Abstract
The present study describes atypical Leydig cell (LC) hyperplasia in 20-week-old Sprague-Dawley rats with low testosterone and high luteinizing hormone levels after prenatal administration of 100 mg/kg/day di(n-butyl) phthalate on days 12 to 21 postconception. Light microscopy revealed LC hyperplasia surrounded by severely degenerated seminiferous tubules. Aggregated LCs had large ovoid nuclei with nucleoli and abundant eosinophilic cytoplasm. Immunohistochemical analysis showed expression of proliferating cell nuclear antigen and vimentin in many hyperplastic LCs. Electron microscopy revealed atypical nuclei, abundant free ribosomes, stripped rough endoplasmic reticulum, intermediate-size filaments, elongated cytoplasmic filopodia, atypical tight junctions, and cilia formations, but smooth endoplasmic reticulum was scarcely observed.
Introduction
A number of uses of di(n-butyl) phthalate (DBP) have been identified including the manufacture of plastics, paints, wood varnishes, and lacquers, used in cosmetics, medical supplies, textiles, propellants, food packaging, dental materials, paper, and foods (OSHA 2009). Although the concentration of DBP in manufactures show a large variability, some studies have described that in polyvinyl chloride the concentration is 8.5% (Kawakami, Isama1, and Matsuoka 2011) and that in cosmetics it is 5.98% (Hubinger et al. 2006). The exposure to phthalate can produce a variety of effects in laboratory animal, but the adverse effects on the development of the testis are the most serious (Mylchreest et al. 2002).
However, after oral administration of DBP to rats, 90 to 96% of administered dose is excreted in urine within 48 hr (Hoppin et al. 2002), but the prenatal exposed DBP exhibits abnormal gonocytes development (Alam et al. 2010). Moreover, prenatal DBP exposures induce testicular dysgenesis in adult rat offspring, but this testicular toxicity is not considered to be the direct influence of prenatal exposed DBP and/or its metabolites (Mylchreest et al. 2002). It has been suggested that Leydig cell (LC) hyperplasia might be an important event in the development of testicular dysgenesis in male animals exposed to DBP (Mylchreest et al. 2002; Barlow, Phillips, and Foster 2004). When male rats are exposed to DBP in utero, it acts as an anti-androgen by decreasing testicular testosterone (T) synthesis, and LC hyperplasia develops in adulthood (Mylchreest et al. 2002; Barlow, Phillips, and Foster 2004; Mahood et al. 2005). Although postnatal exposure to phthalate esters other than DBP has been reported to describe the smooth endoplasmic reticulum (sER) vesiculation and dilation in rat LCs (Jones et al.1993), the detailed morphology of LC hyperplasia induced by prenatal exposure of rats to DBP is unclear.
Materials and Methods
Four female and two male 6-week-old Sprague-Dawley (SD) rats (Japan SLC, Shizuoka, Japan) were housed in a plastic cage on hardwood-chip bedding in a High Efficiency Particulate Air Filter (HEPA) -filtered, mass-air-displacement room with a 12-hr light/12-hr dark cycle at 22 ± 2 °C and 55% ± 5% relative humidity and fed a conventional diet (MF, Oriental Yeast, Osaka, Japan) and water ad libitum. All experimental procedures were conducted with the approval of the Animal Care and Use Committee of the Azabu University School of Veterinary Medicine. Guidelines set by the National Institutes of Health and Public Health Service Policy on the Humane Use and Care of Laboratory Animals were followed at all times. At 7 weeks of age, the rats were housed with 2 females and 1 male per plastic cage. Four pregnant rats were intragastrically administered 100 mg/kg body weight DBP (Aldrich Chemical Co., Milwaukee, WI) in corn oil (Nacalai Tesque Inc., Osaka, Japan) or an equivalent volume of corn oil (~0.5 ml/animal, i.g.) on days 12 through 21 postconception. The dose of phthalate was based on several previous studies that reported observation of adverse effects on fetal rats at a dosage of 100 mg/kg/day (Mylchreest et al. 2002; Barlow, Phillips, and Foster 2004). The offspring were sexed at birth, and litters were reduced so that each dam was left with 10 offspring (5 males and 5 females/dam). Weaning was carried out on day 21 postpartum. At 20 weeks of age, male offspring of DBP-treated and vehicle groups were anesthetized and euthanized by a CO2 overdose. The testes were removed and weighed. For light microscopic observation, the testes were fixed in Bouin’s solution for 24 hr, and 4-µm paraffin sections were cut and stained with hematoxylin and eosin (H&E).
Immunohistochemical expressions of proliferating cell nuclear antigen (PCNA) and vimentin were reacted with an anti-PCNA monoclonal antibody (Novocastra Laboratories, Newcastle, UK; diluted 1:50) and an anti-vimentin monoclonal antibody (Dako, Hamburg, Germany; diluted 1:50) using the avidin–biotin complex (ABC) method, and the sections were then counterstained with Mayer’s hematoxylin. As a negative control, nonimmunized mouse serum was substituted for the primary antibody.
Other representative testicular samples were fixed in 0.1 M phosphate-buffered 1.2% glutaraldehyde for 2 hr and then postfixed in 1.0% osmium tetroxide for 2 hr. After dehydration in graded alcohols, the specimens were embedded in Epon 812. Thin sections were cut on a Porter-Blum MT-IIb ultramicrotome and stained with methylene blue or mounted on formvar-coated slit grids. After double staining with uranyl acetate and lead citrate, the sections were observed with a Hitachi H-2000 electron microscope.
Testicular T and serum luteinizing hormone (LH) levels were measured by radioimmunoassay as described previously (Mylchreest et al. 2002).
For each set of mean values, standard deviation and standard error of means were calculated and compared by Scheffé’s F-test (p < .05) using the computer statistical analysis system Stat View-J 5.0 (Abacus Concepts Co. Ltd.).
Results
Exposure of pregnant dams to DBP did not affect body weight (g), which was similar to that of the vehicle group at the beginning of the experiment and at day 21 postconception (data not shown). Additionally, no decrease in litter size, change in pup survival rates, alteration of sex ratio, or difference in body weights of male pups was found compared to the vehicle group (data not shown). However, at 20 weeks of age, the testicular size and weight of the DBP group were significantly decreased compared to those of the vehicle group (Figure 1).
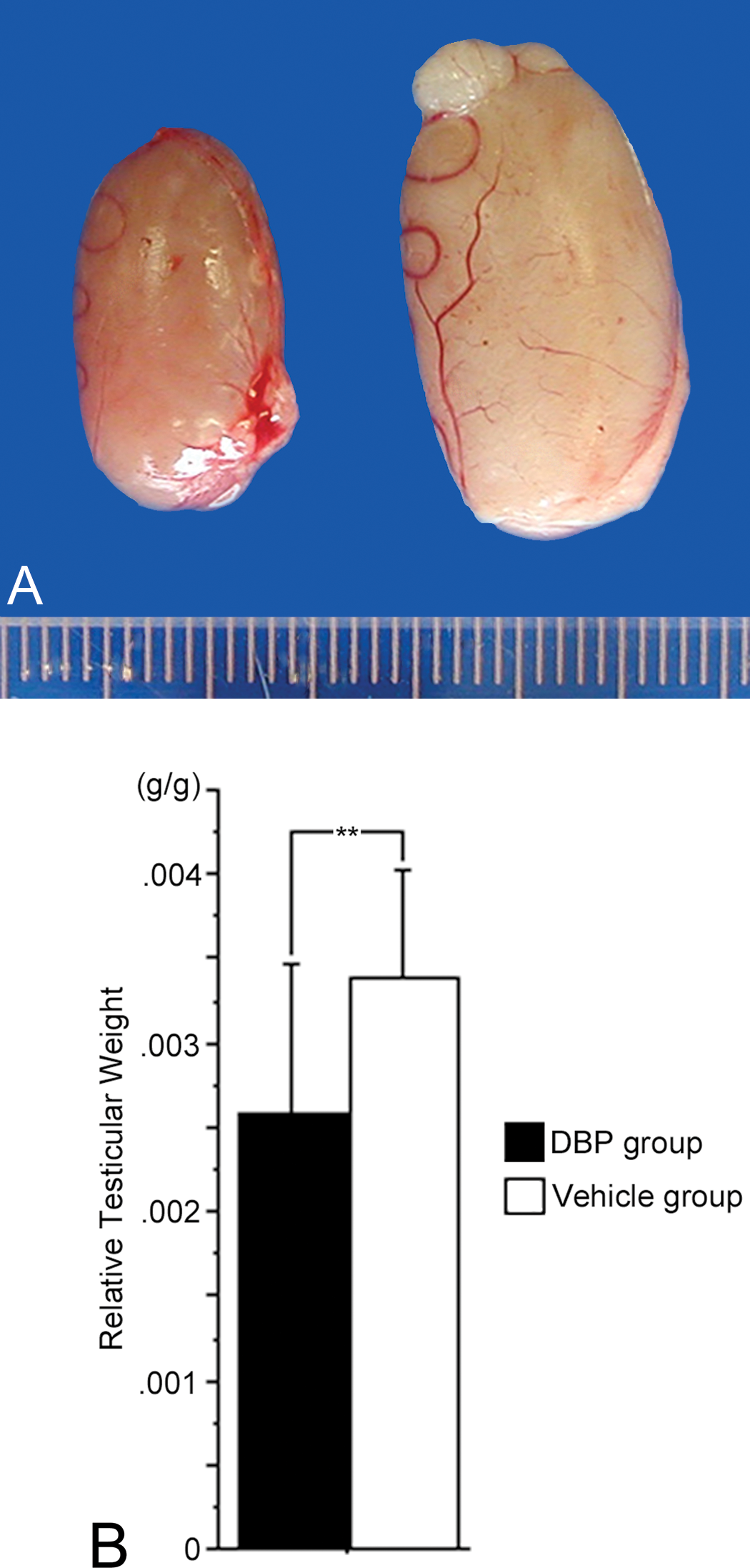
(A) Macroscopic view of testes. A testis of a 20-week-old rat of the DBP group is on the left, and one of the vehicle group is on the right. (B) Average relative testicular weight (testicular weight/body weight) of 20-week-old rats of the DBP group was significantly lower than that of the vehicle group. Relative testicular weight values are mean ± SD for four litters per group. **p < .05.
In the present study, many areas of LC hyperplasia were also observed, although testicular degeneration was not observed in the vehicle group (data not shown). Histological observation revealed the testes of the DBP group showed various degrees of degenerated seminiferous tubules and Sertoli cells lining many tubules, although a minimum of ineffective spermatogenesis occurring in several of the seminiferous tubules was observed (Figure 2). Lesions of LC hyperplasia were not nodular structures but were surrounded by severely degenerated seminiferous tubules. Aggregations of LCs showed large ovoid nuclei with nucleoli and abundant eosinophilic cytoplasm (Figure 2). The LCs comprising hyperplastic lesions were immunohistochemically positive for PCNA and vimentin (Figure 2). These lesions were not, however, categorized by size as LC adenomas (McConnell et al. 1992), consistent with the Society of Toxicologic Pathology (STP) recommendation that the diagnosis of adenoma be made when nodular proliferation of LCs is greater than 3 seminiferous tubules in diameter, while those smaller than that are classified as hyperplasia (McConnell et al. 1992).
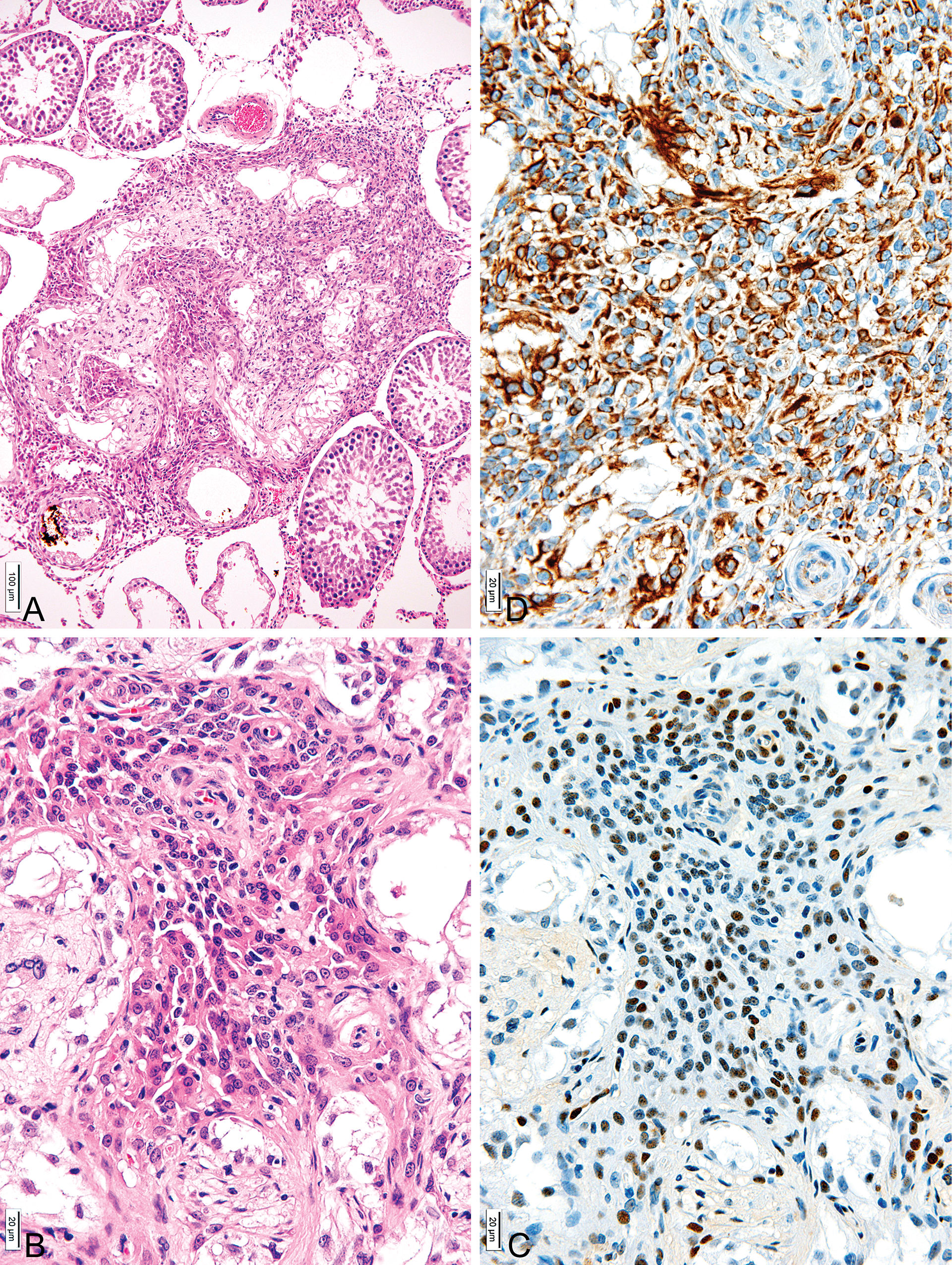
Representative light micrographs of LC hyperplastic lesions of the DBP group. (A) Low power view of lesions of LC hyperplasia without nodular features but surrounded by severely degenerated seminiferous tubules. Ineffective spermatogenesis occurring in some of the seminiferous tubules was also observed. H&E ×90; bar = 100 µm. (B) LC hyperplasias were surrounded by severely degenerated seminiferous tubules. Aggregations of LCs consisted of large ovoid nuclei with nucleoli and abundant eosinophilic cytoplasms. H&E ×350; bar = 20 µm. (C) Immunohistochemical assay for PCNA in a serial section of (A). Cytoplasm of many hyperplastic LCs showed PCNA expression. ABC method ×350; bar = 20 µm. (D) Immunohistochemical assay for vimentin in a serial section of (A). Cytoplasm of many hyperplastic LCs showed vimentin expressions. ABC method ×350; bar = 20 µm.
The ultrastructures of normal rats LCs have been described previously, and therefore an in-depth description will not be given here (Russell and Burguet 1977; Jones et al. 1993). The LCs of the vehicle group at 20 weeks showed centrally placed, round, or ovoid nuclei with marginal heterochromatin and nucleoli (Table 1, Figure 3). The cytoplasm was dominated by abundant sER that isolated diffusely dilated cisterna distributed throughout the cytoplasm, a little rough endoplasmic reticulum (rER), and some microbodies. Well-developed tight junctions were observed (Table 1, Figures 3 and 4). On the other hand, the DBP group showed hyperplastic LCs with atypical nuclei with nucleoli, abundant free ribosomes, some stripped rER, plentiful microfilamentanous components, several bundles of intermediate-sized filaments, many elongated filopodia, atypical tight junctions, and one or two cilia formations, while sER and microbodies were rarely observed (Table 1, Figures 3 and 4).
Comparison of the ultrastructures on testicular Leydig cell with prenatal DBP nd vehicle exposures.

Note. Details are noted on the study; ++, many; +, some; ±, a few; −, none.
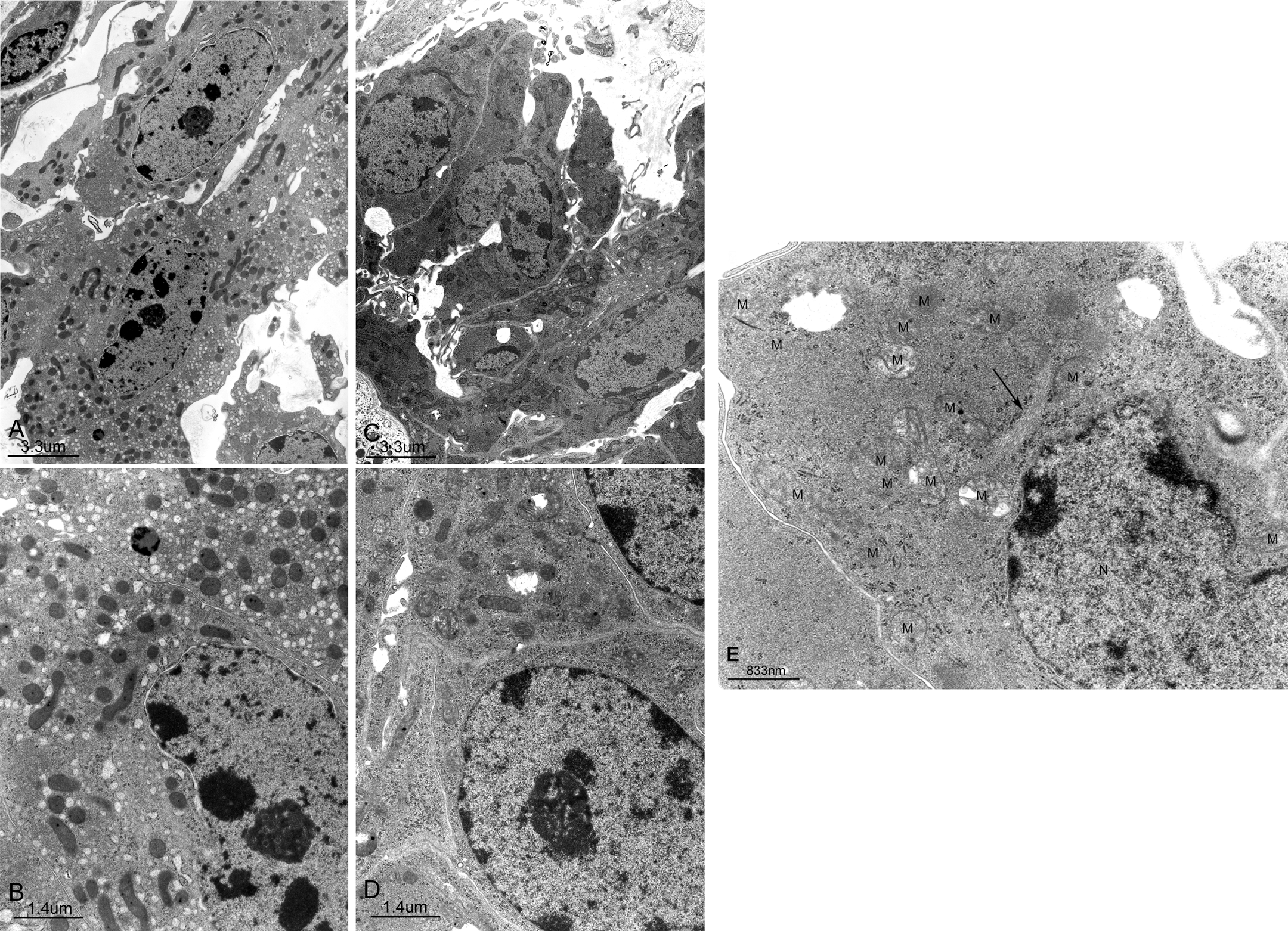
(A,B) Representative electron micrographs of LCs of the vehicle group. Ovoid nuclei, richly developed sER, dilated cisterna, and many mitochondria are observed. (A) ×5,600; bar = 3.3 µm, (B) ×13,000; bar = 1.4 µm. (C,D,E) Representative electron micrographs of LCs of hyperplasic lesion of the DBP group. Atypical nuclei and many mitochondria are observed. (C) ×5,600; bar = 3.3 µm, (D) ×13,000; bar = 1.4 µm. (E) High power view of electron micrographs of cytoplasm of LCs of hyperplasic lesions are occupied by microfilamental components, bundles of intermediated filaments (arrow), many mitochondria (M), stripped rER, and atypical nuclei (N) ×28,000; bar = 833 nm.
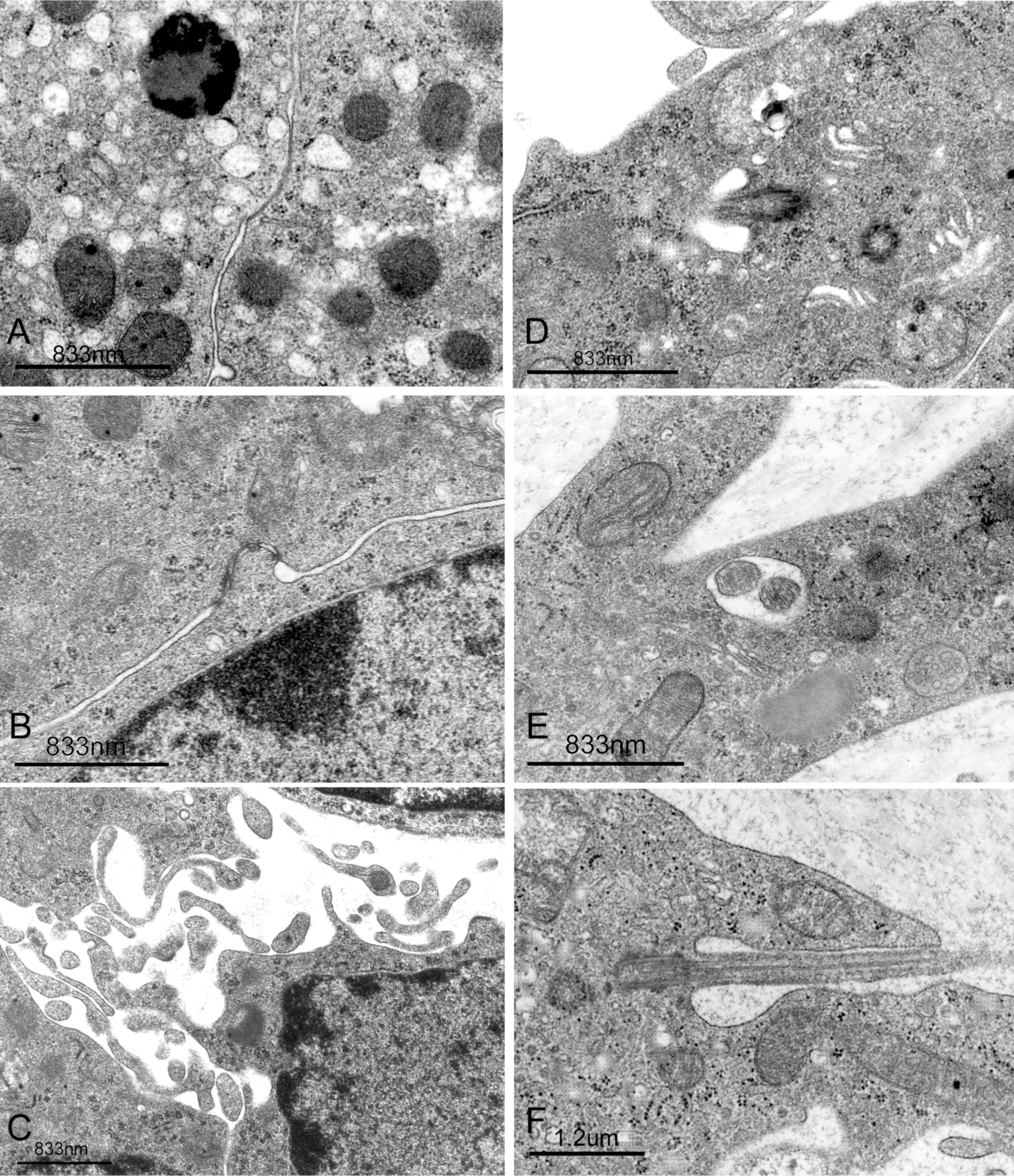
(A) Representative electron micrograph of tight junction (arrow) of LCs of the vehicle group. A typical microbody (arrowhead) is observed; ×28,000; bar = 833 nm. (B) Representative electron micrograph of atypical tight junction (arrow) of LCs of the DBP group; ×28,000; bar = 833 nm. (C) Representative electron micrograph of LCs of the DBP group show many cytoplasmic filopodial projections; ×20,000; bar = 833 nm. (D,E,F) Representative electron micrographs of LCs of the DBP group show cilia formations. (D) Obliquely sectioned cilia (arrow); ×28,000; bar = 833 nm. (E) Cross sections of two cilia (arrow); ×28,000; bar = 833 nm. (F) Longitudinal section of cilia is observed; ×17,500; bar = 1.2 µm.
The testicular T level of the DBP group in this study was significantly lower than that of the vehicle group, and serum LH levels of the DBP group were significantly higher than those of vehicle group (Figure 5).
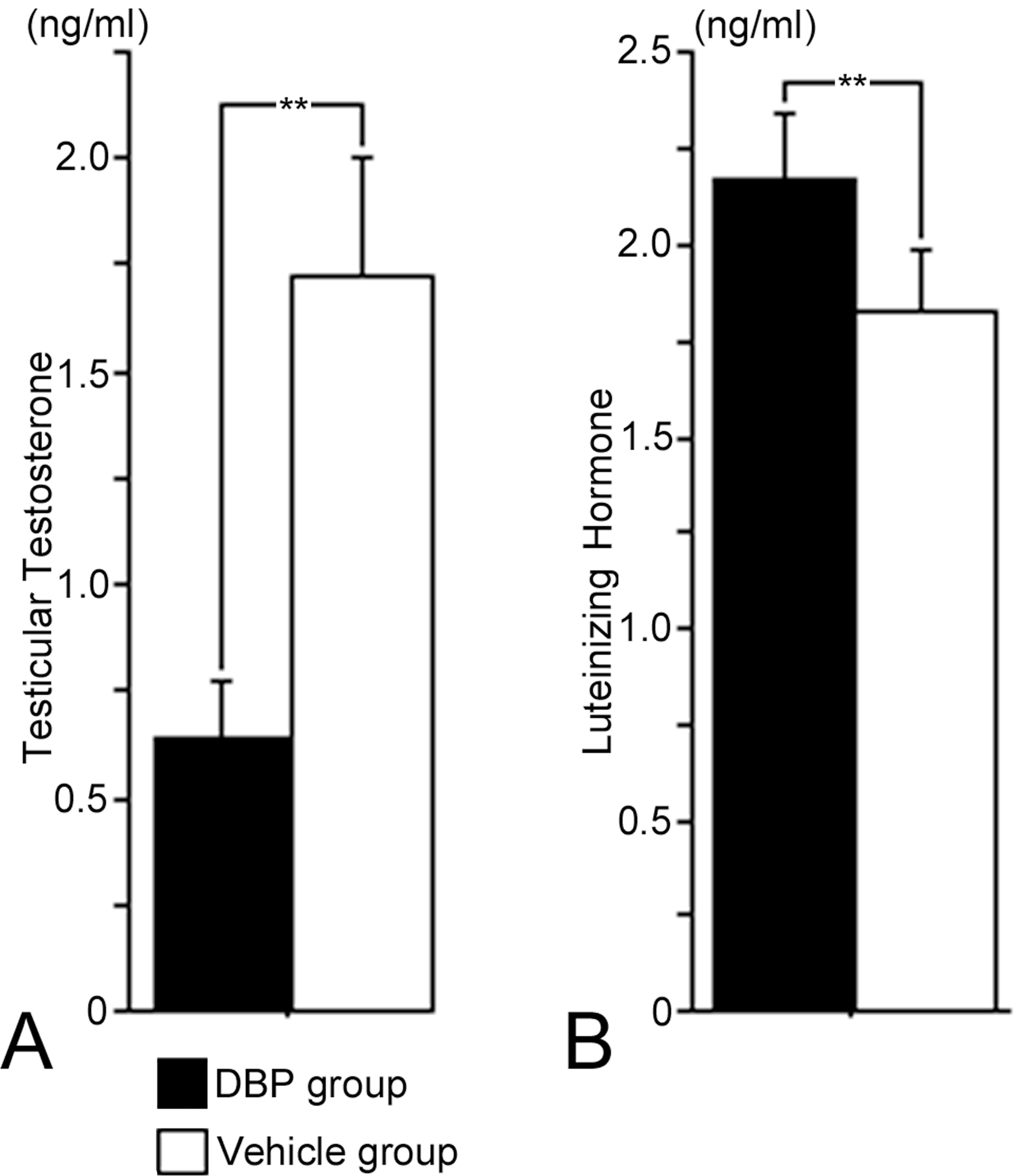
(A) Testicular testosterone levels (ng/ml) and (B) luteinizing hormone levels (ng/ml) of the DBP group were significantly lower than those of the vehicle group. Values are mean ± SD for four litters per group. **p < .05.
Discussion
Although DBP and its major metabolites do not bind to the androgen receptor (Harris et al. 1997; Gray and Furr 2006; Mylchreest et al. 2002), prenatal and/or postnatal exposure of rats to DBP has been characterized as antiandrogenic, causing testicular degeneration and LC hyperplasia in adulthood (Mylchreest et al. 2002; Barlow, Phillips, and Foster 2004).
Nuclear atypia and nucleoli observed in LC hyperplasia in this rat study have also been described in spontaneous human LC hyperplastic lesions (Soderstrom 1987). Although normal LCs have been described as characterized by a large amount of sER, which is considered a higher activity of cholesterol metabolism and T steroidogenesis (Ghadially 1975; Soderstrom 1987; Akingbemi et al. 2001; Barlow, Phillips, and Foster 2004; Hu et al. 2009), and treatment of 6-8 weeks old rats with phthalates (di(2-ethylhexyl) phthalate, di-n-pentyl phthalate, di-n-octyl phthalate, and diethyl phthalate) for 2 days has been showed to be vesiculated and dilated sER in LCs, but not decrease amount of sER (Jones et al. 1993). Because sER was scarcely observed in LCs of hyperplastic lesions in this study, these findings might seem to indicate that the prenatal DBP exposure causes higher testicular toxicity to cholesterol metabolism and T steroidgenesis in LCs compared to other phthalates, and certainly, the testicular T level of the DBP group was significantly lower than that of the vehicle group. Microbodies identified in normal LCs of the rat, mouse, guinea pig, and human that are considered to be involved in T metabolism (Soderstrom 1987; Jones et al. 1993) were not observed in the hyperplastic LCs in this study. Moreover, bundles of intermediate-sized filaments, probably of the vimentin type frequently observed in hyperplastic LCs in this study, were considered compensatory evidence for the decrease in cell organelles, including sER and microbodies (Ghadially 1975; Soderstrom 1987). On the other hand, the increase in the number of cytoplasmic filopodial projections that interdigitated and made contact with adjacent LCs, amount of stripped rER, and number of atypical tight junctions observed in this study suggest that these hyperplastic LCs might show preneoplastic characters. One or two cilia were observed in LCs in this study (Figure 4). Hyperplastic LCs showed cilia formations, which were previously reported in men (Takayama 1981), rabbits (Crabo 1963), and dogs (Connell and Christensen 1975). The significance of cilia formation has not been described in detail, but it has been suggested to play important roles in the control of cell proliferation and in several developmental signaling pathways (Seeley and Nachury 2009). Based on the morphological features, the LC hyperplasia seen in adult male rat offspring following exposure to DBP in utero was considered to be atypical LC hyperplasia.
While chronic LH stimulation has been reported to induce LC hyperplasia in rats (Jegou and Sharpe 1993), some studies have in fact suggested that infertile men may have some degree of LC dysfunction because they have lower serum T values and higher LH levels than controls (Soderstrom 1987; Holm et al. 2003). Low testicular T and high serum LH levels observed in this study might be involved in the induction of LC hyperplasia due to prenatal DBP exposure. However, further study is required to elucidate the detailed mechanisms of induction of LC hyperplasia following prenatal DBP exposure.
Footnotes
Abbreviations
Acknowledgments
This study was partially supported by a research project grant awarded by the Azabu University Research Services Division and the Promotion and Mutual Aid Corporation for Private Schools of Japan; a Grant-in-Aid for Matching Funds Subsidy for Private Universities also provided funding for this project. This study was also supported by Grant-in-Aid (C) #21580371 from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
The authors declare to have no conflicts of interest. This study was supported in part by a Grant-in-Aid from the Ministry of Education, Culture, Sports, Science and Technology of Japan, a Research Project Grant awarded by the Azabu University Research Services Division, and a Promotion and Mutual Aid Corporation for Private Schools of Japan.
