Abstract
Renal tubule lesions often prove troublesome for toxicologic pathologists because of the diverse nature and interrelated cell types within the kidney and the presence of spontaneous lesions with overlapping morphologies similar to those induced by renal toxicants. Although there are a number of guidance documents available citing straightforward diagnostic criteria of tubule lesions for the pathologist to refer to, most are presented without further advice on the when to or to the why and the why not of diagnosing one lesion over another. Documents presenting diagnostic perspectives and recommendations derived from an author’s experience are limited since guidance documents are generally based on descriptive observations. In this Regulatory Forum opinion piece, the authors attempt to dispel confusing renal tubule lesion terminology in laboratory animal species by suggesting histological advice on the recognition and interpretation of these complex entities.
Introduction
Toxicologic pathologists are often faced with the dilemma of how to recognize, diagnose, and interpret complex lesions in a variety of organs. Administration of test compounds may result in changes with overlapping morphological features or may mix with spontaneous background lesions further complicating the pathologist’s role in identifying induced lesions. This is particularly true for the kidney, which is composed of a heterogeneous population of cells and interrelated anatomic tubule segments with different morphological and functional capabilities. Furthermore, the renal nephron may respond quite differently to xenobiotics based on age, sex, and species of the animal to which it is delivered (Sellers and Khan 2005).
The kidney performs many sophisticated physiological processes and is largely responsible for maintaining the normal physiological balance of an individual (Sands and Verlander 2005). The proximal and distal convoluted tubule epithelial cells are responsible for performing many of these complicated processes, which include the major functions of absorption, secretion, metabolism, and acid–base equilibrium. Regardless of chemical class, nephrotoxicity may result in either direct or indirect tubule injury. In some cases, tubule cells may undergo degeneration, necrosis, regeneration, or cell proliferation all at the same time, depending on the test substance administered, dose, and the time on test. Therefore, it is expected that lesions involving the renal tubules often pose the greatest problem in the recognition and interpretation of renal toxicological injury in preclinical studies. While glomerular injury may also occur with xenobiotic administration, the spectrum of changes frequently noted in the glomeruli do not generally share this problem of simultaneous morphologic changes, confounding presence of spontaneous background findings, and/or vehicle effects, which frequently generate confusion for pathologists attempting to interpret these changes in tubules.
Recently, the combined Societies of Toxicologic Pathology published a comprehensive review of suggested nomenclature for the rat and mouse urinary system as part of continuing series of the International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice (INHAND; Frazier et al. 2012). Despite the availability of the INHAND document, other guidance documents (Chandra and Frith [1993] 1994; Hard et al. 1995; Hard et al. 1999), book chapters (Gopinath, Prentice, and Lewis 1987; Greaves 2012; Montgomery and Seely 1990; Short and Goldstein 1992; Wolf and Hard 1996; Seely 1999; Khan and Alden 2002; Gopinath and Mowat 2014), and websites (goRENI and National Toxicology Program [NTP] Atlas of Nonneoplastic Lesions) on similar aspects of kidney lesion identification and nomenclature, it was our impression that there remained some confusion regarding specific pathological tubule entities, particularly those with overlapping histomorphologies. As two of the authors of the INHAND document and from our own collective study and peer review experiences, we felt compelled to try to clarify and provide further guidance on some of these less straightforward pathologic entities. Therefore, it is the purpose of this article to present a brief overview of selected tubule lesions, to discuss their relevance, and more importantly, to present our own opinions concerning which terms should and should not be used in the context of rodent preclinical studies.
Our main objective for this document was to give less experienced pathologists and regulatory reviewers advice and information to help with their decision-making process. We accept that some pathologists, who are more experienced, may disagree with our criteria, recommendations, and opinions. We also understand there are situations that may need to have considerable latitude when diagnosing and interpreting nephrotoxicity. Additionally, our diagnostic criteria, recommendations, and opinions may necessitate change as newer insights and developments emerge. Finally, this opinion piece was not intended to be an extensive and comprehensive discourse and scientific review of the pertinent literature appropriate to the subject at hand. Therefore, we apologize to the many researchers whose scientific contributions are not recognized or were omitted in this article for the sake of brevity.
Basophilia, Tubule: Often Seen and More Often Overused
Changes in the cytoplasmic tinctorial characteristics of proximal renal tubules are one of the most frequently observed manifestations of toxic injury in preclinical toxicity studies. Tubule basophilia is a morphological feature of several changes of the renal tubules in all species (Figures 1 and 2). Although tubule basophilia is one of the hallmark features of spontaneous chronic progressive nephropathy (CPN) in rodents (Gray, Zwieten, and Hollander 1982; Owen and Heywood 1986; Peter, Burek, and van Zwieten 1986), it may also represent persistent low-grade toxicologic insult to tubule cells or may reflect reparative processes when the cell is in the recovery phase (Frazier and Seely 2013).
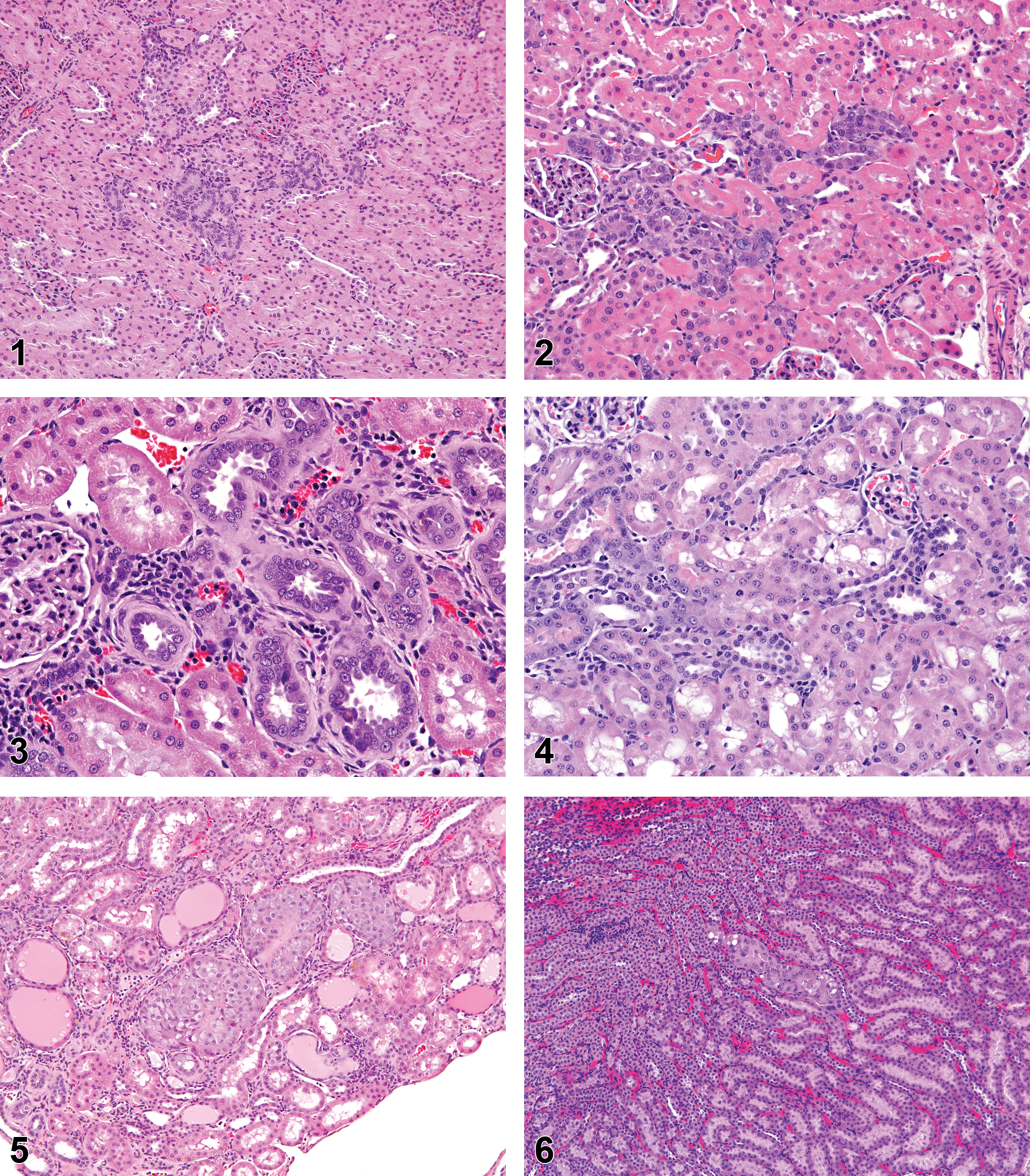
Young male rat. Basophilic appearing tubules observed in the renal cortex. This lesion could be spontaneous or induced and the diagnosis of “Basophila, Tubule” appropriate (hematoxylin and eosin).
Even in nonrodents, tubule basophilia is not specific to a single process. In dogs, minimal focal tubule basophilia can be noted in a large percentage of control beagles but may also be an early manifestation of tubule degeneration (Frazier and Seely 2013). In minipigs, tubule basophilia is one of the most common spontaneous lesions noted in control kidneys (Jeppeson and Skydsgaard 2014), but it is also the primary manifestation of degenerative kidney lesions associated with nephrotoxicants in pigs and may be the only lesion noted at exposures that induce tubule necrosis in other species. Only with persistent or more severe tubule injury in pigs is there evidence of overt tubule necrosis or eventual fibrosis if the underlying basement membrane is disturbed (Swindle et al. 2012). Hence, the diagnostic term “Basophilia, Tubule,” while histologically descriptive and accurate, and a preferred term for INHAND and Standard for the Exchange of Nonclinical Data (SEND; Frazier et al. 2012; Keenan and Goodman 2014), if used indiscriminately, can be potentially confusing to regulatory reviewers, because it can denote either a spontaneous lesion, a degenerative lesion, or a regenerative lesion from previous renal insult. However, clarity can be achieved in most cases, using the appropriate terminology in the proper context of a study by describing the change and its interpretation thoroughly in the pathology narrative report.
Tubule basophilia, as one single component of more complex renal injury, is frequently noted in direct cytotoxic kidney damage, in CPN, in obstructive or retrograde nephropathies, in cases of pyelonephritis, or occasionally with renal phospholipidosis. In these situations where tubule basophilia is a single component of more involved renal pathology (which often encompass a host of morphologic terms), the use of “Basophilia, Tubule” should be discouraged in favor of a more overarching terminology. Rather than list a litany of morphologic diagnoses in tables, which would include basophilia and all of the other constellation of lesions accompanying the particular type of injury, a bridging term such as CPN, obstructive nephropathy, or pyelonephritis can be utilized. These terms, while diagnostic rather than the more preferred descriptive terminology commonly applied to toxicologic lesions, are consistent with current renal INHAND terminology and are included in SEND regulatory terms, based on their commonly recognized morphology and mechanism. In other cases, where no standard or preferred overarching terminology is available, more general terms such as “degeneration” might be more applicable, as “degeneration” covers a variety of other tubule changes occurring in parallel. However, in some studies, there will be a range of lesions noted that vary significantly from one animal to another, where tubule basophilia is present as the major lesion in some animals with necrosis or other types of degeneration or pathology in other animals, perhaps varying from dose to dose. In such cases, pathologists should have the latitude to utilize specific terms such as “tubule basophilia” to demonstrate the continuum of lesions noted from animal to animal and to highlight the lower end of this spectrum.
CPN: Old Condition—Newer Reflections
Historically, morphological components of a peculiar “nephrosis” were recognized as far back as the 1920s in laboratory rat studies examining diet modification (Polrogt, McCollum, and Simmonds 1923; Newburgh and Curtis 1928). In retrospect, we realize that these investigators had been describing what we now recognize as CPN. CPN is one of the most commonly diagnosed rodent renal diseases and its morphological features have been extensively reviewed (Hirokawa 1975; Gray 1977; Barthold 1998, Goldstein, Tarloff, and Hook 1988; Hard and Khan 2004). Usually, advanced stages of CPN pose little diagnostic challenge for most pathologists when the entire morphological spectra of tubule, glomerular, and basement membrane changes are present. Identification of regenerative tubules lined by thickened basement membrane material and tubule hyaline casts are usually enough to make a CPN diagnosis. However, CPN terminology becomes problematic in its earliest stage when changes associated with CPN occur in young rats, particularly those below 12 weeks of age. In these cases, tubule basophilia is often evident without any other components of CPN such as conspicuously thickened basement membranes, nuclear crowding, tubule hyaline casts, or variable mononuclear cell infiltrates (Figure 3). In the case of only CPN-related tubule basophilia, it is appropriate to use “Basophilia, Tubule” as a diagnostic term. In most cases, if the incidence is not test article–related, this should be interpreted as an early indication of CPN. However, tubule basophilia closely associated with other morphological components of CPN should be diagnosed as CPN.
As CPN progresses in severity, complex tubule changes develop, which are somewhat different from typical CPN-related regenerative tubules. These diagnostically challenging tubule changes appear as dilated or cystic, plaque-like or solid, or proliferative tubules (Hard and Seely 2005, 2006). However, with careful examination, these tubules can be identified as part of the CPN complex by their location within foci of CPN and association with prominent basement membrane material. These changes should be recognized as a component of CPN and, consequently, not diagnosed separately. Although some pathologists may be tempted to “split out” individual morphological components of CPN in an attempt to identify chemically induced nephrotoxicity in routine safety studies, this practice is strongly discouraged because of the highly variable and age-dependent aspects of CPN in control and exposed male and female animals.
A well-known effect of compound administration is the potential exacerbation of the incidence and/or severity of CPN in toxicity studies (Hard and Khan 2004; Travlos et al. 2011). Exacerbation of CPN may prohibit the pathologist from identifying other morphologically distinct changes unrelated to CPN. A more challenging situation arises when a chemical entity produces dose responsive effects in the kidney that resemble CPN in some, but not all, respects. Frequently in these cases, there is a notable lack of alteration in the basement membrane, tubule lesions are more widely distributed throughout the kidney and the presence of hyaline casts may be reduced or absent. The toxicologic pathologist on such a study may recognize that some lesions are CPN-related and some are test article–related by the subtle differences from spontaneous disease, but is confronted with a vast array of choices in terminology. CPN, obviously, is not an appropriate term for the entire lesion; and if used by itself, inappropriate or incorrect conclusions regarding risk assessment may be inferred. However, if changes present in the cortical tubules as a whole which might include vacuolation, swelling or cytoplasmic rarefaction in some tubule cells, rare single cell necrosis, or occasional sloughed epithelium in a few foci, inclusion of multiple morphologic diagnoses to the tables becomes cumbersome. The use of multiple diagnoses may also be difficult for regulatory reviewers to assimilate or to assign relevance to, so a lump diagnosis of “degeneration” may be more preferred terminology. The general term of “nephropathy” to denote such xenobiotic lesions should not be used, in order to avoid confusion with CPN.
Degeneration or Regeneration: The Conundrum of “Generation”
While tubule basophilia is one of the most obvious microscopic features of tubule degeneration and regeneration, tubule basophilia should not be construed as always denoting a reparative response and should not be used as a diagnostic term when aspects of regeneration are clearly evident. Tubule degeneration, in addition to having features of tubule basophilia, may be apparent as one or more of the following features: tubule dilation, cytoplasmic swelling or vacuolation, cytoplasmic condensation and eosinophilia, nuclear pyknosis, and cell sloughing. Therefore, when some of these features accompany tubule basophilia, degeneration may be the predominant tissue reaction and “regeneration” is probably not the appropriate terminology.
Following acute injury, acute regeneration may occur, which is characterized by initial flattening and proliferation of the lining tubule epithelial cells to cover an injured portion of the tubule. As previously noted, this may be accompanied by tubule basophilia in these or adjacent tubule cells. Ongoing regeneration is characterized by increased cell number (recognized morphologically by conspicuously increased mitoses or as nuclear crowding), heterogeneity in cell size or shape, or an increase in the nuclear-to-cytoplasmic ratio (Frazier et al. 2012; Figure 4). When such features are found in tubules, the diagnosis of “Regeneration, Tubule” is straightforward and preferred. The interpretation and use of terminology can become more complex when there is little difference in the qualitative assessment of tubule changes between end of treatment and end of recovery phases. In such a situation where lesions are primarily limited to cytoplasmic basophilia either at terminal necropsy or after recovery necropsy, it may not be apparent that tubule basophilia represents a true repair response unless there is a significant reduction in lesion severity. The diagnostic term “Regeneration, Tubule” in this situation becomes less informative, and use of the term “Basophilia, Tubule” might be equally applicable. Choice of the appropriate terminology rests with individual pathologist preference and whether there are sufficient other findings present to warrant a diagnosis of regeneration.
In our experience, the most confusing situation for the toxicologic pathologist in regards to kidney nomenclature arises with the presence of renal tubule regenerative responses noted during the treatment phase of a toxicity study. The presence of cytoplasmic basophilia, cell heterogeneity, flattening, and/or mitoses during treatment phases typically indicate regeneration. This could suggest either continuous cycles of low-grade injury and repair or earlier previous injury with the cell accommodating to biochemical insult and reaching some form of homeostasis with active induction of repair processes while still exposed to drug. Many toxicologists or pathologists have used the term degeneration/regeneration as a morphologic term in such cases. To be completely accurate, if morphologic features of both degeneration and regeneration are present, then perhaps it is better to diagnose both to bring attention to the ongoing tubule injury and repair processes.
Tubule Hyperplasia or Atypical Tubule Hyperplasia (ATH): What’s the Big Deal?
Dorland’s illustrated medical dictionary defines hyperplasia as an abnormal multiplication or increase in the number of normal cells in normal arrangement in a tissue (Dorland 2003). Consequently, this definition could be used to denote a number of tubule lesions observed in rodent kidney sections from preclinical studies. Hyperplasia, in general, also conveys the idea that the lesion may become neoplastic. However, distinguishing between regenerative, nonneoplastic, and preneoplastic hyperplasia is crucial in assessing risk and for regulatory decisions involving carcinogenicity of a lesion (Boorman et al. 2003). Actually, in rodents, the term “Hyperplasia, Tubule” has been used to diagnose all of the above types of hyperplasia by pathologists based on their training, experience, and limitations of laboratory derived lexicons.
Hard et al. (1995) in an earlier guidance document suggested that 2 types of tubule hyperplasia, simple tubule hyperplasia and ATH, were present in the rat. Simple tubule hyperplasia implied a single layer of an increased number of tubule lining epithelial cells contained within the basement membrane while ATH was characterized as a complex proliferation of “atypical” cells beyond the normal single cell layer (Figure 5). Although seemingly straightforward, neither term achieved widespread acceptance leaving the generic term “Hyperplasia, Tubule” to be used to denote changes associated with CPN, regenerative hyperplasia, or nonneoplastic and preneoplastic hyperplasia.
In the INHAND urinary system document, recommendations for tubule hyperplasia and ATH are clearly outlined (Frazier et al. 2012). Therefore, in keeping with the INHAND document, it is recommended that the diagnostic term “Hyperplasia, Tubule” be used only to denote a nonneoplastic proliferation of tubule epithelial cells unrelated to other pathologic entities in the kidney (e.g., CPN, obstructive or retrograde nephropathy) while “ATH” is reserved for those lesions, which are considered to have preneoplastic potential. In the 2-year carcinogenicity studies, rats with advanced CPN will consistently have small foci of simple tubule hyperplasia, and the inclusion of these diagnoses in tables is neither informative nor helpful in determining human carcinogenic risk. In contrast, the inclusion of ATH lesions in tables, especially where unrelated to CPN, may be relevant to human risk assessment.
Correlations with Renal Biomarkers: Moving away from the Microscope
With the recent development of several urine analytes that have shown promise as renal biomarkers of nephrotoxic injury, it is important to mention whether the various types of tubule changes described previously will have correlative clinicopathologic alterations. In fact, histologic changes of CPN, tubule basophilia, tubule degeneration, or tubule regeneration in the cortex may all be associated with slight to moderate increases in urinary albumin, kidney injury molecule-1, neutrophil gelatinase-associated lipocalin/lipocalin 2, osteopontin, and clusterin (CLU; Bonventre et al. 2010; Harpur et al. 2011; Ozer et al. 2010). If the medullary collecting ducts are affected, there may be changes in renal papillary antigen-1 in the rat (Betton et al. 2012). CLU, in particular, has shown more consistent (but still variable) results in demonstrating regeneration of renal tubules. The magnitude of changes in the rat or mouse with any of these 5 markers will vary tremendously according to the degree of injury and may only be reflected in 3- to 5-fold increases when standardized to creatinine when a small percentage of tubules are affected. The dynamic range and magnitude of elevations are not likely to be nearly as significant as with tubule necrosis. When only minimal or focal lesions are noted, such as in early CPN, urinary analytes are likely to fall within normal ranges. It should also be noted that these biomarkers are likely not as helpful in chronic studies. While systematic evaluation has not been performed, individual nonpublished studies have shown several of these tend to slowly decrease from maximum release after a month of treatment (D. Ennulat, personal communication, September 15, 2014). Importantly, it is not possible to definitively differentiate these types of lesions (CPN vs. basophilia vs. regeneration) based on their biomarker profile alone.
Amphophilic-vacuolar (AV) Proliferative Lesions: Something to Get Excited About
The presence of genetically derived renal tumors in the rat is well established. At least 2 animal models exist, the Eker rat and the Nihon rat (Eker 1954; Okimoto et al. 2000). Hyperplastic and neoplastic tubule lesions postulated to be of familial origin have been reported from conventional rat 90-day toxicity studies (Hard et al. 1994; Hall et al. 2007) and from littermates (Thurman et al. 1995). Hard et al. surveyed the NTP database for the spontaneous occurrence of similar distinctive tumors and proposed the name “amphophilic-vacuolar” to distinguish these proliferative lesions from other more common rat proliferative lesions (Hard et al. 2008). More recently, these tumors were surveyed from several laboratories and confirmed that AV tumors arose spontaneously and could be observed in rats as young as 7 to 10 weeks of age (Crabbs et al. 2013).
AV proliferative lesions are characterized by the presence of large, finely granular cells that stain predominantly eosinophilic to amphophilic and contain numerous cytoplasmic vacuoles (Figure 6). Tumors are distinctly identified by their typically solid lobular growth, intervening tracts of fibrovascular stroma, and often lymphoid cell infiltration. Cystic and more basophilic variants are more uncommon (Hard et al. 2008).
Obviously, it is important to recognize AV lesions and report them separately from conventional proliferative lesions since any chemically or xenobiotic-induced reports of AV lesions, to our knowledge, have not been demonstrated.
Regulatory Review: Why Standardization Is Important
For the pharmacology/toxicology regulatory reviewer, it can be extremely difficult to appreciate the subtle connotative distinctions between a drug that causes tubule degeneration, exacerbates CPN, or is associated with renal tubules undergoing regeneration. Prior to INHAND standardization, it was common for differences in terminology between and even within organizations. The same compound may have had different terms applied to similar lesions even from study to study. Consistency in terminology, based on the appropriate mechanism of injury or repair as described previously, should allow regulatory colleagues to understand intuitively which lesion has more or less clinical risk. Regenerative tubules imply partial resolution, which lends a much different perspective than basophilia involved with degeneration alone. CPN has little direct clinical relevance, but exacerbated CPN may still be indicative of low-grade nephrotoxic potential. As noted previously, simple hyperplasia is much less concerning than atypical forms. It should be clear then that each of the different manifestations of tubule alterations elicit a slightly different risk assessment. It is therefore imperative that standardized nonclinical terminology purveys the appropriate message and applicable level of clinical risk so that clinicians and regulatory colleagues are not confused by overlapping or vague nomenclature.
Conclusion
Lesions of the renal tubule can be challenging to diagnose and interpret, particularly when multiple simultaneous processes are occurring. However, there are multiple references available to the toxicologic pathologist to assist in providing the proper terminology and interpretation. Tubule basophilia, degeneration, regeneration, and CPN can all be overlapping and without care in usage of terms, confusion may arise among reviewers, clinicians, or regulatory agencies.
Footnotes
Author Contribution
All authors (JS, KF) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
This Regulatory Forum opinion piece is dedicated to Dr. Gordon C. Hard for his many significant contributions to renal toxicologic pathology.
*This is an opinion article submitted to the Regulatory Forum and does not constitute an official position of the Society of Toxicologic Pathology or the journal Toxicologic Pathology. The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of their respective agencies and organizations. The Regulatory Forum is designed to stimulate broad discussion of topics relevant to regulatory issues in Toxicologic Pathology. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by Experimental Pathology Laboratories, Inc. and GlaxoSmithKline. The authors wish to specifically thank the members of the Regulatory Forum committee for their input, assistance, and encouragement.
