Abstract
Estrogens and androgens affect male and female reproductive systems. Recently, we reported that prenatal di(n-butyl) phthalate (DBP) exposure induced atypical Leydig cells (LCs) hyperplasia during adulthood. The present study investigated the expression of estrogen receptor α (ERα), estrogen receptor β (ERβ), and androgen receptor (AR) in LCs of 5-, 7-, 9-, 14-, and 17-week-old Sprague-Dawley (srl) rats whose dams had been administered DBP intragastrically at 100 mg/kg/day or the vehicle (corn oil) from days 12 to 21 postconception. Immunohistochemical, Western blotting, and reverse transcription polymerase chain reaction analyses revealed that the expressions of ERα, ERβ, and AR proteins and mRNAs in the DBP group were similar to those of the vehicle group at 5 and 7 weeks, but significantly higher ERα and lower ERβ and AR levels were observed in the DBP group at 9 to 17 weeks. The rats prenatally exposed to DBP had seminiferous tubule degeneration and atypical hyperplasia of LCs during adulthood, which was associated with an increase in expression of ERα and a decrease of ERβ and AR in the testis.
Keywords
Introduction
Phthalates are chemicals used as plasticizers in polyvinyl chloride to impart flexibility and durability and comprise up to 40% of the plastic volume. They are classified as environmental endocrine-disrupting chemicals (Occupational Safety and Health Administration [OSHA] 2009). They are found in numerous consumer goods, and because they are not covalently bound to the plastic product, they can leach out of these products over time and be ingested (OSHA 2009). The phthalate esters, including di(n-butyl) phthalate (DBP), displayed no biological activity in a rat uterotropic assay or an in vivo screening test for estrogenicity (Harris et al. 1997; Milligan, Balasubramanian, and Kalita 1998; Zacharewski et al. 1998). However, it has been suggested that DBP has an estrogenic or antiandrogenic effect on the development of the male reproductive system, and the specific primary cellular target of DBP has been considered to be testicular Leydig cells (LCs; Harris et al. 1997; Mylchreest et al. 1999; Parks et al. 2000; Foster et al. 2001; Akingbemi et al. 2003, 2004; Gray and Furr 2006; Alam et al. 2010).
Numerous studies of rodents have shown that prenatal exposure to phthalates reduces the production of fetal LCs and testosterone (T), which results in postnatal male reproductive disorders including altered seminiferous cord formation, multinucleated gonocyte formation, epididymal agenesis, nipple retention, reduced anogenital distance, hypospadias, and cryptorchidism. Further, LCs hyperplasia during adulthood may be an important event in the development of the testicular degeneration process seen in prenatally DBP-exposed male rodents (Mylchreest et al. 2000; Foster et al. 2001; Barlow and Foster 2003; Mahood et al. 2005; Gray and Furr 2006). This has led to the hypothesis of a causal relationship between reduced T and increased luteinizing hormone (LH) production, altered fetal testicular development, and testicular atrophy with increased numbers of LCs in adulthood, but the mechanisms of these changes still remain unknown (McKinnell et al. 2001; Skakkebaek, Rajpert-De, and Main 2001; Lehmann et al. 2004).
The phthalates, including DBP, and their mono metabolites do not bind to the androgen receptor (AR) and have little affinity to estrogen receptor β (ERβ), indicating that they are not direct AR and ERβ antagonists (Harris et al. 1997; Mylchreest, Cattley, and Foster 1998; Gray et al. 1999; Parks et al. 2000; Foster et al. 2001; Akingbemi et al. 2003, 2004). However, DBP was found to be capable of binding to estrogen receptor α (ERα) and enhancing the proliferation of an MCF-7 cell line expressing ERα (Nishihara et al. 2000; Jobling et al. 1995; Harris et al. 1997; Zacharewski et al. 1998). We recently reported that exposure of rats prenatally to DBP induced atypical LCs hyperplasia in adulthood and proposed that fetal LCs are directly affected by DBP in utero. The adult phenotype exposed to DBP developed atypical LCs with high cellular proliferative activity and different estrogen and androgen dependences (Shirai et al. 2013; Wakui et al. 2013). The objective of the present study was to investigate alterations of the hormone receptors ERα, ERβ, and AR on testicular LCs in puberty and adulthood of rats following prenatal exposure to the low level of DBP.
Materials and Methods
Experimental Design
DBP (99.8% pure) was purchased from Aldrich Chemical Co. (Milwaukee, Wisconsin, USA). Eight-week-old time-mated female Sprague-Dawley rats (n = 10) were procured from SRL Co. (Shizuoka, Japan) on gestation day (GD) 0; the day of copulation was confirmed. Upon arrival, animals were distributed into dose groups using body weight randomization. Animals were housed individually in polycarbonate cages containing wood chip bedding in a high-efficiency particulate air-filtered, mass-air-displacement room maintained on a 12-hr light–dark cycle at approximately 22°C ± 2°C with a relative humidity of 55 ± 5%. Animals were fed a conventional diet and had free access to food and water (MF, Oriental Yeast, Osaka, Japan). All experimental procedures were conducted under the approval of the Animal Care and Use Committee at Azabu University School of Veterinary Medicine; medical guidelines established by National Institutes of Health and Public Health Service Policy on the Humane Use and Care of Laboratory Animals were followed. Two groups of pregnant rats (n = 5 per group) were intragastrically administered DBP in ∼0.5 ml corn oil (Nacalai Tesque Inc., Osaka, Japan) per animal at 0 (vehicle group) or 100 mg/kg/day on GDs 12 to 21. Dose solutions were prepared fresh every morning and administered at 9:00 a.m. The regimen was based upon previous studies that illustrated the lowest-observed-adverse-effect level of DBP on fetal male rats at 100 mg/kg/day (Mylchreest, Cattley, and Foster 1998; Barlow and Foster 2003; Shirai et al. 2013; Wakui et al. 2013). Offspring were weighed and sexed at birth. Litters were reduced to 10 offspring, 5 males and 5 females per dam. Weaning was carried out at 21 days postpartum, and pups were then removed from the mothers. Offspring were housed in polycarbonate cages (n = 5 per cage; single sex) with wood chip bedding that was replaced every 5 days. All animals were weighed at birth and again at 5, 7, 9, 14, and 17 weeks of age. For each time point, 5 males were randomly selected from each dose group (1 male from 1 mother of each dose group), weighed, anesthetized, and euthanized by a CO2 overdose. The testes were removed and weighed, and representative samples were fixed in 10% formaldehyde in 0.1 M phosphate buffer for routine light microscopy. The number of LCs was counted at random in >50 16 × 104-μm square areas per section using the computer morphometry program Image-Pro Plus, version 7.0.1 (Media Cybernetics, Inc., Bethesda, MD).
Antibodies
The specificities of the ERα, ERβ, and AR antibodies used were verified by immunolocalization, which has been well characterized (Saunders et al. 1997; Wakui et al. 2011). To detect ERα, we used MC-20 antiserum, a mouse anti-ER antibody (Santa Cruz Biotechnology, Santa Cruz, CA) that has no affinity for ERβ (Saunders et al. 1997; Wakui et al. 2011) as the primary serum. To detect ERβ, we used PA1-310 antiserum, a rabbit anti-ER antibody (Affinity BioReagents, Golden, CO) that was generated against amino acids in the C-terminal region of rat ERβ corresponding to 19 amino acids and has only 6% amino acid homology with rat ERα. To detect AR, we used PA1-111 antiserum, a rabbit anti-AR antibody (Affinity BioReagents), corresponding to the 21 N-terminal amino acids of rat and human AR. These antibodies were affinity purified by column chromatography and their immunolocalizations were confirmed in the nuclei of rat ERα-, ERβ-, and AR-positive cells (Prins, Birch, and Geene 1991; Saunders et al. 1997; Muto et al. 2002; Li et al. 2009; Wakui et al. 2011).
Immunohistochemical Analysis
Immunohistochemical expressions of ERα, ERβ, and AR were analyzed using the avidin-biotin complex (ABC) method. After deparaffinization and hydration, 4-μm thick sections were treated sequentially with 0.3% hydrogen peroxide for 10 min, then blocked with 10% goat or horse serum in phosphate-buffered saline (PBS, 10 mM potassium phosphate, 150 mM sodium chloride, and pH 7.4) for 20 min. Then, sections were rinsed in PBS and treated with primary antibodies for ERα (1:100; Santa Cruz Biotechnology), ERβ (1:50; Affinity BioReagents), or AR (1:50; Affinity BioReagents). Primary antibodies were detected with biotinylated IgG (Vector Laboratories, Burlingame, CA; diluted 1:100), followed by ABC-peroxidase staining (Vector Laboratories, Burlingame, CA) and diaminobenzidine (Sigma-Aldrich, St. Louis, MO). Sections were counterstained with Mayer’s hematoxylin. As a negative control, nonimmunized serum was substituted for the each primary antibody. The expression index of each immunostain reaction of LCs was measured at random in >50 16 × 104-μm square areas per section, and total number of immunostain reaction LCs were measured at random in >800 cells per each section and the indices were expressed as percentage values using the computer morphometry program Image-Pro Plus, version 7.0.1 (Media Cybernetics, Inc.).
Western Blot Analysis
Rat testicular tissues were homogenized in 50 mM Tris-hydrochloride (HCl), 150 mM potassium chloride (pH 7.4), 1% Triton X100, and 0.25 mM phenylmethanesulfonyl fluoride (PMSF) and centrifuged at 8,000 × g for 30 min at 4°C. The supernatant obtained was centrifuged at 100,000 × g for 90 min at 4°C. The pellet was suspended in 50 mM Tris-HCl (pH 7.4), 1% Triton X100, and 1 mM PMSF, and protein concentrations were determined by using a bicinchoninic acid protein assay reagent kit (Pierce, Rockford, IL) with bovine serum albumin as a standard. Microsomal samples (10 µg each) underwent electrophoresis on a 10% sodium dodecyl sulfate–polyacrylamide gel. The proteins were transferred for 2 hr to a nitrocellulose membrane that was blocked by immersing it in 5% nonfat dried milk in PBS with 0.1% (v/v) Tween 20. Western blot analysis was performed by using anti-ERα antibody (1:1,000; Santa Cruz Biotechnology), anti-ERβ antibody (1:1,000; Affinity BioReagents), anti-AR antibody (1:1,000; Affinity BioReagents), and anti-β-actin antibody (1:2,000; A5441, Sigma Aldrich) diluted in PBS with 0.1% (v/v) Tween 20 and incubated 1 hr at room temperature on an orbital shaker. After being washed 3 times in PBS with 0.1% (v/v) Tween 20, they were incubated with horseradish peroxidase–conjugated anti-rabbit antibody (1:2,500; Amersham Biosciences, Piscataway, NJ) for 1 hr on an orbital shaker. After being washed 3 times in PBS with 0.1% (v/v) Tween 20, signals were detected using chemiluminescence (ECL Plus Western Blotting Detection System, Amersham Biosciences). Protein levels were quantified by densitometric scanning (Image-Pro Plus, Media Cybernetics, San Diego, CA). For better comparison and to compensate for possible unequal loading, ERα, ERβ, and AR protein levels in each sample were normalized to that of the housekeeping protein β-actin.
Reverse Transcription Polymerase Chain Reaction (RT-PCR) Analysis
For each RNA sample, 100 ng of testis was used as the template for first-strand cDNA synthesis using a TaqMan Reverse Transcription kit (PE Applied Biosystems, Foster City, CA), according to the manufacturer’s 2-step protocol. Controls included for each reaction were the RNA sample without reverse transcriptase (RNA − RT) and no RNA with reverse transcriptase (no RNA + RT). The final reaction mixes for reverse transcription included 1 × TaqMan RT buffer; 5.5 mM MgCl2; 500 mM each dATP, dGTP, and dCTP; 1 mM dTTP; 0.25 µM random hexamers; 1.25 U/ml Murine Leukemia virus RT; and 0.4 U RNase inhibitor (PE Applied Biosystems). Quantitative analyses of target gene (ERα, ERβ, and AR) mRNA levels were performed by real-time quantitative PCR (ABI Prism 7700 Sequence Detection System, PE Applied Biosystems) with TaqMan chemicals and probes. The TaqMan probes and primers for target genes were assay-on-demand gene expression products that were custom synthesized by PE Applied Biosystems and oligonucleotides with a fluorescent reporter and quencher dyes attached (Erα: Rn 01430445_ml; ERβ: Rn 00688791_ml; AR: Rn 00573309_ml). Optimal primer, probe, and cDNA concentrations were determined in a separate set of experiments to ensure that both target gene and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) fragments were amplified with equal efficiency. PCR reactions were performed with first-strand cDNA (2 ml) from each sample, a Universal PCR Master Mix kit (PE Applied Biosystems), 250 nM TaqMan probe, 0.16 U AmpErase UNG (uracil N-glycosylase), and 900 nM forward and reverse primers of the target gene and GAPDH. Three measurements per sample were performed in each of the 2 independent experiments. Results were analyzed with the ABI Sequence Detector software version 1.7 (PE Applied Biosystems). For relative quantification of target gene expression, the standard curve method was applied. The calibrated standard curve of each target gene cDNA and GAPDH amplification plots were examined at 5 different dilutions (containing 100, 50, 25, 10, and 5 ng) of total RNA samples that were obtained from each PCR product using a TOPO II TA Cloning Kit (Invitrogen, Carlsbad, CA) according to the manufacturer’s recommendations. The target gene’s normalized value was determined by dividing the average target gene value by the average GAPDH value. The standard deviation (SD) of the quotient was calculated from the SD of the target gene and GAPDH using the following formula:
The normalized target gene value is a unitless number that can be used to compare the relative amounts of the target genes in different samples. One way to make this comparison is to designate one of the samples as a calibrator. In this study, testis of the vehicle-treated control group was designated as the calibrator, and the average target gene value was divided by the average calibrator value according to the manufacturer’s instructions for quantification of relative gene expression.
Hormone Analysis
Testicular T and serum LH levels were measured by radio immunoassays as previously described (Wakui et al. 2013).
Statistical Analysis
For each data set, the mean value, SD, and standard error of the mean were calculated and compared by Scheffé’s F test using the computer statistical analysis system Stat View-J 5.0. A p value of less than .05 was regarded as statistically significant (Abacus Concepts, Piscataway, NJ).
Results
Gavage of pregnant dams with 100 mg/kg/day DBP from GD 12 to 21 did not affect their body weights (g), which were similar in control and DBP-treated dams at the beginning of the experiment and also at the end. Additionally, no decrease in litter size or pup survival, alteration of sex ratio, or difference in body weights of male pups compared to controls on any day examined were found (data not shown). Until the pups were 7 weeks old, the relative testicular weights of DBP groups were similar to those of the vehicle group, while testicular weights showed a significant decrease after 9 weeks (data not shown). Light microscopic observations revealed that the number of LCs in the DBP group was significantly higher than that of the vehicle groups at 9 to 17 weeks (Figure 1). However, the testicular T level of the DBP group was significantly lower than that of vehicle group from 5 to 17 weeks old. The serum LH level of the DBP group was significantly lower than that of the vehicle group at 5 and 7 weeks old, and it was significantly increased at 9 to 17 weeks old compared to that of vehicle group (Figure 1).
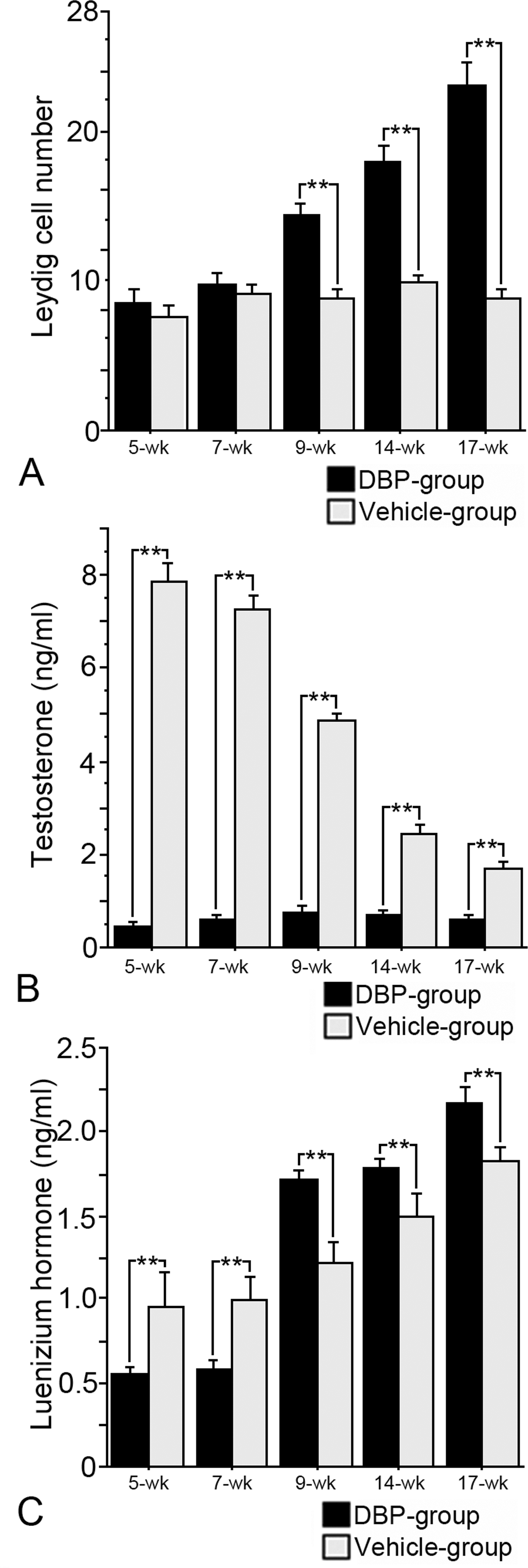
(A) Leydig cell number per unit square (16 × 104 µm) for rats (5–17 weeks old) following exposure on gestation days 12–21 to 100 mg DBP/kg/day (DBP group) or corn oil (vehicle group). (B) Testicular testosterone (ng/ml) and (C) serum luteinizing hormone (ng/ml) —levels from rats (5–17 weeks old) following exposure on gestation days 12–21 to 100 mg DBP/kg/day (DBP group) or corn oil (vehicle group). Values represent mean ± SEM. **p < .05, Scheffé’s F test. DBP = di(n-butyl) phthalate; SEM = standard error mean.
Light and electron microscopic observations of LCs in rats prenatally treated with 100 mg/kg/day DBP and normal rats at 5 to 17 weeks old have been described previously (Shirai et al. 2013; Wakui et al. 2013), and therefore an in-depth description will not be given here. Microscopic observations of testicular structures of the DBP group at 5 and 7 weeks old were similar to those of the vehicle group, but as the DBP group aged, it showed gradually increasing testicular atrophy with widespread germ cell loss and an increase in the number of LCs. This lesion became severe testicular degeneration and dysplastic LCs hyperplasia at 9 to 17 weeks of age.
Immunohistochemical expression (Figures 2– 5) and western blot analysis (Figure 6) of ERα, ERβ, and AR in testes of the DBP and vehicle groups were similar at 5 and 7 weeks, but ERα increased and ERβ and AR decreased in the DBP group at 9 to 17 weeks. RT-PCR analysis revealed that expressions of ERα, ERβ, and AR mRNAs of the DBP group were similar to those of the vehicle group at 5 and 7 weeks, but significantly higher ERα mRNA, and lower ERβ and AR mRNA levels were seen in the DBP group at 9 to 17 weeks of age (Figure 7).
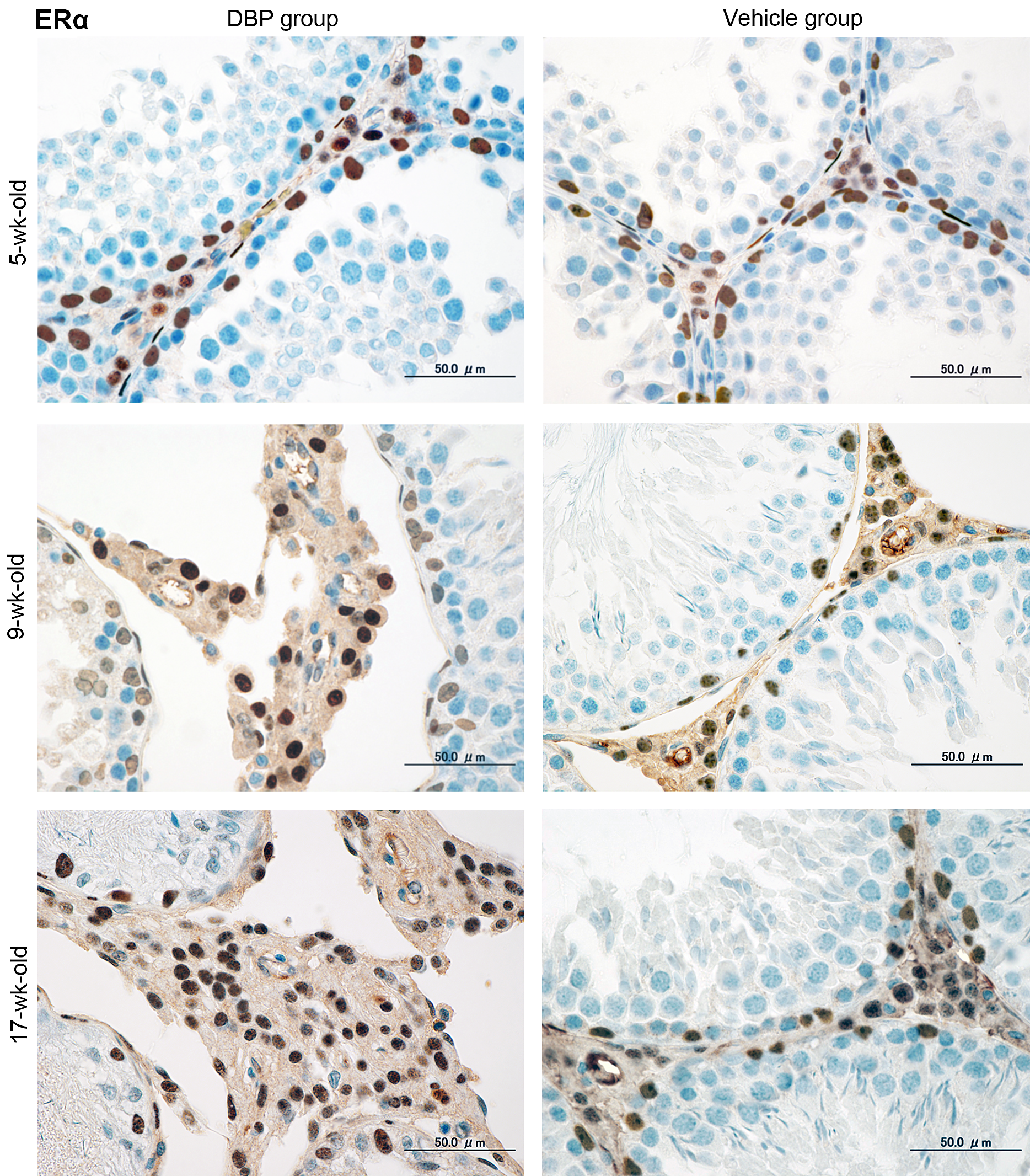
Representative immunohistochemical assays for ERα in rat testes at 5, 9, and 17 weeks old following exposure on gestation days 12–21 to 100 mg DBP/kg/day (DBP group) or corn oil (vehicle group). Three serial sections were used to analyze ERα antibody by an immunohistochemical method. Brown black immunohistochemical stains localized at the nuclei of Leydig cells, Sertoli cells, and peritubular cells. The number of Leydig cells increases with age, and Leydig cell hyperplasia with degenerated seminiferous tubules are seen in 9-week-old rats of the DBP group. Bar = 50 µm. DBP = di(n-butyl) phthalate; ERα = estrogen receptor α.
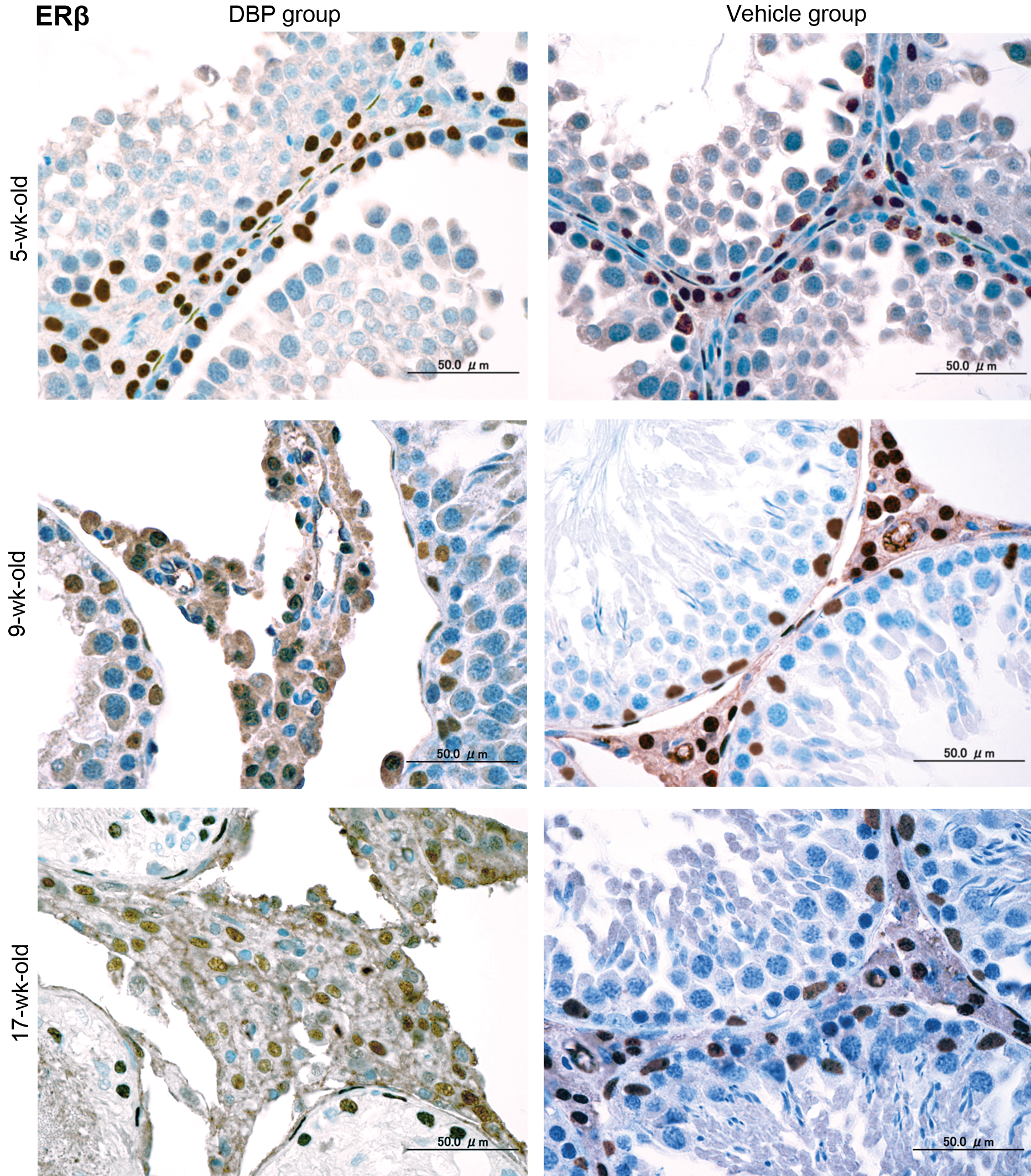
Representative immunohistochemical assays for ERβ in rat testes at 5, 9, and 17 weeks old following exposure on gestation days 12–21 to 100 mg DBP/kg/day (DBP group) or corn oil (vehicle group). Three serial sections were used to analyze ERβ antibody by an immunohistochemical method. Brown black immunohistochemical stains localized at the nuclei of Leydig cells, Sertoli cells, and peritubular cells. The number of Leydig cells increases with age, and Leydig cell hyperplasia with degenerated seminiferous tubules are seen in 9-week-old rats of the DBP group. Bar = 50 µm. DBP = di(n-butyl) phthalate; ERβ = estrogen receptor β.
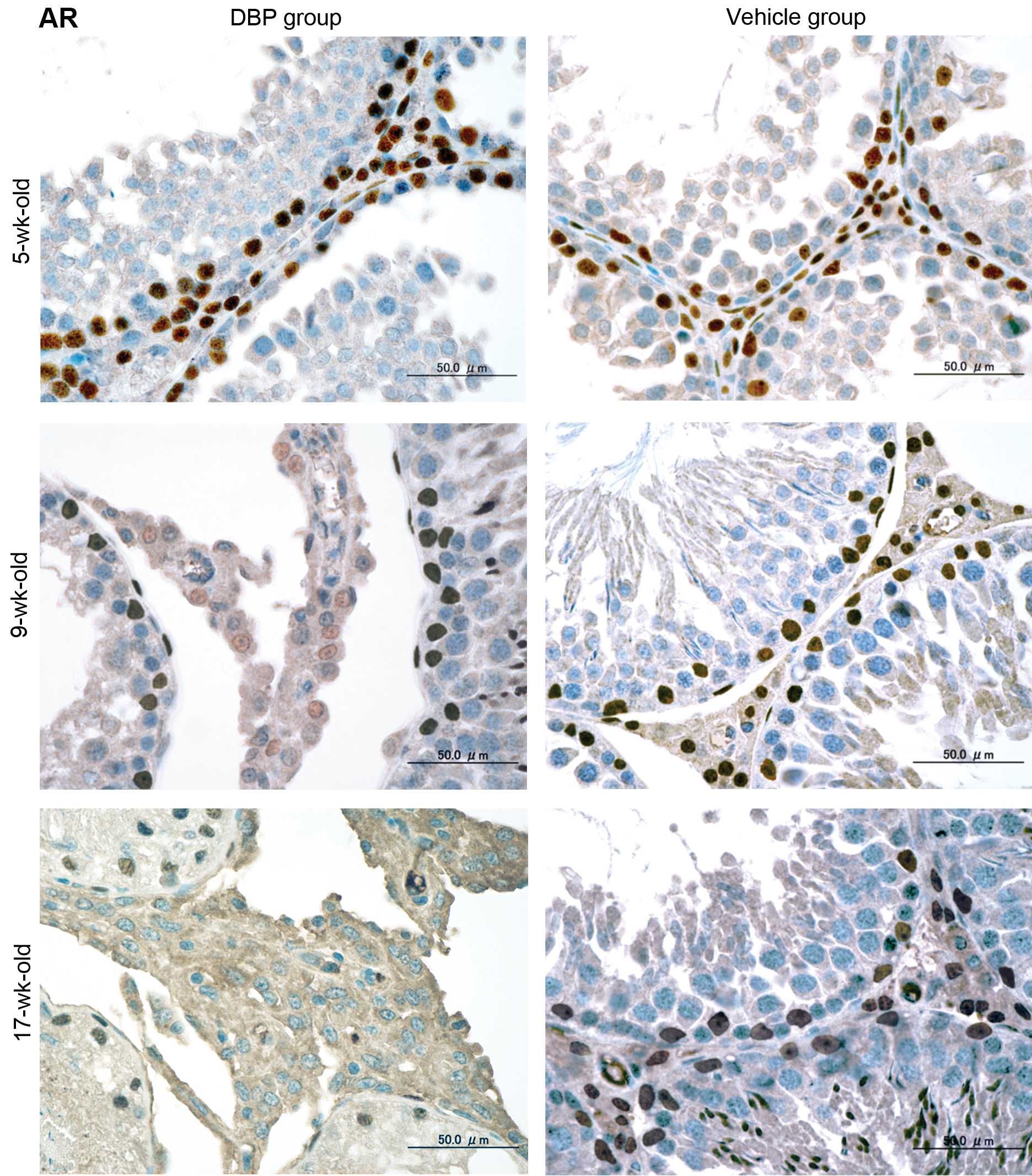
Representative immunohistochemical assays for AR in rat testes at 5, 9, and 17 weeks old following exposure on gestation days 12–21 to 100 mg DBP/kg/day (DBP group) or corn oil (vehicle group). Three serial sections were used to analyze AR antibody by an immunohistochemical method. Brown black immunohistochemical stains localized at the nuclei of Leydig cells, Sertoli cells, and peritubular cells. The number of Leydig cells increases with age, and Leydig cell hyperplasia with degenerated seminiferous tubules are seen in 9-week-old rats of the DBP group. Bar = 50 µm. AR = androgen receptor; DBP = di(n-butyl) phthalate.
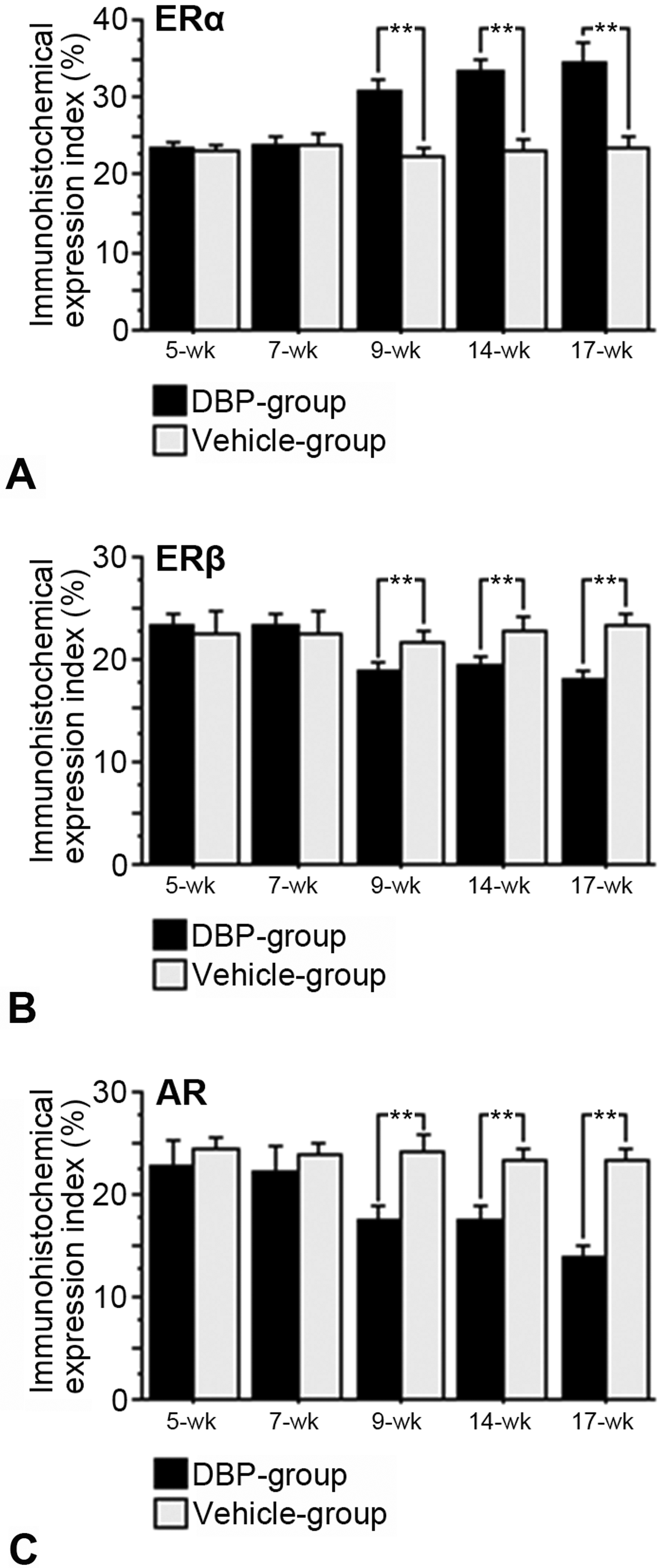
The panels A, B, and C show the immunohistochemical expression index of (A) ERα, (B) ERβ, and (C) AR immunostain reactions of Leydig cells. Immunohistochemical expression index of ERα, ERβ, and AR in Leydig cells of the DBP and vehicle groups were similar at 5 and 7 weeks of age, while expression of ERα is more intense, and those of ERβ and AR are lower at 9, 14, and 17 weeks of the DBP group compared to the vehicle group. Results are obtained by screening samples from 5 rats of each group. Values represent the mean ± SD. **p < .05 by Scheffé’s F test. AR = androgen receptor; DBP = di(n-butyl) phthalate; ERα = estrogen receptor α, ERβ = estrogen receptor β; SD = standard deviation.
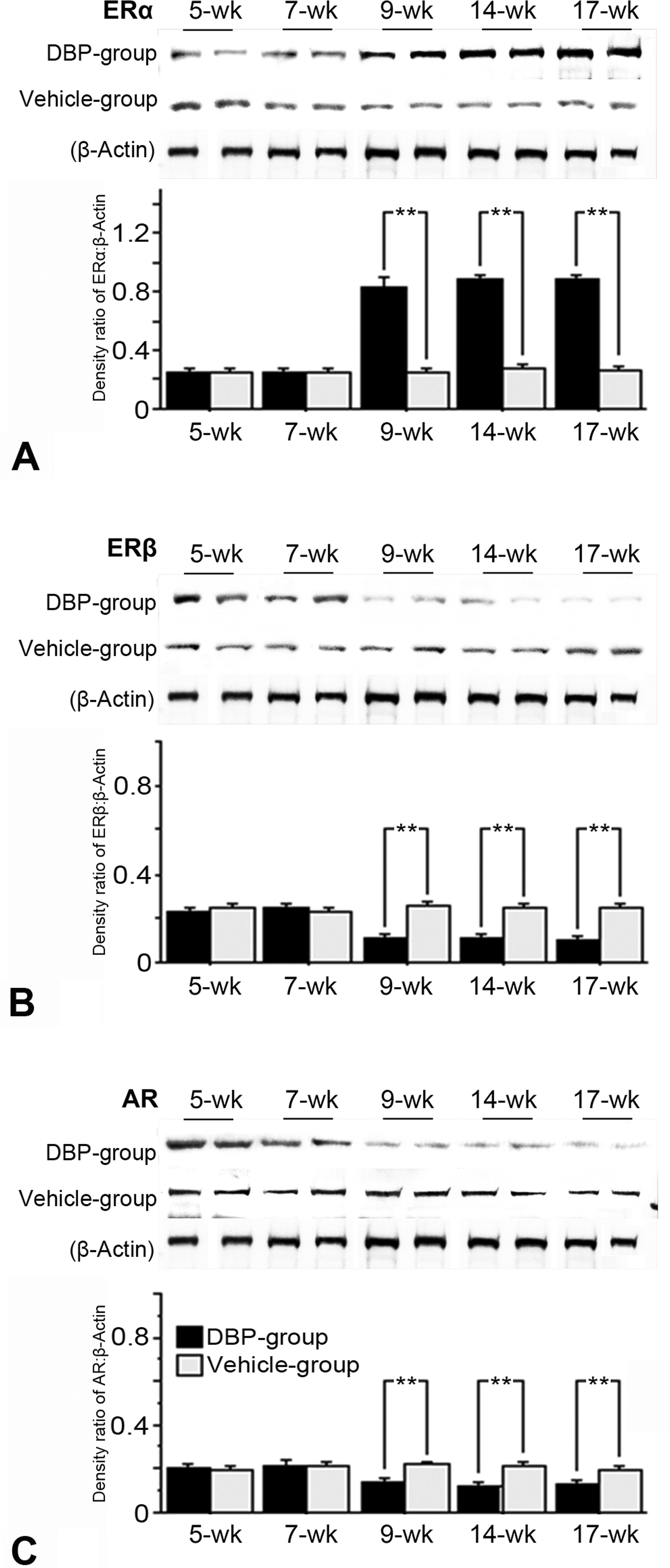
Representative Western blots of (A) ERα, (B) ERβ, and (C) AR in rat testes at 5, 7, 9, 14, and 17 weeks old following exposure on gestation days 12–21 to 100 mg DBP/kg/day (DBP group) or corn oil (vehicle group); results from 2 rats of each group are shown. The protein concentration was determined using a bicinchoninic acid protein assay reagent kit (Pierce) with bovine serum albumin as the standard. Microsome samples (10 µg) were applied for Western blotting F
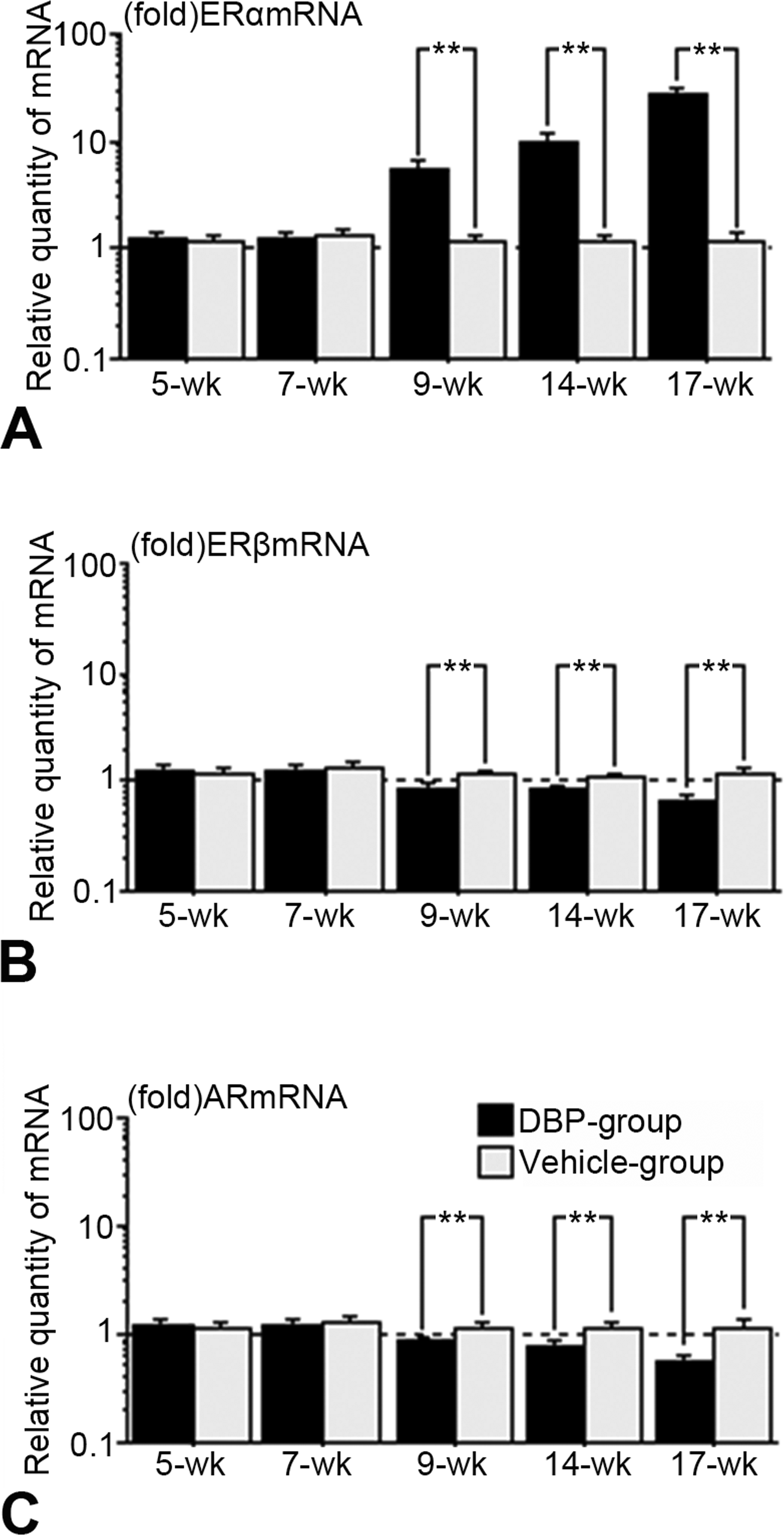
(A) ERα, (B) ERβ, and (C) AR mRNA levels in rat testes at 5, 7, 9, 14, and 17 weeks old following exposure on gestation days 12–21 to 100 mg DBP/kg/day (DBP group) or corn oil (vehicle group). The indicated mRNA levels were determined by real-time quantitative RT-PCR and analyzed using the standard curve method: 1-fold of the relative quantity of mRNA of the vehicle-treated normal testis was designated as the calibrator value. Each ERα, ERβ, or AR mRNA value was normalized to that of the endogenous housekeeping gene GAPDH in each tissue. The results were obtained by screening samples from 5 rats of each group at each time point. ERα, ERβ, and AR mRNA levels of the DBP group and the vehicle group were similar at 5 and 7 weeks of age. The amount of ERα mRNA in the DBP group was significantly (**p < .05, Scheffé’s F test) greater than that of the vehicle group; amounts of ERβ mRNA and AR mRNA in the DBP group were significantly lower (**p < .05, Scheffé’s F test) than those of the vehicle group at 9, 14, and 17 weeks of age. Values represent the mean ± SEM. AR = androgen receptor; DBP = di(n-butyl) F
Discussion
A primary target of phthalates, including DBP, is thought to be LCs, which produce the male primary steroid hormone T and ordinarily express ERα, ERβ, and AR and whose autocrine or paracrine activity might play a role in the regulation of germ cell maturation (Harris et al. 1997; Paech et al. 1997; Mylchreest et al. 1999; Parks et al. 2000; Akingbemi et al. 2001, 2003, 2004; Foster et al. 2001; Gray et al. 2001; Selva et al. 2004; Alam et al. 2010).
Two distinct generations of LCs have been identified in postnatal rats, premature LCs and adult LCs. The latter are distributed within almost all testes after 8 weeks postnatally, and are essential for sexual maturation, male mating behavior, and fertility (Chen, Ge, and Zirikin 2009; Wu et al. 2010). The direct effects of DBP on adult LCs have been described as a decreased in testicular T level and a compensatory increase in serum LH level (Pan et al. 2006). The direct effects of DBP on premature LCs have been described: 4-week-old juvenile rats exposed to DBP for 30 days (Ryu et al. 2007) and 3-week-old rats exposed to diethylhexyl phthalate for 28 days (Akingbemi et al. 2001) had increased testicular T and serum LH levels. Moreover, 5- to 14-day-old juvenile rats exposed to DBP (Kim et al. 2004) or 4-week-old rats treated 30 days with DBP (Ryu et al. 2007) have been reported to show downregulation of testicular ERβ and AR, although the level of testicular ERα was unchanged or unclear.
On the other hand, the effect of prenatal phthalate exposure on adult LCs was thought to be different from that of postnatal exposure (Mylchreest, Cattley, and Foster 1998; Barlow and Foster 2003; Shirai et al. 2012; Wakui et al. 2013). Because DBP is rapidly transferred across the placenta to embryos (Clewell et al. 2008) and the elimination half-life of phthalate monoesters is approximately 24 hr, exposure ends shortly after birth, and then the DBP is cleared from rat offspring (Hoppin et al. 2002; Barlow and Foster 2003). The prenatal exposure to DBP does not directly affect adult LCs, but we have suggested that atypical adult LCs hyperplasia following prenatal DBP exposure was derived from the fetal LCs affected in utero by DBP (Shirai et al. 2013; Wakui et al. 2013).
In the present prenatal DBP exposure study, the low T levels in adulthood are probably explained by the reduction in smooth endoplasmic reticulum that has been observed morphologically (Shirai et al. 2013; Wakui et al. 2013). High LH levels with LC hyperplasia in adulthood following prenatal DBP exposure have been considered a compensatory reaction triggered by T insufficiency (Mylchreest, Cattley, and Foster 1998; Gray et al. 1999; Mylchreest et al. 1999, 2002; Shultz et al. 2001; Akingbemi et al. 2003; Barlow, Phillips, and Foster 2004). It has been reported that T downregulation enhances ERα activation in LCs (Delbes et al. 2005), and the present study documented that hyperplasia of adult LCs following prenatal DBP exposure might be involved in an upregulation of ERα pathway activity. However, the detailed mechanism concerning how LC hyperplasia would be a direct response to activation of ERα is still unclear.
In conclusion, the present study proposes that prenatal exposure to DBP has effects on adult rat ERα, ERβ, and AR of LCs, and that the hyperplasia of LCs might be involved in the increase in LH levels, the low T biosynthesis, and the high expression of ERα on LCs. It has been previously described that prenatal exposure to DBP induced LCs hyperplasia at 18 months of age in rat (Barlow, Phillips, and Foster 2004). Since ERβ and AR are expressed in Sertoli cells and germ cells as well as LCs (O’Donnell et al. 2001; Hess 2003), the fact that levels of ERβ and AR have decreased in the Western blot and RT-PCR could be explained by the seminiferous tubular degeneration and germ cell loss that occurred in the older animals. The immunohistochemical changes in the receptors in the LCs could be a response to the low T biosynthesis rather than a causative factor for the hyperplasia. Prenatal DBP exposure might extend beyond the reproductive organs, to include the systemic physiology, because ERα is expressed in other tissues, for example, the efferent ducts and initial part of the epididymis, the cardiovascular system and bones (Gustafsson 1999; Mueller and Korach 2001; Simoncini et al. 2002; Hess 2003; Arias-Loza et al. 2006; Ropero et al. 2006; Bolego et al. 2010). Moreover, there is a need to find out the detailed cellular localization of ERα, ERβ, and AR within the testis following prenatal exposure to DBP and their roles in testicular development and normal physiology. Additional studies are therefore required to better assess the risk to humans regarding concomitant prenatal exposure to environmentally relevant levels of DBP and other phthalates.
Footnotes
Acknowledgment
The authors thank M. Sigomoto, M. Ohwada, K. Sakurai, and M. Hayashi for technical assistances and Katherine Ono for critical reading and editing the article.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed of the following financial support for the research, authorship, and/or publication of this article: A research project grant award by the Azabu University Research Services Division, and the Promotion and Mutual Aid Corporation for Private Schools of Japan, Grant-in-Aid for Matching Fund Subsidy for Private Universities provided funding for this project. This study was supported by Grants-in-Aid (C) #25450473 from the Ministry of Education, Culture, Sports, Science and Technology of Japan.

