Abstract
Clinically, girls appear to be more sensitive than boys to the cardiotoxic effects of doxorubicin, whereas the opposite may be true for adults. To identify and characterize potential sex-related differences, adult male and female spontaneously hypertensive rats (SHR; some ovariectomized [OVX]) received 1 mg/kg of doxorubicin or saline iv weekly for 9, 10, or 12 weeks. Weight gain was slower in treated males. Serum concentrations of cholesterol and triglycerides increased and those of albumin decreased in both sexes, but changes were more pronounced in treated males. Treated males had significantly more severe cardiomyopathy scores and higher serum levels of cTnT than females. The increased cardiotoxicity was accompanied by higher numbers of cardiac mast cells (MCs) and percentage of cardiac MCs undergoing degranulation. Doxorubicin-treated OVX animals had significantly increased numbers of cardiac MCs, more severe myocardial lesions, and elevated serum concentrations of cTnT compared to doxorubicin-treated normal female SHR. The severity of cardiac lesions in the OVX female was similar to that observed in doxorubicin-treated males. This study demonstrated the presence of sex-related differences in the cardiotoxic effects elicited by doxorubicin and identified variations in the level of cardiac MC activity as a factor which could possibly contribute to the male-female dissimilarity.
Keywords
Introduction
The incidence of cardiovascular disease is lower in premenopausal women than in men (Ogita et al. 2003). The lower incidence of cardiac disease in women has been attributed to the protective effects of estrogen. In contrast, diastolic dysfunction is more prevalent in postmenopausal hypertensive women than in similarly aged men (Mori et al. 2011), presumably due to lower estrogen levels in this patient population. In prepubertal girls, who also have low concentrations of estrogen, doxorubicin-induced reductions in ventricular contractility were more common and severe than in boys (Lipshultz et al. 1995; Krischer et al. 1997). In fact, the incidence of early and late doxorubicin-induced cardiomyopathy is greater in prepubertal girls than in boys of the same age.
Experimental studies examining the influence of sex on anthracycline-related cardiomyopathy and nephropathy have been inconclusive. Van Hoesel et al. (1986) and Sakemi et al. (1996) reported that anthracycline-associated nephropathy in female rats was less severe than in males. Julicher et al. (1988) also found that changes in body weight, urine albumin concentrations, and the incidence of ascites in females treated with doxorubicin were not as profound as that occurring in male rats. These investigators suggested that the reduced toxicity may occur because female rats have a higher capacity for cellular defenses against reactive free radicals than male rats (Julicher et al. 1984, 1988).
Studies that included ovariectomized (OVX) female Sprague-Dawley or OVX female Wistar rats showed that renal alterations were more severe in OVX females than in normal females (Montilla et al. 2000; Munoz-Castaneda, Tunez, et al. 2005; Munoz-Castaneda, Muntane, et al. 2005; Munoz-Castaneda, Montilla, et al. 2005; Munoz-Castaneda et al. 2006; Sakemi et al. 1997). The occurrence of more severe nephropathy was attributed to reduced concentrations of estrogen (Sakemi et al. 1997) resulting in increased cellular oxidative stress and reactive oxygen species (Montilla et al. 2000; Munoz-Castaneda et al. 2006).
Mast cells (MCs) and macrophages contain estrogen and androgen receptors (Frisancho-Kiss et al. 2007). Stimulating estrogen receptors lowers the proliferation of MCs and macrophages, slows the release of histamine, and reduces the concentrations of proinflammatory cytokines, whereas testosterone has the opposite effects. More recently, Lu et al. (2012) reported that female sex hormone–related cardioprotection in the heart with volume overload is caused by an estrogen-altered cardiac MC phenotype, and/or an estrogen-related prevention of MC activation. Cardiac MCs in spontaneously hypertensive rats (SHRs) have also been reported to facilitate myocardial dysfunction (Shiota et al. 2003).
In the present study, we used an SHR model that included adult normal male and female as well as OVX female rats to evaluate the influence of sex on doxorubicin-induced cardiomyopathy and nephropathy (Herman et al. 1985, 1999; Zhang et al. 1996). In particular, we assessed the relationship of cardiac and renal MCs with doxorubicin-induced toxicity.
Materials and Methods
Animals
The protocol was approved by the Institutional Animal Care and Use committee of the FDA White Oak Animal Program (Silver Spring, MD). Animal care procedures conformed to the 1996 Institute of Laboratory Animal Resources Guide for the Care and Use of Laboratory Animals (National Research Council 1996).
The study included 33 adult male and 29 female SHRs (12 weeks old) and 10 OVX female SHRs (12 weeks old) obtained from Harland Breeding Facilities (Indianapolis, IN). The animals were housed individually and given rodent chow and water ad libitum.
Study Initiation Procedures
The study was divided into four experiments that were intended to be of equal 12-week treatment duration. In Experiment 1, 6 male and 5 female SHR were treated with Dox. Severe toxicity (3/6 male deaths at 9 weeks) led us to terminate dosing of males at 10 weeks. Females were dosed for the intended 12 weeks. To prevent excess toxicity (particularly male SHR) in the remaining experiments, the maximum dosing period was shortened to 9 or 10 weeks. In Experiment 2, both male and female rats (5 each) were treated for 10 weeks. In Experiment 3, 6 male and 4 female rats were treated for 9 and 10 weeks, respectively. All 3 experiments included groups of male and female control animals (5–6/group) which were given iv saline weekly for the appropriate dosing period. In Experiment 4, 10 female rats were OVX by the vendor 4 weeks before the 12-week shipment age. After the 1-week acclimation period, treatment commenced with 1-mg/kg/wk doxorubicin (5 animals) or saline (5 animals) weekly for 10 weeks. In all experiments, treatment commenced after a 1-week acclimation period.
All rats were injected weekly with 1-mg/kg doxorubicin (Drug Synthesis and Chemistry Branch, NCI, NIH, Bethesda, MD) or saline. The doxorubicin was prepared in normal saline at a dose volume of 1.0 mg/ml just before use (0.1 ml/100 g body weight). Rats in control groups received the same volume of normal saline. Injections were given through a saline-containing, 27-gauge venocath inserted into a tail vein through a small-gauge needle, which minimized the risk of doxorubicin extravasation into surrounding tissue. Each animal was weighed weekly. In Experiment 1 only, systolic blood pressure was monitored with a noninvasive tail cuff method (IITC Life Science Instruments, Woodland, CA) before starting treatment and prior to necropsy (1 week after the end of treatment).
Study Termination Procedures
Studies were terminated 1 week after the final dosing (at 9, 10, or 12 weeks). The animals were anesthetized with isoflurane and euthanized by exsanguination by means of a needle-tipped catheter inserted into the inferior vena cava. Plasma samples for hematology determinations (Experiments 2 and 3) were collected in heparin-containing syringes. Serum samples obtained from centrifuged clotted blood were used for measuring concentrations of cardiac troponin T (cTnT; all animals) and blood urea nitrogen, creatinine, cholesterol, triglycerides, and albumin (Experiments 2 and 3).
Necropsy Procedures
During necropsy, all major organs were examined grossly. The entire heart and the left kidney were excised. Each heart and kidney was cut sagitally and the two portions of the bisected tissues fixed in 10% neutral buffed formalin. One-half of the heart and kidney tissues were embedded in paraffin, sectioned at a thickness of 5 µm and stained with hematoxylin and eosin (H&E). The remaining heart and kidney tissues were embedded in glycol methacrylate plastic resin. Sections (1-µm thick) of the plastic embedded heart tissue were stained with alkaline toluidine blue.
Histopathologic Evaluation
Sixty-four longitudinally cut heart and kidney sections (two each toluidine blue stained and H&E stained) contained representative structures that included all three layers of the heart (endothelium, myocardium, and epicardium) and three portions of the kidney (cortex, medulla and pelvis). These tissue sections from the hearts and kidneys of all animals were evaluated blindly by pathologists (J.Z. and H.H.) without knowledge of cardiac troponin T and clinical chemistry analysis. Cardiac and renal MCs were assessed by J.Z. using toluidine blue–stained plastic sections.
The severity of the myocardial lesions was graded on a 6-point, ordinal scale ranging from 0 to 3, based on the percentage of myocytes displaying myofibrillar loss and cytoplasmic vacuolization: 0, normal; 1, 5% or less; 1.5, 6% to 15%; 2, 16% to 25%; 2.5, 26% to 35%; and 3, 35% or more (Billingham 1991). The severity of renal lesions (tubular dilatation and damage, deposition of protein casts within the tubular lamina, and glomerular vacuolization) was graded on a 5-point ordinal scale of from 0 to 4 (Zhang et al. 1996).
Method for Counting MCs
Cardiac MCs were counted in toluidine blue–stained tissue sections. The counting was performed in 50 microscopic fields of heart tissue selected without bias at a magnification of ×200. The resultant data were expressed as mean (SD) number of MCs per mm2 of tissue section. MC degranulation was identified by extruded granules and the appearance of ruptured cell membranes. A ratio of the number of degranulated MCs to the number of intact MCs was expressed as mean (SD) percentage of the total MC per mm2 of the section.
Morphological changes in MCs were assessed microscopically on the basis of our previous experience (Knapton et al. 2010). MCs were distinguished from other inflammatory cells by their shape, size, and cellular contents. Inactivated MCs are readily observed as angular or elongated dense cells containing dark perinuclear granular material. Their borders are smooth and regular. Activated MCs are similar, except they have become swollen and more rounded in shape. MC degranulation was identified by extruded granules and the appearance of ruptured cell membranes. Degranulating MCs characteristically have an irregular border as granules are released from the cytoplasm. Also, degranulating MCs become lighter in color as the perinuclear granules are released into the surrounding tissue.
We did not count renal MCs because they are not evenly distributed in the kidney. Renal MCs in the toluidine blue–stained sections were present in the connective tissue of the capsule, in the perirenal fat on the convex portion of the kidney, and in the fat tissue on the concave hilum of the renal sinus but were absent in the interstice of the cortex and medulla. These findings are consistent with those of Ehara and Shigematsu (2003), who reported that animals with renal diseases did not have MCs in the interstitium.
Blood Sample Analysis
Terminal serum clinical chemistry analysis (Experiments 2 and 3) was performed on the VetScan model #200-1000 using the Comprehensive Diagnostic and Large Animal rotors (Abaxis Inc., Union City, CA). Blood cell counts and other hematological values (Experiments 2 and 3) were determined with the VetScan HMT (Abaxis).
The Cardiac Troponin T Assay
Blood samples were centrifuged (3,000 × g for 15 min), and the serum was frozen at −40°C until assayed for concentrations of cTnT (Elecsys Stat; Roche Diagnostics, Indianapolis, IN) by the laboratory of Children’s Hospital, Harvard Medical School, Boston, MA. Technicians were blinded to the treatment.
Statistical Methods
The Tukey–Kramer multiple comparisons test was used to assess differences between treatment groups in the mean values of final body weight, mean arterial pressure (Experiment 1 only), and hematological, cardiac troponin T, and blood analyte values. Differences in myocardial and renal lesion scores among the various treatment groups were assessed with the Kruskal–Walis Test for nonparametric data. Alpha was set at .05, and all tests were two-tailed. The GraphPad Instat statistical software package (GraphPad Software, Inc, San Diego, CA) was used in all analyses.
Results
General Toxicity and Blood Pressure
Severe toxicity among males in Experiment 1 (3 died by 9 weeks of dosing) led us to shorten the planned maximum dosing periods from 12 weeks to 9 or 10 weeks. A majority of the 14 surviving doxorubicin-treated males in Experiments 1, 2, and 3 were less active than controls and had rough hair coats. Necropsy revealed that many had excess pericardial and peritoneal fluid. Terminal blood samples appeared milky. The kidneys from these treated males were lighter in color than those from control males.
All doxorubicin-treated normal female rats remained in good condition and survived to the end of all experiments. Excess pericardial and peritoneal fluid was again found in 4 of the 15 doxorubicin-treated normal females. The kidneys were lighter in color than those from control normal females, but the discoloration was less evident in the normal females than in the males. The centrifuged blood from 7 doxorubicin-treated normal females appeared slightly milky.
Although the animals were of similar age (12 weeks old at the beginning of the dosing period), the pretreatment mean body weight of all males was significantly higher than the mean of all females (324 ± 21 grams vs. 193 ± 10 grams, p < .05). Doxorubicin-treated males gained significantly less weight than did control males (mean increase 104% vs. 117%, respectively, p < .05). Control females increased body weight by 114%, whereas the increase in doxorubicin-treated females was only 105% (p < .05).
Blood pressure was monitored only in Experiment 1. Initial blood pressure was similar in male and normal female rats. Mean (SD) baseline blood pressure was 185 (15) mmHg for doxorubicin-treated males, 175 (6) mmHg for doxorubicin-treated normal females, 162 (12) mmHg for control males, and 170 (6) mmHg for control normal females. Mean (SD) terminal systolic blood pressures were significantly higher: 194 (9) mmHg for control males and 216 (13) mmHg for control normal females. Terminal systolic blood pressure was also higher in doxorubicin-treated normal females (213 [16] mmHg). The poor condition of doxorubicin-treated males precluded measuring terminal blood pressure.
Hematology and Clinical Chemistry Changes
In Experiment 2, white blood cell counts significantly decreased (3.0 [0.5] and 4.4 [0.6] × 103 µl) and platelet counts significantly increased (755 [224] and 485 [112] × 103 µl; p < .05) in doxorubicin-treated versus control normal females. In Experiment 3, red blood cell counts and hematocrit were significantly decreased (6.69 [0.36] and 7.78 [0.04] × 107 µl, 32.3 [2.0] and 37.6 [0.3]%) and platelet counts were significantly increased (927 [20] vs. 547 [97] × 103 µl) in doxorubicin-treated compared to control normal females (p < .05).
In Experiment 2, the concentrations of a number of analytes were altered in doxorubicin-treated males and normal females (increases in blood urea nitrogen, creatinine, cholesterol, and triglycerides and decreases in albumin; Table 1). Changes in blood analytes in Experiment 3 were similar to those in Experiment 2. The most profound changes in both experiments were in the concentrations of cholesterol and triglycerides (Table 1). In doxorubicin-treated males, mean terminal serum cholesterol concentrations were 6.3 to 8.6 times higher than those of control animals. Significant increases in cholesterol among doxorubicin-treated normal females were from 2.4 to 4.5 times higher than the values from control normal females. The maximum concentrations in normal females were significantly less than the concentrations determined in doxorubicin-treated males (p < .05; Table 1). The effect of doxorubicin on serum triglyceride concentrations was similar to that seen with serum cholesterol (Table 1).
Clinical chemistry values for spontaneously hypertensive rats before and after 9 to 10 weeks of treatment with 1-mg/kg doxorubicin/week or saline.
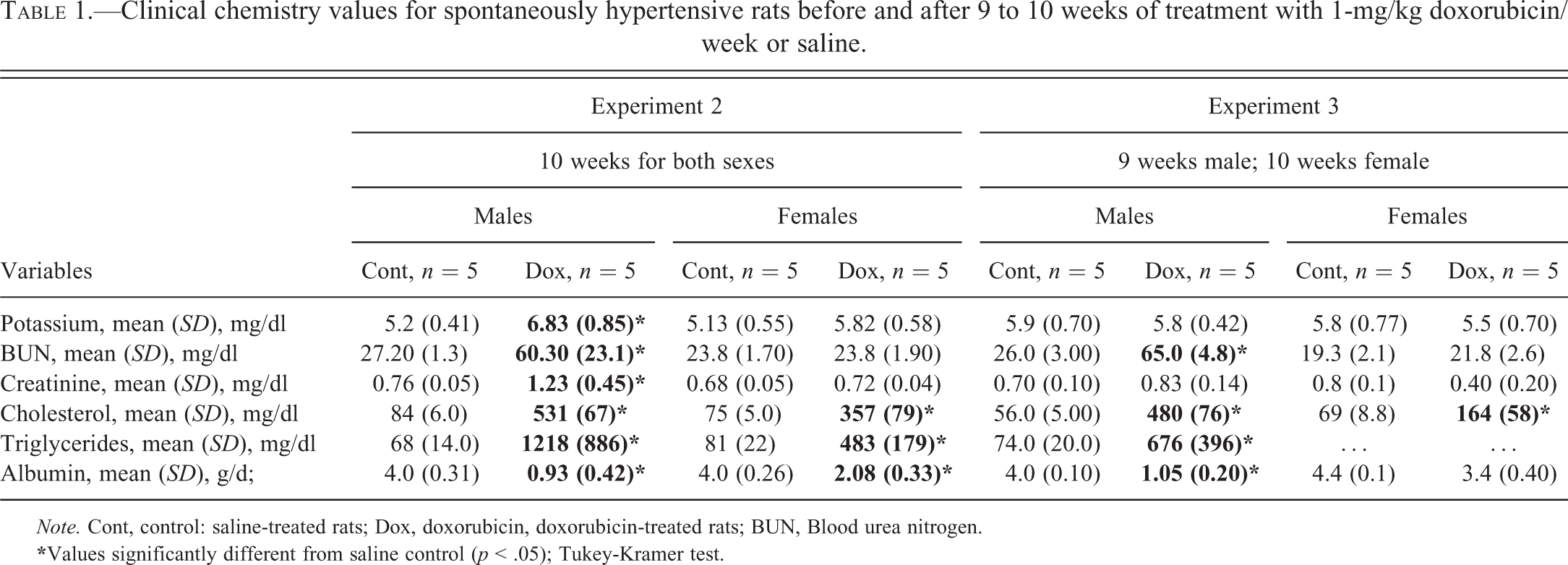
Note. Cont, control: saline-treated rats; Dox, doxorubicin, doxorubicin-treated rats; BUN, Blood urea nitrogen.
Histopathology Results
Myocardial Tissue
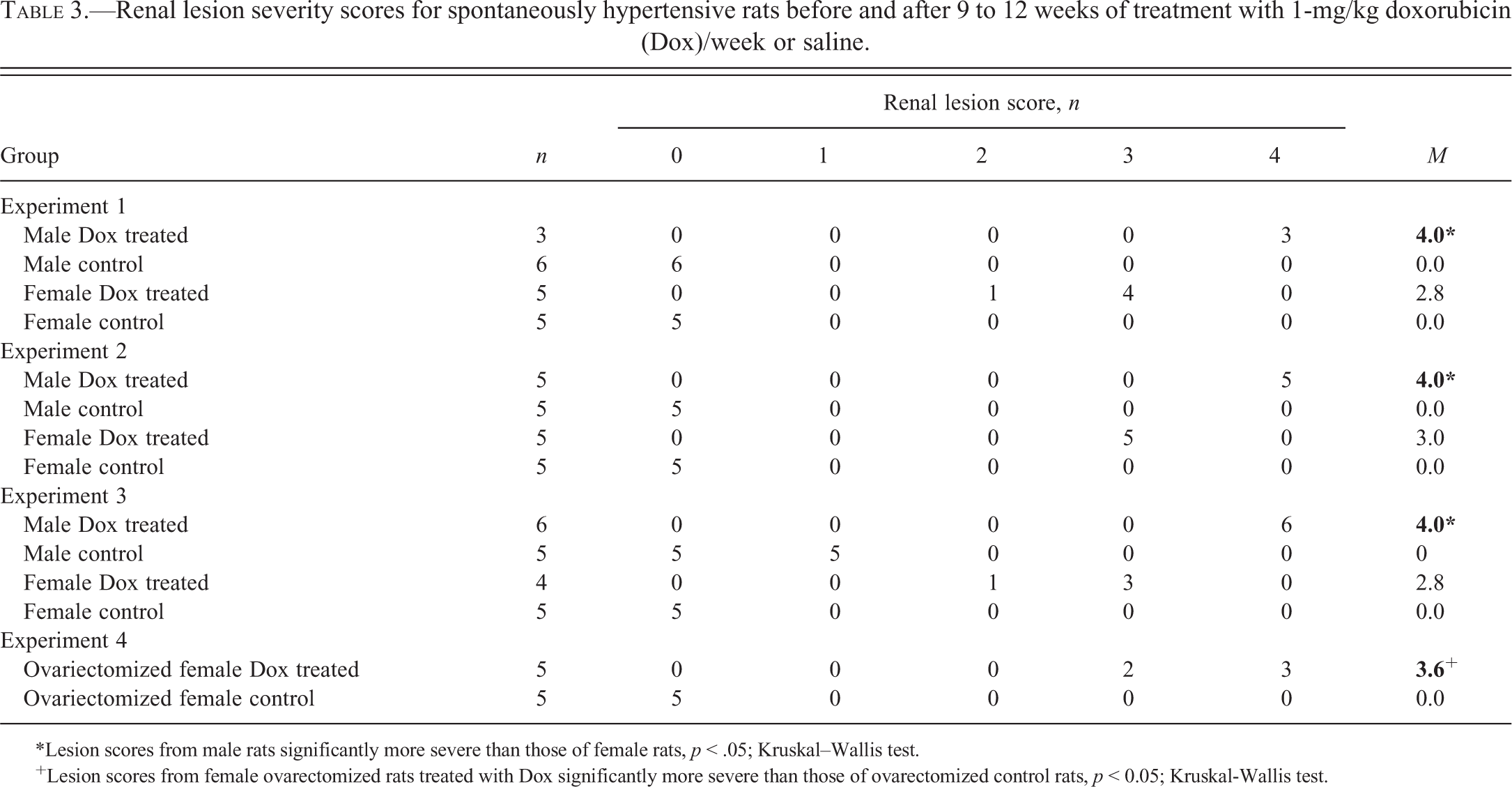
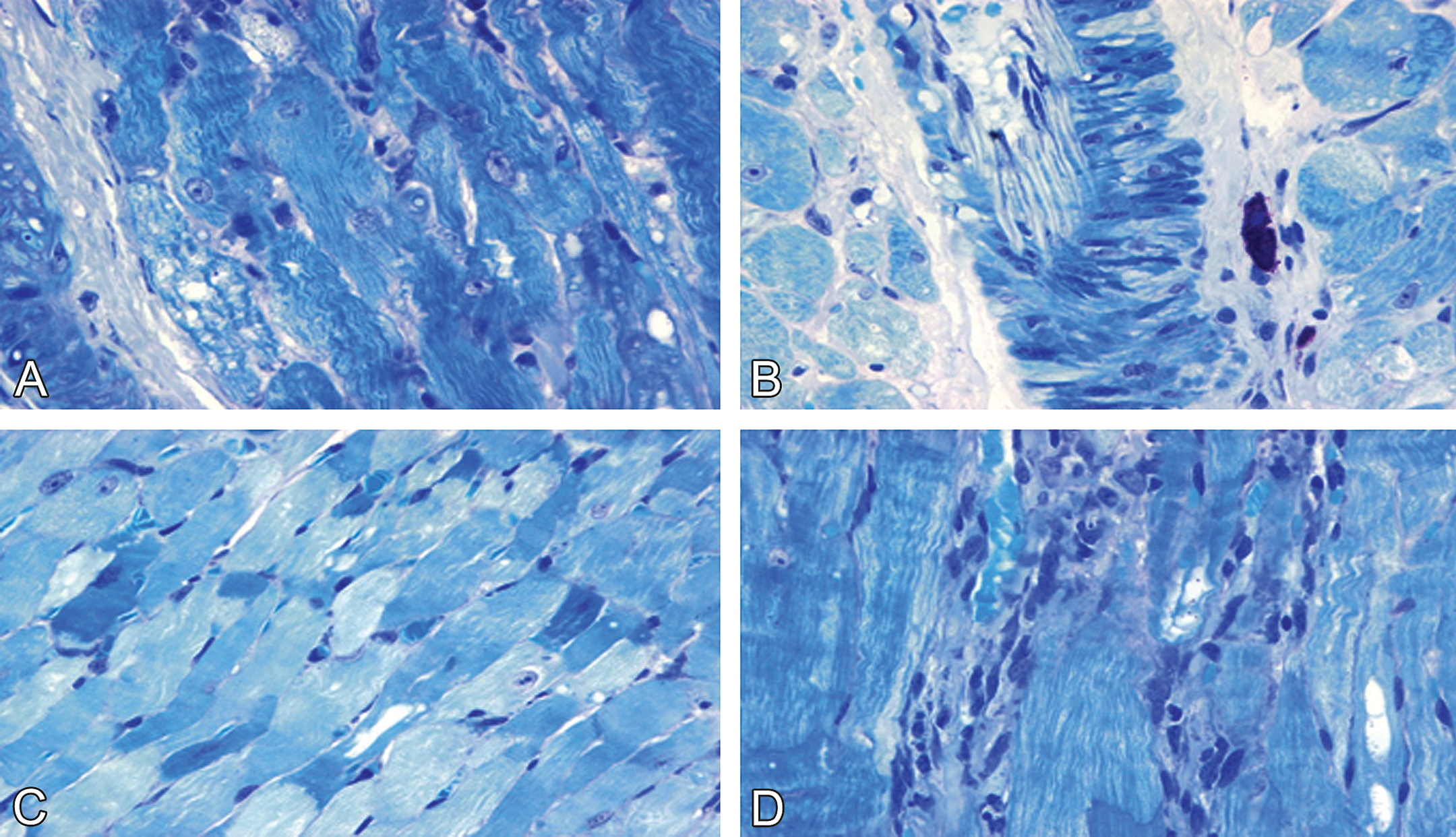
In Experiments 1 through 3, control males and normal females showed no cardiac damage (Figure 1A–B); however, cytoplasmic vacuolization and myofibrillar loss were found in doxorubicin-treated males and normal females (Figure 1C–D). Further, these doxorubicin-induced myocardial lesions were more severe in males than in normal females (Figure 1C–D). This difference in severity also occurred in Experiment 1, even though females received a higher cumulative dose of doxorubicin (12 mg/kg) than did males (9–10 mg/kg).
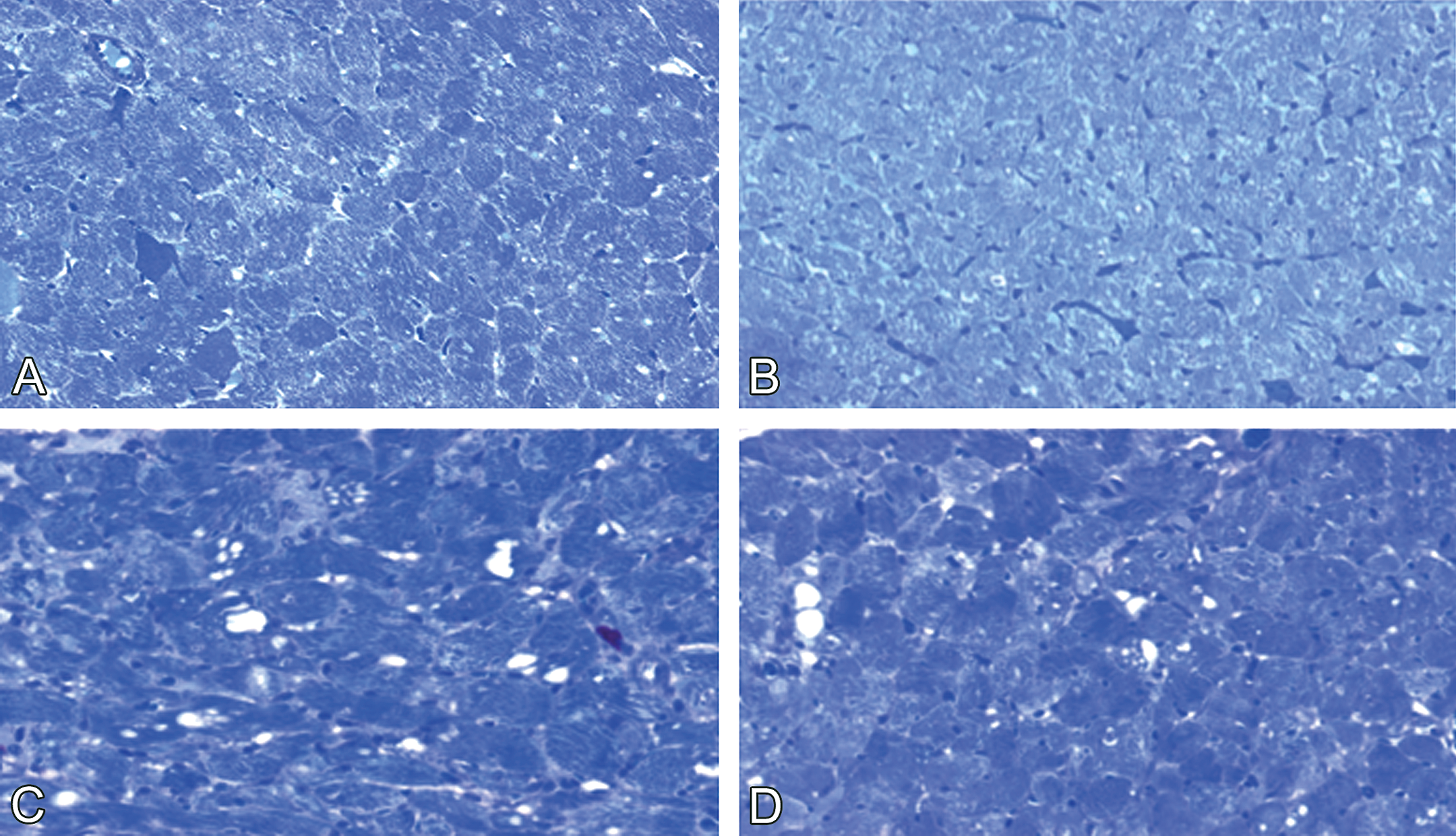
Doxorubicin-induced cardiotoxicity in male and female spontaneously hypertensive rats. (A) Normal myocyte morphology in male and (B) female control rats. (C) Cytoplasmic vacuolation and myofibrillar loss are more severe in male (1 mg/kg doxorubicin/week × 10 weeks) than in (D) female (1 mg/kg doxorubicin/week × 12 weeks) (D) rats. Toluidine blue stain, ×400.
Overall, cardiomyopathy scores were higher in doxorubicin-treated males (mean lesion scores ranged from 2.1 through 2.6) than in treated normal females (mean lesion scores ranged from 1.3 to 1.8, p < .05; Table 2). The significance of the difference in myocardial lesion severity between male and normal female SHR was based on analysis by the Mann–Whitney test for nonparametric data.
Cardiac lesion severity scores for spontaneously hypertensive rats before and after 9 to 12 weeks of treatment with 1 mg/kg doxorubicin (Dox)/week or saline.
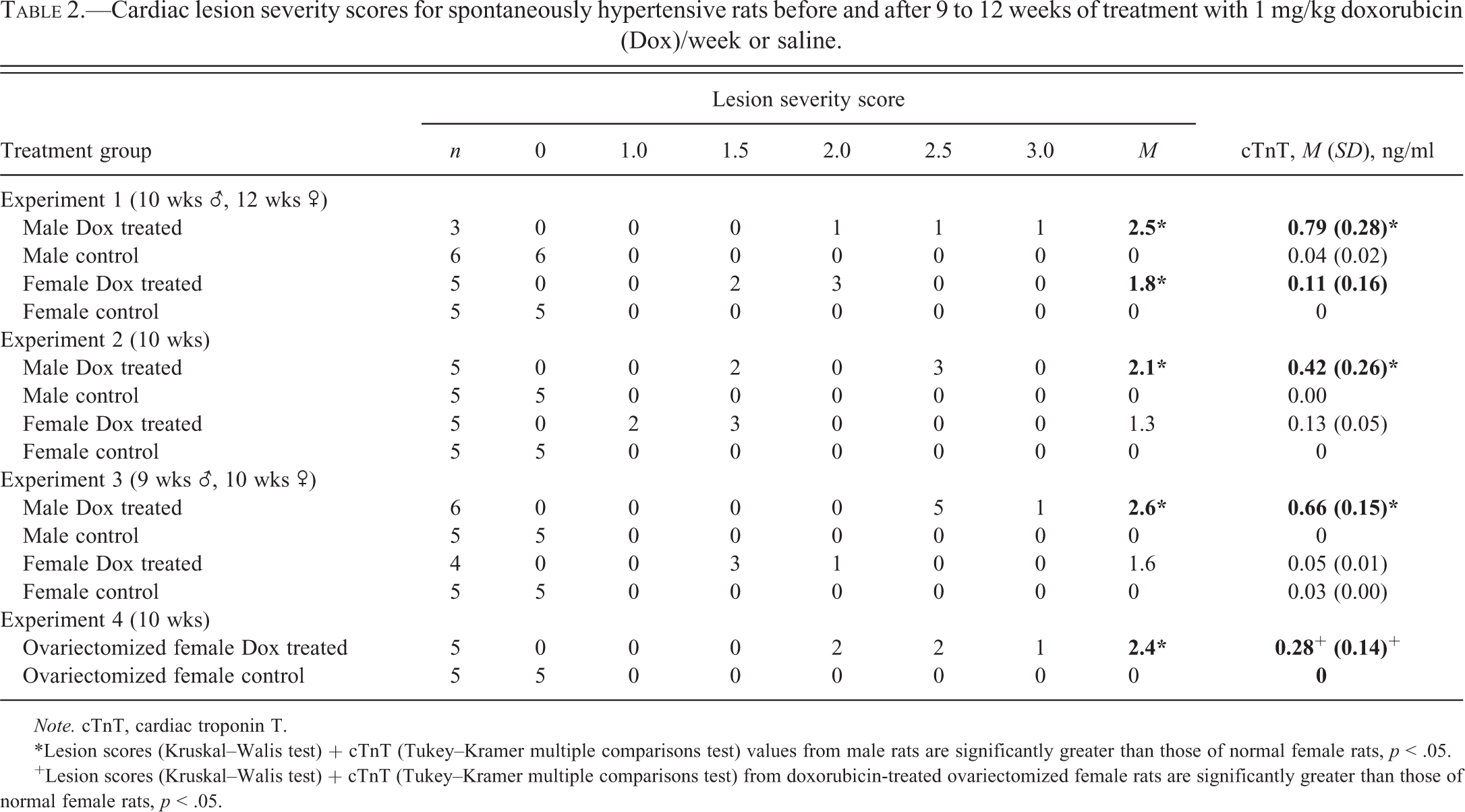
Note. cTnT, cardiac troponin T.
*Lesion scores (Kruskal–Walis test) + cTnT (Tukey–Kramer multiple comparisons test) values from male rats are significantly greater than those of normal female rats, p < .05.
+Lesion scores (Kruskal–Walis test) + cTnT (Tukey–Kramer multiple comparisons test) from doxorubicin-treated ovariectomized female rats are significantly greater than those of normal female rats, p < .05.
Renal Tissue
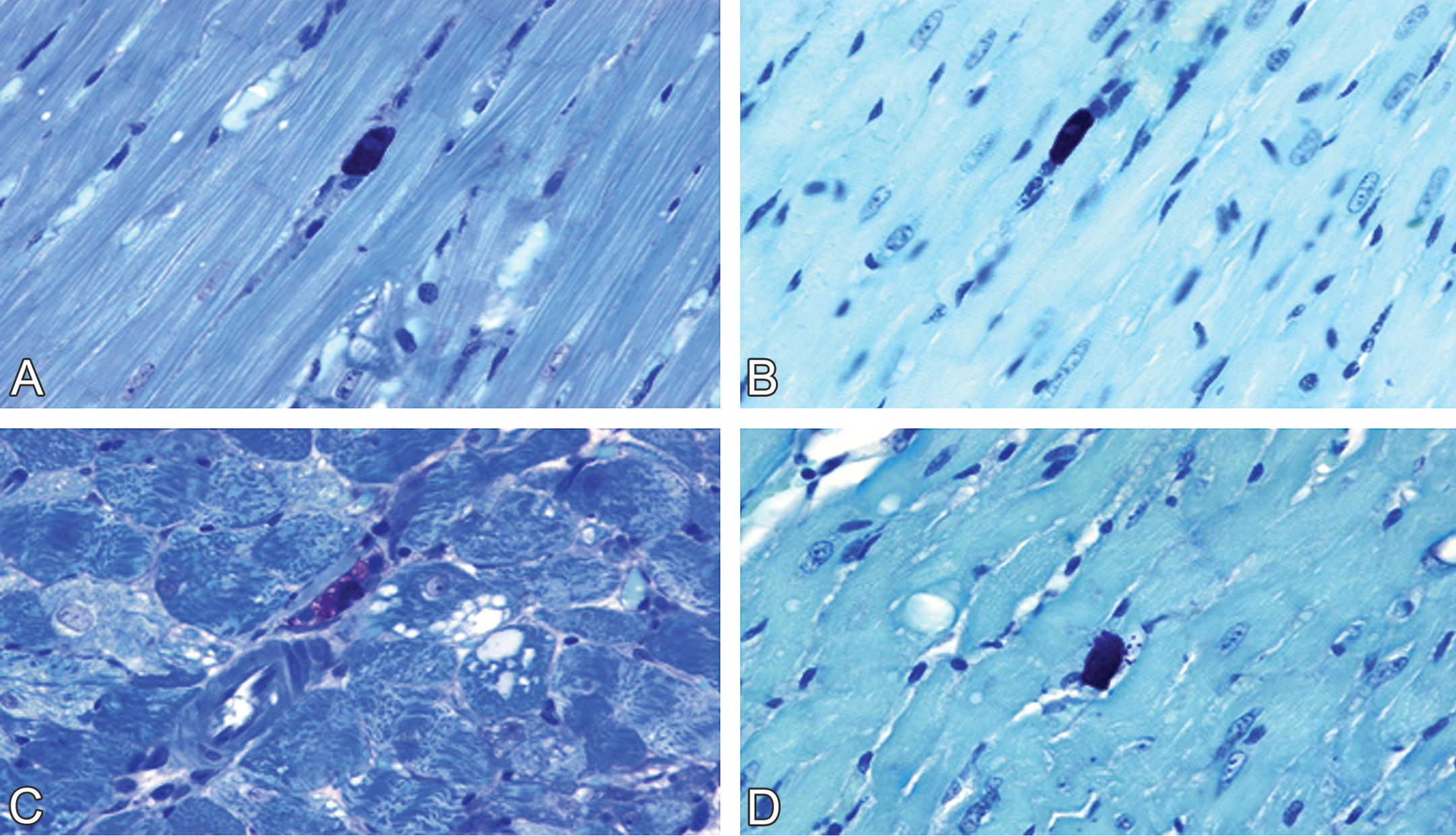
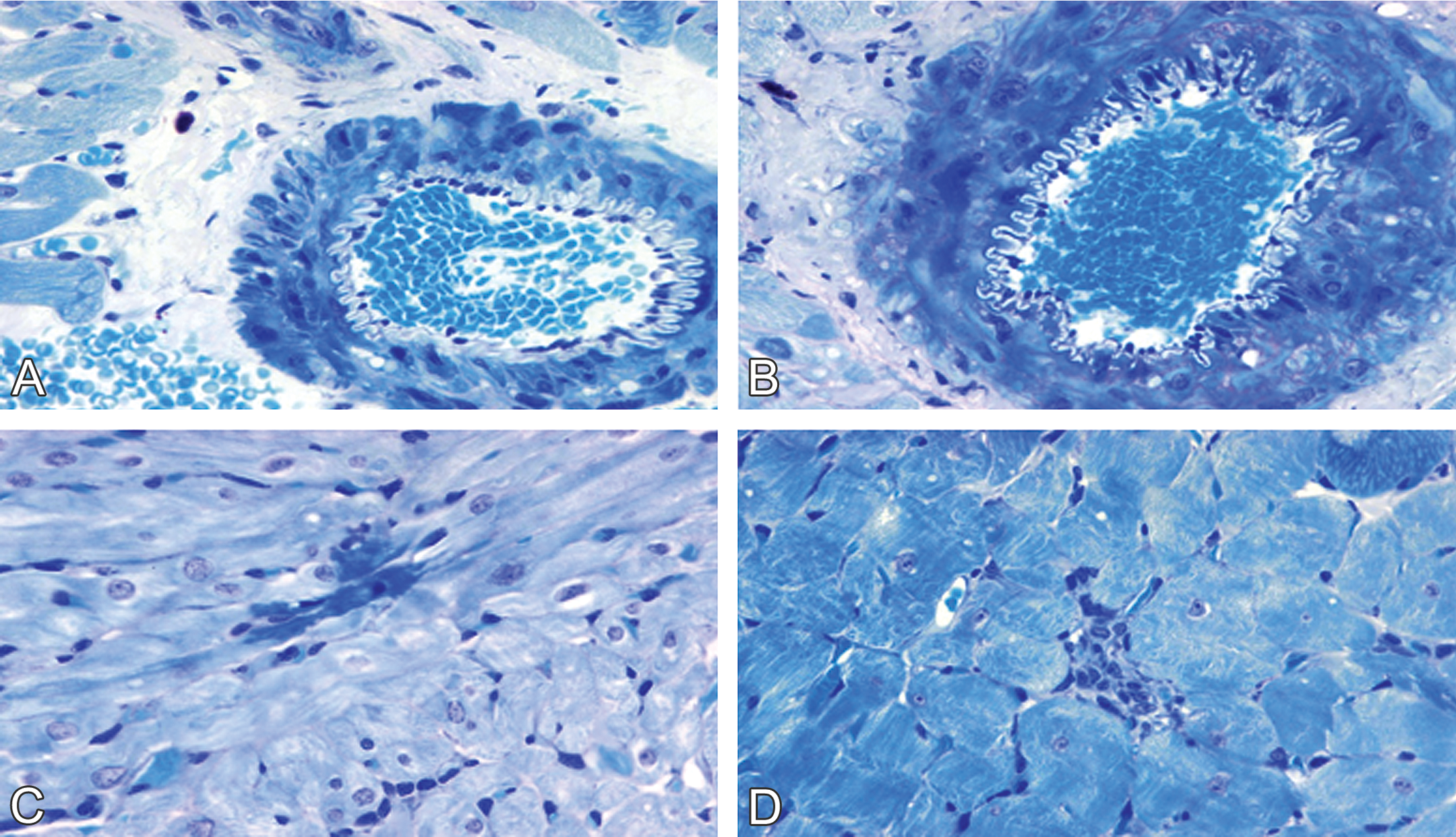
Renal injury was absent from control males and normal females (Figure 2A–B), but glomerular vacuolization and sclerosis, dilatation of the tubules, protein casts in the tubular lumens, and interstitial lymphocytic infiltration were present in doxorubicin-treated males and normal females (Figure 2C–D). Nephropathy scores were significantly greater in doxorubicin-treated males (mean lesion score, 4) than in doxorubicin-treated normal females (mean lesion score, 2.8–3.0; p < .05; Table 3) as determined by the Mann–Whitney test.
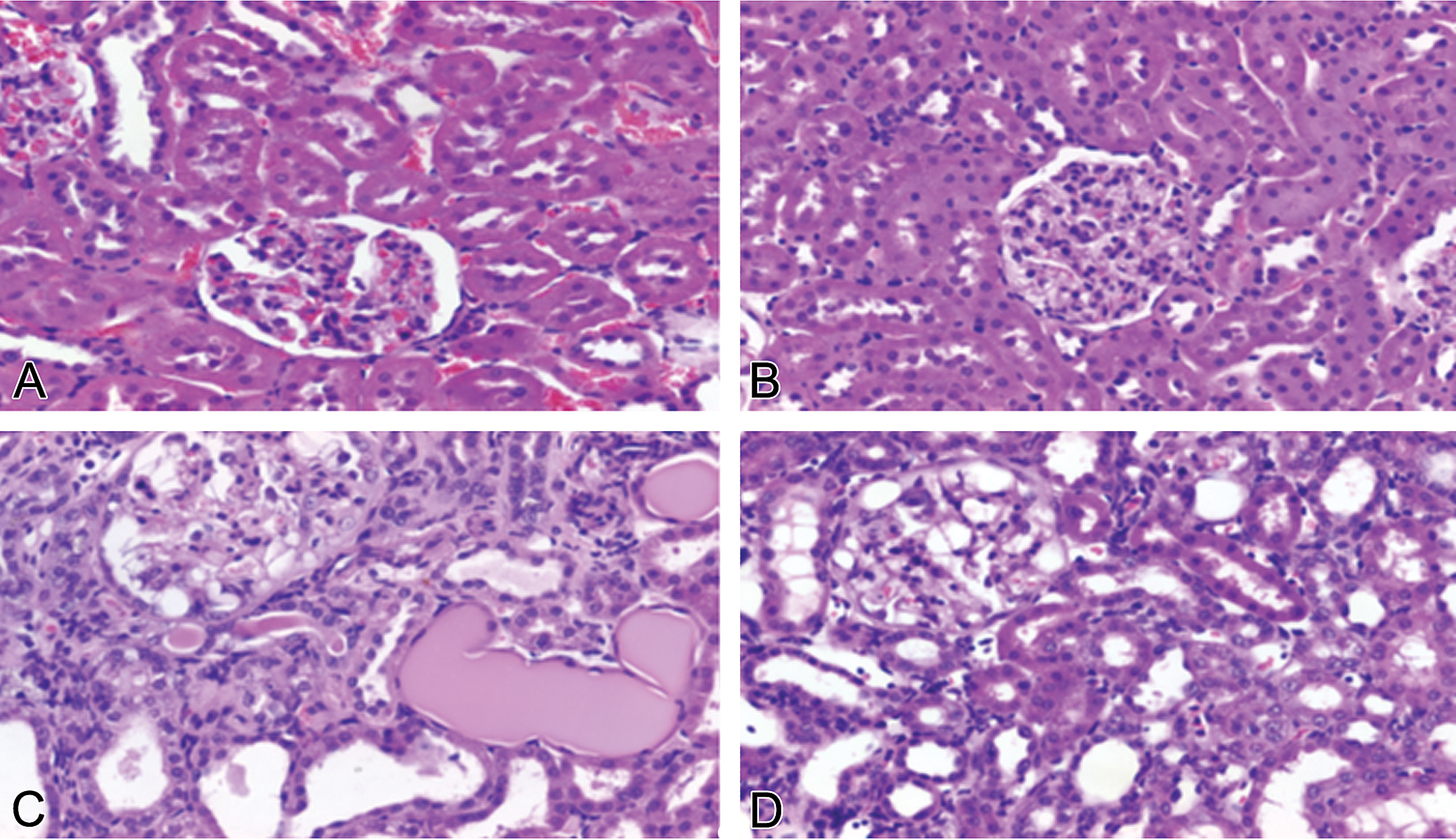
Doxorubicin-induced nephrotoxicity in male and female spontaneously hypertensive rats. Normal tubular and glomerular morphology in (A) male and in (B) female control rats. (C) Doxorubicin-induced nephrotoxicity in a male rat (1 mg/kg/week × 10 weeks) was characterized by severe glomerular vacuolization, tubular dilatation, and protein casts in tubular lumens. Tubular epithelia damage, including necrosis (small spots suggestive of apoptotic bodies, beneath the glomerulus) and degeneration (cytoplasmic vacuoles, on the left bottom corner) are also visible. (D) Doxorubicin-induced nephrotoxicity in female rats (1 mg/kg/week × 12 weeks) was characterized by moderate glomerular vacuolization and mild tubular dilatation (hematoxylin & eosin stain).
Renal lesion severity scores for spontaneously hypertensive rats before and after 9 to 12 weeks of treatment with 1-mg/kg doxorubicin (Dox)/week or saline.
*Lesion scores from male rats significantly more severe than those of female rats, p < .05; Kruskal–Wallis test.
+Lesion scores from female ovarectomized rats treated with Dox significantly more severe than those of ovarectomized control rats, p < 0.05; Kruskal-Wallis test.
MCs Evaluation
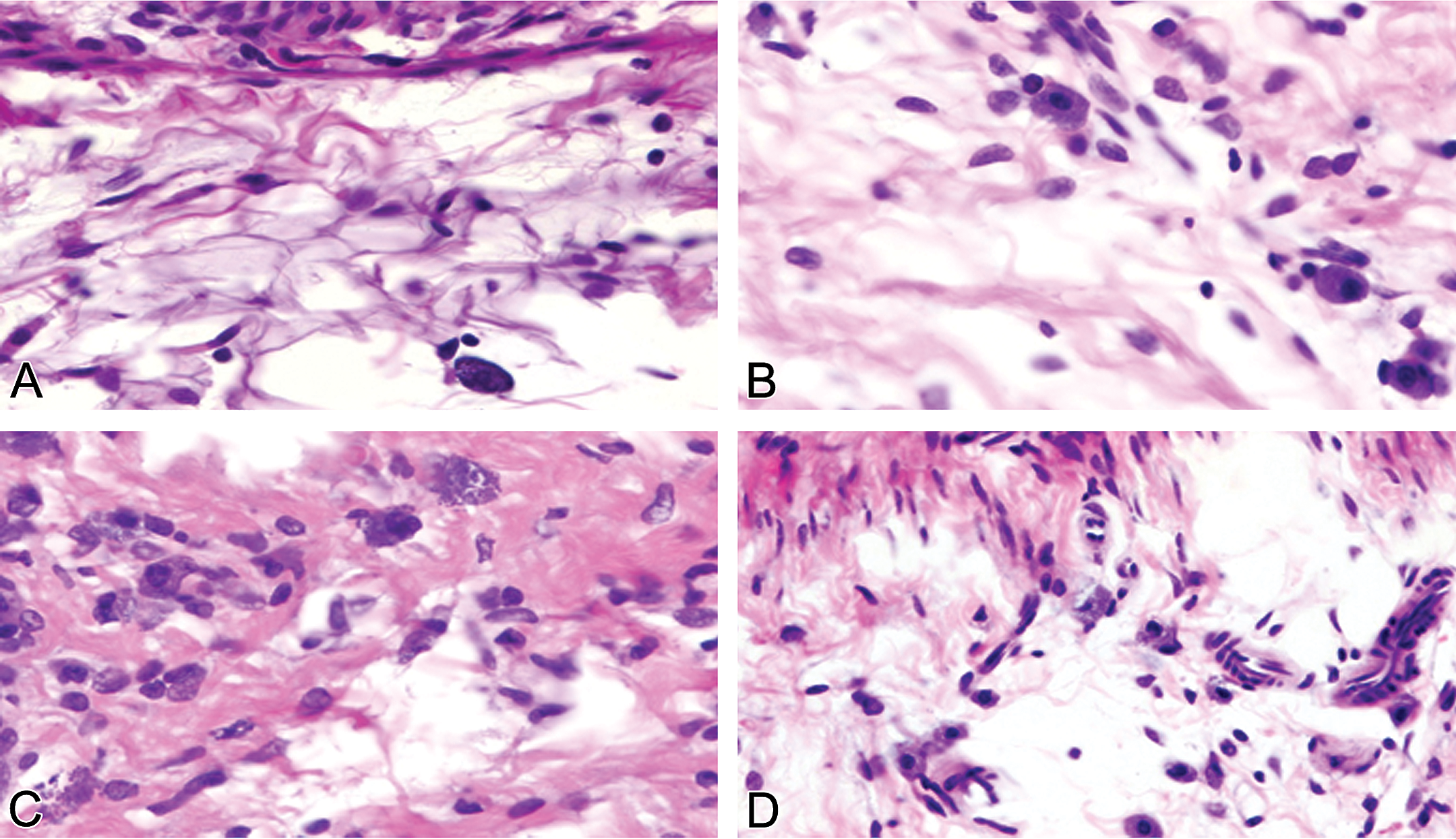
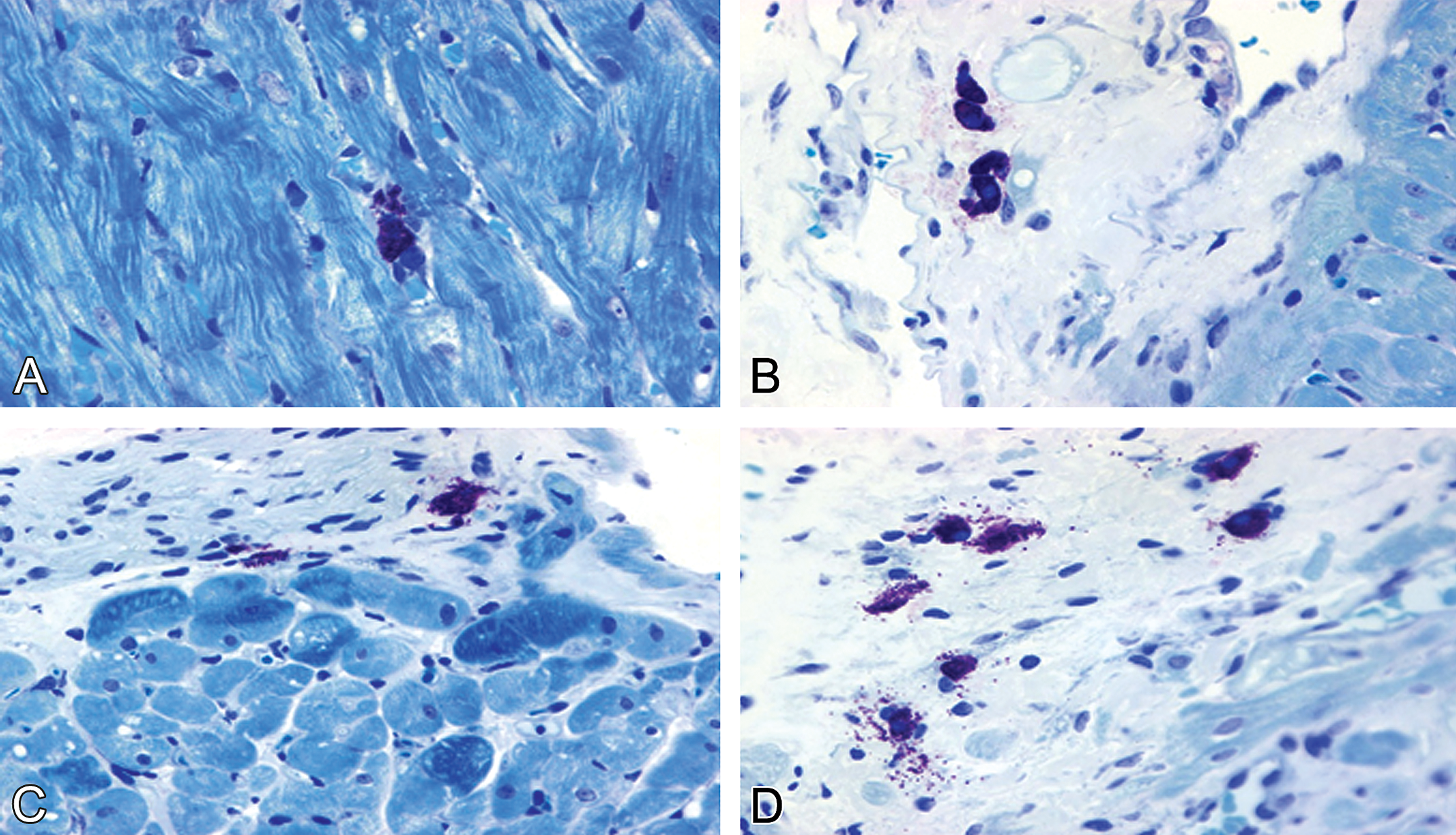
Morphological alterations of cardiac and renal MCs were evaluated on the tissue sections from control male and normal female rats. In control males and normal females, the morphology of resident cardiac and renal MCs was intact (Figures 3A–B and 4 A–B). In doxorubicin-treated rats, the association between MC degranulation and myocardial injury was more pronounced in males than in normal females (Figure 3C–D). In doxorubicin-treated males, the mean (SD) number of cardiac MCs (1.56 [0.18]/mm2) and the percentage of MC degranulation (0.64 [0.15]/mm2) was significantly higher than that in doxorubicin-treated normal females (number of MCs, 0.75 [0.24]/mm2; percentage of MC degranulation, 0.3 [0.05]/mm2; Table 4).
Representative photomicrographs comparing different degrees of cardiac mast cell degranulation in male and female rats. Quiescent cardiac mast cells show intact morphology without myocardial injury in saline-treated (A) male and (B) female. (C) A high incidence of cardiac mast cell degranulation in doxorubicin-treated male in association with severe myocyte damage. (D) Reduced incidence of cardiac mast cell degranulation in doxorubicin-treated female associated with mild myocyte damage. (A and D) Toluidine blue–stained plastic sections; (B and D) toluidine blue–stained paraffin sections. All images are ×630.
Number of cardiac mast cells and percentage of mast cell degranulation per cubic millimeter of tissue sections in normal male, normal female, and ovariectomized female rats treated with or without doxorubicin.

Statistical significance determined by the Tukey–Kramer multiple comparisons test.
*Significantly different compared to male rats given saline (p < .001).
**Significantly different compared to normal female rats given doxorubicin (p < .001).
***Significantly different compared to normal female rats given doxorubicin (p < .01).
In control males and normal females, renal MC morphology was normal (Figure 4 A–B). In doxorubicin-treated rats, renal MC degranulation was more common in males than in females (Figure 4C–D).
Representative photomicrographs comparing different degrees of renal mast cell degranulation in male and female rats. Intact renal mast cells with normal renal morphology in saline-treated (A) male and (B) female rats. Renal mast cell degranulation enhanced more in doxorubicin-treated (C) male than in (D) doxorubicin-treated females. A–D, H&E stain. All images are ×630.
OVX Female Rats Treated with Saline
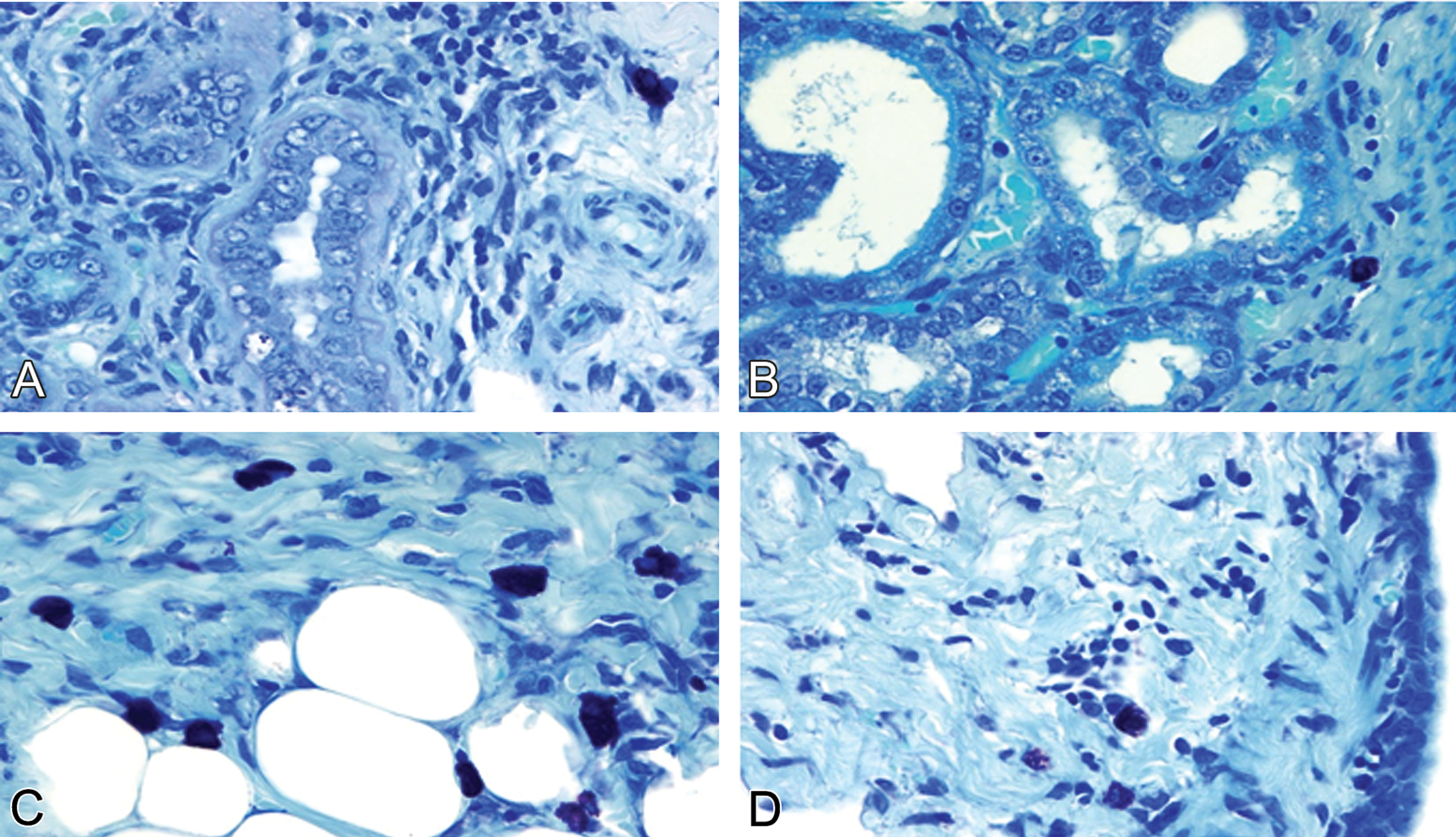
In OVX control females, the mean (SD) number of cardiac MCs (1.41 [0.29]/mm2; Table 4) and the percentage of MC degranulation (0.34 [0.13]/mm2) were significantly higher than these values in control males and normal females (p < .05). Mild MC degranulation was indicated by morphological alterations including rupture of the cell membranes and individual granules outside the cytoplasm. The characteristic cardiac morphology in OVX females indicated cardiac remodeling (fibrosis, apoptosis, and interstitial infiltration with mononuclear cells) and vascular remodeling (endothelial cell hypertrophy and apoptosis of vascular smooth muscle cells; Figures 5 and 6). However, the characteristic features of doxorubicin-induced cardiotoxicity (myofibrillar loss and cytoplasmic vacuoles) were not present and are therefore not included in Table 3.
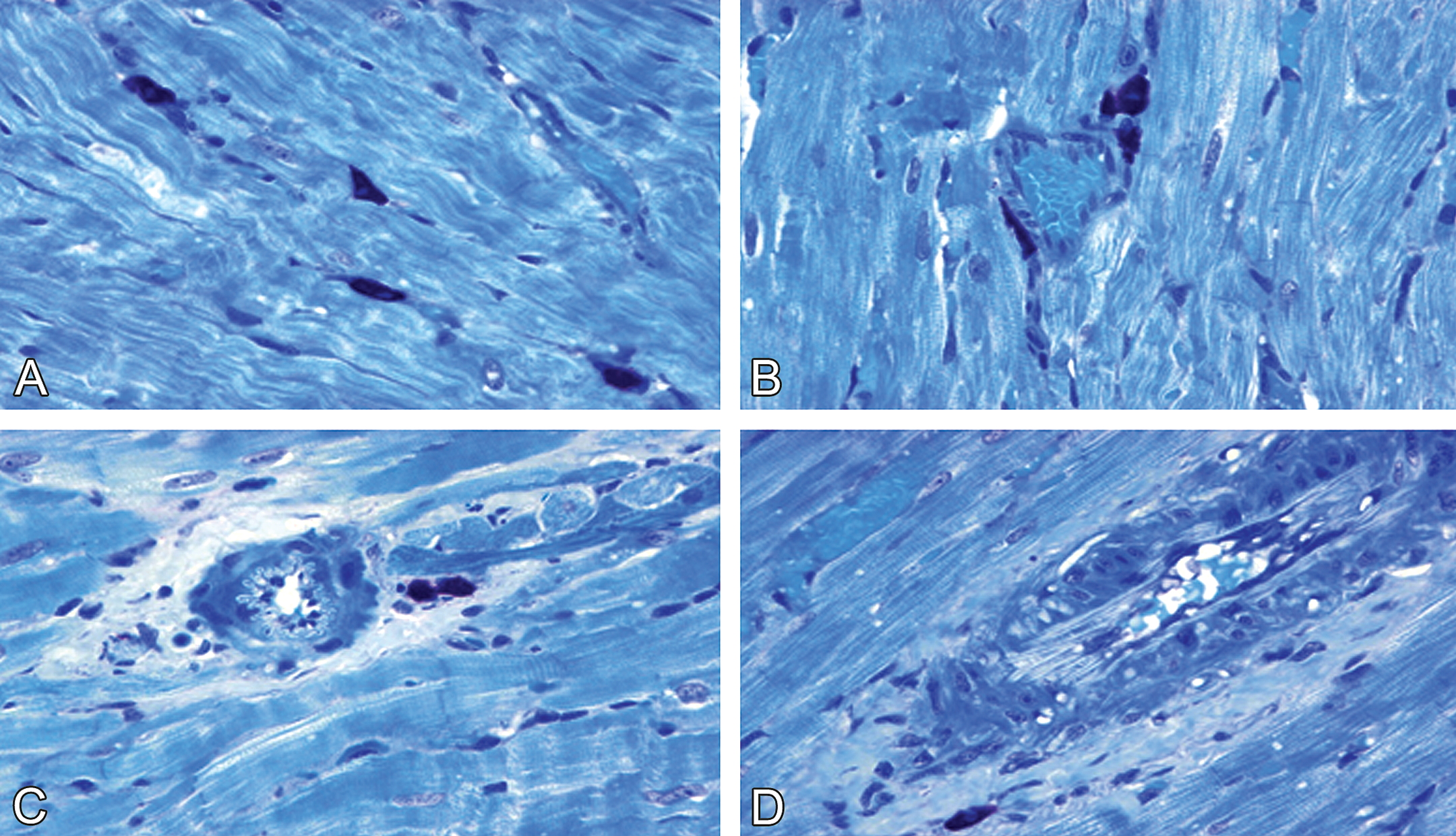
Representative photomicrographs of ovariectomy-induced cardiac and vascular remodeling in saline-treated female rats. (A) Apparent increase in numbers of cardiac mast cells in the interstitium. (B) Remarkable increase of cardiac mast cell numbers in the perivascular space of a small vein. (C) Cardiac mast cell degranulation in association with perivascular fibrosis and the wall thickening of an arteriole, and (D) a small artery. (A–D) Toluidine blue–stained plastic sections, all images are ×630.
Representative photomicrographs of ovariectomy-induced cardiac and vascular remodeling in saline-treated female rats. Cardiac mast cell degranulation in association with (A and B) perivascular fibrosis and the wall thickening of medium-sized arteries, (C) cardiomyocyte apoptosis, and (D) increased numbers of interstitial histocytes. (A–D) Toluidine blue–stained plastic sections, all images are ×630.
OVX Female Rats Treated with Doxorubicin
In doxorubicin-treated OVX females, cardiac and vascular remodeling occurred in association with characteristic doxorubicin-induced cardiomyopathy (only the severity of these doxorubicin-induced lesions is summarized in Table 2 and shown in Figures 7 and 8). The degree of change in cardiac MC degranulation was striking: numerous MC granules were diffused to almost of all interstices, particularly in the extended areas of cardiac fibrosis and the perineural areas (Figures 7 and 8). The characteristic doxorubicin-induced injuries (myofibrillar loss and cytoplasmic vacuoles) were almost comparable in severity to those observed in doxorubicin-treated male rats (Figures 7 and 8). All 5 doxorubicin-treated rats had lesion scores of 2 or 3 while none of the control had such lesion severity scores (p < .05; Table 2). Endothelial cell hypertrophy (data not shown), apoptosis, and cytoplasmic vacuoles of vascular smooth muscle cells were observed in the small coronary arteries (Figures 7 and 8), and cardiomyocyte apoptosis and interstitial inflammation were more apparent (Figures 7 and 8).
Representative photomicrographs of exacerbated doxorubicin-related cardiotoxicity and aggrandized doxorubicin-related cardiac mast cell degranulation in ovariectomized female rats. (A–D) Progression of cardiac mast cell degranulation from an individual cell to multiple cells. Interstitial fibrosis increases with the degree of mast cell degranulation. The granules vary in the color, density, and distance from the nuclear of mast cells. (A–D) Toluidine blue–stained plastic sections, all images ×630.
Representative photomicrographs of exacerbated doxorubicin-related cardiotoxicity and aggrandized doxorubicin-related cardiac mast cell degranulation in ovariectomized female rats. (A) Severe cytoplasmic vacuoles and myofibrillar loss in association with intracytoplasmic granules of mast cells (note the very tiny granules in the vacuoles). (B) Mast cell degranulation in association with severe vascular remodeling (smooth muscle cell vacuolization and apoptosis), (C) increased number of apoptotic myocytes, and (D) increased infiltration of cardiac histocytes and pronounced cytoplasmic vacuoles containing individual granules. (A–D) Toluidine blue–stained plastic sections, all images ×630.
Renal MC Degranulation in OVX and Control Female Rats
A combination of MC degranulation along with renal fibrosis, interstitial inflammation, and tubular epithelial cell vacuolization was noted in the kidneys from doxorubicin-treated males (Figures 9 and 10). In doxorubicin-treated normal females, renal MC degranulation occurred in association with mild interstitial fibrosis (Figures 9 and 10). In contrast, both renal MC numbers and degranulation increased in female SHR after ovariectomy alone. Evidence of renal fibrosis and inflammation was also apparent in these kidneys (Figures 9 and 10). These alterations, along with tubular cell injury, were more prevalent in the kidneys from doxorubicin-treated OVX females (Figures 9 and 10).
Representative photomicrographs demonstrating renal mast cell-mediated nephropathy. (A) doxorubicin-treated normal male rats. Renal cell degranulation, tubular epithelial cell vacuolization, interstitial fibrosis, and inflammation are evident (note significant thickening of fibrous tissue surrounding tubules). (B) Doxorubicin-treated normal female rats. Interstitial fibrosis was mild. No significant inflammation and increased fibrous tissue surrounding tubules. (C and D) Saline-treated ovariectomized female rats show increased numbers of activated and degranulated renal mast cells. (A–D) Toluidine blue–stained paraffin-embedded tissue sections, all images ×630.
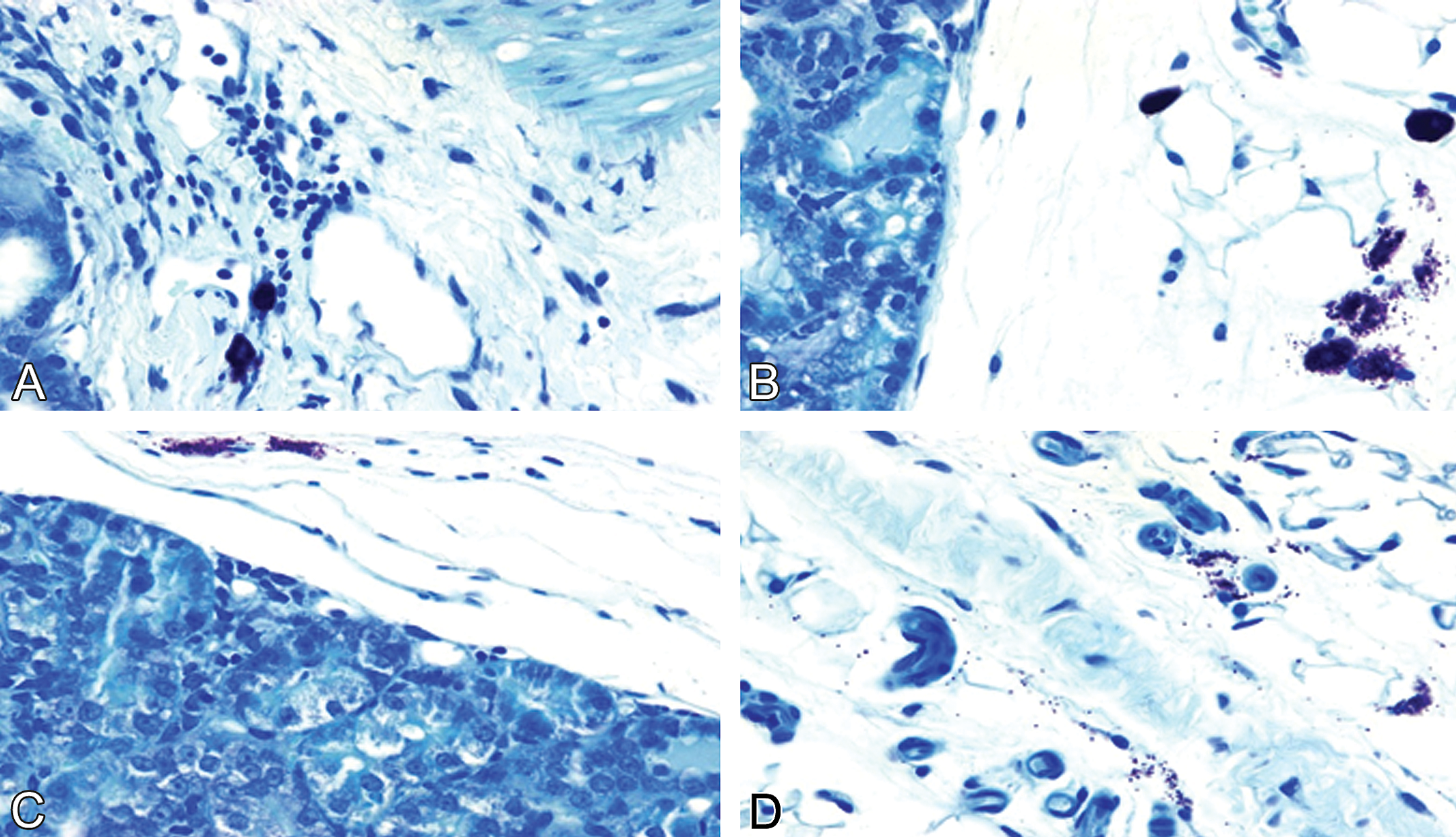
Representative photomicrographs demonstrating renal mast cell–mediated nephropathy. (A–D) Doxorubicin-treated ovariectomized female rats show increased interstitial fibrosis and inflammation in association with high a degree of renal mast cell degranulation. (A–D) Toluidine blue–stained paraffin-embedded tissue sections, all images ×630.
Serum Concentrations of Cardiac Troponin T
Data on the serum levels of cTnT in male and normal female SHR that received saline or doxorubicin are summarized in the right column of Table 2. Cardiac troponin T concentrations were increased in all doxorubicin-treated male and normal animals. In each experiment, these values were significantly higher in doxorubicin-treated males than doxorubicin-treated normal females (p < .05). The higher concentrations of cTnT in doxorubicin-treated males were associated with the more severe myocardial changes observed in these animals. In Experiment 4, serum concentrations of cTnT and myocardial lesion severity scores were significantly higher in doxorubicin-treated OVX females than in doxorubicin-treated normal females.
Discussion
Sex-related Differences in Doxorubicin-induced Cardiomyopathy
In this study of male and female SHRs, including females with and without ovariectomy, we sought to identify the extent and characteristics of any sex-related differences in susceptibility to the cardiac and other toxic effects of doxorubicin. We evaluated cardiac MCs in rats treated with doxorubicin along with those of nontreated control rats and found that the number of cardiac MCs and the percentage of cardiac MCs undergoing degranulation increased with the severity of cardiomyopathy and the elevation of serum cTnT concentrations. This analysis also indicated that the level of cardiac and renal MC activity in females was less pronounced than in males, a factor that could contribute to the observed sex-related differences in susceptibility to the toxic effects of doxorubicin.
Sex differences in the sensitivity to certain toxic effects of doxorubicin in rats, such as impaired weight gain and nephropathy, have been reported before (Van Hoesel et al. 1986; Julicher et al. 1984, 1988). However, the pathogenesis of such sex-related differences in susceptibility to doxorubicin is not completely understood.
We have reported previously that doxorubicin-damaged cardiomyocytes are associated with increased serum cTnT concentrations in adult male SHRs (Zhang et al. 1996; Herman et al. 1999). The present study confirmed this association in both sexes of SHR but extended these findings to include a reduced level of cardiotoxicity in female SHR. In addition, the present study found that certain clinical signs of toxicity such as increased concentrations of cholesterol, triglycerides, blood urea nitrogen, and creatinine and decreased concentrations of albumin occurred in both sexes of SHR but again were more profound in male than in female SHRs. These findings indicate that distinct sex-related differences in susceptibility to the cardiac and other toxic effects of doxorubicin occur in this rat model.
A potential factor responsible for such sex-related differences may involve dissimilarities in MC activity (e.g., MC activation or degranulation) as noted in the present study. MC degranulation in the heart is an adverse effect that can induce myocyte injury and provoke cardiac hypertrophy (Estensen 1985). Recently, Levick et al. (2011) suggested that the reduced incidence of cardiac disease observed in premenopausal women could at least be partially due to the effects of hormones such as estrogen on cardiac MC function. The concentrations of female sex hormones were not monitored in the present study and thus it is not possible to ascertain whether the presence of estrogen was responsible for modifying MC activation or degranulation and thereby attenuating the severity of doxorubicin-induced cardiac alterations in normal female SHR.
Cardiac dendritic cells function as antigen-presenting cells in tissues such as the heart (Zhang et al. 1993). These cells and along with cardiac MCs may exert comparable biological activity because both are bone marrow derived, are located in the myocardial interstitium, and are activated by exogenous agents (Zhang et al. 1993; Nakano et al. 2009). Doxorubicin increases the numbers of cardiac dendritic cells, as well as CD4 T helper lymphocytes and macrophages in the same SHR model used in the present study (Zhang, Herman, and Ferrans 1993, 1994). In these studies, which included only male SHR, the numbers of cardiac dendritic cells, T helper lymphocytes, and macrophages increased in association with the severity scores of doxorubicin-induced cardiomyopathy. The status of these types of cells was not evaluated in the present study. However, it is conceivable that the sex-related variations in severity of doxorubicin-induced myocardial alterations, observed in the present study, could also involve differences in the activity and in the numbers of antigen-presenting cells (such as cardiac dendritic cells, macrophages) in conjunction with MCs.
Ovariectomy Leads to Cardiac and Vascular Remodeling
We found that in untreated OVX female rats, the mean number of cardiac MCs (1.01/mm2) and the percentage of cell degranulation (0.25/mm2) were significantly greater than those in normal untreated females (0.64/mm2 for cell numbers and 0.12/mm2 for percentage of cell degranulation) and in untreated males (0.68/mm2 for cell numbers and 0.21/mm2 for percentage of cell degranulation). Morphological alterations in the hearts from OVX rats consisted of myocardial fibrosis, cardiomyocyte apoptosis, macrophage infiltration, endothelial cell hypertrophy suggestive of activation, and vascular smooth muscle cell apoptosis. It should be noted that these myocardial changes occurred without shifts in the levels of cTnT. The pathogenic mechanism by which ovariectomy alone causes the changes in myocardial morphology observed in female SHR remains to be clarified but could be related to the effects of estrogen depletion.
Estrogen has long been thought to exert antioxidative effects (Ozgonul et al. 2003). Munoz-Castaneda et al. (2006) have suggested that ovariectomy-induced loss of female sex hormones increases oxidative stress levels of sufficient magnitude to cause myocyte alterations. OVX animals have been reported to have significantly lower concentrations of the antioxidative enzymes glutathione peroxidase, superoxide dismutase, catalase, and nitrite–nitrate concentrations in heart tissue and lower erythrocyte counts than those of normal female rats (Munoz-Castaneda et al. 2006; Ha 2004; Oge et al. 2003). Another indication of enhanced oxidative stress due to ovariectomy is the increase in concentrations of malonaldehyde (Ha 2004). All though such markers were not monitored in the present experiments, the earlier studies suggest that increased oxidative stress, presumably from the depletion of estrogens, may be important in the pathogenesis of OVX-induced cardiovascular remodeling.
Recently, Lu et al. (2102) reported that ovariectomy can also lead to phenotypic changes in MCs, such that cells from females almost mimic the MC phenotype of males. These investigators also reported that in addition to ovariectomy-induced changes in MC phenotype, the resulting decreases in estrogen levels may alter the activity of other estrogen responsive cells that synthesize factors (endothelin-1, stem cell factor, and tumor necrosis factor-α) that participate in activation of MCs, increases in MC density, and induction of myocardial inflammation. Of note, the plasma concentration of 17β-estradiol was significantly lower in OVX rats than in intact female rats (Lu et al. 2012). Thus, myocardial changes in OVX animals may be initiated by changes in MC phenotype and in the activity of other immune responsive cells induced by reduced concentrations of female sex hormones (Lu et al. 2102).
Ovariectomy Exacerbated Doxorubicin-related Cardiomyopathy in Female Rats
The present study also found that ovariectomy exacerbated doxorubicin-induced cardiotoxicity. Munoz-Castaneda, Montilla, et al. (2005) reported that OVX female Wistar rats exposed to doxorubicin had greater increases in oxidative stress levels (higher lipid peroxides and protein carbonylation), as well greater declines in glutathione peroxidase, superoxide dismutase, and catalase in heart tissue and erythrocytes than in saline-treated OVX rats or in doxorubicin-treated normal Wistar female rats. These investigators also reported that 17 β-estradiol reduced oxidative stress and cardiomyopathy in doxorubicin-treated OVX female rats (Munoz-Castaneda, Montilla, et al. 2005). These observations indicate that the effects of increased oxidative stress resulting from ovariectomy alone may be greatly enhanced by exposure to doxorubicin. It appears that estrogenic hormones can exert cardioprotective activity by increasing the expression of antioxidant defense mechanisms and decreasing the generation of damaging reactive oxygen species (Borras et al. 2003; Satoh et al. 2007).
Age-dependent Doxorubicin-related Cardiomyopathy
The influence of sex and age on the incidence of nontarget-organ toxicity is an important concern in the clinical use of cancer chemotherapeutic agents, such as the anthracyclines. In the present study, the sensitivity of adult rats to the cardiotoxic effects of doxorubicin clearly differed between the sexes. The risk of doxorubicin-induced cardiomyopathy in adults also increases with age. However, children appear to be more susceptible to these cardiac effects than are adults (von Hoff et al. 1979). In contrast to the situation with adult males, young girls are more susceptible to the adverse cardiac effects of doxorubicin than are boys (Lipshultz et al. 1995, 2010).
Why doxorubicin is more cardiotoxic in girls than in boys is unknown. Differences in body fat might affect doxorubicin toxicity by altering the metabolism or volume of distribution (Lipshultz et al. 1995). Children treated with doxorubicin when they were 4 years or younger seem to be especially susceptible to cardiac alterations (Lipshultz et al. 2005). However, body fat differences may not be a primary factor because sexual dimorphism in fat patterning starts later, between the ages of 5 and 7 years (Webster-Gandy, Warren, and Henry 2003). Male–female differences in oxidative stress levels could also explain the observed differential sensitivity to the cardiotoxic effects of doxorubicin (Lipshultz et al. 1995). However, in studies of both humans and SHRs, Sartori-Valinotti et al. (2007) found no consistent sex difference in tissue oxidative stress levels. These studies were of adult humans and 15-week-old adult SHRs; whether the same relationship exists at an earlier age is unknown. The use of only adult SHR also precluded evaluation of age in the present study.
Estrogen concentrations and MC density can vary with age. Rakusan et al. (1990) reported age-related differences in MC density in male Sprague-Dawley rats (the lowest density being in the hearts of the youngest rats). Shiota et al. (2003) evaluated cardiac MCs during the entire life span of male SHRs (from birth to 20 months). Cardiac MCs were detected at all ages, but prohypertrophic cytokines and proliferating growth factors varied with age. At the age of 2 weeks (the prehypertensive stage), an increased number of cardiac MCs correlated with the increased expression of c-kit and soluble stem cell factor, and MCs contained tumor necrosis factor-α, and increased nuclear factor kappa-B and interleukin-6. At the age of 12 months (the cardiac hypertrophy stage) and at the age of 20 months (the heart failure stage), the levels of transforming growth factor-β and basic fibroblast growth factor significantly increased, and the hearts displayed increased myocardial fibrosis. Hellstrom and Holmgren (1950) reported the number of MCs in the different portions of the heart obtained from human cardiac specimens is strikingly low up to the age of 2 years. Later, the number sharply rises, and then drops continually with increasing age. These studies included mainly males and thus it was not possible to definitively determine whether sex-related differences in cardiac MC quality and/or quantity existed.
Sex-related Differences in Doxorubicin-related Nephropathy
In rats, the kidney is susceptible to doxorubicin toxicity. Nephrotoxicity, presenting as a nephrotic syndrome, has been reported in rats (Cortes et al. 1975; Herman et al. 1985; Van Hoesel et al. 1984, 1986). In male rats, the extent and severity of nephrotoxicity steadily increased with time and cumulative dose of doxorubicin (Van Hoesel et al. 1984, 1986). However, renal alterations in female rats developed more slowly and were less severe than those of males (Van Hoesel et al. 1984, 1986). We found that in treated rats, nephropathy scores were greater in males than that in females. Renal MC degranulation was also more frequent in treated males than in females. Sakemi et al. (1996, 1997) examined doxorubicin-induced renal alterations in castrated male and OVX female Spague-Dawley rats. Male rats had more severe nephropathy than did female rats. Castration reduced the severity of doxorubicin-induced renal alterations to that in normal female rats, indicating that hormones such as testosterone and estrogen could be important in modulating the sensitivity to doxorubicin-induced nephropathy. Montilla et al. (2000) noted that OVX rats had increased oxidative stress and hyperlipidemia, and exposure to doxorubicin increased this oxidative stress and exacerbated the renal injury. Our results confirm these observations and indicate that greater renal MC degranulation could be important in facilitating the occurrence of doxorubicin-induced renal injury in these animals. Clinically, abnormal MC function has been associated with the development of renal tissue fibrosis and renal deterioration in glomerular diseases (Mack and Rosenkranz 2009).
Conclusions
Our results clearly show that the severity of doxorubicin-induced toxicity in the heart and kidney is influenced by sex. Adult female SHRs had significantly less severe myocardial lesions than did similarly treated male SHRs. These observations were verified both by cardiac pathology and by monitoring serum concentrations of cTnT. Administering doxorubicin to OVX female SHRs caused changes similar to those in male SHRs (significant increases in MC degranulation, in cardiomyopathy and nephropathy scores, and in serum concentrations of cTnT). These findings indicate that ovariectomy-mediated depletion of female sex hormones, possibly by altering cellular oxidative status, can enhance the toxic effects of doxorubicin in tissues, such as the heart and kidney. Hormone-induced alterations of MCs and other types of immune effector cells (e.g. cardiac dendritic cells, macrophages, and lymphocytes) also could be an important component of sex-related differences in susceptibility to the toxic effects of doxorubicin.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
