Abstract
Renal tubular inclusion bodies are rarely associated with drug administration. The authors describe the finding of renal cortical tubular intranuclear and intracytoplasmic inclusion bodies associated with the oral administration of a norepinephrine/serotonin reuptake inhibitor (NSRI) test article in Sprague-Dawley (SD) rats. Rats were given an NSRI daily for 4 weeks, and kidney histopathologic, ultrastructural pathology, and immunohistochemical examinations were performed. Round eosinophilic intranuclear inclusion bodies were observed histologically in the tubular epithelial cells of the renal cortex in male and female SD rats given the NSRI compound. No evidence of degeneration or necrosis was noted in the inclusion-containing renal cells. By ultrastructural pathology, inclusion bodies consisted of finely granular, amorphous, and uniformly stained nonmembrane-bound material. By immunohistochemistry, inclusion bodies stained positive for
Keywords
Introduction
The test article in this toxicology study is a norepinephreine (NE)/serotonin (5-HT) reuptake inhibitor (NSRI) that was developed and investigated for the treatment of fibromyalgia (FM). NSRI increases both NE and 5-HT levels via binding to presynaptic reuptake transporters in the central nervous system (CNS). FM is a chronic pain disorder that is characterized by widespread musculoskeletal pain without demonstrable structural abnormalities or evidence-based pathophysiological mechanisms (Lawson 2002). Characteristic clinical features of FM include headaches, migraines, fatigue, depression, and morning stiffness (Lawson 2002). The exact etiology of FM is currently unknown. Among the several hypotheses proposed regarding FM pathogenesis, abnormal physiological processing of nociceptive neurons resulting in increased pain perception has also been proposed (Nijs and Van Houdenhove 2009). In fact, alterations in central pain processing have been demonstrated in FM patients, and therefore have been suggested to play a role in the etiopathogenesis of FM (Gracely et al. 2002; Staud et al. 2001).
The primary neuronal transmitters involved in pain perception and processing are 5-HT and NE. Therefore, 5-HT and NE have been implicated in the mediation of analgesic mechanisms via the descending inhibitory pain pathways in the CNS. It is proposed that dysfunction of 5-HT and NE pathways is a potential mechanism for the chronic pain in FM patients. The theory is that tricyclic antidepressant’s pharmacologic efficacy in FM is related to inhibiting reuptake of 5-HT and NE into the neuronal terminal and thereby reducing pain perception and processing in patients with FM (Lawson 2002). Indeed, pharmacologic studies with antidepressants showed that inhibition of both the 5-HT and NE reuptake transporters were more effective in treating FM than inhibition of either transporter alone (Goldenberg et al. 1996). Additionally, some clinical studies showed that using dual 5-HT and NE reuptake inhibitors, duloxetine and milnacipran, demonstrated efficacy in patients with FM, providing pain relief and psychotherapeutic benefits (Arnold et al. 2004; Kyle, Dugan, and Testerman 2010). Thus, NSRI inhibitor was developed by Pfizer Worldwide Research and Development to treat FM.
The physiologic function of
To assess the preclinical safety profile of the NSRI test article, a 4-week toxicology study was conducted in Sprague-Dawley (SD) male and female rats. An unexpected finding at the end of the study was the presence of inclusion bodies in the renal tubular epithelial cells without tissue damage or inflammation as assessed by light microscopic examination. Inclusion bodies in the renal tubular epithelium are typically seen in some viral infections or lead poisoning, but rarely associated with drug administration. Similar inclusions have been reported in the literature in male and female Fisher 344 rats given propiverine (Dietrich et al. 2008). Propiverine is an anti-cholinergic drug used to treat neurogenic detrusor overactivity and overactive bladder (Alloussi et al. 2010). To the best of our knowledge, this article is the first description of intranuclear and intracytoplasmic inclusion bodies in renal tubular epithelial cells associated with NSRI test article administration in SD rats. The relevance of such kidney inclusion bodies to human safety risk assessment is discussed.
Materials and Methods
Animals
Male and female Sprague-Dawley [(Crl:CD(SD)] rats obtained from Charles River Laboratories were used for this 4-week toxicology study. Animals were 7 to 8 weeks old and weighed 150 to 275 g at the start of dosing. Clinically acceptable animals were assigned to dose groups using a computer-assisted randomization procedure. Animals were individually housed in stainless steel cages. Standard procedures and conditions were applied for animal care, feeding, and maintenance of room, caging, and environment. Animals were fed Certified Rodent Diet ad libitum, and water was supplied ad libitum via an automatic watering system. Animals were fasted overnight prior to blood collection for clinical laboratory measurements at scheduled necropsy. This study was conducted in accordance with current guidelines for animal welfare (National Research Council, 1996). The procedures used in this study were reviewed and approved by the Institutional Animal Care and Use Committee.
Experimental Design
Toxicity study animals (10 rats/sex/group) and toxicokinetics (TK) study animals (4/sex/group) received the test article, a dual inhibitor of NE and 5-HT reuptake, NSRI, which was synthesized by Pfizer Worldwide Research and Development. Dosing suspensions were prepared approximately weekly in aqueous 1% methylcellulose at doses of 3, 10, or 30 mg/kg/day. Animals were dosed once daily by oral gavage for 4 weeks. Control animals received the vehicle alone.
Experimental Procedures
Clinical signs, ophthalmic examination, body weight and food consumption
Animals were observed twice daily for clinical signs. Ophthalmic examination was conducted once during the predosing and once during the last 7 days of dosing. Body weights were determined once prior to dosing and weekly thereafter. Food consumption was determined weekly and at scheduled termination.
Clinical pathology
Hematological and clinical chemistry parameters were evaluated on day 28 at scheduled termination in animals designated for main study toxicologic assessment. Blood samples were analyzed for hematology parameters (absolute and differential blood counts). Serum was analyzed for clinical chemistry parameters: glucose, total cholesterol, total bilirubin, total protein, albumin, globulin, albumin/globulin ratio, phosphorus, calcium, chloride, sodium, potassium, creatinine, aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, and urea nitrogen.
Toxicokinetics (TK)
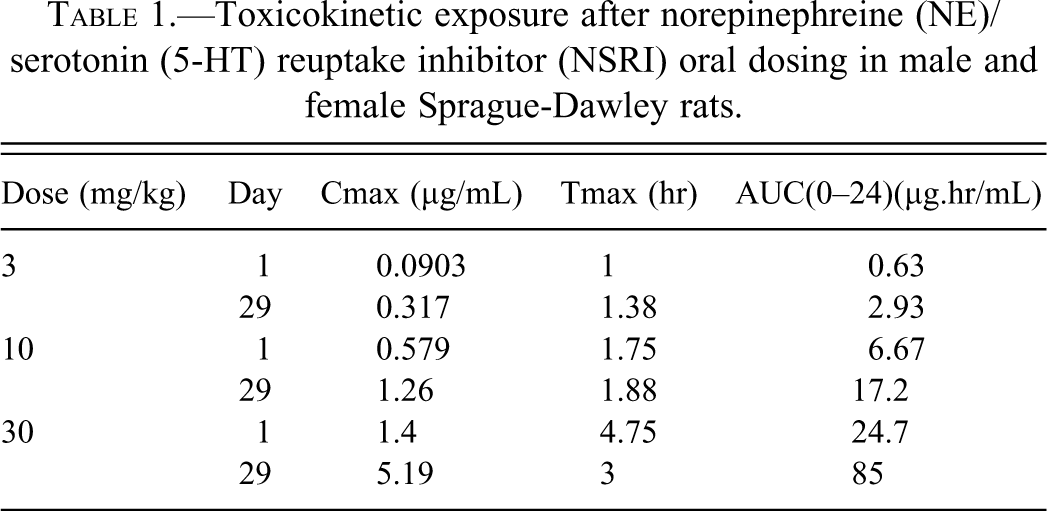
Toxicokinetic parameters were assessed on days 1 and 29 from animals designated for TK assessment. Following blood sampling, animals designated for TK assessment were euthanized and discarded. No other procedures were conducted on these animals. Serum samples were analyzed for NSRI drug concentration using a validated Liquid chromatographic-tandem mass spectrometric (LC/MS/MS) method. The TK parameters of maximum concentration (Cmax), time to Cmax (Tmax), and area under the concentration–time curve (AUC) were determined.
Anatomic Pathology
Organ weights and ratios, gross pathology, histopathology
Necropsies were performed on all animals designated for main study toxicologic assessment. The brain, kidneys, and liver were weighed at scheduled necropsy. Organ-to-body weight and organ-to-brain weight ratios were calculated. Representative samples of the kidneys and liver were collected at necropsy, fixed in 10% buffered formalin, and processed for light microscopic examination. Kidney and liver tissues from all animals were sectioned, stained with hematoxylin and eosin (H&E), and evaluated microscopically.
Immunohistochemistry for DAO
To analyze the protein expression of DAO, 4 µm sections were cut from formalin-fixed, paraffin-embedded blocks, mounted on positively charged glass slides, dried, and then loaded on the automated immunostainer (room temperature using a Ventana Discovery [Ventana Medical Systems, Tucson, AZ]). Slides were deparaffinized and then rehydrated. Enzymatic antigen retrieval was completed with a Ventana specialty solution Protease 2 (Ventana Medical Systems). Endogenous Biotin was blocked using the A/B Kit (Ventana medical Systems) Sections were incubated 4 min each in Avidin and Biotin steps.
Automation included exposure to 100 µL of primary anti-DAO antibody (Rockland Inc.) diluted at 1:1,000 (90 µg/ml) with reagent diluent (Ventana Medical Systems) at room temperature for 120 min. 100 µl of the Secondary Antibody a biotinylated anti-sheep (Vector Labs) diluted at 1:200 with reagent diluent (Ventana Medical Systems) was applied to each section for 30 min at room temperature. Sections were again rinsed and chromagenically labeled using the DABmap Detection Kit substrate solutions (Ventana Medical Systems), followed by counterstaining with Hematoxylin and then Bluing Reagent for 4 min each, removed from the autostainer, washed in warm water, dehydrated through graded alcohol, cleared in xylene, and cover slipped. Control reactions included (1) sections incubated with the omission of primary antibody and processed as mentioned above and (2) sections incubated with normal sheep Immunoglobulin G (IgG) (90 µg/ml) instead of the primary antibody and processed as above. In addition, periodic acid Schiff (PAS) and acid-fast histochemical staining were performed on kidney sections.
Ultrastructural Pathology
Minced tissue samples were fixed in 2.5% glutaraldehyde + 2% formaldehyde in 0.1M sodium phosphate buffer, post-fixed in 0.1M sodium phosphate buffered 1% osmium tetroxide, dehydrated through a graded ethanol series, and embedded in epoxy resin. Sections (approximately 0.6 µm thick) were prepared from two randomly selected epoxy resin tissue-bearing blocks of each selected animal, stained with Toluidine Blue O, and examined using light microscopy. Blocks were trimmed based on light microscopic findings, and sections (approximately 70 nm thick) were prepared from each of the two selected blocks of each animal, stained with uranyl acetate and lead citrate examined in a Hitachi H-7000 transmission electron microscope. Digital images were acquired (AMT, Inc.) and image files were transferred to a secure image server, and appended to the present report. All processes were performed under Good Laboratory Practices guidelines and according to standard operational procedures.
Results
Clinical Signs, Ophthalmic Examination, Body Weight, and Food Consumption
There was no significant effect on body weight at doses ≤ 10 mg/kg/day. Body weight was decreased up to 11% in both sexes at 30 mg/kg/day during the study period. Food consumption was decreased initially but recovered over time. All other parameters (clinical signs and ophthalmic examination) were within normal limits.
Clinical Pathology
No significant NSRI-related changes were observed in hematology parameters. Mean serum total cholesterol was increased 27% in 30 mg/kg/day males and 29% and 62% in 10 and 30 mg/kg/day females, respectively. All other clinical chemistry parameters were within normal limits.
Toxicokinetics
In general, systemic exposure increased with increasing dose (Table 1). Exposure to the NSRI compound at all doses was higher on day 29 compared to day 1. The overall mean AUC (0–24) values ranged from 1.6- to 3.7-fold higher, while overall mean Cmax values ranged from 1.2- to 2.7-fold higher on day 29 compared to day 1. No test article levels were determined in plasma samples obtained from controls.
Toxicokinetic exposure after norepinephreine (NE)/serotonin (5-HT) reuptake inhibitor (NSRI) oral dosing in male and female Sprague-Dawley rats.
Anatomic Pathology
Kidney and liver weights and ratios and gross pathology
When compared to controls, no changes in absolute kidney or liver weights or relative ratios (relative to body or brain) or gross pathology observations were noted in the kidneys or liver at necropsy.
Histopathology, ultrastructural pathology, and immuno-histochemistry
NSRI-related light microscopic changes were evident in kidney sections from rats at ≥10 mg/kg/day. Light microscopic changes in the kidney were characterized by the presence of intranuclear and cytoplasmic inclusion bodies in tubular epithelial cells of the renal cortex but not in the medulla. Inclusions were eosinophilic, round, and surrounded by a clear halo consistent with chromatin margination (Figure 1). Inclusions were PAS and acid-fast negative. This acidic nature of such inclusions was most consistent with protein content. Renal tubular epithelial cells containing these inclusions did not exhibit any features suggestive of cellular degeneration, necrosis, or inflammation, and kidneys were otherwise within normal limits.
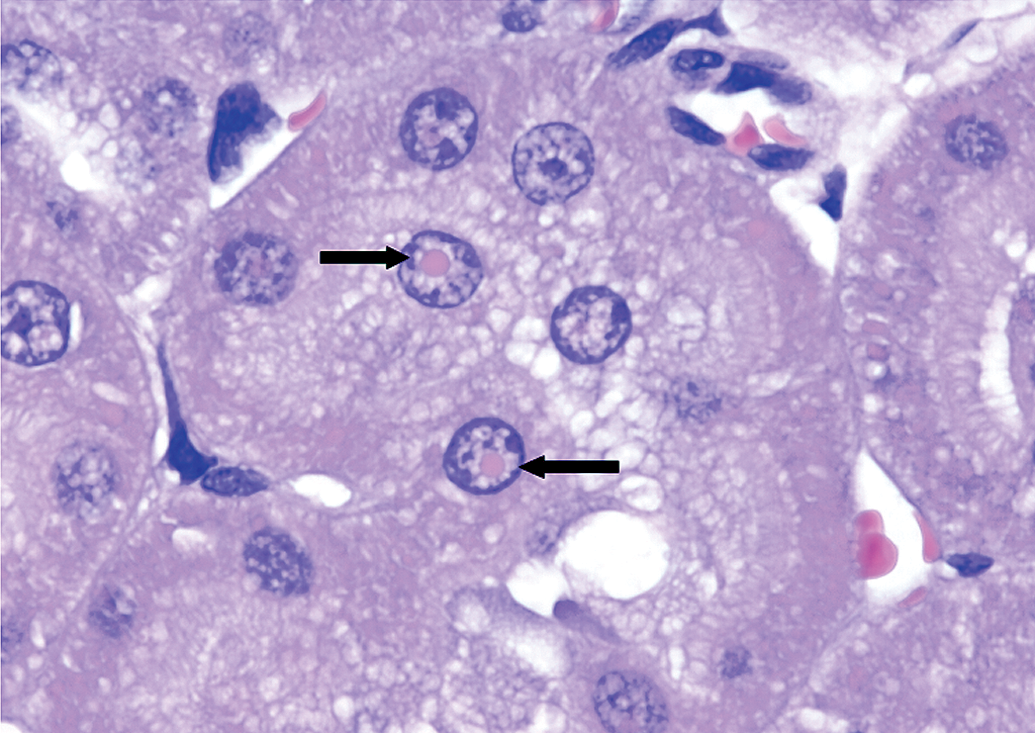
Kidney histopathology section from a Sprague-Dawley rat treated with a norepinephreine (NE)/serotonin (5-HT) reuptake inhibitor (NSRI). Note cytoplasimc and intranuclear inclusion bodies (arrows). H&E. 40×.
By immunohistochemical examination, inclusions were strongly positive for
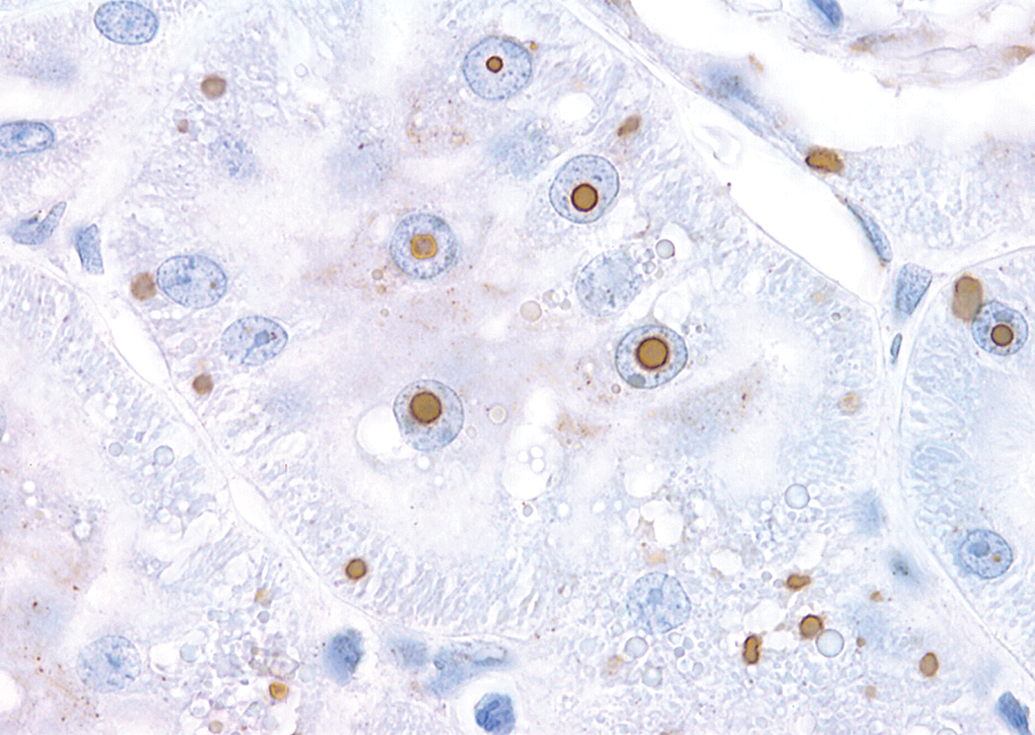
Kidney section immunohistochemical staining from a Sprague-Dawley rat treated with a norepinephreine (NE)/serotonin (5-HT) reuptake inhibitor (NSRI). Note strongly DAO-positive intranuclear and intracytoplasmic inclusion bodies evident in the renal tubular epithelial cells. 40×.
By ultrastructural pathologic examination, intranuclear renal cortical tubular epithelium inclusions consisted of nonmembrane-bound finely granular, amorphous, and uniformly stained material of moderate electron density that displaced the nucleoli and surrounding chromatin to the periphery (Figure 3A and B). Inclusions of similar morphology were also identified in the cytoplasm. There was no evidence of damage or alterations to any of the cellular organelles.
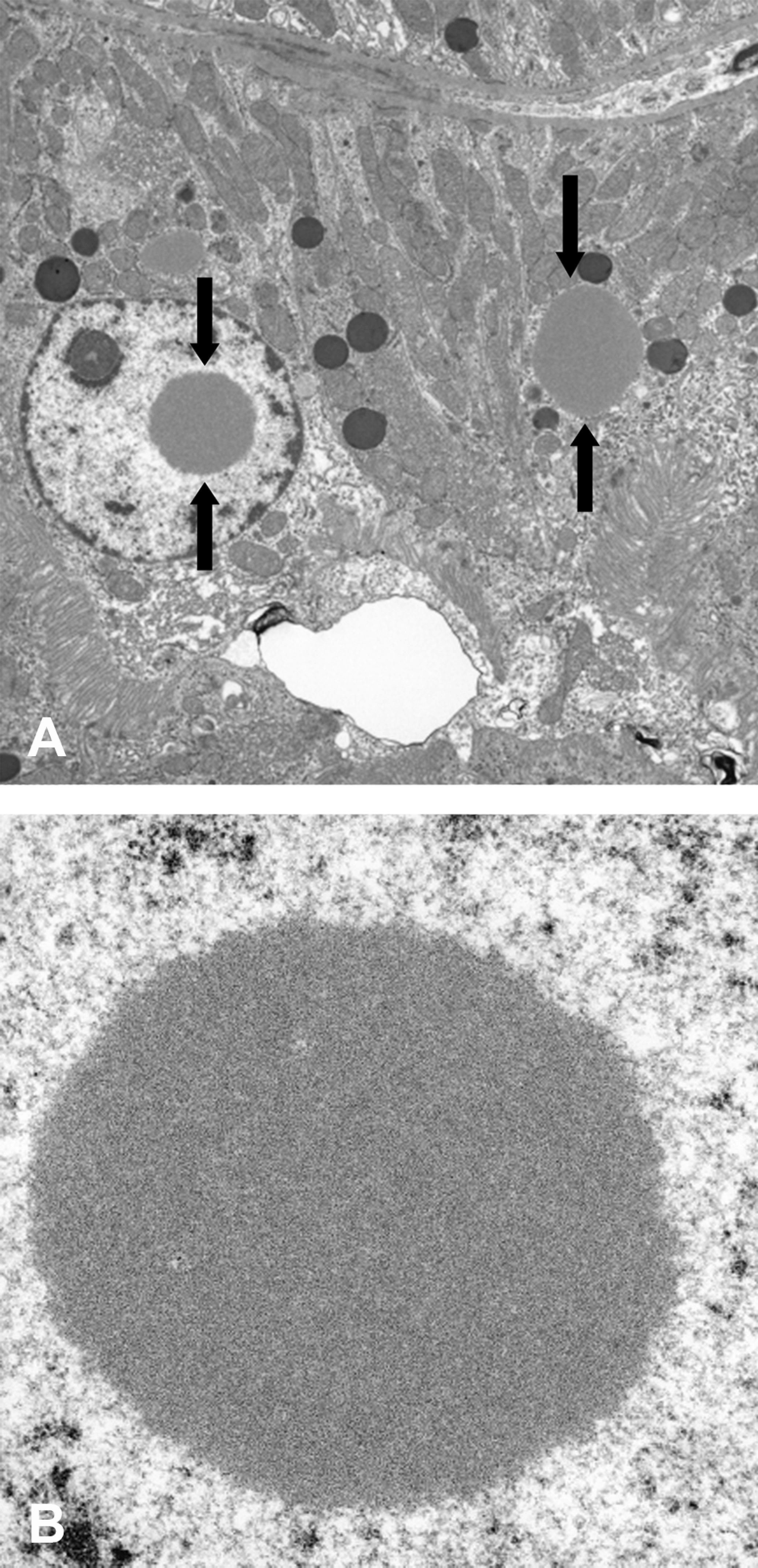
Electron photomicrograph of kidney from a Sprague-Dawley rat treated with a norepinephreine (NE)/serotonin (5-HT) reuptake inhibitor (NSRI). (A) Note intranuclear and intracytoplasmic renal Inclusions. 2,100×. (B) inclusions consisted of nonmembrane-bound finely granular, amorphous, and uniformly stained material of moderate electron density that displaced the nucleoli and surrounding chromatin to the periphery. 11,000×.
Discussion
A test article with dual NSRI inhibition in SD rats was associated with intranuclear and cytoplasmic inclusion bodies in renal tubules in the kidney as assessed by histopathology, histochemistry, immunohistochemistry, and ultrastructural pathology. These inclusions were identified by immunohistochemistry as DAO-positive. The presence of these inclusions in tubular epithelial cells was not associated with pathologic changes or adverse renal function effects as assessed by kidney weights, clinical chemistry parameters, and renal tubular epithelium morphology. The inclusions were not associated with any morphological evidence of renal tubular cellular injury at the light or electron microscopic level. There is no literature, to the best of our knowledge, on the presence of such drug-induced inclusions with an NSRI drug or in the SD rat strain used in safety assessment studies. Published literature indicates that DAO-positive intranuclear and cytoplasmic inclusions were observed in the renal tubular epithelial cells of the F344 rat strain dosed with another drug, propiverine, for up to 52 weeks (Nakano et al. 1989; Yamashita et al. 1990; Day, Heussner, and O’Brien 2006; Gramatte et al. 2006; Heussner et al. 2006; Runkel et al. 2006). Similar to our study with the NSRI test article in SD rats, the presence of these inclusions was not associated with morphological or pathological evidence of renal damage in F344 rats (Yamashita et al. 1990; Day, Heussner, and O’Brien 2006; Gramatte et al. 2006; Runkel et al. 2006).
Immunohistochemical staining identified these inclusions as DAO-positive. DAO is an enzyme that catalyzes oxidative deamination of
There is a DAO-deficient LEA/SENDAI rat model (Maekawa et al. 2005; Konno et al. 2009). Although there is significant deficiency in DAO levels, this model lacks renal pathological changes. In propiverine studies in F344 rats, DAO-positive inclusions were restricted to in the nuclei and the cytosol of the renal cortical proximal tubules of both sexes (Dietrich et al. 2008). No pathological lesions in the liver or kidney, beyond the excessive renal accumulation of cytosolic and nuclear DAO-positive inclusions, were observed in either male or female F344 rats following propiverine treatment. This is consistent with our data. An inbred mutant strain (ddY/DAO) lacking DAO activity was established (Konno et al. 2010). These mice lack any evidence of pathologic abnormalities in the kidney (Knno et al. 2010).
Hepatic clearance is the major route of elimination for our NSRI compound. Propiverine metabolism is similar to our NSRI compound. Propiverine is rapidly absorbed and undergoes extensive and first pass metabolism in the liver to the main metabolite, propiverine N-oxide (Dietrich et al. 2008). It is interesting to note that the renal inclusions seen with propiverine in rats were not seen in B6C3F1 mice (Dietrich et al. 2008). Additionally, no inclusions were seen with propiverine in dogs. Consistent with these data, in another study with NSRI in dogs no inclusions were observed. DAO-positive inclusions in the kidneys of rats only but not dogs or mice, despite high homology to mice, strongly suggest that these are rat-specific drug-induced renal inclusions. We did not have a recovery arm in our study. However, partial recovery of propiverine-induced inclusions after a 5-week recovery period suggests that these test article–induced inclusions would be reversible (Nakano et al. 1989). Therefore, these renal findings appear to be species-specific, reversible, and not relevant to human safety risk assessment. To the best of our knowledge, this article is the first description of intranuclear and intracytoplasmic inclusion bodies in renal tubular epithelial cells associated with NSRI test article administration in SD rats.
Footnotes
The authors declared the following potential conflict of interest: The authors are employed by Pfizer Inc.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Pfizer Inc.
