Abstract
Transgenic organisms that express fluorescent proteins are used frequently for in vivo visualization of proteins and cells. The phenotype of a transgenic medaka (Oryzias latipes) strain that expresses a red fluorescent protein (RFP) in hepatocytes was characterized using light and fluorescence microscopy, immunohistochemistry, and transmission electron microscopy (TEM). Expression of RFP was first detected by confocal fluorescence microscopy in the location of the liver bud of live medaka embryos at 60 hr postfertilization (developmental stage 27). Subsequently, RFP signal was observed exclusively in hepatocytes throughout life using fluorescence microscopy in live fish and immunohistochemistry in formalin-fixed, paraffin-embedded liver sections. As the fish aged, prominent intracytoplasmic eosinophilic inclusions immunoreactive for RFP were observed by light microscopy and were correlated with membrane-bound electron dense inclusions on TEM. These results define the onset and location of RFP expression in the Tg(zf.L-fabp:DsRed) medaka and characterize a histologic phenotype that results from RFP expression in hepatocytes.
Introduction
The construction of transgenic organisms that express a fluorescent protein has revolutionized cell biology by enabling in vivo labeling of gene expression in cells and animals. To study the mechanism of hepatic response to injury, we obtained a transgenic Japanese medaka (Oryzias latipes), Tg(zf.L-fabp:DsRed) that expresses a red fluorescent protein (RFP) specifically in hepatocytes, because no specific immunohistochemical marker is available for fish hepatocytes (Van Wettere 2012). This transgenic medaka expresses an RFP under the regulatory control of the 2.8-kb fragment of the zebrafish liver fatty acid binding protein (L-FABP) promoter. FABPs are small cytosolic lipid chaperone proteins (∼15 kDa) that are expressed in most tissues, and they are involved in intracellular fatty acid trafficking. Nine FABP genes have been discovered in mammals and were named according to the tissues in which they were first discovered (Storch and Thumser 2010). Most FABPs are expressed in multiple tissues; however, the L-FABP is expressed exclusively in the liver (Denovan-Wright et al. 2000; Her et al. 2003; Storch and Thumser 2010). Information about the phenotype of this transgenic medaka has not been published, and the observation of large eosinophilic cytoplasmic inclusions in hepatocytes of adult medaka led us to investigate the specific spatial and temporal expression of RFP in the Tg(zf.L-fabp:DsRed) medaka fish line.
Materials and Method
Medaka Culture
The founder Tg(zf.L-fabp:DsRed) medaka were obtained from Drs. Zeng Zhiqiang and Hong Yunhan, Department of Biological Sciences, National University of Singapore. This stable transgenic line showed standard Mendelian inheritance from the F2 generation onward, and offspring were screened by observation of larvae for RFP expression in the liver using a fluorescent stereomicroscope (Leica MZ16F, Leica Microsystems, Heerbrugg, Switzerland). The Tg(zf.L-fabp:DsRed) medaka line was housed under recirculating freshwater aquaculture conditions, with charcoal filtration and UV treatment used in each passage at the Department of Molecular and Environmental Toxicology, North Carolina State University (NCSU), Raleigh, NC. Water temperature and pH were monitored daily and maintained at 26 ± 2°C and 7 ± 0.5, respectively. The light–dark cycle was 16 hr light, 8 hr dark. Dry food (Otohime B1, Reed Mariculture, Campbell, CA) was fed 4 times per day through automated feeders, and newly hatched Artemia nauplii were fed once daily.
In the culture conditions described above, medaka spawned daily and embryonated eggs (10–30 per mass) were separated from each other by gentle rolling on a moistened paper towel surface to disrupt the attachment filaments joining individual eggs. Groups of individual embryonated eggs were suspended in a 2% saline solution made from Instant Ocean (Instant Ocean® salts, Aquarium System Inc. Mentor, OH) in a 100× 15-mm petri dish and incubated at 26 ± 1°C with 16 hr light, 8 hr dark light–dark cycle (Iwamatsu 2004). After 5 days, the 2% saline solution was replaced with 1× embryo rearing medium (ERM) containing 17.1-mM NaCl and, in micromolars, 272 CaCl2 2H2O, 402 KCl, and 661 MgSO4·7H2O through hatching (usually at 9 days postfertilization [dpf]). After hatching, the larvae were fed once daily with dry food (Otohime B1) and maintained for up to 15 dpf in petri dishes containing 0.3 × ERM and then transferred to 130 × 70 × 120-mm plastic containers filled with water from the recirculating freshwater aquaculture system. At 60 dpf, the fish were transferred to 40 L tanks and maintained under recirculating freshwater aquaculture conditions for the remaining grow out period. The ERM or culture system water in the petri dish and in the container that held the eggs/larvae was renewed every 2 days. Once the larvae were transferred to 40 L tanks, they were fed as described previously for the adult line. Animal care and use were in conformity with protocols approved by the NCSU Institutional Animal Care and Use Committee in accordance with the National Academy of Sciences Guide for the Care and Use of Laboratory Animals.
Laser Scanning Confocal Fluorescence Microscopy
Embryos were dechorionated using standard procedure (Porazinski, Wang, and Furutani-Seiki 2010), anesthetized with a 10-μm tricainemethane sulfonate (MS-222, Argent Laboratories, Redmond, WA) solution, and immobilized in a 1% solution of low melt agarose (Apex Bioresearch product, Genesee Scientific, San Diego, CA) in ERM in depression well glass petri dishes (Glass bottom culture dishes, Mat Tek Corporation, Ashland, MA) and oriented in right lateral recumbency. Embryos were then imaged live with laser scanning confocal fluorescence microscopy (LSCM—Zeiss LSM 710 NLO 34 channel system, Carl Zeiss MicroImaging GmbH, Jena, Germany) with ZEN 2009 version 5.5 SP1 image acquisition software (Carl Zeiss), 561-nm laser, and Carl Zeiss C-apochromat 10×/0.3 DICI with different magnification levels. Images of the embryos were obtained at 55, 60, 67, 79, and 103 hr postfertilization (hpf).
Histology and Electron Microscopy
Embryos or larvae were collected at 2, 3, 4, 5, 6, 7, 12, 15, or 20 dpf, euthanized with an overdose of tricaine methanesulfonate (300 mg/L; MS-222, Argent Laboratories), examined under fluorescent microscopy for RFP expression (Leica MZ16F, Leica Microsystems), and fixed in 4% paraformaldehyde for 24 hr. The embryos were dechorionated using standard procedure and transferred to 70% ethanol for histopathology. At least 15 embryos or larvae of each age were examined by light microscopy. Additionally evaluated were whole adult fish and livers from approximately 3-, 6-, 12-, and 18-month-old control fish used in other unrelated experiments and from older broodstock medaka. Five fish in each age group were examined. Adult fish were euthanized with an overdose of tricaine methanesulfonate (300 mg/L) and the coelom was incised along the ventral midline to enhance fixative penetration. The fish were fixed whole in 4% paraformaldehyde for 24 hr, demineralized in 10% formic acid for 24 hr, and transferred to 70% ethanol for histopathology. The livers from 3 additional 6-month-old control fish were inspected macroscopically, harvested, and cut into 2 approximately equal-sized pieces. One piece was fixed in 4% paraformaldehyde for 24 hr and transferred to 70% ethanol for histopathology. The other piece was cut into 1 × 1-mm samples and fixed in 4F:1G fixative (4% formaldehyde and 1% glutaraldehyde buffered in monobasic sodium phosphate, pH 7.2–7.4) for transmission electron microscopy (TEM; McDowell and Trump 1976).
Paraformaldehyde-fixed livers, embryos, larvae, and whole fish were processed and embedded in paraffin according to routine histologic techniques. Sections of 5-µm thickness were mounted on glass slides, stained with hematoxylin and eosin (H&E), and examined by light microscopy. Randomly oriented sections were cut through the embryos, while median and paramedian sections were made through the larvae and older fish.
Liver samples fixed in 4F:1G fixative were dehydrated in alcohol, embedded in Spurr resin, and processed for TEM (Dykstra 1993). To determine whether structures of interest were in planar sections of the block face, semi-thin sections, 0.5 μm thick, stained with 1% toluidine blue in 1% sodium borate, were examined under a light microscope. Ultrathin sections, 90 nm thick, of appropriate blocks were stained with uranyl acetate and lead citrate and examined with a FEI/Philips EM 208S transmission electron microscope (Laboratory for Advanced Electron and Light Optical Methods, NCSU).
Immunohistochemistry
To assess RFP expression in the liver and other organs, immunohistochemistry for RFP was performed in at least 5 sections per embryo, larva, or adult fish stage examined. The RFP antibody was a rabbit polyclonal antibody that recognizes the full-length amino acid sequence of the mushroom polyp coral Discosoma RFP (Abcam Inc, Cambridge, MA. Catalogue # ab34771). Tissue sections were deparaffinized in xylene, rehydrated in a graded series of ethanol, and rinsed in distilled water. Antigen retrieval was performed by heating (99°C) the slides in a 10-mM sodium citrate solution at pH 6 for 10 min in a vegetable steamer (Oster 5712 food steamer, Maitland, FL). Following a cool down period of 10 min in the warm sodium citrate buffer, the slides were rinsed in 1× Tris-buffered saline with 0.05% Tween (TBST). Goat serum was applied for 20 min to prevent nonspecific binding of the secondary antibody. Tissues were incubated with the RFP antibody (1/500 dilution) for 30 min at room temperature. The slides were rinsed in 1 × TBST and then incubated with the secondary antibody for 20 min (BioGenex San Ramon, CA). After a wash in 1× TBST, tissue sections were then treated for 20 min with streptavidin alkaline phosphatase (BioGenex San Ramon, CA). Following a final wash in 1× TBST, development was achieved by treatment of tissue with VECTOR Red Alkaline Phosphatase Substrate Kit (Vector Lab, Burlingame, CA) for 1 to 5 min. Levamisole was added to the VECTOR Red Alkaline Phosphatase Substrate Kit to block endogenous alkaline phosphatase activity (Vector Lab, Burlingame, CA). After a wash in tap water, the slides were counterstained with Mayer’s hematoxylin for 20 to 40 sec and cover slipped. For negative controls, the primary antibody was omitted and rabbit nonimmune serum was applied. For positive controls, sections of liver from adult Tg(zf.L-fabp:DsRed) medaka were run at the same time.
Results
Tg(zf.L-fabp:DsRed) Medaka Liver Phenotype
Livers of the Tg(zf.L-fabp:DsRed) medaka had a distinct red color compared to the brown color of outbred orange red medaka (Figure 1A). The difference in coloration was most evident in 3-month-old and older fish. Except for the coloration, no significant macroscopic changes were noted in the examined embryos, larvae, or adult fish. No microscopic changes were observed in the 2- to 20-day-old embryos/larvae. In the 3- to 18-month-old fish, deeply eosinophilic globules ranging from 2 to 10 µm in diameter and eosinophilic acicular inclusions ranging from 2 × 3 µm to 5 × 20 µm were present in the cytoplasm of hepatocytes (Figure 1B and C). The amount of eosinophilic inclusions varied greatly among livers but inclusions were more frequent and numerous in livers of 12- and 18-month-old fish. Material with similar staining characteristics filled the cytoplasm of few to moderate numbers of scattered perisinusoidal macrophages (Figure 1B). The macrophages filled with this eosinophilic material were most abundant in the 12- and 18-month-old fish as well. Occasional and incidental small areas of spongiosis hepatis or cystic degeneration were observed in the 12- and 18-month-old medaka.
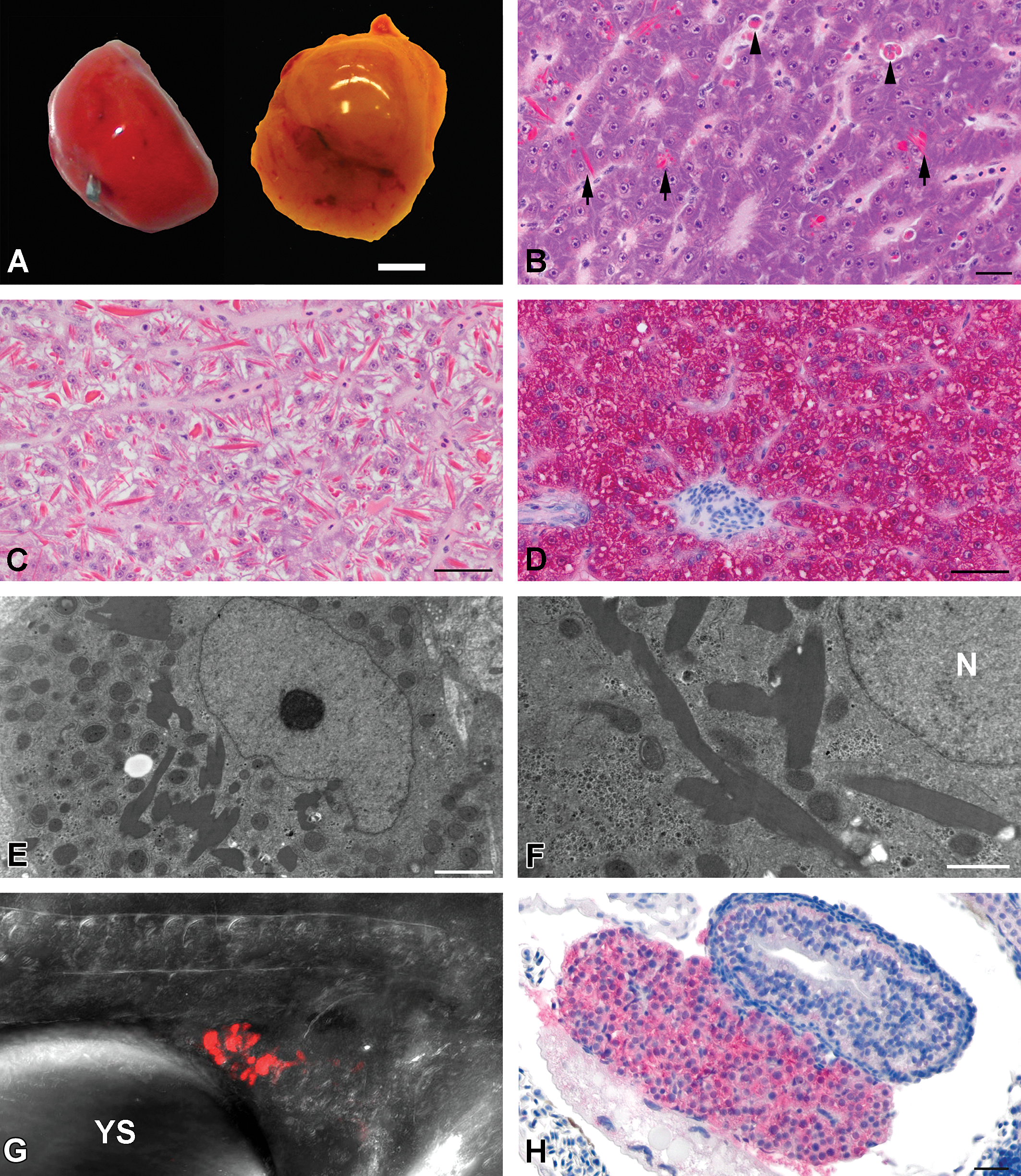
(A): Gross appearance of the liver of an adult Tg(zf.L-fabp:DsRed) medaka (left) and an adult orange red strain medaka (right). Note the red color of the transgenic fish liver compared to that of an outbred nontransgenic medaka fish strain. Bar = 1 mm. (B): Histology of an adult Tg(zf.L-fabp:DsRed) female medaka liver. Note the deeply eosinophilic globules and rhomboid inclusions in the cytoplasm of hepatocytes (arrow). Some macrophages contain similar deeply eosinophilic material (arrow head). Hematoxylin and eosin stain. Bar = 30 µm 60×. (C): Histology of an adult Tg(zf.L-fabp:DsRed) male medaka liver with pronounced accumulation of intracytoplasmic eosinophilic inclusions in hepatocytes. This level of RFP accumulation was mainly observed in 12- and 18-month-old fish. Hematoxylin and eosin stain.
In 12-, 15-, and 20-dpf larvae, and 3, 6, 12, and 18-month-old fish, all hepatocytes demonstrated diffuse cytoplasmic immunoreactivity for RFP (Figure 1D). Intracytoplasmic immunoreactivity for RFP was also observed in some macrophages in 3-, 6-, 12-, and 18-month-old fish; livers of older fish contained more immunoreactive macrophages. With the exception of macrophages, immunoreactivity for RFP was not observed in cells other than hepatocytes in multiple sagittal sections of whole embryos, larvae, and adult fish.
The intracytoplasmic inclusions in hepatocytes were further characterized by TEM. Hepatocytes contained intracytoplasmic membrane-bound inclusions composed of homogenous moderately electron-dense material (Figure 1E and 1F). The form and size were variable and ranged from less than 0.5 × 0.5-µm irregular globules up to 3 × 15-µm rhomboid or acicular inclusions. At high magnification, an approximately 5-nm wide membrane surrounding the electron dense inclusion was visible. Multifocally, similar electron dense material distended rough endoplasmic reticulum cisternae in hepatocytes. No other unusual ultrastructural features were observed in the hepatocytes. Electron-dense inclusions or other changes were not observed in biliary epithelial cells, including those lining bile preductules; nor were they observed in endothelial cells.
Onset of L-FABP–RFP Expression in Embryo
Dechorionated embryos were evaluated by LSCM to detect the onset of RFP expression in the developing liver. Red fluorescence was not detected in 55-hpf embryos (stage 26) but at 60-hpf (stage 27) red fluorescence appeared in the region of the liver bud (Figure 1G). The cell mass displaying red fluorescence became larger over time as observed at the 67-, 79-, and 103-hpf time points. Red fluorescence was restricted to the liver bud and was not observed in other tissues.
The onset of RFP expression was also evaluated using immunohistochemistry in 2- to 7-dpf embryos. At 4 dpf, a small mass of vacuolated cells resembling hepatocytes was already clearly recognizable in H&E stained sections but diffuse cytoplasmic RFP immunoreactivity was first observed in 6-day-old embryos (Figure 1H).
Discussion
Transgenic organisms expressing fluorescent proteins are frequently employed for in vivo labeling of temporal and spatial gene expression analysis and cell-tracking studies. Here, we report the phenotype of a transgenic medaka fish that expresses an RFP reporter driven by a 2.8-kb fragment of the zebrafish L-FABP promoter. Examination by light microscopy of adult Tg(zf.L-fabp:DsRed) medaka revealed eosinophilic inclusions in hepatocytes and led to investigation into the nature of these inclusions. In this study, we demonstrate temporal onset of expression and exclusive spatial expression of RFP in Tg(zf.L-fabp:DsRed) medaka hepatocytes using light and fluorescent microscopy, immunohistochemistry, and TEM.
Accumulations of progressively larger amounts of RFP over time were observed by light microscopy in some hepatocytes, especially in older fish. This observation may suggest a deficit in RFP intracellular trafficking due to impaired exportation from the rough endoplasmic reticulum (RER) or inadequate lysosomal degradation. The observation by TEM of electron-dense globular and acicular inclusions in hepatocytes correlated well with the light microscopy and RFP immunohistochemical findings. This electron-dense material was located in dilated rough endoplasmic reticulum cisternae and possibly in other organelles, although only accumulation in rough endoplasmic reticulum could be clearly identified. Correlation between the light microscopic and ultrastructural appearance with the specific RFP immunoreactivity in histologic sections provides strong indirect evidence that the material observed by TEM is RFP. However, immunogold labeling would be needed to confirm that this homogeneous electron-dense material is truly RFP. As noted above, some hepatic macrophages in older fish contained moderate amounts of RFP protein, which suggests they had phagocytized hepatocellular debris, likely from normal cell turn over. No apparent deleterious morphological effects or reproductive effects resulting from the RFP expression and accumulation in hepatocytes were observed in this study. However, specific reproductive and hepatocyte metabolic testing were not performed.
In medaka, the liver anlage is reported to develop at 2 dpf and achieves its adult phenotype by 16 dpf (Hinton et al. 2008; Iwamatsu 2004). The results of this study indicate that expression of L-FABP in medaka hepatocytes/hepatoblasts begins approximately 2.5 dpf (60 hpf—stage 27). Interestingly, although expression of RFP could be detected as early as 2.5 dpf by LSCM, RFP positive cells could only be detected by immunohistochemistry from 6 dpf. It is likely that the size of the hepatoblastic mass and the level of RFP protein expressed in hepatoblasts before 6 dpf are perhaps too low to be detected by immunohistochemistry. In previous work, in vivo imaging of the liver bud at 9 dpf revealed sinusoidal blood flow and fluorescent bile in the intrahepatic biliary system (Hardman, Kullman, and Hinton 2008; Hardman et al. 2008; Hinton et al. 2004). Together, these observations indicate that hepatocyte and biliary cells are functional during early liver development.
Given the restricted expression of RFP in hepatocytes, this transgenic medaka would be useful to identify hepatocytes in vivo or in tissue sections for studies involving organogenesis, response to injury, or carcinogenesis. Tg(zf.L-fabp:DsRed) medaka have been used to study cellular events associated with hepatic regeneration in medaka, and a similar double transgenic line Tg(lfabp:DsRed; elaA:EGFP) has been used in developmental biology and regeneration studies of the liver in zebrafish (Korzh et al. 2008; Li et al. 2012; Van Wettere et al. 2013). The lfabp:DsRed construct could also be used to develop transgenic lines with a cre-lox recombination system to perform fate mapping or targeted ablation studies.
Overall, the results of this study define the onset and location of RFP expression in the Tg(zf.L-fabp:DsRed) medaka fish line and characterize the histologic phenotype induced by the RFP expression in hepatocytes. This information will be useful in interpreting the hepatic morphological changes when this transgenic medaka line is used to study the mechanism of liver response to injury and hepatocarcinogenesis.
Footnotes
Acknowledgments
We are indebted to Dr. Zeng Zhiqiang and Dr. Hong Yunhan, Department of Biological Sciences, National University of Singapore, for providing the founder transgenic medaka for our colony. We also thank Gwijun Kwon for help with the medaka embryo collection and maintenance. We are grateful to Sandra Horton, Monica Matmeuller, Laura Shewmon, and the staff of the Histopathology Laboratory as well as Jeanette Shipley-Phillips from the Laboratory for Advanced Electron and Light Optical Methods at the NCSU-CVM for their expertise. We would like to acknowledge the invaluable help of Jeff Tucker with the LSCM at the National Institute of Environmental Health Sciences.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
