Abstract
Alloxan-induced diabetic rats showed proliferative changes in the forestomach, accompanied by chronic inflammation, and one lesion progress to squamous cell carcinoma (SCC) without distant metastasis. The authors demonstrated that these lesions might be caused by Candida albicans infection. Antimicrobial therapy, particularly tetracycline treatment, has been blamed for a reduction in the number of competing bacterial organisms, which is frequently mentioned as a cause of candidiasis. The objective of this study is to ascertain whether or not tetracycline treatment can accelerate early-onset of C. albicans infection and the proliferative changes in this diabetic model. Alloxan-induced diabetic rats were given chlorinated water (AL group) and tetracycline solution (0.1% during week 1 and 0.01% thereafter) as drinking water (AT group). They were sacrificed after 25 weeks of drinking the treated water. The infection rate with C. albicans in the AT group was significantly higher than in the AL group. The incidence and severity of the squamous cell hyperplasia were enhanced in the AT group compared to the AL group. The proliferative lesions were consistently accompanied by inflammation and C. albicans infection in both groups. SCC was detected in one case in the AT group. These findings demonstrate that tetracycline induces C. albicans infection and enhances forestomach proliferative lesions in alloxan-induced diabetic rats.
Introduction
Multiple cancers frequently occur in the upper digestive tract (Berg, Schottenfeld, and Ritter 1970; Vrabec 1979). One possible explanation is that specific infection stimulates the normal epithelium to initiate inflammation and/or promotes carcinogenesis. Bacteria, particularly Helicobacter pylori, are strongly related as etiological factors in stomach cancers. However, there is less evidence for an association between fungi and cancer. A possible association between Candida colonization and alimentary tract neoplasia was reported (Cawson 1969), and Candida albicans was much more frequently detected in the biofilm of squamous cell carcinoma (SCC) than in normal regions (Nagy et al. 1998). Earlier human studies also showed a statistically significant association between fungal invasion and epithelial dysplasia (Barrett, Kingsmill, and Speight 1998; Roed-Petersen, Renstrup, and Pindborg 1970).
Our previous study reported that aged alloxan-induced diabetic rats frequently have proliferative lesions of the squamous epithelium in the forestomach and that those lesions progress to SCC (Kodama et al. 2006). Proliferative changes were regularly accompanied with a severe inflammation of the mucosal epithelium along with an infection with filamentous fungi and/or bacterial colonies. By fungal and bacterial culture, they were identified as C. albicans, Lactobacillus sp., Bacillus licheniformis, and Escherichia coli, respectively (Kodama et al. 2006). The antifungal agent itraconazole reduced the degree of the mucosal hyperplasia, inflammatory changes, and Candida infection in alloxan-induced diabetic rats (Sano et al. 2009a). Therefore, C. albicans infection was an important factor in the pathogenesis of mucosal proliferation and inflammation (Kodama et al. 2006; Sano et al. 2009a, 2009b).
The major Candida species responsible for upper alimentary tract infection is C. albicans, a polymorphic organism that resides as a commensal in the human gastrointestinal tract (Walsh and Merz 1986). When the balance of the normal flora is disrupted or the immune defenses are compromised, Candida species can become pathogenic. It was well known that antimicrobial therapy, particularly tetracycline treatment, is frequently mentioned as a cause of human oral candidiasis and induces similar lesions in C. albicans-inoculated experimental animals (Samaranayake and Samaranayake 2001; Seelig 1966). The objective of this study was to ascertain whether only tetracycline treatment can cause the early-onset of C. albicans infection and induction of proliferative changes in alloxan-induced diabetic rats.
Materials and Methods
Animals and Diets
Female WBN/Kob rats were obtained from Japan SLC, Inc. (Shizuoka, Japan). They were reared in a barrier-sustained animal room maintained at a temperature of 24 ± 2°C and a relative humidity of 60% ± 20%, with 12-hr light/dark cycles and ventilated at least 12 times/hr by sterilized fresh air. All the rats were housed and reared in aluminum mesh cages. To protect against infection, the cages were changed once or more each week. Rats were given a pelleted diet (CRF-1; Oriental Yeast, Tokyo, Japan). All procedures for animal handling and experimental treatments were in accordance with the Guidelines for the Care and Use of Laboratory Animals of the Committee for Animal Experiments of Setsunan University and the Japanese Association for Laboratory Animal Science.
Glucosuria and Glycemia Monitoring
Fresh urine samples were collected in metabolism cages. Urinary glucose levels were measured semiquantitatively, using a urine test paper (Wako Pure Chemical Industries, Osaka, Japan) every day from day 1 to day 3 after alloxan dosing, once every week for 1 month after the first week, and once every month thereafter, from the fresh urine obtained from alloxan-induced diabetic rats. Blood glucose levels were also measured semiquantitatively by the glucose oxidase method (Glutest E; Sanwa Kagaku, Aichi, Japan) once every month from the fourth week after dosing, using blood samples from the tail vein. Samples of blood from the tail vein and fresh urine were collected from 1:00 to 4:00 p.m.
Experimental Design
A total of thirty female WBN/Kob rats were divided into two groups at 10 weeks of age. All rats, aged 10 weeks, were given a single dose of alloxan (Sigma-Aldrich Japan, Tokyo, Japan) via the tail vein at a dosage level of 40 mg/kg body weight. The concentrations were set up as a given dose according to which a rat survives for a long period of time after developing signs of diabetes and which induces continuous glucosuria.
The rats in the alloxan-treated group (AL group) were given chlorinated water ad libitum. The alloxan- and tetracycline-treated rats (AT group) received a 0.1% tetracycline hydrochloride solution as drinking water ad libitum during week 1 of the study and this was reduced to a 0.01% solution thereafter. The rats were sacrificed at the 35th week of age.
Histopathological Analysis
One moribund animal (a rat of the AL group) was sacrificed at 13 weeks of age. The remaining twenty-nine rats were killed by exsanguination from the abdominal aorta under deep anesthesia at the end of each scheduled period. The entire alimentary tract was immediately removed following necropsy. The organs of the thirty rats were immersed in 10% phosphate-buffered formalin solution immediately after necropsy.
Fixed organs were trimmed, dehydrated by automated processor, and embedded in paraffin wax. Sections (4 µm thick) of tissue specimens were stained with hematoxylin–eosin for histopathological examination.
Special staining methods such as a Gram stain for general pathogens were carried out on all the sections.
Immunohistochemical Analysis
Immunohistochemical confirmation of C. albicans and cell proliferation was conducted for representative forestomach sections. The sections were deparaffinized in xylene and rehydrated through graded ethanol at room temperature. Rehydrated sections were digested by pepsin for 20 min at 37°C to retrieve antigen. Solutions and washes were prepared between the various steps using 0.05 M Tris buffer saline (TBS, pH 7.6) with 0.01% Tween 20. Nonspecific endogenous peroxidase activity was blocked by exposure to 0.03% hydrogen peroxide in 100% methanol for 5 min and masking was conducted with 1% bovine serum albumin in phosphate-buffered saline for 5 min at room temperature. Incubation was carried out overnight at 4°C with anti–C. albicans (diluted 1:400, MAB806; Chemicon, Temecula, CA, USA) mouse monoclonal antibody or anti-Ki67 rabbit monoclonal antibody (diluted 1:500, SP6; Epitomics, Burlingame, CA, USA). The slides were subsequently rinsed with TBS plus Tween 20, treated for 30 min at room temperature with Histofine simple stain rat MAX PO (M) (Nichirei, Tokyo, Japan) or a stain system kit DAKO LSAB 2 kit/HRP (DAKO Japan, Kyoto, Japan), rinsed with TBS plus Tween 20, incubated in diaminobenzidine solution containing 0.01% hydrogen peroxide for the peroxidase coloring reaction, and counterstained with Mayer’s hematoxylin. Staining was negatively controlled by substituting the mouse isotype immunoglobulin, diluted to the same concentration, for the primary antibody.
Statistical Analysis
Fisher’s exact test and the unpaired Student’s t test were used for statistical analysis of the body weight data.
Results
General Conditions and Monitoring of Glucosuria and Glycemia
One rat in the AT group showed only transiently high blood glucose levels after alloxan dosing, this animal showed normoglycemia (blood glucose <120 mg/dL) during the experimental period. In all the remaining alloxan-treated rats, severe hyperglycemia (>300 mg/dL) and glucosuria (>500 mg/dL) continued for 25 weeks from the day of alloxan injection to the time of scheduled necropsy or being found moribund. The body weights of all the rats in the AL and AT groups decreased within several days of injection, and the average body weight of these two groups were 182.1 ± 15.3 g and 187.9 ± 14.8 g, respectively. One rat of the AL group was necropsied within 13 weeks of dosing because it became moribund.
Histopathology
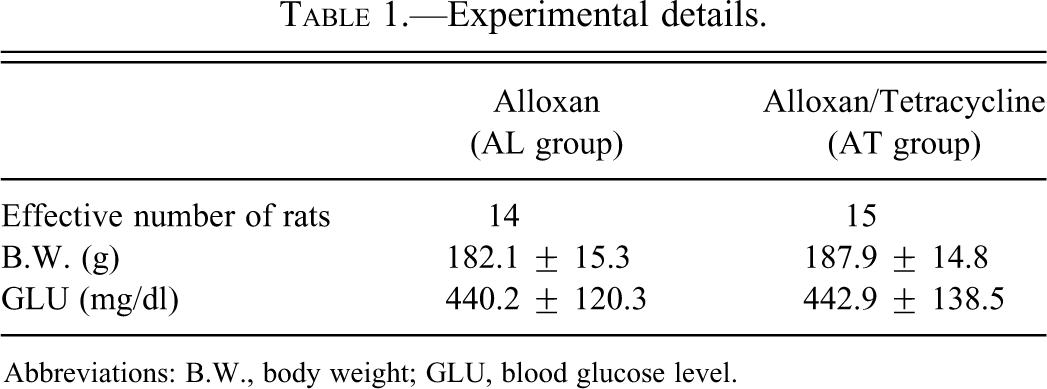
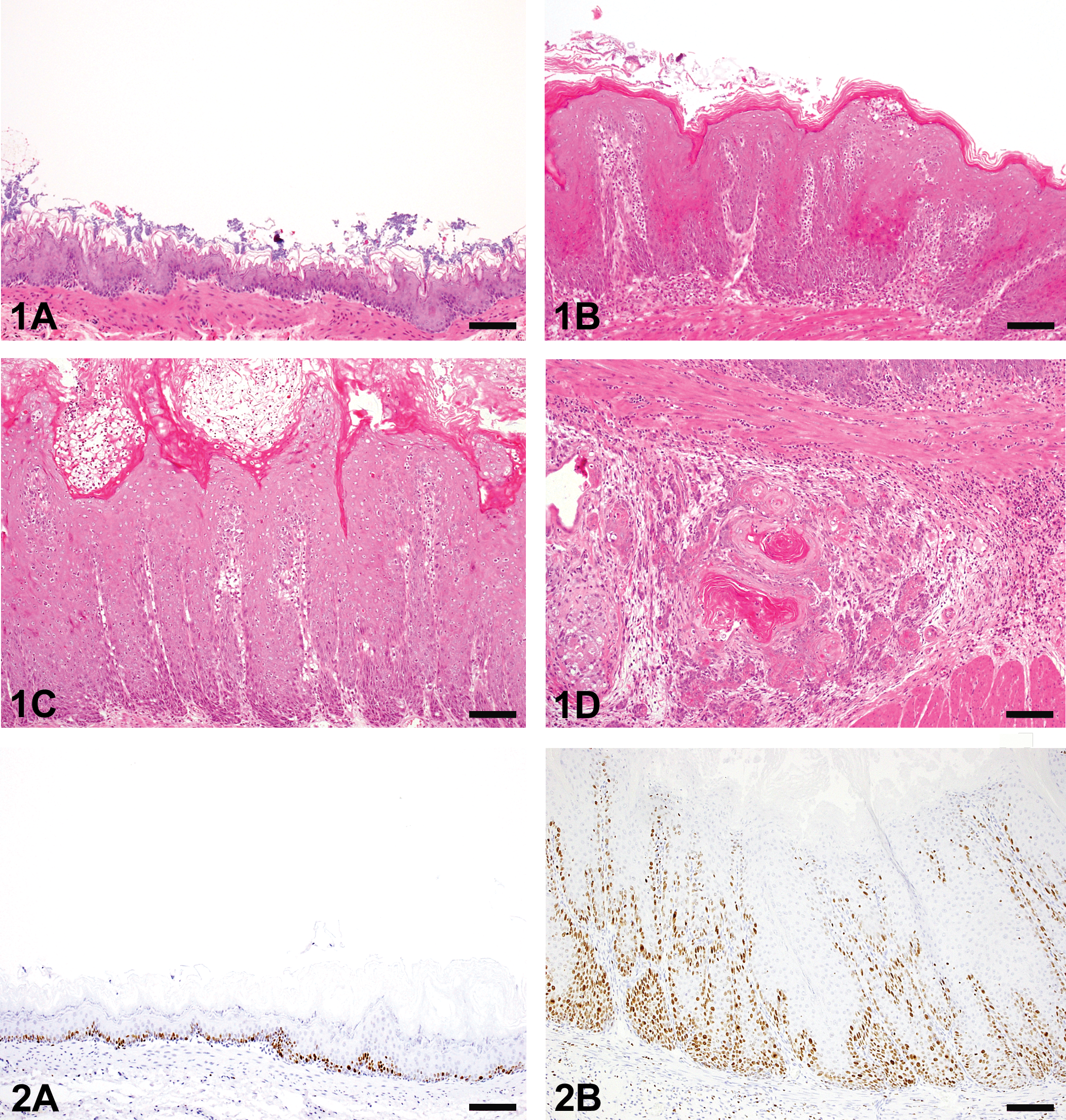
Squamous hyperplastic change in the forestomach was detected in both the AL and the AT groups (Table 1). The incidence and severity of the proliferative lesions were significantly enhanced in the AT group compared to the AL group (Table 2). The proliferative changes were only detected in the forestomach of 4 rats (28.6%) in the AL group (Figure 1A, B), whereas almost all the rats (14/15, 93.3%) in the AT group developed squamous cell hyperplasia to varying degrees and this progressed to SCC in 1 case (Figure 1C, D; Table 2). SCC showed only local invasion in the submucosa and the metastatic focus was not detected by macroscopic examination. Two types of inflammatory change were observed in hyperplastic mucosa (Figure 1B, C). Accumulation of neutrophils and/or mucosal ulceration invariably accompanied with the hyperplastic mucosal epithelium and some ulcerative lesions reached the submucosal layer. Lymphocytes and plasma cells were also observed in lamina propria and submucosal layer. Squamous cell hyperplasia and inflammatory changes were most prominent on the side of the limiting ridge of the stomach.
Experimental details.
Abbreviations: B.W., body weight; GLU, blood glucose level.
(A) Normal forestomach mucosa in rats of the alloxan-treated group (AL) group. Scale bar, 200 µm. (B) Slight hyperplasia of mucosal squamous epithelium in rats of the AL group. Neutrophils accumulate in mucosal epithelial layer, and varying numbers of lymphocytes infiltrate throughout the entire submucosal layer. Scale bar, 200 µm. (C) Severe hyperplasia of forestomach mucosal squamous epithelium in rats of the alloxan- and tetracycline-treated rats (AT) group. Scale bar, 200 µm. (D) Squamous cell carcinoma (SCC) of the forestomach in the AT group. Scale bar, 200 µm.
Histopathologic findings in upper alimentary tract.
The severity of the proliferative lesions in the forestomach squamous epithelium was judged from the thickness of the mucosal epithelium: +, slight change; ++, moderate change; +++, severe change.
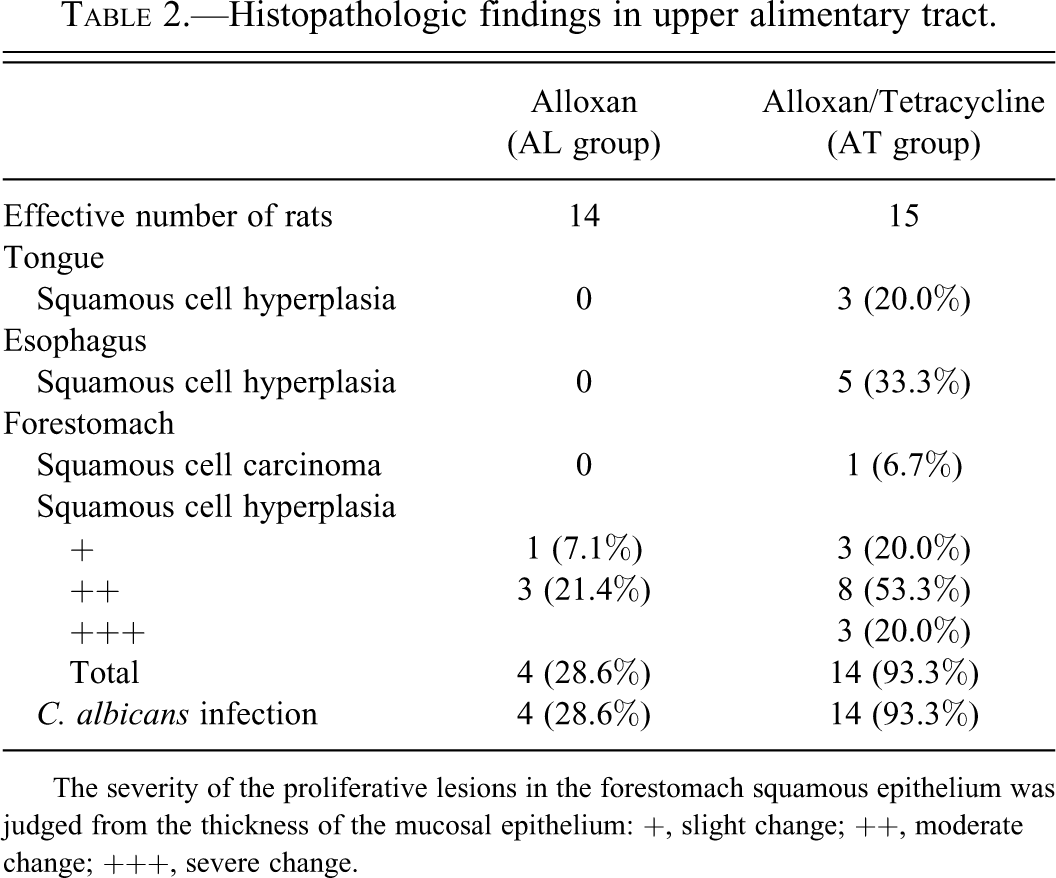
The Ki67-positive cells of the forestomach were increased with the severity of the hyperplasia in both the groups (Figure 2A, B). In organs other than the forestomach, the proliferative change in the mucosal squamous epithelium was only observed on the tongue (20.0%) and esophagus (33.3%) of the AT group (Figure 3A, B; Table 2).
(A) Hyperplasia of tongue mucosal squamous epithelium in rats of the alloxan- and tetracycline-treated rats (AT) group. Inflammatory cells accumulate from mucosal epithelial layer to the submucosal layer. Scale bar, 200 µm. (B) Hyperplasia of esophagus mucosal squamous epithelium (arrow) in rats of the AT group. Scale bar, 500 µm.
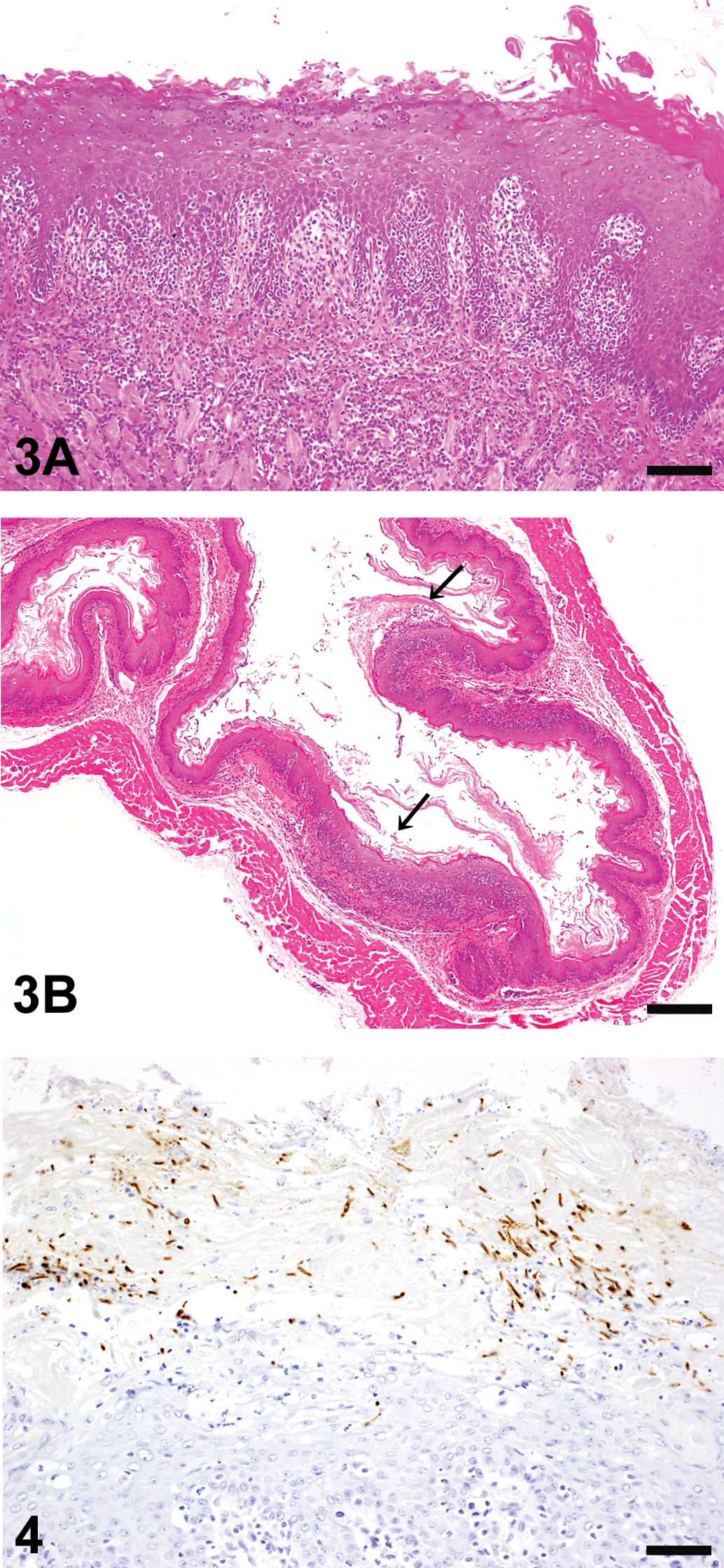
Ulcerated mucosal epithelium with bacterial or fungal infection was observed in both the groups. Filamentous fungi showing dimorphism such as yeast forms and mycelial forms were also frequent in the superficial mucosal layer in almost all the diabetic rats of the AT group (14/15, 93.3%) and 4 rats of the AL group (28.6%). All of the fungi in the hyperkeratotic lesions of the mucosa were positive for C. albicans antibody in both the groups (Figure 4). In addition, rod-shaped gram-positive bacteria were also seen on the mucosal surface in both the groups with or without hyperplasia. A nondiabetic rat in the AT group and the ten diabetic rats of AL group without hyperplastic mucosa showed neither hyperplastic mucosal changes nor fungus infection in the forestomach.
Discussion
We have reported in our previous report that long lasting diabetic conditions accelerated by alloxan-dosing induced proliferative changes in the mucosal epithelial cells that progressed to SCC in an inbred strain of rats (WBN/Kob) and recommended this experimental system as a suitable model for the study of the chronic inflammation-related promotion of carcinogenesis. However, induction of these severe proliferative changes or SCC took at least a year in our previous reports (Kodama et al. 2006; Sano et al. 2009a, 2009b). The present study revealed that treatment with tetracycline alone induced C. albicans infection and enhanced mucosal proliferation in the upper alimentary tract (tongue, esophagus, and forestomach) in alloxan-induced diabetic rats.
Antibiotic therapy, particularly tetracycline therapy, is frequently mentioned as a cause of human oral candidiasis (Samaranayake and Samaranayake 2001; Seelig 1966). Furthermore, a previous in vivo study showed that the tetracycline solution apparently affected the size of the lesions that were caused by C. albicans inoculation, while it demonstrated no significant differences in the number of animals infected both grossly and histologically (Allen et al. 1985). The role of tetracycline treatment was explained as being that reduction in the number of competing bacterial organisms is thought to allow an overgrowth of the yeasts and result in an overt fungal infection (Helstrom and Balish 1979). In our study, the incidence and severity of the mucosal proliferative lesions in the AT group were apparently high in comparison to those in the AL group. However, the results of special staining in our study did not clarify the alterations in the bacterial flora of the forestomach mucosa between the AL and AT groups. The clear role of the treatment with tetracycline is unknown, but tetracycline treatment must aid in the induction of candida infection. In addition, uncontrolled diabetes mellitus is well known to predispose individuals to oral candidiasis (Bartholomew, Rodu, and Bell 1987). Streptozotocin-induced diabetic rats given a single lingual inoculation of C. albicans remained positive for the yeast throughout the 40-week study (Dourov and Coremans-Pelseneer 1987). Hyperglycemia limits neutrophil function including phagocytotic ability. With the oxidative killing ability of the neutrophils hindered, diabetics may not be able to clear pathogens as well as nondiabetics (de Leon et al. 2002; Nagy et al. 1998; Wilson et al. 1987). Furthermore, a depletion of memory CD4+ cells was also described in diabetics (Moutschen, Scheen, and Lefebvre 1992). Likewise, in diabetic mice, it was shown that a depression in primary and secondary immune responses was due to altered T-cell function and phagocytic activity in macrophages (Saiki et al. 1980). Thus, we believe tetracycline treatment and long lasting diabetic conditions induced earlier and more severe lesions in this study.
C. albicans infection–related proliferative changes have a propensity to undergo malignant transformation. A recent epidemiological study suggested a significant correlation between oral squamous cell carcinoma and the degree of oral yeast carriage (McCullough et al. 2002). C. albicans was much more frequently detected in the biofilm of oral SCC (Bhatia et al. 1989; Nagy et al. 1998) and might also lead to carcinoma in epidemiological and in vitro studies (Renstrup 1970; Sitheeque and Samaranayake 2003; Williams et al. 2001). Some C. albicans biotypes were able to produce the potent carcinogen N-nitrosobenzylmethylamine, which has been reported to be capable of inducing tumors in the tongue, esophagus, and forestomach of rats (Krogh, Hald, and Holmstrup 1987; Lijinsky et al. 1982). Another researcher has suggested that infection by C. albicans is more important for carcinogenesis, because it has a promoter function in relation to the 4-nitroquinoline-1-oxide (4NQO)–induced oral mucosal tumors (O’Grady and Reade 1992). As well as histological evidence of dysplasia in the mice exposed to both 4NQO and C. albicans, and there was increased Ki-67 and p16 expression in comparison with mice treated with 4NQO alone or C. albicans alone, suggesting that Candida may lead to genetic alterations (Dwivedi, Mallya, and Dongari-Bagtzoglou 2009). Ki-67 is a cell cycle–associated nuclear protein, used as a proliferation marker and linked to malignancies (Schluter et al. 1993). In our study, the Ki67-positive cells of the forestomach increased with the severity of the hyperplasia. These data supported our previous study and demonstrated that C. albicans infection plays an important role in the transition from normal to hyperplastic mucosa.
It became clear that tetracycline treatment can easily cause C. albicans infection and enhance the Candida-related mucosal proliferation in the upper alimentary tract (tongue, esophagus, and forestomach) in alloxan-induced diabetic rats. Furthermore, alloxan-induced diabetic rats treated with tetracycline are useful for identifying the risk of C. albicans infection–related carcinogenesis under the diabetic condition.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
