Abstract
Ethylene glycol monomethyl ether (EGME) or atrazine induces luteal cell hypertrophy in rats. Our previous study suggested that EGME stimulates both new and old corpora lutea (CL), while atrazine stimulates new CL. Bromocriptine (BRC) is known to suppress the luteolysis in rats. This study investigated the light- and electron-microscopic luteal changes induced by EGME, atrazine, or BRC. Female rats were treated with EGME (300 mg/kg/day), BRC (2 mg/kg/day), EGME and BRC (EGME + BRC), or atrazine (300 mg/kg/day) for 7 days. Luteal cell hypertrophy induced by EGME, EGME + BRC, and atrazine was subclassified into the following two types: CL hypertrophy, vacuolated type (CL-V) characterized by intracytoplasmic fine vacuoles, and CL hypertrophy, eosinophilic type (CL-E) characterized by eosinophilic and abundant cytoplasm. The proportions of CL-V and CL-E were different among the treatments. BRC-treated old CL showed lower proportion of endothelial cells and fibroblasts than normal old CL. Ultrastructural observation revealed that the luteal cells of CL-V contained abundant lipid droplets, whereas those of CL-E in EGME and EGME + BRC groups showed uniformly well-developed smooth endoplasmic reticulum. No clear ultrastructural difference was observed between the control CL and atrazine-treated CL-E. These results indicate that EGME, atrazine, and BRC have differential luteal morphological effects.
Introduction
There is growing interest in the possible health threat posed by substances in the environment, food, and consumer products that interfere with reproductive function (Yuan and Foley 2002). The female reproductive organs are regulated across the estrous cycle by the hypothalamic–pituitary–gonadal (HPG) system through complex feedback loops. These feedback mechanisms are perturbed by ovarian toxicants including ethylene glycol monomethyl ether (EGME), atrazine, and bromocriptine (BRC) (Kumazawa et al. 2009; Taketa et al. 2011a). In rodents, ovarian toxicants produce abnormal hormone secretion, disrupted estrous cycles, and identifiable histopathological changes in the reproductive tract; and detailed histopathological evaluations often lead to their mode of actions (Sanbuissho et al. 2009).
EGME and atrazine induce luteal hypertrophy following repeated administration (Davis et al. 1997; Taketa et al. 2010, 2011a). EGME, which is widely used in various industrial products such as detergents, adversely affects both the male and female reproductive systems (Johanson 2000; Welsch 2005). In the female reproductive system, EGME and its active metabolite, 2-methoxy acetic acid (MAA), induce the hypersecretion of progesterone (P4) from luteal cells both in vivo and in vitro (Almekinder et al. 1997; Davis et al. 1997; Taketa et al. 2011a). Atrazine, a chlorotriazine herbicide, is a potent endocrine disruptor that promotes mammary tumor growth in female rats (Eldridge et al. 1994) and alters the central nervous system regulation of the reproductive system in mammals (Cooper et al. 2000; Eldridge et al. 1999; Foradori et al. 2009). Recently, atrazine has been shown to stimulate steroidogenesis and increase P4 secretion in vitro and in vivo (Fraites et al. 2009; Tinfo et al. 2011).
BRC, a dopamine D2 agonist that inhibits prolactin (PRL) release in the anterior pituitary, is also known to affect luteal function in rats (Gaytan et al. 2001). In rats, BRC suppresses luteolysis and induces an increase in the number of corpora lutea (CL) without affecting the estrous cycle (Gaytan et al. 2001; Kumazawa et al. 2009).
Rodents, including rats and mice, have features that do not form a truly functional CL during the ovarian cycle unless mating results in pseudopregnancy or pregnancy (Stouffer 2006). In rats, CL are classified into two main types: new CL, which are newly formed by the current ovulation, and old CL, which are CL remaining from prior estrous cycles (Bowen and Keyes 2000). These types of CL are generated and regress over several cycles. New and old CL are morphologically distinguishable at each estrous stage, and new CL drastically change their morphology and function of P4 secretion during the estrous cycle (Taketa et al. 2011b; Yoshida et al. 2009). Old CL do not have the function of P4 secretion but morphologically can be classified into distinct types at the estrous stage during regression (Taketa et al. 2011b).
In rats, PRL has both luteotrophic and luteolytic actions depending on the stage of the estrous cycle or in the pregnant or pseudopregnant state. During mating, cervical stimulation activates a neuroendocrine reflex of circadian nocturnal PRL surges, which maintain CL and its secretion of P4 (Rehm et al. 2007). Therefore, sustained PRL secretion preserves functional CL. Meanwhile, since preovulatory PRL surge in the rat estrous cycle induces regression of nonfunctional CL, sustained inhibition of PRL secretion prevents luteolysis (Gaytan et al. 2001; Rehm et al. 2007).
We have previously shown that EGME and atrazine, respectively, induces luteal cell hypertrophy and increases P4 secretion in their cells with and without PRL hypersecretion (Taketa et al. 2011a). Furthermore, EGME increased P4 synthesis factor expression both in new and in old CL, whereas atrazine stimulated P4 synthesis factors expression in new CL. These results indicated that EGME stimulates luteal steroidogenesis in both PRL-dependent and PRL-independent manners, while atrazine stimulates luteal steroidogenesis in a PRL-independent manner. Since the molecular biological effect in the CL was different between EGME and atrazine treatments, it suggested that these chemicals may also have different morphological influences on the CL. However, these influences have not yet been examined. Additionally, the luteal histopathological effect of BRC is still not fully understood. The present study describes the detailed morphological characterization of EGME, atrazine, or BRC on the CL by light and electron microscopy. Since BRC is the strong inhibitor of PRL release, this study also examines the PRL-independent morphological luteal effect of EGME under a PRL-inhibitory state by coadministration of EGME and BRC to normal estrous cycling rats.
Materials and Methods
Animals
Female 8-week-old Sprague-Dawley (Crl:CD) rats were purchased from Charles River Laboratories Japan, Inc. (Yokohama, Japan). After 2 weeks of acclimation, they were housed in a clean air room maintained at 23°C to 25°C and a relative humidity of 50% to 60% with a 12-hr light cycle. Commercial rodent chow (CRF-1; Oriental Yeast Co., Ltd., Tokyo, Japan) and drinking water were available ad libitum. Estrous cycle stage was determined each morning by vaginal smear throughout the experimental period. Only animals displaying 4-day estrous cycles were included in the experiments. The animal protocols were reviewed and approved by the Animal Care and Use Committee of the National Institute of Health Sciences, Japan.
Chemicals and Reagents
EGME, purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan), was dissolved in water for injection. Atrazine, purchased from Tokyo Chemical Industry (Tokyo, Japan), was suspended in 0.5 w/v% methyl cellulose 400 solution (Wako Pure Chemical Industries, Ltd.). BRC, purchased from Sigma-Aldrich (St. Louis, MO), was dissolved in 70% ethanol diluted with saline. Control animals received 0.5 w/v% methyl cellulose 400 solution by gavage and 70% ethanol diluted with saline by subcutaneous administration.
Study Design
EGME (300 mg/kg/day) or atrazine (300 mg/kg/day) was administered by gavage and BRC (2 mg/kg/day) was subcutaneously administered to rats once per day for 7 days. For evaluating the effects of EGME on the luteal cells in the absence of PRL, the animals were coadministrated with EGME (300 mg/kg/day, gavage) and BRC (2 mg/kg/day, subcutaneously; EGME + BRC) once per day for 7 days. Effective doses of EGME, atrazine, and BRC were selected based on previous reports (Dodo et al. 2009; Kumazawa et al. 2009; Shibayama et al. 2009; Taketa et al. 2011a). On the morning after the last treatment, the animals were euthanized by decapitation (n = 4–5/group). Considering the estrous cyclicity, the administration to all animals was started at the proestrus stage and the control and BRC-treated animals were euthanized at the diestrus stage after 7-day treatment. Since repeated administration of EGME, EGME + BRC, or atrazine disrupts the estrous cycle, animals in these groups did not synchronize to stage of the estrous cycle at necropsy.
Light Microscopy and Electron Microscopy
For light microscopic examination, unilateral ovary was fixed in 10 vol% neutral-buffered formalin and processed with hematoxylin and eosin (H&E) staining. The number of total CL and two types of hypertrophic CL were counted in two sections of the ovary. The vagina and uterus were also microscopically examined to confirm the estrous stage.
For ultrastructural analysis, contralateral CL was separately removed from the other side of the ovary used above for light microscopy and was fixed in 3% glutaraldehyde in 0.1 M phosphate buffer solution (PBS) for 24 hr. After rinsing in the same buffer, the CL was postfixed in 1% osmium tetroxide (OsO4) in 0.1 M PBS for 2 hr. The tissue was then dehydrated through a graded series of ethanol and embedded in epon812. One-micron sections were stained with toluidine blue, the CL types (CL-V or CL-E) were distinguished, and selected blocks were thin sectioned. The grids were stained with 1% uranyl acetate for 20 min at room temperature (RT), rinsed 3 times with distilled water (DW), stained with lead citrate (Millonig 1961) for 10 min at RT, rinsed 3 times with DW, and air-dried. The grids were examined with the JEM-1400 Electron Microscope (JEOL Ltd., Tokyo, Japan) and photomicrographs were obtained with an ORIUS CCD camera (Gatan, Inc., Pleasanton, CA).
Results
Light Microscopic Luteal Changes by EGME, Atrazine, or BRC Treatment
During the diestrus stage, the control group produced luteal cells of the new CL, which exhibited pale eosinophilic staining in the cytoplasm with sparse and fine vacuolation (Figure 1a); while the luteal cells of the old CL exhibited strong eosinophilic staining in the cytoplasm and the additional presence of interstitial cells, such as endothelial cells and fibroblasts (Figure 1b). In the BRC group, the luteal features of the new CL were similar to the control group (Figure 1c). Although all BRC-treated animals represented normal estrous cyclicity and the luteal cell shape and size of the old CL in the BRC group showed no apparent changes compared to the control group, the proportion of interstitial cells in the old CL of the BRC group was lower than that in the old CL of the control group (Figure 1d). In the EGME, EGME + BRC, and atrazine groups, a large number of luteal cells were hypertrophic and the CL were categorized into two types according to the hypertrophic luteal cell types: CL hypertrophy, vacuolated type (CL-V) and CL hypertrophy, eosinophilic type (CL-E). It was hard to distinguish the new CL from the old CL because of the luteal cell hypertrophy. The luteal cells of CL-V contained rich and fine vacuoles in cytoplasm (Figure 2a, c, and e), whereas those of CL-E had eosinophilic and abundant cytoplasm (Figure 2b, d, and f). Furthermore, each luteal cell of both CL types had distinct cell borders. The individual luteal cells of CL-V and CL-E in the EGME + BRC group were more hypertrophic than those in the EGME and atrazine groups (Figure 2). In the EGME and EGME + BRC groups, CL-E were more frequently observed than CL-V, while both CL-V and CL-E were uniformly observed in the atrazine group (Table 1). Interstitial cells in CL-V were sparse, whereas some CL-E contained many interstitial cells in the EGME, EGME + BRC, and atrazine groups.
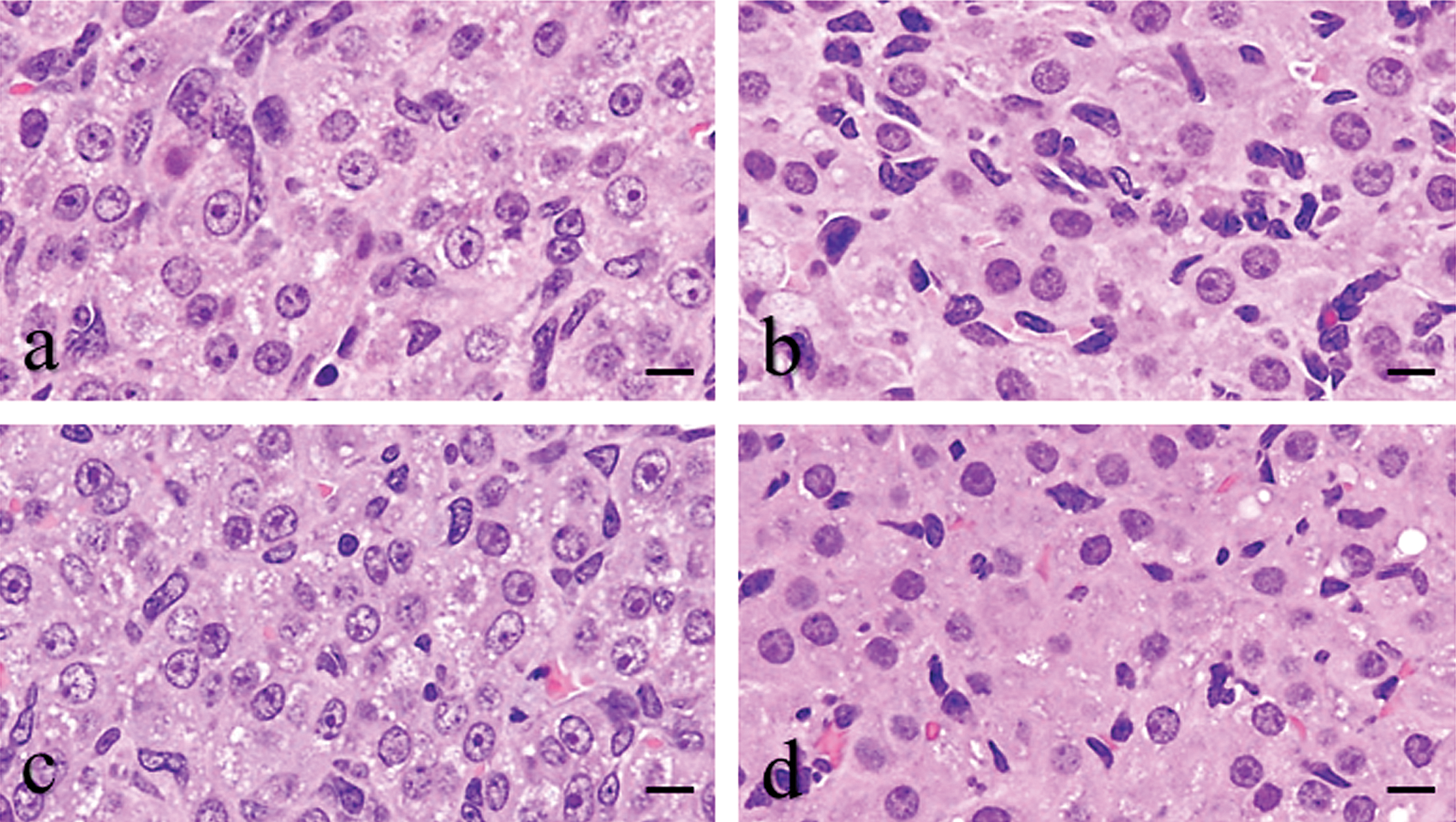
Light microscopic changes in new corpora lutea (CL) (a) and old CL (b) in the control group at the diestrus stage, and CL treated by BRC (c and d) for 7 days. The bromocriptine (BRC)-treated new CL (c) was not different from the control new CL (a) whereas the old CL showed less interstitial cells (d) compared to the control CL (b). H&E staining. Bars represent 10 µm.
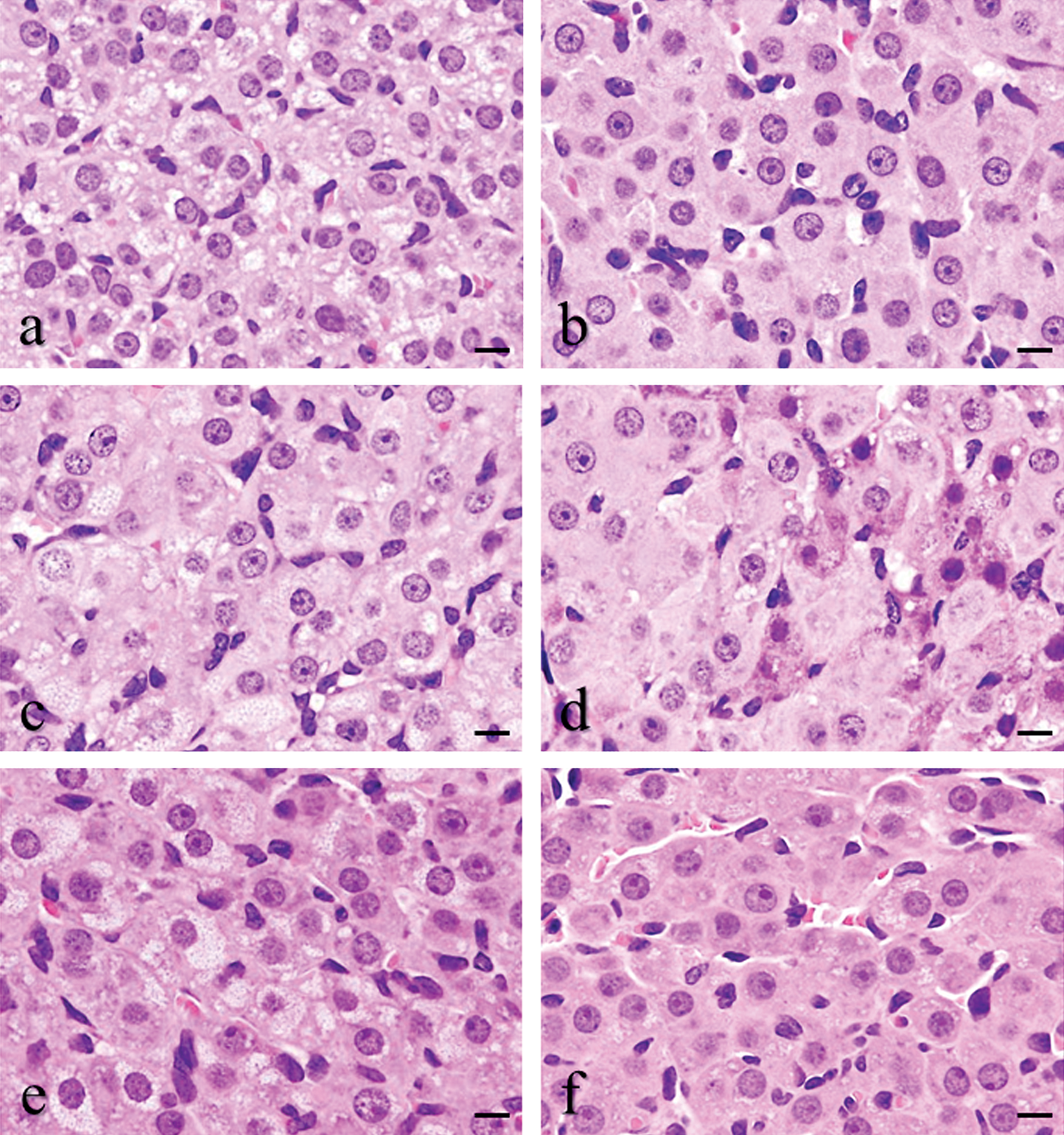
Light microscopic changes in corpora lutea (CL) treated by ethylene glycol monomethyl ether (EGME) (a and b), EGME + bromocriptine (BRC) (c and d), or atrazine (e and f) for 7 days. The hypertrophic luteal cells of CL-V had fine vacuolated cytoplasm by EGME (a), EGME + BRC (c), or atrazine (e) treatment. The hypertrophic luteal cells of CL-E contained eosinophilic and abundant cytoplasm by EGME (B), EGME + BRC (d), or atrazine (f) treatment. H&E staining. Bars represent 10 µm.
Number and percentage of CL-V and CL-E in total CL

Values are presented as the mean ± SEM (n = 3–4/group). The number of CL was counted in two sections of the ovary. − = not applicable; BRC = bromocriptine; EGME = ethylene glycol monomethyl ether; CL-V = corpora lutea, vacuolated type; CL-E = corpora lutea, eosinophilic type. SEM = standard error of the mean.
Ultrastructural Luteal Changes by EGME, Atrazine, or BRC Treatment
There was no obvious luteal ultrastructural difference between new CL and old CL at the diestrus stage in the control group. During the diestrus stage, lipid droplets, smooth and rough endoplasmic reticulum (SER and RER, respectively), and mitochondria were uniformly distributed in the luteal cells of the control group (Figures 3a and 4a). The luteal cells of CL-V in the EGME, EGME + BRC, and atrazine groups contained abundant lipid droplets (Figure 3c-e). Although the large-sized of lipid droplets tended to be observed in the EGME-treated and EGME + BRC-treated luteal cells of CL-V, they were judged to be within normal ranges because of the various sizes of lipid droplets in the control luteal cells (Figure 3a, c, and d). The atrazine-treated luteal cells of CL-V showed similar-sized lipid droplets to the control group (Figure 3a and e). The luteal cells of CL-E in the EGME and EGME + BRC groups were characterized by uniformly well-developed SER (Figure 4c and d) and occasional abundant mitochondria. No obvious luteal changes were found in the atrazine-treated CL-E (Figure 4e). The ultrastructure of the luteal cells of the BRC group was similar to the control group (Figures 3a, b and 4a, b).
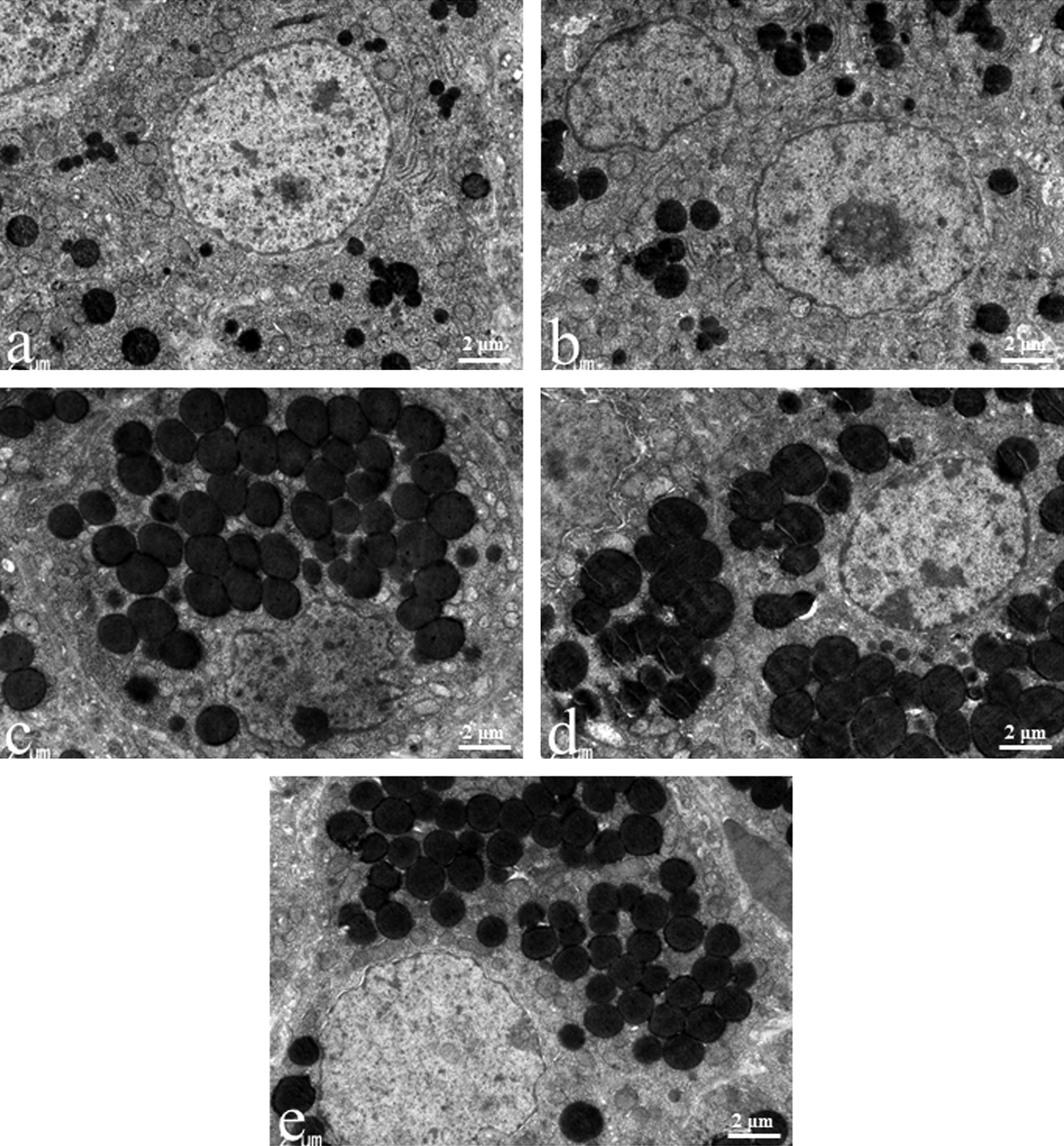
Ultrastructural luteal changes induced by treatment with bromocriptine (BRC), ethylene glycol monomethyl ether (EGME), EGME + BRC, or atrazine. The hypertrophic luteal cells of corpora lutea, vacuolated type (CL-V) contained abundant lipid droplets following EGME (c), EGME + BRC (d), or atrazine (e) treatment compared to the control new CL at the diestrus stage (a). BRC-treated new CL (b) was not different from the control new CL. Magnification 8,000×.
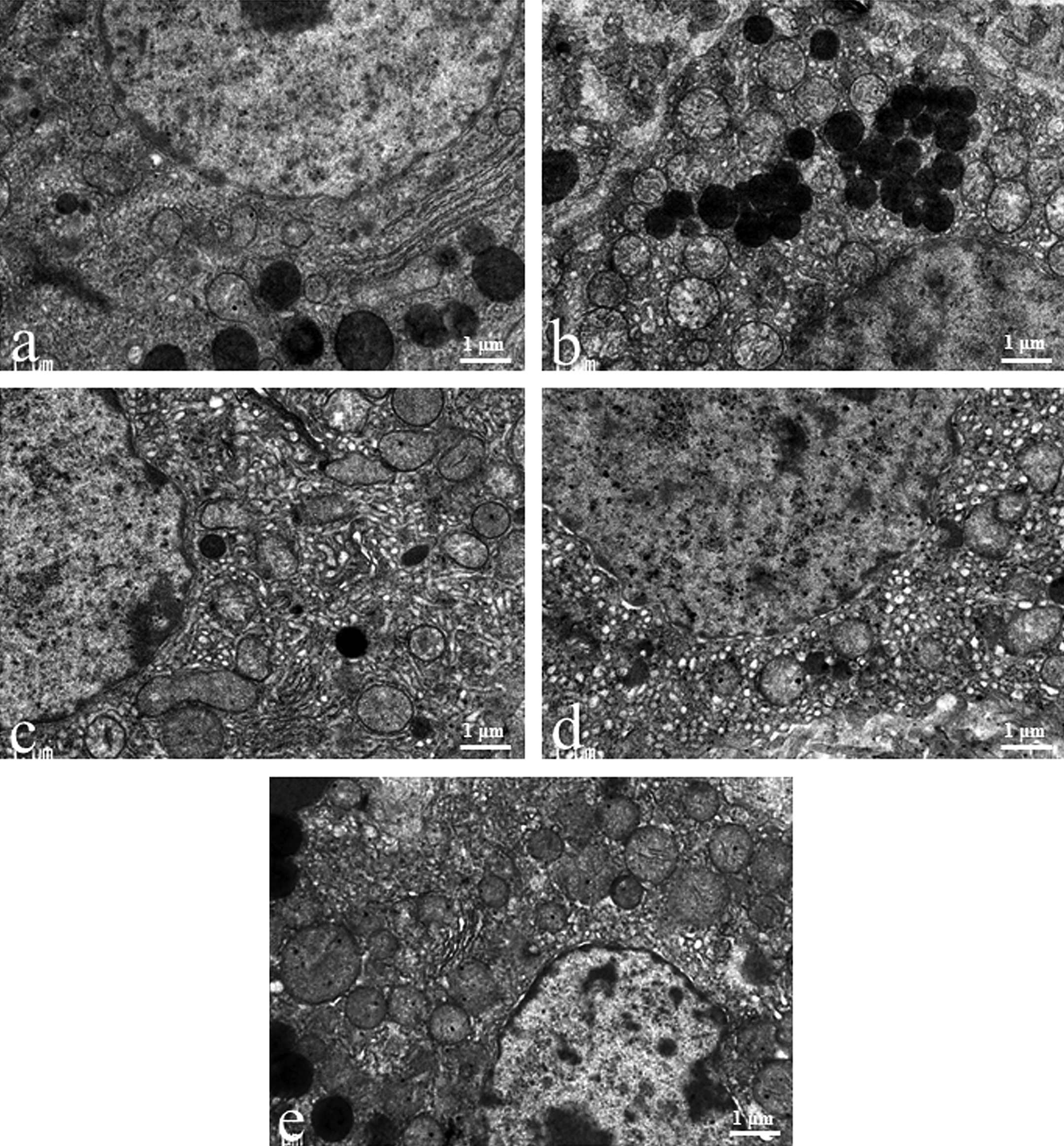
Ultrastructural luteal changes induced by treatment with bromocriptine (BRC), ethylene glycol monomethyl ether (EGME), EGME + BRC, or atrazine. The hypertrophic luteal cells of corpora lutea, eosinophilic type (CL-E) contained well-developed smooth endoplasmic reticulum following EGME (c) or EGME + BRC (d) treatment compared to the control new CL at the diestrus stage (a). BRC-treated new CL (b) or atrazine-treated CL-E (e) was not obviously different from the control new CL. Magnification ×15,000.
Discussion
This study focused on light- and electron-microscopic changes of the luteal cells induced by EGME, atrazine, or BRC in rats. A large number of CL representing luteal cell hypertrophy by 7 days of treatment with EGME or atrazine indicate that EGME and atrazine affect both new and old CL. This change was also observed by coadministration of EGME and BRC, indicating that EGME morphologically affects luteal cells without PRL release. Considering our previous study that the concurrent administration of EGME with BRC stimulated P4 secretion, upregulated steroidogenic factor expression levels, and downregulated luteolytic factor expression levels in luteal cells without PRL hypersecretion (Taketa et al. 2011a), the main luteal stimulatory effect of EGME seems to be independent of PRL. In the EGME, EGME + BRC, and atrazine groups, the CL hypertrophy was classified into two types by light microscopy, CL-V and CL-E. The proportions of CL-V and CL-E were different among the treatment groups, suggesting that chemically induced luteal hypertrophy does not mean similar morphology. Although the low proportion of interstitial cells in the BRC-treated old CL indicates that BRC treatment inhibited regression of old CL, their luteal cells were not hypertrophic. This change was presumably caused by the inhibition of the PRL surge that initiates structural luteolysis in rats, by the D2 agonistic effect (Gaytan et al. 2001). Since the animals had passed only one complete estrous cycle during the administration, the increase in CL number by BRC treatment was not obvious. The increase in the number and size of CL-V and CL-E by the EGME + BRC treatment in comparison to the EGME-only treatment was considered to be due to the luteolysis inhibitory effect of BRC.
Electron microscopic observation revealed that luteal hypertrophy of CL-V in the EGME, EGME + BRC, and atrazine groups was caused by excessive storage of lipid droplets. On the contrary, luteal hypertrophy of CL-E in the EGME and EGME + BRC groups resulted from excessive SER development. Occasional increase in mitochondria suggested the possibility that EGME may also affect mitochondrial function and development in CL-E. In the pregnant rats, luteal cells became enlarged and the amount of SER greatly increased by 7 to 10 days of pregnancy (Long 1973). Since the increase in PRL secretion is observed in the first 10 days of pregnancy (Egli et al. 2010), PRL seems to enhance SER development in the new CL. Considering the same luteal change in the EGME + BRC-treated CL-E, EGME may also stimulate luteal SER development in a PRL-independent manner. The CL reached a maximum size and had maximal P4 secretory activity at approximately 16 to 17 days of pregnancy, and the luteal cells exhibited strong proliferation of SER membranes (Long 1973; Meyer and Bruce 1984). These reports indicate that luteal cell hypertrophy in the functional CL is typically due to the development of SER. Therefore, EGME-induced CL-E, which contained well-developed SER, support sufficient P4 secretory function. On the other hand, lipid accumulation in the CL was seen only in early pregnancy in rats (Long 1973). These observations indicate a possibility that luteal morphology might be altered by the duration of the stimulatory period. That is, short-term or intermittent stimulation may result in lipid accumulation to uptake the source of cholesterol, whereas long-term or continuous stimulation seems to induce SER development for the activation of P4 synthesis in the luteal cells. It was previously shown that glucocorticoids stimulated the accumulation of lipid droplets and steroidogenesis with increased 20α-hydroxysteroid dehydrogenase (20α-HSD) secretion in rat CL (Towns et al. 1999). This previous report and the present study suggest that CL-V also have the capacity for steroid synthesis though their P4 secretion capacity may be lower than CL-E due to the increase in 20α-HSD.
The origin of CL-V and CL-E is still not fully understood. From our previous study, EGME elicited luteal changes in both the new and old CL, with the new CL being the principal target (Taketa et al. 2011a). Given the possibility of high P4 synthesis capacity in CL-E, the CL-E may have originated from the new CL in EGME-treated and EGME + BRC-treated CL. However, light microscopic changes revealed many interstitial cells in several CL-E, indicating that some CL-E may have been from the old CL. The scarce interstitial cells in CL-V suggest that the new CL seem to be the origin of EGME-treated and EGME + BRC-treated CL-V. We have already shown the increase in 3β-hydroxysteroid dehydrogenase (3β-HSD) expression in the old CL by EGME treatment (Taketa et al. 2011a). Since 3β-HSD localizes in the SER (Stocco et al. 2007), it is probable that some of the CL-E, which contains abundant SER, originated from old CL in the EGME and EGME + BRC groups.
Considering the scarce interstitial cells and high proportion of atrazine-treated CL-V in the present study and new CL was the main stimulatory target of atrazine in our previous study (Taketa et al. 2011a), atrazine-treated CL-V was assumed to be derived from new CL. This observation suggests that atrazine stimulates the accumulation of lipid droplets in the new CL. It is still unclear why atrazine-treated CL-E became hypertrophic without apparent ultrastructural change. One possibility is that the atrazine-treated CL-E was not functionally activated. Since atrazine did not stimulate old CL in our previous study, the atrazine-induced CL-E in the present study might be derived from old CL (Taketa et al. 2011a). The further morphological investigations about atrazine-induced CL-E are required.
In our previous study, EGME induced luteal hypertrophy at higher than 100 mg/kg/day (Taketa et al. 2010). Similarly, in vitro, EGME stimulates P4 secretion at concentrations more than 100 mg/kg in human and rat luteal cells (Almekinder et al. 1997; Davis et al. 1997). From these data, a similar affect level of EGME in luteal cells between human and rat could be predicted. The luteal toxicity of atrazine in human has been undetermined. Although the affecting dose level of 300 mg/kg/day in this study seems to be quite high compared to the human exposure level of atrazine reported in United States (1.8–6.1 µg/kg/day; Gammon et al. 2005), its potency of luteal toxicity should be noted. Further investigations about luteal toxicity of EGME and atrazine in humans are required.
In conclusion, the morphological differences of CL-V and CL-E in response to EGME and atrazine treatment reflect a distinct capacity of each CL type for steroidogenesis. However, the present study is a short-term investigation of luteal toxicity in response to EGME and atrazine; therefore, further studies investigating the long-term morphological effects are required.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by Health and Labour Sciences Research Grants for Risk Chemical Substances from the Ministry of Health, Labour and Welfare, Japan [H22-Toxicol-003].
Acknowledgments
We thank Ms. Tomomi Morikawa, Ms. Ayako Kaneko, and Ms. Yoshimi Komatsu for their excellent technical assistance. We also appreciate Dr. Satoru Hosokawa and Dr. Akira Inomata (Drug Safety Japan, Global Drug Safety, Biopharmaceutical Assessments Core Function Unit, Eisai Product Creation Systems, Eisai Co., Ltd.) for their helpful suggestions.
