Abstract
When 100 mg/kg/day of di(n-butyl) phthalate (DBP) was intragastrically administered to pregnant Sprague-Dawley rats throughout gestation days 12 to 21, the male pups had similar body weights with no apparent physical differences (e.g., litter size, sex ratio) compared to that of the vehicle group. However, prominent age-related morphological alterations in the smooth endoplasmic reticulum (sER) of testicular Leydig cells (LCs) were observed once these animals reached puberty. At weeks 5 to 7, the abundant sER with non-dilated cisternae was distributed in LCs. Subsequently, although the number of LCs significantly increased, the amount of sER was significantly decreased at 9 to 14 weeks of age and had disappeared at 17 weeks. In contrast, the number of LCs and the amount of sER in LCs of the lower dose groups (10, 30, and 50 mg/kg/day) were similar to those of the vehicle group. Further, serum testosterone levels in the 100 mg/kg dose group were significantly lower during 5 to 17 weeks of age. While their luteinizing hormone (LH) level was significantly lower at 5 to 7 weeks of age, it became significantly higher during 9 to 17 weeks. The amount of sER in LCs decreased with age with the increase in LCs proliferation and serum LH levels in rat exposed in utero to DBP in a dose-dependent manner.
Keywords
Introduction
Since the 1930s, phthalate diesters have been widely used as solvents (C1-C2; methyl and ethyl esters), plasticizers (C3-C13), film formers, emulsifying agents, surfactants, and added to adhesives, glues, pharmaceutical enteric coatings, paints, and printing inks. A common phthalate ester, ortho-di-butyl-phthalate (DBP) is found in many over-the-counter personal care products such as nail polish, cosmetics, shampoo, deodorants, perfumes, and hair spray (Occupational Safety and Health Administration [OSHA] 2009). DBP hydrolyzes in vivo (Figure 1) to produce mono-butyl-ester (MBP) which may undergo phase I oxidation to give oxidized mono butyl ester (MBP-O) or become glucuronidated (phase II metabolism) to form (MBP-G). MBP-O has also been shown to undergo α and β oxidation to ultimately produce ortho-phthalic acid (OPA). It has been reported that 90 to 96% of an oral DBP dose is excreted (parent and metabolites) via urine within 48 hr (Hoppin et al. 2002; Alam et al. 2010). Incorporating data from a variety of different research groups, an extensive physiologically based pharmacokinetic model in pregnant and fetal rats has been reported (Clewell et al. 2008). The model successfully predicted levels of MBP and MBP-G in fetal plasma and testes. It has been shown that DBP administered at 100 mg/kg/day or greater during gestation results in higher levels of MBP via saturation of uridine 5′-diphospho-glucuronosyl-transferase (Clewell et al. 2008; Alam et al. 2010).
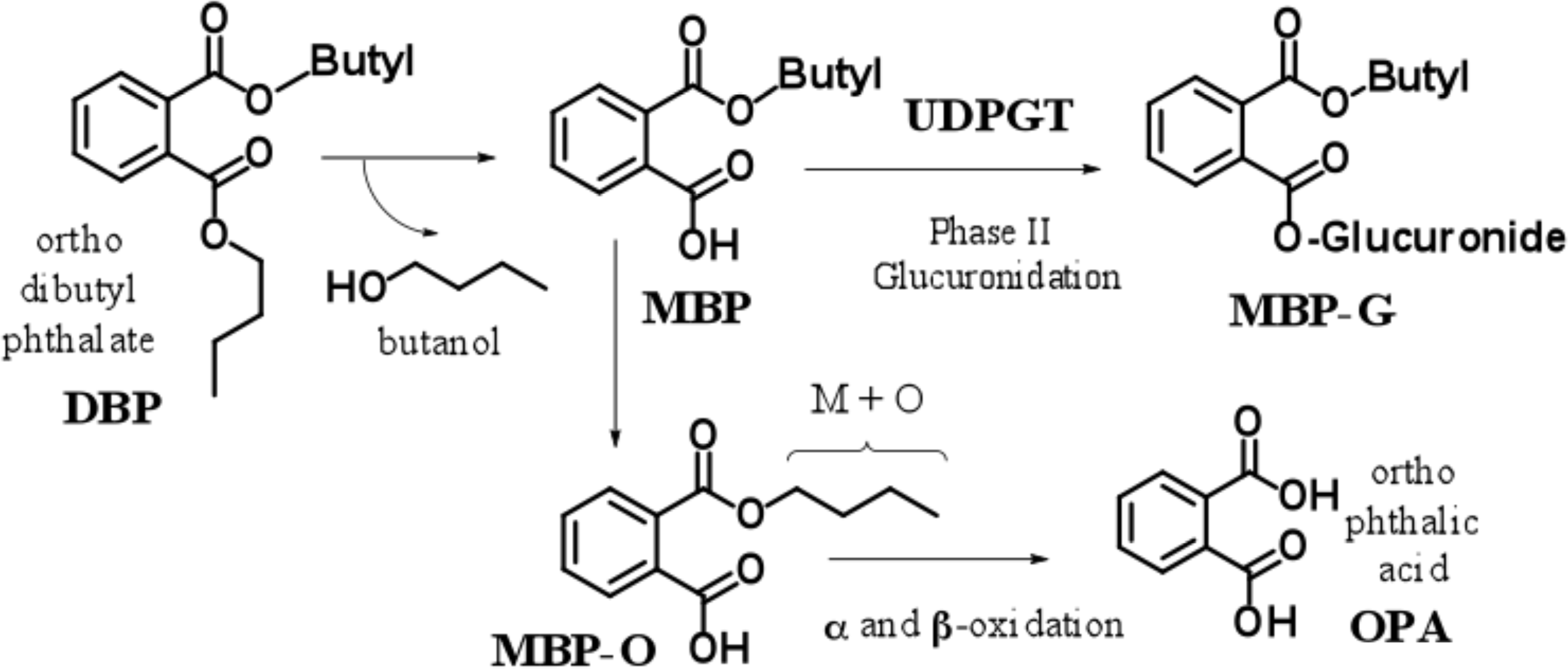
Biotransformation of ortho-dibutyl phtalate.
Therefore, the experimental data appear to support the notion that the metabolite MBP acts as the compound predominantly responsible for observed anti-androgenic effects in vivo. MBP has been shown to inhibit fetal rat testes testosterone (T) production (Parks et al. 2000; Akingbemi et al. 2004). In normal testis, the Leydig cells (LCs) are located between the seminiferous tubules in cell clusters and the shapes of LCs vary from round to elongated or even fusiform. Normal steroidogenic LCs are characterized by a large amount of cystic dilated smooth endoplasmic reticulum (sER) in the cytoplasm (Mylchreest et al. 2002). When male rats are exposed in utero to DBP during the late fetal period, fetal testicular T synthesis decreases with testicular dysgenesis and LCs aggregation (Mylchreest et al. 2002; Barlow and Foster 2003). Exposure of rats in utero to DBP (≥100 mg/kg) has been reported to induce LC hyperplasia in adulthood (Mylchreest et al. 2002; Mahood et al. 2007; Wakui et al. 2012). Hyperplasia differs from hypertrophy in that the adaptive cell change in hypertrophy is due only to increased cellular size, while in hyperplasia the cellular number is increased (Kumar et al. 2010). Although the details are not fully elucidated (Mahood et al. 2007), various reports support that (1) fetal steroid hormones and genes are altered (Howdeshell et al. 2007), (2) the presence of glucocorticoids can amplify these abnormalities (Drake et al. 2009), (3) a correlation between inhibition of the sterol regulatory element-binding protein 2-dependent gene expression pathways exists, and (4) suppression of steroidogenesis by DBP and/or MBP via fetal LCs occurs in primates as well as in rodents (Hallmark et al. 2007). Recently, we reported prominent postnatal changes in LCs of rats following in utero exposure to DBP. They included the disappearance of sER with LC hyperplasia in adulthood by light and electron microscope observation (Wakui et al. 2012). However, age-related morphological changes in sER of LCs following in utero exposure of male rats to DBP have not been previously described.
Materials and Methods
DBP (99.8% pure) was purchased from Aldrich Chemical Co. (Milwaukee, WI). Eight-week-old time-mated female Sprague-Dawley rats (n = 20) were procured from SRL Co. (Shizuoka, Japan) on gestation day 0; the day of copulation was confirmed. Upon arrival, animals were distributed into dose groups using body weight randomization. Rats were individually housed in polycarbonate cages containing wood chip bedding in a high efficiency particulate air (HEPA)-filtered, mass-air-displacement room maintained on a 12-hr light–dark cycle at approximately 22 ± 2 °C with a relative humidity of 55 ± 5%. Animals were fed a conventional diet and had free access to food and water (MF, Oriental Yeast, Osaka, Japan). All experimental procedures were conducted under approval of the Animal Care and Use Committee approval at Azabu University School of Veterinary Medicine; medical guidelines established by National Institutes of Health and Public Health Service Policy on the Humane Use and Care of Laboratory Animals were followed. Five groups of pregnant rats (n = 4 per group) were intragastrically (i.g.) administered DBP in ∼0.5 ml corn oil (Nacalai Tesque Inc., Osaka, Japan)/animal at 0 (vehicle group) mg, 10 mg, 30 mg, 50 mg, or 100 mg/kg/day on gestation days 12 to 21. Dose solutions were prepared fresh every morning and administered at 9:00 am. The regimen was based upon previous studies that illustrated adverse effects of DBP on fetal male rats at 100 mg/kg/day (Mylchreest et al. 1999; Barlow and Foster, 2003). Offspring were weighed and sexed at birth. Litters were reduced to leave 8 offspring, 4 males and 4 females per dam, with a mother not given DBP or corn oil. Weaning was carried out at 21 days postpartum, and pups were then removed from mothers. Offspring were housed in polycarbonate cages (n = 4 per cage; single sex) with wood chips as bedding that was replaced every 5 days. All animals were weighed at birth and again at 5, 7, 9, 14, and 17 weeks of age. For each time point, one male from each dose group was randomly selected, weighed, anesthetized, and euthanized via CO2 overdose. The testes were removed, weighed, and representative samples fixed in 0.1 M phosphate buffered containing 1.2% glutaraldehyde (2 hr); post fixed in 1.0% osmium tetroxide (2 hr). After dehydration in graded alcohols, specimens were embedded in Epon 812 (TAAB, Berkshire, UK). Sections (1 µm) were cut using a Porter-Blum MT-IIb ultramicrotome and stained with methylene blue. To count LCs, light microscope images were captured on a 40× objective on an Olympus BX53 microscope with CCD attachment DP73 (Olympus Co., Tokyo, Japan). Because LCs are located within the interstitial tissue areas without seminiferous tubules, the number of LCs per unit square of interstitial tissue areas (104 µm2) were calculated using Image-Pro Plus v.7.0.1 (Media Cybernetics, CA) by examining 50 randomly selected sites in each group to avoid a sampling bias. Then, for the ultrastructural study, thin sections were cut on a Porter-Blum MT-IIb ultramicrotome and mounted on formvar (polyvinyl formal)-coated slit grids. After double staining with uranyl acetate and lead citrate, sections were inspected using an electron microscope (H500H, Hitachi, Tokyo, Japan). Because sER is an amorphous organelle, a confirmed analytical method for quantifying sER has not been established. In this study, to semiquantify as amount of sER, the unit circles (1 µm diameter) were overlaid to avoid overlap, for each sER mass in 40 electron micrographs of the cytoplasmic regions in LCs at 15,000× magnification were randomly taken from each group to avoid a sampling bias. The number of unit circles per unit square (5 µm × 5 µm) were used to quantify the relative amounts of sER in LCs (Figure 5 and 6). Serum testosterone (T) and luteinizing hormone (LH) levels were measured by radio immunoassays as previously described (Fisher et al. 2003; Wakui et al. 2007). For each set of mean values, standard deviation and standard error of means were computed and compared via Scheffé’s F-test (p < .05) using Stat View-J 5.0 v.5.0 (Abacus Concepts, Berkeley, CA).
Results
Compared to the vehicle group, no statistical differences in average body weight (g), litter size, pup survival, or sex ratio for all four dose groups, 10 mg, 30 mg, 50 mg, and 100 mg DBP/kg/day, were observed (data not shown). At postbirth weeks 5, 7, 9, 14, and 17, testicular weights for the 10 mg, 30 mg, and 50 mg/kg dose groups were similar to that for the vehicle group. However, the 100 mg/kg dose group displayed a statistically significant decrease in testicular weight after 9 weeks of age (Figure 2). Light microscopic observations revealed that the number of LCs in the 100 mg/kg dose group was significantly higher than that of the vehicle group at 9, 14, and 17 weeks; all other dose groups at all ages were similar to that of the vehicle group (Figure 2). Because electron microscopic observations of normal rat LCs have been previously described, an in-depth description will not be given (Russell and Burguet 1977; Jones et al. 1993). In general, normal LCs were centrally placed with a round to ovoid nucleus containing marginal heterochromatin, and abundantly dominated by sER; they were diffusely isolated, with dilated cisternae, and distributed throughout the cytoplasm (Figures 3 and 4). Compared to the vehicle group, distinctive morphological changes were observed in the 100 mg/kg dose group. At 5 to 7 weeks of age (i.e., puberty), the 100 mg/kg dose group had abundant sER with a complicated arrangement of non-dilated cisternae and little stripped rough endoplasmic reticulum (Figures 3 and 4). The amount of sER in LCs was significantly decreased at 9 to 14 weeks of age, and sER were not observed at 17 weeks of age (Figures 3 –6). However, mitochondrial swelling was not encountered, and the Golgi apparatus displayed no observable change (Figures 3 –5). From 5 to 17 weeks of age, the 10 mg, 30 mg, and 50 mg/kg dose groups exhibited T and LH levels similar to that of the vehicle group (Figure 7), whereas the 100 mg/kg dose group had significantly lower T levels (Figure 7). The LH level of the 100 mg/kg dose group was significantly lower at 5 and 7 weeks of age and significantly higher at 9 to 17 weeks of age compared to that of the vehicle group (Figure 7).
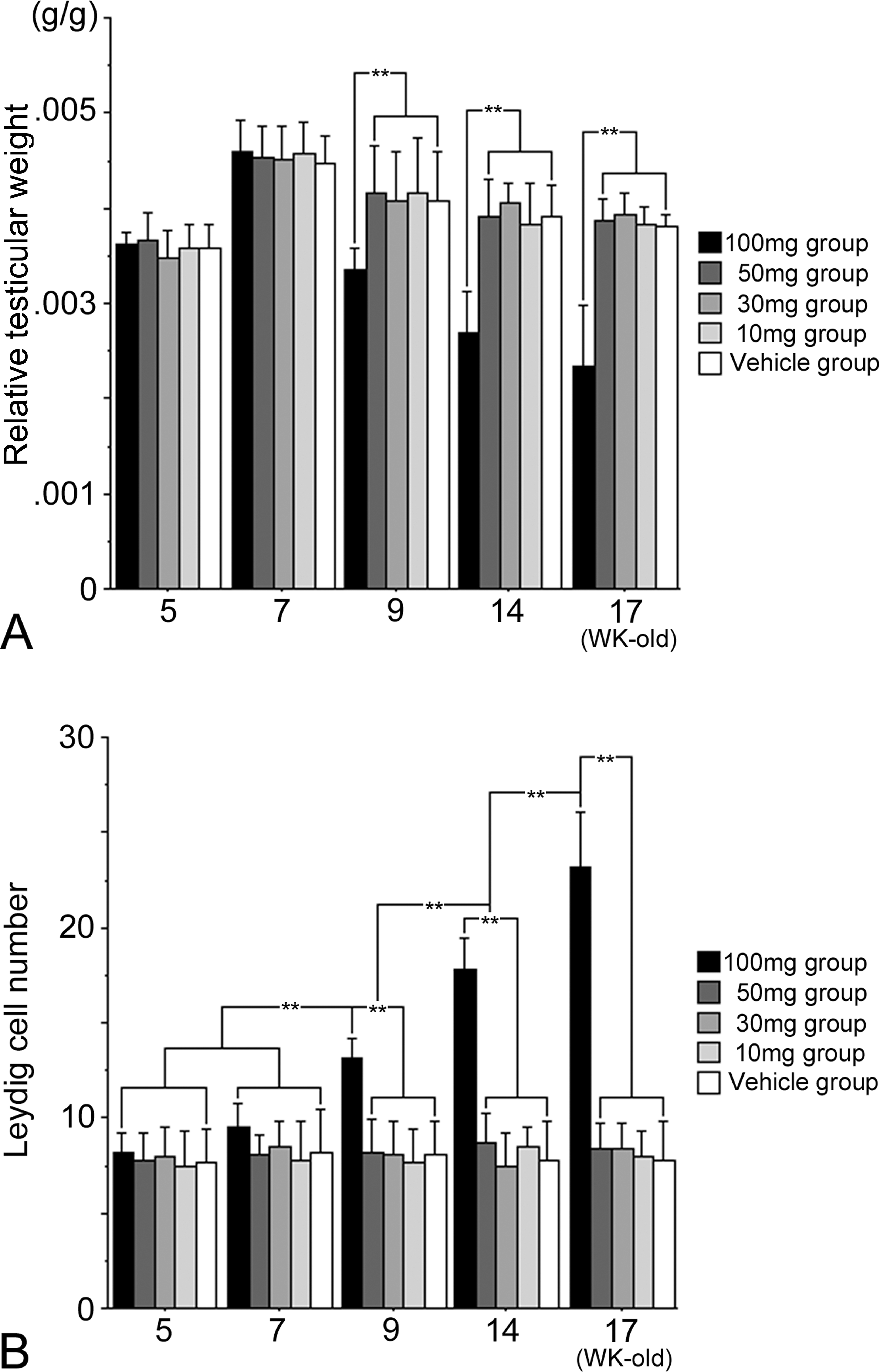
(A). Relative testicular weight (testicular weight/body weight) and (B). Leydig cell number per unit square (104 µm2) for rats (5–17 weeks old) exposed in utero to DBP or corn oil/kg/day throughout gestation days 12 to 21. Mean ± SEM (standard error of mean) for 4 animals per group. **p < .05.
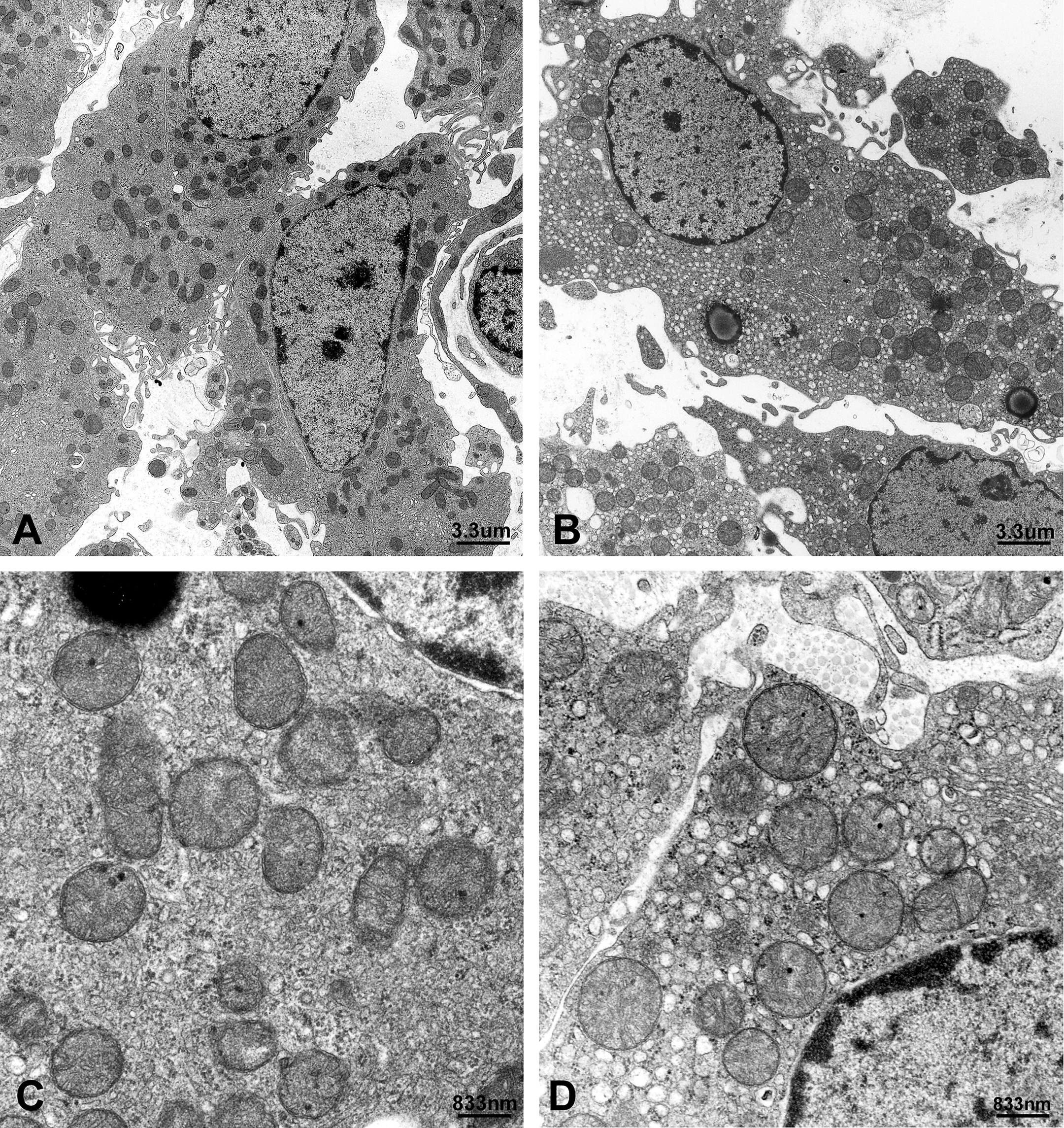
Representative electron micrographs of Leydig cells stained with uranyl acetate and lead citrate. (A). Seven-week-old rats exposed in utero to 100 mg DBP/kg/day or (B). corn oil as the vehicle throughout gestation days 12 to 21. Bar = 3.3 µm. (C) High power view of (A) displaying abundant and complicated arrangements of smooth endoplasmic reticulum (sER) with abundant non-dilated cisternae. (D) High power view of (B) illustrating abundant sER with dilated cisternae. Bar = 833 nm.
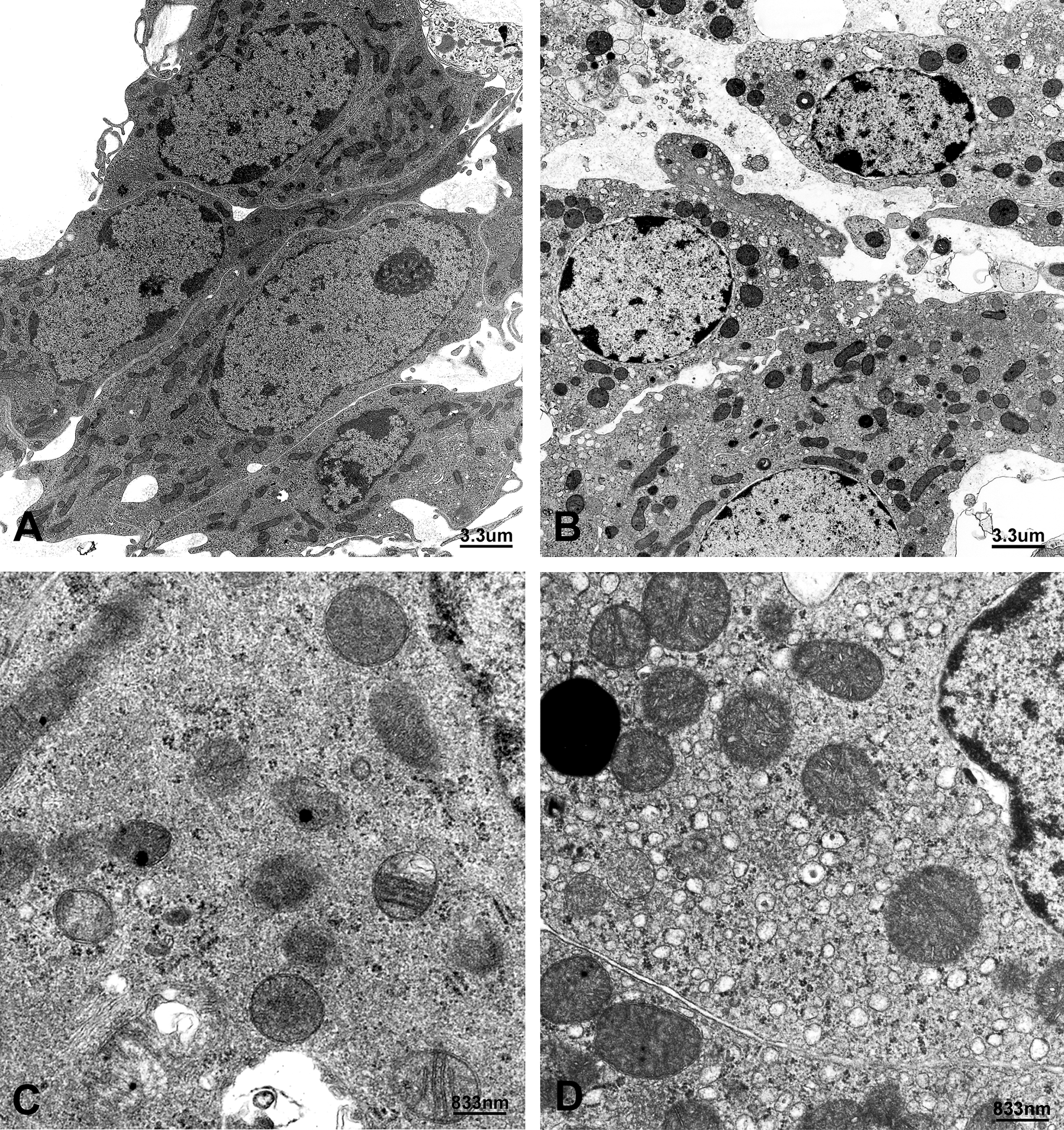
Representative electron micrographs of Leydig cells stained with uranyl acetate and lead citrate. (A). Seventeen-week-old rats exposed in utero to 100 mg DBP/kg/day or (B). corn oil throughout gestation days 12 to 21. Bar = 3.3 µm. (C). High-power view of (A) displaying amorphous materials, free ribosomes, and little stripped rough endoplasmic reticulum. (D). High-power view of (B) showing abundant smooth endoplasmic reticulum with dilated cisternae. Bar = 833 nm.
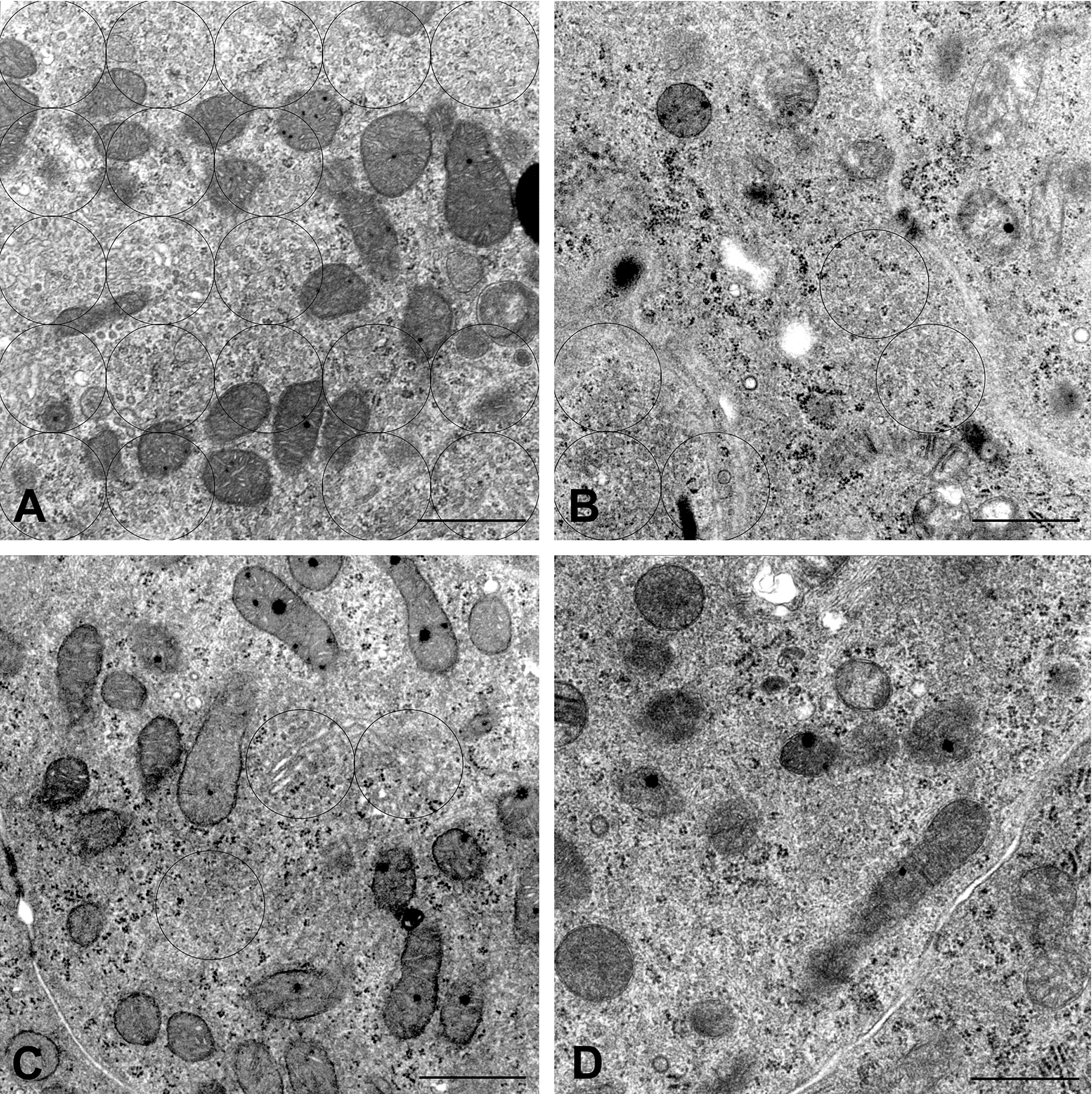
Representative electron micrographs used to calculate the relative amounts of smooth endoplasmic reticulum (sER) in cytoplasmic regions of Leydig cells in rats exposed in utero to 100 mg DBP/kg/day throughout gestation days 12 to 21. The 1-µm diameter circles (black line) were overlaid on the sER masses. (A). At 7 weeks, (B). 9 weeks, (C). 14 weeks, and (D). 17 weeks of age. Uranyl acetate and lead citrate stain; bar = 1 µm.
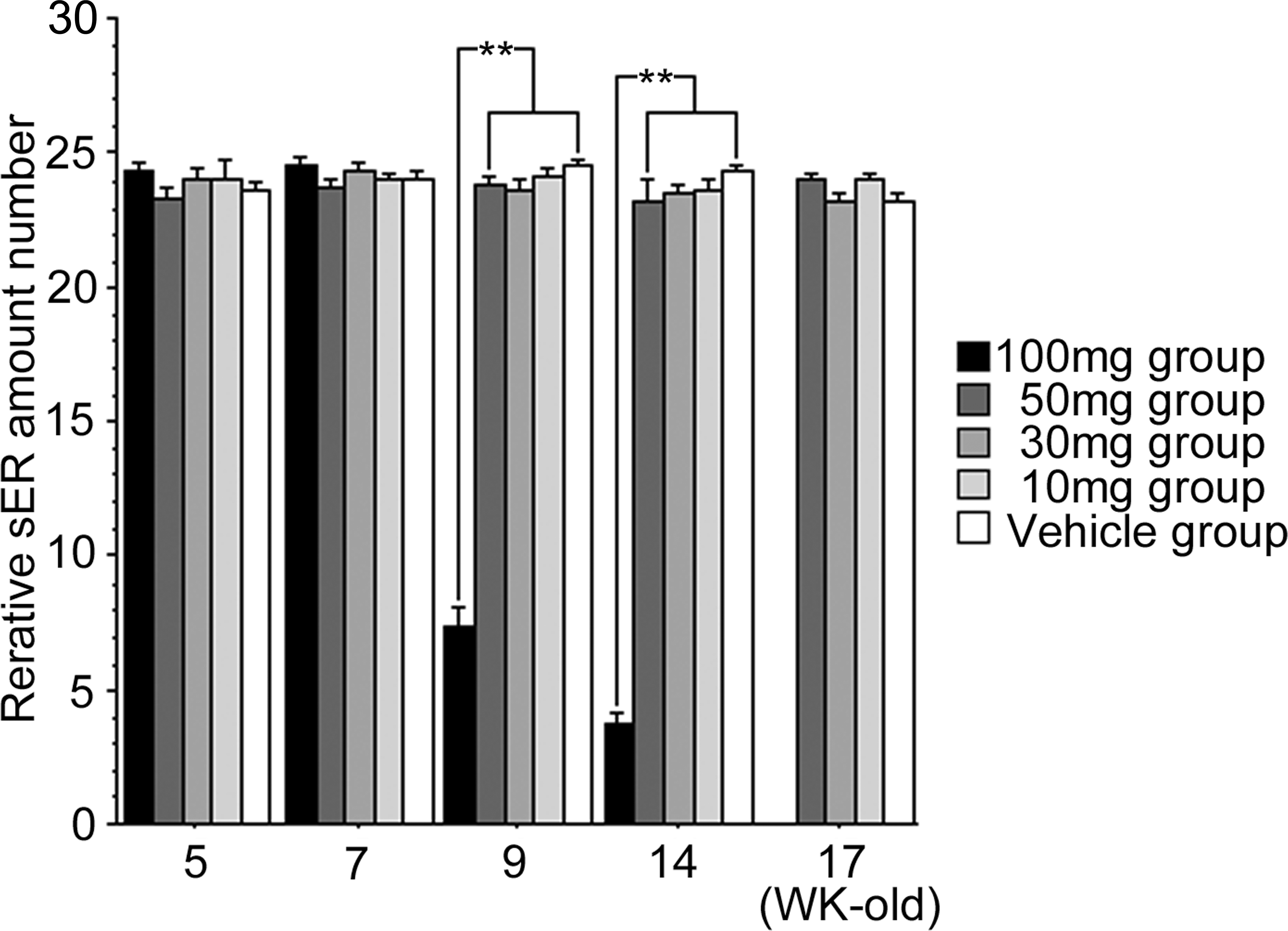
Relative amounts of smooth endoplasmic reticulum (sER) in Leydig cells from 5 to 17 weeks old rats exposed in utero to DBP/kg/day or corn oil (vehicle group) throughout gestation days 12 to 21. Mean ± SEM for 4 animals per group. **p < .05.
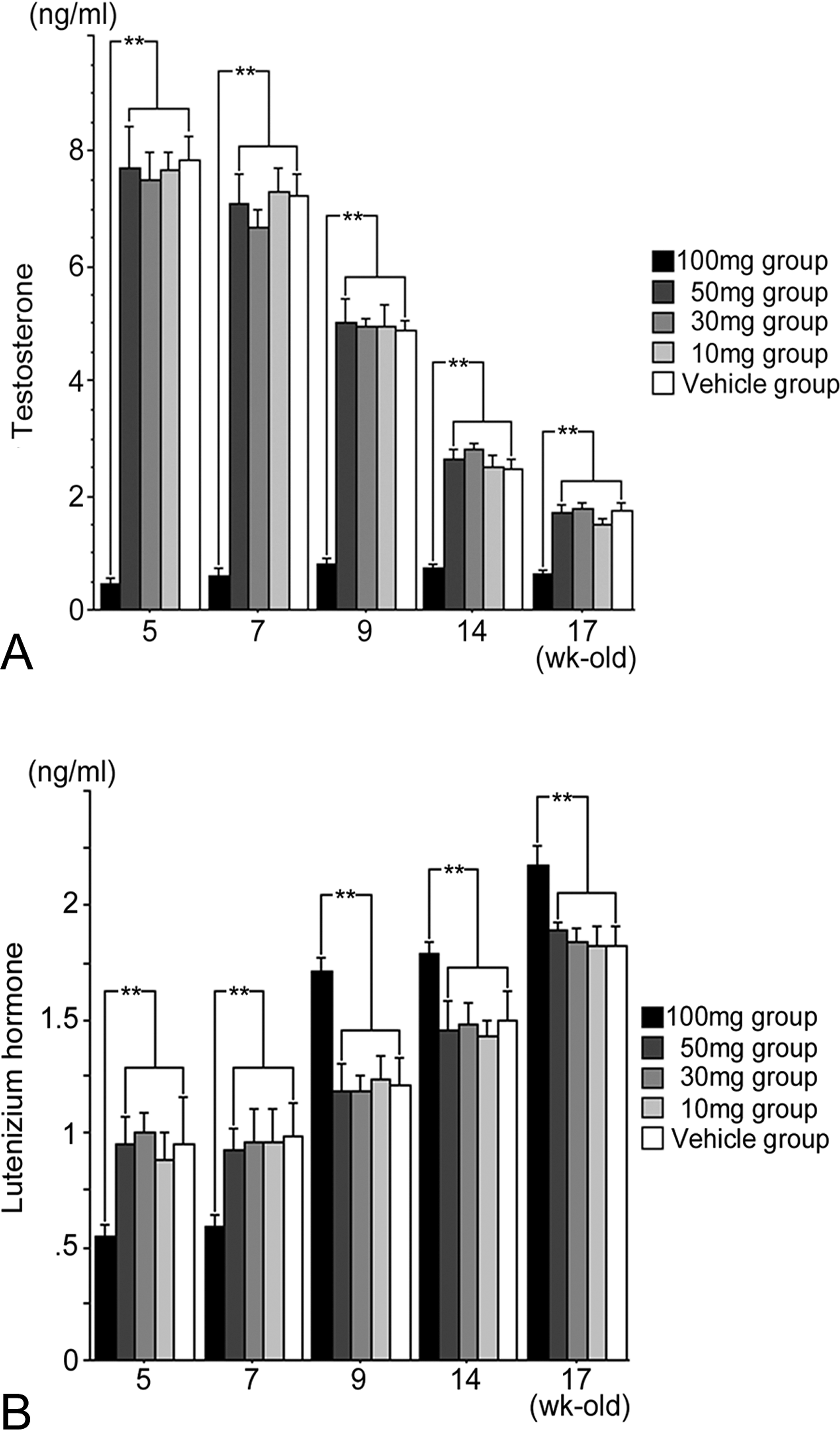
(A). Testicular testosterone (ng/ml) and (B). luteinizing hormone (ng/ml) levels from rats (5–17 weeks old) exposed in utero to DBP/kg/day or corn oil (vehicle group) throughout gestation days 12 to 21. Mean ± SEM for 4 animals per group. **p < .05.
Discussion
Postnatal exposure of male rats to high levels of DBP (≥100 mg/kg) induces testicular dysgenesis resulting in increased numbers of LCs; unchanged and decreased testicular steroidogenesis have also been reported (Kim et al. 2004; Alam et al. 2010). In addition, ultrastructural studies showed that treating rats (at 6 to 8 weeks old) i.g. with ortho-phthalates—di(2-ethylhexyl) phthalate, di-n-pentyl phthalate, di-n-octyl phthalate, or diethyl phthalate—for two days induced vesiculation and dilation of sER in LCs, while the overall sER quantity was unchanged (Jones et al. 1993). In this study, DBP given i.g. daily during gestation days 12 to 21 induced LC hyperplasia without hypertrophy in the highest dose group (100 mg/kg) at 9 weeks of age; they had non-dilated cisternal sER and were morphologically deficit in steroidogenesis. Upon reaching adulthood (i.e., 9 weeks post birth), the amount of sER in LCs of the 100 mg/kg dose group exhibited a gradual age-related decrease that disappeared by 17 weeks of age. Hence, these data showed time-dependent morphological effects on sER in LCs of rats following in utero DBP exposure.
Orally administered DBP is rapidly metabolized and excreted in urine within 48 hr (Hoppin et al. 2002; Alam et al. 2010). It has been suggested that in utero DBP exposure directly affects fetal rat LCs (Mylchreest et al. 2002; Barlow and Foster, 2003). Previous rat studies have reported that LC numbers were unchanged during the juvenile to pubertal periods, but significantly increased after 90 days of age. Some studies have proposed a compensatory reaction to testicular steroidogenesis triggered by T insufficiency (Mylchreest et al. 2002; Barlow and Foster, 2003; Hu et al. 2009). In the present study, the male pups receiving 100 mg DBP/kg from gestation days 12 to 21 exhibited a significant decrease in T levels even at 5 to 17 weeks of age, but showed a significant increase in the number of LCs after 9 weeks old, and the amount of sER correlated with the increased LH levels. Human studies have suggested that LC hyperplasia occurred in patients due to lower serum T values and higher LH levels (Booth et al. 1987; Holm et al. 2003).
In conclusion, the present study revealed that administration of 100 mg DBP/kg/day to male rats in utero from days 12 to 21 of gestation was associated with abundant sER in non-dilated cisternae in LCs at puberty and significantly decreased sER in LCs in adulthood (i.e., after 9 weeks of age). These experimental observations appeared to be linked to low T and high LH levels. While the complete molecular mechanisms and apparently dose-dependent effects were not fully elucidated, links between the alteration of sER and decrease ability to synthesized T with increased receptivity to LH warrant additional research.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported via a research project grant awarded by the Azabu University Research Services Division and the Promotion and Mutual Aid Corporation for Private Schools of Japan; a Grant-in-Aid for Matching Funds Subsidy for Private Universities provided funding for this project. This study was also supported by Grant-in-Aid (C) #21580371 from the Ministry of Education, Culture, Sports, Science and Technology of Japan..
Abbreviations
Acknowledgment
The authors thank Dr. Katherine Ono for critical reading and editing the article.
