwards et al. 1995; German, Hall, and Day 1999; Locher et al. 2001). Several special stains have been used to determine the presence of mast cells in the intestinal mucosa of dogs with idiopathic IBD. However, potentially more meaningful is the functional assessment of mast cell degranulation. One of the most important mediators released by mast cells during degranulation is histamine. Unfortunately, histamine is not very stable but is quickly metabolized by deamine oxidase to imidazole acetic acid or histamine N-methyltransferase to N-methylhistamine (NMH), which can be quantified in serum, urine, or even feces using gas chromatography–mass spectrometry (Ruaux et al. 2009). Fecal NMH concentrations have been shown to be significantly higher in dogs with protein-losing enteropathy (Berghoff, Suchodolski, and Steiner 2008; Berghoff et al. 2013).
Serum or Fecal S100 Protein Concentrations
S100 proteins are a group of calcium-binding proteins that are being expressed by a wide variety of cells (e.g., neutrophils and many cancer cells) and that have several functions. Calprotectin is a complex of S100A8 and S100A9 and is generally considered to be a marker of neutrophilic inflammation. Calprotectin concentrations have been shown to be increased in serum and feces from humans with IBD (Costa et al. 2005; Moum, Jahnsen, and Bernklev 2009). Recently, an immunoassay for the measurement of calprotectin concentrations in serum, feces, or other biological samples has been developed and analytically validated and initial studies have shown serum and fecal calprotectin concentrations to be significantly increased in dogs with chronic enteropathies (Heilmann, Suchodolski, and Steiner 2008; Heilmann et al. 2012). Similarly, S100A12 has also been shown to be significantly increased in both serum and feces from dogs with chronic enteropathies (Heilmann et al. 2011). However, further studies are needed to determine the clinical utility of these markers for diagnosing specific types of chronic enteropathies or monitoring disease progression and/or treatment response in dogs with such diseases. Also, initial studies would suggest that the assay developed and analytically validated for use in dogs can also be used for biological samples from cats, but further studies are required to confirm these findings.
Marker Assays to Assess the Exocrine Pancreas
Serum Lipase and Amylase Activities
Lipase and amylase activities have been measured in serum and sometimes other biological fluids such as peritoneal fluid from humans and veterinary species for several decades. Several assays are available for the measurement of both enzymes and in general sensitivities and specificities for one assay cannot be directly applied to another assay. For example, most serum lipase assays utilize a diacylglycerol as a substrate, but other assays use a synthetic compound, resorufin, which is considered to be more specific for the lipolytic activity of pancreatic lipase (Vorderwülbecke, Kieslich, and Erdmann 1992). The most significant disadvantage for the measurement of both serum amylase and lipase activity is that cells from many different tissues synthesize and secrete amylases and lipases, and assays that measure serum activities of these enzymes cannot differentiate between enzymes originating from these different tissues, thus limiting specificity of this assay for the exocrine pancreas. For example, in humans and dogs, gastric lipase is a very important lipase, digesting approximately one-third of all fat consumed (Carriere et al. 1993). One study reported that serum lipase activity is not significantly decreased in dogs with EPI, a condition where more than 90% of exocrine pancreatic reserve have been lost (Steiner, Rutz, and Williams 2006). At the same time, sensitivity of serum lipase or amylase activity for the diagnosis of human, canine, and feline pancreatitis is generally considered to be very poor, thus limiting the overall diagnostic accuracy of these assays. Thus, in the author’s opinion, measurement of serum amylase and/or lipase should be limited to species where better assays to assess pancreatic inflammation are not available.
Serum PLI Concentration
As mentioned, many different tissues synthesize and secrete lipases. In contrast to catalytic assays for the measurement of lipase activity, use of immunoassays does allow for the specific measurement of lipase originated from the exocrine pancreas. Immunoassays for the measurement of serum concentrations of pancreatic lipase (Spec cPL® in dogs and Spec fPL® in cats) have been developed and validated (Huth et al. 2010; Steiner, Medinger, and Williams 1996; Steiner, Gumminger, and Williams 2001; Steiner and Williams 2000a). Serum pancreatic lipase immunoreactivity (PLI) has been shown to be highly specific for exocrine pancreatic function (Steiner 2008). Also, the sensitivity of different minimally invasive diagnostic tests was compared in dogs with pancreatitis. The sensitivity of serum trypsin-like immuoreactivity (TLI) concentration was below 35%, that of serum lipase activity less than 55%, and ultrasound 68%. In contrast, the sensitivity of serum canine pancreatic lipase immunoreactivity (cPLI) concentration for pancreatitis was above 80% (Steiner 2008). Clinical studies in cats have shown similar results.
Thus, serum PLI concentration is the most sensitive and specific diagnostic test for pancreatitis currently available in dogs and cats. Also, recently, patient-side tests for the semiquantitative assessment of PLI concentration, SNAP® cPL, and SNAP® fPL have been introduced. A negative SNAP test virtually rules out pancreatitis. A positive test result needs to be followed up by measurement of a Spec PL in the laboratory to confirm the diagnosis of pancreatitis and to obtain a quantitative result that will allow monitoring the progress of the patient.
Unfortunately, PLI assays are species specific and currently assays for rodents, which are frequently used for studies of experimental pancreatitis, are not available.
Serum TLI Concentration
Pancreatic acinar cells synthesize a wide variety of active digestive enzymes (e.g., amylase, lipase) but also inactive zymogens (e.g., trypsinogen, chymotrypsinogen, proelastase; Rinderknecht 1993). These enzymes and zymogens are being collected in so-called zymogen granules and are then being released by way of exocytosis onto the luminal side of pancreatic acinar cells and leave the pancreas down the pancreatic duct. However, a small amount of zymogen granules is being released into the vascular space, containing a small amount of trypsinogen that can then be measured by immunoassays (Steiner 2008). Since these assays detect trypsinogen, trypsin, and even some molecules of trypsin that have been captured by proteinase inhibitors, these assays are called TLI. In dogs and cats with EPI, serum TLI concentration is dramatically decreased (Steiner and Williams 2000b; Williams and Batt 1986). Also, pancreatic inflammation (i.e., pancreatitis) leads to an increased release of trypsinogen and trypsin into the vascular space, leading to increased serum TLI concentrations. While serum TLI concentration is considered rather specific for the exocrine pancreas in healthy individuals, it appears that TLI assays lack specificity in individuals with certain GI conditions. In humans with chronic enteropathies, increased serum TLI concentrations have been anecdotally reported. Some have suggested that this is due to synthesis of small amounts of trypsinogen by enterocytes. However, this has not been verified or described for other species.
It is also important to note that assays for the measurement of TLI are highly species-specific and thus specific assays have to be used for each species. Currently, TLI assays are widely available for dogs, cats, and humans.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
