Abstract
Ethylene glycol monomethyl ether (EGME) is a known reproductive toxicant that induces luteal hypertrophy in rat ovaries. In this study, we characterized the histopathological features of corpora lutea (CL) from EGME–treated rats and compared them with normal CL formation and regression. Normally cycling female Sprague-Dawley rats were treated with 5-bromo-2′-deoxyuridine (BrdU) intraperitoneally on the morning of estrus and their ovaries were examined 1 (metestrus), 4 (estrus), 8 (estrus), or 12 (estrus) days later to observe the transition of BrdU-labeled cells within in the CL. CL at each time point of estrus stage were classified into 4 types: Type I (newly formed CL), Type II (mature CL), Type III (regressing CL), and Type IV (residual CL). CL almost fully regressed within 4 estrus cycles. In contrast, in female rats given EGME orally (30, 100, or 300 mg/kg for 2 or 4 weeks), luteal cells were hypertrophic with abundant cytoplasm. Although the size of CL varied, all CL in EGME–treated rats had histological features similar to Type II CL, but they were more hypertrophic with less apoptosis. These results suggest that EGME has a luteal hypertrophic effect on all CL phases, including regression.
Introduction
Ethylene glycol monomethyl ether (EGME), an important organic solvent in paints, printing inks, thinners, and photoresists, causes significant hematologic and central nervous system disturbances following occupational exposure (Hardin 1983). In addition, adverse effects on the male and female reproductive systems have been reported (Anderson et al. 1987; Chapin et al. 1984; Foster et al. 1983; Johanson 2000; National Toxicology Program 1993; Welsch 2005). In the female reproductive system, EGME and its active metabolite, 2-methoxy acetic acid (MAA), have been shown to induce hypersecretion of progesterone from ovarian luteal cells both in vivo and in vitro (Almekinder et al. 1997; Davis et al. 1997). Recently, we reported that EGME–treated rats showed prolonged estrus cycle, ovulatory inhibition, and corpora lutea (CL) hypertrophy characterized by round to polygonal luteal cells with abundant vacuolated cytoplasm (Dodo et al. 2009). Davis et al. (1997) reported that 300 mg/kg of EGME elevated serum progesterone in rats within 32 hours after dosing, whereas serum estradiol, follicle stimulating hormone (FSH), luteinizing hormone (LH), and prolactin (PRL) remained at baseline levels, suggesting the enlarged CL was attributed to inhibition of luteal cell death. Given these observations, there is the possibility that luteal hypertrophy induced by EGME is a result of the persistence of CL that arise from normal estrus cycling during EGME treatment. However, in this scenario, it is not clear which type of CL is affected by EGME.
In normal rat ovaries, multiple types of CL are generated and regress over several estrus cycles. In spite of some comprehensive reports illustrating luteal appearance at each stage of the estrus cycle (Hubscher et al. 2005; Westwood 2008; Yoshida et al. 2009; Yuan and Foley 2002), there are few reports describing luteal morphology during normal cycling. For accurate morphological evaluation of ovarian toxicants, it is essential to characterize normal luteal morphology during CL generation and regression.
In the present study, we investigated changes in luteal morphology during regression using the 5-bromo-2′-deoxyuridine (BrdU) pulse-chase labeling method. Granulosa cells of antral follicles just before ovulation, which are actively dividing during BrdU administration, are labeled positive (Gaytan et al. 1996). Since luteal cells do not proliferate after ovulation (Stocco et al. 2007), existing luteal cells are BrdU negative, and the BrdU–positive cells from the ovulatory follicle that were transformed into luteal cells in the CL can be followed through regression and defined luteal stages. In addition, interstitial cells, including endothelial cells and fibroblasts in newly formed CL at the estrus stage, could also be followed. We then morphologically classified luteal types in normally cycling rats and used this classification system to further elucidate the mechanism of EGME–induced luteal hypertrophy.
Materials and Methods
Animals
Nonpregnant, mature female Sprague-Dawley (Crl:CD (SD)) rats from Charles River Laboratories Japan, Inc. (Shiga, Japan) were used. 8-week-old and 11-week-old rats were used in experiments 1 and 2, respectively. After 1 week of acclimation, they were housed in a clean air room maintained at 20°C–23°C and a relative humidity of 48–69% with a 12-hour light cycle, and provided approximately 14 g/day of CRF-1 commercial rodent chow (Oriental Yeast Co., Ltd., Tokyo, Japan). Potable water was provided to animals in all studies ad libitum. Estrus cycle stages were determined every morning by vaginal smear throughout the experimental period. The study protocol was approved by the Laboratory Animal Care and Use Committee and was performed in compliance with the Laboratory Animal Policy of Eisai Co., Ltd.
Chemicals
Ethylene glycol monomethyl ether (EGME) was obtained from Wako Pure Chemical Industries, Ltd. (Osaka, Japan), and was dissolved in water for injection in a volume of 5 mL/kg body weight.
Study Design
Experiment 1: Luteal Classification of Normal Estrus Cycling Rats
To investigate the morphological changes from CL formation to regression in normally cycling rats, the animals were intraperitoneally administered 5-bromo-2′-deoxyuridine (BrdU) (B1575; Tokyo Chemical Industry Co., Ltd., Japan) at a dose of 50 mg/kg body weight on the morning of estrus (about 9:00 AM). The estrus cycle was confirmed by vaginal cytology to be normal during the experimental period. BrdU is readily incorporated into nuclei during the DNA synthetic phase of the cell cycle (S-phase) and is detected immunohistochemically with an anti-BrdU antibody. The rats were euthanized by exsanguination from the abdominal aorta under isoflurane anesthesia on 1 (metestrus), 4, 8, or 12 days (latter 3 points were estrus) (n = 3 or 4 per time point for each group) after the BrdU treatment. The ovaries, uterus, and vagina were fixed in 10 vol% neutral-buffered formalin and embedded in paraffin wax. Paraffin sections were stained with hematoxylin and eosin (H&E) and Masson’s trichrome for microscopic examination. The sections were also subjected to immunohistochemical demonstration of BrdU pulse-labeled cells. In BrdU immunostaining, ovarian sections were deparaffinized, heated in Target retrieval solution (Dako Corporation, Glostrup, Denmark) at 121°C for 15 min, washed in distilled water, treated with 90% methanol containing 3% H2O2 for 5 min at room temperature (RT), and washed 3 times in phosphate-buffered saline (PBS). Sections were then incubated with BrdU antibody (×100, M0744; Dako) for 1 hour at RT. After washing 3 times in PBS, the sections were then incubated with the secondary antibody (EnVision + Dual Link System-HRP, Dako) for 1 hour at RT. The reaction products were visualized with 3.3′-diaminobenzidine (DAB, Muto Pure Chemical Co., Ltd., Tokyo, Japan). The sections were counterstained with Mayer’s hematoxylin. To test the specificity of immunostaining, negative controls were run without the primary antibodies. In the present study, using BrdU pulse-chase labeling, CL of normal rats at estrus stage were described and classified into the 4 morphologic types (Types I–IV), from newly formed (Type I) to fully regressed (Type IV).
Experiment 2: Histopathological Examination of the Ovaries in EGME-treated Rats
EGME was orally administered to female rats (10 animals/group) at dose levels of 0, 30, 100, or 300 mg/kg for 2 or 4 weeks. Dose levels were selected based on the relevant literature (Davis et al. 1997; National Toxicology Program 1993). A control group received an equivalent volume (5 mL/kg) of water for injection. The animals were observed daily for general condition, and body weights and food consumption were measured once every 2 to 4 days. On the next day of the last treatment, animals were anesthetized with isoflurane and euthanized by exsanguination from the abdominal aorta. The ovaries, vagina, and uterus were fixed in 10 vol% neutral-buffered formalin and embedded in paraffin. Both ovaries were transversely dissected to examine their maximum areas. The paraffin sections were stained with H&E, and silver impregnation and Masson’s trichrome staining for the detail examination of the interstitium in CL. To detect apoptotic cells, immunostaining of single-stranded DNA (ssDNA) was performed according to the procedure described above. Antigen retrieval treatment was performed with Liberate Antibody Binding (L.A.B.) solution (Polysciences, Inc., Warrington, PA, USA) for 10 min at RT. For the first antibody treatment, ssDNA antibody (×800, A4506; Dako) was used for 1 hour at RT.
Results
Luteal Classification of Normally Cycling Rats
At 1 day after the BrdU labeling (at metestrus stage), the granulosa cells of antral follicles were positive (Figures 1a and 1b).
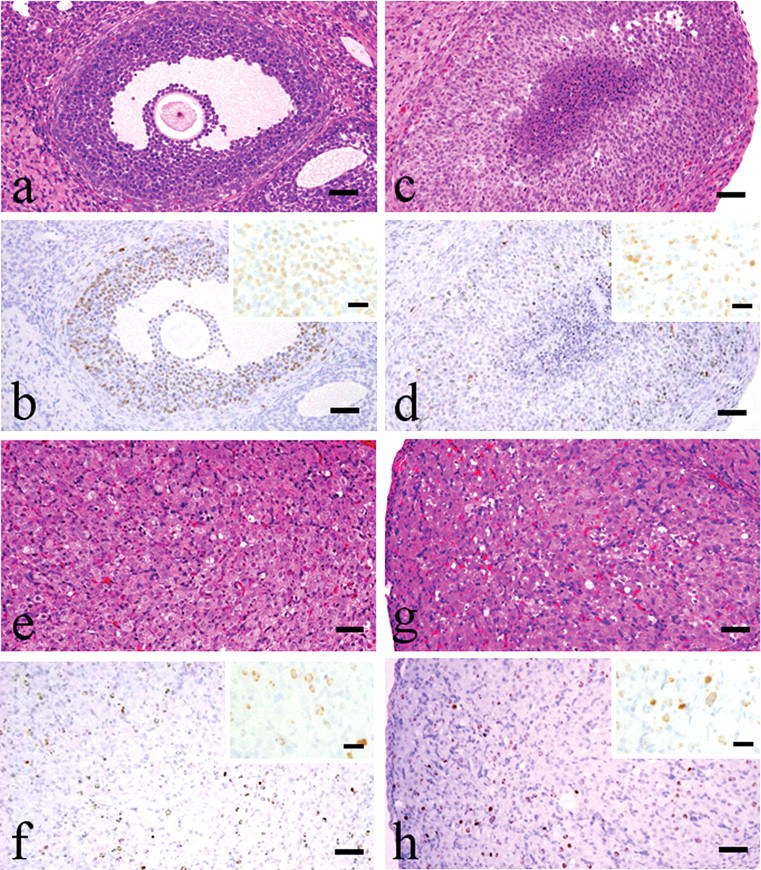
Histological characterization of CL by chasing BrdU-positive granulosa and luteal cells. (a and b) One day after BrdU labeling, granulosa cells of antral follicles were positive for anti-BrdU antibody. (c and d) The BrdU-positive granulosa cells became the newly formed CL (Type I CL) after 1 estrus cycle (4 days) of the labeling. (e and f) CL attained their largest size after the 2 cycles post labeling (8 days) (Type II CL) (g and h). After 3 cycles of post labeling (12 days), CL became smaller, with morphology typical of regression (Type III CL). Bars represent 50 µm, and bars in small windows represent 20 µm.
At 4 days after BrdU labeling (at estrus stage), the luteal cells of new CL formed shortly after ovulation were BrdU-positive after the first estrus cycle (Figures 1c and 1d). Newly formed CL were composed of luteal cells with a small amount of basophilic cytoplasm, and they occasionally had a central fluid-filled cavity (Type I: newly formed CL). In preexisting CL, luteal cells were negative and interstitial cells were positive for BrdU. Granulosa cells in antral follicles were also BrdU positive.
At 8 days after labeling (at estrus stage after the second estrus cycle), CL reached maximum size and were characterized by luteal cells with abundant eosinophilic cytoplasm and distinct cell borders, and indistinct interstitial cells (Type II: mature CL) (Figures 1e and 1f). Luteal cells of Types I and II CL, and interstitial cells in some other CL, were also positive for BrdU.
At 12 days after labeling (at estrus stage after the third estrus cycle), the CL were smaller than at Day 8, they had conspicuous interstitium, and luteal cell borders were slightly indistinct (Type III: regressing CL) (Figures 1g and 1h). BrdU-positive cells gradually decreased as the cycles passed. The luteal cells of Type I CL were almost all negative for BrdU, but luteal cells of Type II and Type III CL and interstitial cells in CL that were further regressed than Type III CL were still positive for BrdU.
Since the interstitial cells of Type I and II CL were positive for BrdU on 1 day after BrdU labeling (Figures 2a and 2b), these CL could be traced for over 3 estrus cycles. The main components of interstitial cells were endothelial cells and fibroblasts. The CL after the third estrus cycle (after 8 or 12 days of labeling) had more conspicuous interstitium and were smaller than Type III CL (Type IV: residual CL) (Figures 2e and 2f). At 12 days after labeling, almost all Type IV CL were regressed after the 4th estrus cycle (Figures 2g and 2h).
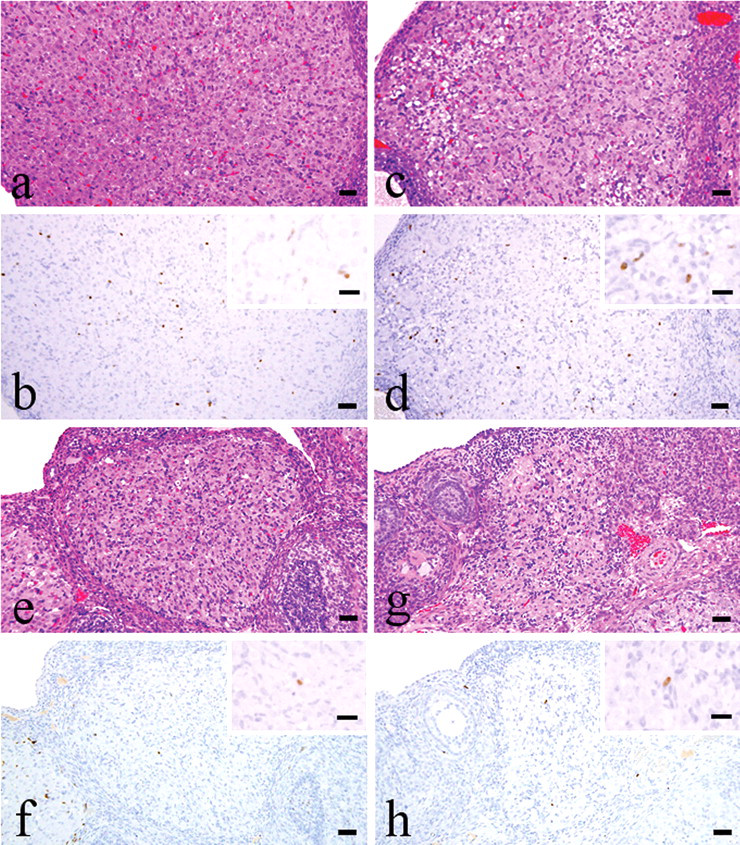
Histological characterization of CL by chasing BrdU-positive interstitial cells of CL (a and b). One day after BrdU labeling, interstitial cells of Types I and II CL were positive for anti-BrdU antibody (Type II CL) (c and d). The BrdU-positive Type III CL were observed after 1 or 2 estrus cycles (4 or 8 days) post labeling (e and f). After 2 or 3 cycles (8 or 12 days) post labeling, the 3-cycles–passed CL showed more conspicuous interstitium and smaller size than Type III CL (Type IV CL) (g and h). After 4 estrus cycles of the new formation (on Day 12 after labeling), CL had almost completed regression. Bars represent 50 µm.
Ethylene Glycol Monomethyl Ether–treated CL
EGME induced continuous diestrus at ≥ 100 mg/kg after 2-week administration and resulted in prolonged estrus interval and decreased frequency of estrus, as determined by vaginal cytology. After 4-week administration, similar changes were noted at ≥ 100 mg/kg. There were no significant changes in ovarian weights after 2- or 4-week administration.
Histopathologically at 2 weeks after administration, ovarian changes were observed at ≥ 100 mg/kg and characterized by luteal hypertrophy and increased numbers of large atretic follicles with many pyknotic cells and a thin epithelial cell layer. No newly formed CL (Type I CL) were present (Figure 3 ). Morphologically hypertrophic CL contained round to polygonal luteal cells with abundant vacuolated cytoplasm, and ssDNA immunostaining showed only rare apoptotic cells (Figure 4 ).
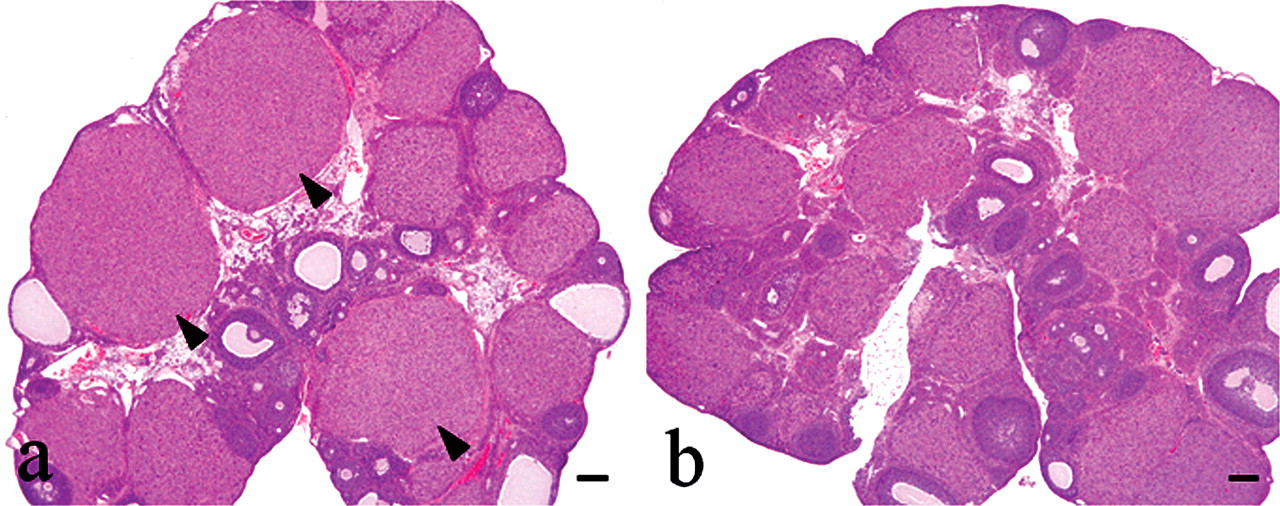
H&E staining in ovaries treated with vehicle or 300 mg/kg EGME for 2 weeks. (a) The EGME–treated ovaries contain well-organized and hypertrophic CL (arrowheads). (b) A variety of luteal morphologies are observed in control animals. Bars represent 200 µm.
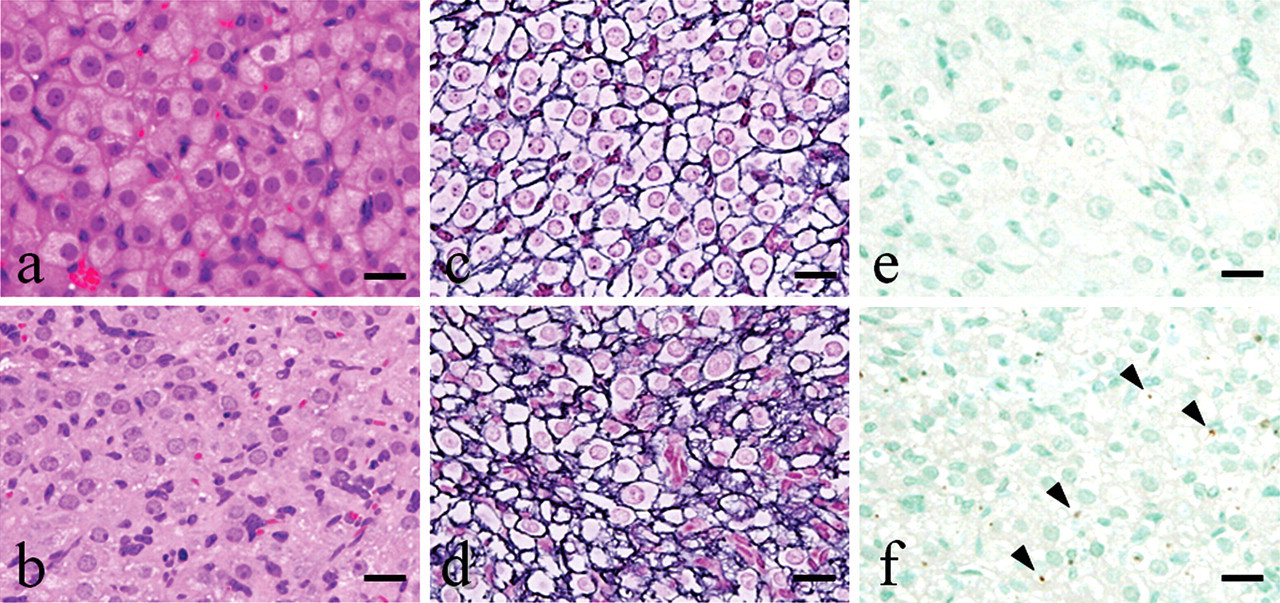
EGME–induced luteal morphological changes with H&E staining (a and b), silver impregnation staining (c and d), and anti-ssDNA immunostaining (e and f). Luteal cells become hypertrophied with abundant vacuolated cytoplasm (a) and distinct cell borders (c), and there is a paucity of apoptotic cells (e) with EGME treatment. Luteal cells at estrus of concurrent controls show a variety of cell size (b), indistinct cell borders (d), and frequent apoptotic cells (arrowheads; f). Bars represent 20 µm.
The most characteristic findings in EGME–treated ovaries were the presence of small, irregular CL composed of well-developed luteal cells, few apoptotic cells, and conspicuous interstitial tissues for 2- and 4-week treatments. These morphologies of the interstitium were identical to regressing Type III and Type IV CL by Masson’s trichrome staining (Figure 5 ). There were no histopathological differences in CL between the 2-week and 4-week studies, but large atretic follicles were more conspicuous after the 4-week treatment. There were dose-related changes in 100 and 300 mg/kg groups, but no morphological changes in CL were observed in the 30 mg/kg group. No histopathological changes were observed in small follicles including primordial and primary follicles, medium follicles, or preantral follicles at any dose. Changes in other reproductive organs, mucinous degeneration of the vagina characterized by incomplete basal stratum and accumulation of cell debris, and endometrial hyperplasia in the uterus were observed at 100 and/or 300 mg/kg and were considered secondary to the ovarian changes.
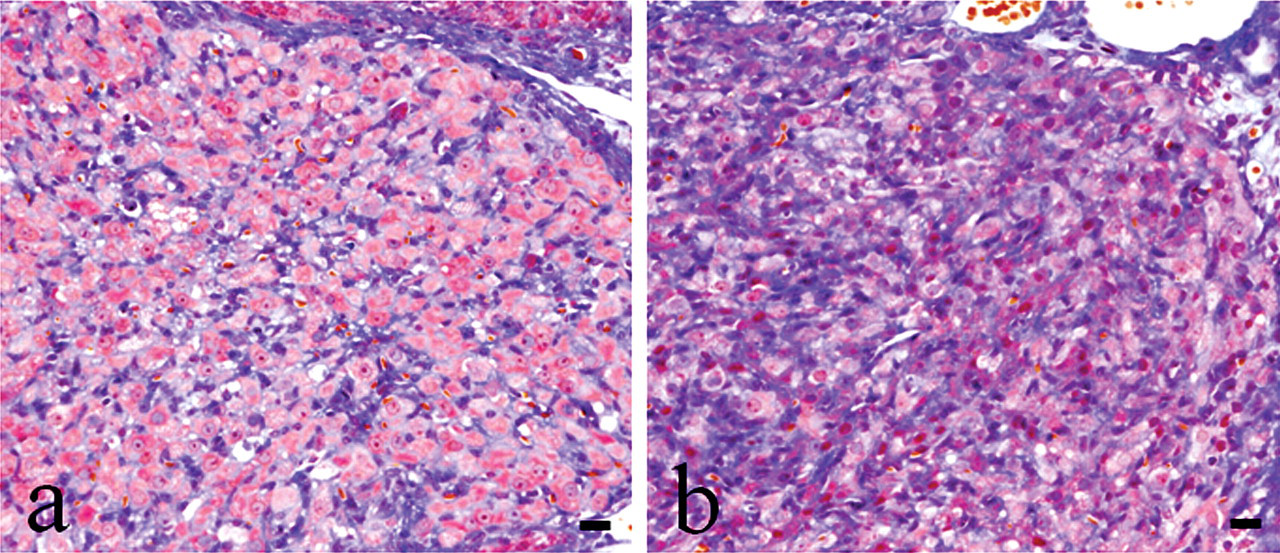
Histological features of the EGME–treated regressing CL with Masson’s trichrome staining. The histological comparison of EGME-treated Type III CL (a) and Type III CL of concurrent controls (b) showed that EGME-treated luteal cells have abundant vacuolated cytoplasm and clear cell borders, even though they had obvious interstitium and indistinct silhouettes. Bars represent 20 µm.
Discussion
In the present study, morphological classification of normal CL from formation through regression was accomplished using H&E microscopic examination, immunohistochemistry, and BrdU pulse-chase labeling method. This classification scheme was then used to characterize the effects of EGME on the female reproductive system in the rat, predominantly on ovarian CL.
The BrdU pulse-labeling method is a well-described and useful tool for assessing cellular proliferation (Gratzner 1982; Rosiepan et al. 1994; Soriano et al. 1991). Gaytan et al. (1996) previously reported the proliferative activity in the different ovarian compartments using this method, and they showed that granulosa and theca cells in Graafian follicles, and luteal interstitial cells were positive for BrdU. Our results are consistent with this previous report, with BrdU-positive cells observed in granulosa cells of antral follicles and interstitial cells of CL when BrdU was pulsed at the stage of estrus. Using this method, followed by serial sacrifice, we were able to trace BrdU-positive follicular and luteal cells though the normal estrus cycle and develop a useful system for classification of the morphological changes and characteristics of CL over time. An overview of the resultant luteal classification study is presented in Figure 6 .
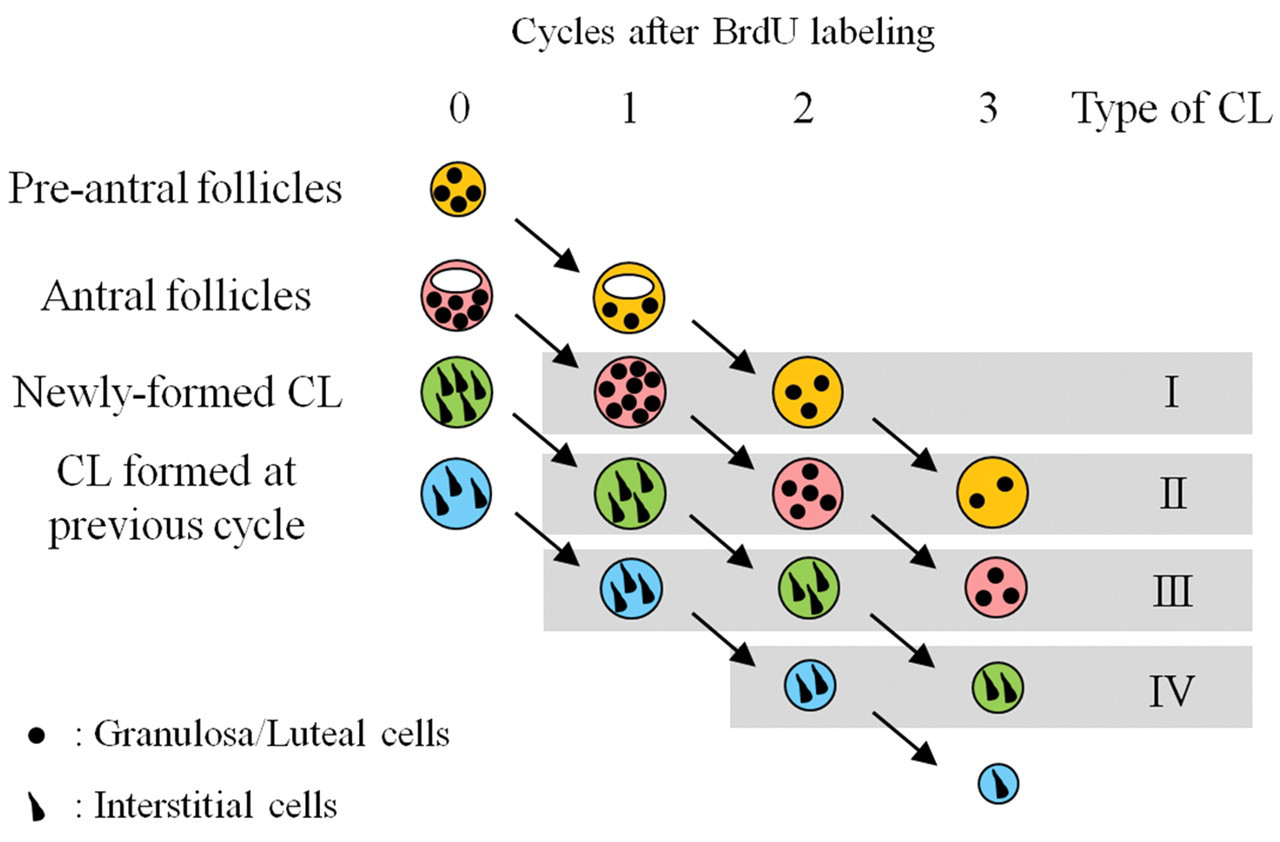
Overview of luteal classification as determined by BrdU pulse-labeling. This figure shows the transition of BrdU pulse-labeled cells treated on the morning of estrus. Granulosa cells in preantral and antral follicles (filled circles) were positive for BrdU at labeling (0 cycles after labeling), and they could be traced by Type II or Type III CL after 3 cycles of labeling, respectively. Interstitial cells of CL (filled wedge shapes) were positive for BrdU in newly formed and 1-cycle–passed CL at BrdU labeling, and they could also be traced after 3 cycles of the BrdU labeling. The grizzled zones show each type of CL that we classified in this study. Follicles and/or CL of the same color indicate the same origin of follicles or CL.
Based on the observations in this study, we confirmed that the lifespan of a CL in the rat, from formation to regression, spanned 4 estrus cycles, and we were able to classify CL into 4 morphologically distinct types at each estrus stage. The present study also demonstrated that the CL type at estrus could be classified using light microscopy without BrdU labeling. Previously, Imai et al. (1967) classified the CL at various stages of the estrus cycle in mice into 4 types, namely, Type I (newly formed CL, characterized by luteal cells with wide cell spacing and no cellular cytoplasm); Type II (CL formed at the previous ovulation, with small cellular cytoplasm); Type III (CL formed at the latest ovulation, characterized by cells with well-defined cytoplasm that were in close contact); and Type IV (regressing CL, with indistinct luteal cell borders and cytoplasm). Their classification was different from the present results in rats, and this discrepancy may be a result of differences in definition of CL classification: we focused on the histological transition of CL at the time of repeat estrus after BrdU pulse-labeling, whereas they focused on the histological features of CL at each stage of the estrus cycle. To our knowledge, our study is the first report characterizing the morphological transition of CL, from newly formed to complete regression, in rodents using the BrdU pulse-chase labeling method. However, as luteal morphology differs from stage to stage within 1 estrus cycle, additional studies are needed to characterize luteal morphology characteristics at each stage of the estrus cycle in the rat. Yoshida et al. (2009) provided a practical guide to ovarian morphology in rats with normal cycling. They classified CL into 2 types, currently formed CL and previously formed CL, and characterized them at each stage of the estrus cycle. Their classification was very useful for rough evaluation of luteal toxicants, and our present classification of CL, which refers to the morphology of luteal development and regression, enables more detailed evaluation of ovarian toxicants.
In the present study, EGME-induced luteal hypertrophy in the rat was characterized microscopically by luteal cells with well-developed cytoplasm, regardless of luteal size and interstitial component, and decreased ssDNA–positive apoptotic cells as compared to untreated rats. Although the mechanisms of EGME-induced luteal hypertrophy have not been fully determined, low PRL level might play an important role. Davis et al. (1997) showed that EGME treatment secondarily suppressed the preovulatory PRL surge by sustaining high levels of progesterone. Therefore, we hypothesize that EGME–induced luteal hypertrophy is a result of direct luteal stimulation and a secondary decrease in luteal apoptosis. The preovulatory PRL surge at proestrus induces Fas-ligand expression in the T lymphocytes of the CL, which acts on luteal cells expressing Fas and induces luteal cell apoptosis by activating the Fas pathway (Kuranaga et al. 1999; Sugino and Okuda 2007). The vaginal mucification and hyperplasia of luminal epithelium in the endometrium observed in the present study also indicated high levels of progesterone (Nagaoka et al. 2002; Rehm et al. 2007; Yuan 1991). In recent years, Henley et al. (2009) reported that MAA, the primary metabolite of EGME, had histone deacetylase (HDAC)–inhibitory activities and imparted antiestrogenic effects on endogenous ER signaling in vivo and in vitro. Their data suggested that the reproductive abnormalities by EGME treatment may in part result from attenuation of endogenous ER-mediated signaling.
The EGME treatment–induced hypertrophic CL could not be classified into any of the types of CL observed in normally cycling rats. The morphological features in most CL formed after EGME treatment were similar to Type II CL in our classification. However, they were more hypertrophied with less apoptosis than Type II CL. In addition, CL that contained interstitial morphology consistent with regressing Type III and Type IV CL at the time of EGME treatment also contained hypertrophic luteal cells with abundant vacuolated cytoplasm and distinct cell borders. These results suggest that EGME affected not only mature CL, but also regressing CL. EGME has been known to stimulate luteal progesterone secretion in vitro (Almekinder et al. 1997; Davis et al. 1997). Thus, it was speculated that luteal hypertrophy induced by EGME was a result of both luteal stimulation and suppression of luteolysis.
In addition to the luteal changes, an increase of atretic follicles was also observed in most animals in both the 2- and 4-week treatment groups, and this observation was more conspicuous in the 4-week treatment group. Davis et al. (1997) had reported that the CL were hypertrophied without any follicular pathological changes in EGME-treated rats by Day 8, and they suggested the increase in size could be attributed to a decrease of luteal cell death. They concluded that elevated progesterone, induced by EGME, was the primary mediator of the subsequent reproductive alterations, including suppressed cyclicity, suppressed estradiol and pituitary hormones, and vaginal mucification similar to that which occurs in pregnant or pseudopregnant rats. The increased number of atretic follicles in the present study was considered a secondary change owing to prolonged disruption of the estrus cycle and anovulation.
Although no morphological effect in CL was observed at 30 mg/kg of EGME in the present study, our previous study showed disturbance of estrus cyclicity at ≥ 30 mg/kg of EGME in the fertility study (Dodo et al. 2009). However, Davis et al. (1997) reported that EGME had no effect on estrus cyclicity less than 100 mg/kg in a 1-week study. The reason for this apparent discrepancy in sensitivity is unclear, but it might be a result of the difference of age of rats used, and their feeding conditions, and dosing periods. Thus, our results indicated the possibility that EGME acts as an ovarian toxicant in rats at lower dose levels than expected. Further studies will be needed for better understanding of environmental risk assessment of EGME.
In conclusion, in rats, CL are formed and regress over 4 estrus cycles and can be classified into 4 morphologically distinct types corresponding to each subsequent estrus period. It is highly probable that luteal hypertrophy induced by EGME treatment is a result of a combination of a direct stimulatory effect of EGME on luteal cells and suppression of luteolysis in regressing and mature CL.
Footnotes
Acknowledgments
We thank Ms. Michiyo Shimada, Ms. Mayu Ozeki, and Ms. Sachiko Tsukada for their excellent technical assistance. We also appreciate Dr. Yvonne Van Gessel for her critical review and helpful comments.
