Abstract
Male and female Syrian hamsters of the strain Han: AURA from the Fraunhofer Institute for Toxicology and Experimental Medicine breeding colony were maintained as control animals for five toxicity/carcinogenicity studies and were examined for the presence of neoplastic disease either when they died or when the study terminated. In total, 250 male animals and 250 female animals in three carcinogenicity studies were analyzed as well as the results of a 6-month study (fifty males and fifty females) and a 12-month study (fifty males and fifty females). In the 24-month studies, in the male animals, twenty-five organs were affected with malignant and benign tumors. In the female animals, twenty-four organs were affected with malignant and benign tumors. The most frequently affected tissue in the three 24-month studies was the adrenal gland where 141 cortical adenomas (56.4%) were noted in 250 male animals and 115 cortical adenomas (46%) noted in 250 female animals. In addition, a high incidence of squamous cell papilloma in the vagina was noted in female hamsters on the 24-month studies. A moderate incidence of squamous cell papilloma of the nonglandular forestomach, benign granulosa cell tumor of the ovary, lymphoma of the hemopoietic system, endometrial stromal polyp, and adenocarcinoma of the uterus and islet cell adenoma of the pancreas were also observed on the 24-month studies.
Currently, rats and mice are used in far greater numbers in biomedical research than Syrian hamsters (Mesocricetus auratus; Strandberg 1987). Information about the incidence of spontaneously occurring tumors in Syrian hamsters is essential if Syrian hamsters are to be used for acute and chronic toxicity studies (Pour et al. 1976a). Initially, researchers believed that hamsters had a low incidence of spontaneous tumors (Homburger and Bernfeld 1985; Strandberg 1987). Gradually, as more reports of spontaneous tumors in hamsters appeared, it was realized that this was not the case (Homburger and Bernfeld 1985); however, some workers still believe that the number of tumors in hamsters is low (Kamino et al. 2001a). The interpretation of tumor incidences in hamster carcinogenicity studies depends on a good, working knowledge of the background neoplastic and nonneoplastic lesions of the species involved (Pour et al. 1976a)
Hamsters are occasionally chosen as the candidate species for carcinogenicity studies in preclinical studies. The reasons for this include the fact that the hamster lung has proved to be particularly resistant to inhaled materials known to be carcinogenic in the rat (Mohr and Dungworth 1988). The hamster is the preferred rodent species chosen for peroxisome proliferator–activated receptor alpha (PPAR-α) carcinogenicity studies because the compounds being tested are known to be liver enzyme inducers which may result in liver tumors in the mouse and the hamster is thought to be less likely to develop liver tumors (Choudhury et al. 2000).
Materials and Methods
Animals
Male and female Syrian hamsters of the strain Han: AURA from the Fraunhofer ITEM breeding colony were maintained as control animals for toxicity/carcinogenicity studies. The hamsters were aged approximately 6 weeks at the start of the studies. They were housed individually in Makrolon (polycarbonate; EBECO, Germany) type-III cages with absorbent softwood as bedding material in the cages (Ssniff ¾, Ssniff Spezialdiäten, Soest, Germany). Animal room temperatures were generally maintained at 22 ± 2°C and 40–70% relative humidity with a 12-hr light/dark cycle controlled by an automatic timing device. All hamsters had free access to tap water (Hannover city water supplier, Germany) in Makrolon bottles (approximately 150 ml) with sipper tubes and were fed a hamster diet (V 2144) supplied by Ssniff Spezialdiäten. Drinking water and diet were subjected to routine chemical analysis to monitor for contaminants and offered fresh at weekly intervals. Animal health was always monitored by a veterinary officer. All studies were conducted according to Good Laboratory Practice (GLP) and following the regulations of the German animal protection law.
Histopathology
Complete necropsies were performed on all decedent and terminal animals in all of the studies. Moribund animals were always immediately removed from the study and sacrificed and necropsied, thus autolysis was never encountered. Harvested tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 3–4 µm, and stained with hematoxylin and eosin. Trimming was done according to Ruehl-Fehlert et al. (2003), Kittel et al. (2004), and Morawietz et al. (2004). A complete histopathological examination including the larynx, the teeth of the upper jaw, and macroscopic observations was conducted in all animals. The bones (nasal and paranasal cavities, sternum, femur, knee joint, and proximal tibia) were routinely processed, decalcified prior to embedding and also histologically examined in all hamsters (in all studies). The abdominal and thoracic cavities were only grossly examined and examined histopathologically in the case of a positive macroscopic finding.
Study Design
This article is based on three hamster 24-month carcinogenicity studies conducted at the Fraunhofer Institute for Toxicology and Experimental Medicine, Germany, during the period 1999–2009. The first study contained thirty control male and thirty control female animals, the second study contained sixty control males and sixty vehicle control males as well as sixty control females and sixty vehicle control females. The third study contained 100 female and 100 male control animals. The results from these three studies have been presented separately and then presented as combined data so that there are 250 male and 250 female animals in total in the three 24-month studies group. Information was gathered from control groups only. All control animals were cage controls; however, in the second 24-month carcinogenicity study, there was also a control group that received the vehicle. One hundred and twenty hamsters (sixty males and sixty females) received a vehicle control.
The vehicle was prepared by dissolving 40 g hydroxypropylmethylcellulose in 1,000 ml of water from a purification system (Millipore; = 4% hydroxypropylmethylcellulose solution). Ten to fifteen drops of Med Antifoam C/1,000 ml were added to the solution. The animals were treated with the vehicle daily by oral gavage for 730 consecutive days, that is, until the day before necropsy. All other control animals in this and all other studies were untreated cage controls.
In addition, the results of a 6-month study (fifty cage control males and fifty cage control females) and a 12-month study (fifty cage control males and fifty cage control females) were analyzed. The incidence of tumors was defined as low (<10/250), moderate (10–50/250), and high (>50/250).
Results
Unscheduled Deaths and Decedents
No unscheduled deaths occurred during the 6-month study. Two male hamsters were euthanatized during the 12-month study. The hamsters died on day 351 and 364, respectively. The causes of death were bacterial septicemia and metastasizing hemangiosarcoma, respectively.
The first 24-month study produced mortality rates for male/female hamsters of 1%/0% at 365 days and 14%/8% at 546 days and 64%/71% at 728 days. The second 24-month study produced mortality rates for male/female cage control hamsters of 2%/0% at day 360, 5%/8% at day 540, 13%/28% at day 660, and 25%/58% at day 748. In addition, the second 24-month study produced mortality rates for male/female vehicle control hamsters of 2%/0% at day 360, 5%/15% at day 540, and 12%/47% and 22%/73% at day 748. The third 24-month study produced mortality rates for male/female hamsters of 0%/3% at day 364, 0%/10% at day 532, 3%/60% at day 644, and 23%/87% at day 728. In all three studies, left atrial thrombosis was a frequent cause of death in these animals and occasionally tumor development was considered the cause of death.
The incidence of tumors in the 6-month study, 12-month study, first 24-month study, second 24-month study, third 24-month study, and combined 24-month studies are given in Tables 1, 2, 3, 4, 5 and 6, respectively. A greater number of female hamsters died in the 24-month studies than male hamsters, and left atrial thrombosis was a frequent cause of death in these animals and occasionally tumor development was considered the cause of death.
The incidence of tumors in control Han: AURA Syrian hamsters from the 6-month study.

[B], Benign tumor.
Incidence of tumors in control Han: AURA Syrian hamsters from the 12-month study.
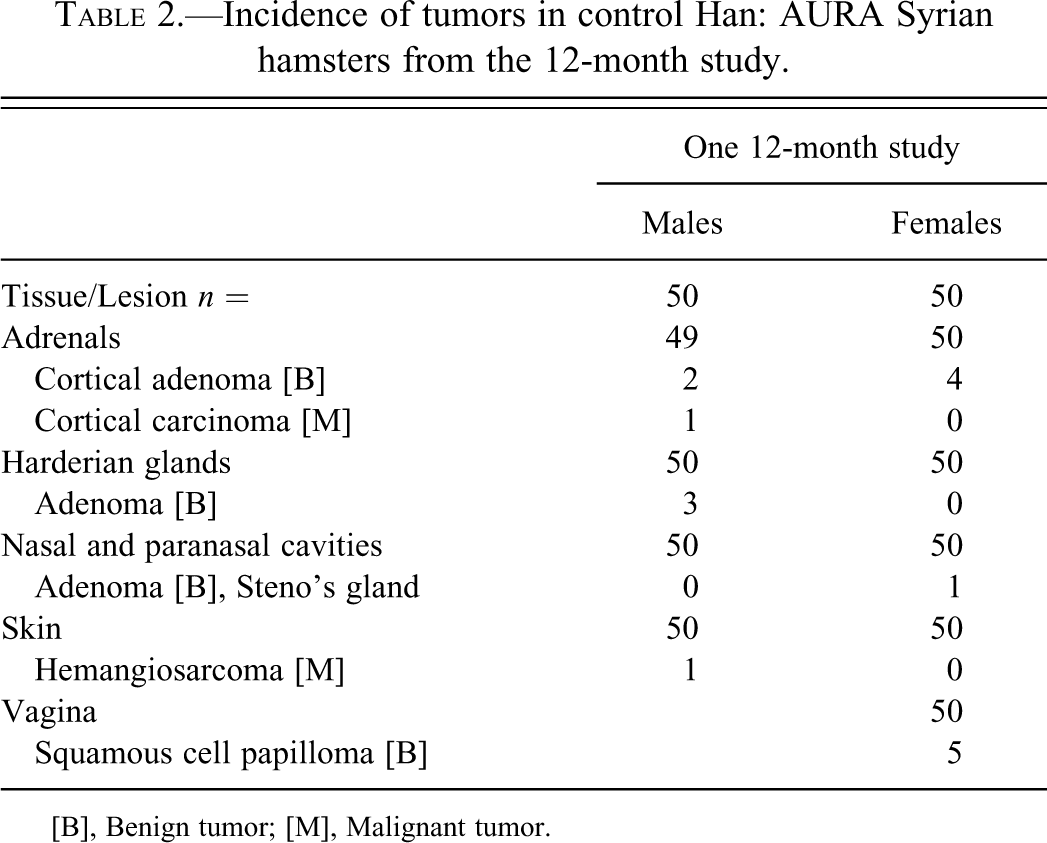
[B], Benign tumor; [M], Malignant tumor.
Incidence of tumors in control Han: AURA Syrian hamsters in the first 24-month study.

[B], Benign tumor; [M] Malignant tumor.
aTissues missing due to small size of tissues and difficulty in accessing tissues at necropsy.
Incidence of tumors in control Han: AURA Syrian hamsters in the second 24-month study.
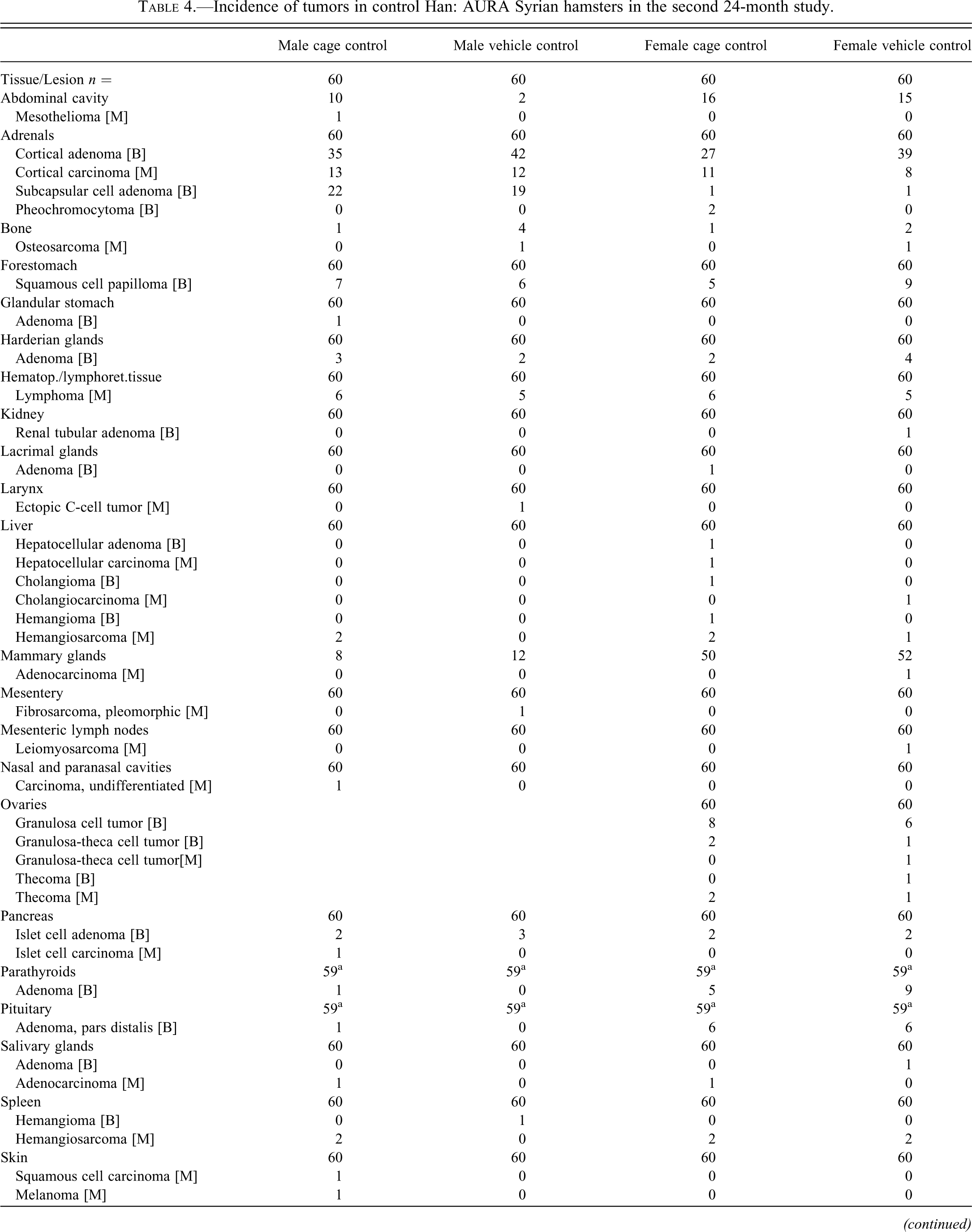
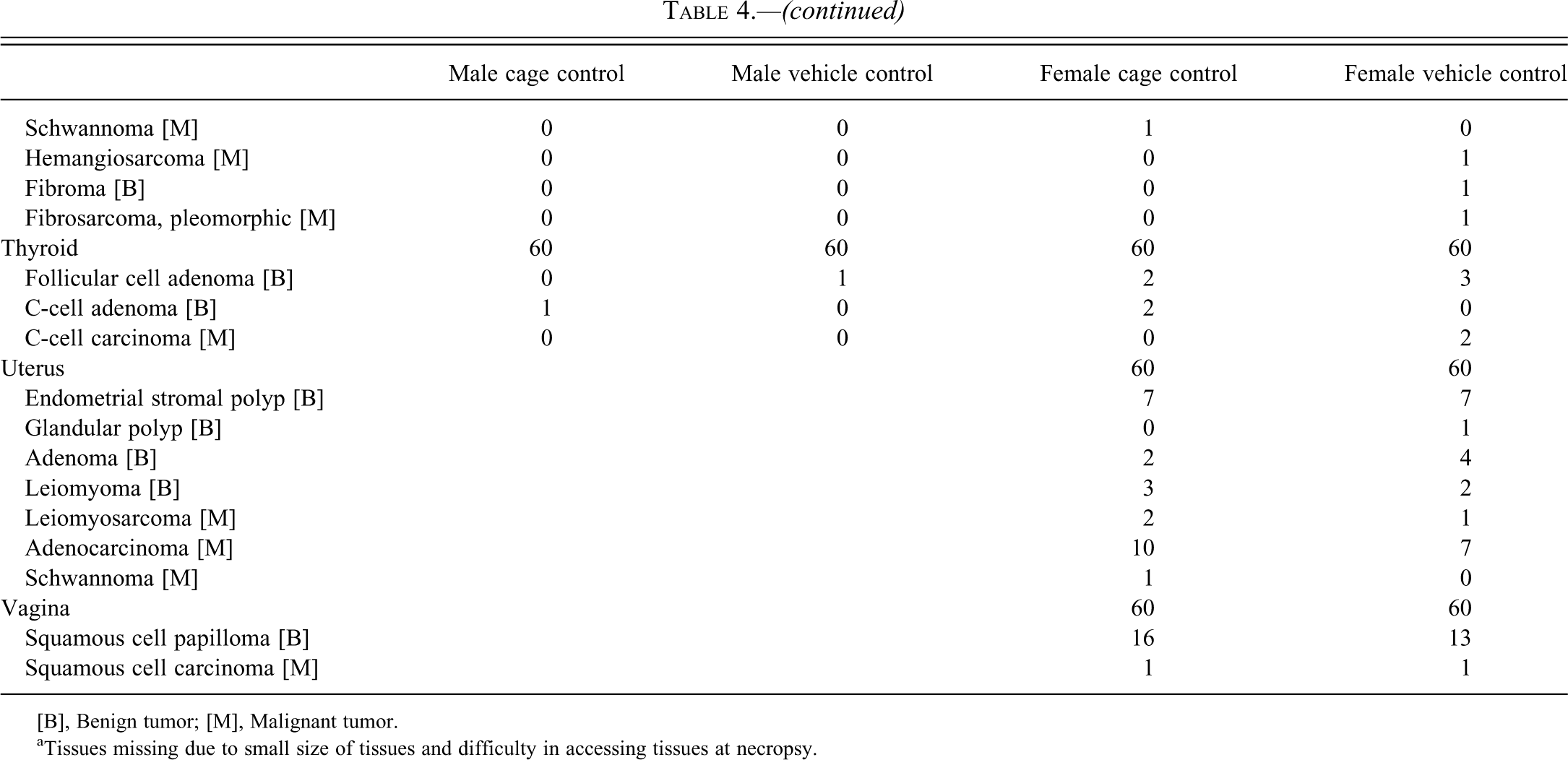
[B], Benign tumor; [M], Malignant tumor.
aTissues missing due to small size of tissues and difficulty in accessing tissues at necropsy.
Incidence of tumors in control Han: AURA Syrian hamsters in the third 24-month study.


[B], Benign tumor; [M], Malignant tumor.
aTissues missing due to small size of tissues and difficulty in accessing tissues at necropsy.
Incidence of tumors in control Han: AURA Syrian hamsters in three 24-month studies.


[B], Benign tumor; [M] Malignant tumor.
aTissues missing due to small size of tissues and difficulty in accessing tissues at necropsy.
In the first 24-month study, twenty-four of the thirty male animals had tumors and twenty-nine of the thirty female animals had tumors. Twenty-two male animals had benign tumors and twenty-two of the thirty female animals had benign tumors. Eight of the thirty male animals had malignant tumors and seventeen of the thirty animals had malignant tumors. In the second 24-month study, fifty-six of the sixty cage control, male animals and fifty-five of the sixty vehicle control, male animals had tumors and fifty-two of the sixty cage control female animals and fifty-six of the sixty vehicle control female animals had tumors. Forty-seven of the sixty cage control, male animals and fifty of the sixty vehicle control, male animals had benign tumors and forty-seven of the sixty cage control, female animals and fifty of the sixty vehicle control, female animals had benign tumors. Twenty-five of the sixty cage control, male animals and nineteen of the sixty vehicle control, male animals had malignant tumors and twenty-eight of the sixty cage control, female animals and twenty-nine of the sixty vehicle control, female animals had malignant tumors.
In the third 24-month study, 85 of the100 control male animals and 81 of the 100 control female animals had tumors. Of these, 72 of the 100 control, male animals and 66 of the 100 control, female animals had benign tumors and 38 of the 100 control, male animals and 45 of the 100 control female animals had malignant tumors.
In the 24-month studies, in the male animals, twenty-five organs were affected with malignant and benign tumors. In the female animals, twenty-four organs were affected with malignant and benign tumors. In male animals, 285 benign tumors and 104 malignant tumors were reported. In female animals, 366 benign tumors and 147 malignant tumors were reported.
The most frequently affected tissue in the 24-month studies was the adrenal gland where a total of 141 (56.4%) cortical adenomas (Figure 1) were noted in male animals and a total of 115 cortical adenomas (46%) noted in female animals. In addition, a total of fifty-five cortical carcinomas (Figure 2) in male animals and a total of thirty-three cortical carcinomas in female animals were noted. A total of sixty-one subcapsular cell adenomas in male animals and a total of three subcapsular cell adenomas in female animals were also observed. A total of three benign pheochromocytomas were noted in female animals. Furthermore, cortical adenoma was observed in one female hamster on the 6-month study and cortical adenomas (two in male animals and four in female animals) and carcinomas (one in a male animal) were noted in animals on the 12-month study.
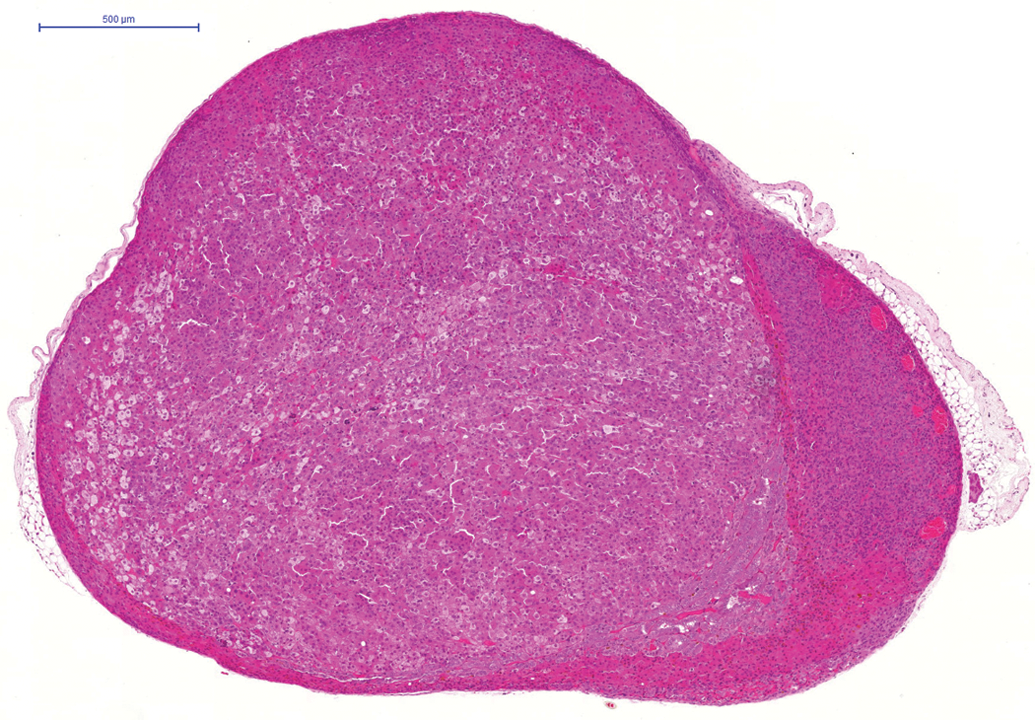
Cortical adenoma in adrenal gland of hamster on a 24-month study. H and E.
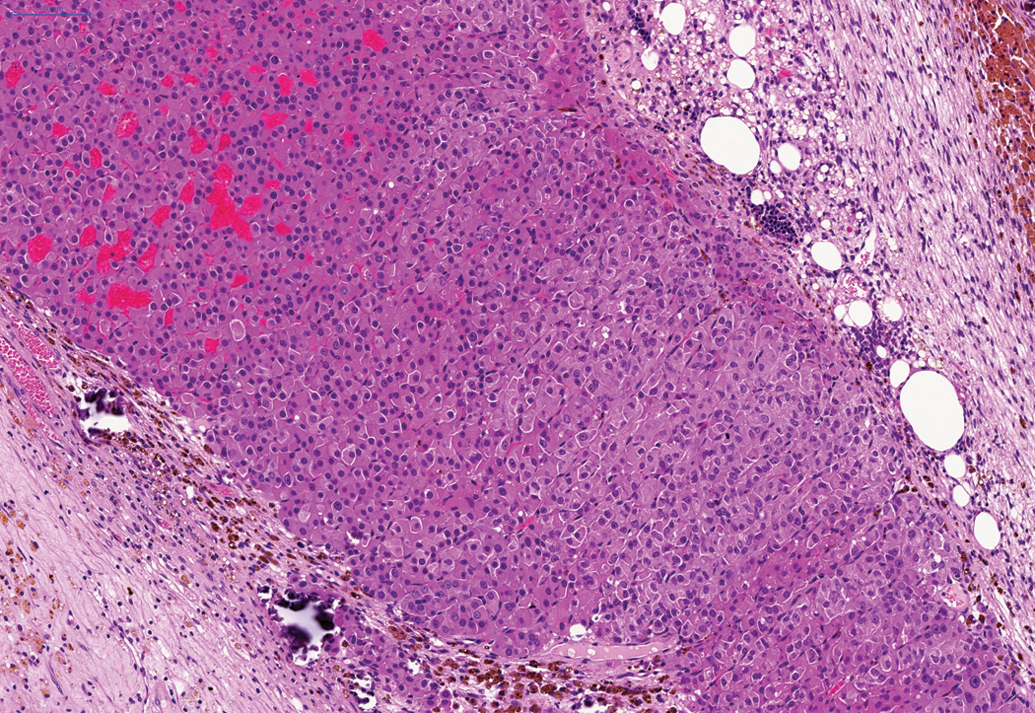
Cortical carcinoma in adrenal gland of hamster on a 24-month study. H and E.
Other tumors with a low to moderate incidence in the 24-month studies included a total of 26 of the 250 male animals and a total of 22 of the 250 female animals which demonstrated squamous cell papilloma of the nonglandular forestomach (Figure 3). In addition, a total of 20 of the 241 female animals demonstrated adenomas of the pituitary and a total of 24 of the 242 female animals demonstrated adenoma of the parathyroid (Figure 4). Furthermore, a total of 21 of the 250 male animals showed adenomas of the Harderian gland (24-month studies) and a total of 3 of the 50 male animals on the 12-month study showed adenoma of the Harderian gland. Also, a total of 11 of the 250 male animals demonstrated adenoma of the islet cells of the pancreas (Figure 5). In the hematopoietic and lymphoreticular systems, a total of 15 of the 250 male hamsters and a total of 21 of the 250 female hamsters displayed malignant lymphoma.
Squamous cell papilloma in forestomach of hamster on a 24-month study. H and E.
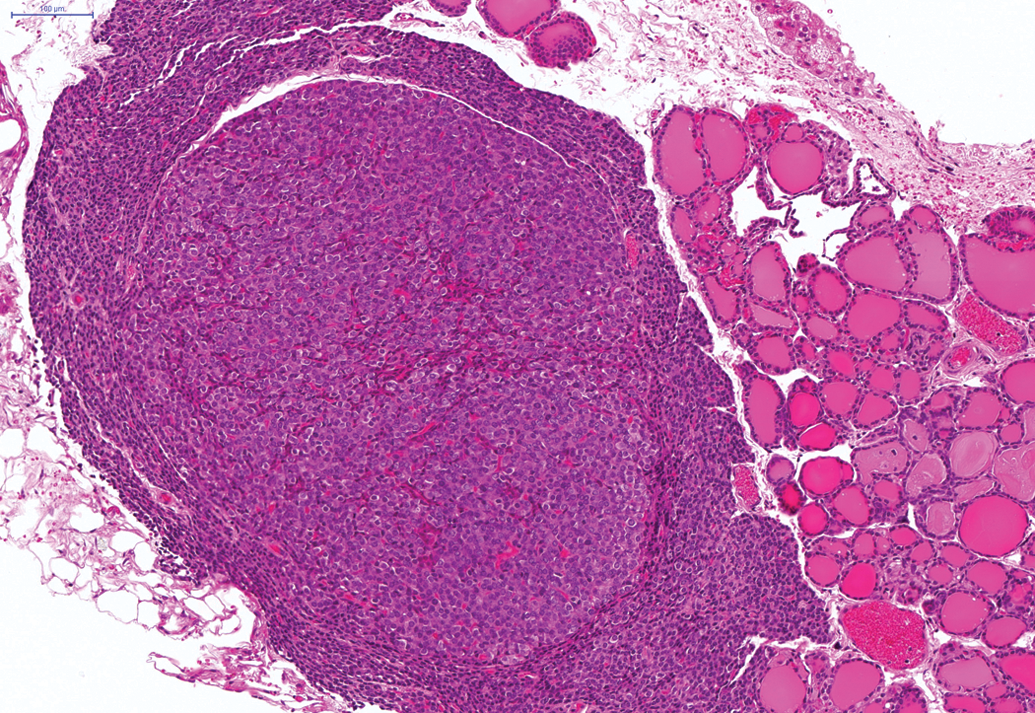
Adenoma of the parathyroids in hamster on a 24-month study. H and E.
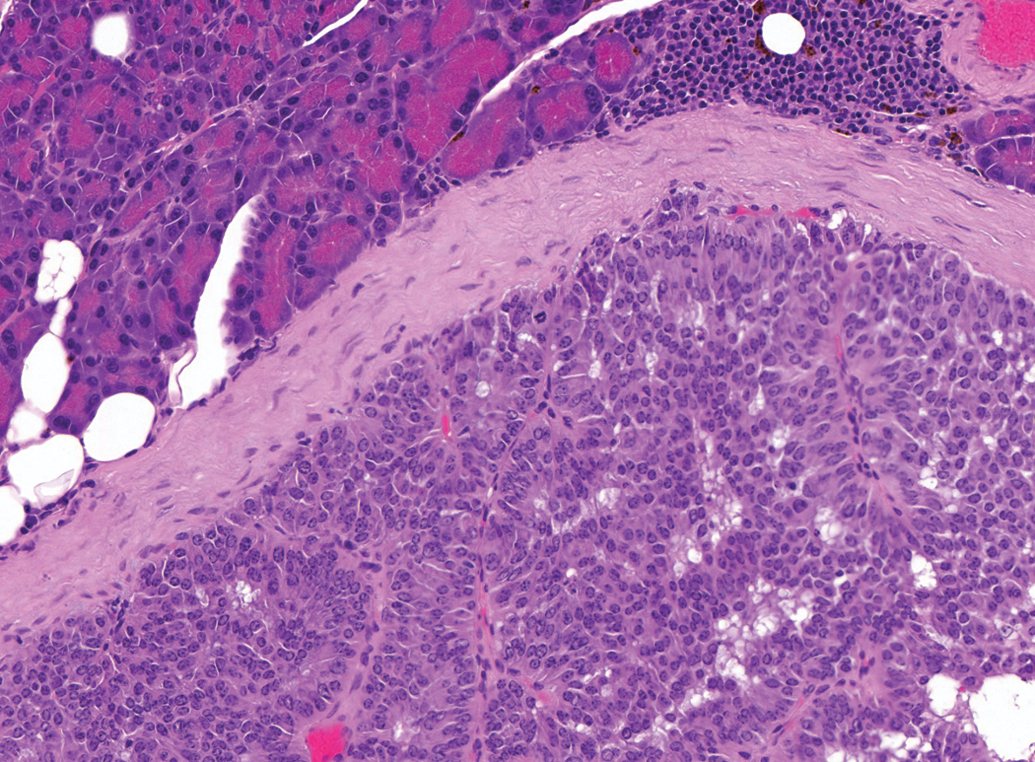
Islet cell adenoma in pancreas of hamster on the 6-month study. H and E. ×100.
In female animals on the 24-month studies, a moderate incidence of benign granulosa cell tumors (Figure 6) was noted in the ovaries (total of 25 of the 250) and a moderate incidence (total of 28 of the 250) of female animals showed endometrial stromal polyps and a total of 34 of the 250 female animals demonstrated adenocarcinoma of the uterus (Figure 7). In female animals in the 24-month studies, a high incidence of squamous cell papilloma (Figure 8) was noted in the vagina (total of 57 of the 250 animals) and this finding was also present in the 12-month study, where 5 of the 50 squamous cell papillomas were noted in female animals.
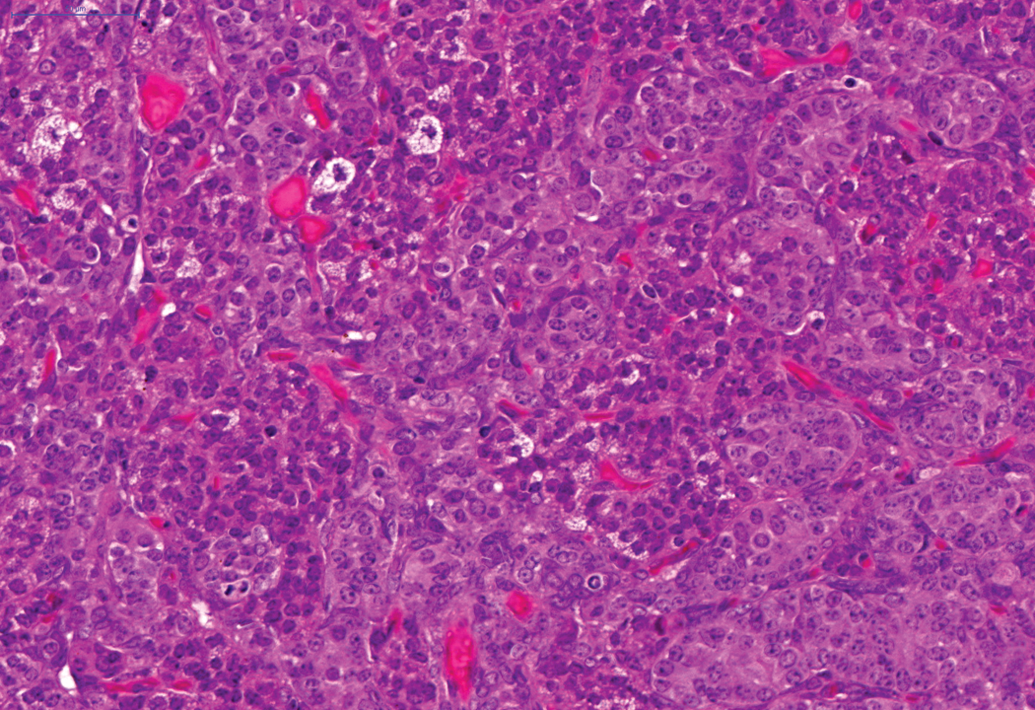
Benign granulosa cell tumor in ovary on a 24-month study. H and E.
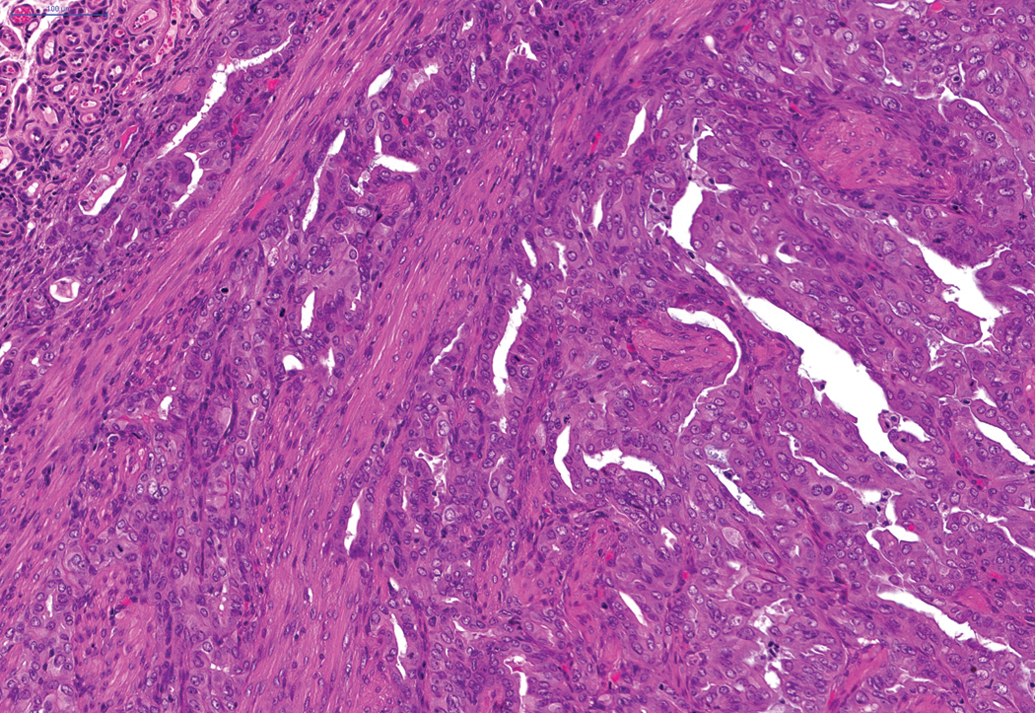
Adenocarcinoma in the uterus of a female hamster on a 24-month study. H and E.
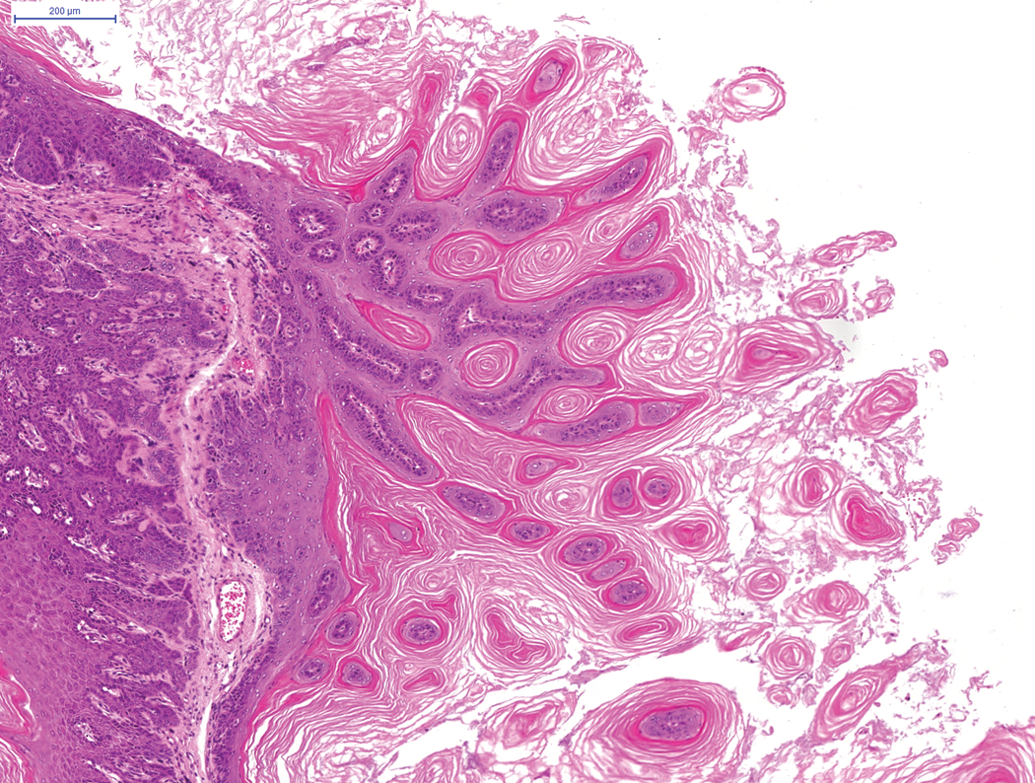
Squamous cell papilloma in hamster vagina on a 24-month study. H and E.
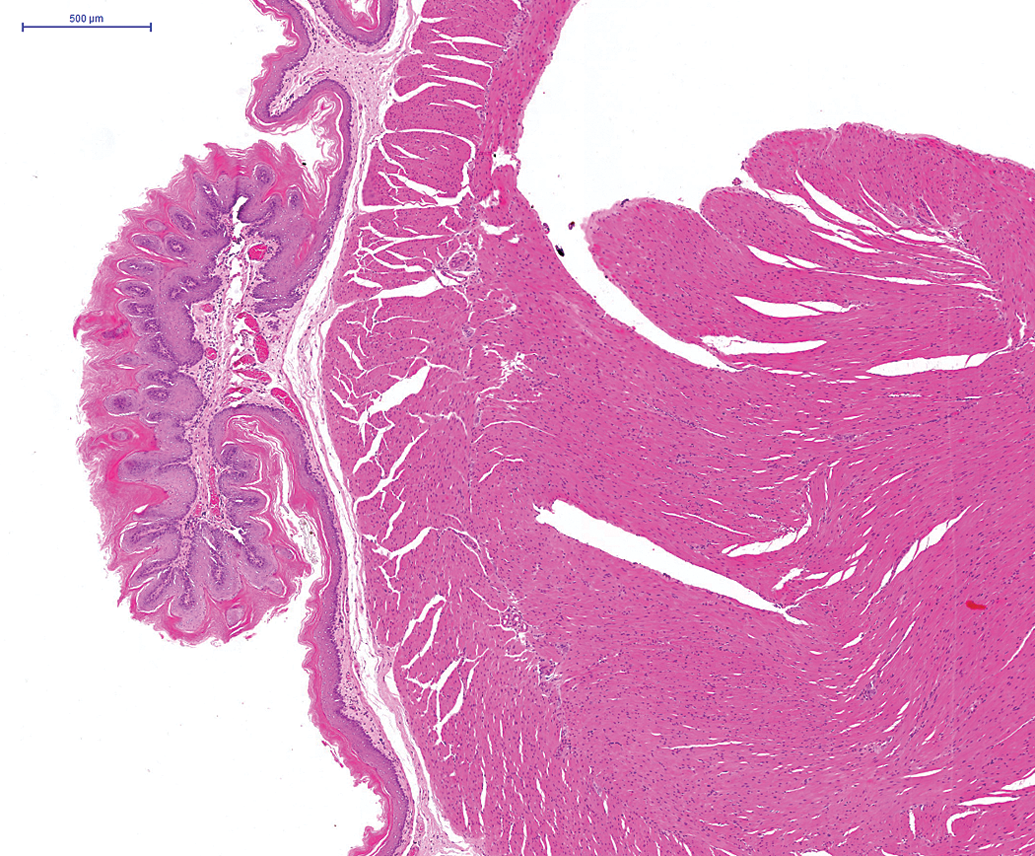
In male animals, an adenoma was noted in the epididymis of one animal on the second 24-month study. In addition, a renal carcinoma was noted on the third 24-month study (Figure 9) and an adenoma in Steno’s gland in the nasal cavity was reported on the 12-month and an adenoma and carcinoma of the nasal and paranasal cavities were reported in the 24-month studies (Figure 10). Benign and malignant ectopic C-cell tumors were noted in both male and female animals in the trachea and larynx in the 24-month studies (Figure 11).
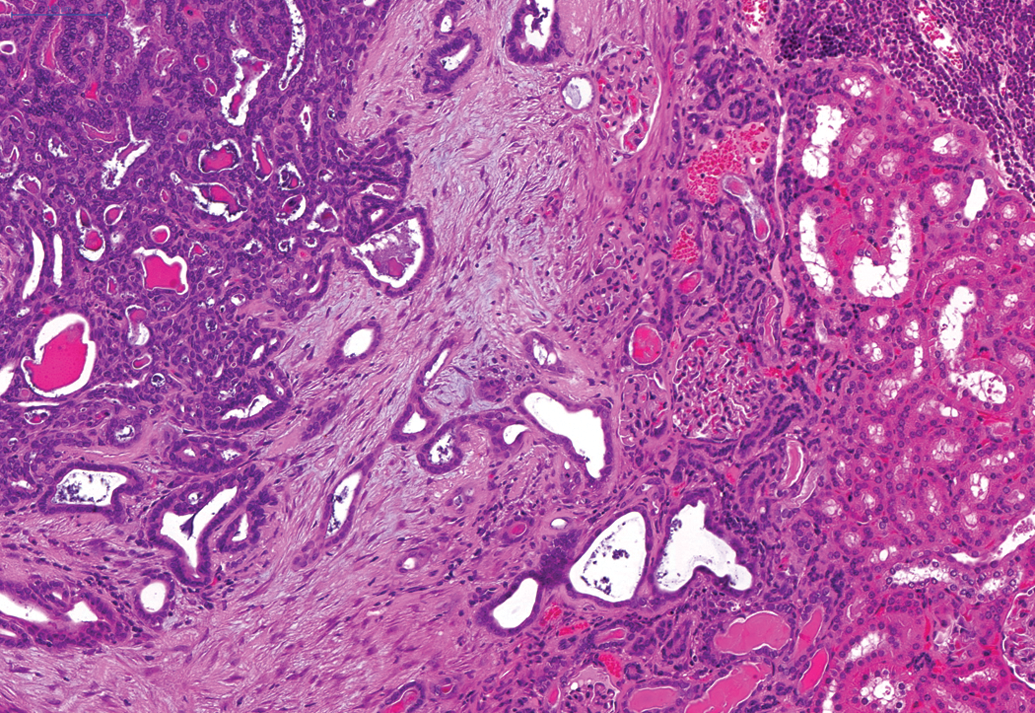
Renal tubular carcinoma present in hamster kidney on a 24-month study. H and E.
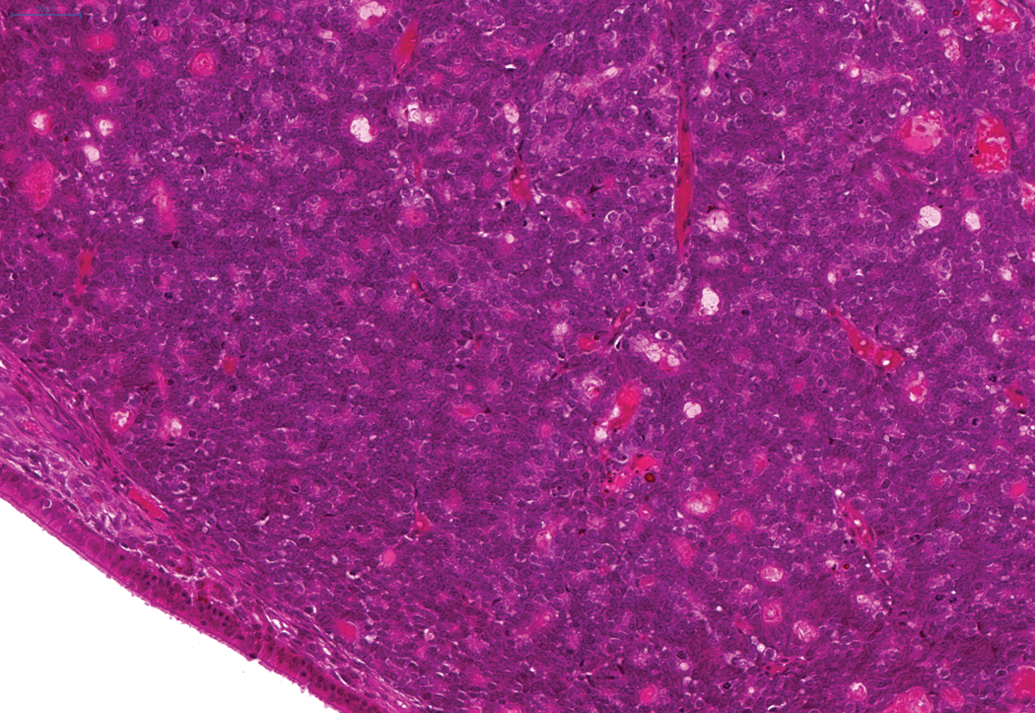
Adenoma present in Steno’s gland in the nasal cavity of a hamster on a 24-month study. H and E.
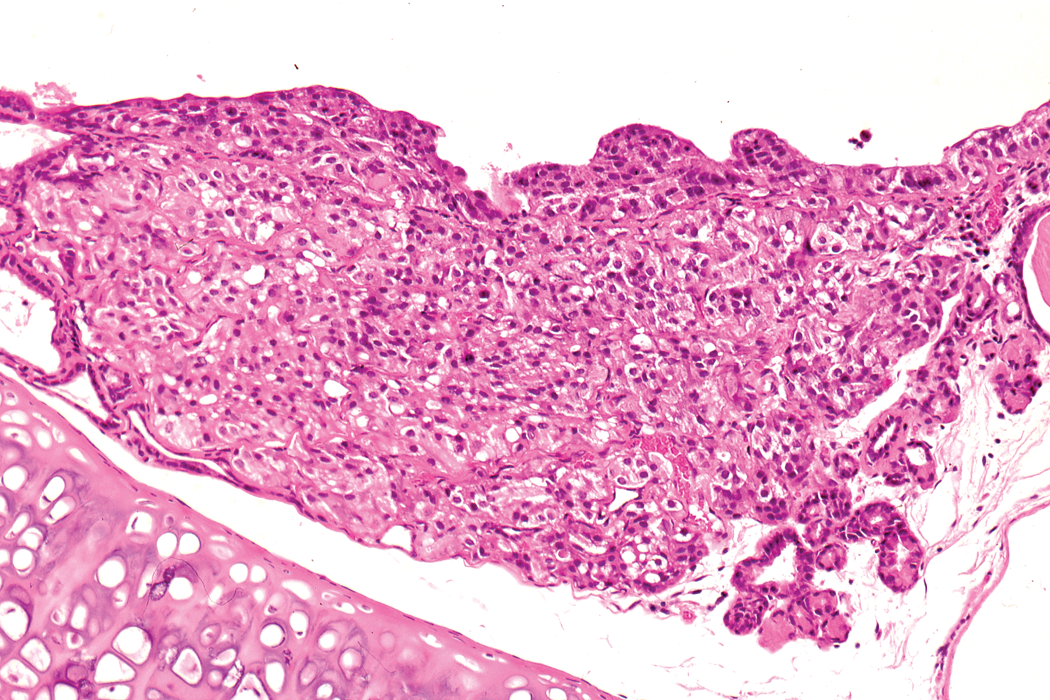
Ectopic C-cell tumor, benign, in the larynx of a hamster on a 24-month study. H and E. ×100.
Discussion
It is important to have information on the background tumors of hamsters, so users may compare different breeds, evaluate the worth of hamster carcinogenesis models, and eventually allow extrapolation of observations to humans (Pour et al. 1976a). In our study, male hamsters outlived female hamsters, whereas there was a higher incidence of tumors in female hamsters. The majority of the tumors in females were benign, which is in line with a study by Pour et al. (1976a), where males also outlived females that had a higher incidence of benign tumors. These results are in contrast to those in a study reported by Kirkman (1962), where the incidence of tumors in male hamsters was higher than in females.
In our 24-month studies, benign tumors (adenoma of nasal and paranasal cavities), malignant tumors (undifferentiated carcinoma of nasal and paranasal cavities), and ectopic C-cell tumors of the trachea and larynx were reported in low numbers in the respiratory tracts of hamsters. Furthermore, adenoma of Steno's glands of nasal and paranasal cavities occurred in the 12-month study, but no respiratory tumors in animals on the 6-month study were reported. Pour et al. (1976d) demonstrated a low incidence of respiratory tract tumors in two colonies of Syrian hamsters. These tumors were predominantly benign and found in the nasal cavity, trachea, and lungs (Pour et al. 1976d). The incidence of spontaneous tumors of the nasal cavity of the Syrian hamster is less than 0.1% (Pour et al. 1976d). The prevalence of spontaneous tumors in the lung of the Syrian hamster is low (Mohr et al. 1996).
The presence of spontaneous ectopic C-cell or neuroendocrine cell hyperplasia or benign or malignant tumors of these cells in the larynx (Pour 1985) or the trachea (Ernst et al. 1995) may be unique to hamsters. The frequency varies considerably between colonies of hamsters (Mohr et al. 1996). Pour (1985) reported an incidence of 9% in the larynx of Syrian hamsters. Ernst et al. (1995) have confirmed the neuroendocrine origin of these cells and state that they occur in the larynx and upper trachea in close proximity to the thyroid gland. Ernst et al. (1995) demonstrated that the neuroendocrine cells in these laryngeal and tracheal locations are positive for calcitonin and calcitonin gene–related peptide, neuron-specific enolase, and serotonin using immunohistochemistry. Benign tumors of neuroendocrine cells do not display invasion, whereas malignant tumors of these cells demonstrate submucosal and luminal invasion and metastasis to the lung (Ernst et al. 1995). We favor the term ectopic C-cell hyperplasia/adenoma/carcinoma rather than neuroendocrine cell hyperplasia/tumor since we consider the origin of these cells to be the C-cells from the thyroid. It is likely that the tumors arise from rests of C-cells which failed to migrate to the thyroid. The site of origin at the laryngotracheal junction, the morphology, and the immunohistochemistry of these cells appear to support this hypothesis (Ernst et al. 1995).
In our 24-month studies, tumors of the gastrointestinal system included adenoma of the glandular stomach, squamous cell papilloma and carcinoma of the forestomach, adenoma and adenocarcinoma of the salivary gland, and hemangiosarcoma and odontoma of the teeth. There was no particular sex bias in the occurrence of these tumors. A moderate incidence of squamous cell papilloma in the forestomach of male and female hamsters in our 24-month study occurred. In the gastrointestinal system, Pour et al. (1976d) reported that all digestive tract neoplasms occurred largely in male and female animals of two strains of Syrian hamsters. The tumors described were generally forestomach papillomas and intestinal adenocarcinomas. Spontaneous tumors of the oral cavity, buccal pouch, salivary glands, and esophagus are infrequent in the hamster (Takahashi and Okamiya 1996c), however, squamous cell papillomas of the forestomach are reported to be more common (Takahashi and Okamiya 1996c). In addition, numerous polyps and squamous cell papillomas of the hamster stomach are noted by Van Hoosier and Trentin (1979). The cause of the moderate incidence of squamous cell papillomas in the forestomach of hamsters is not known. No gastrointestinal tumors were present in hamsters in the 6- and 12-month studies.
In our 24-month studies, only one adenoma was reported in the glandular stomach of a hamster. Tumors of the glandular stomach are rarely observed in hamsters (Takahashi and Okamiya 1996a). Pour et al. (1976a) reported an absence of spontaneous tumors in the glandular stomach of hamsters derived from two colonies of Syrian hamsters. Connective tissue tumors such as leiomyomas and leiomyosarcomas may be encountered in the smooth muscle of the gastrointestinal system; however, these tumors are rare. In our 24-month studies, a leiomyosarcoma was reported in the cecum and a hemangioma was reported in the colon. The cecum and colon are reported to be predilection sites for intestinal adenomas and adenocarcinomas (Takahashi and Okamiya 1996a), but this was not our experience. Tumors composed of granular cells in the intestinal walls of white hamsters have been described by Pour et al. (1973). Aggregates of granular cells were considered to be a nonneoplastic (hyperplastic) finding in our study and this was occasionally observed in the serosa of the small and large intestine (results not shown). Tumors of the exocrine pancreas are extremely rare in the hamster (Pour and Tomioka 1996) in contrast to the high incidence of tumors in the endocrine pancreas.
Moore et al. (1996) state that although foci of hepatocyte alteration and cystic bile ducts are common in the livers of older hamsters, hepatocellular carcinomas, and cholangiocarcinomas are nevertheless rare in the livers of hamsters. This was also our experience with respect to liver tumors, although hemangiomas and hemangiosarcomas were slightly more common in our 24-month studies. No hepatic tumors were reported in our 6- and 12-month studies. Pour et al. (1976d) describe hepatocellular adenoma and carcinoma of liver, cholangioma and cholangiocarcinoma of the liver, liver hemangioendotheliomas, and gallbladder polyps and pancreatic duct adenomas largely in the male and female animals of two strains of Syrian hamsters.
In common with previous studies (Pour et al. 1976b), granulosa cell tumors (benign) were the most common ovarian tumor in female hamsters in our 24-month studies. Other ovarian tumors in our 24-month studies included malignant granulosa cell tumors, benign thecoma and malignant thecoma and a malignant granulosa-theca cell tumor. In contrast to Pour et al. (1979), the incidence of squamous cell papillomas in the vagina of hamsters was high in our 24-month studies, but the incidence of squamous cell carcinoma of the vagina was low. Squamous cell papilloma of the vagina was also observed in our 12-month study, but not in the 6-month study. Furthermore, there was a moderate incidence of endometrial stomal polyp and adenocarcinoma in the uterus in our 24-month studies. Endometrial adenocarcinoma has been previously reported in older Syrian hamsters (Pour et al. 1976b; Van Hoosier and Trentin 1979). Other uterine tumors in our 24-month studies included adenoma of the uterus, leiomyoma, glandular polyp, leiomyosarcoma and malignant schwannoma. No ovarian or uterine tumors occurred in our 6-month or 12-month hamster studies. Spontaneous adenomas of the epididymis are rare (Pour et al. 1976b; Ribas and Mostofi 1996) and in our 24-month studies, only one adenoma of the epididymis was reported.
One malignant melanoma was reported in the skin of a male hamster in our 24-month studies. Other skin tumors observed in our 24-month studies included squamous cell carcinoma, rhabdomyosarcoma, malignant schwannoma, hemangiosarcoma, fibroma, and pleomorphic fibrosarcoma. No skin tumors were reported in our 6-month study; however, a hemangiosarcoma of the skin was reported in a male hamster in our 12-month study. In general, spontaneous tumors of the skin in the hamster are rare except for malignant melanoma which is not uncommon in certain hamster strains (Ghadially and Ghadially 1996). For instance, malignant melanomas occurred only in cream and white hamsters in one study looking at cream, white, and albino hamster strains (Pour et al. 1979), however, this is not surprising since melanomas only occur in pigmented cell lines and thus not in albino hamsters (Strandberg 1987). Hamsters have small pigmented spots and the costovertebral spot or flank organ (Ghadially and Ghadially 1996). Ghadially and Ghadially (1996) state that the small pigmented spots are not susceptible to melanoma formation. Spontaneous malignant tumors of the mammary gland are very rare in hamsters (Fernandez et al. 1996) and only one adenocarcinoma of the mammary gland was reported in a female hamster in our 24-month studies.
In our 24-month studies, we noted a renal tubular adenoma in a male and female hamster and a renal tubular carcinoma in a male hamster. Naturally occurring kidney tumors are uncommon in the Syrian golden hamster (Tsuda et al. 1996).
In common with Pour et al. (1976a, 1976b), most tumors in our 24-month studies originated in the endocrine system. In our 24-month studies, there was a moderate incidence of parathyroid adenomas in female hamsters. C-cell adenomas and carcinomas and follicular cell adenomas were reported in the thyroid in both sexes, moderate levels of islet cell adenomas occurred in the pancreas in male hamsters, high levels of cortical adenoma, carcinoma and subcapsular cell adenoma occurred in the adrenals of male and female hamsters, and we also noted a moderate incidence of pituitary adenomas in female hamsters. In addition, one endocrine adenoma of the adrenal cortex was noted in one female hamster in our 6-month study and adenoma and carcinoma of the adrenal cortex were noted in both male and female hamsters in our 12-month study. Pancreatic islet cell neoplasms are most common in cream hamsters, adrenal gland tumors most common in white hamsters and thyroid gland tumors most common in albino hamsters (Pour et al. 1979). Thyroid tumors were observed more often in white, male hamsters and adrenal tumors were noted in a higher incidence in female hamsters of all three strains (Pour et al. 1979). Van Hoosier and Trentin (1979) observed a moderate incidence of parathyroid adenomas in their study. Other workers have noted the preponderance of tumors in the adrenal cortex of the hamster (Russfield 1966). In two long-term studies, an inhalation and transplacental carcinogenicity study, respectively, about 35% of female hamsters and 55% of male hamsters developed tumors of the adrenal cortex (Dasenbrock et al.). In hamsters kept for life, adenomas and carcinomas of the adrenal gland are the most frequently observed tumors in both sexes and in the early stages of life (Kamino et al. 2001b).
Tumors of soft tissues include vascular tumors (hemangioma and hemangiosarcoma). These tumors are common in the hamster (Berman 1996) and were noted in the liver, spleen, teeth, colon, and skin in our 24-month studies. In addition, a hemangiosarcoma was noted in the skin of a female hamster in the 12-month study. Soft tissue tumors also include mesenchymal tumors such as leiomyomas, leiomyosarcomas, schwannomas, not otherwise specified (NOS) sarcomas, fibroma, and pleomorphic fibrosarcoma. These were noted in the thoracic cavity (malignant schwannoma and sarcoma, NOS), mesentery (pleomorphic fibrosarcoma), skin (fibroma, malignant schwannoma), and uterus (malignant schwannoma, leiomyoma, and leiomyosarcoma) in our 24-month studies. In addition, a malignant mesothelioma was observed in a male hamster in the abdominal cavity. Malignant mesothelioma has been reported in a Syrian hamster in the abdomen, testes, and liver (Adaska and Carbone 1994).
Tumors of bone include osteosarcoma and osteoma and three osteosarcomas and one osteoma were noted on our 24-month studies. Bone tumors are rare in hamsters and few chondromas have been reported in the hamster (Brown et al. 1993).
In our 24-month studies, the most frequent hematopoietic tumors included histiocytic sarcoma and malignant lymphoma. There was also a moderate incidence of malignant lymphoma in both male and female hamsters on our 24-month studies. Tumors of lymphoreticular/hematopoietic origin are among the most common spontaneous tumors in aged hamsters and appear to be the most frequent malignant tumor in the Syrian hamster (Barthold 1996). In a study of spontaneous tumors in domestic Syrian hamsters, plasmacytomas and malignant lymphomas were the most common tumors (Kondo et al. 2008) and Pour et al. (1976c) reported that malignant lymphomas developed in high incidences in both Eppley and Hannover colonies of Syrian hamsters. These tumors tend to occur in hamsters which are one year of age or older (Barthold 1996). An extramedullary plasmacytoma has been reported in the salivary gland of two Syrian hamsters (Munday et al. 2005).
No tumors of the nervous system were observed in animals on the 24-month studies in common with Pour et al. (1976c) and primary tumors of the nervous system of the Syrian hamster are reported to be extremely rare (Ernst et al. 1991). However, adenoma of the Harderian glands (12- and 24-month studies, male animals) and adenoma and adenocarcinoma of the lacrimal gland (24-month studies) were observed in small numbers in the hamsters on our studies. Pour et al. (1976c) observed Harderian gland adenomas in equal numbers in both Eppley and Hannover colonies of Syrian hamsters, with a predominance in male hamsters.
In conclusion, we report on the incidence of neoplasms in hamsters on 6-, 12-, and 24-month studies, and we advocate the use of the term ectopic C-cell hyperplasia/adenoma/carcinoma for the neuroendocrine cell hyperplasia/tumors noted at the laryngotracheal junction in hamsters. Background information about the incidence of tumors in Syrian hamsters on long-term studies is useful to both toxicologists and toxicological pathologists and will prove vital for decisions about the suitability of the Syrian hamster in long-term safety studies.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
