Abstract
Previous reports investigating the mechanisms of galactosamine toxicity have discussed the presence of responders and nonresponders after intraperitoneal (IP) administration of a toxic dose. The incidence of nonresponders has been reported to be as high as 47%. To rule out inadvertent intestinal, solid organ, or subcutaneous injection as at least a partial cause for the variability, we performed midline incisions and dosed 10 rats via a flexible catheter, with a toxic dose of galactosamine. Results were compared to a previous range finding study with IP-injected rats. As opposed to the IP-injected rats that had a roughly 50% response rate (based on serum alanine aminotransferase [ALT] elevation) and 100% of the midline incision catheter-instilled rats had elevations in ALT. Saline controls had no elevations. Histopathologic examination of livers from 5 midline-incisioned rats euthanized 48 hr after dosing with the lowest ALT responses revealed portal eosinophilic infiltrates and biliary hypertrophy/hyperplasia contiguous with areas of necrosis. Examination of 5 rats with the highest ALT elevations euthanized 10 days post dose revealed similar lesions to be resolving. We conclude that a significant contribution to variability in response to IP-injected galactosamine and possibly other investigative drugs is inadvertent misinjection of all or part of the dose.
Material and Method
In the original dose range finding study, six 10- to 11-week-old female Brown Norway rats were obtained from Charles River Laboratories and after 1 week of acclimatization, they were divided into three groups of 2 rats per group. Groups 1, 2, and 3 received single intraperitoneal (IP) injections of D-(+)-galactosamine hydrochloride (purchased from Sigma-Aldrich, 3050 Spruce St., St. Louis, MO) at 250 mg/kg, 450 mg/kg, and 750 mg/kg, respectively, in a dose volume of 5 ml/kg administered with a 23-gauge needle. Blood samples were drawn pretreatment via tail vein phlebotomy and at 24 and 48 hr post dose to evaluate alanine aminotransferase (ALT) levels. Animals were euthanized after 48 hr and livers harvested for histopathologic evaluation.
In the second study, ten 10- to 11-week-old female Brown Norway rats were obtained from Charles River Laboratories and were acclimatized for 6 days. At the end of this period, rats were anesthetized with isoflurane, shaved along the ventral abdomen, and the surgical field prepared by three cleansings with chlorhexidine surgical solution followed each time by rinses with sterile water. Using a sterile surgical field and aseptic technique, a veterinary surgeon made a 1-cm incision on the midline halfway between the xiphoid process and the umbilicus extending to the peritoneum. Once the peritoneum was visible, a tiny stab incision no more than 0.2 cm in length was made and a sterile flexible catheter inserted a short distance into the abdomen. The dose-filled syringe was discharged through the catheter into the abdomen. The abdominal wall was closed with suture, and the skin was closed with surgical staples. All 10 rats received a single 750-mg/kg dose of D-(+)-galactosamine hydrochloride (Sigma-Aldrich) in a dose volume of 5 ml/kg. A separate group of 3 female stock Brown Norway rats received the same surgical procedure and were dosed with sterile saline in the same dose volume. Blood was drawn prior to treatment to establish an individual animal baseline for ALT and again 24 hr after dosing. Based on ALT elevations, the 5 rats with the most elevated ALT levels were selected to survive to day 10 at which point they were euthanized by exsanguination under isoflurane anesthesia and livers harvested for histopathology. Five rats with the lowest ALT elevations were euthanized after 48 hr and examined histopathologically as were the three saline control rats. All rats had terminal blood samples taken for ALT evaluation.
The care of animals on this study was according to the National Institutes of Health procedures as described in the U.S. Public Health Service Policy on Humane Care and Use of Laboratory Animals (Office of Laboratory Animal Welfare; April 2002). The study protocols were approved by the local animal care and use committee.
Results
In the first dose range finding study (Table 1), 50% of animals treated by IP injection had ALT elevations above the baseline range.
Serum alanine aminotransferase (U/L) levels prior to treatment and after a single D-(+)-galactosamine hydrochloride intraperitoneal injection with a 23-gauge needle in female Brown Norway rats.
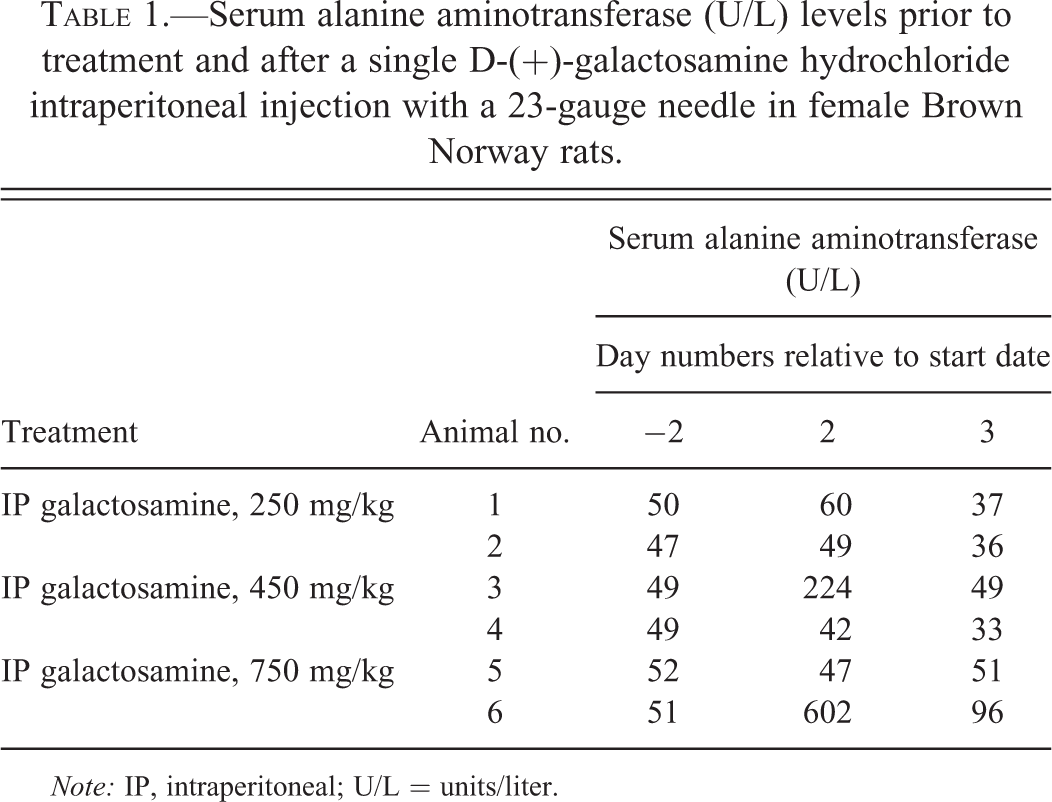
Note: IP, intraperitoneal; U/L = units/liter.
In the midline incision study (Table 2), pretreatment ALT levels were consistently within a narrow range and rose in all animals 24 hr after treatment.
Serum alanine aminotransferase (U/L) levels prior to treatment and after a single D-(+)-galactosamine hydrochloride installation by flexible catheter through a ventral midline incision in female Brown Norway rats.
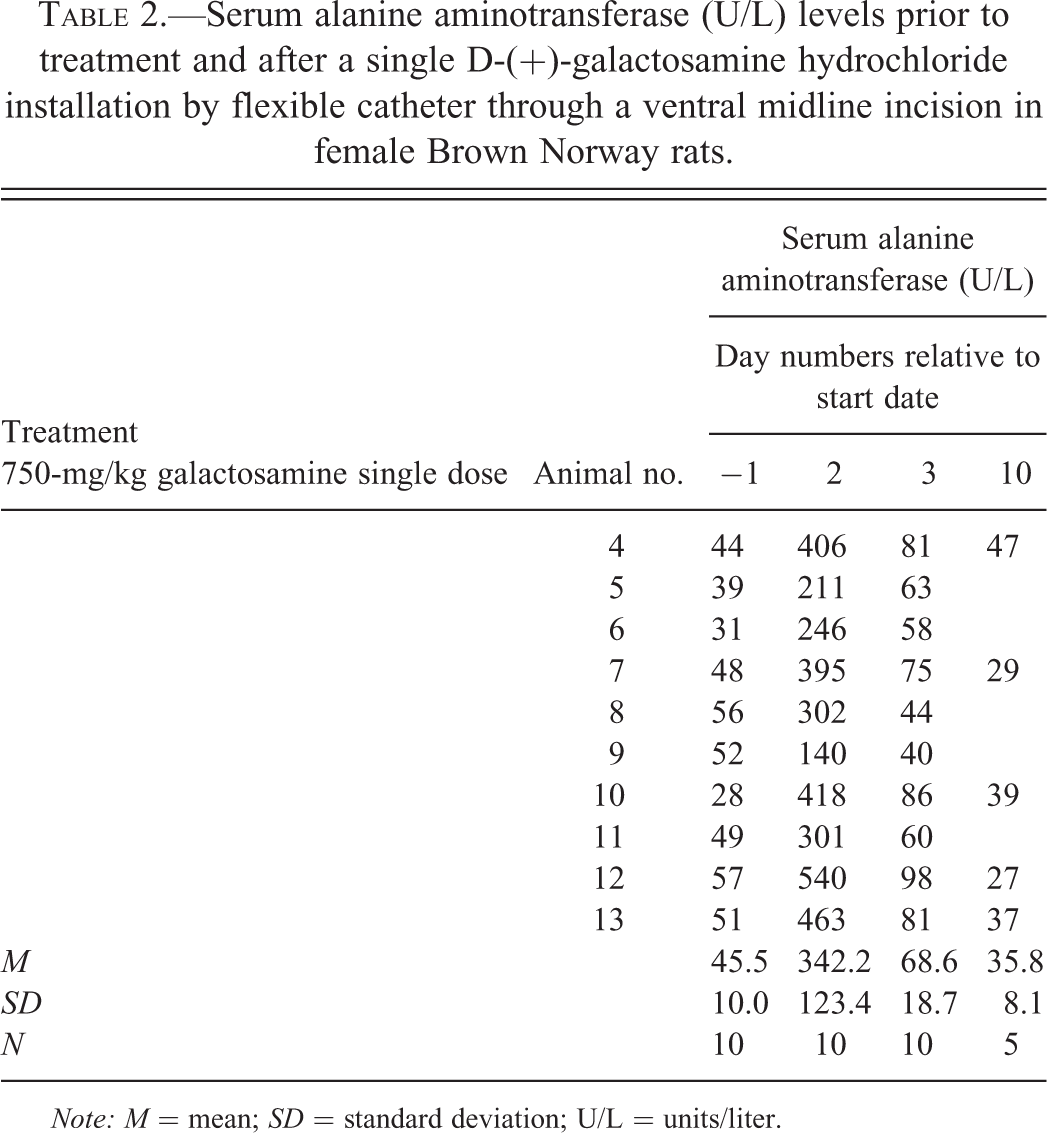
Note: M = mean; SD = standard deviation; U/L = units/liter.
All ALT elevations were normal or returning to the normal range by 48 hr after galactosamine administration. All the animals euthanized 9 days after dosing had normal range ALT values.
Typical histopathologic findings in animals with ALT elevations did not differ between surgically instilled and IP-injected galactosamine-treated livers and included portal eosinophilic infiltrates and biliary hypertrophy and hyperplasia contiguous with areas of necrosis with associated histiocytic/eosinophilic infiltrates. These features are presented in Figures 1 and 2. In galactosamine-dosed rats without ALT elevations, eosinophilic infiltrates or minimal necrosis were present indicating some exposure by IP injection had occurred in most cases but perhaps not complete delivery of the full dose into the peritoneal cavity.
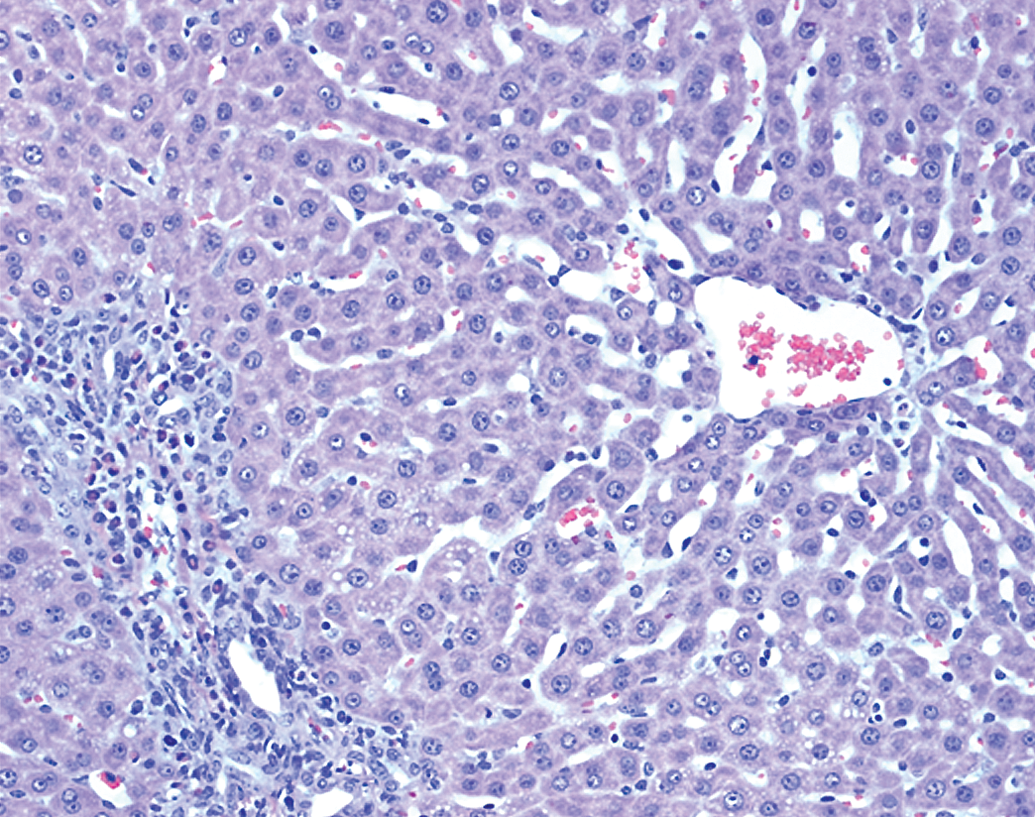
Galactosamine 750-mg/kg single dose delivered via 23-gauge needle. Liver section 48 hr after dosing. Minor increase in eosinophils in portal area and absence of ALT elevation at 24 hr indicate probable partial misdose. H&E (250×). ALT = alanine aminotransferase; H&E = hematoxylin and eosin.
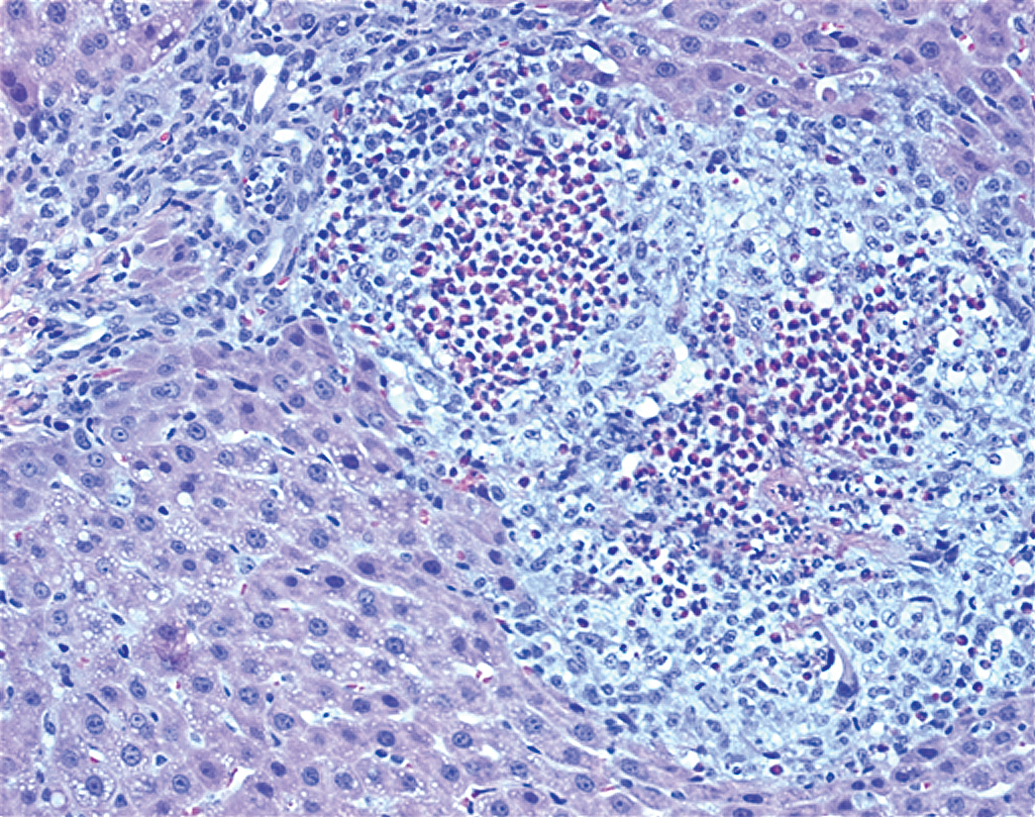
IP galactosamine 750-mg/kg single dose delivered through an abdominal incision via flexible catheter. Lesions are 48 hr post dosing showing periportal area of necrosis with mixed eosinophil and histiocytic infiltrate and biliary hypertrophy and hyperplasia. H&E (250×). H&E = hematoxylin and eosin; IP = intraperitoneal.
A tabular listing of histopathologic findings for each animal from the dose range finding study reveals a correlation of ALT with the most severe hepatic lesions (Table 3).
Tabular listing of histopathologic findings by animal with reference to Serum ALT.

Note: ALT = alanine aminotransferase. X = examined for lesion but lesion not present.
Grade 1 = minimal; Grade 2 = mild; Grade 3 = moderate; Grade 4 = marked.
Discussion
Previous reports investigating the mechanism(s) of galactosamine toxicity in rats have discussed the presence of responders and nonresponders after IP administration of a toxic but nonlethal dose (Coen et al. 2009, 2012). The incidence of nonresponders has been reported to be as high as 47%. Recent studies using the rat as a model in our laboratory also found a 50% response rate after a single IP injection of toxic and subtoxic doses. The nature of this variability was reported by Coen et al. (2009, 2012) to be associated with the presence of parent and metabolites within the intestinal lumen in nonresponders while responders had metabolites predominantly in urine. Although galactosamine is used as an acute hepatic toxin for nonclinical investigative studies, the issue of the presence of responders and nonresponders for any pharmaceutical is problematic, especially when idiosyncratic toxicity is the issue. Therefore, the nature of the variable phenotype must be carefully researched. A recent report by Masson et al. (2011) examining factors contributing to variability in IP-dosed galactosamine metabolic profiles concluded, “For aqueous extracts, technical variability was two to four times lower than within group interanimal variability. Similar results were obtained for organic extracts for the galactosamine group.” Numerous other toxins and investigative drugs have shown similar variability in response after IP injections. A recent study of IP ketamine–xylazine dosing had response rates varying from 30 to 85% with a mean response rate of 56.7%. Older studies of IP injection site failure report failure rates ranging from 1–2% up to 10–20% (Steward et al. 1968; Gaines Das and North, 2007; Miner, Koehler and Greenway, 1969). Because of the association of galactosamine nonresponders with intestinal metabolites, we felt that misadministration of all or part of the galactosamine dose into the intestine or other sites during IP injection should be completely ruled out. To rule out inadvertent intestinal injection as at least a partial cause for the variability in hepatic galactosamine response, we performed midline incisions and dosed via a flexible catheter in 10 rats with a toxic but nonlethal dose of galactosamine and compared the results to 3 saline-treated rats dosed by the same route and to a previous range finding study with intraperitoneally injected rats. As opposed to the intraperitoneally injected rats, which had a roughly 50% response rate (as measured by ALT elevation) to the toxic galactosamine dose, 100% of the midline incision, catheter-instilled rats had significant elevations in ALT. None of the midline-incisioned, saline controls had ALT elevations. Histopathologic examination of livers from 5 midline-incisioned rats euthanized 48 hr after dosing with the lowest ALT responses revealed portal eosinophilic infiltrates and biliary hypertrophy and hyperplasia contiguous with areas of necrosis with associated histiocytic/eosinophilic infiltrates. Examination of the 5 rats with the greatest ALT elevations euthanized 9 days post dose revealed similar lesions to be resolved or resolving but with persistent portal eosinophilia in most animals. Histopathology of livers from the range-finding rats revealed similar lesions that correlated with the severity of ALT elevations.
Clearly, this experiment is not conclusive proof that misadministration of galactosamine during IP dosing is solely responsible for the variability in responses seen by ourselves and others. Indeed, it has been suggested that administration of anesthesia may contribute to enhanced galactosamine toxicity, and anesthesia (or relative hypoxia and enhanced endotoxin absorption) as an additive effect to galactosamine toxicity cannot be ruled out by our study design. That being said, the models in question where anesthesia was described as increasing toxicity were designed for a large animal model of fulminant liver failure and used bolus intravenous (IV) dosing of 1,000 g/kg and either prolonged halothane anesthesia (over a period of 3–4 days) or neuroleptanalgesic combination of a tranquilizer and an opioid for 72 hr. Notably, animals dosed IV (Kalpana et al. 1999) with or without anesthesia had elevated ALT levels indicating that IV dosing, bypassing the pitfalls of IP dosing, had sufficient exposure to raise ALT and thus be counted as a responder even if the severity of the response was less than that with concurrent anesthesia (Diaz-Buxo et al. 1997). We conclude that a significant contribution to variability in response to intraperitoneally injected galactosamine and possibly other investigative drugs given by this route is misadministration of the drug.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
