Abstract
To investigate effects of short-term mercury (Hg) exposure in tilapia (Oreochromis niloticus) including histopathological changes, Hg bioaccumulation, and protective role of metallothionein (MT) in different exposure routes, adult tilapias were intraperitoneally injected, orally intubated, or semistatically exposed to 0.5, 1, 2, 5 µg/g mercuric chloride. Histopathology, autometallography (AMG), inductive coupled plasma–atomic emission spectrometry (ICP-AES), and MT immunohistochemistry were determined at 0, 3, 6, 9, 12, and 15 days postexposure. Microscopic lesions were observed in the kidney, hepatopancreas, spleen, and intestine. AMG positive grains were found in renal tubule epithelium, melanomacrophage centers (MMCs), and intestinal epithelium of treated tilapias. Hg concentrations measured by ICP-AES in abdominal visceral organs were significantly higher than in other organs. All exposure routes caused lesions of increasing severity and Hg accumulations in a dose-dependent manner. Semistatic groups produced the highest intensity of lesions, AMG positive staining, as well as total Hg concentrations. Positive MT expression in renal tubule epithelium, pancreatic acini, and splenic MMCs was observed only in semistatic groups. The semistatic exposure route demonstrated the most significant microscopic lesions, Hg bioaccumulation, and MT expression.
Introduction
Mercury (Hg) is a highly toxic heavy metal that can cause numerous health problems and is classified as a carcinogen and mutagen (Agency for Toxic Substances and Diseases Registry [ATSDR] 1999; Bernard et al. 2001; Zahir et al. 2005). Humans may be exposed to environmental Hg in several ways, e.g., vapor inhalation, accidental direct ingestion, and consumption of Hg-contaminated food, especially fish and seafood (Oliveira-Ribeiro, Guimaräes, and Pffeiffer 1996; ATSDR 1999; Oliveira-Ribeiro et al. 1999). Therefore, humans might get high levels of Hg from bioaccumulation via contaminated food because humans are at the top of the food chain (Raldúa et al. 2007).
Fish from natural water sources are directly exposed to high concentrations of many toxic substances and chemical agents including Hg. Information on Hg levels in various species of fish has been reported, with concerns regarding human consumption (ATSDR 1999).
Using fish from natural water source as an environmental biomarker may help assess the risk of Hg consumption (Oliveira-Ribeiro et al. 2002; Jewett and Duffey 2007; Raldúa et al. 2007). Tilapia (Oreochromis niloticus) is a widely distributed fresh water fish that is important in global aquaculture, especially in Thailand (Maclean et al. 2002; Fishery Statistic Analysis and Research Group 2005). Environmental toxicological studies have used tilapia as the animal model in many studies because of its tolerance to water pollution (Chan 1995; Lam et al. 1998; Maclean et al. 2002; Atli and Canli 2003; Cheung, Lam, and Chan 2004).
Autometallography (AMG) is a potent histochemical staining procedure that is used to localize heavy metals in tissues and cells of fish and other species (Danscher 1984; Danscher et al. 2000; Loumbourdis and Danscher 2004; Alvarado et al. 2006). Several Hg toxicity studies used AMG to detect small amounts of Hg in the cells of various species (Danscher and Norgaard 1985; Woshner et al. 2002; Danscher and Stoltenberg 2006). However, we can find no study that uses this technique in tilapia. To quantify Hg accumulation in organs or tissues, the inductive coupled plasma–atomic emission spectrometry (ICP-AES) analytical method was used. ICP-AES has high sensitivity and precision and is used to accurately determine total Hg and other heavy metal concentrations in fish tissues (WHO 2003)
Metallothionein (MT) is a low-molecular-weight (6,000–7,000) and cystein-rich protein with high affinity for divalent cations such as Ag2+, Cd2+, Cu2+, Zn2+, and Hg2+ (Lau et al. 2001; Shimada et al. 2005; Wu et al. 2008; Gao et al. 2009). Although the role of MT is still unclear, it is believed that MT is involved in the regulation of essential metals such as Zn and detoxification of nonessential metal ions such as Cd, Pb, and Hg (Chan 1995; Cheung, Lam, and Chan 2004). Studies have shown that heavy metals can induce the MT gene and protein in several fish species and may reduce potential toxicity of heavy metal residues. Therefore, MT may be used as a biomarker for heavy metal contamination in polluted waters (Quirós et al. 2007).
Information on Hg toxicology in various fish species has been reported (WHO 1991a, 1991b). However, there is limited information in Hg toxicity in tilapia. Therefore, the purpose of this study was to describe the acute pathological effects caused by exposure of tilapia to Hg via different routes, and to determine the protective role of MT, during the acute exposure stage. Also, the distribution and accumulation of Hg in various tissues of tilapia were investigated by histochemical AMG and ICP-AES.
Materials and Methods
Experimental Animal
Adult tilapias (O. niloticus), 25 to 50 g in weight and 9 to 12 cm in length, were purchased from Veterinary Medical Aquatic Research Center (VMARC), Chulalongkorn University. Each group of 25 was maintained in 62.5-L glass aquaria filled with 50-L tap water. Fish were acclimated for more than 1 week prior to beginning the experiment. Air pump with aquarium foam sponge filter was used for the aeration system. Water temperature, pH, and dissolved oxygen (DO) were measured daily during the experiment. Average temperature was 27.14 ± 0.39°C, pH was 7.30 ± 0.11, and DO was 9.56 ± 0.60 mg/l. All fish were fed twice daily at 3 to 5% body weight with commercially food throughout the experimental period. Tap water in the aquaria was changed every 2 days. All procedures were approved by the ethics committee of Chulalongkorn University Animal Care and Use Committee.
Experimental Design
The tilapias were randomly divided into 1 control and 12 exposure groups of 25 animals each. A total of 0.5 ml of 0.5, 1, 2, or 5 µg/g (ppm) of mercuric chloride (HgCl2, Sigma-Aldrich, USA) was intraperitoneally (IP) injected once to fish of 4 exposure groups (Stewart et al. 2011). Another four groups were orally intubated once with 0.5 ml of 0.5, 1, 2, and 5 µg/g of HgCl2, respectively (Tashjian and Hung 2005). The rest, which were the semistatic exposure groups, were kept in aquaria that contained 0.5, 1, 2, or 5 ppm HgCl2, respectively (OECD No. 23 2000). One aquarium of 25 tilapias was kept as an unexposed control group in the same condition. Animals in each group were sacrificed at 0, 3, 6, 9, 12, or 15 days of exposure.
Sample Collections
The fish were anesthetized by rapid cooling and euthanized by decapitation. Kidney, liver, spleen, gill, gut (stomach and intestine), brain, and muscle were collected and preserved in 10% neutral buffered formalin. The tissues were paraffin-embedded by accepted, routine methods. After paraffin embedding, 4 µm sections were cut and stained with H&E (Kienan 2008) for microscopic evaluation, AMG staining, and immunohistochemistry. The fresh tissues of gill, abdominal organs, and muscle were collected at 3, 9, and 15 days postexposure and kept in −80°C for ICP-AES process.
AMG
Tissue sections from various organs were evaluated for the presence of intracellular Hg deposition. After deparaffinization, other metal residues were eliminated by incubating the sections with 1% potassium cyanide for 2 hr and rinsed well with tap water and distilled water (DW). The sections were incubated with physical developer (50% Arabic gum, 50% citrate buffer, 5.6% hydroquinone, and 17% AgNO3) to silver amplification for 1 hr in automatic shaker at 26°C and then with 10% sodium thiosulfate and Farmer’s solution (20% sodium thiosulfate and 7.5% potassium ferric cyanide) to eliminate silver residues. Thereafter, the sections were rinsed in tap water and counterstained with Mayer’s hematoxylin. The positive reactions resulted in yellow-brown to black silver grains in the cells. The sections were observed by light microscopy to identify cell types and locations of silver grains. Positive staining and intensity of silver grains in each cell were scored (Shimada et al. 2005).
Immunohistochemistry
Immunohistochemistry was performed to detect MT protein, using monoclonal mouse MT antibody (MT1; E9, Dako®, Denmark) as the primary antibody with EnVision™ detection system (Dako®, Denmark). After deparaffinization, the sections were treated with citrate buffered pH 6.0 for 20 min at 121°C by autoclave and microwave heat at 700 W for 5 min for antigen retrieval process. The sections were incubated with 3% H2O2 in methanol to quench endogenous peroxidase for 30 min at room temperature and then with 1% BSA for 30 min at 37°C to inhibit nonspecific reactions. Thereafter, the sections were reacted overnight at 4°C with mouse antihorse MT monoclonal antibody diluted 1:50. The biotinylated antimouse IgG antibody and EnVision polymer (Dako REAL™ EnVision™ detection system) reacted to sections as a secondary antibody at room temperature for 45 min. The positive reactions resulted in brown staining with the substrate 3,3′-diaminobenzidine tetrahydrochloride, and the sections were counterstained with Mayer’s hematoxylin for 30 sec. Human ductal carcinoma section that gave intense cytoplasmic staining was used as positive control (Shimada et al. 2005).
ICP-AES
After lyophilization, the tissue samples were weighed and ground. The dry samples were digested with 10 ml of nitric acid:hydrochloric acid (10:1) overnight and evaporated at 60°C until reaching 1 ml of solution. The samples were cooled to room temperature and diluted with 9 ml of DW. Hg levels were measured with ICP-AES (Perkin Elmer model PLASMA-100). The Hg levels are expressed in parts per million on a dry weight basis (ppm/mg; Subramanian 1996).
Statistical Analysis
Statistical analysis for total Hg levels in fish tissues measured by ICP-AES was analyzed using one-way analysis of variance (ANOVA) and followed by Newman–Keuls multiple comparison test (p < .05).
Results
Clinical and Macroscopic Findings
Among oral-treated animals, there were no exposure-related clinical signs at any observation time. Tilapias in the 2 and 5 ppm semistatic exposure groups showed respiratory distress characterized by swimming to the surface, rapid opercular movements, ataxia, incoordination, dyspnea, and death after a few hours of exposure. Some fish in the 2 and 5 ppm IP groups showed enlarged abdomen after 3 days exposure. Fish with enlarged abdomen had severe purulent peritonitis with adhesions of visceral organs.
Microscopic Findings
Various organs including trunk of the kidney, liver, spleen, and gastrointestinal tract had remarkable lesions; Table 1 compares the degree of severity of these lesions between groups.
Summary of severity of major histopathological lesions in early stage.

Note. − No remarkable lesions, + mild, ++ moderate, and +++ severe histopathological lesions.
Trunk of the kidney from the control fish showed normal renal corpuscles characterized by vascular capillary network of glomerulus surrounded by Bowman’s capsule, and tubules that have basal nuclei and fine vacuolated columnar epithelium with interstitial capillaries (Figure 1A). However, treated animals had hydropic degeneration and massive necrosis of the tubular epithelium with hyaline droplets (Figure 1B) and pleomorphic crystal deposition. The most severe lesions were observed in the semistatic exposure groups followed by the IP and oral administration groups, respectively. Severe degeneration and necrosis of the renal tubular epithelium in early exposure stages of the semistatic exposure groups gradually declined to a milder severity at the end of the experiment. The severity of the lesions was not remarkably different among exposure concentrations. In the IP and oral administration groups, there were no differences in severity of the lesions among concentration throughout the experiment. Mild-to-moderate numbers of immature nephrons and regeneration of renal tubule epithelium were found in the semistatic and the IP exposure groups. In the semistatic groups, mild lesions were observed throughout the experiment, but in the IP exposure groups the lesions were found from day 6 until the end of experiment.
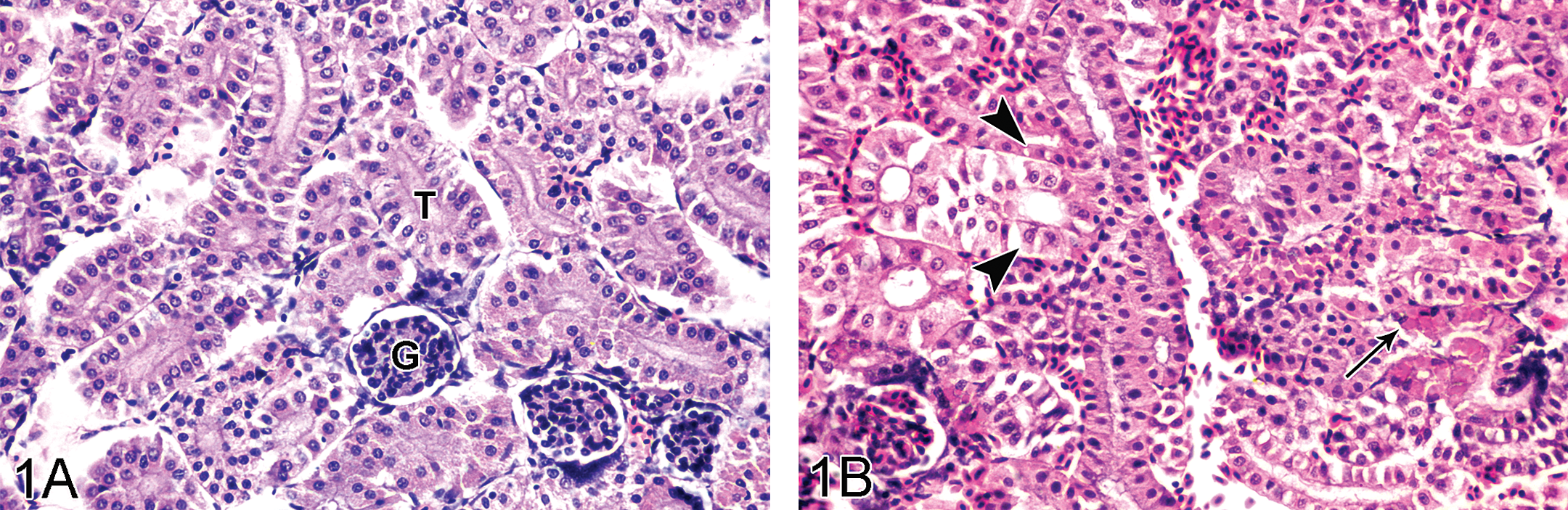
A: Trunk kidney of control tilapia showing normal renal corpuscles characterized by vascular capillary network of glomerulus (G) and renal tubules (T) that have basal nuclei and tiny vacuolated columnar epithelium, H&E stain, ×390. B: Severe tubulonephrosis (arrowheads) with hyaline droplets (arrow) was observed in 2 ppm semistatic exposure tilapia on day 3, H&E stain, ×390.
The liver of tilapias treated with Hg showed hydropic degeneration of the hepatocytes and xenobiotic-induced megalocytotic hepatocytes. Fewer hepatocytes with lipid vacuoles were observed in the exposure groups (Figure 2B) compared to the controls, in which cytoplasmic lipid vacuolation was very prominent (Figure 2A). In the semistatic and IP exposure groups, fat loss in hepatocytes was severe from day 3 until the end of experiment. Hyaline droplets accumulations in the hepatocytes were also found on day 12 of the semistatic exposure groups.
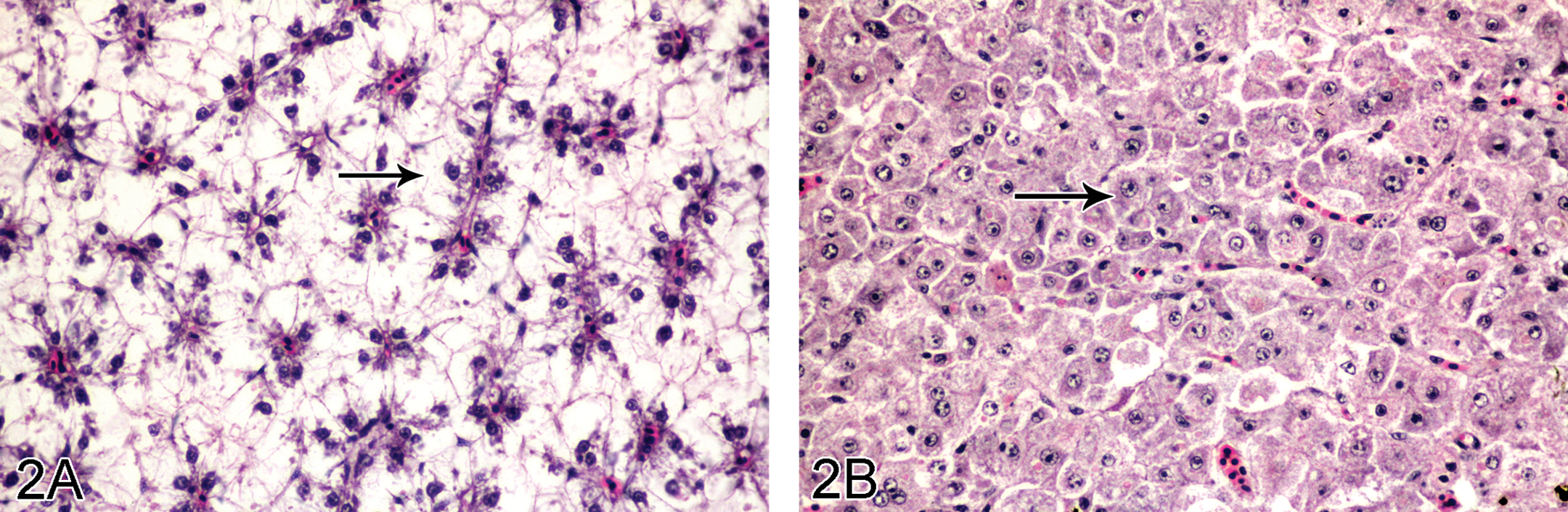
A: Hepatocytes of control tilapia with high fat content in the cytoplasm (arrows). Arrowheads: hepatic nuclei, H&E stain, ×390. B: Severe loss of lipid in hepatocytes characterized by low-fat vacuolation in the cytoplasm (arrows) was found in 1 ppm semistatic exposure tilapia on day 15. Arrowheads: hepatic nuclei. H&E stain, ×390.
Pancreatic atrophy was characterized by decreased number of acini and appeared as remnants around hepatic blood vessels. This was found in all the experimental groups and was of mild-to-moderate severity in the IP and semistatic exposure groups throughout the experiment.
Density of melanomacrophage centers (MMCs) in spleen of the control groups was generally low (Figure 3A). Increase in the number of MMCs in spleen was obviously observed in the experimental groups compared to the controls (Figure 3B). Moderate-to-severe lesions were observed from day 1 in the semistatic and day 3 in the IP and oral exposure groups until the end of the experiment. The reduction of lymphocytes in the spleen (lymphoid depletion) was also found in high dose of the semistatic exposure groups.
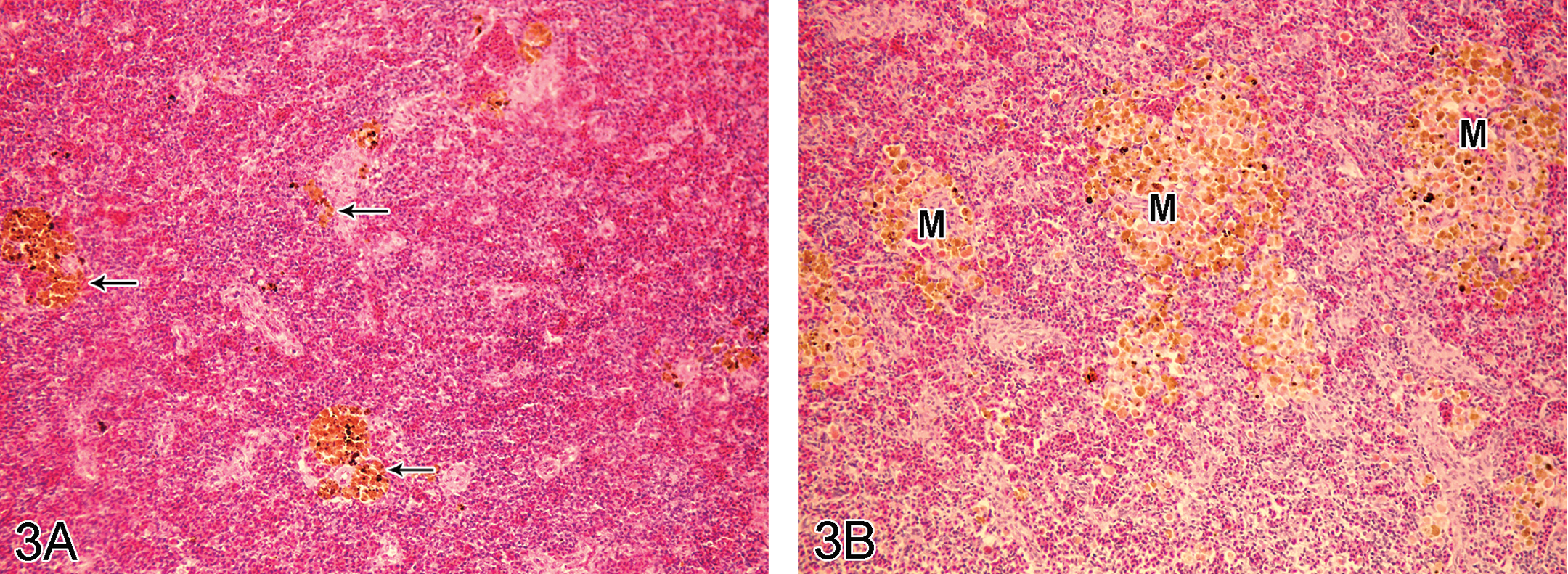
A: Spleen of control tilapia with low density of melanomacrophage centers (arrows), H&E stain, ×190. B: Melanomacrophage center (M) aggregation was observed in spleen of 0.5 ppm semistatic exposure tilapia on day 12, H&E stain, ×190.
Mild-to-moderate diffuse goblet cell proliferation and mononuclear cell infiltration in intestinal lamina propria and submucosa were found in the oral and semistatic exposure groups but were more severe in the semistatic groups. Necrosis of gastric epithelium with mononuclear cells infiltration in lamina propria of the high dose, 2 ppm, oral group after 3 days postexposure was also found.
There were no remarkable lesions in gills, brain, and skeletal muscle that were attributed to Hg exposure by any route/concentration.
AMG
Hg deposition and yellow-brown to black positive silver grains were found in the cytoplasm of the renal tubule epithelium (Figure 4), pancreatic acini (Figure 5), MMCs of the spleen (Figure 6), and intestinal epithelium (Figure 7) of the experimental groups. Degrees and distribution of Hg positive cells were different between the organs and days after exposure compared to the controls (Table 2). After 3 days postexposure, positive silver grains were observed mainly in the IP and semistatic exposure groups, but this was minimal in the oral exposure groups. The largest number of positive cells appeared in the 5 ppm Hg exposure groups, followed by 2, 1, and 0.5 ppm, respectively. By contrast, no evidence of positive silver grains was found in gill, brain, and skeletal muscle of the exposure groups, as well as in any tissue of fish from the control group.
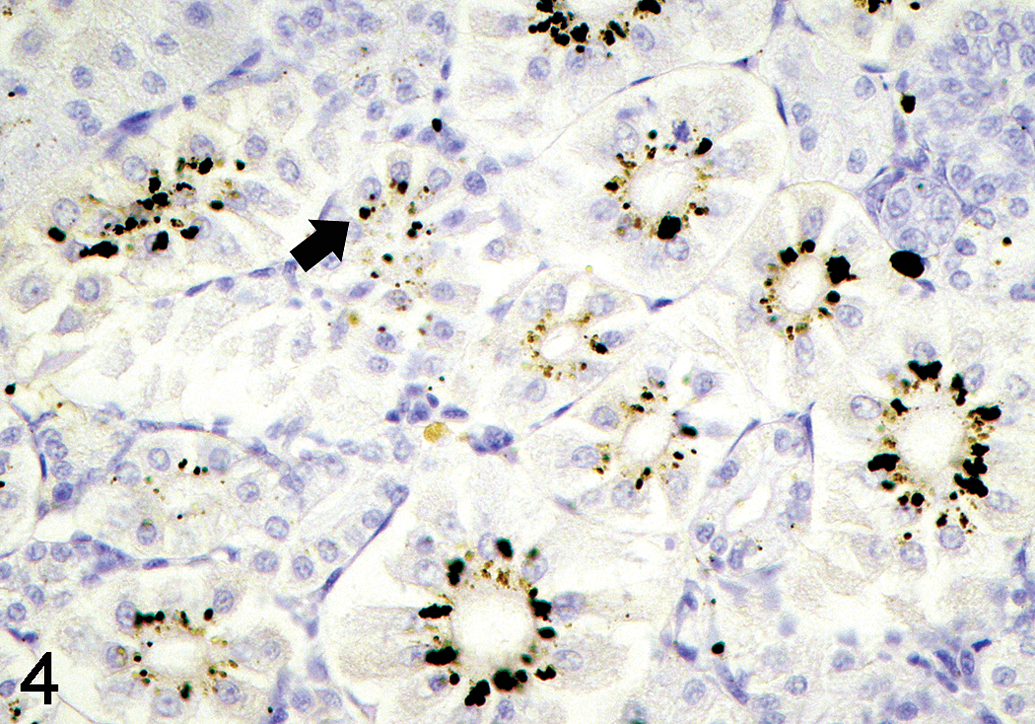
Silver grains (solid arrow) deposit in the renal tubular epithelial cells of 1 ppm semistatic exposure tilapia on day 15, autometallography (AMG) stain, ×420.
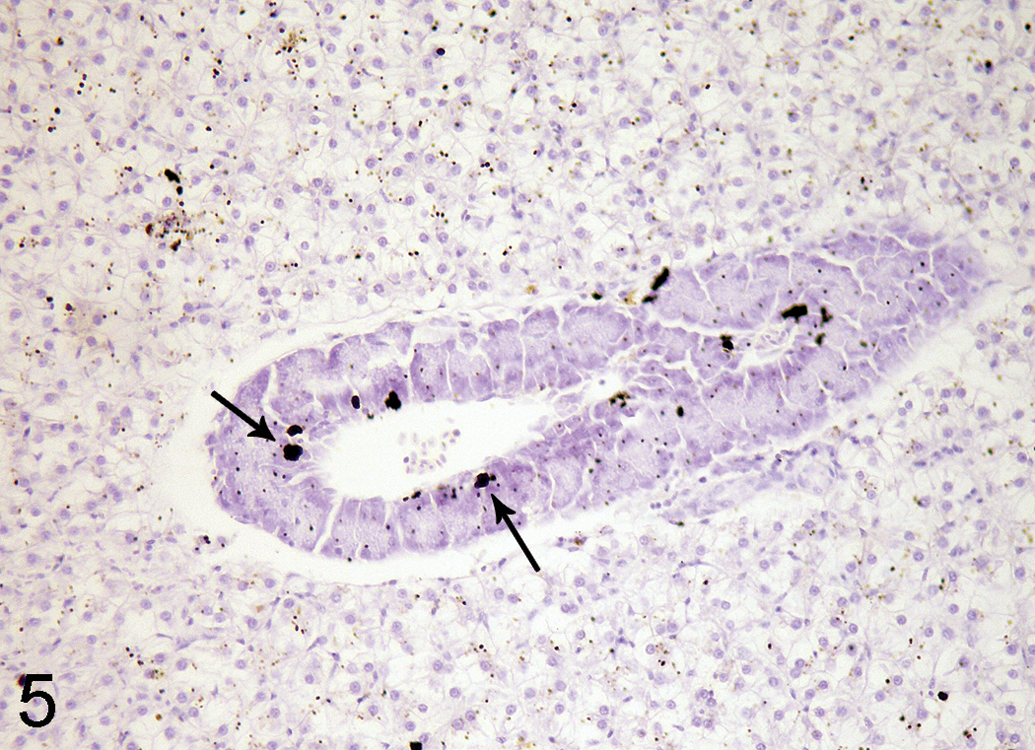
Silver grains (arrows) deposit in the pancreatic acinar cells in hepatopancreas of 0.5 ppm semistatic exposure tilapia on day 3, autometallography (AMG) stain, ×190.
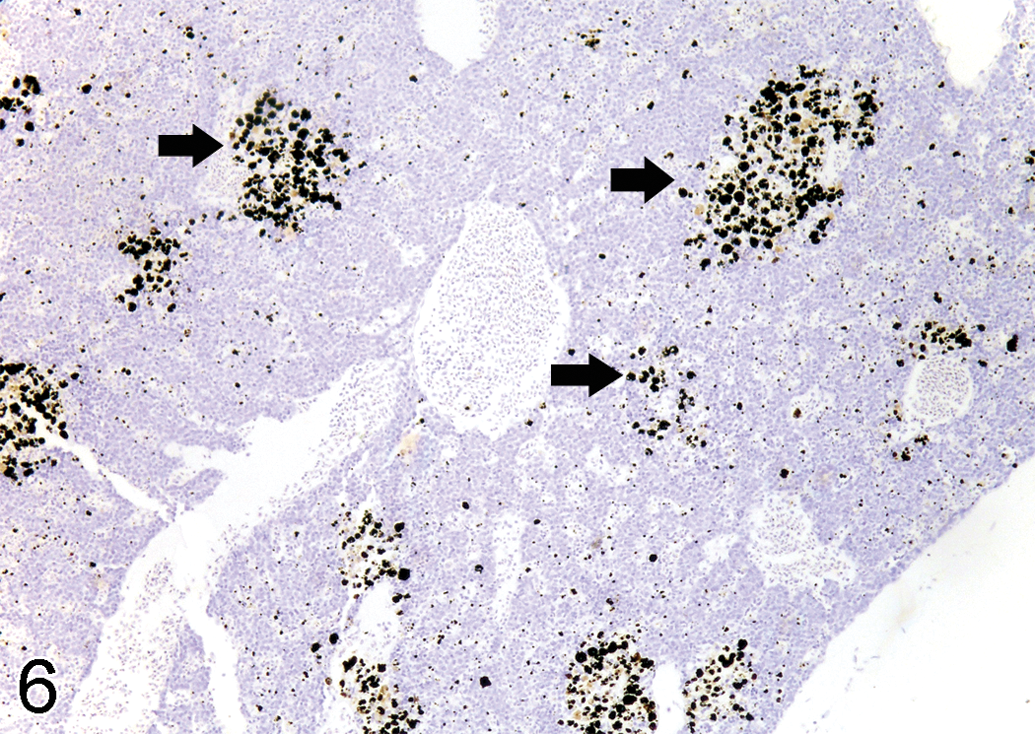
Silver grains (solid arrows) deposit in the melanomacrophage centers in spleen of 1 ppm semistatic exposure tilapia on day 15, autometallography (AMG) stain, ×190.
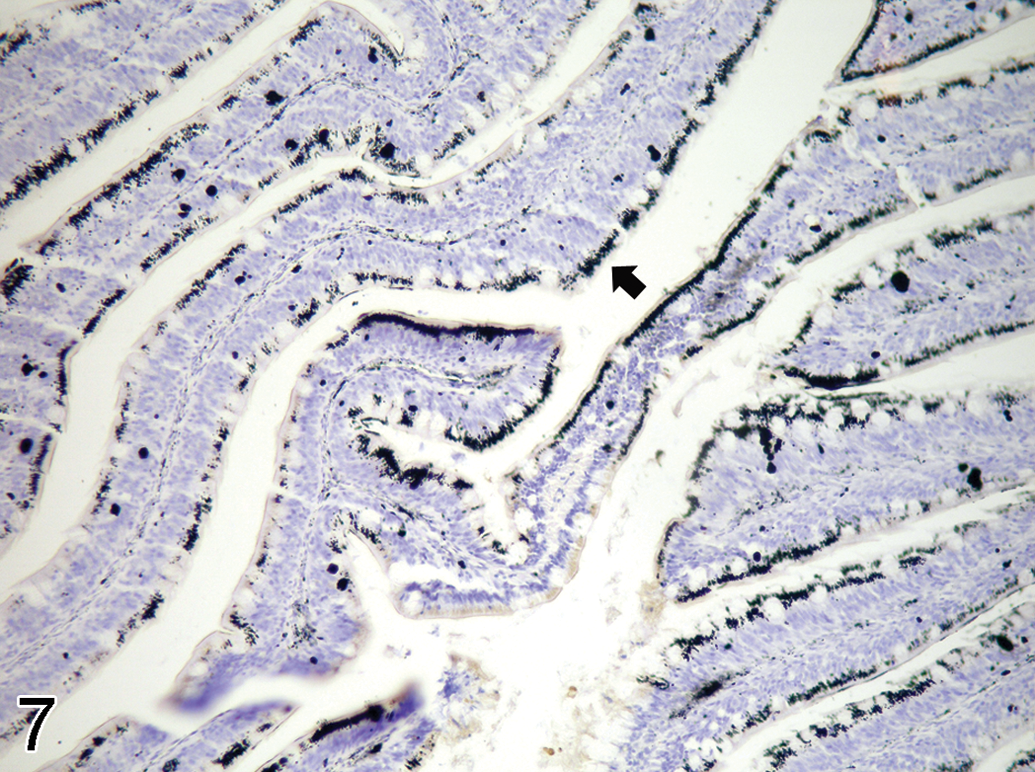
Silver grains (solid arrow) deposit in the intestinal epithelial cells of 1 ppm semistatic exposure tilapia on day 15, autometallography (AMG) stain, ×290.
Summary of intensity of silver grains in tissues by autometallography.
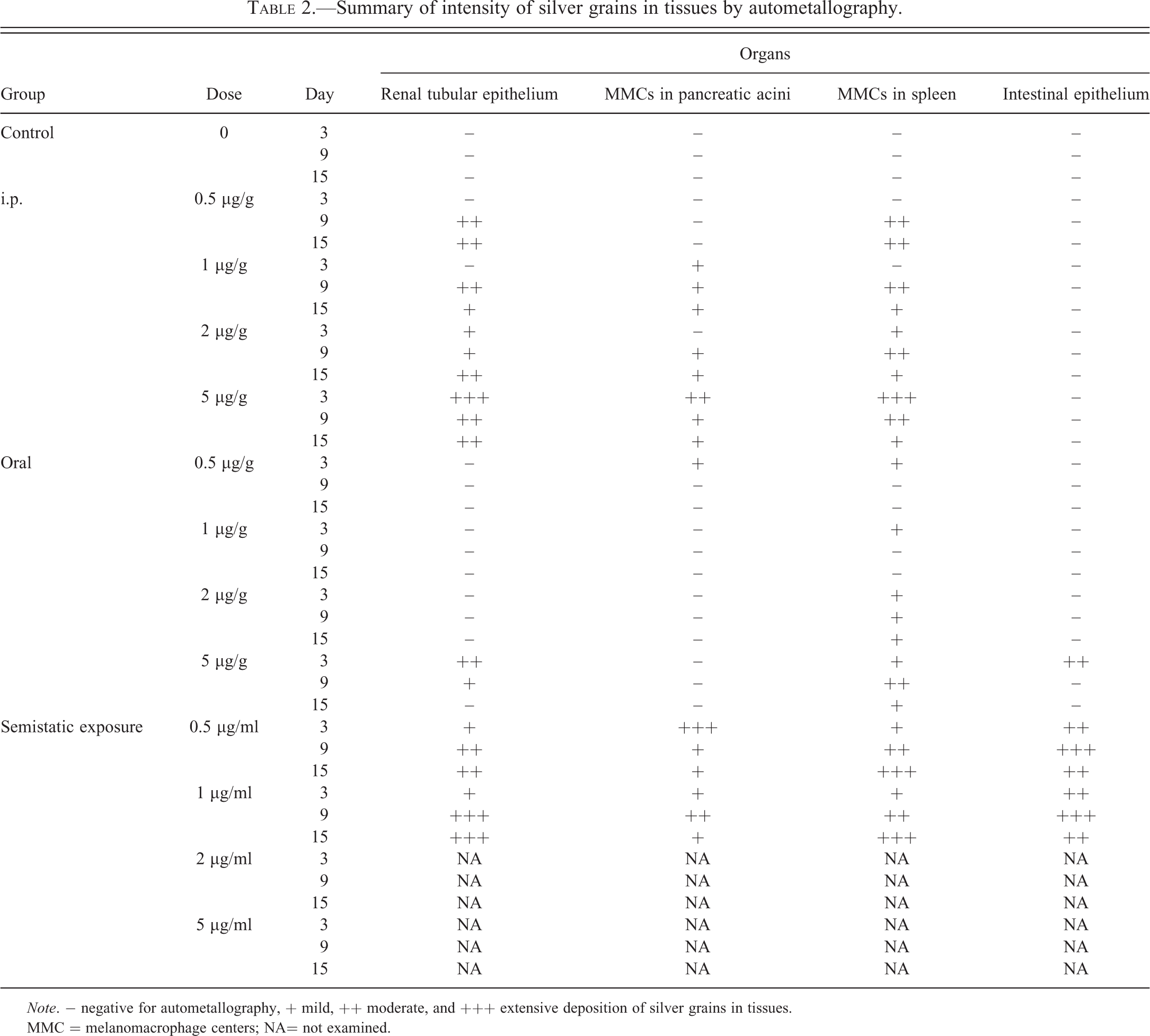
Note. − negative for autometallography, + mild, ++ moderate, and +++ extensive deposition of silver grains in tissues.
MMC = melanomacrophage centers; NA= not examined.
MT Expression
Intense intracytoplasmic MT protein expression was observed in the 0.5 and 1 ppm semistatic exposure groups. However, MT protein expression of the 1 ppm groups was more intense than the 0.5 ppm groups. In the 0.5 ppm semistatic exposure group, the MT protein expression was detected in cytoplasm of renal tubular epithelium (Figure 8) after 9 days exposure, proliferative splenic MMCs, and atrophic pancreatic acinar cells at 15 days exposure. In the 1 ppm semistatic exposure groups, the positive stains were observed in trunk kidney after day 6 and proliferative splenic MMCs at day 15. In the control group and the other exposure groups, MT protein was not detectable in any tissue samples.
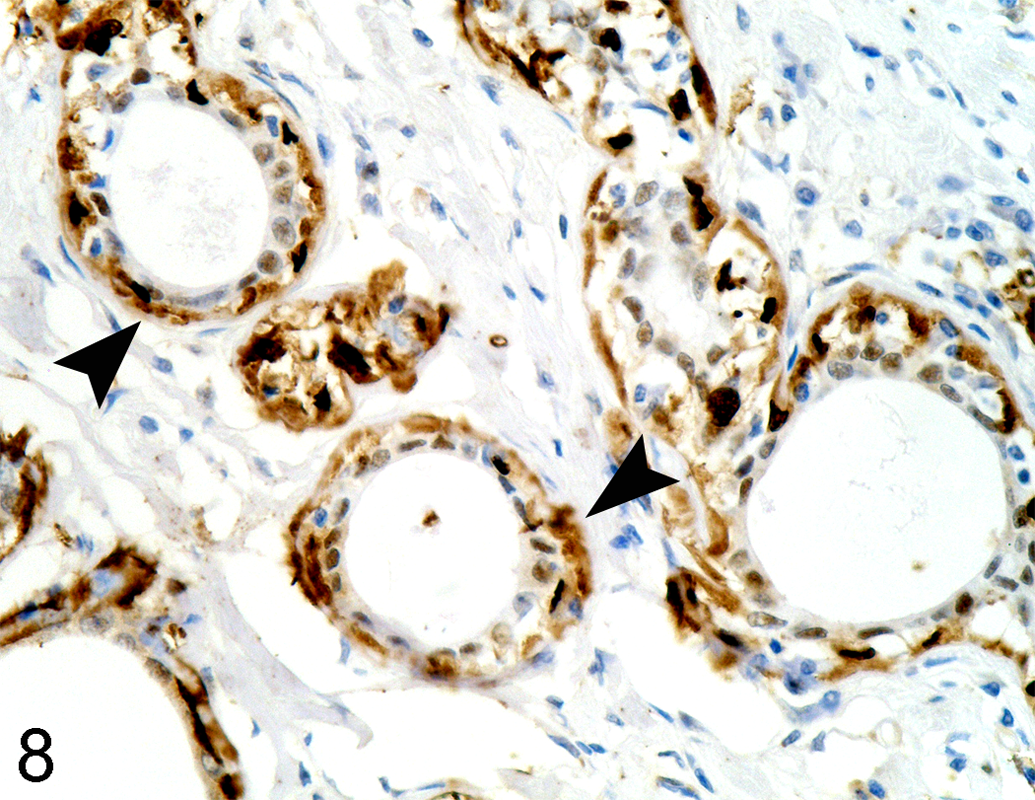
0.5 ppm HgCl2 semistatic exposure tilapia on day 15 showed MT expression (solid arrowheads) in renal tubular epithelial cells, MT immunohistochemistry, ×390.
ICP-AES
Table 3 presents total Hg concentrations of the gills, abdominal visceral organs, and skeletal muscle in ppm/g dry weight. The cutoff values of total Hg concentrations of the control group in three major organs throughout the experiment were less than 0.2 ppm/g dry weight. Total Hg concentrations of visceral organs were higher than gills and skeletal muscle. Total Hg concentrations of the exposure groups were higher than the control at any time point. In IP and oral exposure, total Hg concentration gradually decreased until the end of the experiment. However, total Hg concentration of the semistatic exposure groups were almost increased by time until the end of the experiment. The highest total Hg concentration was detected in the semistatic exposure groups, followed by the IP and oral exposure groups, respectively. In IP exposure, all concentrations of the treated groups were significantly different between abdominal organs and other organs at day 3 (p < .05), but were not significantly different at day 9 and day 15. Moreover, there were statistically significant differences of total Hg concentrations in abdominal organs among day 3, 9, and 15 while day 3 groups had the highest concentration and gradually decreased by time. In oral exposure, the 2 ppm treatment group was significantly different from the other doses. There were significant differences between visceral organs in abdomen and other organs on day 3, day 9, and day 15 (p < .05). When comparing between the same organs of 2 ppm exposure groups, abdominal viscera revealed a significant difference in Hg concentration among 3, 9, and 15 days (p < .05). Total Hg concentration of the gills in 2 ppm groups on day 9 was also significantly different from day 3 and day 15 (p < .05). In the semistatic exposure tilapia groups, 1 ppm group had the highest concentration compared to the other exposure groups. Hg concentration of abdominal organs was significantly different from the other organs when compared among the exposure times (p < .05).
Total Hg concentration in various tissues (ppm/g dry weight) of various exposure routes.
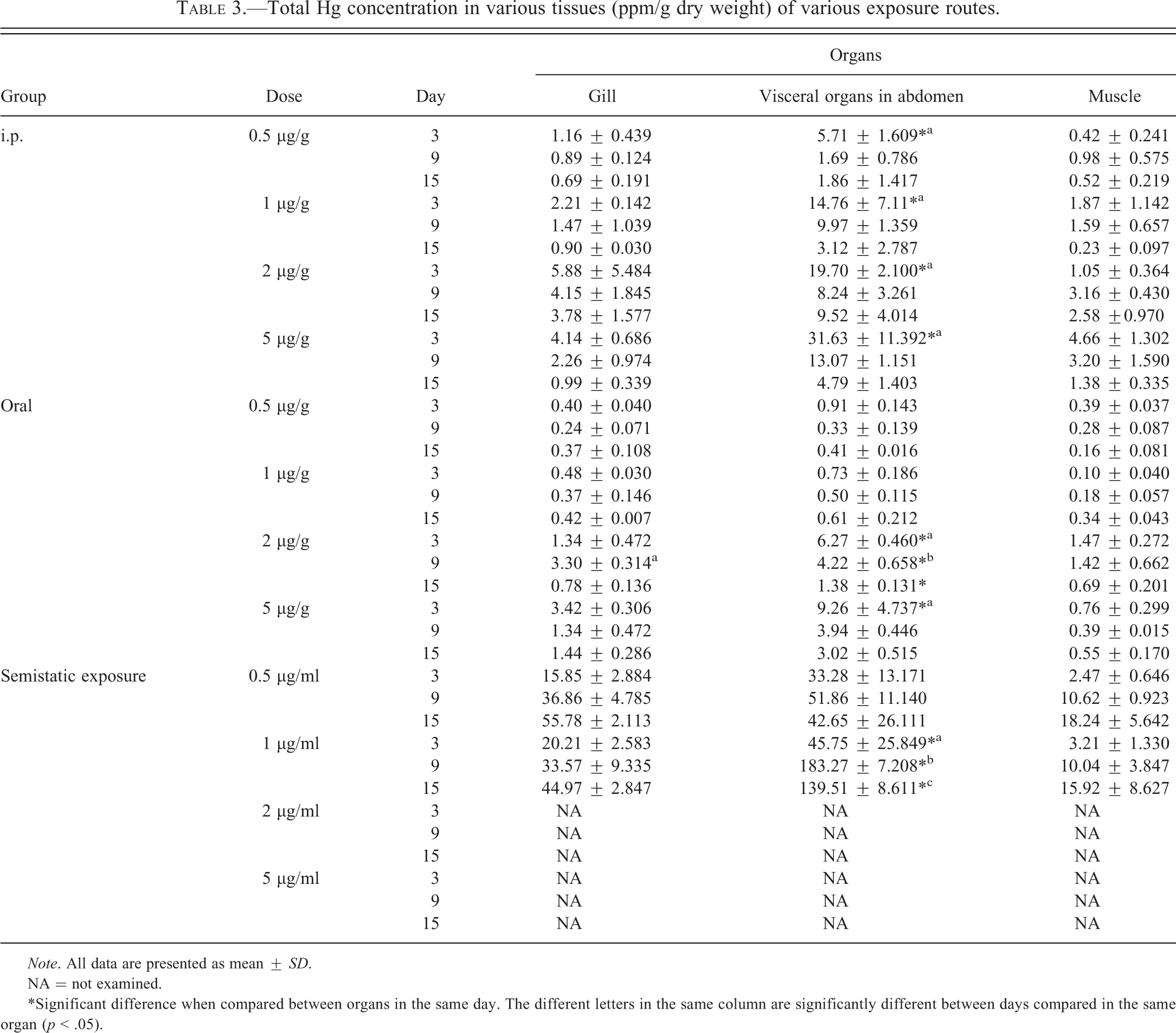
Note. All data are presented as mean ± SD.
NA = not examined.
*Significant difference when compared between organs in the same day. The different letters in the same column are significantly different between days compared in the same organ (p < .05).
Discussion
Mean mortality rate in 2 and 5 ppm (µg/l) HgCl2 semistatic exposure groups was 100% after 2 hr on the first day of the experiment. The value of 24-hr mean lethal concentration (LC50–24 hr) of Hg2+ was estimated at an exposure dose of 2 ppm. The LC50–24 hr was similar to those reported by Charuwan-Somsiri (1982). However, the value was higher than those reported in other studies (Ishikawa, Ranzani-paiva, and Lombardi 2007; Kaoud and Mekawy 2011). The higher values in this study and Charuwan-Somsiri’s report might be related to older and larger size of the test organism. Adult tilapias were used as the test organism in our study and Charuwan-Somsiri’s report (1982) compared with other studies that obtained lower values of LC50 using fingerling as the test organisms (Ishikawa, Ranzani-paiva, and Lombardi 2007; Kaoud and Mekawy 2011). Older and larger aquatic organisms are quite resistant to toxicants (Buhk 1997; Boening 2000). Acute toxic signs including respiratory distress, tremor, ataxia, incoordination of movements, dyspnea, and death were observed obviously in higher Hg concentrations (2 and 5 ppm) and the semistatic exposure groups. Similar toxic signs have been reported in other studies in tilapias (Ishikawa, Ranzani-paiva, and Lombardi 2007; Kaoud and Mekawy 2011) and other fish (Bano and Hasan 1990).
Lesions of acute Hg toxicity in tilapia were observed mainly in the kidney, hepatopancreas, and spleen. Renal tubule degeneration and necrosis, with hyaline droplets, was observed. The effect of Hg could be found after day 1 in high dose of the semistatic exposure route, similar to the study in channel catfish (Ictalurus punctatus; Kendall 1975). Loss of fat storage in degenerative hepatocytes was found mainly in treated tilapias. Loss of fat reserve cells was also reported in arctic charr (Salvelinus alpinus) and Northern pike (Esox lucius) exposed to inorganic Hg (Oliveira-Ribeiro et al. 2002; Drevnick, Sandheinrich, and Oris 2006). In the current study, there was no evidence of hepatic necrosis in the control or Hg exposure groups, unlike another study in arctic charr associated with Methyl Hg exposure, which showed severe necrosis of hepatocytes (Oliveira-Ribeiro et al. 2002). In Hg-exposed bleaks, there were multifocal granulomas, macrovesicular steatosis, nuclear pyknosis of hepatocytes with bile duct proliferation (Raldúa et al. 2007), but no evidence of these lesions was found in this study. Pancreatic atrophy also appeared after being exposed to Hg in this study, similar to the study in catfish (Ictalurus punctatus) exposed to Hg2+ (Kendall 1977). Also, alteration of gastrointestinal organs was observed in treated tilapias in this study. Similar lesions have been described in Liza parsia by Pandey, Mohamed, and George (1994). Increase of MMCs in the spleen was observed in all the experimental groups. The pathogenesis of the increasing of MMCs may involve an increase in phagocytic activity due to tissue damage by Hg and macrophages, which is the first line of protection of Hg intoxification (Loumbourdis and Dancher 2004). Moreover, MMCs contained melanin which is well known for its ability to neutralize free radicals and cations associated with heavy metals (Robert and Ellis 1978; Agius 1985). Therefore, the high density of MMCs in spleen could possibly be used as a potent biomarker for Hg contamination in fish (Mela et al. 2007).
The highest positive autometallographic results were observed in the semistatic exposure groups followed by the IP and oral groups, respectively. The high positive Hg staining identified in affected organs of the semistatic exposure groups was observed without variation in the exposure concentration and time because this exposure experiment was a repeat-exposure model in which tilapia received Hg2+ constantly until the end of experiment. In the IP exposure routes, the results showed intense positivity in renal epithelial cells and MMCs of spleen, but there were no positive silver grains in intestinal mucosa. These results indicated that Hg2+ might directly be absorbed into circulation and distributed into target organs including the kidney and spleen similar to the distribution of Hg in mammals (Zahir et al. 2005). According to previous publications, Hg has great affinity for gill tissue because the gill is a principal route for entering the body (Allen, Min, and Keong 1988; Filenko et al. 1988). No AMG positive silver grains were observed in gill tissues of tilapias exposed to Hg in this study, although the high Hg level was detected by ICP-AES. The AMG positive grains detected in gill have been reported in Cu-exposed turbot (Scophthalmus maximus; Alvarado et al. 2006). Nevertheless, there is no report of AMG staining of the gill of Hg-exposed fish. Danscher and Stoltenberg (2006) suggested that different types of heavy metals might demonstrate different autometallographic patterns in different kind of tissues.
The accumulation of Hg in tissues was confirmed by ICP-AES. The highest Hg levels in affected tissues were found in the semistatic exposure groups followed by abdominal visceral organs and muscle, respectively. In the IP and oral exposure groups, total Hg levels decreased with exposure time, probably since both of these experiments were single time exposure models. In contrast, the semistatic exposure groups, which were multiple repeated exposure experiments, showed the increasing pattern of Hg levels in the tissues with exposure time. When comparing the total Hg levels among the organs, the highest levels were in abdominal visceral organs followed by gill and muscle, respectively. Abdominal visceral organs, including the kidney and liver, have been reported as the major organs for Hg bioaccumulation in several studies (Oliveira-Ribeiro, Guimaräes, and Pffeiffer 1996). Therefore, it is not surprising that abdominal visceral organs were detected with the highest total Hg levels in this study. The high bioaccumulation of Hg in the gill revealed in the present study is similar to other Hg toxicity experiments (Filenko et al. 1988; Allen, Min, and Keong 1988). Muscle was the tissue that accumulated less Hg in tilapia in the present work. Several studies reported that inorganic Hg did not present a great affinity to muscle tissue (WHO 1991b; Oliveira-Ribeiro, Guimaräes, and Pffeiffer 1996). Moreover, organic Hg is the most important form that accumulates in muscle in nature (WHO 2003).
Several laboratory and field studies noted that MT played an important role in heavy metal homeostasis and detoxification in aquatic animals. Expression of MT in tissues has been used as an indicator for environmental monitoring of heavy metal exposure in many aquatic organisms (Chan 1995; Hamilton and Mehrle 1986; Haux & Forlin 1988; Alvarado et al. 2006). In the present study, MT protein expression was observed only in the semistatic exposure groups, but no evidence was detected in the IP and oral experiments. These results suggested that the concentration of Hg in single exposure of the IP and oral groups was possibly not enough to induce the MT protection response in treated tilapias. To use MT expression as the indicator for biological monitoring in fish, the MT induction time is necessary to know how long the animals will respond after heavy metal exposure. The MT induction time has been determined in various fish species exposed to heavy metals. Difference in the species of fish, types of heavy metal, and temperature zone produced difference in time course of MT induction (Baksi, Libbus, and Frazier 1988; Hogstrand and Haux 1990). A significant induction of MT mRNA levels in the liver and gills of heavy metal–exposed tilapia was observed at week 3 (Lam et al. 1998). In the current study, the MT positive expression was detected after day 6 until day 15.
In conclusion, short-term exposure to inorganic Hg can produce lesions of acute Hg toxicity in the kidney, spleen, and hepatopancreas of tilapias. AMG may be used to determine the bioaccumulation of Hg in fish because of its specificity and practicality. MT protein expression using immunohistochemistry could serve as biomarker for protection system of fish related to Hg exposure. According to the different exposure models in the present study, the semistatic exposure might be a suitable route to study Hg toxicology in tilapia because this model demonstrated significant histopathological lesions and bioaccumulation. In addition, this model more closely emulates the heavy meal exposure of fish in natural water sources.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank the staffs from the Department of Veterinary Public Health, Research Center for Bioscience in Animal Production, Veterinary Pathology Unit, Ms. Anunya Srinonate, Mr. Supradit Wangnaitham, and Mr. Sitthichock Lacharoje, and also the VMARC.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Research Council of Thailand in 2007.
