Abstract
MicroRNA (miR)-9-5 p has been shown to affect lung cancer progression and lung fibrosis, but the efficacy of miR-9-5 p in acute lung injury (ALI) remained indefinite. The study was performed to probe the modulating mechanism of miR-9-5 p in ALI via regulating macrophage polarization. The ALI mouse model was established and blood samples of ALI patients were obtained. MiR-9-5 p levels in ALI mice and ALI patients were detected. Mouse pulmonary macrophages were extracted from bronchoalveolar lavage fluid and polarized into M1 and M2 macrophages. Intervention of miR-9-5 p expression was performed to observe the effects on M1 polarization and M2 polarization in lung macrophages, inflammatory factors in BALF, wet/dry weight ratio (W/D) in lung tissues, myeloperoxidase (MPO) activity in lung tissues, and lung tissue lesion condition. MiR-9-5 p levels were elevated in the lung tissues of ALI mice and ALI patients. MiR-9-5 p silencing could repress lung macrophages in ALI mice polarized toward the M1 phenotype and promoted the polarization toward the M2 phenotype, reduced the lung lesions, the lung water content, and the secretion levels of the pro-inflammatory factors TNF-α, IL-6, and IL-1β in BALF, increased the secretion of the anti-inflammatory factor IL-10, as well as impeded the MPO activity in the lung tissues of ALI mice. MiR-9-5 p deletion ameliorates LPS-induced inflammatory infiltration in lung tissues via inhibiting the polarization of mouse lung macrophages to the M1 phenotype and promoting the polarization to the M2 phenotype.
Keywords
Introduction
Acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) are a series of lung changes resulting from various lung damages, commonly leading to high morbidity. 1 ALI is featured by acute respiratory absence with tachypnea, cyanosis refractory to oxygen, degenerative lung compliance, and diffuse alveolar infiltrates on chest X-ray. 2 The inducers of ALI include various diseases, such as sepsis, pneumonia, trauma, acute pancreatitis, aspiration of gastric contents, and near-drowning. 3 Most patients who are afflicted by ALI frequently develop diseases like short-term dyspnea all the way and refractory respiratory failure. 4 As a life-threatening clinical syndrome, the critical care and therapeutic modalities of ALI still encounter challenging obstacles. 5 Thus, it is essential to investigate promising therapeutic targets for ALI.
MicroRNAs (miRNAs) are small noncoding RNAs that function as effective modifiers of multiple respiratory diseases. Specifically, some miRNAs have been reported to improve cellular damage and mitigate immune response by targeting specific molecules in lung-related diseases. 6 For instance, it has been found that miR-9-5 p exhibits a high level in both lung cancer and lung fibrosis, thus promoting the malignant progression of the disease.7,8 In non-small lung cancer cells, miR-9-5 p also exhibits a high level. 9 Macrophages are primary orchestrators in the pathogenesis of ALI, and the modulation of macrophage polarization affects the ALI prognosis. 10 The M1 and M2 macrophage phenotypes are implicated in the pro-inflammatory and pro-fibrogenic signatures in ALI. 11 A previous study has documented that miR-9-5 p promotes M1 macrophage polarization and inhibits M2 macrophage polarization. 12 Nevertheless, the efficacy of miR-9-5 p in ALI remains to be elucidated. The study was designed to assess the efficiency of miR-9-5 p in ALI via the modulation of macrophage polarization, thus offering insightful therapeutic modalities for ALI treatment.
Materials and Methods
Ethics Statement
The study operations for clinical samples were approved by the ethics committee of The Second Affiliated Hospital of Shandong First Medical University. All the participants have provided informed consent. The animal protocol was approved by the Institutional Animal Care and Use Committee of The Second Affiliated Hospital of Shandong First Medical University.
Clinical Data Collection
Sixty-eight patients with ALI treated in The Second Affiliated Hospital of Shandong First Medical University were recruited and provided peripheral blood samples. The collected peripheral blood samples were centrifuged at 3000 r/min for 10 min, and the serum was kept at −20°C for subsequent reverse transcription quantitative polymerase chain reaction (RT-qPCR) and enzyme-linked immunosorbent assay (ELISA) testing. All ALI patients met the 1994 American/European Consensus Conference definition: (1) Patients displayed high-risk factors for direct lung injury or indirect injury; (2) Acute onset, heart failure and fluid overload did not explain the respiratory failure, nor did they have hypertension pulmonary edema; (3) Specific test data included hypoxemia: PaO2/FiO2 ≤ 300 mmHg; (4) Chest imaging changes with infiltrative shadows in both lungs on chest X-ray or computed tomography scan images; (5) Excluded: pulmonary arterial wedge pressure ≤ 18 mmHg or left atrial hypertension. Clinical data of ALI patients were complete. Healthy individuals for a physical examination in our hospital were also recruited (n = 30).
Establishment and Treatment of ALI Mouse Models
Healthy specific pathogen-free grade ICR mice (aging 4 w, weighing 18-25 g) were offered by the Laboratory Animal Center of Shandong University (Shandong, China). ICR mice were used to establish ALI mouse models of septic shock. Under sterile conditions, coliform lipopolysaccharide (LPS) (Sigma Aldrich, St. Louis, USA) was instilled into the trachea of mice (5 mg/kg) to establish the ALI mouse models. The normal group mice (10) were instilled with the same amount of normal saline.
ALI modeled mice were randomly classified into 3 groups (10 mice/group): ALI group, antagomiR negative control (NC) group (ALI mice injected with antagomiR NC via tail vein), and miR-9-5 p antagomiR group (ALI mice injected with miR-9-5 p antagomiR via tail vein). Two mice of the ALI group died during the modeling process, three of the NC group died during modeling, and no death occurred in other groups. miR-9-5 p antagomir and antagomiR NC were obtained from Dharmacon Corporation (Lafayette, CO, USA). A total of 20 μg oligonucleotides required by each group were diluted with RNA free H2O to 5 μg/μL, and added with 3.2 μL in vivo-jet Polyethyleneimine (PEI) (PT-201-10G, Polyplus transfer, Strasbourg, France), and 5% glucose solution to fill the volume to 50 μL for transfection complex preparation, and followed by 15-min incubation. Then, 1 h after modeling, the mice were subjected to transfection complex injection through the tail vein. The mice in the normal group were injected with 5% glucose solution. 13
Bronchoalveolar Lavage Fluid (BALF) Collection
The mice were treated for 48 h, and then anesthetized with a mixture of ketamine (87.5 mg/kg) and toluene thiazide (12.5 mg/kg). The mice were euthanized, and underwent neck dissection. The trachea was fully exposed and intubated. The entire lung was isolated. The right lung of mice was ligated with silk thread and harvested, and in the guide tube, the left lung was slowly infused with 3 mL 4°C precooled PBS, and BALF was slowly backdrawn after 30 s and repeated three times. The collected BALF was subjected to 20-min centrifugation at 3000 r/min and 4°C. The supernatant was preserved at −80°C. 14
Isolation of Alveolar Macrophages (AMs) via BALF
BALF was subjected to 5-min centrifugation at 500 × g. Precipitated cells were collected, which were washed with Roswell Park Memorial Institute (RPMI)-1640 medium containing 10% fetal bovine serum (FBS) and 0.1% 100 × penicillin-streptomycin solution twice. Cells were resuspended to obtain the cell suspension. Subsequently, cells were seeded in 12-well plates (2 × 105 cells per well). The cells were subjected to 90-min incubation to allow the cells to adhere fully to the wall. The unadhered cells were eliminated by PBS washing twice, and the adhered cells were AMs.
For cell treatment, RPMI-1640 containing 10% FBS and 1% penicillin was used for AMs culture. When the cells reached 70%-80% confluence, the cells were digested. The cell density was adjusted to 5 × 104 cells/mL and seeded into 24-well plates. The miR-9-5 p mimic, miR-9-5 p inhibitor and their NC (Han Biotechnology Co. Ltd., Shanghai, China) were transfected into AMs by the Lipofectamine 2000 reagent (Invitrogen, CA, USA).
AM M1/M2 Polarization Culture in Vitro
AM M1 or M2 polarization was conducted with reference to previous literature. 15 Specifically, LPS (1 μg/mL) and interferon (IFN)-γ (100 ng/mL) or interleukin (IL)-4 (40 ng/mL) and IL-13 (20 ng/mL) were supplemented in the culture medium and then co-incubated with AMs for 48 h. Primary AM was polarized towards M1 type by LPS and IFN -γ, while AM was polarized towards M2 by IL-4 and IL-13 stimulation.
ELISA
The inflammatory factors IL-6, IL-10, tumor necrosis factor (TNF)-α, and IL-1β levels in BALF were tested. The following ELISA kits (all from Abcam, Cambridge, MA, USA) were used: IL-6 (ab100712), IL-10 (ab46103), TNF-α (ab100747), and IL-1β (ab197742). ELISA kits were subjected to 20-min equilibration for wash solution preparation. After dissolution, the reaction plate was added with 100 μL standard samples for standard curve drawing. Then, 100 μL samples were added to the reaction wells and incubated for 90 min. After washing, samples were added to 100 μL antibody solution with biotin, and cultured for 1 h. After washing, 100 μL fresh enzyme combination reaction working solution were added with samples and followed by 60-min incubation. The plate was added with 100 μL substrate, and followed by 15-min incubation. The reaction termination was achieved by stop solution (2 mol/L H2SO4). The optical density (OD) value was evaluated at 450 nm by a microplate reader (BioTek Instruments Inc., Winooski, VT, USA).
Determination of the Wet-To-Dry Weight Ratio (W/D) of the Lungs
After the mice were sacrificed, the lungs were isolated. Part of the right lung tissues were removed with absorbent paper to eliminate the blood from lung surface and weighed, which was the wet weight. The lungs were then put in a 80°C oven for 48 h. The lungs were weighed again and the value was recorded, which was the dry weight. W/D ratio = wet weight/dry weight.
Myeloperoxidase Activity in Lung Tissues
MPO is a functional and activation marker of neutrophils, and the changes in its level and activity represent the functional and active status of neutrophil polymorphonuclear leukocytes. When there is damage to the lung tissue, MPO is produced in abundance, leading to inflammation. Mouse lung tissues were well ground in PBS through a homogenizer to get a 10% lung tissue homogenate conforming to the MPO kit (Shanghai Enzyme Link Biotechnology Co., Ltd., Shanghai, China). The OD was measured at 450 nm using a spectrophotometer. MPO levels in lung tissues were calculated from the standards and expressed in U/g.
H&E Staining
After fixation of the lung tissues with 10% neutral formaldehyde solution over 24 h, the fixative solution was washed off with distilled water after fixation. Lung tissues were subjected to conventional gradients of alcohol (ethanol concentrations of 70%, 80%, 90%, 95%, and 100%) for 1 min/time, followed by xylene permeabilization for 2 times for 5 min/time, wax immersion and paraffin embedding, and finally sliced into 4 μm section. The sections were put at 80°C for 1 h for quick drying. After cooling, the sections were dewaxed with xylene (twice, 5 min/times), and rehydrated with gradient alcohol (100%, 95%, 90%, 80%, and 75%) and distilled water (each for 3 min), followed by staining with hematoxylin (Beijing Solaibao Technology Co., Ltd., Beijing, China) for 4 min. Subsequently, the sections were subjected to 10-s fractionation with hydrochloric acid alcohol, 2-min staining with eosin solution (Shanghai Bogu Biotechnology Co., Ltd., Shanghai, China), gradient alcohol dehydration, and xylene permeabilization twice. The slides were air-dried in the fume hood, neutral gum drops were placed on the slides to seal them, and the images were observed in the light microscope (Shanghai Cai Kang Optical Instruments Co. Shanghai, China) for pathological changes. The results were calculated by the following four-item scores: (i) alveolar congestion, (ii) hemorrhage, (iii) alveolar and interstitial inflammation (interstitial neutrophil colonization or infiltration), and (iv) degree of pulmonary edema (alveolar wall thickening or hyaline membrane formation). Scoring criteria: 0, minimal injury; 1, mild injury; 2, moderate injury; 3, severe injury; 4, most severe injury. The sum of the items was the lung injury score, with a total score of 16.
Immunofluorescence Assay
Tissue sections were dewaxed and placed in citrate buffer. Samples were boiled 3 times in the microwave for 5 min for antigen recovery. Thereafter, the tissue sections were incubated with anti-CD68 (1:100, ab283654, Abcam), iNOS (1:100, ab15323, Abcam), and CD206 (1:200, DF4149, affbiotech, Cambridge, UK) for 24 h. Samples were subjected to incubation with goat anti-rabbit IgG H&L (1:100, ab6717, Abcam) secondary antibody for 1 h. Tissues were counter-stained with 4’,6-diamidino-2-phenylindole (DAPI) for 20 min without light exposure, and sealed with anti-fluorescence quenching agent. The observation was made using a fluorescent microscope (Olympus Corporation, Tokyo, Japan). Positive cells in the micrographs were counted through the Image J software (National Institutes of Health, Maryland, USA). Five random fields of view were checked.
RT-qPCR
Total RNA extraction was performed by the Trizol kit (Invitrogen, Carlsbad, CA, USA). Reverse transcribe of RNA into complementary DNA was achieved through the PrimeScript RT kit (Takara Biotechnology, Dalian, China) and One-Step PrimeScript miRNA cDNA Synthesis Kit (Takara Biotechnology). The reaction solution was taken for fluorescence PCR as the instructions of SYBR® Premix Ex TaqTM II kit (Takara). Fluorescece PCR was performed on an ABI PRISM® 7300 system (ABI, CA, USA). Primers were synthesized by Wuhan Bojie Bioengineering Co., Ltd. (Wuhan, China). β-actin was the target internal reference, and the primer sequences are presented in Supplementary Table 1. The 2−ΔΔCt method was applied for gene level assessment.
Statistical Analysis
All tests were performed using SPSS 20.0 (IBM Corp. Armonk, NY, USA) and the GraphPad software version 7.0. All measurement data were represented as mean ± standard deviation. A t-test was employed for data analysis of two groups, one-way analysis of variance was adopted for data comparison in multiple groups and Tukey’s tests were implemented. The chi-square test and Fisher’s exact test were used for correlation analysis of the clinical baseline characteristics. The results of the data were statistically significant when P < .05.
Results
ALI Patients Display Increased miR-9-5p Levels and Severe Inflammatory Infiltration
To observe miR-9-5 p efficacy in clinical ALI patients, we collected blood samples from ALI patients and adopted RT-qPCR to assess miR-9-5 p expression. First, we observed no significant differences between patients and healthy controls at baseline levels including gender, age, body max index, history of smoking and alcohol consumption, and other factors (Supplementary Table 2). MiR-9-5 p levels were significantly high in ALI patients (Figure 1(a)). ELISA assay indicated that ALI patients exhibited elevated serum levels of inflammatory factors TNF-α, IL-1β, IL-6 and decreased IL-10 level (Figures 1(b) to (e)). ALI patients display increased miR-9-5 p levels and severe lung inflammatory cell infiltration. A, miR-9-5 p levels in serum was assessed by RT-qPCR; B-E, for TNF-α, IL-1β, IL-6, and IL-10 levels in serum were tested by ELISA. *P < .05 vs. the healthy group.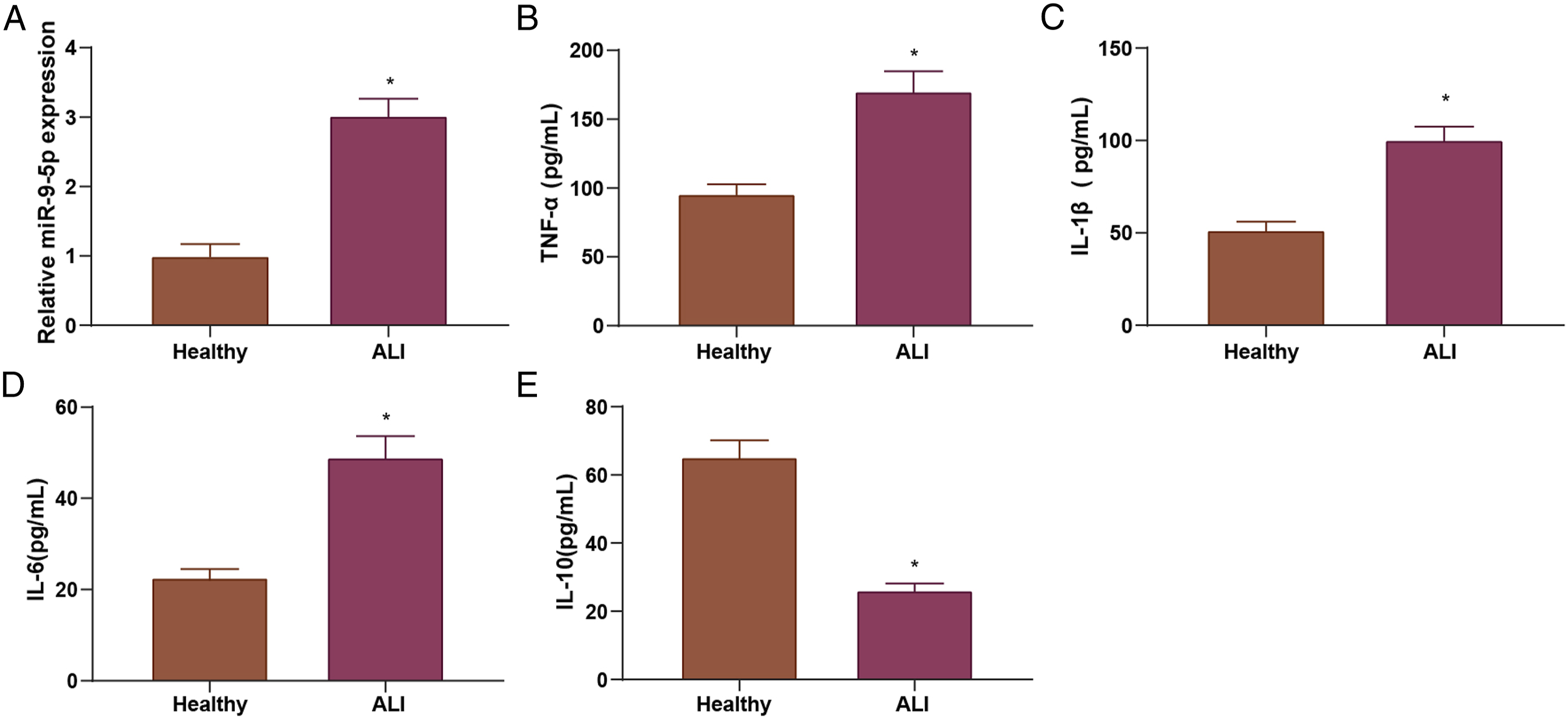
LPS-Induced ALI Mice Exhibit Deteriorated Inflammatory Infiltration in Lung Tissues
To assess the inflammatory infiltration in the lung tissues of LPS-induced ALI mice, the histopathological changes in normal mouse lung and ALI mouse lung were observed by HE staining. The results indicated that the degree of lung tissue damage was significantly exacerbated in LPS-induced ALI mice in comparison with the normal mice. Specifically, the ALI mice had more alveolar structural destruction, alveolar rupture and fusion, and the alveoli exhibited obvious congestion and hemorrhage, widened interstitium, many inflammatory cells infiltrating the alveolar cavity and interstitium, and high lung tissue damage scores, whereas the normal mice had basically normal lung tissue structure and low lung tissue damage scores (Figures 2(a) and (b)). The W/D assessment results indicated the degree of pulmonary edema, and MPO activity was an important indicator of neutrophil function and aggregation degree, indirectly reflecting the degree of inflammatory infiltration of lung tissues. The effect of LPS on the inflammatory response in the lungs of ALI mice could be reflected by measuring the changes of W/D and MPO activity. The results revealed that both lung W/D ratio and MPO activity in lung tissues were elevated in ALI mice in comparison to normal mice (Figures 2(c) and (d)). The ELISA assay showed that TNF-α, IL-1β, and IL-6 levels were elevated while IL-10 level was depleted in LPS-induced ALI mice in comparison to normal mice (Figure 2(e)). LPS-induced ALI mice exhibit inflammatory infiltration in lung tissues. A, lung tissue sections of ALI mice were observed by H&E staining; B, lung tissue damage score of ALI mice was calculated; C, W/D ratio was assessed; D, lung tissue MPO activity of ALI mice was analyzed; E, inflammatory factor expression in BALF of ALI mice was tested by ELISA, *P < .05 vs. the normal group.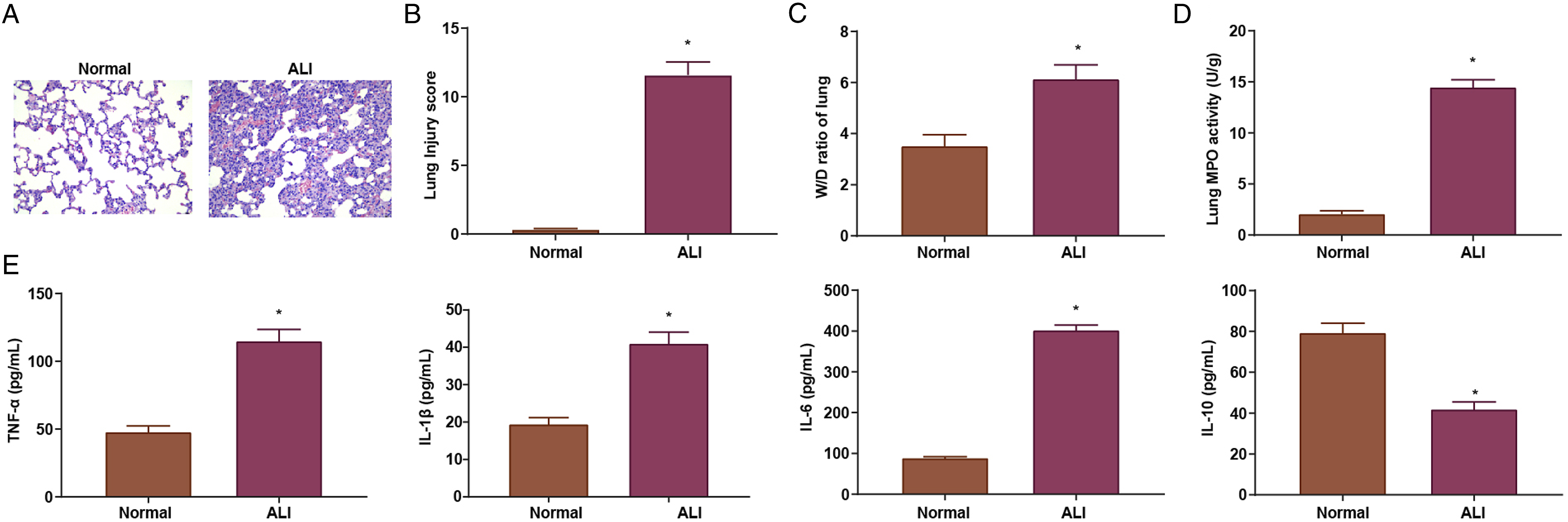
The above outcomes reflected that the ALI mouse model was successfully constructed and ALI mice displayed aggravated inflammatory response.
LPS-Induced ALI Mouse Exhibits Increased miR-9-5p Levels and Exacerbated M1 Macrophage Polarization
To assess miR-9-5 p levels and M1/M2 macrophage polarization in ALI mice, we first examined the macrophage signature factors of M1 macrophage markers iNOS, CD86 and M2 macrophage markers Arg1 and CD206 as well as miR-9-5p levels by RT-qPCR. The outcomes revealed that, in the lung tissue of LPS-treated ALI mice, Arg1 and CD206 levels were reduced, while iNOS, CD86 and miR-9-5p levels were augmented (Figures 3(a) and (b)). Lung macrophage M1 (CD68/iNOS/DAPI) and M2 (CD68/CD206/DAPI) expression was examined by immunofluorescence staining, showing that the CD68+ iNOS+ cells in lung tissues was elevated and that of CD68+CD206+ cells in lung tissues was depleted in ALI mice (Figure 3(c)). LPS-induced ALI mice exhibited increased miR-9-5 p expression and exacerbated M1 macrophage polarization. A, M1-M2 macrophage-related factor levels in ALI mice were assessed by RT-qPCR; B, miR-9-5 p levels in ALI mice were tested by RT-qPCR; C, M1 (CD68/iNOS/DAPI) and M2 (CD68/CD206/DAPI) expression in lung macrophages were detected by immunofluorescence assay; *P < .05 vs. the normal group.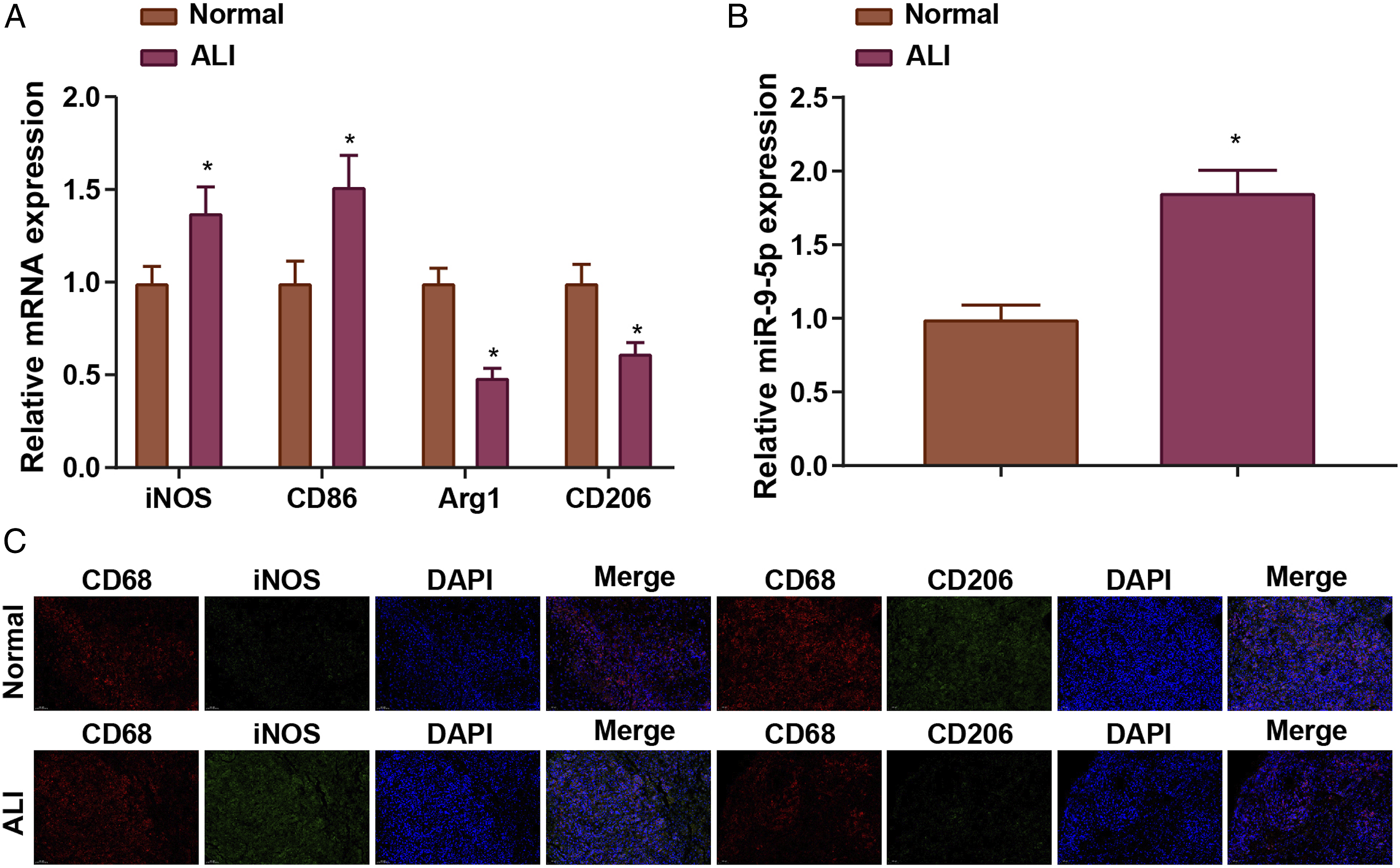
Regulation of Alveolar Macrophage Polarization by miR-9-5p
To clarify whether miR-9-5 p could promote M1/M2 polarization of AMs, we obtained AMs by isolation and then promoted M1 polarization of AMs by LPS and IFN-γ treatment, and promoted M2 polarization of AMs by IL-4 and IL-13 treatment. The detection of marker genes of M1 and M2 AMs were assessed by RT-qPCR. The findings indicated that LPS and IFN-γ treatment promoted M1 marker genes iNOS and CD86 levels in AMs, whereas M2 macrophage markers Arg1 and CD206 levels were reduced; in contrast, IL-4 and IL-13 treatment suppressed iNOS and CD86 levels, while levels of Arg1 and CD206 were elevated (Figures 4(a) and (b)). RT-qPCR also uncovered that LPS and IFN-γ treatment increased miR-9-5 p levels in AMs, while IL-4 and IL-13 treatment inhibited miR-9-5 p expression in AMs (Figures 4(c) and (d)). Regulation of alveolar macrophage polarization by miR-9-5 p. A/B, M1-type marker genes and M2-type marker genes levels in alveolar macrophages by LPS and IFN-γ stimulation were tested by RT-qPCR; C/D, M1-type marker genes and M2-type marker genes levels in alveolar macrophages by IL-4 and IL-13 stimulation were tested by RT-qPCR; E/F, M1/M2 macrophage marker gene levels were assessed by RT-qPCR after miR-9-5 p mimic or miR-9-5 p inhibitor treatment; *P < .05 vs. the mimic NC group; #P < .05 vs the inhibitor NC group.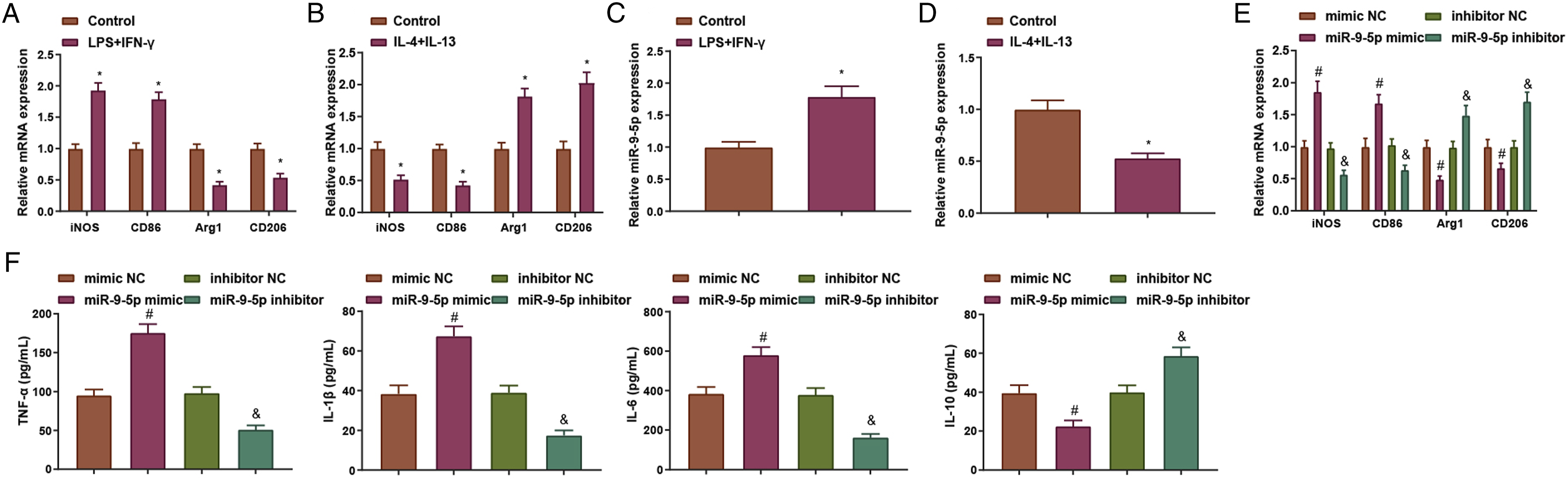
Further, AMs were transfected with miR-9-5 p mimic or miR-9-5 p inhibitor for M1/M2 polarization. RT-qPCR assay showed that miR-9-5 p mimic promoted the expression of markers iNOS and CD86 in M1 macrophages, while restricted Arg1 and CD206 levels; whereas miR-9-5 p inhibitor treatment inhibited iNOS and CD86 levels in M1 macrophages but promoted the Arg1 and CD206 levels (Figure 4(e)). ELISA assay unveiled that miR-9-5 p mimic treatment promoted TNF-α, IL-6, and IL-1β levels, and suppressed IL-10 level. miR-9-5p inhibitor treatment exhibited an inverse trend (Figure 4(f)). The above discoveries disclosed that miR-9-5 p inhibition promoted M2-type polarization of AMs.
MiR-9-5p Reduction Ameliorates LPS-Induced ALI by Promoting M2 Polarization
To figure out the ameliorative effect of miR-9-5 p on LPS-induced ALI, we treated the ALI mouse model with miR-9-5 p antagomiR. The mice were divided into the normal group, ALI group, antagomiR NC group and miR-9-5 p antagomiR group. The results demonstrated that miR-9-5 p levels were repressed after treated with miR-9-5 p antagomiR (Figure 5(a)). After miR-9-5 p silencing in ALI mice, HE staining results showed that pathological inflammatory damage in lung tissues was mitigated, and pathological sections showed reduced inflammatory cell infiltration, hemorrhage and a decrement in damage score, as well as in MPO activity and W/D in lung tissues (Figures 5(b) to (e)). ELISA assay that examined the levels of inflammatory factors in BALF revealed that miR-9-5 p silencing repressed TNF-α, IL-1β, and IL-6 levels and increased IL-10 level (Figure 5(f)). MiR-9-5 p reduction ameliorated LPS-induced ALI by promoting M2 polarization. A, for miR-9-5 p expression level in ALI mice was detected by RT-qPCR after miR-9-5 p silencing; B, lung tissue sections were observed by H&E staining after miR-9-5 p silencing; C, lung tissue injury score were calculated after miR-9-5 p silencing; D, W/D ratio was assessed after miR-9-5 p silencing; E, lung tissue MPO activity was analyzed after miR-9-5 p silencing; F, inflammatory factor expression in BALF was detected by ELISA after miR-9-5 p silencing. *P < .05 vs. the normal group; #P < .05 vs. the antagomiR NC group.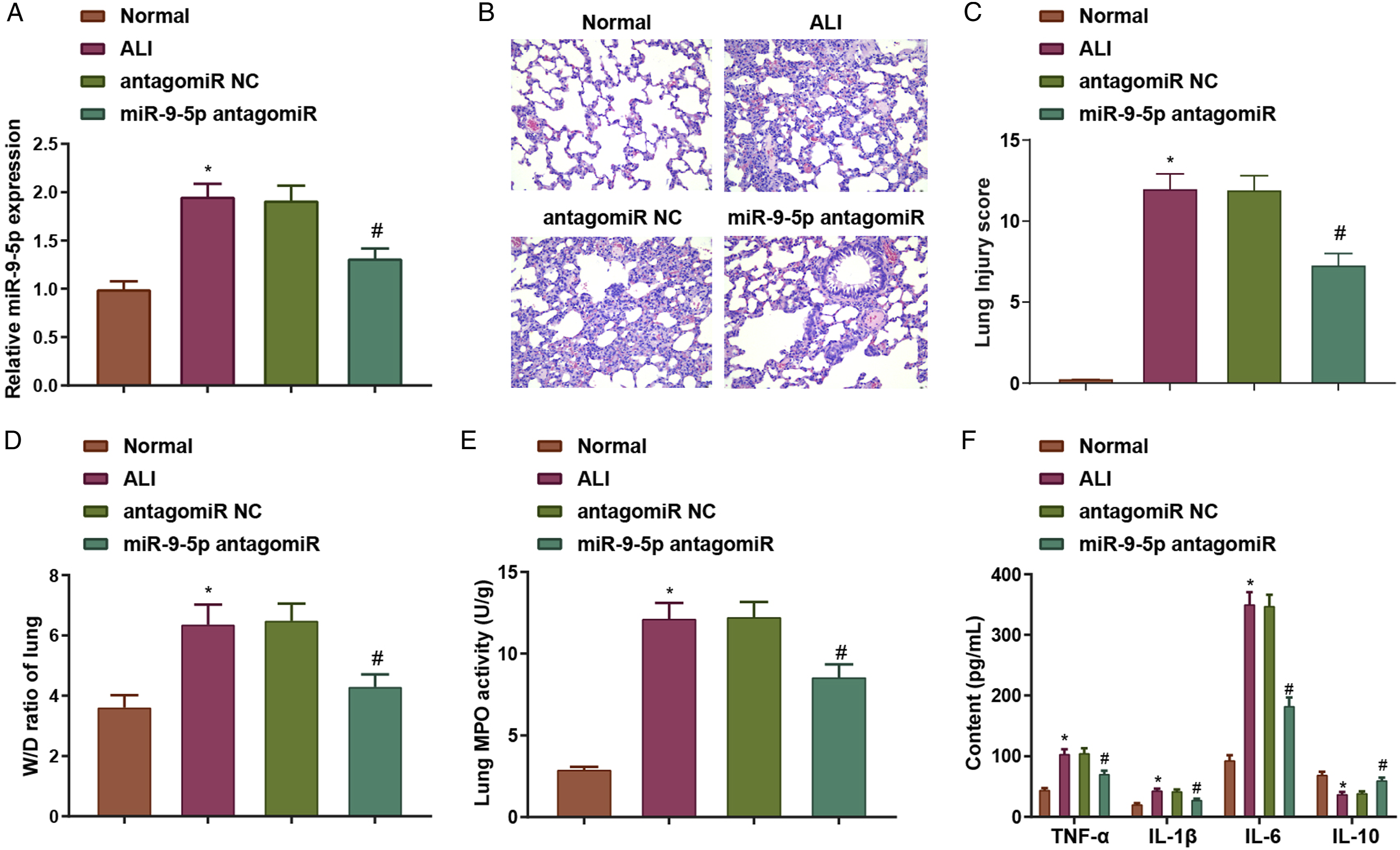
Thereafter, AM characteristic factors iNOS, CD86, Arg-1 and CD206 levels in each group of mice were tested by RT-qPCR, which reflected that miR-9-5 p antagomiR-treated mice inhibited the expression of iNOS and CD86 yet promoted the expression of Arg-1 and CD206 (Figure 6(a)). The results of immunohistochemical staining indicated that miR-9-5 p antagomiR treatment inhibited the CD68+iNOS+ cells in lung tissues and promoted CD68+CD206+ cells in lung tissues (Figure 6(b)). miR-9-5 p reduction ameliorated LPS-induced ALI by promoting M2 polarization. A, iNOS, CD86, Arg-1, and CD206 levels in alveolar macrophages were detected by RT-qPCR after miR-9-5 p silencing; B, the fluorescence of M1/M2 type markers was assessed by immunohistochemical staining after miR-9-5 p silencing. *P < .05 vs. the normal group; #P < .05 vs. the antagomiR NC group.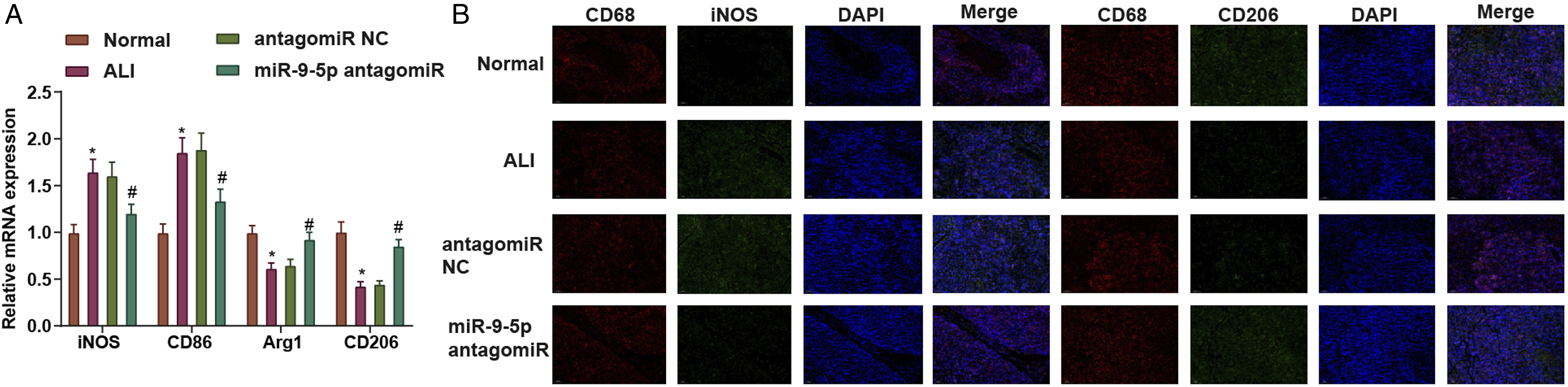
These findings unraveled that miR-9-5 p deficiency promoted M2 polarization in AMs and thereby alleviated LPS-induced ALI.
Discussion
ALI and ARDS are featured by a severe inflammatory response within the lungs and severely exacerbate gas exchange as a result of alveolar-capillary barrier disruption and pulmonary edema. However, currently there are limited viable molecular-based ALI therapies. 16 To address the obstacles, the study focused on the mechanism of miR-9-5 p in ALI development. Collectively, miR-9-5 p deficiency contributes to relieving inflammatory response in lung tissues of ALI mice via the regulation of macrophage polarization.
MiR-9-5 p has been found to exert effects on multiple lung-related diseases. Tang et al. have reported that miR-9-5 p is elevated in lung adenocarcinoma tissues in comparison with the para-carcinoma tissues. The overexpression of miR-9-5 p is positively correlated with the metastasis and advance stage of lung adenocarcinoma. 8 Similarly, in non-small cell lung cancer, hsa-miR-9-5 p has been explored to display higher level in the miRNA-gene regulatory mechanism. 17 The same findings have also been validated by Yang et al., who have elucidated that serum miR-9-5 p is augmented in non-small-cell lung cancer patients compared with normal controls. Functionally, miR-9-5 p augmentation is implicated in positive lymphatic metastasis and distal metastasis. 18 High levels of miR-9-5 p have also been detected in lung specimens from patients with idiopathic pulmonary fibrosis as well as in stage I lung adenocarcinoma tissues.18,19 Previous researchers have further elucidated the pro-inflammatory roles of miR-9-5 p regulatory network in chronic obstructive pulmonary disease. 20 Partly in consistent with previous findings, our study manifested that miR-9-5 p levels were augmented in ALI patients and LPS-induced ALI mice. MiR-9-5 p elevation is associated with deteriorated inflammatory infiltration.
MiR-9-5 p has also been revealed to affect the macrophage polarization and inflammatory responses in various diseases. For instance, the enforced miR-9-5 p expression can stimulate M1 cell polarization during osteoarthritis progression. 21 Lu et al. have found that M1 polarization leads to the enrichment of miR-9-5 p expression, involving in immune responses and signal transduction. 22 As for the effects of miR-9-5 p depletion, Ma et al. have elucidated that miR-9-5 p is accounted for inducing proinflammatory mechanism in sepsis, whereas the impairment of miR-9-5 p is effective to stimulate M2 macrophage polarization. 23 In addition, miR-9-5 p deficiency contributes to repressing pulmonary fibrosis. 24 In chondrocytes, the promotive inflammatory response in can be partly counteracted by miR-9-5 p reduction, uncovering the improving impacts of miR-9-5 p silencing in chondrocytes. 25 More specifically, in vivo decrement of miR-9-5 p by a specific antagomir in a murine acute myocardial infarction model effectively improve fibrosis and mitigate inflammatory response, thus promoting post-myocardial infarction heart function. 26 In line with previous discoveries, the study disclosed that miR-9-5 p silencing could repress the inflammatory infiltration in ALI mice by reducing TNF-α, IL-1β, and IL-6 levels and elevating IL-10 levels. As for macrophage polarization, the study has also validated that miR-9-5 p depletion promoted M2 macrophage polarization through increasing CD68/CD206/DAPI levels.
In summary, this study demonstrated that miR-9-5 p is expressed at a high level in ALI patients and ALI mice. MiR-9-5 p deficiency could mitigate inflammatory infiltration in ALI mice by repression of M1 macrophage polarization and promotion of M2 macrophage polarization. By highlighting the regulatory efficacy of miR-9-5 p in ALI, this study may pave the pathway for ALI treatment. Nevertheless, the potential molecular network of miR-9-5 p and its downstream genes should be further probed in future work.
Supplemental Material
Supplemental Material - MicroRNA-9-5p Is Involved in Lipopolysaccharide-Induced Acute Lung Injury Via the Regulation of Macrophage Polarization
Supplemental Material for MicroRNA-9-5p Is Involved in Lipopolysaccharide-Induced Acute Lung Injury Via the Regulation of Macrophage Polarization by Hao Li, Weimi Hu, Yueyue Lin, Tengxiao Xu, Xianjing Zhang, and Chen Wang in International Journal of Toxicology
Footnotes
Acknowledgement
We thank the associate editor and the reviewers for their useful feedback that improved this paper.
Author Contributions
Li, H. contributed to conception, contributed to acquisition, and critically revised manuscript Hu, W. contributed to design and critically revised manuscript; Lin, Y. contributed to acquisition and analysis and critically revised manuscript; Xu, T. contributed to analysis and critically revised manuscript; Zhang, X. contributed to conception and critically revised manuscript; Wang, C. contributed to design, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
