Abstract
Because precise information as to the toxicity of vanadium is required for practical use of vanadium compounds as antidiabetic drugs, we examined vanadium toxicity in mice fed normal diet or high-fat diet (C57BL/6N, male, 7 weeks) by oral administration of ammonium metavanadate (AMV) with a maximum dose of 20 mgV/kg/day. Marked lipid accumulation in hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines and severe degeneration, necrosis, and loss of mucosal epithelial cells in the small intestine were observed. These pathological changes were more severe in mice fed high-fat diet than mice fed normal diet, and the intensity of the changes increased with increase in the administered dose of AMV. By electron microscopy, the number and size of lipid droplets in hepatocytes were increased. In the small intestine, a TUNEL assay showed a decreased number of positive cells, and positive cells for acrolein immunohistochemistry were observed specifically in the mucosal epithelial cells indicating degeneration and necrosis in the AMV-treated group, suggesting that a possible factor responsible for cell necrosis in the small intestine could be oxidative stress. In conclusion, AMV may impair cellular lipid metabolism, resulting in lipid accumulation, and induce mucosal epithelial cell necrosis in the small intestine.
Introduction
Vanadium is a transition element that exists widely on earth and is contained in several foods (Habib and Ibrahim 2011), water (Kato et al. 2004; Sullivan and Leavey 2011) particulates in air, fuel oils, and coal (Nriagu 1998). Vanadium compounds have been proposed as new antidiabetic drugs because of their insulin mimetic/enhancing effects both in vitro and in vivo (Marzban and McNeill 2003). The potential importance of the vanadium compounds as orally active insulin-mimetic agents has been described (Heyliger, Tahiliani, and McNeill 1985). Several subsequent reports showed that vanadium treatment decreases the plasma glucose level in different animal models of both type 1 diabetes (Blondel, Bailbe, and Portha 1989; Ramanadham et al. 1989; Sekar, Li, and Shechter 1996) and type 2 diabetes (Ding et al. 2001). In type 2 diabetes and experimental models of insulin resistance, vanadium lowers plasma insulin levels and improves insulin sensitivity (Bhanot and McNeill 1994; Brichard, Pottier, and Henquin 1989; Cohen et al. 1995). Hence, those compounds are potential candidates for oral therapy in both type 1 and type 2 diabetes.
Despite numerous studies, the mechanisms by which vanadium mediates its metabolic effects in vivo are still not completely understood (Marzban and McNeill 2003). Moreover, a number of toxicities and death have been reported following oral and intraperitoneal vanadium administration to animals (McNeill 2000). Vanadium compounds caused gastrointestinal problems, diarrhea, dehydration (Heyliger, Tahiliani, and McNeill 1985; McNeill 2000), hepatic disorder (Al-Bayati et al. 1989), necrosis of hepatocytes with fatty cell infiltration and vacuolation (Uche, Obianime, and Gogo-Abite 2008), and necrosis of renal tubules (Wei et al. 1982). Furthermore, effects of vanadium compounds on various enzyme activities have also been reported; for example, vanadium compounds inhibit Na+-K+ ATPase (Cantley et al. 1977; Dubyak and Kleinzeller 1980), ribonuclease, adenylate kinase, and phosphofructokinase (Macara, Kustin, and Cantley 1980). Precise information concerning the pharmacokinetics and toxicity of vanadium is required for practical use of vanadium compounds as antidiabetic drugs.
A previous study has shown the difference in the toxic effects of vanadium between two groups of mice treated with ammonium metavanadate (AMV; NH4VO3; 1, 5, 10, 20 mg/V/kg); mice fed high-fat diet showed severe clinical and pathological changes with a decreased survival rate and normal diet mice group showed no signs of toxicities after treatment with AMV at 1 to 5mgV/kg for 10 days. Mortality was markedly increased in the high-fat diet group compared with the normal diet group when animals were treated with AMV at 20 mg/V/kg for 1 to 5 days, suggesting that the vanadium compounds may affect lipid metabolism (Hasegawa T., personal communication). The purpose of this study was to study the vanadium toxicity in mice with emphasis on the effect of vanadium treatment on lipid metabolism.
Materials and Methods
Animals
Six-week-old male C57BL/6N mice, weighing 20 to 23 g and obtained from Japan SLC, Inc. (Shizuoka, Japan), were kept in cages and allowed to be acclimatize to the new environment for a week. The animals were kept at around 25°C and divided into two groups. One group was fed pelleted normal CE-2 diet (CLEA JAPAN, Inc.) and the other group was fed pelleted High-Fat Diet 32 (CLEA JAPAN, Inc.). Each diet and water were available ad libitum throughout the experiment. The experiments were performed in accordance with the Ethics Committee for Animal Experiments, Yamanashi Institute of Environmental Sciences (ECAE-01-2011).
Vanadium Administration
AMV (Kanto Chemical Co., Inc., Japan) was used in this study. AMV was dissolved in distilled water and administered to the CE-2 group at 0, 10, and 20 mgV/kg/day for 1 to 4 days and to the high-fat diet 32 group at 0, 2, 5, and 10 mgV/kg/day for 1 to 5 days by stomach tube; the dose levels were used according to the previous report on the effect of vanadate on the animal model of diabetes mellitus (Ding et al. 2001). Five mice were used per administered dose.
Hematological Examination
Blood was collected from the surviving animals (n = 3–5) 24 hr after the last administration from the abdominal portion of vena cava under ether anesthesia and analyzed immediately by FUJI DRI-CHEM 3500 (Fujifilm, Tokyo, Japan). Statistical analysis was performed by analysis of variance and multiple comparisons.
Histopathology
Necropsy was conducted 24 hr after the last administration. Tissues from the liver, spleen, kidney, heart, lung, brain, stomach, pancreas, small (at the level of duodenum and ileum) and large (at the level of colon) intestines, bone marrow, triceps brachii muscle, and quadriceps femoris muscle were collected and fixed in 10% neutral-buffered formalin. These tissues were routinely processed and embedded in paraffin for histopathological examination. Sections of approximately 3 µm in thickness were cut and stained with hematoxylin and eosin.
Sudan Black B stain
Sudan Black B staining was performed on 10% neutral-buffered formalin fixed tissues from the liver, kidney, and small and large intestines of the CE-2 control, 10-mgV, and 20-mgV groups (for 3 days, each group) and of the high-fat diet control, 2-mgV, 5-mgV, and 10-mgV groups (for 3 days, each group). These tissues were embedded in embedding medium for frozen specimens (Sakura Finetek Co., Ltd., Tokyo, Japan) and then frozen in liquid nitrogen. Sections of approximately 8 µm in thickness were cut with a Leica CM1850 UV (Leica Micosystems K.K., Tokyo, Japan) and stained with Sudan Black B (Reagenzien Merck, Germany).
Electron Microscopy
Electron microscopy was performed on glutaraldehyde fixed liver tissues from the CE-2 control, 10-mgV, and 20-mgV groups (for 3 days, each group). The liver tissues were cut into 1 to 2 mm3 cubes. The small blocks were rinsed in 0.1 M phosphate-buffered saline (PBS, pH = 7.4), postfixed for 1 hr in 1% osmium tetroxide, dehydrated in alcohol, and embedded in epoxy resin. Semithin (1-µm thick) sections were stained with 1% toluidine blue to select and locate interesting areas for electron microscopic examination. Ultrathin sections stained with uranyl acetate and lead citrate were examined under a TEM-100CX electron microscope (JEOL Ltd., Tokyo, Japan).
Immunohistochemistry: Immunoperoxidase Method
Immunohistochemical analysis was performed on the tissues from CE-2 fed groups of mice with (1) a polymer method for antiactive caspase-3 antibody (rabbit polyclonal, diluted at 1:350, Promega, Madison) as a marker of apoptosis and (2) a Histofine MOUSESTAIN Kit (Nichirei Biosciences Inc., Tokyo, Japan) for antihexanoyl-lysine antibody (mouse monoclonal, diluted at 1:75, NIKKEN Seil, Tokyo, Japan) and antiacrolein monoclonal antibody (mouse monoclonal, diluted at 1:100, NIKKEN Seil, Tokyo, Japan) as markers of lipid peroxidation resulting from oxidative stress. After deparaffinization, to improve the binding of antibodies, the sections were transferred into citric acid buffer and boiled for 20 min at 98°C in a microwave oven, according to a standard microwave treatment protocol. After this treatment, the procedures described below were performed for each primary antibody.
Polymer Method
After blocking endogenous peroxidase activity with 3% hydrogen peroxide, sections were preincubeted with normal goat serum for 5 min in a microwave oven. Then sections were reacted with the primary antibody for 10 min in a microwave oven. After microwave treatment, sections were treated with polymer reagents (Dako, Glostrup, Denmark) for 5 min in a microwave oven.
Histofine MOUSESTAIN Kit
After blocking endogenous peroxidase activity with 3% hydrogen peroxide, sections were treated with blocking reagent A for 60 min at normal temperature. Then the sections were washed with PBS and reacted with the primary antibody for 10 min in a microwave oven, followed by washing with PBS and treatment with blocking reagent B for 10 min at normal temperature. The sections were then washed with PBS again and treated by applying drops of Simple Stain Mouse MAX-PO (M) to sections for 10 min at normal temperature.
After the procedures in (1) or (2), sections were washed with PBS. The reaction was visualized using 3,3′-diaminobenzidine tetrahydrochloride as a chromogen. Sections were counterstained lightly with hematoxylin.
TUNEL Assay
The TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) assay was performed on 10% neutral-buffered formalin fixed tissues from the liver, kidney, and small intestine of the CE-2 control, 10-mgV, and 20-mgV groups (for 3 days, each group) with a TACS 2 TdT-DAB In Situ Apoptosis Detection kit (Trevigen, Inc., Gaithersburg, MD). After deparaffinization, the sections were treated with proteinase K (Dako) for 5 min and soaked in quenching solution for 5 min. Then the sections were washed with PBS and soaked in TdT labeling buffer for 5 min. The sections were reacted with labeling reaction mix containing Mn2+ for 60 min, 37°C. The reaction was stopped with TdT stop buffer for 5 min. After washing with distilled water, Strep-HRP solution was dropped over the sections for 10 min at 37°C. Then the sections were washed with PBS. The reaction was visualized using 3,3′-diaminobenzidine tetrahydrochloride as a chromogen. The sections were counterstained lightly with methyl green.
Results
Survival Rate and Body Weight Changes
CE-2 group
Body weight decreased day by day, and 2 mice died on the day of the fourth administration in the 20-mgV group. No difference was observed in the control and 10-mgV groups (Figure 1).
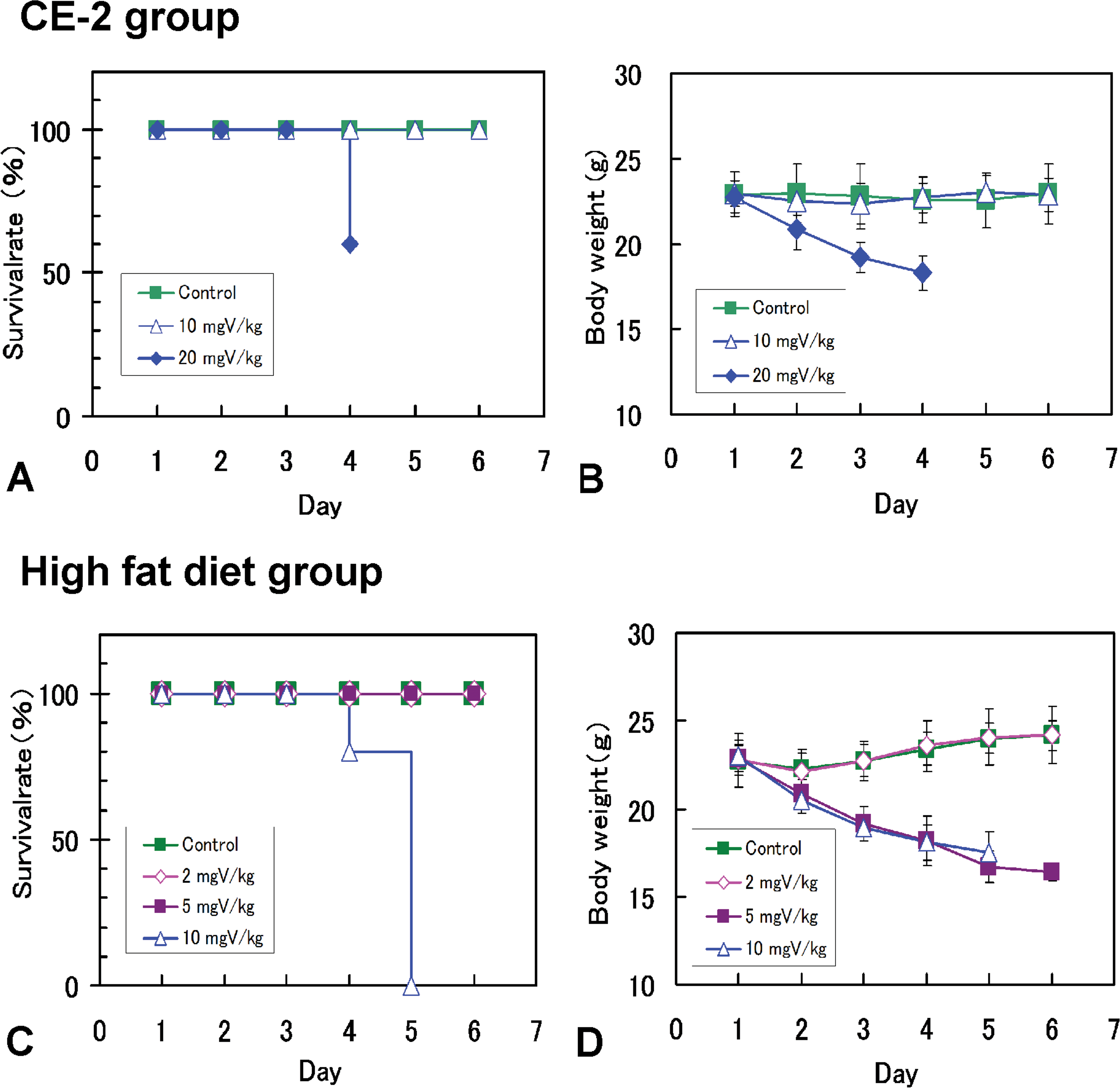
Changes of survival rate and body weight in the CE-2 and high-fat diet groups.
High-fat diet group
Body weight decreased day by day in the 5-mgV and 10-mgV groups. In the 10-mgV group, mice began to die on the day of the third administration, and all remaining mice died on the day of the fourth administration. No difference was observed between the control, 2-mgV, and 5-mgV groups in survival rate (Figure 1).
Clinical Sings
CE-2 group
Diarrhea began to be observed on the day of the second administration, and all mice showed diarrhea after the third and fourth administration in the 20-mgV group. No signs were observed in the 10-mgV group.
High-fat diet group
Diarrhea was observed on the day of the third administration and onward in the 5-mgV group, and on the day of the second administration and onward in the 10-mgV group. No clinical signs were observed in the control and 2-mgV groups.
Hematological Examination
CE-2 group
After the third administration in the 20-mgV group, aspartate aminotransferase (AST), alanine aminotransferase (ALT), blood urea nitrogen (BUN), creatine phosphokinase (CPK), and creatine phosphokinase-MB (CKMB) were increased compared with the control group (Table 1).
Results of hematological examination.
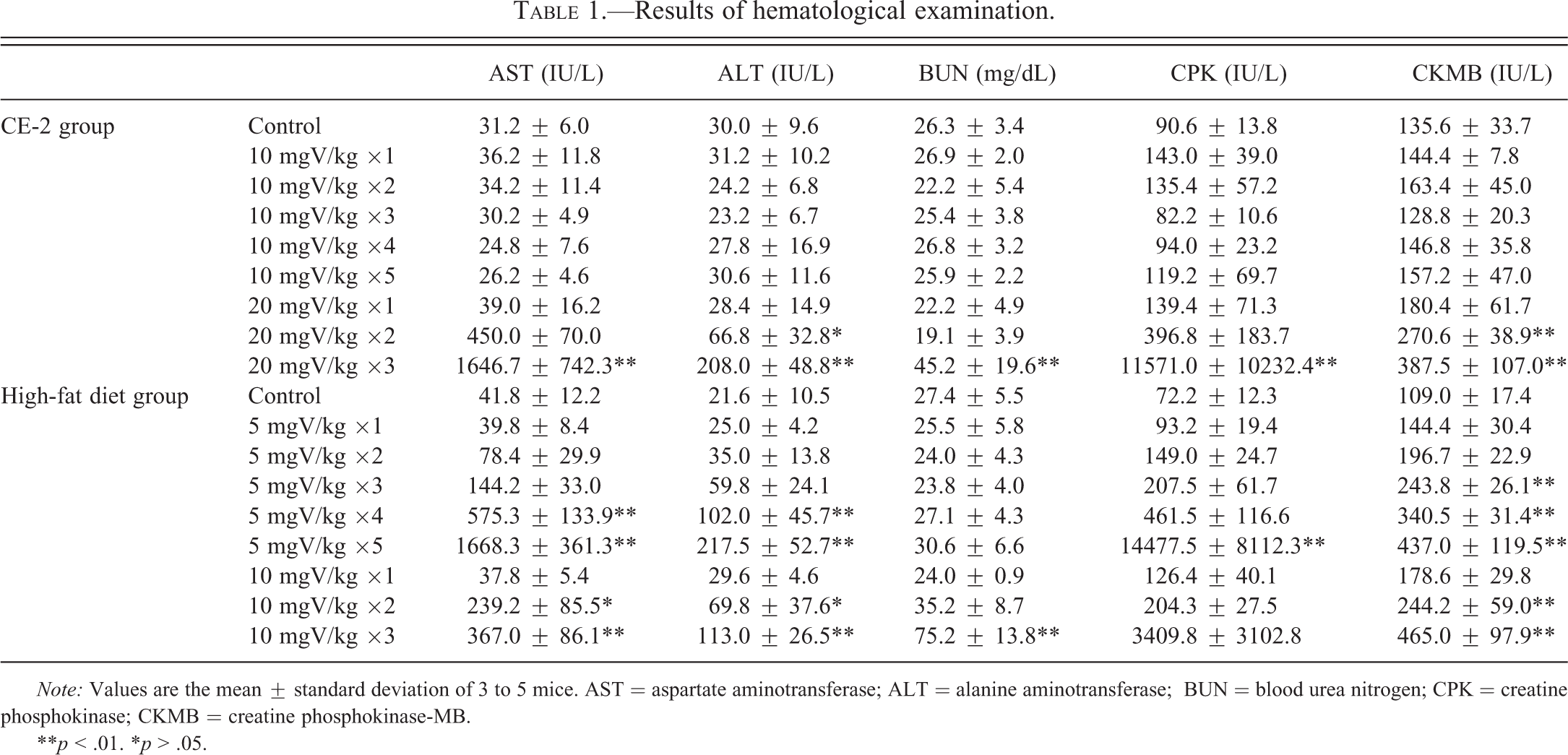
Note: Values are the mean ± standard deviation of 3 to 5 mice. AST = aspartate aminotransferase; ALT = alanine aminotransferase; BUN = blood urea nitrogen; CPK = creatine phosphokinase; CKMB = creatine phosphokinase-MB.
**p < .01. *p > .05.
High-fat diet group
A marked increase in AST and ALT was observed after the third administration and onward in the 5-mgV group, and after the second administration and onward in the 10-mgV group (Table 1). A marked increase in CPK and CKMB was also observed after the third administration in the 5-mgV group and after the first administration in the 10-mgV group. BUN was significantly increased after the third administration in the 10-mgV group compared with the control group.
Gross Morphology
CE-2 group
Atrophy of the spleen was observed after the third administration in the 10-mgV and 20-mgV groups. A pale color in the liver and enlargement by distension of the gall bladder and small and large intestines were observed after the second administration in the 20-mgV group (Figure 2B).
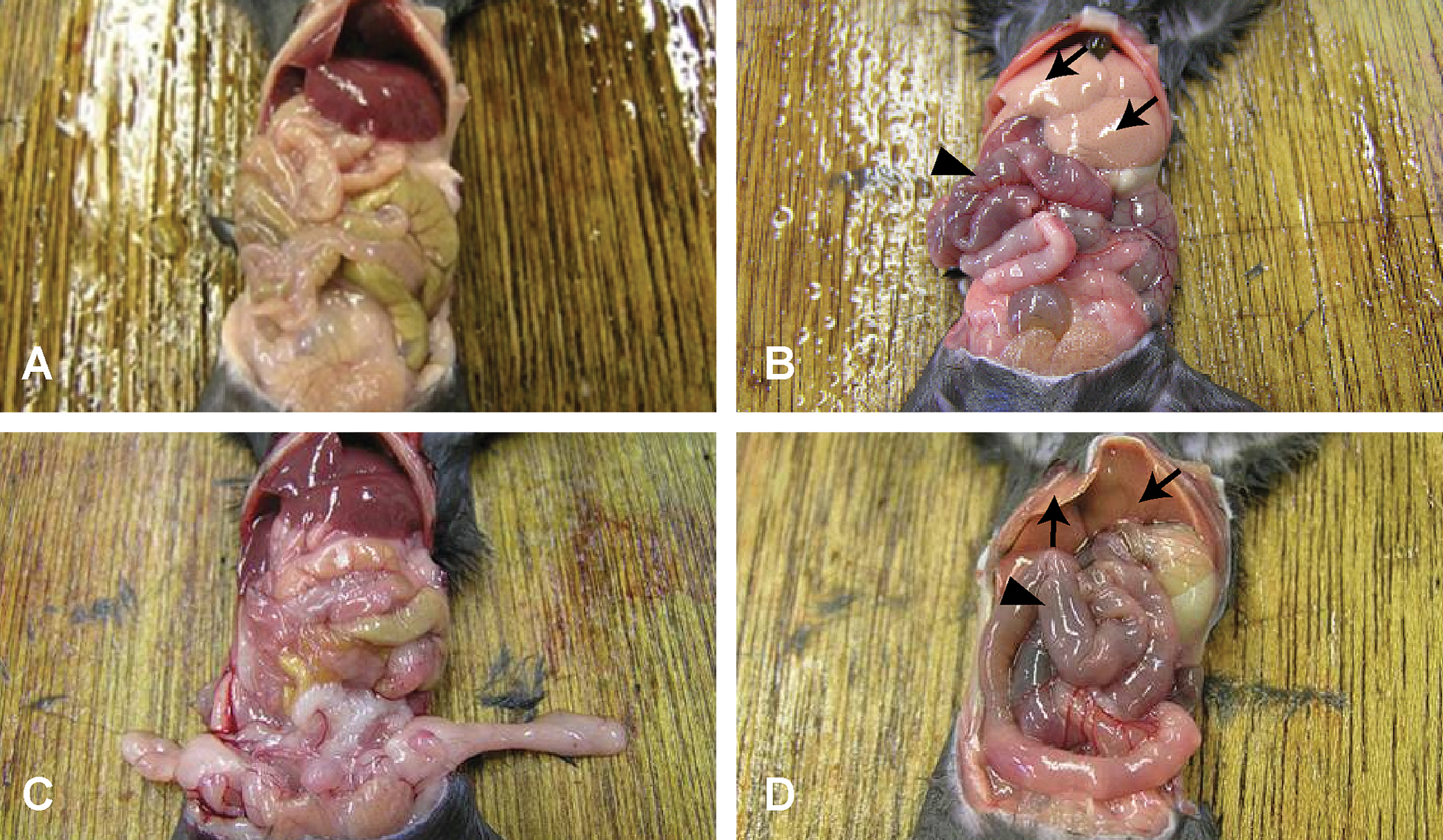
Gross morphology in the CE-2 group (A: control, B: 20 mgV/kg/day, for 3 days) and the high-fat diet group (C: control, D: 10 mgV/kg/day, for 3 days). Pale change of the liver (B, D, arrows), swelling and dark reddish change of the intestinal tract (B, D, arrowheads) are observed in both groups of mice treated with ammonium metavanadate.
High-fat diet group
Atrophy of the spleen, a pale change in the liver, and enlargement by distension of the gall bladder and small and large intestines were observed after the third administration in the 5-mgV group and after the second administration in the 10-mgV group (Figure 2D). No significant gross changes were observed in the 2-mgV group.
Histopathology
CE-2 group
In the 10-mgV group (for 3 days), finely vacuolated cytoplasm of hepatocytes and renal epithelial cells was observed. In the small and large intestines, respectively, focal shortening of intestinal villi and mild dilatation of crypts was observed. Mild atrophy of lymph follicles was observed in spleen. In the 20-mgV group (for 3 days), finely vacuolated cytoplasm of hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines was observed (Figure 3). In the small and large intestines, respectively, moderate to marked dilatation of crypts and degeneration and necrosis of mucosal epithelial cells was observed (Figure 4). Marked atrophy of lymph follicles and loss of lymphocytes were observed in the spleen. Moderate to marked congestion was observed in the liver, kidney, and heart. No significant changes were observed in the other tissues examined.
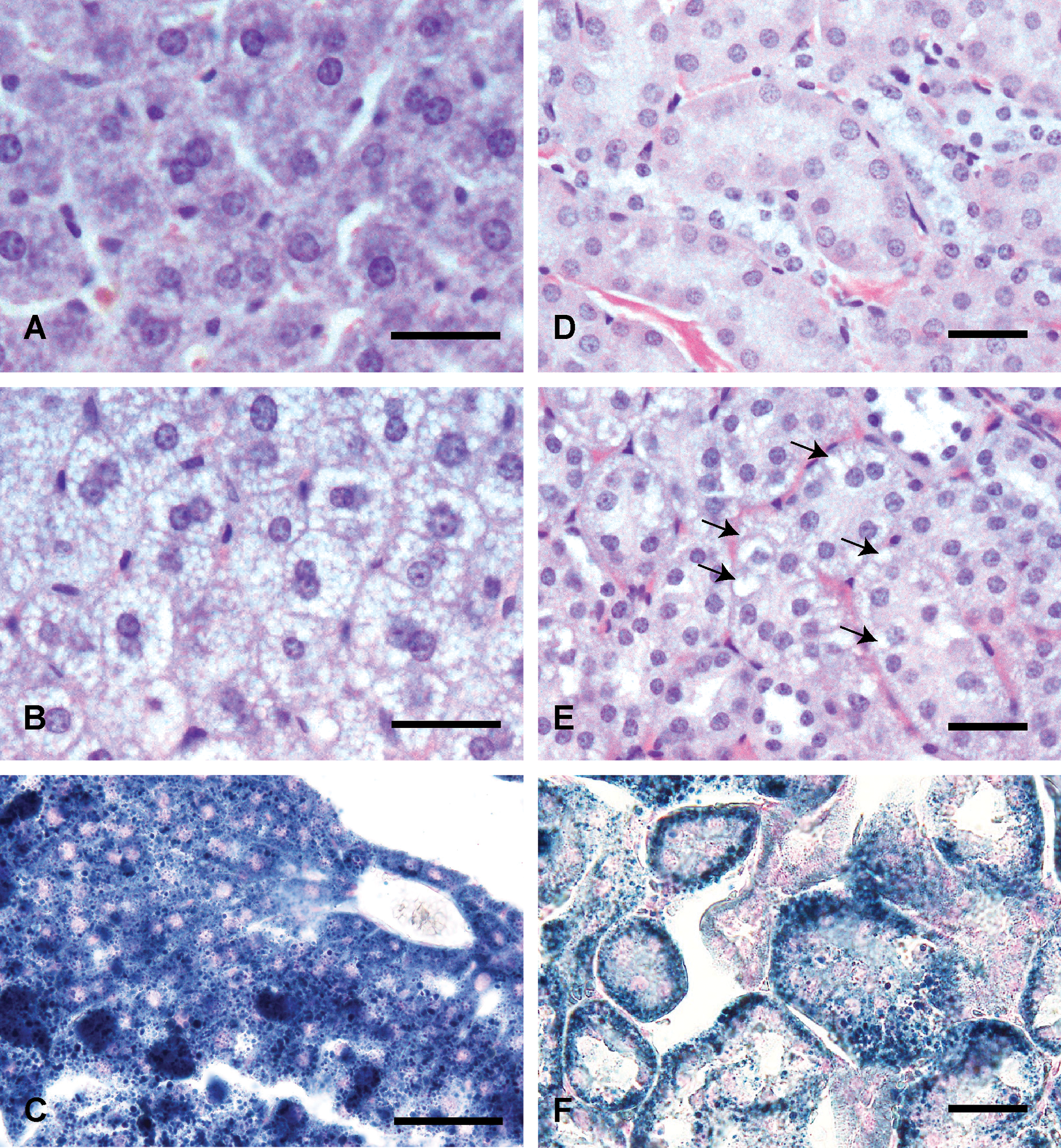
Continued
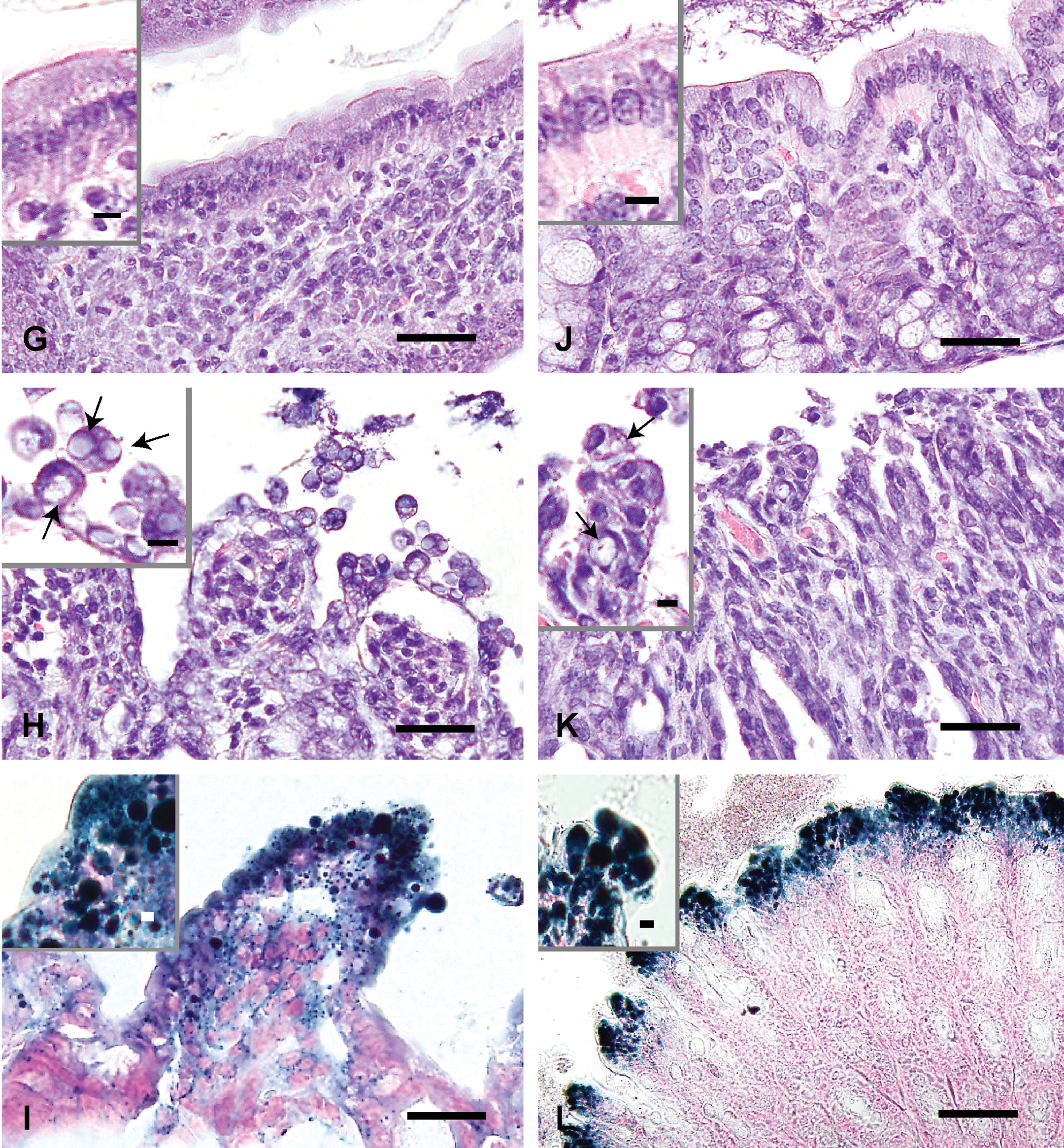
Finely vacuolated cytoplasm of hepatocytes (B), renal epithelial cells (E, arrows), mucosal epithelial cells of the small (H, arrows) and large (K, arrows) intestines is observed in the CE-2 20-mgV group (for 3 days). Intensity and the number of Sudan Black B-positive lipid droplets (C, F, I, L) are similar to those of vacuoles observed in each tissues stained with HE stain (B, E, H, K). HE stain (A, B, D, E, G, H, J, K). Sudan Black B stain (C, F, I, L). Liver (A), kidney (D), small (G: ileum) and large (J: colon) intestines from the CE-2 control group. Liver (B, C), kidney (E, F), small (H, I: ileum) and large (K, L: colon) intestines from the CE-2 20-mgV group (for 3 days). Bars = 30 µm. Bars in insets = 5 µm.
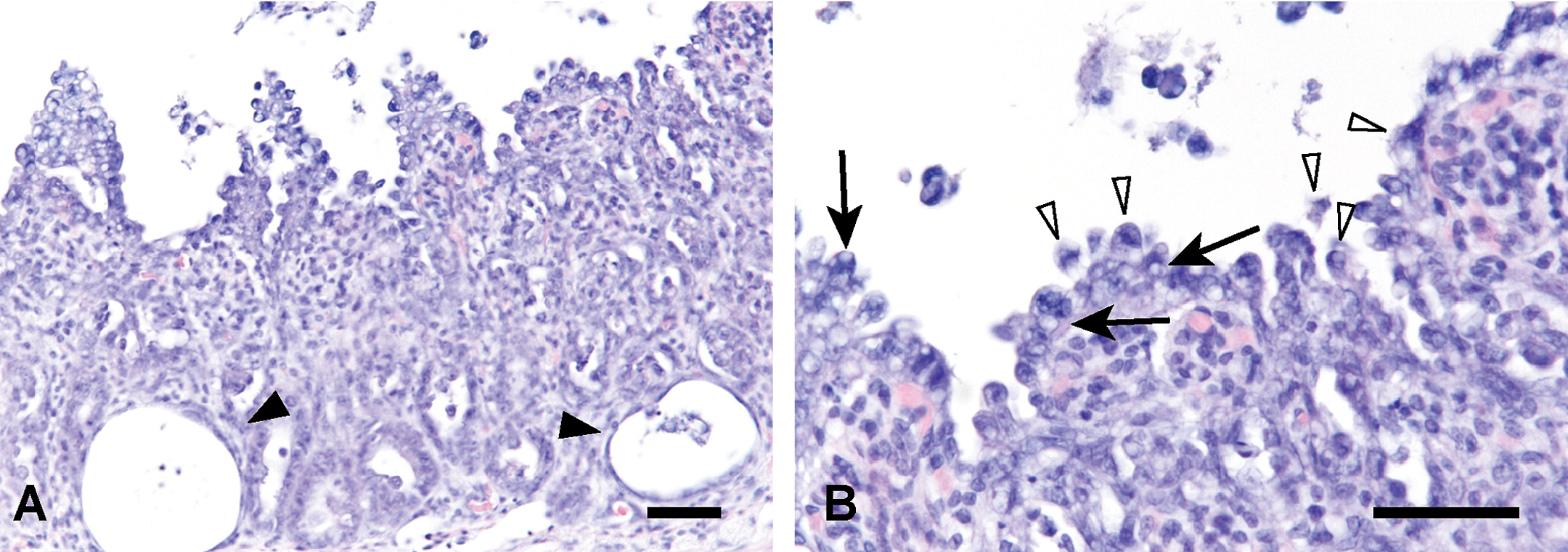
Small intestine (ileum) from the CE-2 20-mgV group (for 3 days) shows severe degeneration, loss and necrosis of mucosal epithelial cells, and dilatation of crypts (A, arrowheads). Fine vacuolization (B, arrows) and necrosis (B, arrowheads) in mucosal epithelial cells are observed. B is higher magnification of A. HE stain. Bars = 30 µm.
High-fat diet group
Histological changes were similar between the CE-2 group at 20 mgV and the high-fat group at 10-mgV group (for 3 days) in the liver, kidney, and small and large intestines (Figure 5). In the kidney, mild to moderate fine vacuolization of the cytoplasm in addition to atrophy of renal epithelial cells were observed (Figure 5). Additionally, a marked decrease in the number of zymogen granules in the pancreas and degeneration and coagulative necrosis in cardiac muscle (1 of the 5 animals examined) were observed. Mild to marked congestion was observed in the liver, kidney, and heart. In the 5-mgV group (for 3 days), finely vacuolated cytoplasm of hepatocytes (Figure 5) was observed as observed in the 10-mgV group (for 3 days). Pathological changes in the kidney and small and large intestines in the 2-mgV and 5-mgV groups (for 3 days, each group) were less severe than those in the 10-mgV group (for 3 days).
Continued
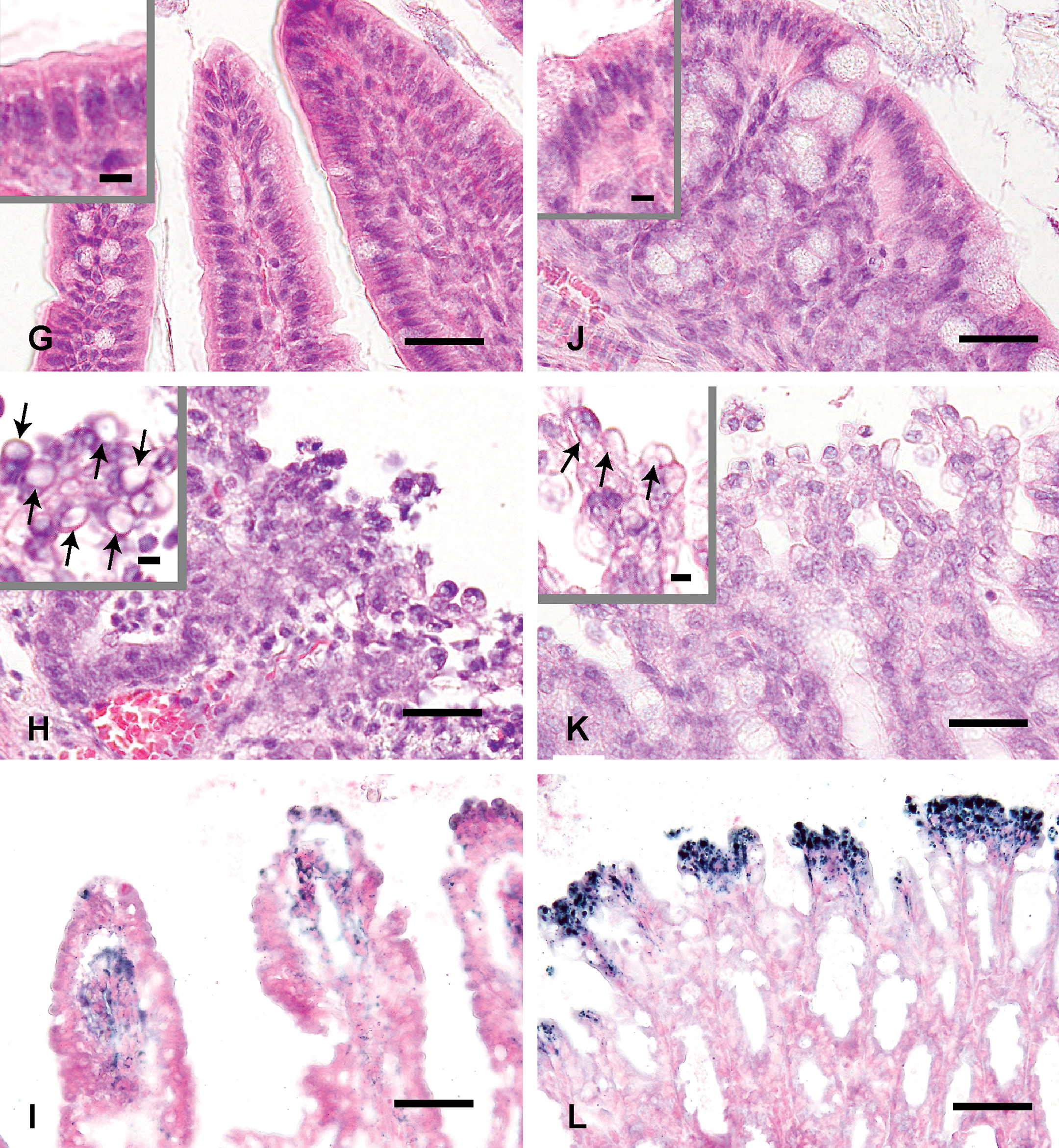
Finely vacuolated cytoplasm of hepatocytes (B), renal epithelial cells (E, arrows), mucosal epithelial cells of the small (H, arrows) and large (K, arrows) intestines is observed in the high-fat diet 10-mgV group (for 3 days). Intensity and the number of Sudan Black B-positive lipid droplets (C, F, I, L) are similar to those of vacuoles observed in each tissues stained with HE stain (B, E, H, K). HE stain (A, B, D, E, G, H, J, K). Sudan Black B stain (C, F, I, L). Liver (A), kidney (D), small (G: ileum) and large (J: colon) intestines from the high-fat diet control group. Liver (B, C), kidney (E, F), small (G, H: ileum) and large (J, K: colon) intestines from the high-fat diet 10-mgV group (for 3 days). Bars = 30 µm. Bars in insets = 5 µm.
Sudan Black B stain
CE-2 group
In the 20-mgV group (for 3 days), a large number of Sudan Black B-positive droplets were observed in hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines (Figure 3). In the small intestine, Sudan Black B-positive droplets were also observed in the degenerative mucosal epithelial cells (Figure 3). In the 10-mgV group (for 3 days), Sudan Black B-positive droplets were also observed in hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines; the numbers of positive droplets were smaller than those in the 20-mgV group (for 3 days).
High-fat diet group
In the 10-mgV group (for 3 days), a large number of Sudan Black B-positive droplets were observed in hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines (Figure 5). In the small intestine, Sudan Black B-positive droplets were also observed in the degenerative mucosal epithelial cells (Figure 5). The vacuolar change, Sudan Black B-positive findings and degenerative changes in the 10-mgV group (for 3 days) were more severe than those observed in the CE-2 10-mgV group (for 3 days; Figure 6).
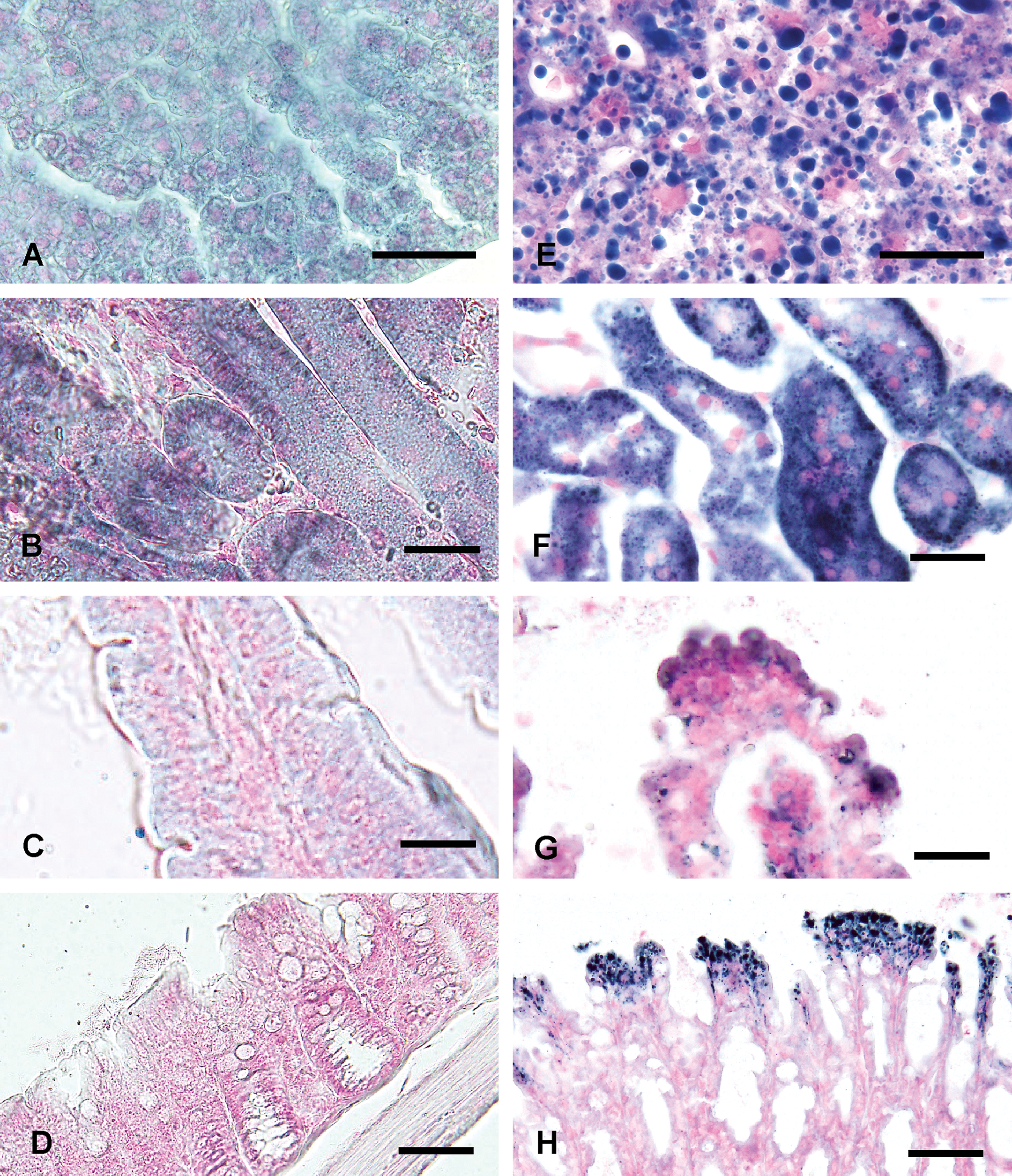
Comparison of lipid accumulation between the CE-2 group (A, B, C, D) and the high-fat diet group (E, F, G, H) treated with ammonium metavanadate at the same dose and number of administrations (10 mgV/kg/day, for 3 days). Lipid accumulation and degeneration of each tissue in the high-fat diet group are more severe than those of the CE-2 group. Liver (A, E), kidney (B, F), small (C, G: ileum) and large (D, H: colon) intestines stained with Sudan Black B stain. Bars = 30 µm.
Electron Microscopy
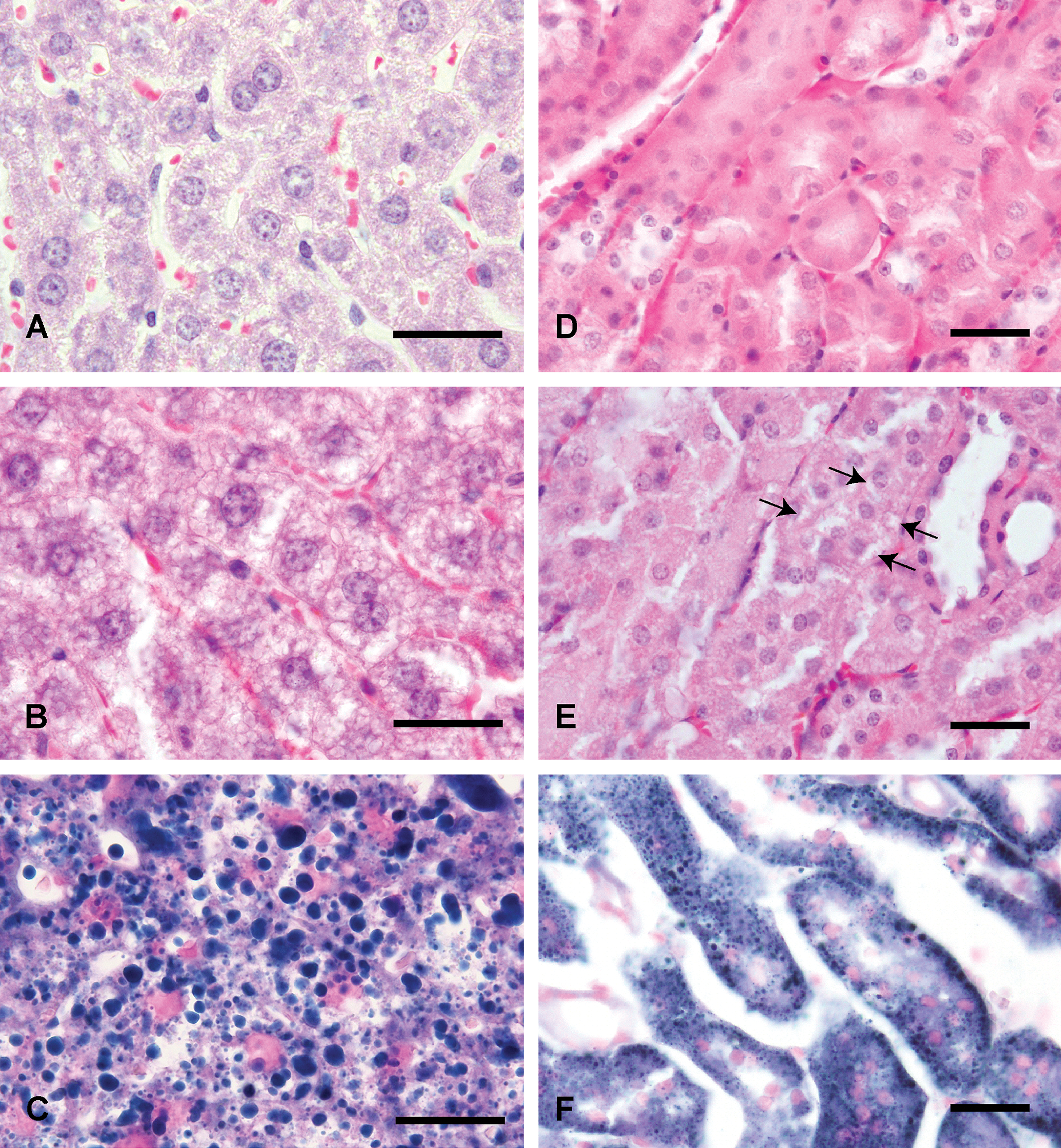
In the CE-2 control group, electron dense homogenous round structures (lipid droplets [LDs]), 1.9 to 2.3 µm in size, surrounded by a single membrane were occasionally observed in the cytoplasm of hepatocytes (Figure 7). Fine black granules (LD-associated proteins) were attached to the surface of the membrane of the round structure (Figure 7). In the CE-2 20-mgV group (for 3 days), low electron-dense spherical structures, 1.6 to 6.6 µm in size, surrounded by a single rough membrane were observed diffusely in the cytoplasm (Figure 8). Tiny vacuolar structures, 0.1 to 0.3 µm in size, were observed on the rough membranes surrounding these structures (Figure 8). These spherical structures frequently exhibited assembly and fusion-like morphologies (Figure 8). A large number of mitochondria were noted compared with those observed in the control group (Figure 8).
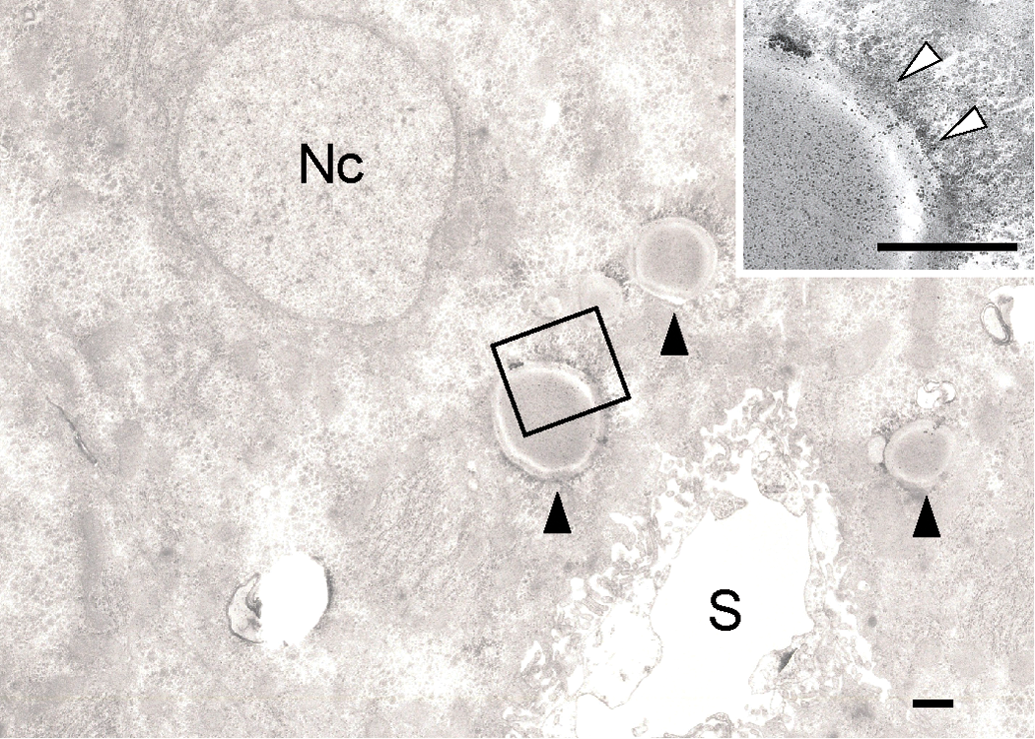
Transmission electron microscopy of hepatocytes of CE-2 control group showing electron dense homogeneous round structures (lipid droplets, black arrowheads) surrounded by a single membrane in the cytoplasm. Black fine granules (lipid droplet–associated proteins, white arrowheads) are attached on the surface of the membrane of the round structures. Inset is higher magnification of rectangle. Nc: nucleus, S: sinusoid. Bars = 1 µm.
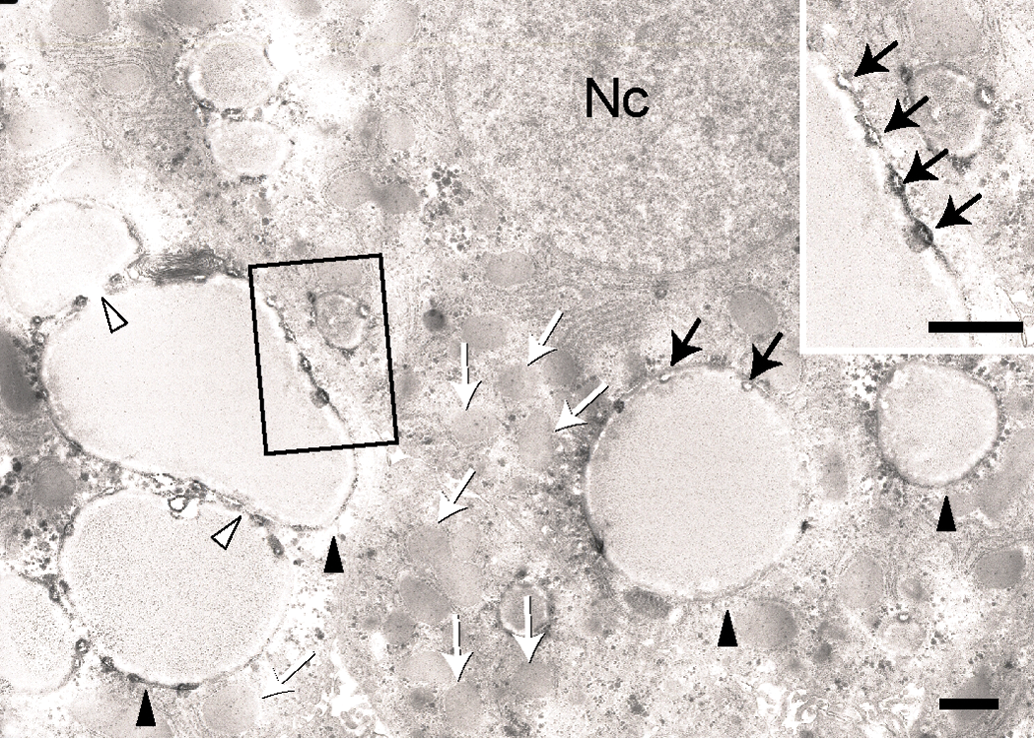
Transmission electron microscopy of hepatocytes of CE-2 20-mgV group (for 3 days) showing that the number and the size of the lipid droplets are larger (black arrowheads) and the density of lipid contents are lighter than those in control. Assembly and/or fusion-like morphology of the lipid droplets are frequently observed (white arrowheads). Tiny vacuolar structures (black arrows) are observed on the rough membranes surrounding these lipid droplets. Note densely packed mitochondria (white arrows) in the cytoplasm. Inset is high magnification of rectangle. Nc: nucleus, S: sinusoid. Bars = 1 µm.
Immunohistochemistry
Caspase-3
In the CE-2 control group, positive reactions were detected in the cytoplasm of mucosal epithelial cells of the small intestine; the positive reactions were detected at the tip of the villi. In the CE-2 10-mgV and 20-mgV groups (for 3 days, each group), the numbers of positive cells appeared to be same as those observed in the control group. In the liver and kidney, no positive reactions were detected.
Hexanoyl-lysine
In the CE-2 control and CE-2 10-mgV and 20-mgV groups (for 3 days, each group), no positive reactions were detected in the liver, kidney, and small and large intestines.
Acrolein
In the CE-2 20-mgV group (for 3 days), positive reactions were detected in the cytoplasm of mucosal epithelial cells of the small intestine; the positive reactions were located at the tip of the villi showing degeneration and loss in the small intestine (Figure 9). No positive reactions were detected in the liver, kidney, and large intestine.
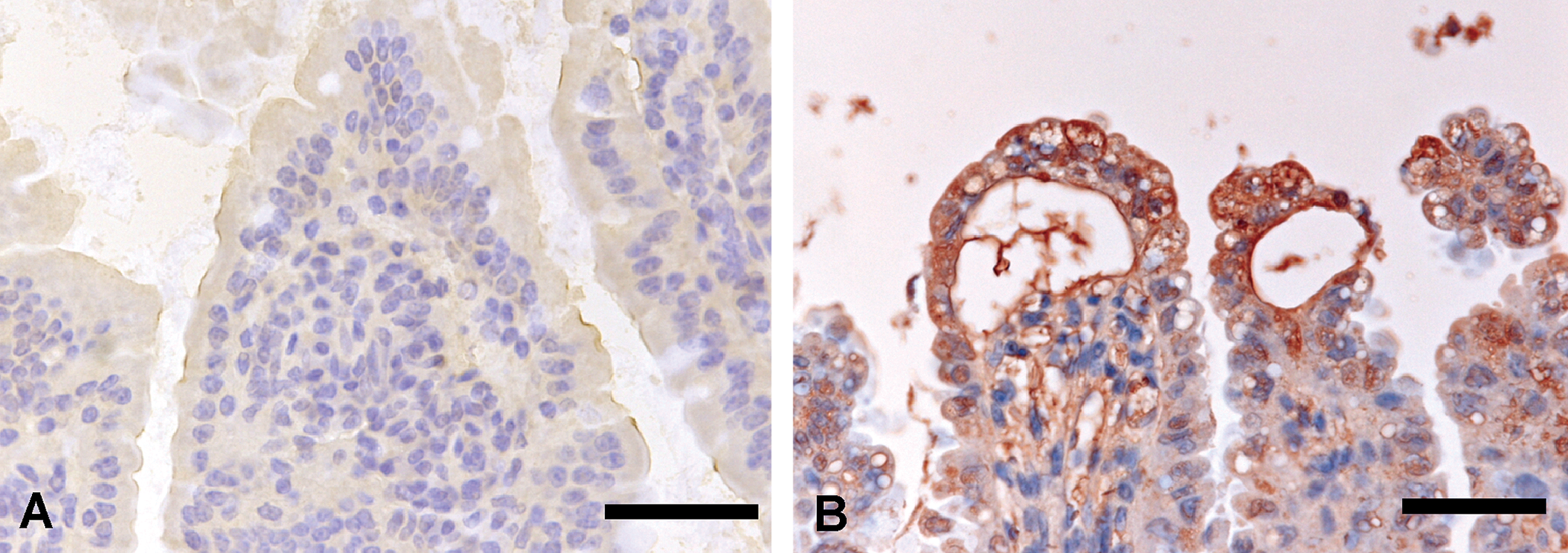
Acrolein immunohistochemistry of the small intestine (ileum) from the CE-2 control group (A) and from the 20-mgV group (for 3 days) (B). Positive reactions are observed in the cytoplasm of mucosal epithelial cells at the tip of the villi showing degeneration and loss (B). Bars = 30 µm.
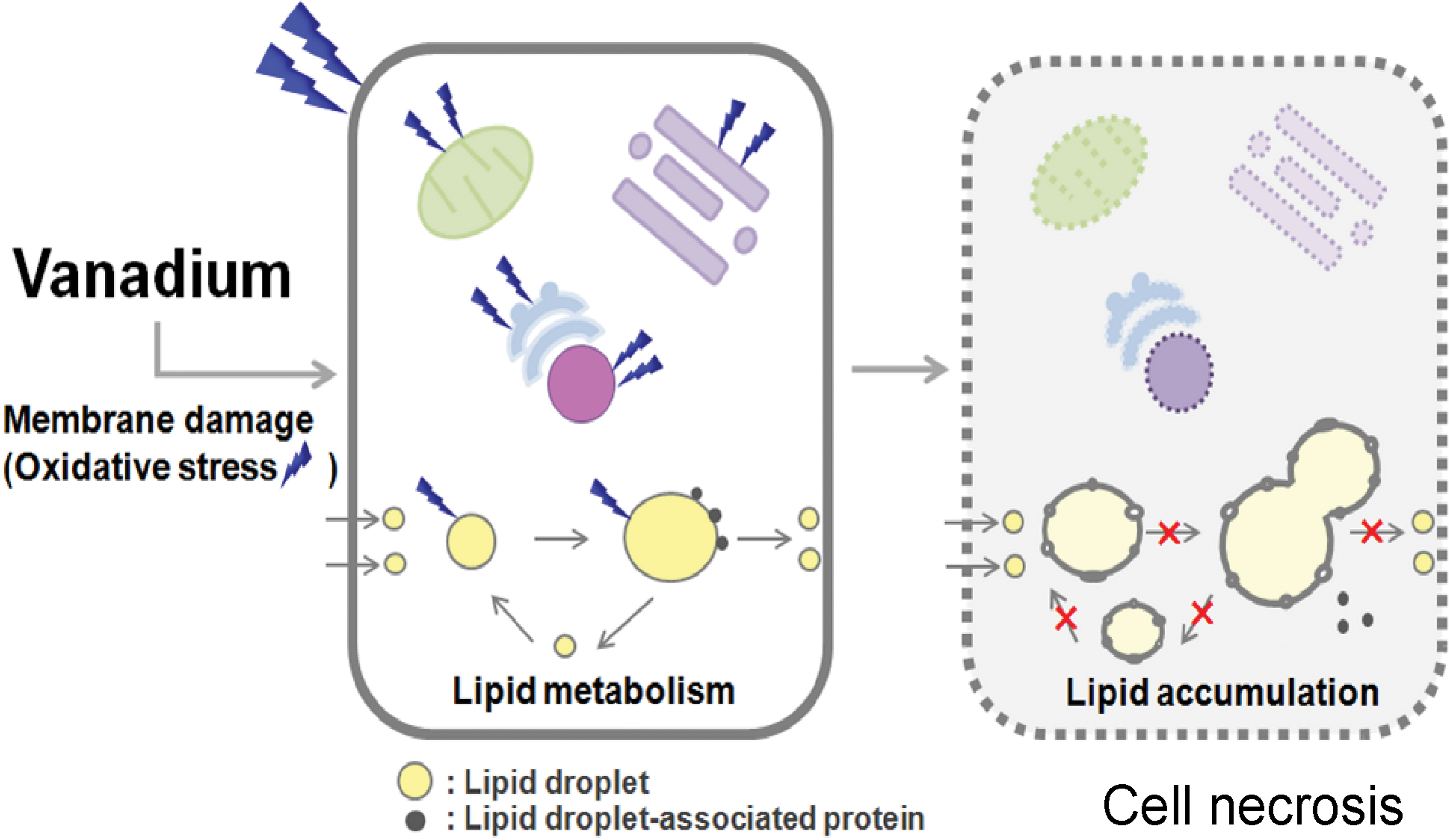
Schematic diagram on the hypothesis of the vanadium toxicity. Vanadium may induce damage of cellular and subcellular membranes directly and/or indirectly through oxidative stress: one of the possible factors responsible for the cellular lipid metabolism disorder and cell necrosis could be membrane damage induced by vanadium.
TUNEL Assay
In the small intestine of the CE-2 control group, positive reactions were detected in the nuclei of mucosal epithelial cells at the tip of the villi. The number of positive reactions in nuclei of mucosal epithelial cells at the tip of the villi was smaller in the CE-2 10-mgV and 20-mgV groups (for 3 days, each group) compared with the number observed in the control group.
Discussion
In the CE-2 group, finely vacuolated cytoplasm was observed in the hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines; the severity of vacuolization by light microscopic examination increased with the dose and number of administrations of vanadium. Vacuoles were positive for Sudan Black B staining, consistent with lipid. In the high-fat diet group, the same degenerative changes were observed after treatment with the smaller given dose. Hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines, in which vacuolar changes were observed, have functions of absorption, metabolization, and excretion of lipid substances. Therefore, AMV may have impaired the process of these cellular functions, resulting in lipid accumulation in tissues.
Lipids taken into normal hepatocytes are hypothesized to be metabolized in the following two courses after hatching or budding from the endoplasmic reticulum (ER; Fujimoto et al. 2008; Olofsson et al. 2009): formation of (1) cytosolic LDs and (2) very-low-density lipoproteins (VLDLs). LDs are depots of neutral lipid that exist virtually in any kind of cells. In hepatocytes and small intestinal epithelial cells, VLDLs are secreted into the blood stream via the secretory pathway of the cell (Olofsson et al. 2009). The newly formed (primordial) LDs have a diameter of 0.1 to 0.4 µm (Marchesan et al. 2003), and the mature LDs present in a cell can be more than 10 to 50 times larger (Boström et al. 2005). VLDLs have a diameter of 50 to 80 nm (Pavelka and Roth 2005). Following analysis of the light microscopic results showing marked lipid accumulation in hepatocytes, an electron microscope study was performed with the focus on the liver, which is the main organ of lipid metabolism. In the CE-2 control group, electron-dense homogeneous round structures surrounded by a single membrane were occasionally observed in the cytoplasm; black fine granules, presumably LD-associated proteins, were shown to be attaching to the surface of the membrane. Based on the size and morphology of these features, these round structures observed in the hepatocytes of the CE-2 control group mice in this study may be considered to be LDs rather than VLDLs.
In the CE-2 20-mgV group (for 3 days), the number and size of LDs were increased compared with those of the control group. Additionally, in this administration group, the density of LD contents appeared to decrease and assembly and/or fusion-like findings of LDs were frequently observed. These changes may have occurred in association with altered cellular lipid metabolism such as inhibition of lipolysis (Degani et al. 1981; Shisheva and Shechter 1992) or stimulation of lipogenesis (Fantus et al. 1989; Shisheva and Shechter 1992), both of which are mediated by insulin-mimetic effects of the administered vanadium compounds. LD storage occurs to a variable extent in most cells and today is regarded as a key process in cellular turnover of lipids (Olofsson et al. 2009). AMV may cause inhibition of lipid turnover not only in hepatocytes but also in the other tissues rich in functions of absorption, metabolization, and excretion of biological macromolecules.
Another possible mechanism for lipid accumulation in hepatocytes could be secondary binding inhibition of LD-associated proteins to the surface of the LD resulting from injury of the LD membrane induced by vanadium treatment and mediated by oxidative stress. LD is made of a highly hydrophobic lipid ester core and a phospholipid monolayer surface (Fujimoto et al. 2008). Several specific proteins known as LD-associated proteins present on the surface of the LD (Fujimoto et al. 2008; Olofsson et al. 2009). In hepatocytes from the CE-2 20-mgV group (for 3 days), membranes surrounding LDs were rough and showed formation of tiny vacuolar structures, whereas in hepatocytes from the CE-2 control group, membranes surrounding LDs were smooth at the electron microscopic level. These changes indicate that ammonium metavanatate induced damage to the LD membrane directly and/or indirectly, such as by oxidative stress (Figure 10). In addition, the damage of the LD membrane may cause inhibition of the binding of LD-associated proteins to the LD surface. Formation, maintenance, modification, and involution of LDs are regulated by a variety of proteins such as LD-associated proteins (Fujimoto et al. 2008). Thus, impairment of regulation for LDs mediated by LD-associated proteins resulting from damage of the LD membrane induced by AMV may cause lipid accumulation in hepatocytes.
In this study, marked degeneration, necrosis, and loss of mucosal epithelial cells of the small intestine were observed. These findings could be responsible for the severe diarrhea observed in the CE-2 20-mgV group and the high-fat diet 5-mgV and 10-mgV groups. To identify the mechanism of the small intestinal pathological changes resulting from vanadium administration, small intestines from the CE-2 group were examined with the emphasis on apoptosis and necrosis as the cause of cell death. In this study, no difference was observed in caspase-3 immunohistochemical findings in mucosal epithelial cells of the small intestine between the CE-2 control group and CE-2-treated group. In addition, positive cells were decreased in the CE-2-treated group in the TUNEL assay. These findings suggest that apoptotic event could not be directly involved in cell death of the intestinal mucosal epithelial cells in this study.
Another possible mechanism responsible for the cell necrosis of the small intestinal epithelial cells could be oxidative stress induced directly or indirectly by vanadium treatment. Overproduction of reactive oxygen species (ROS) through either endogenous or exogenous insults is harmful to living organisms and is termed oxidative stress. ROS are known to damage not only DNA but also additional cellular components such as cellular and subcellular membranes consisting of proteins and phospholipids, leading to cell necrosis (Mussali-Galante et al. 2007). In addition, vanadium may participate in the reactions involving formation of the free radicals, which are a type of the ROS (Cortizo et al. 2000; Farber, Kyle, and Coleman 1990). In this study, positive cells for acrolein immunohistochemistry, which is one of the markers for ROS-induced cellular lipid injury, were observed specifically on the mucosal epithelial cells showing degeneration and necrosis in the small intestine in the CE-2 20-mgV group. The control and treated groups at the other doses showed negative immunoreactivity. This finding indicates that cell necrosis of mucosal epithelial cells in the small intestine may have been associated with lipid peroxidation by ROS production by AMV (Figure 10). In the several lipids existing in the organisms, phospholipids, a component of the LD membrane, are the most reactive substance with free radicals (Toledo, Sniderman, and Kelley 2006). In the CE-2 20-mgV group, a large number of LDs were observed in mucosal epithelial cells of the small intestine. Therefore, ROS-induced lipid peroxidation, especially for phospholipids, resulting from vanadium treatment, may be involved in the development of cell degeneration in the intestinal mucosal membrane.
Additional pathological changes included marked atrophy of the spleen with loss of its lymph follicles in both the CE-2 and high-fat diet groups; the degree of the changes showed no correlation with the dose and number of administrations of AMV. In a mouse of high-fat diet group fed the maximum dose, degeneration and coagulative necrosis in cardiac muscle were observed. In the pancreas, a decrease in the number of zymogen granules in all examined mice was observed. Vanadium compound–induced pathological changes in splenic lymph follicles differ among reports, with one showing atrophy (Cui et al. 2011) and the other showing hyperplasia (Fortoul et al. 2008). An increase in lipid peroxidation rate in cardiac tissue after AMV administration has been reported (Soares et al. 2007). Vanadium may also affect organs that do not have a specific physiological function in absorption, metabolism, and excretion of lipids.
There was no difference in serum triglyceride and total cholesterol levels between the CE-2 control group and CE-2-treated group. However, an apparent increase in the serum triglyceride level and decrease in the serum total cholesterol level were observed with the increase in the given dose of AMV in high-fat diet group. There are some studies that have reported no alteration in the serum levels of triglycerides and cholesterol in normal animals treated with vanadium compounds (Pepato et al. 2008). On the other hand, it is known that the presence of hepatic steatosis is associated with elevated serum triglycerides (Toledo, Sniderman, and Kelley 2006); an excess amount of lipid accumulation in hepatocytes by impaired lipid metabolism downregulates the hepatocellular uptake of serum triglyceride. In high-fat diet group, accumulation of lipids in hepatocytes was severe in animals fed an increased dose of AMV. Elevated serum triglycerides and severe lipid accumulation, observed in high-fat diet group, may have been induced by AMV treatment. In the process of examining vanadium toxicity in this study, secondary malabsorption of diet components including fat caused by damage to intestinal mucosal epithelial cells should also have been considered as one of the underlying mechanisms of the alteration in biochemical serum parameters.
There is lack of data on several aspects of the health effects of vanadium compounds; the toxicity of vanadium depends on its physicochemical state, particularly on its valence state and solubility (Venkataraman and Sudha 2005). Vanadium pentoxide and sodium metavanadate have a toxicity rating of 5, equivalent to a probable lethal oral dose in humans of 5 to 50 mg/kg (Gosselin, Smith, and Hodge 1984). LD50 values for sodium metavanadate administered by gavage to rats and mice are 41 mg V/kg and 31 mg V/kg, respectively (Agency for Toxic Substances and Disease Registry [ATSDR] 1990). Present study using AMV demonstrated that in the high-fat diet 10-mgV/kg group, mice began to die on the day of the third administration, and all the remaining mice died on the day of the fourth administration, suggesting probable lethal oral dose in mice would be high-fat diet 5- to 10-mgV/kg. In this study, changes at the histological level were observed in all groups of animals administered AMV (CE-2 10 and 20 mg/kg, high-fat diet 2, 5, and 10 mg/kg) with or without clinical and gross morphological changes. Study with lower doses of AMV would be required to determine the dose for the pharmacologic use. Factors including fat level in food would be taken into account as an additional risk factor in the development of the vanadium toxicity.
In conclusion, this study demonstrated that marked lipid accumulation in hepatocytes, renal epithelial cells, and mucosal epithelial cells of the small and large intestines developed in accordance with the increase in the administered dose of AMV. Severe degeneration, necrosis, and loss of mucosal epithelial cells in the small intestine were also associated with these changes. These changes were more severe in the high-fat diet group than in the CE-2 group. Based on these findings, AMV may impair cellular lipid metabolism, resulting in lipid accumulation, and induce mucosal epithelial cell necrosis in the small intestine.
Footnotes
Acknowledgments
This work was supported in part by Special Project (Assessment and Control of Dust Emission in Degraded Drylands of East Asia), MEXT, Japan. The author thanks Ms. E. Kawahara at Tottori University for her excellent assistance in electron microscopy.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author were supported by Special Project (Assessment and Control of Dust Emission in Degraded Drylands of East Asia), MEXT, Japan.
