Abstract
Pregnant rats were treated with 0.3 and 0.6 mg cadmium (CdCl2)/kg injected subcutaneously on a daily basis from gestational day 7 to day 15 (organogenesis period). One control group was not injected and other received saline. The 45-day-old offspring were tested in a step-down inhibitory avoidance to evaluate short-term and long-term memory and in a radial maze for the study of spatial memory. These studies showed that gestational exposure to 0.6 mg Cd/kg produced in the male offspring a significant impairment in the retention of long-term memory evaluated 24 hours after training in the step-down inhibitory avoidance. The radial maze also demonstrated that the male offspring prenatally exposed to 0.6 mg Cd presented a significant deficit in the retention of spatial memory evaluated 42 days after training. These results demonstrate that the exposure to Cd during organogenesis may affect the retention of some types of memory.
Introduction
Cadmium (Cd) is a toxic heavy metal widely distributed in the environment. The industrial uses of Cd have been identified as a major cause of widespread dispersion of the metal. Due to this, Cd has been found to contaminate food, air, and water. 1 -3 Since tobacco plants accumulate Cd from soils, cigarette smoke is another significant source of Cd exposure. 4,5 The extremely long biological half-life of Cd essentially makes it a cumulative toxin. Several clinical and experimental studies have demonstrated that acute and chronic Cd exposure produced development toxicity such as impairment of fetal growth, defects in the development of different organs, and fetal death. 3,6,7 This metal may cross the placental barrier and may directly affect fetal development. 8,9
It is well known that Cd is also neurotoxic. Cadmium produces neurotoxicity with a complex pathology that includes neurological dysfunction 10 ; behavioral alterations 11 ; histopathological damage with extensive hemorrhagic lesions, destruction of fibers, edema, and pyknotic cells 12 ; and changes in neurochemistry. 13 Cadmium exposure in humans produces central nervous system (CNS) alterations such as headache, fatigue, sleep disturbances, tremor, loss of appetite and weight, and memory deficits. 14,15 In a study using cell culture of human neuroblasts, high concentration of Cd inhibited the growth and the viability of neuroblasts inducing morphological and cytoskeletal alterations as well as apoptotic cell death. 16 In cortical neurons obtained from fetal rats, Cd also induced either apoptosis or necrosis depending on the Cd concentration. 17,18 In juvenile mice, Cd exposure by drinking water for 10 days produced histopathology alterations of cerebral cortex in a dose-dependent manner. 19
Experimental studies also indicate that neonatal exposure to Cd leads to potent neurotoxicity. 20,21 The intraventricular administration of Cd in newborn rats produced greater concentrations of Cd in the brain causing not only morphological but also functional damage. 22 The acute subcutaneus administration of Cd to rats on postnatal day (PD) 5 caused a decrease in the weight of the whole brain and cerebellum, loss of neurons, and reactive gliosis. 23
Although the severity of prenatal exposure to high doses of Cd (intravenous injection of 1.25-1.35 mg/kg) has been widely studied, 24,25 there is little information on the teratogenic effects of prenatal exposure to low levels of Cd. Part of this evidence shows that prenatal exposure to Cd causes long-lasting behavioral and pharmacological alterations in the offspring at doses (3.5-14 mg/kg in water and 0.6-2.0 mg/kg injected subcutaneously) that have no overt toxic effects on the dams. 26 -28 In these studies, the authors observed delay in the development of some reflex, altered motor activity, impaired memory processes, and alterations in several behaviors in offspring prenatally exposed to Cd.
In view of the above, the purpose of the present work was to study the effects of prenatal exposure to Cd on the CNS functionality. The doses used, 0.3 and 0.6 mg Cd/kg, were smaller than those that would cause maternal toxicity and observable adverse effects in the embryos. 29,30 Offspring exposed with these doses during organogenesis were examined in their memory performance. In the 45-day-old offspring, short-term memory (STM) and long-term memory (LTM) were evaluated using a step-down inhibitory avoidance task, and spatial memory was also determined using a radial maze.
Materials and Methods
Material
Cadmium chloride (CdCl2·2.5H2O; molecular weight 228.34; purity 99.5%) was supplied by J. T. Baker Fisher Scientific. USA.
Animals
Parent animals were male and nulliparous female Wistar rats (90-120 days of age) from our own breeding center. They were maintained under constant temperature (22°C ± 1°C) and humidity (50%-60%) conditions in a 12-hour light/12-hour dark cycle (lights on at 7:00 hours) and with food and water ad libitum. In the evening of the proestrus day, they were housed overnight with male rats. The presence of spermatozoa in the vaginal smears was registered as an index of pregnancy and was referred to as gestational day 0 (GD 0). Females were then weighed and housed with other pregnant females until GD 7 when they were randomly assigned to one of the 4 following groups: control group (CG, n = 12), saline group (SG, n = 14), Cd-treated group with 0.3 mg/kg (Cd 0.3, n = 13), and Cd-treated group with 0.6 mg/kg (Cd 0.6, n = 12). From GD 7 to GD 15, experimental groups were daily injected subcutaneously with 0.3 or 0.6 mg Cd (CdCl2)/kg, the SG received an identical volume of saline solution, and the CG received no injections. Maternal weight gain during different periods was registered.
Within 24 hours after delivery, all pups were weighed and litters were randomly culled to 5 males and 5 females whenever possible, and the following data were evaluated: length of gestation, litter size, and body weight of pups at PD 1, 4, 7, 14, and 21. On PD 21, offspring were weaned and housed in groups of 6 rats according to sex and treatment. At the 45 days of age, 1 male and 1 female from each litter were used for the same memory task.
The care and the handling of the animals were in accordance with the internationally accepted standard Guide for the Care and Use of Laboratory Animals as adopted and promulgated by the National Institute of Health. 31 Experimental designs were also approved by the local standard for protecting animal’s welfare, Institutional Committee for the Care and Use of Experimental Animals (Protocol Number 003/2014, 004/2015 and 005/2014).
Step-Down Inhibitory Avoidance
The inhibitory avoidance apparatus was a box with a floor consisting of parallel nonrusting steel bars. A 2.5-cm high platform was placed on the left end of the box. Latency of the rats to step down placing the 4 paws on the grid was measured. In the training session, the animals were gently placed on the platform and received a 0.6 mA, 2-second scrambled foot shock immediately after they stepped down placing their 4 paws on the grid. Test sessions were carried out 1.5 (STM) and 24 hours (LTM) after training. They were exactly like the training session, except that the foot shock was omitted. A 180-second ceiling was imposed on test session latency measurements. In the test sessions, step-down latency was used as measure of retention. 32
Radial Maze
In this experiment, an 8-arm radial maze with an octagonal center platform (diameter: 40 cm) was used. Each of the 8 arms was 60-cm long and 9-cm wide. The maze was elevated 90 cm above the floor with good visibility of room cues (posters, chairs, etc). During the training and testing sessions, the same 3 arms selected at random were maintained with food (corn flakes). All rats were submitted to food restriction, and training started when the rats reached 85% of their weight. In individual trials, each animal explored the maze until it found the food in the 3 arms, and 2 types of errors were scored: (1) reference memory errors, when the rat entered a nonrewarded arm, and (2) working memory errors, when a rewarded arm was revisited. The animals were trained for 15 days (2 trials/day) and tested again 7 (R1), 21 (R2), and 42 (R3) days after the last day of training to evaluate the retention of spatial memory. 33,34
Statistics
The data about the dams and their litters were analyzed by 1-way analysis of variance (ANOVA). The step-down inhibitory avoidance task required nonparametric statistics. Data from each sex were considered separately to detect sex-dependent differences. Each session (training, STM, and LTM) was analyzed by Kruskal-Wallis test followed by Mann-Whitney U test. Data from radial maze task were analyzed by 3-way ANOVA (group × sex × trial) with trials (training and testing sessions) as a repeated measure, and comparisons between groups in each trial were realized using the t tests for independent samples. Differences were considered significant when P < 0.05.
Results
In agreement with our previous study, 28 there were no statistical differences in body weight between the groups of dams on GD 0, in weight gain during the 4 different periods registered, in gestational length, or in litter size. Also, Cd treatment during pregnancy did not affect the body weight of pups at PD 1, 4, 7, 14, and 21 (data not shown). No visible teratogenic malformation was observed in any of the groups.
In the first memory evaluation, the step-down inhibitory avoidance test showed that the prenatal Cd exposure did not affect STM because no significant differences between groups were observed in the latencies measured in the test sessions in both female and male offspring (Figures 1 and 2). However, when LTM was analyzed, the gestational exposure to 0.6 mg Cd produced in the male offspring a significant impairment in the retention of this type of memory (Figure 2). The latencies of these rats to step down on the grid were lower to those of the other groups (P < 0.05) when measured 24 hours after training. The female offspring prenatally treated with Cd did not show alterations in this type of memory; the latencies measured in the test sessions were similar in all groups.
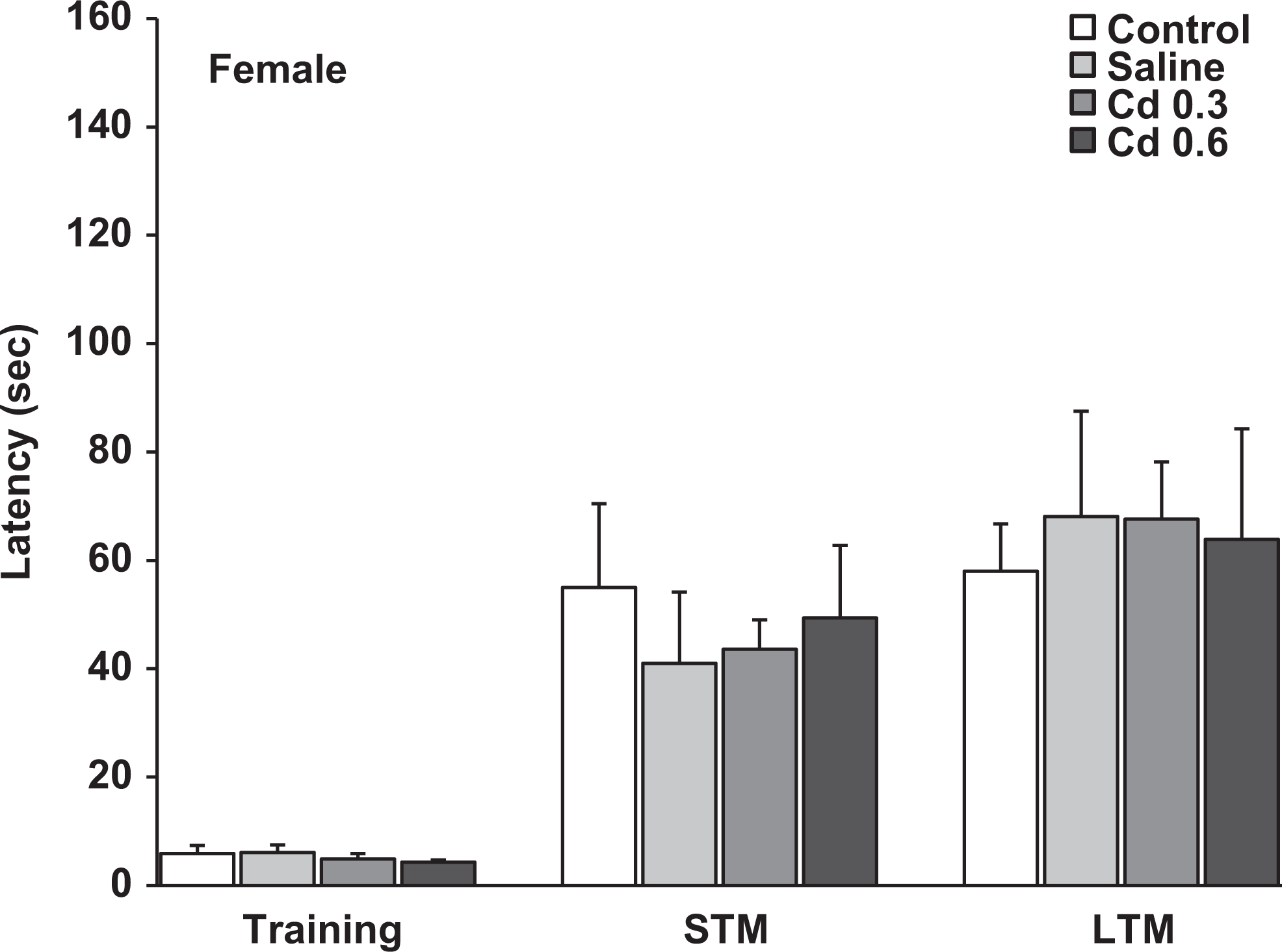
Latencies measured in the step-down inhibitory avoidance during the training session and the evaluation of short-term memory (1.5 hours after training) and long-term memory (24 hours after training) in the female offspring of the different groups. Data represent the mean ± standard error of the mean (SEM) latency in seconds of 9 to 10 animals per group.
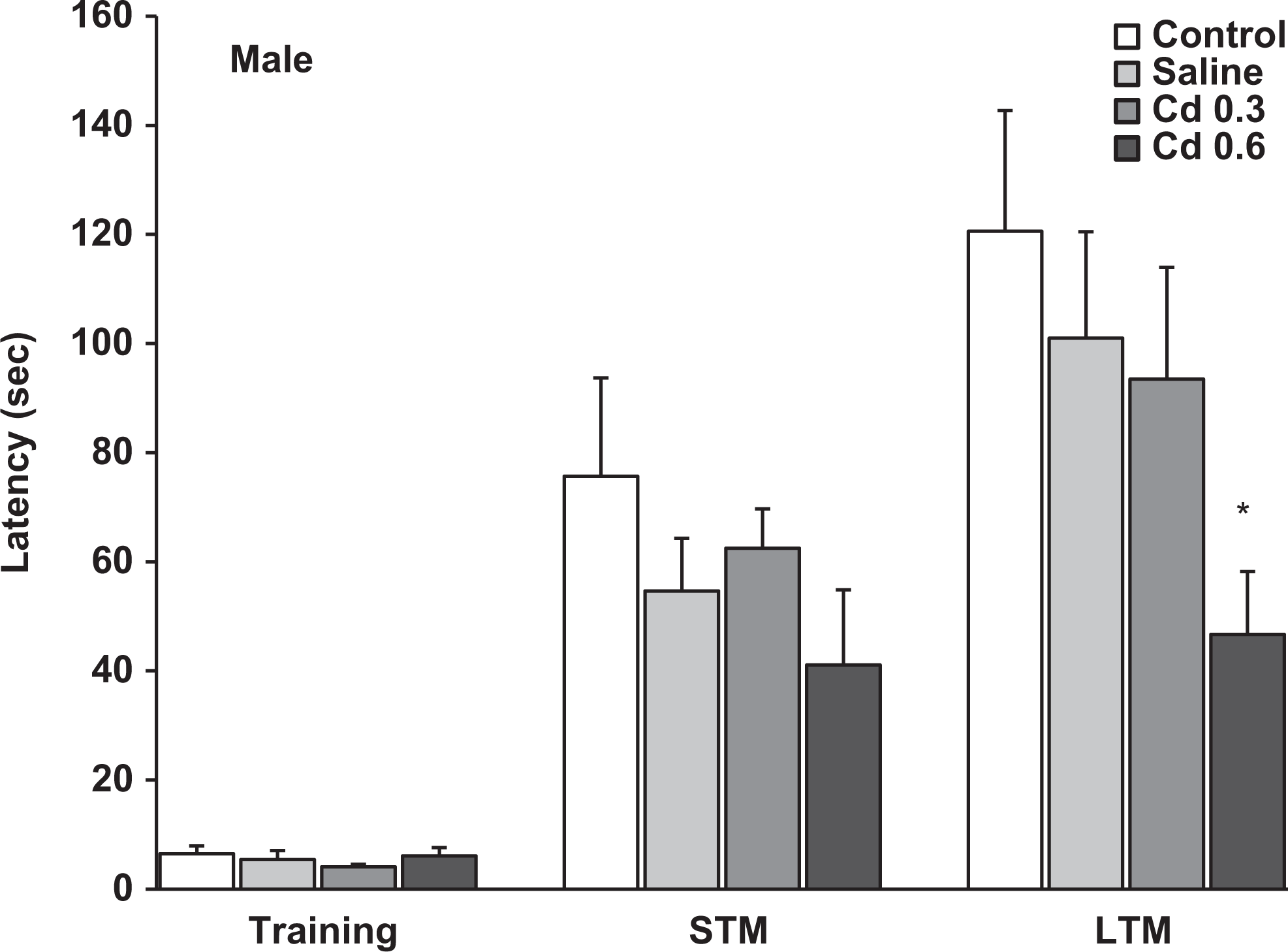
Latencies measured in the step-down inhibitory avoidance during the training session and the evaluation of short-term memory (1.5 hours after training) and long-term memory (24 hours after training) in the male offspring of the different groups. Data represent the mean ± standard error of the mean (SEM) latency in seconds of 9 to 10 animals per group. *P < 0.05, compared to control, saline, and Cd 0.3 groups.
The spatial memory evaluated in the radial maze was analyzed using 3-way ANOVA (group × sex × trial) with trials as a repeated measure. The analysis revealed a significant effect of group, F 3, 69 = 4.6, P < 0.005, and a repeated-measures trial effect, F 17, 1,173 = 128.4, P < 0.001, in the reference memory errors. In the working memory errors, the ANOVA also showed a significant effect of group, F 3, 69 = 3.7, P < 0.02, and a repeated-measures trial effect, F 17, 1,173 = 8.6, P < 0.001. There was no sex and interaction between group effects in both reference and working memory errors. Figures 3 and 4 show the reference memory and the working memory errors, respectively, during training sessions in the male offspring. As can be seen, a similar performance was observed in all groups on different days of training. However, when we evaluated the retention of memory 7, 21, and 42 days after training, the males prenatally exposed to 0.6 mg Cd presented a significant impairment in the retention of spatial memory 42 days after training. As seen in Figure 3, these rats showed a significant increase in the reference memory errors in R3 (42 days after training) compared to the other groups (P < 0.05). Figure 4 shows that males exposed to 0.6 mg Cd also committed significantly more working memory errors than the control and saline groups in R3 (P < 0.02). The evaluation of spatial memory in the female offspring evidenced no alterations in either training or testing sessions in the radial maze task (Figures 5 and 6).
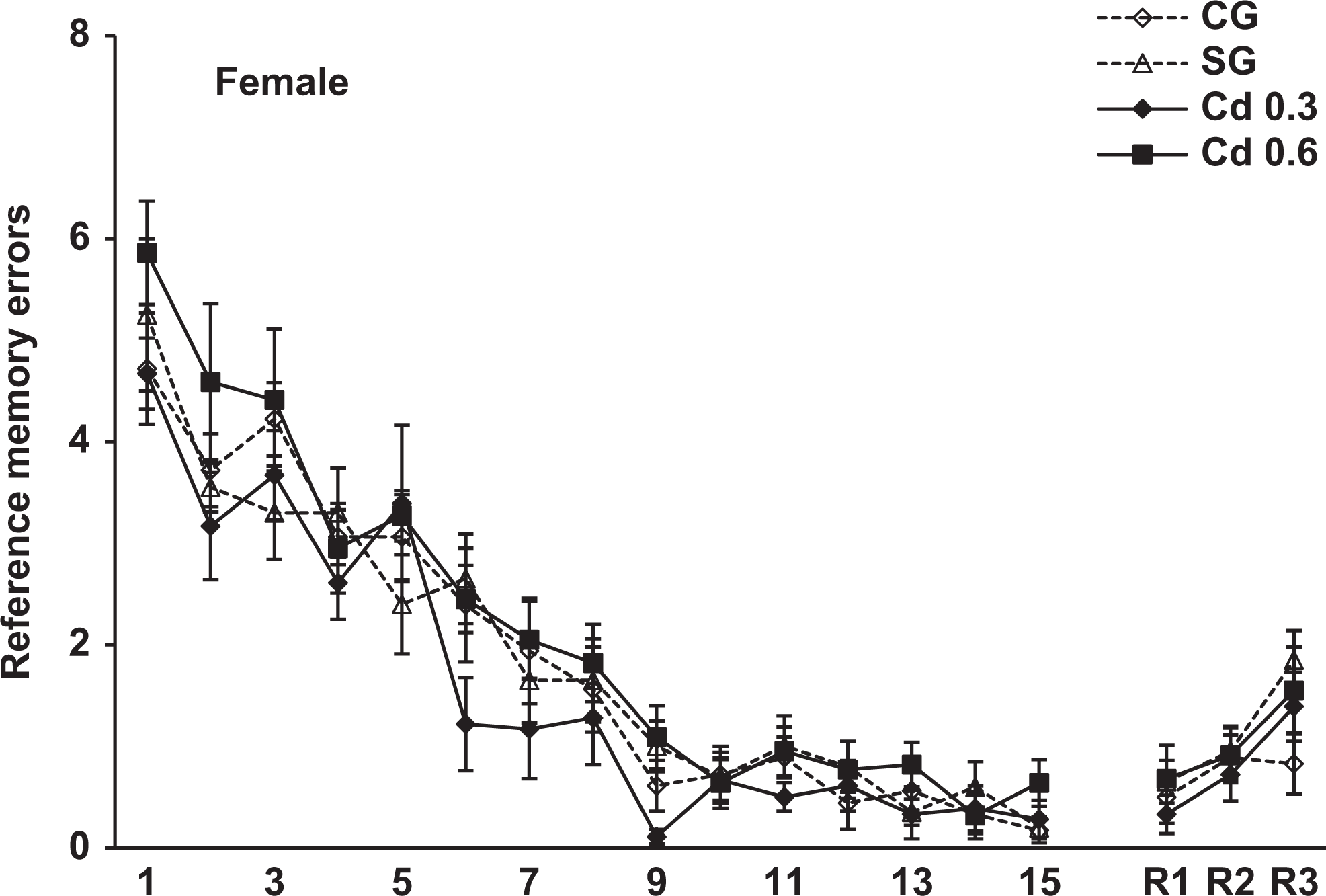
Reference memory errors scored during the 15-day training and 7 (R1), 21 (R2) and 42 (R3) days after training in the radial maze in the female offspring of the different groups. Data represent the mean errors of 8-11 animals per group.
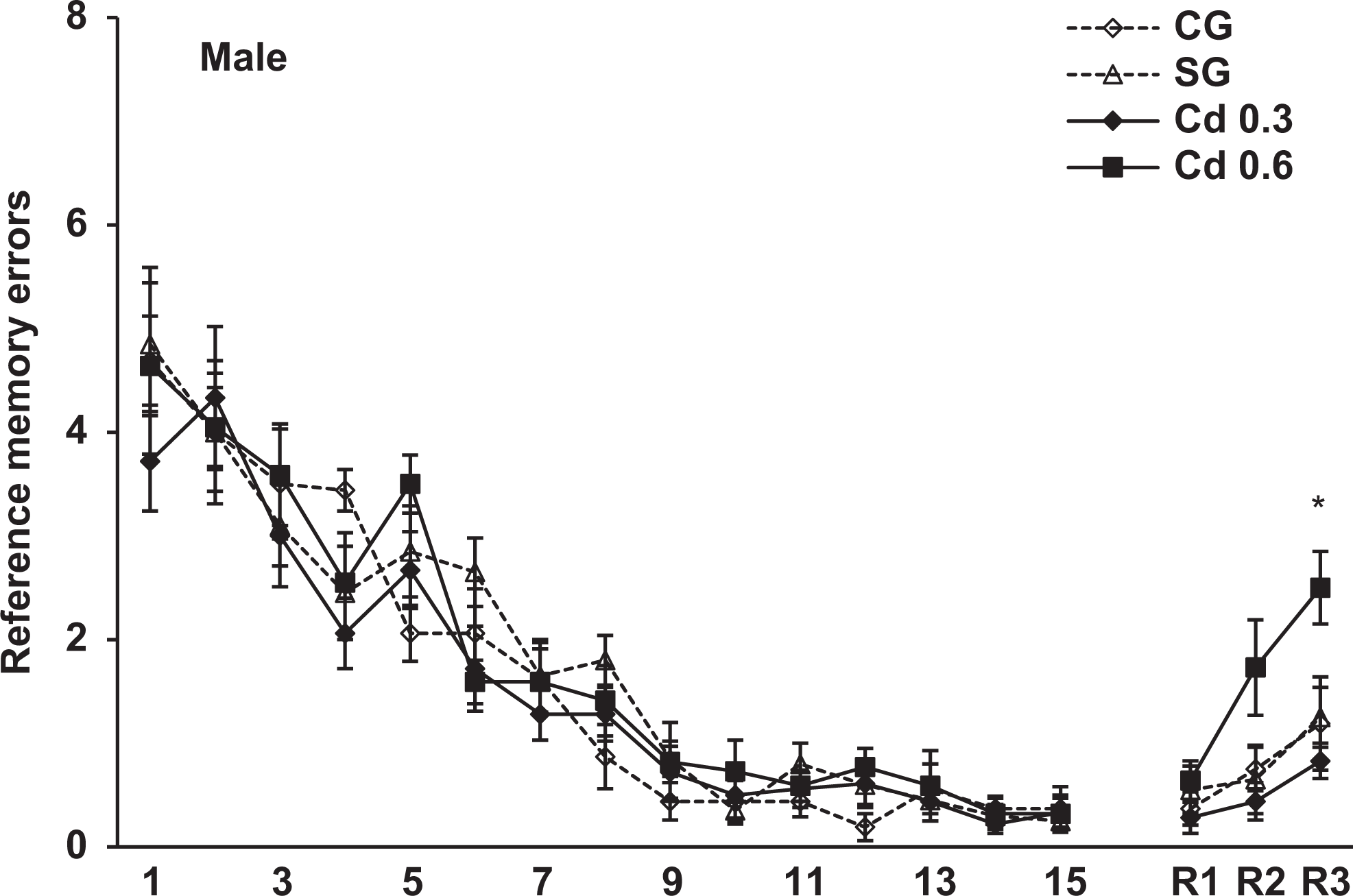
Reference memory errors scored during the 15-day training and 7 (R1), 21 (R2) and 42 (R3) days after training in the radial maze in the male offspring of the different groups. Data represent the mean errors of 8-11 animals per group. * p < 0.05, compared to control, saline and Cd 0.3 groups.
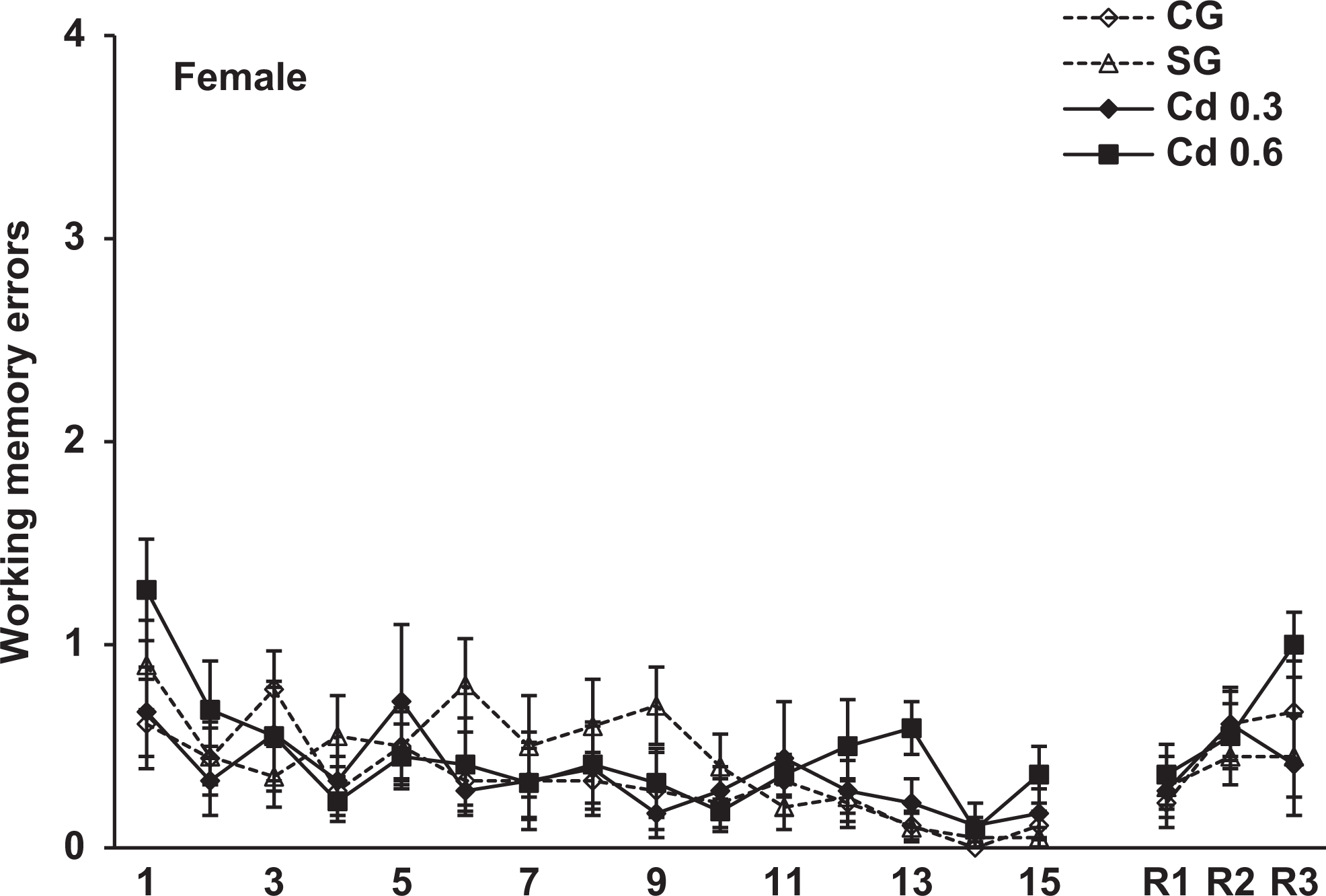
Working memory errors scored during the 15-day training and 7 (R1), 21 (R2) and 42 (R3) days after training in the radial maze in the female offspring of the different groups. Data represent the mean errors of 8-11 animals per group.
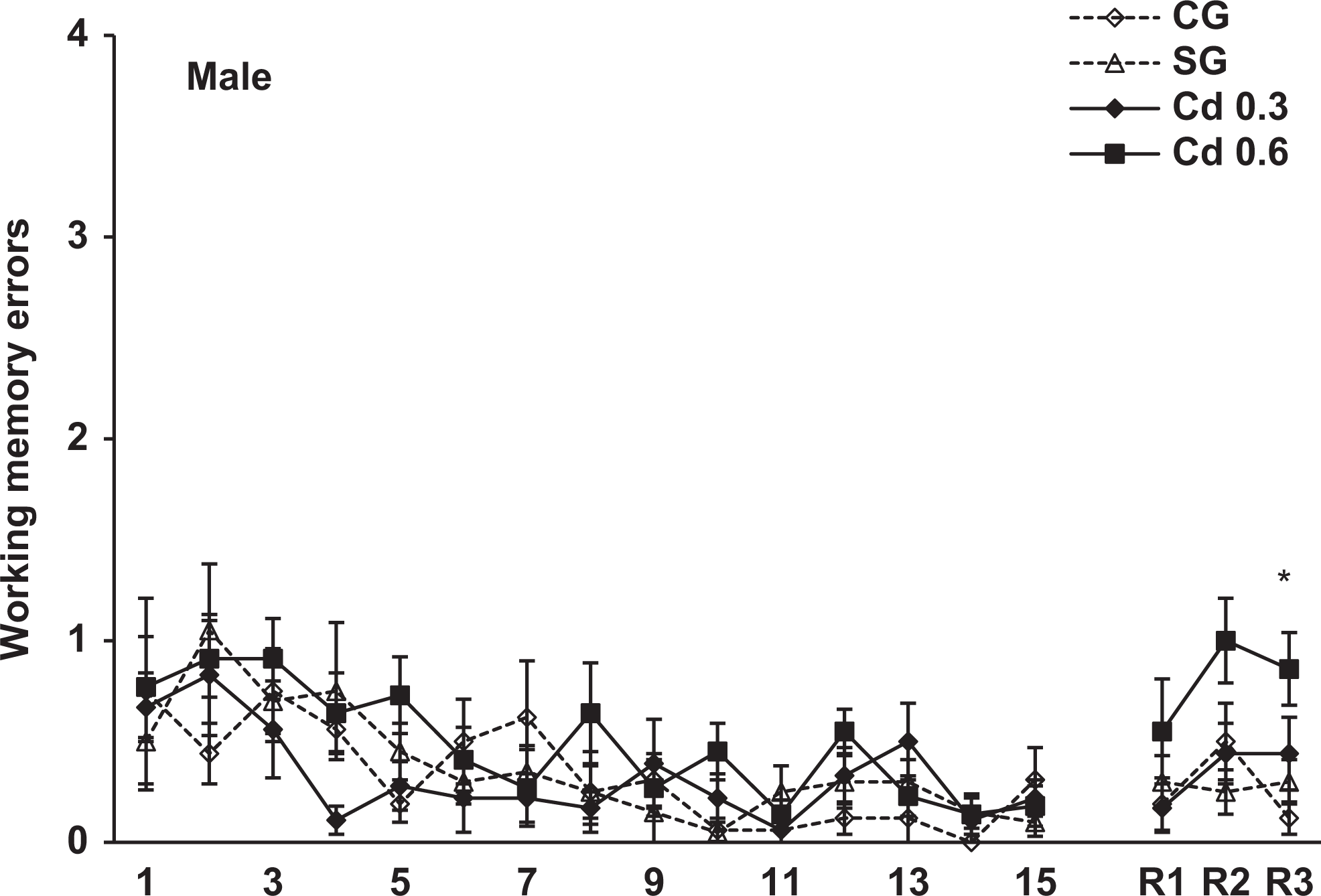
Working memory errors scored during the 15-day training and 7 (R1), 21 (R2) and 42 (R3) days after training in the radial maze in the male offspring of the different groups. Data represent the mean errors of 8-11 animals per group. * p < 0.02, compared to control and saline groups.
Discussion
The results of this study demonstrated that prenatal exposure to Cd during organogenesis impairs the retention of memory evaluated in both tasks. In the step-down inhibitory avoidance, we observed that the male offspring of dams exposed to 0.6 mg Cd during organogenesis evidenced deficits in the retention of LTM. Furthermore, the males prenatally exposed to 0.6 mg Cd also showed impairment in the retention of spatial memory evaluated in the radial maze.
As mentioned previously, Cd exposure in humans produced several CNS alterations, among them, memory deficits. Some rare epidemiological studies in children have found a positive correlation between Cd levels and cognition deficits, emotional disorders, 35 -37 and high incidence of learning disabilities. 38,39 Experimental Cd exposure in rats also demonstrated learning and memory deficits in different tasks. After a single exposure to Cd in 6-day-old rats, while the acquisition of a passive avoidance task was normal, the Cd-treated animals had a deficit in retention of this task 3 days after acquisition. 40 The Cd exposure in infancy also appears to affect social memory processes in adulthood, since they failed to learn the identity of a strange rat in a social recognition test. 41 In other study, female lactating mice were exposed to Cd for 7 days just after delivery, and the pups in their adulthood showed significant impairment of memory in step-down test and Morris water maze. 42
Relatively few investigations have been conducted on long-lasting behavioral alterations in offspring prenatally exposed to Cd. In a recent work, we observed that the exposure to 0.6 mg Cd during organogenesis produced a delay in the development of the righting reflex and of the cliff aversion in the pups and a lower anxiety level in 45- and 90-day-old offspring. 28 Other studies have shown that prenatal Cd exposure led to altered motor activity, increased aggressive behavior, altered drinking behavior, and impaired acquisition and retention of a conditioned escape test and the social memory processes. 26,27
In relation to the possible biological mechanisms underlying Cd-related memory impairment, such as that of LTM and spatial memory observed in this work, some evidence suggests that Cd treatment would produce alterations in brain areas and neurotransmitter systems involved in memory regulation. As we have mentioned, Cd is neurotoxic; even at low doses, it produces adverse effects on the developing CNS. 10,43 Some studies have demonstrated that the pathologies of the CNS are produced as a consequence of high blood–brain barrier permeability to Cd, resulting in greater concentrations of Cd in the brain. 14 Another work showed that the hippocampus accumulates the divalent metals to a greater extent than do other parts of the brain, brain region that plays an important role in memory consolidation. 44 Furthermore, other authors also observed that, in rats, Cd impairs cell viability on immature hippocampus slices 45 and glutamatergic synaptic transmission in hippocampal CA1 area. 46 Several studies suggest that the stimulation of cholinergic and glutamatergic systems facilitates learning and memory. 47 -50 Other neurotransmitter systems, such as the GABAergic and serotonergic, exert an inhibitory influence on learning and memory. 51 -53
Cadmium can also affect cholinergic transmission 54 because it modifies acetylcholine release, 55 impairs choline uptake, 56 and reduces acetylcholine concentrations in several brain areas, including the hippocampus. 57 As a consequence of gestational exposure to 10 mg Cd acetate per liter of drinking water during the whole pregnancy, the levels of serotonin and their metabolite, 5-hydroxyindolacetic acid, increased in all areas of newborn brains. 58
On the other hand, some evidences indicate that Cd increases oxidative damage in CNS. Cadmium can initiate apoptosis process in distinct brain regions via generation of oxidative stress. 59 The ability of Cd to induce oxidative stress in brain cells is related to the induction of reactive oxygen species, lipid peroxidation, and reduction in antioxidant enzyme activity. 60,61 Also, rat pups exposed to Cd gestationally and lactationally through dams show changes in their brain antioxidant defense mechanisms at critical periods of development. 43
All this evidence indicates that exposure to Cd could produce alterations in brain areas or neurotransmitter systems that participate in memory regulation. Modifications in some of these areas or systems may also be responsible for the impairment in the retention of LTM and spatial memory that we observed in the male offspring prenatally exposed to Cd. Furthermore, different alterations in neurotransmitter systems could explain why in the step-down inhibitory avoidance the gestational exposure to Cd impairment the retention of memory measured 24 hours after training while in the radial maze, the impairment we observed 42 days after training. Step-down inhibitory avoidance involves learning that is usually acquired in one single trial; otherwise, the radial maze learning results in long experiments with slowly decaying error scores. Besides these differences in both tasks, the most influential neurotransmitters on passive avoidance are dopamine and glutamate, while glutamatergic and cholinergic activity predominantly affected the radial maze performance with very low level of dopamine influence. 50
This greater vulnerability of the male offspring to Cd is difficult to interpret. Considering that the hippocampus development is sexually dimorphic, 62 prenatal exposure to Cd could differentially affect the hippocampus or other brain regions involved in memory regulation, depending on the sex, and determining the greater susceptibility of the male offspring to memory deficits. On the other hand, animal studies suggest that organizational effects of gonadal hormones on brain development are responsible for the sex differences in cognitive performance that manifest as adults. 63 Thus, another possibility is that Cd produced alterations in gonadal steroids during critical stages of development, thus causing sex differences in spatial memory.
In conclusion, our results indicated that exposure to Cd during organogenesis produced impairment in the retention of LTM and spatial memory in the young male offspring. Study of neurotransmitter systems and the ability of Cd to induce oxidative stress on brain areas that participate in memory regulation is needed to clarify the possible mechanisms involved in the observed memory deficits.
Footnotes
Author Contributions
Fernanda Gumilar contributed to acquisition, analysis, and interpretation and critically revised the manuscript. Cristina Bras, Patricia Aggio, Sergio Dominguez, Mariana Bartos, Cristina Gallegos, and Nina Mónaco contributed to acquisition and analysis and drafted the manuscript. Alejandra Minetti contributed to conception and design and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the Secretaría General de Ciencia y Tecnología of Universidad Nacional del Sur under Grant (24/ZB44).
