Abstract
The goal of this article is to evaluate a recently published subchronic inhalation study with carbon nanofibers in rats and discuss the importance of a weight-of-evidence (WOE) framework for determining no adverse effect levels (NOAELs). In this Organization for Economic Cooperation and Development (OECD) 413 guideline inhalation study with VGCF™-H carbon nanofibers (CNFs), rats were exposed to 0, 0.54, 2.5 or 25 mg/m3 CNF for 13 weeks. The standard toxicology experimental design was supplemented with bronchoalveolar lavage (BAL) and respiratory cell proliferation (CP) endpoints. BAL fluid (BALF) recovery of inflammatory cells and mediators (i.e., BALF– lactate dehydrogenase [LDH], microprotein [MTP], and alkaline phosphatase [ALKP] levels) were increased only at 25 mg/m3, 1 day after exposure. No differences versus control values in were measured at 0.54 or 2.5 mg/m3 exposure concentrations for any BAL fluid endpoints. Approximately 90% (2.5 and 25 mg/m3) of the BAL-recovered macrophages contained CNF. CP indices at 25 mg/m3 were increased in the airways, lung parenchyma, and subpleural regions, but no increases in CP versus controls were measured at 0.54 or 2.5 mg/m3. Based upon histopathology criteria, the NOAEL was set at 0.54 mg/m3, because at 2.5 mg/m3, “minimal cellular inflammation” of the airways/lung parenchyma was noted by the study pathologist; while the 25 mg/m3 exposure concentration produced slight inflammation and occasional interstitial thickening. In contrast, none of the more sensitive pulmonary biomarkers such as BAL fluid inflammation/cytotoxicity biomarkers or CP turnover results at 2.5 mg/m3 were different from air-exposed controls. Given the absence of convergence of the histopathological observations versus more quantitative measures at 2.5 mg/m3, it is recommended that more comprehensive guidance measures be implemented for setting adverse effect levels in (nano)particulate, subchronic inhalation studies including a WOE approach for establishing no adverse effect levels; and a suggestion that some findings should be viewed as normal physiological adaptations (e.g., normal macrophage phagocytic responses—minimal inflammation) to long-term particulate inhalation exposures.
Keywords
Introduction
VGCF™-H is a vapor-grown carbon nanofiber (CNF) and a member of the family of carbon nanostructures. Suggested commercial functions for these high-performance materials include battery applications and strengthening of polymer composites. From a physicochemical standpoint, CNFs are carbon allotropes and belong to the multiwall carbon nanotube (MWCNT) family (DeLorme et al. 2012). Although similar in chemical composition to carbon nanotubes, CNFs have some physicochemical characteristics that may present a reduced toxicological potency relative to CNT inhalation exposures. These include a limited specific surface area metric of the CNF particulate (14 m2/g vs. 250 [CNT] m2/g); diminished trace catalyst metal content, and decreased aspect ratios. This conclusion is based upon current knowledge of fiber/nanoparticle toxicology tenets and relates to potential impacts of enhanced particle surface area indices, role of metal catalysts to produce reactive oxidant species, and the influence of fiber-type aspect ratios (Donaldson et al. 2006; 2011).
Recently, we reported on the toxicological results of a subchronic inhalation toxicity study with CNFs in male and female rats (DeLorme et al. 2012). In this study, 4 groups of rats per sex were exposed 6 hr/day, 5 d/week nose-only, to aerosols of 0, 0.54, 2.5, or 25 mg/m3 CNF over a 90-day period. Following the end of exposures, rats were assessed via standard toxicology, clinical chemical, and histopathological endpoints. Moreover, to gain greater mechanistic insights into the potential effects of inhaling CNF on the respiratory tract, additional investigative methodologies were added to the standard experimental design of the study. For instance, bronchoalveolar lavage fluid (BALF) analyses were designed to focus on identification of pulmonary inflammatory cells and mediators following long-term particle exposures. In addition, using BrdU cell proliferation (CP) endpoints, cell injury and repair activity in several anatomical compartments of the respiratory tract (including terminal bronchiole, lung parenchymal/alveolar duct, and subpleural/mesothelial regions) were assessed and quantified.
The results of the study demonstrated that inhalation exposures of rats to 0.54, 2.5, and 25 mg/m3 CNFs produced minor but detectable accumulation of extrapulmonary fibers in various organs including liver, kidney, and brain (choroid plexus) but without adverse tissue effects. At 2.5 and 25 mg/m3 CNF, some histopathological evidence of inflammatory responses in the terminal bronchiole and lung parenchymal regions of the respiratory tract were noted wherein fiber-laden alveolar macrophages had accumulated. This morphological finding was characterized by minimal infiltrates of inflammatory cells in rats exposed to 2.5 mg/m3 CNF; while inflammation, along with some thickening of interstitial walls, and hyperplasia of type II epithelial cells, was graded as “slight” for the 25 mg/m3 concentration by the study pathologist. In addition, a nonspecific inflammatory response was noted in the nasal passages, and this was regarded as a nonadverse, adaptive effect.
In contrast to the histopathological assessments at the two highest exposure concentrations, a variety of BALF and CP endpoint increases versus controls were noted only at 25 mg/m3 VGCFTM-H but were not different from control values at either of the 0.54 or 2.5 mg/m3 exposure concentrations. The lack of significant findings versus air-exposed controls at the 2.5 mg/m3 concentration included measurements of the following BALF endpoints: cellular inflammation, lactate dehydrogenase (LDH), microprotein (MTP), and alkaline phosphatase (ALKP). Microscopic BrdU immunostaining CP studies were conducted on respiratory tract anatomical sites that included the following locations: terminal bronchiolar regions; lung parenchyma/alveolar ducts; and subpleural regions and mesothelial cell components of the visceral pleura.
The no observed adverse effect level (NOAEL) for CNF was conservatively interpreted to be 0.54 mg/m3 for male and female rats, based solely on the minimal inflammation assessment in the terminal bronchiole and lung parenchymal areas of the 2.5 mg/m3 exposures, and a small increase in lung weights versus controls (i.e., 10%) in the females. Accordingly, this brief article is designed to provide an alternative viewpoint and discussion of the results from the DeLorme et al. (2012) study, as they relate to NOAEL assessment.
Given the apparent lack of concurrence in the study results, it is important to consider that (1) the histopathology observations at the 2.5 mg/m3 exposure level did not correlate with any of the CP and/or BALF results at that exposure concentration; (2) a “minimal inflammation” response (including macrophages and neutrophils) might be considered a physiologically normal, pulmonary adaptive response to a 13-week inhalation exposure to particulates. Accordingly, analysis of the study findings raises some important and meaningful questions:
In the absence of correlative findings of BAL fluid and cellular proliferation analyses, could the minimal inflammatory response assessment represent an adaptive rather than an adverse response, following 90 days of inhalation exposure?
Should the identification/designation of an adverse effect level (and therefore determination of corresponding NOAELs) in a study require a consideration of all the requisite data; that is, a weight-of-evidence (WOE) approach, and not simply a single adverse finding?
The determination of NOAEL is generally used as a point of departure from which a variety of assessment factors are applied for setting health-based occupational or community exposure levels. Therefore, conclusions drawn from the study findings require consideration of all of the relevant data generated, concomitant with the severity of adverse effects measured in the study.
Methods
Test Substance
The test material VGCFTM-H CNF was supplied by the sponsor, Showa Denko KK, Tokyo, Japan. A transmission electron micrograph as well as chemical composition and other physicochemical characteristics of CNFs are illustrated in Figure 1 and Table 1.
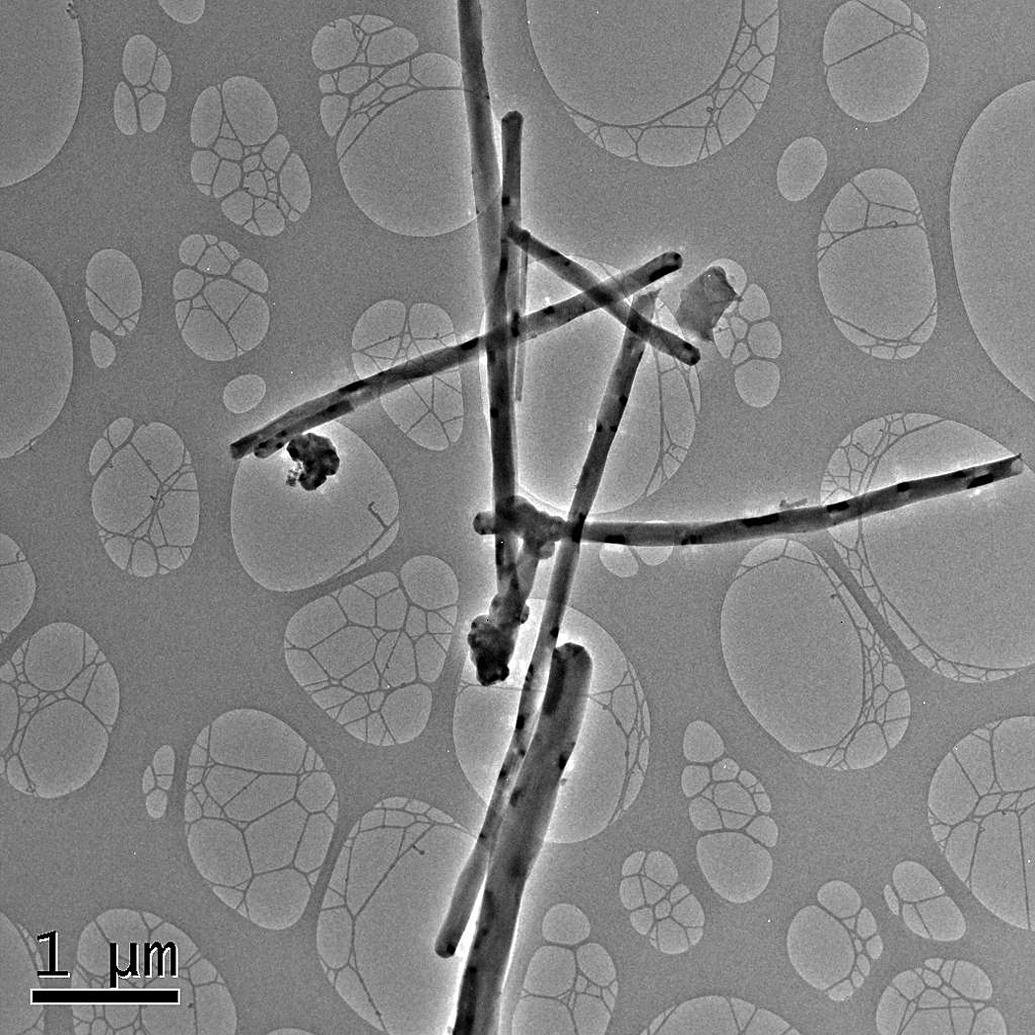
Transmission electron micrograph of aerosolized VGCF™-H CNFs on filter samples taken from the 25 mg/m3 exposure atmosphere demonstrating a nonlinear nature of the fibers and the possible agglomeration of fibers at the fiber ends.
Ninety-day inhalation exposure study with carbon nanofibers—VGCF™-H characterization.

Abbreviations: GSD = geometric standard deviation; MMAD = mass median aerodynamic diameter; ICP-AES = Inductively coupled plasma- atomic emission spectroscopy.
General Experimental Design
The design of this study has recently been reported in detail (DeLorme et al. 2012). Briefly, 4 groups each of male and female Sprague Dawley rats were exposed nose only to target concentrations of 0, 0.5, 2.5, or 25 mg/m3 of VGCFTM-H CNFs for 13 weeks (6 hr/day, 5 days/week). Groups of rats from each exposure level were sacrificed 1 day after the end of 65 exposures. Additionally, a recovery group of males and females exposed to the 0 or 25 mg/m3 concentrations were studied at 3 months postexposure to assess reversibility of any observed toxic effects.
In addition to standard clinical, chemical, and hematological endpoints, 10 animals from each of the exposure groups were sacrificed, necropsied, and examined for gross and microscopy pathology evaluations. Additional groups of 5 rats per sex per exposure concentration were sacrificed 1 day after the final exposure for BALF studies and other groups for BrdU CP studies.
BAL Studies
The methodology for conducting BAL fluid studies has previously been reported (Warheit et al. 1997; DeLorme et al. 2012). Briefly, pulmonary inflammatory responses were assessed based on quantification of cellular differentials in cytocentrifuge preparations of BAL-recovered cells. Biochemical assays were performed on lavage-recovered fluid and analyzed for LDH (a measure of general cytotoxicty); BAL fluid MTP concentrations (as a measure of enhanced permeability of vascular proteins); and BAL fluid alkaline phosphatase (as a measure of alveolar type II cell cytotoxicity). Previous studies have demonstrated an excellent correlation between BAL fluid inflammatory mediators, cytotoxicity indicators, and lung morphology/histopathological effects (Warheit et al. 2007a, 2007b; Sayes, Reed, and Warheit 2007).
CP Studies
The methodologies for implementing CP studies on lung tissues have previously been described elsewhere (Warheit, Hartsky, and Frame 1996; DeLorme et al. 2012). A minimum number of 500 to 1,000 cells/anatomical region/rat were counted each in the following anatomical compartments of the lung: (1) airway and terminal bronchiolar regions; (2) lung parenchymal and alveolar duct regions; and (3) subpleural regions, including mesothelial cells on the visceral pleural surface. Note that greater than 90% of the cells counted in the subpleural/mesothelial region were located in the subpleural compartment; versus <10% identified as mesothelial cells. The airway terminal bronchiole compartment was defined as airway epithelial cells proximal to alveolar ducts. Alveolar duct/lung parenchymal compartments were identified as those cells located distal to the terminal bronchioles—commencing at the alveolar duct bifurcation through the first three alveolar ducts. The cells were counted in random fashion as one moved distally. The subpleural/mesothelial region was defined as the population of cells contained within the distal alveolar duct regions adjacent to the visceral pleural surface.
Statistical significance was judged at the p < .05 level as described previously (DeLorme et al. 2012). Separate analyses were performed on the data generated for each sex, for each anatomical compartment.
Light Microscopy—Histopathology of Respiratory Tract–Lung Tissue
Briefly, parasagittal sections from the right cranial and caudal lobes and regions of the left lobe were dehydrated in ethanol and sectioned for histology, embedded in paraffin, cut, and mounted on glass slides. The slides were stained with an anti-BrdU antibody and counterstained with aqueous hematoxylin. Multiple areas of lung sections were sampled, and the original DeLorme et al. (2012) study was evaluated by a pathologist and the original study was peer reviewed. The severity of lesions was graded on a severity scoring basis of 1 to 4 (i.e., minimal, slight, moderate, and marked). For this publication, light micrographs from the BrdU lung histopathology set were taken from selected slide sections from the mid- and high-exposure concentration groups.
Results
BAL Fluid Studies
The number of BAL-recovered cells from the lungs of VGCFTM-H exposed rats was not significantly increased among any of the groups (data not shown). Ninety-day exposures to CNF produced small but significantly enhanced inflammatory responses characterized by the presence of neutrophils (11–12%) at the high exposure concentration (i.e., 25 mg/m3). The percentage of neutrophils in the 2.5 mg/m3 exposure concentration was <5%, which is in the range of normal control values, thus indicating a lack of pulmonary cellular inflammatory responses following 13 weeks of exposure (Figure 2; Figure 3A–D).
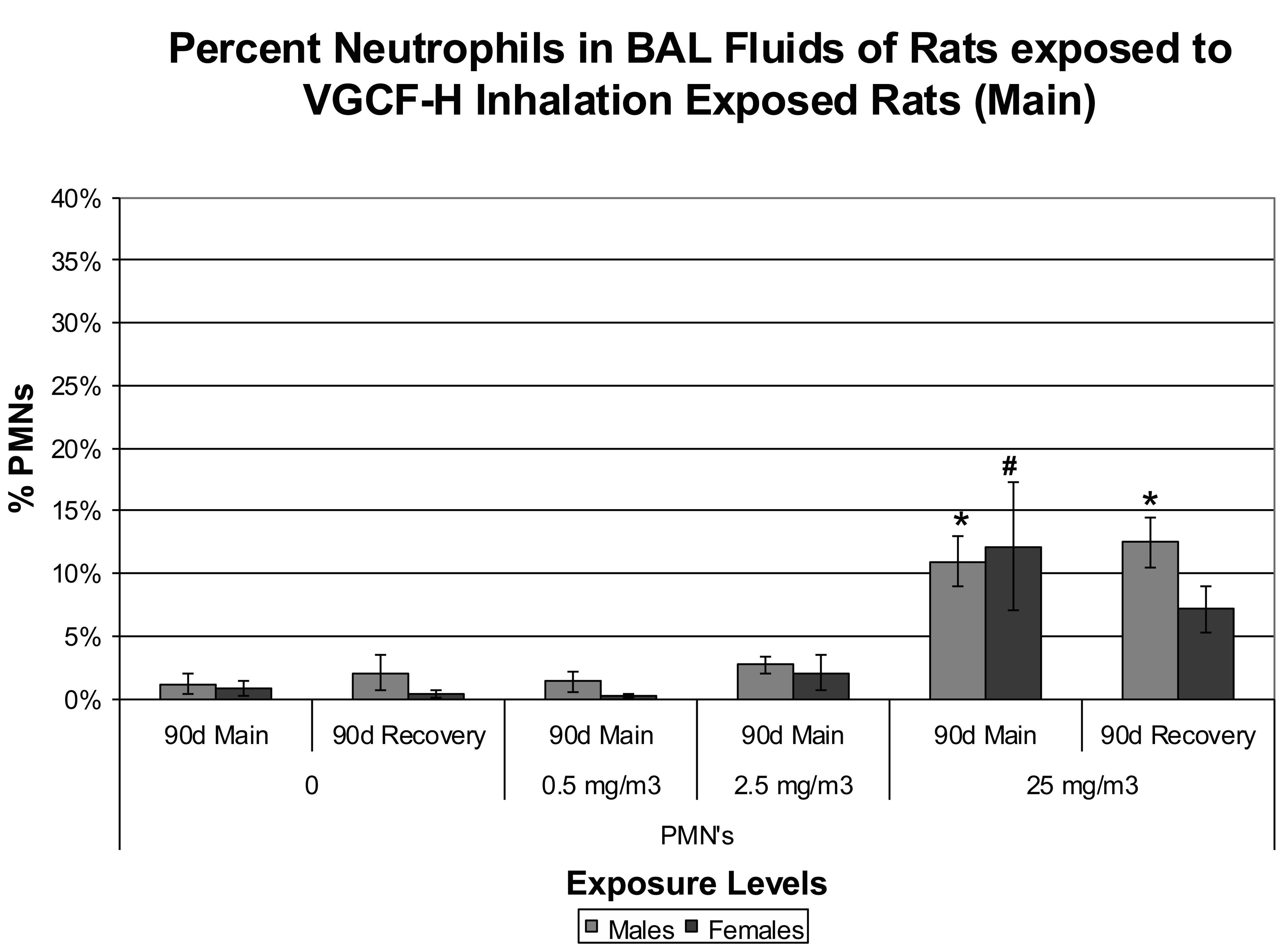
Bronchoalveolar lavage fluid data representing percentages of neutrophils recovered from the lungs of male and female rats exposed to carbon nanofiber ([CNF] data modeled from DeLorme et al. 2012). *p or #p < .05.
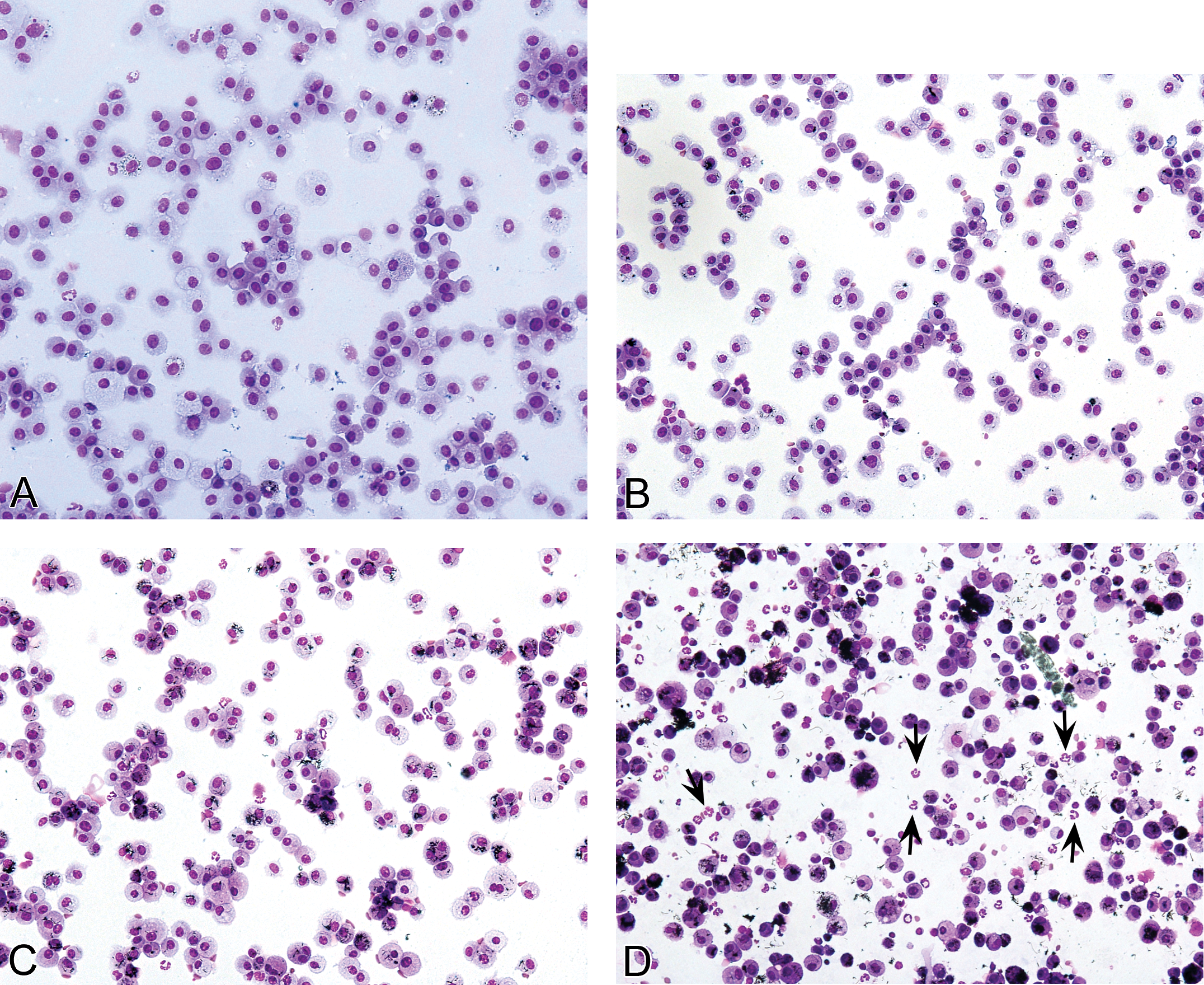
A, Cytocentrifuge cellular preparations of bronchoalveolar lavaged cells recovered from an air control male rat. Note that the >98% of recovered cells were identified as pulmonary macrophages (20× magnification × objective). B, Cytocentrifuge cellular preparations of bronchoalveolar lavaged cells recovered from a male rat exposed to 0.5 mg/m3 VGCF™-H carbon nanofibers (CNFs). Note that >98% of recovered cells were identified as pulmonary macrophages. In addition 60–70% of recovered macrophages contained CNF test material (20× magnification). C, Cytocentrifuge cellular preparations of bronchoalveolar lavaged cells recovered from a male rat exposed to 2.5 mg/m3 CNFs. Note that >97% of recovered cells were identified as pulmonary macrophages. Moreover, 90% of recovered macrophages had phagocytized CNF test material (20× magnification). D, Cytocentrifuge cellular preparations of bronchoalveolar lavaged cells recovered from a male rat exposed to 25 mg/m3 CNFs. Note the minor inflammatory response as represented by ∼11% neutrophils (arrows). Greater than 90% of the macrophages were phagocytic, that is, containing CNFs (20× magnification).
At the 1-day sacrifice period following the 13-week exposure, the BAL fluid LDH, MTP, and ALKP levels were significantly increased versus control values in male and female rats exposed to 25 mg/m3 CNF. However, no differences versus controls were measured for those endpoints in the 0.54 or 2.5 mg/m3 exposed groups (see Table 2).
Bronchoalveolar lavage fluid values.
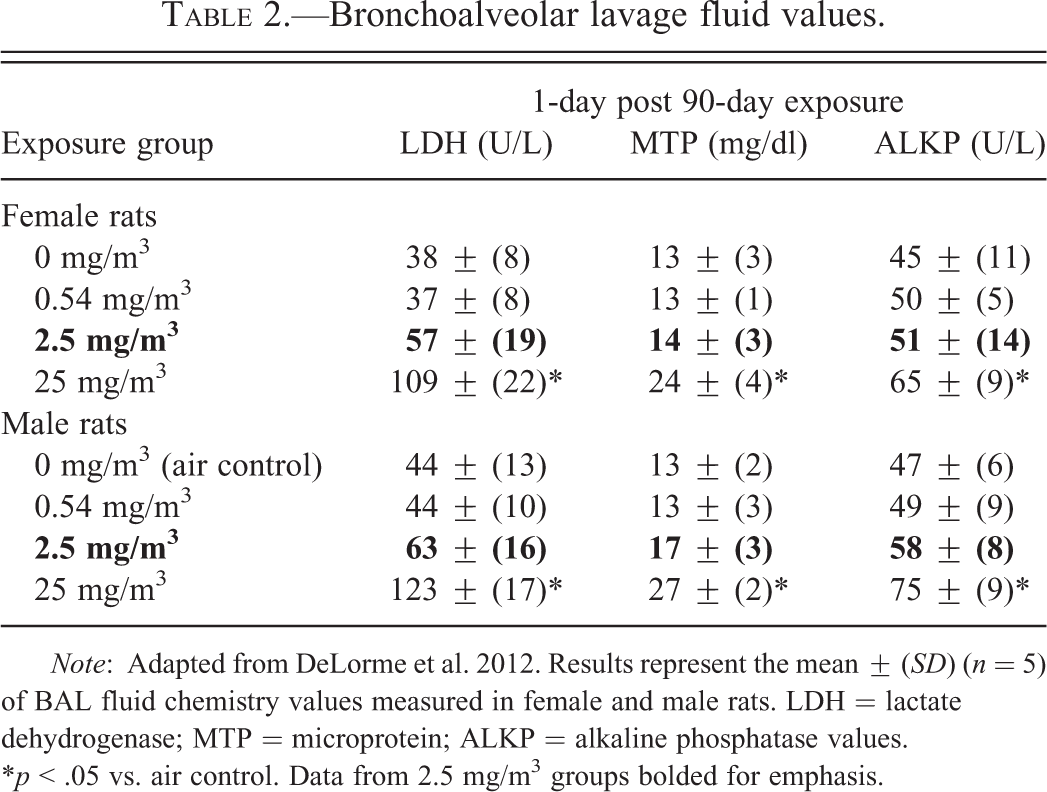
Note: Adapted from DeLorme et al. 2012. Results represent the mean ± (SD) (n = 5) of BAL fluid chemistry values measured in female and male rats. LDH = lactate dehydrogenase; MTP = microprotein; ALKP = alkaline phosphatase values. *p < .05 vs. air control. Data from 2.5 mg/m3 groups bolded for emphasis.
Thirteen-week exposures to 25 mg/m3 VGCFTM-H nanofibers produced significant increases in CP indices in airway/terminal bronchiolar, lung parenchymal (alveolar ducts), and subpleural regions, as determined by enhanced BrdU immunostaining versus air control values. Positive immunostained cells were often located in close proximity to particulate-containing alveolar macrophages. In contrast to the findings at the high-exposure concentration (25 mg/m3), no significant increases in CP indices versus controls were measured in rats exposed to either 0.54 or 2.5 mg/m3 CNF test substance (see Table 3).
BrdU cell proliferation in anatomical locations of respiratory tract.
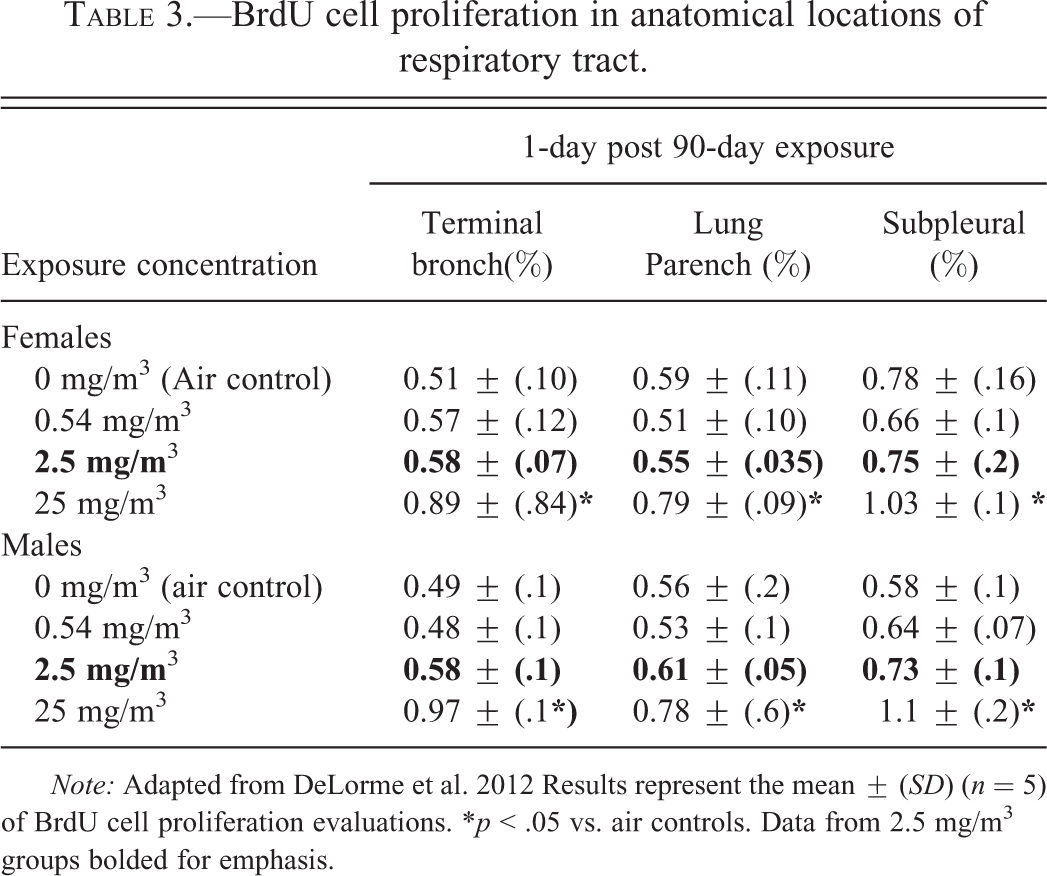
Note: Adapted from DeLorme et al. 2012 Results represent the mean ± (SD) (n = 5) of BrdU cell proliferation evaluations. *p < .05 vs. air controls. Data from 2.5 mg/m3 groups bolded for emphasis.
A small but significant increase (10%) in lung weights versus controls was measured in female rats but not in male rats exposed to 2.5 mg/m3. Significant lung weight increases versus controls were measured in male and female rats exposed to 25 mg/m3 CNF.
Histopathology
Following the end of 13-week aerosol exposures, extrapulmonary nanofibers were infrequently identified in the brain (choroid plexus), heart, liver, kidneys, spleen, intestinal tract, and mediastinal lymph nodes. The appearance of extrapulmonary fibers in organs was rare and the CNF test material was viewed by light microscopy only because the particulates were birefringent when polarized light was utilized. These observations indicate that small amounts of inhaled CNF test material had translocated from airspace to the systemic circulation. Perhaps more importantly, no adverse histopathological effects were observed in any systemic organs.
Exposure of rats to concentrations of 0.54, 2.5, or 25 mg/m3 produced a concentration-related accumulation of CNF within alveolar macrophages, and at 2.5 and 25 mg/m3, a subacute to chronic inflammation of the terminal bronchiole and lung parenchymal regions of the lung. In rats exposed to 2.5 mg/m3 CNF, microscopic assessments were characterized by the study pathologist as minimal subacute inflammation of the terminal bronchiole and lung parenchymal areas in rats exposed to 2.5 mg/m3 (grade 1 severity—7/10 each for males and females; see Figure 4A–C). In rats exposed to 25 mg/m3 CNF, histopathological observations included a slight subacute to chronic inflammation of the terminal airways and lung parenchymal areas, along with some thickening of interstitial walls, and hypertrophy/hyperplasia of type II pneumocytes (grade 2 severity—10/10 each for males and females; see Figure 5A–C).
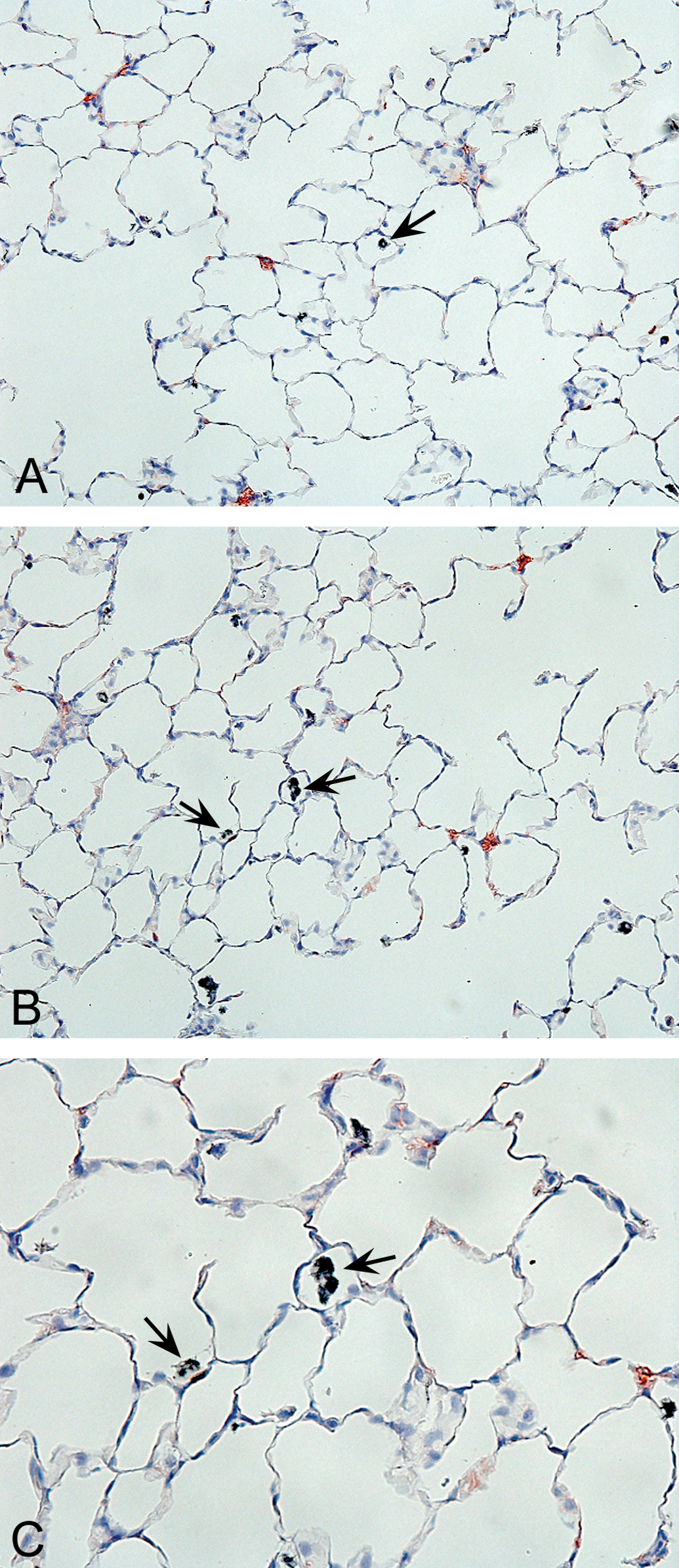
A, Light micrograph—low magnification lung tissue section from a male rat exposed to 2.5 mg/m3 VGCF™-H carbon nanofibers (CNFs) for 90 days, demonstrating occasional CNF-containing macrophages (arrows) within alveoli. Note the apparent normal lung architecture following 13 weeks of dust exposure (10× magnification). B, Light micrograph—higher magnification of lung tissue section from a male rat exposed to 2.5 mg/m3 VGCFTM-H CNFs for 90 days. This representative lung section demonstrates phagocytosis of CNF particulates by pulmonary macrophages in alveolar regions (note the black pigment-containing cells—see the arrows), which may have been responsible, in part, for the assessment of minimal inflammation by the study pathologist (20× magnification). C, Light micrograph—higher magnification of Figure 4B—a lung tissue section from a male rat exposed to 2.5 mg/m3 VGCFTM-H CNFs for 90 days. This representative lung section demonstrates phagocytosis of CNF particulates by pulmonary macrophages within alveoli (note the black pigment-containing cells—see the arrows) and denotes a normal physiological pulmonary response to the inhaled CNF test material (40× magnification).
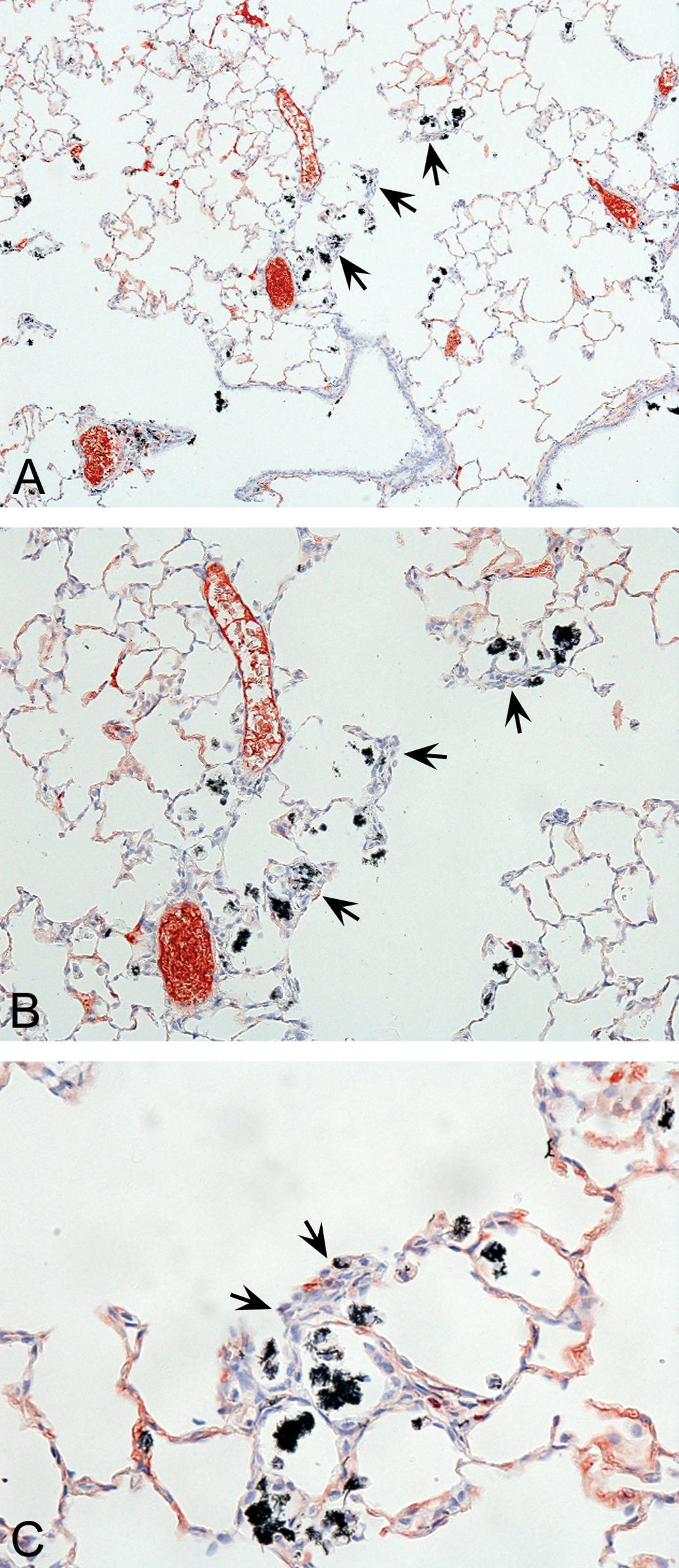
A, Representative lung tissue section from a male rat exposed to 25 mg/m3 VGCF™-H carbon nanofibers (CNFs) for 90 days. Terminal bronchioles and corresponding alveolar ducts are illustrated. Note the slight inflammation and phagocytic pulmonary macrophages (black pigment-containing material — see arrows) which have accumulated in alveolar duct regions. (10× magnification). B, Higher magnification of Figure 5A from a lung tissue section from a male rat exposed to 25 mg/m3 VGCFTM-H CNFs for 90 days. The accumulation of phagocytic macrophages located within alveolar ducts is more prominent in this light micrograph. Also note the slight thickening of alveolar walls in areas adjacent to accumulations of CNF-containing macrophages — see arrows (20× magnification). C, Higher magnification of a lung tissue section from a male rat exposed to 25 mg/m3 VGCFTM-H CNFs for 90 days. The accumulation of phagocytic macrophages (cells containing black-pigmented test material) is apparent within alveolar ducts. Also note the slight thickening of alveolar walls and hyperplastic cellular responses in areas adjacent to the accumulated macrophages — see arrows (40× magnification).
In the nasal passages, a nonspecific reaction to exposure was noted, characterized by the appearance of eosinophilic hyaline droplets in the nasal mucosa. These effects were viewed by the study pathologist as an adaptive and nonadverse response.
Discussion
Ninety-day inhalation exposures of CNF in male and female rats generally produced mild toxicological responses, which were focused almost exclusively on the respiratory tract. Other than documenting translocation of inhaled CNF to extrapulmonary sites and organs, no adverse toxicological effects were measured or observed outside the respiratory tract. These measurements included standard, toxicological, clinical/chemical, hematological, and histopathological evaluations.
Among all of the parameters measured in this study, only exposures to the highest CNF concentration, 25 mg/m3, produced low level but sustained pulmonary inflammatory responses (i.e., 11–12% neutrophils recovered in BAL fluids). Cellular inflammatory responses were correlated with significant increases versus controls in several cytotoxicity biomarker endpoints; as evidenced by enhanced BAL fluid LDH, MTP, and ALKP activity (Table 2). CP evaluations in the groups exposed to 25 mg/m3 CNF also revealed significantly enhanced cell turnover indices in airway, lung parenchymal, and subpleural regions of the respiratory tract (Table 3). The augmented changes versus controls in BALF and BrdU CP endpoints in rats exposed to 25 mg/m3 CNF correlated well with the finding of increased lung weights in male and female rats and the microscopic observation of slight inflammation and occasional interstitial wall thickening.
However, in contrast to the results measured at the high concentration, exposures to 2.5 mg/m3 CNF produced no significant increases versus controls in other sensitive biomarkers such as BAL fluid parameters or CP endpoints.
To be more specific, it is noteworthy that 90-day exposures at 2.5 mg/m3 produced no increased neutrophil inflammatory responses, despite the fact that 90% of BAL-recovered pulmonary macrophages contained nanofibers. Moreover, other sensitive measures of cytotoxicity and cell turnover results in various anatomical compartments of the respiratory tract were not different from control values. In contrast to the BAL fluid and CP results, lung tissue microscopy observations in the rats exposed to 2.5 mg/m3 revealed a finding of subacute minimal inflammation—concomitant with a 10% increase in female (but not male) lung weights. Indeed, it seems likely that the cellular components of this “minimal inflammatory response” were represented, in part, by the presence of pulmonary macrophages containing CNF, which had accumulated at sites of nanofiber deposition (see Figure 4A–C). Accordingly, this finding should be viewed as an adaptive physiological response to subchronic exposures to inhaled dusts.
Most standard Organization for Economic Cooperation and Development (OECD) 413 guideline studies for inhaled particulates or nanoparticulates do not specify inclusion of additional, supplementary studies such as BAL and CP evaluations. However, the implementation of these methods in the study experimental design, along with the traditional toxicological endpoints, serves to provide a more rigorous evaluation of the potential hazards of the inhaled test substance. Indeed the more robust study design is likely to foster greater mechanistic insights, resulting in a more thorough interpretation of the biological effects. A recent OECD Working Party on Manufactured Nanomaterials meeting was conducted to revisit the issue of guidance for conducting inhalation toxicity testing on nanomaterials. The recommendations in the report of the WPMN Expert Meeting on Inhalation Testing were the following:
The inclusion of BAL studies as essential complementary components to traditional subchronic inhalation toxicity (OECD 413) studies with particles and nanoparticles.
A strong consideration of CP studies (of various components of the respiratory tract) as recommended supplementary/complementary components to traditional subchronic inhalation toxicity (OECD 413) studies with particles and nanoparticles.
Development of better guidance criteria for establishing NOAELs in subchronic inhalation toxicity studies (i.e., OECD 413) with particles and nanoparticles as referenced in the OECD report (OECD 2012).
In previously reported pulmonary bioassay studies, the integration of BAL and CP assessments has been utilized as important screening assays to gauge the lung toxicity of inhaled or intratracheally instilled particulates (Warheit et al. 1997, 2007a, 2007b; Sayes, Reed, and Warheit 2007). In general, the BAL fluid and lung CP biomarkers have correlated well with morphological evaluations of particle-exposed lung tissues in rats and provided a more detailed, eclectic, and comprehensive toxicological assessment (Warheit et al. 1997, 2007a, 2007b). Apart from the implementation of morphological and biochemical assessments of lung injury following pulmonary exposures, other fundamental aspects of a study design provide critical components for assessing hazards following exposures. In this regard, essential experimental design components of a successful pulmonary bioassay study should include a dose-response and time course regimen, as well as the incorporation of benchmark control (positive and/or negative) particulates/test samples as key reference points, to facilitate an accurate interpretation of the toxicity findings. In past studies, results of the histopathology observations generally were consistent and provided convergence with the other measured pulmonary toxicological endpoints such as BAL biomarkers and CP biomarkers (Warheit et al. 1997, 2007a, 2007b).
The concept that cellular responses in affected organs due to long-term exposures may be considered adaptive effects rather than adverse findings is not unprecedented. In a published U.S. EPA Health Effects guidance document on hepatocellular hypertrophic effects, it was noted that increased liver size or weights may be reflective of an adaptation response, which is not necessarily an adverse effect. Accordingly, it was recommended that a WOE approach should be implemented to evaluate whether the liver effects are adverse or represent a normal physiological response to repeated test substance exposures. The suggested approach should consider the experimental design and duration of the study and could include indicators such as clinical chemistry (e.g., decreased albumin or increased alanine aminotransferase, alkaline phosphatase, etc.) endpoints, as well as histopathological effects, and expected physiological responses (US EPA 2002). It should be noted that in the CNF inhalation toxicity study discussed herein, the more sensitive BALF-derived inflammation and cytotoxicity biomarkers and CP endpoints were not different when comparing the mid-exposure levels versus control values. Only the histopathological (i.e., minimal inflammation) evaluation was considered an adverse (albeit minor) response. In light of all of the study findings, should this microscopic assessment be considered an adverse or adaptive response?
To summarize the key learnings of a subchronic inhalation study with CNFs, a lack of concurrence in morphological, biochemical, and CP results was noted at the mid-exposure level (2.5 mg/m3). This analysis was important, because it served to identify a discordance in the study findings, as the histopathological observations seemed to outweigh some other measurements, when identifying the adverse exposure effect level of the study. Indeed, the lack of convergence between the histopathological findings (minimal inflammation) and the biochemical and CP (no significant effects) results raises important general questions about the necessary criteria required for adequate interpretation of study results. This hazard issue has important ramifications, since the experimental design of this study was particularly robust. As a consequence, it is recommended that there should be a serious and wide-ranging discussion regarding guidance on two primary issues:
the need for an integrated assessment of all study endpoints in determining a NOAEL—with equal weight placed on functional/ mechanistic/ morphological endpoints; and
the need for consistency in understanding adaptive versus adverse responses for inhaled particulate exposures.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The Authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Showa Denko KK, Tokyo, Japan.
Abbreviations
Acknowledgments
This study was sponsored by Showa Denko KK, Tokyo, Japan. The following individuals made significant technical contributions to the conduct and implementation of this study: Dr. Deborah Banas, Dr. Randy Frame, Dr. Toshihiro Arai, Dr. Yukihiro Muro, John Nojag, Bill Ellis Morgan Golt, David Grinstead, Elizabeth Wilkinson, Lisa Lewis, Carolyn Lloyd, Steve Records, Don Hildabrant, Antidio Lorenzo, Jeff Holt, Melissa Fallers, and Tracey White. Dr. Shekhar Subramoney supplied the TEM micrographs. Drs. Jun Ogawa, Matt Matsumoto, Greg Sykes, and Robert Roy provided helpful insights into the preparation of the manuscript. Mark Greenwood also provided important insights into the preparation of the manuscript. The authors also want to acknowledge Beth Mahler for incorporating the figures into the manuscript as well as the thorough, outstanding, and insightful comments provided by the reviewers of this manuscript, which clearly have contributed to the significant improvement in the writing style and presentation of this article.
