Abstract
The development process of myelination varies between region and species. Fully myelinated fibers are required if mammalian neural circuits are to function normally. Histology samples at staggered time points throughout the study were examined at days 4, 5, 7, 8, 10, 14, 17, 24, 37, and 44. We suggest that the development of myelin in the juvenile rodent brain can be conveniently separated into 3 phases. Evaluation of myelin basic protein–stained sections of the areas of brain that contain the elements of the developing limbic system over the sensitive period from postnatal day (PND) 14 to 34 may provide an insight into possible toxicity that may lead to cognition and learning issues in adults. We will hope to develop this notion further in the future. The precise chronology of the development of the blood–brain barrier in rats has yet to be established; thus, there is potential for significant exposure of the juvenile brain to chemicals that do not cross the blood–brain barrier in the adult. Thus, it is suggested that evaluation of myelin development should probably be extended to all new chemical entities intended for pediatric use, and not just those that are intended for central nervous system use.
Introduction
Myelination is required if mammalian neural circuits are to function normally. The development process of myelination varies between region and species. In man, myelination begins in the last trimester and continues through to adolescence. In rats, myelination predominantly occurs postnatally (Wiggins 1982; Paus et al. 1990). Disruption of myelin has been documented as a result of malnutrition as well as a number of toxicants including anticonvulsants, ethanol, heavy metals, and cholesterol inhibitors (Rodier 1990; Herring and Konradi 2011).
As such, the process of myelination may be affected by the administration of substances during the course of juvenile toxicity studies. It has been documented that myelin formation in the brain begins at around postnatal day (PND) 10 in the rat, with the maximal rate of myelin accumulation occurring around PND 20; however, myelin accumulation does continue into adulthood, albeit at a decreasing rate (Doretto et al. 2011).
The process of myelination has similarities in all species, beginning in the spinal cord and proceeding rostrally. Within the cerebral lobes, myelin development occurs initially in the primary motor and sensory areas of the cerebral cortex and corpus callosum before spreading outward.
Although the early myelin development has been investigated with immunofluorescent techniques (Bjelke and Seiger 1989), this investigation covers a greater number of time points and uses formalin-fixed paraffin embedded tissue. The recognition and understanding of the normal, regional process of myelination of the brain in the juvenile rat is necessary for the pathologist to be able to correctly interpret treatment related toxicity. An awareness of how this relates to human development is essential with respect to risk assessment and predictions of possible clinical manifestations of changes to the juvenile rodent brain. This article describes the findings from a detailed investigation of the early regional myelination in the rat brain and, in particular, the early period of development (PND 4–10) which has been largely neglected up to now.
The histology samples used for this investigation were derived from a study designed to assess the toxicity of Vigabatrin and reported elsewhere (Rasmussen et al. Juvenile Studies in Rats Using Vigabatrin in preparation). The brain tissue obtained from that study was used for this further investigation of the regional development of myelin in juvenile rats, with the kind permission of H. Lundbeck A/S, Ottiliavej 3, DK-2500 Valby, Denmark.
Materials and Methods
Sufficient juvenile rats of the Crl:CD(SD) strain were cross fostered soon after birth onto dams such that no siblings were present in the same litter and were allocated to the study.
In order to satisfy other requirements of the regulatory study, the animals were divided into 3 separate subsets, with dosing of the subsets being staggered for ease of management. Subset 1 was treated from PND 4, subset 2 from PND 7, and subset 3 from PND 14. Each subset consisted of 25 animals of each sex, with 5 animals of each dose being sacrificed at the following time points: 8 hr (± 30 min), 1 day, 3 days, 10 days, and 30 days.
As a result of this design, samples were available at the following ages PND 4, 5, 7, 8, 10, 14, 17, 24, 37, and 44 (Table 1).
Disposition of animals on study.
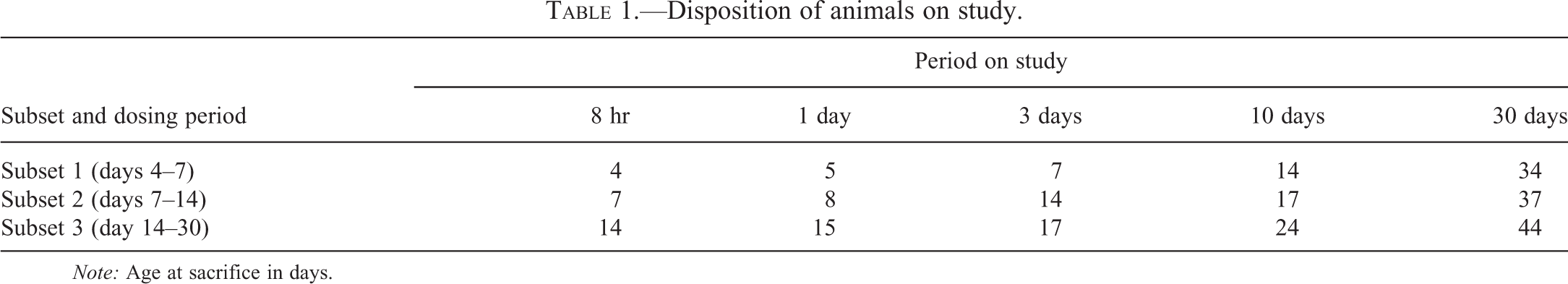
Note: Age at sacrifice in days.
Timing of myelin development in selected areas.
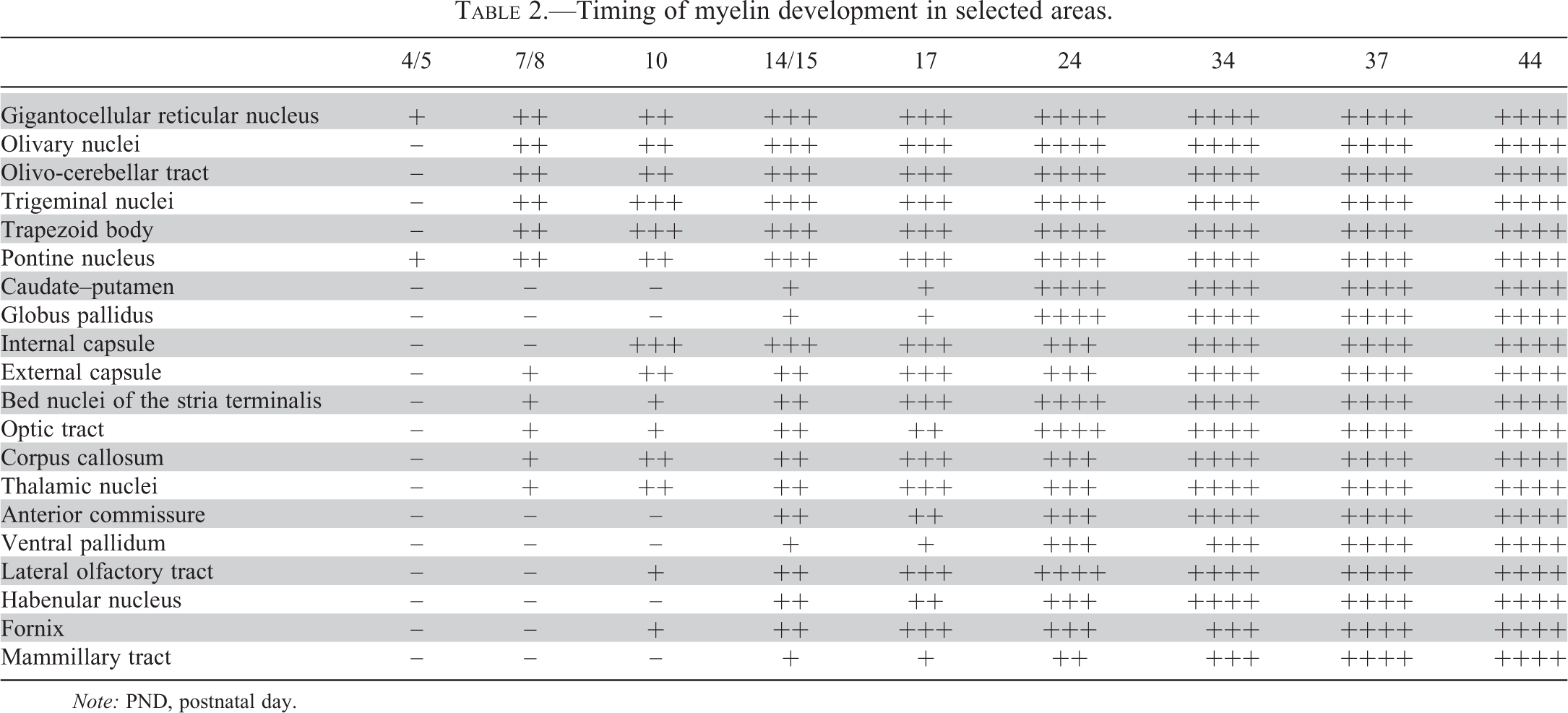
Note: PND, postnatal day.
The brains were weighed and then fixed in formalin, and embedded in wax block prior to sectioning at around 4 to 5 μm for histological examination. Three different stains were employed; hematoxylin and eosin, toluidine blue, and myelin basic protein C stains. Hematoxylin and eosin and toluidine blue stains were performed according to a standard protocol (Bancroft and Stevens 1996). As the stains for hematoxylin and eosin and toluidine blue proved unsuitable for assessment of myelin status, there will be no further discussion of these stains.
For the myelin basic protein stain, wax-embedded sections of brain were stained immunohistochemically on a Ventana Discovery XT automated staining platform. The primary antibody was Myelin Basic Protein Antibody (abcam ab40390), prepared at a 1:50 dilution using reaction buffer as the diluent. Following incubation, the secondary antibody (Swine Anti Rabbit Immunoglobulins, DAKO Z0196) was titrated using a 1:50 dilution with the reaction buffer as the diluent. Following further incubation, the sections were counterstained with hematoxylin.
The sections of the brain were prepared with reference to the standard methods (Rao et al. 2011). Six sections were taken for microscopic examination. The examination included, but was not limited to, the following areas:
The frontal, parietal, and piriform cortex, including the basal forebrain, septal nuclei, and anterior portion of the caudate–putamen; The frontal, parietal, and temporal cortex, including an anterior level of the thalamus/hypothalamus, amygdala, and either the fimbria/fornix or the anterior (dorsal) hippocampus; The parietal, temporal, and entorhinal cortex, including a posterior level of the thalamus and/or the anterior midbrain and posterior (dorsal plus ventral) hippocampus; The occipital lobes including the middle to posterior portion of the midbrain; The middle of the cerebellum with underlying pons; The posterior portion of the cerebellum with underlying medulla oblongata.
Animals were housed, maintained, and handled according to international standards of animal health and welfare in an AALAC (The Association and Accreditation of Laboratory Animal Care International)-accredited facility, and all experimental procedures were approved by the ethics committee of Sequani Limited.
Results
Day 4
Although the majority of the brain is myelin-free, there was some early myelin development in the gigantocellular reticular nucleus and in the oral part of the pontine reticular nucleus (Figure 1A).
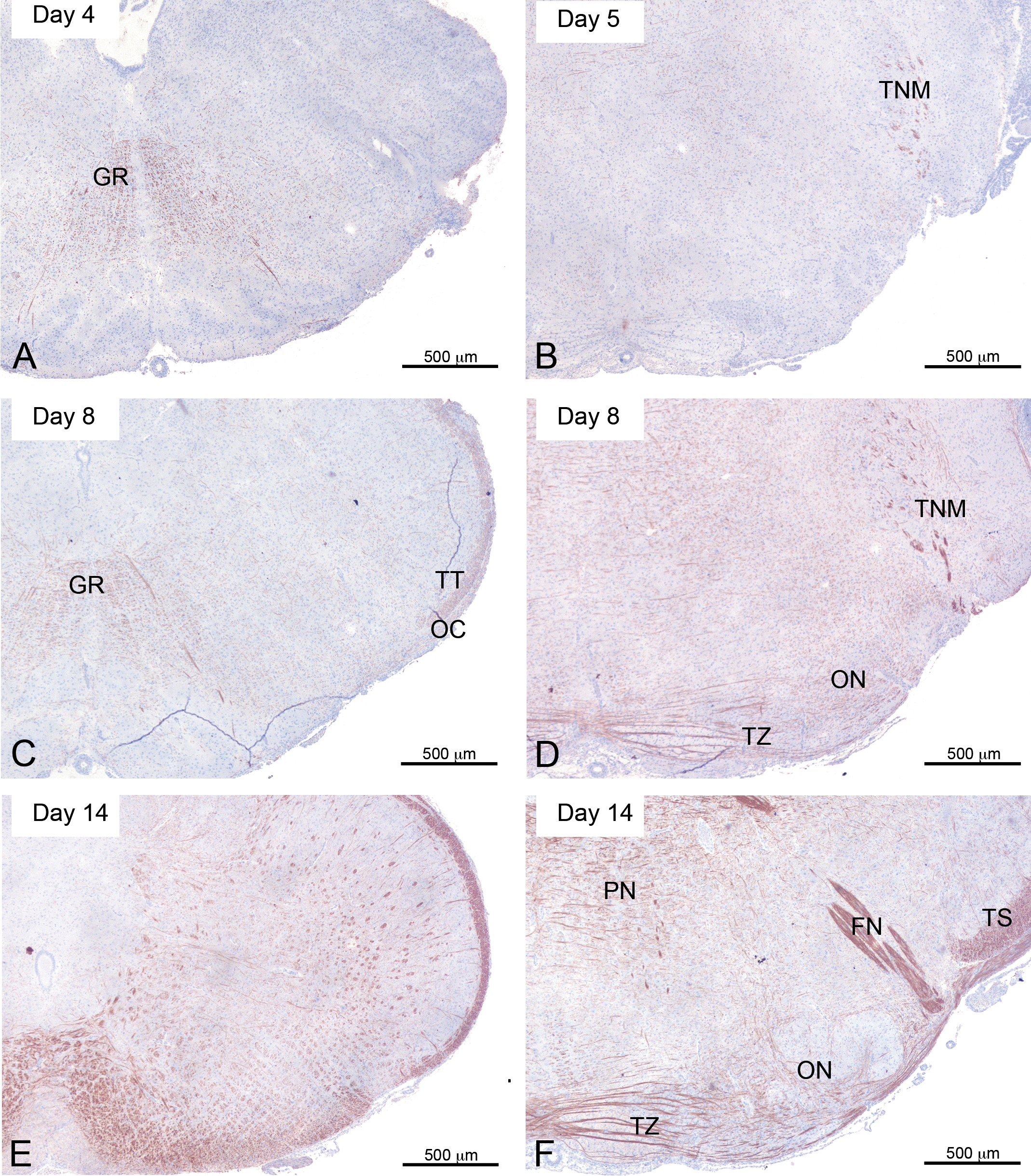
Development of myelination of the medulla oblongata and pons regions from day 4 to day 14. A, C, and E are the region of the Medulla oblongata at days 4, 8, and 14, respectively. The gigantocellular reticular nucleus already well myelinated at day 4 and developing trigeminal tract is clear at day 8. B, D, and F are the region of the pons at days 5, 8, and 14, respectively. Note: GR = gigantocellular reticular nucleus; ON = superior olivary nuclei; TT = spinal trigeminal tracts; OC = olivo-cerebellar tract; TN = spinal trigeminal nuclei; TNM = motor nuclei of the trigeminal nerve; TS = sensory root of the trigeminal nerve; TZ = trapezoid body; PN = pontine nucleus; FN = subcoeruleus nuclei, with heavily stained tracts of the facial nerve passing through.
Day 5
The area that seems to develop fairly rapidly over these 2 days is the trigeminal tract, which is another area that is likely to be important in suckling (Figure 1B). A few fibers are myelinated at PND 4, but myelination moves rostrally along the tract quickly and by PND 5 this area is clearly quite well myelinated.
Day 7
By PND 7, there were the first indications of myelinated fibers in the midbrain and forebrain. At this stage, there were traces of myelin formation, mostly in the external capsule of the corpus callosum, but also in the mesencephalon. The staining was quite sparse and no region was obviously myelinating at this stage. In the mid-cerebellar region and the medulla oblongata, the areas of the trigeminal tract and gigantocellular reticular nucleus were well myelinated, as was the intermediate reticular nucleus. This nucleus is the source of the neurons that provide the inspiratory drive to hypoglossal motoneurons (Chamberlin et al. 2002).
Day 8
In the midbrain and the forebrain, there was a slow incremental increase in myelin in the corpus callosum. The start of the myelination process in the thalamus, which was becoming evident by this time point, was characterized by light myelin basic protein staining in the thalamic nuclei.
There appeared to be rapid development of the mesencephalon between PND 7 and 8, where the presence of myelin in the mesencephalon reticular formation was now clear, in contrast to the sparse myelin content present earlier.
The reticular formation of the mesencephalon is a region in the brainstem that is involved in multiple tasks such as regulating the sleep–wake cycle and filtering incoming stimuli and, perhaps importantly, is part of a network that allows the space–time coordination of target-selecting rapid eye movements, clearly an area that will ideally be fully developed in time for eye-opening on PND 11.
There were also the first indications of myelin in the cerebellum. With increasing myelination, nonmyelinated areas were now beginning to stand out from the background, with the central gray area in particular now becoming apparent due to the lack of myelin in this region.
Another area that becomes significantly myelinated at this time is the region concerned with auditory perception: the trapezoid and the olivary nuclei (Figure 1D). This collection of nuclei functions in multiple aspects of hearing and is an area that displays a significant interspecies variation, being quite large in rodents and less so in primates and man (Kulesza, Spirou, and Berrebi 2003). By this age, the trigeminal tract appeared to be fully myelinated.
Day 10
The amount of myelin in the corpus callosum was slowly increasing, and the lateral olfactory tract had become relatively well myelinated in just 2 days. There were also clear signs of myelin formation in the caudate–putamen and the ventral hippocampal commissure. The presence of myelin in the thalamic nuclei had intensified from the earlier time points (Figure 2A and D).
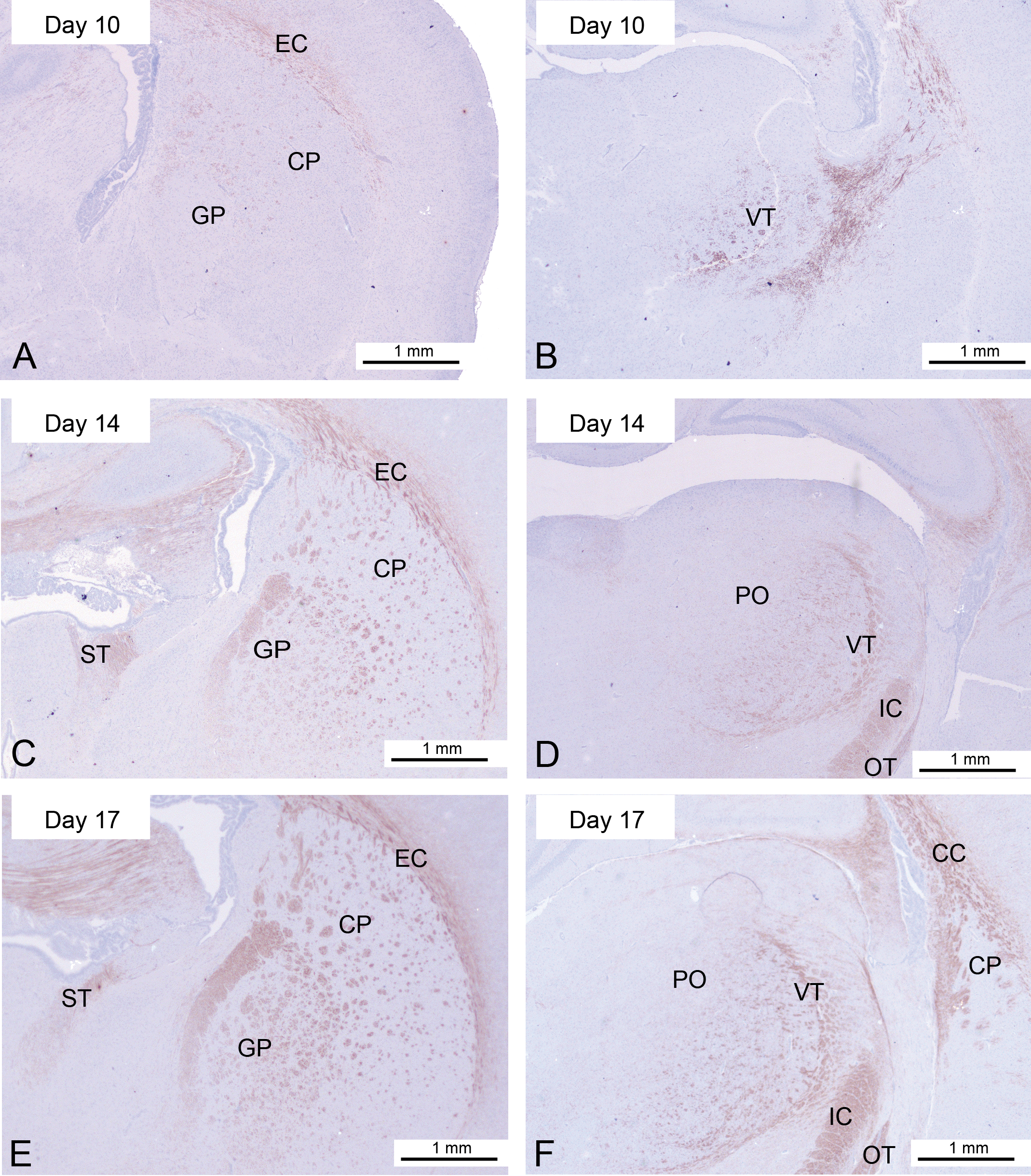
Myelination from days 10, 14, and 17 of parts of the basal ganglia (A, C, and E) and the thalamic nuclei (B, D, and F). Note: CP = caudate putamen; GP = globus pallidus; IC = internal capsule; EC = external capsule; BN = bed nuclei; OT = optic tract; CC = corpus callosum; PO = postthalamic nuclei; VT = ventral thalamic nuclei; ST = basal nuclei of the stria terminalis.
With the exception of the central gray area, the pons was substantially myelinated, and the cerebellum was myelinated in all areas to some extent, with the 8th cerebellar lobule appearing to myelinate first, with the other lobules lagging somewhat.
Day 14/15
In the pons, the myelinated status of the trigeminal tracts and the facial nerve were close to the levels seen in the adult (Figure 1F). Although myelination of the facial nerve was not noted prior to PND 14, it seems inconceivable that this process in the facial nerve occurred in totality between PND 10 and 14. The area in which the facial nerve develops is relatively narrow and the failure to observe early stage development of this nerve is probably a result of inadequate sampling. Logically, one would expect the facial nerve to develop on a similar schedule to the trigeminal nerve.
Between PND 10 and 14, there was a rapid increase in the amount of myelin present in the forebrain and midbrain. The corpus callosum was well myelinated throughout as was the lateral olfactory tract. In addition, the anterior commissure and lateral septal nuclei were now clearly starting to myelinate. Both these areas have been related to the development of olfaction in the rat and are interestingly reported to be a region where a sexual dimorphism exists, with males having larger anterior commissures than females (Noonan et al. 1998). However, as far as could be determined from the relatively small number of animals examined, there appeared to be no developmental dimorphism, development occurring seemingly in parallel in both sexes.
The lateral septal nuclei occupy one of the most strategically important positions in the forebrain, connecting the structures of the limbic system with different sites of the brain stem. These nuclei not only participate in the regulation but also implement overall coordination and modulation of various visceral and somatic functions. They also have a chronoregulatory structure that is responsible for biorhythmologic organization (Zamorski, Myslitski, and Pishak 1998).
The ventral thalamic nuclei are now well nucleated, and for the first time myelin was appearing in the laterodorsal thalamic nuclei. The myelin was spreading into the subthalamic nuclei, although it was still quite sparse. The optic tracts were now well myelinated (Figure 2D and F).
Myelin staining in the pons had become more intense and indicated the spread of myelin to the remaining cerebellar lobes. In the medulla oblongata, myelination looked to be fairly complete. At this comparatively early stage, the presence of myelin was greatest in the areas of the trigeminal and facial nerve tracts and the trapezoidal nuclei (Figure 1F).
Day 17
Because of the extent of myelination in the midbrain, the internal capsule, globus pallidum, and caudate–putamen had now become prominent (Figure 2C and F). Another area in the midbrain that was clearly becoming increasingly myelinated at this stage was the habenular nuclei (Figure 3C).
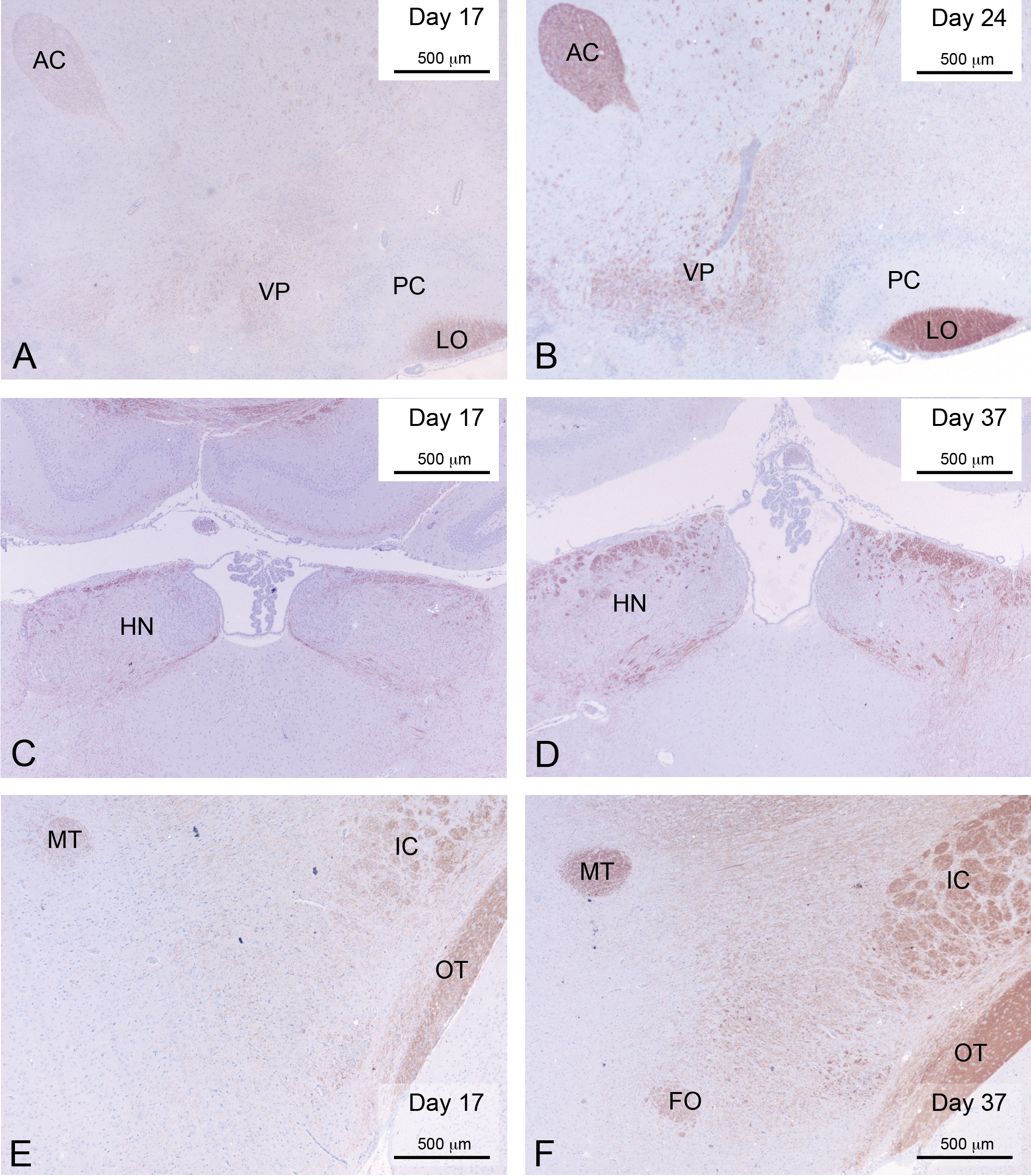
Myelination of the limbic system. Plates A and B show the development of the anterior commissure, the ventral pallidum, and lateral ophthalmic tract at days 17 and 24. Plates C and D show the habenular nucleus at days 17 and 37. Plates E and F show the mammillary tract and fornix of the hypothalamus at days 17 and 37. Note: AC = anterior commissure; VP = ventral pallidum; LO = lateral olfactory tract; CP = caudate putamen; PC = piriform cortex; HN = habenular nucleus; MT = mammillary tract; F = fornix of the hypothalamus; IC = internal capsule; OT = optic tract.
In the pons and medulla oblongata, all the areas that are myelinated in the adult were now myelinated to a greater or lesser extent. Between PND 17 and adulthood, the intensity of myelin staining increased, suggesting a maturation of the process, probably involving a thickening of the myelin sheaths.
Day 24
By PND 24 of age, almost all areas that are myelinated in the adult rat are now fully myelinated. There are, however, a few late developing areas that are only now beginning to myelinate.
Two areas that are only sparsely myelinated at this stage are the posterior tracts of the fornix as they traverse the hypothalamus and the mammillothalamic tract. In fact, neither of these areas appears to be fully myelinated before about PND 44 (Figure 3E and F).
Day 37–44
The continued development of the limbic system is the main feature of development during this phase.
The last 2 areas to develop appear to be the posterior tracts of the fornix as it passes through the hypothalamus, which looks to myelinate a little earlier than the mammillothalamic tract. The fornix is a C-shaped bundle of axons in the brain and carries signals from the hippocampus to the hypothalamus. It is a difficult structure to imagine from the 2 dimensional information that exists in a histological preparation. The fibers begin as the fimbria of the hippocampus on each side of the brain and separate into 2 tracts called the crus of the fornix. The bundles of fibers come together in the midline of the brain, forming the body of the fornix. The body of the fornix travels anteriorly and divides again near the anterior commissure. The posterior fibers continue through the hypothalamus to the mammillary bodies, then to the anterior nuclei of thalamus, which maps to cingulate cortex. The anterior fibers end at the septal nuclei and nucleus accumbens of each half of the brain. We were not able to adequately characterize the myelination of the fornix with the sections that were available. Given the complexity of this structure, it would probably be necessary to do step serial sections through the midbrain to assess this properly. However, there was a strong impression that the fimbria myelinated early and then the myelination moved back through the crus and through the hypothalamus until the mammillothalamic tract became the last part to become myelinated.
Discussion
Our current state of knowledge suggests that myelination of the hindbrain occurs first and thereafter the process moves rostrally (Bjelke and Seiger 1989). We would suggest that it is not that myelination just moves forward but that areas develop as required and there is evidence to support the idea that there are not 1 or 2 centers for the start of myelination, rather that myelination occurs in response to a functional demand, with the areas related to early nursing, such as suckling reflex, developing first (birth to PND 10), followed by other motor and sensory regions (PND 10–24), and finally learning areas (PND 17–37). It may well be that evolution has ensured that these areas are strategically positioned to permit efficient myelination of the juvenile brain, but it would seem that an assessment of myelination as a purely anatomical driven event may be oversimplistic.
As noted in the introduction, it is widely accepted that myelination of the rat brain begins around PND 10. Our investigation suggests that there is, in fact, some substantial deposition of myelin in some areas of the pons and medulla oblongata between PND 4 and 8, although there is comparatively little myelin in the midbrain and forebrain in the early days of life.
Specifically, there is clearly some early myelin development in the reticular formation, particularly the gigantocellular reticular nucleus and in the oral part of the pontine reticular nucleus, followed fairly rapidly by myelination of the trigeminal tract. At a slightly later stage, the intermediate reticular nucleus develops myelin. The gigantocellular nucleus excites the hypoglossal nucleus. This nucleus is the source of the neurons that provide the inspiratory drive to hypoglossal motoneurons (Yang, Chan, and Chan 1995). These are areas that are likely to be instrumental in successful suckling reflexes and, as suckling is the prime function of the neonatal rat, areas that may be associated with this process are of a high priority and it seems entirely logical that these areas are the first to myelinate. The oral part of the pontine nucleus is involved with sleep patterns and is a less obvious candidate for early myelination (Watson, Lydic, and Baghdoyan 2007).
At this early stage of life, the other areas that appear to be developing rapidly are those sensory areas associated with sight and sound, although interestingly, olfactory regions seem to lag a little, going through a rapid period of development around PND 14 to 15.
Clearly, these sensory areas are well developed in time for eye opening on PND 11. It may therefore be possible to postulate that hearing also becomes more acute at around this time. Many of the tracts associated with optic and auditory perception are almost fully myelinated by PND 15.
Although not an area that is subject to myelination, it is of interest to note the data reported for layers 2 and 3 of the piriform cortex. These are areas that receive input from the whiskers and are particularly receptive to input between PND 14 to 20 (Stern, Maravall, and Svoboda 2001). Layers 2 and 3 are very plastic areas and it has been found that trimming all contralateral whiskers from 9-day-old rats disrupts the development of these areas (Maravall, Stern, and Svoboda 2004). Again there is a sound logic to the timing of this development as this period immediately follows the beginning of active whisker exploration (Welker 1964).
PND 10 to 17 is the period in which the development of the basal ganglia and thalamic nuclei appear to dominate. Over this period, the corpus callosum, cerebellum, thalamic and subthalamic nuclei, caudate–putamen, and globus pallidum seem to be myelinating in concert. Also becoming myelinated at this time are the internal and external capsules and the bed nuclei of the stria terminalis (Figure 2). Although this is an area that has cognitive and sensory functions, given the young rat is now becoming ambulatory, it would seem likely that the important motor and learning functions of the area are the driving force behind the development of this region over this time period. Toward the latter stage of the 2nd week of age, the development of the bed nuclei of the stria terminalis is quite dramatic. Interestingly, this region has been connected with anxiety in response to threat monitoring (Somerville, Whalen, and Kelley 2010) and is also thought to promote behavioral inhibition in response to unfamiliar individuals (Fox et al. 2010). It seems sensible that as the young animal moves further away from the parental care zone it needs to recognize what may represent potentially dangerous situations. At this stage, the olfactory tract is well myelinated. This is around the same time as the layers of the pyriform cortex begin to develop. The comparatively late development of these areas that receive input from the whiskers and of the olfactory regions would seem to be consistent with the end of the weaning period, at around PND 21, and the beginning of active whisker exploration and search for food.
The components of the limbic system appear to be the last parts of the white matter to myelinate. They consist of a complex bilateral set of nuclei under the cerebrum. These include the hippocampus, amygdalae, anterior thalamic nuclei, septum, limbic cortex, and fornix. Together they seem to support a variety of functions including emotion, behavior, motivation, long-term memory, and olfaction.
With respect to the limbic system, the first parts to myelinate are the anterior commisure and lateral septal nuclei, both of which start to develop myelin at around PND 14. Shortly after this, the habenular nuclei and ventral pallidum begin to myelinate. The habenular nuclei have been shown to be involved in many functions, including pain processing, reproductive behavior, nutrition, sleep–wake cycles, stress responses, and learning (Andres, During, and Veh 1999). Recently, a further function of the lateral habenula with respect to reward processing has been elucidated, in particular with regard to encoding negative feedback or negative rewards (Matsumoto and Hikosaka 2007). If one imagines that at this stage of development the young rodent may be taking its first steps in investigation of the immediate environment, then it is tempting to imagine the logic of an association between the development of areas of the brain that are involved with learning and the stage of development that is associated with a sudden increase in the amount of external stimuli that are being experienced by animals of this age.
After PND 24, the continued development of the limbic system is the main feature, all the other areas of the white matter appearing to be well myelinated by this age. The limbic system seems to be fully developed by PND 37, with the last 2 areas to develop being the fornix of the hypothalamus and the mammillothalamic tract, with the fornix developing slightly in advance of the mammillothalamic tract. The contribution of the mammillary region to several classes of learning and memory has been the subject of a fair degree of research over the years. There is considerable evidence that lesions of this region of the brain impair performance of tasks that require memory for locations that an animal has visited, and learning tasks that require the animal to form an association between a place and a stimulus embedded within it. It would seem that the mammillary region may play a selective role in certain types of spatial learning and memory (Sziklas and Petrides 1998; Vann and Aggleton 2004).
Again there is logic to the memory and learning areas being the last to develop as the adolescent rat begins to forage further afield and therefore there is an increased requirement for spatial recognition and learning that is not necessary earlier in life.
In terms of toxic mechanisms that may potentially affect myelination, the oligodendrocyte itself is clearly a likely target. These cells are among the most vulnerable cells in the CNS and are particularly susceptible to oxidative damage, mitochondrial toxicity, and excitatory damage. It has been estimated that during the peak of the myelination process, an oligodendrocyte can elaborate about 3 times its weight in membrane per day, and the mature oligodendrocyte may support membrane up to 100 times the weight of its cell body (McTigue and Tripathi 2008; Mi et al. 2005; Bradl and Lassman 2010). It is self-evident that cells with this level of metabolic activity may be vulnerable at several levels, as high metabolic rates necessitate consumption of large amounts of oxygen and ATP.
One outstanding question that remains largely unanswered is the ability of oligodendrocytes to “catch up” if the toxicant is withdrawn. It has been established that oligodendrocytes in the young animal have only a brief period of time for myelination and are relatively incapable of myelinating once they are mature (Watkins et al. 2008). Moreover, the ensheathment of multiple axons by a single oligodendrocyte is a highly coordinated event: oligodendrocytes do not ensheath different axons sequentially at different time points, but the process is completed within a brief time window. In the rat, this is typically within 12 to 18 hr (Watkins et al. 2008). Neither do oligodendrocytes randomly wrap any neuronal processes, but preferentially select axons with diameters above 0.2 μm (Simons and Trajkovic 2006).
The process of myelination is well regulated and the onset of CNS myelination in normal development would appear to be determined by the degree of neuronal differentiation and not by the timing of any intrinsic hard-wired mechanism (Brinkmann et al. 2008). One essential signal for the onset of myelination seems to be provided by the electrical activity of neurons (Groves et al. 1993). This perhaps suggests a mechanism of toxicity related to what appears to be a common effect of many anticonvulsants. As their activity is predicated on dampening of adverse neuronal activity, it seems probable that concomitant dampening of normal neuronal activity is likely to remove some of the stimulation necessary for normal myelination.
This putative mechanism has potentially predictable consequences that could manifest as effects of toxicants in the young rat. Assuming that poorly myelinated areas will perform less effectively than normally myelinated areas, one could perhaps hypothesize that effects resulting in reduced deposition of myelin in the 1st week of life may adversely affect the suckling capability of the neonatal rat. Under these circumstances, the animal is unlikely to thrive and in all probability would not survive. Although a serious consequence for a rodent, this may be of limited relevance to man as the comparable areas are probably well myelinated at birth in man. Specific data on myelination in human infant brains do not appear to be available. However, neuroendocrine development takes place in utero in man (complete by 7 months in utero) but is postnatal in rats. Furthermore, the rat brain is not anatomically comparable to the human neonate until PND 10 of age (Romijn, Hofman, and Gramsbergen 1991; Downes and Burns 2008) and comparisons of postnatal CNS development, based on functional measures, also indicate that 10-day-old rats have a similar level of development to human infants at birth (Wood, Beyer, and Cappon 2003).
Effects on myelination in slightly older rats (1–2 weeks of age) constitute what we consider to be the second phase of myelination and is likely to affect motor activity and special senses, changes for which are routinely tested in toxicity studies in juvenile animals; although the shortcomings of the functional observational battery suggest that this may be insufficiently sensitive for all but the most marked of toxic changes on the brain. Adverse effects on the deposition of myelin in the limbic systems would be likely to affect learning and cognitive processes. Effects on basic learning and memory function can be easily investigated in rats and other animals, but more subtle effects on cognitive processes are more difficult to assess in animals.
In summary, the process of myelination in the rodent brain can be conveniently separated into 3 phases. Any adverse effect on a specific phase is likely to be reflected in abnormal function of the system under development. Changes to the process of myelination in the early days of rodent life are probably not particularly relevant to man, and given the concern over the effects of early life drug intervention on cognition and learning, and the lack of current testing paradigms for many such changes, it may suggest that particular concern should be paid to toxicity that results in aberrant myelin status of the limbic system. This may suggest that in toxicity studies conducted in juvenile rats, myelin basic protein stained sections should be routinely evaluated for the areas of brain that contain the elements of the developing limbic system over the sensitive period from PND 14 to 34. The anterior commissure and habenular nuclei may represent better opportunities for study over this time period as the tracts of the fornix in the hypothalamus and the mammillothalamic tract only fully develop toward the end of this window. We hope to develop this notion further in the future.
In addition, as there is some doubt as to the chronology of the development of the blood brain barrier, there is some logic for the possibility of extending the investigation of disturbances to the myelination process over the period PND 14 to 34 for all new chemical entities intended for pediatric use, and not just those that are intended for CNS use. There is also a lack of knowledge regarding the plasticity of the young brain and the extent to which an effect on myelination in early life may be recoverable by mature oligodendrocytes. Clearly further work is required to clarify the effects of environment and interventions on the development of the juvenile brain so that we may be better positioned to perform an adequate risk assessment of drugs in use for children.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
