Abstract
Differences in cancer incidences between men and women are often explained by either differences in environmental exposures or by influences of sex hormones. However, there are few studies on intrinsic gender differences in susceptibility to chemical carcinogens. We have analyzed the National Toxicology Program (NTP) database for sex differences in rat responses to chemical carcinogens. We found that the odds that male rat bioassays were assigned a higher level of evidence than female rat bioassays was 1.69 (p < .001). Of 278 carcinogenic chemicals in the database, 201 (72%) exhibited statistical gender differences (p ≤ .05) in at least one nonreproductive organ. One hundred thirty of these 201 chemicals induced gender-specific tumors in male rats and 59 in female rats. Sixty-eight chemicals induced tumors in males but no tumors in females. Less than one third (i.e., 19 chemicals) induced tumors in females but not males. Male-specific tumors included pancreatic and skin tumors, and female-specific tumors included lung tumors. For some tumor sites, these differences in gender susceptibility can be associated with literature data on sex hormone receptor expression. In conclusion, gender-specific tumors were common. The male dominance is in line with recent human data, and the male susceptibility to carcinogens should be further studied.
Introduction
Excluding reproductive organs, cancer incidences are often different in men and women (Cook et al. 2009; Edgren et al. 2012). The fact that mammary and prostate cancers are among the most common human cancer types suggests a fundamental influence of sex hormones (c.f. Lin and Giovannucci 2010). The role of sex hormones in cancer development has been extensively studied (Anderson 2005; Folkerd and Dowsett 2010) along with the role of hormonal replacement therapy (Santen et al. 2010). However, gender differences in response to xenobiotic carcinogens may also contribute. Such differences can often be related to gender differences in exposure to carcinogens in, for example, work places. Lifestyle or nutritional factors may also influence gender-specific responses, as discussed previously (Kirsch-Volders et al. 2010). Of importance for the present work is that it can be assumed that there are intrinsic differences in susceptibility to xenobiotic carcinogens as, for example, discussed for kidney tumors in male rats (Leavens and Borghoff 2009) or for human lung tumors (Uppstad et al. 2011; Edgren et al. 2012). This aspect of carcinogenesis, as well as gender disparities in general, has been surprisingly little studied (Gochfeld 2007; Cook et al. 2009; Kirsch-Volders et al. 2010; Edgren et al. 2012).
In an effort to get an overview of a possible gender difference in susceptibility to chemical carcinogens, we have analyzed the results obtained in 2-year bioassays published in the National Toxicology Program (NTP) database.
The relevance of rodent carcinogenesis for humans has been discussed in numerous publications (Maronpot, Flake, and Huff 2004; Anisimov, Ukraintseva, and Yashin 2005; Hoenerhoff et al. 2009). Although relevance has been demonstrated in some cases but not in others, animal models remain important in predicting human risk for carcinogens (Huff 2010). A recent example is animal data influencing formaldehyde risk assessment (Andersen et al. 2010). Here, we present rat (mainly Fisher 344) data, which generally are considered more comparable for human cancer than mouse data (Uppstad et al. 2011). Thus, there is little similarity in the spectrum of spontaneous tumors developed by humans compared with mice and rats, but in contrast to mice and similar to humans, rats develop epithelial tumors more frequently (Anisimov et al. 2005). Furthermore, in a time window above the female reproductive period, the overall cancer risk is higher in males than in females in both rats and humans (Anisimov, Ukraintseva, and Yashin 2005; International Agency for Research on Cancer 2010). It is also crucial to emphasize that cancer epidemiology has inherent weaknesses. These include problems associated with decades of latency periods, problems associated with exposure assessments and the complexity of interacting exposures, dietary differences, and the relative rarity of tumors at a specific site. These weaknesses indicate that, for example, negative epidemiological data for a given chemical and a given tumor site do not necessarily exclude human risks (Huff, Jacobson, and Davis 2008).
Cancer tests typically employ male and female rats, mimicking lifetime exposure to carcinogens (NTP 2009). In contrast to practically all human observational studies, exposures, diets and environmental factors are very well controlled. For example, in NTP tests, male and female rats are treated identically, and although doses are not adjusted to give, for example, equal internal dose, doses are adjusted to weight for each sex. Thus, it can be expected that the NTP database is informative concerning fundamental intrinsic aspects of gender differences in chemical carcinogenesis.
We here present an evaluation of NTP chronic bioassays published between 1976 and 2009. Our hypothesis is that carcinogens might interact with sex hormones and that such interactions should be reflected in the NTP database. Such interactions have been suggested by Edgren et al. (2012) and studied experimentally in, for example, bladder cancer (Miyamoto et al. 2007). It was assumed that even weak carcinogenic responses might be informative when analyzing gender-specific trends among several bioassays, so we included not only assays with clear evidence and some evidence but also assays with equivocal evidence in most analysis. Of 278 carcinogenic chemicals identified by NTP, 201 (72%) showed gender differences, and we found that males had more tumors. In this presentation, we will focus on commonly affected tumor sites in nonreproductive organs, not on individual chemicals.
Methods
The NTP Database and 2-Year Cancer Bioassays
The list of NTP peer-reviewed technical reports (http://ntp.niehs.nih.gov/) is arranged by report number and includes carcinogenicity conclusions and links to the technical report abstracts, levels of evidence, and results and status reports. Levels of evidence include clear evidence, some evidence, equivocal evidence, and no evidence. Clear evidence is conferred on chemicals that cause dose-related (1) increase of malignant neoplasms, (2) increase of a combination of malignant and benign neoplasms, or (3) marked increase of benign neoplasms if there is an indication from this or other studies of the ability of such tumors to progress to malignancy. Some evidence indicates a chemical-related increased incidence of neoplasms (malignant, benign, or combined) in which the strength of the response is less than that required for clear evidence. Equivocal evidence indicates a marginal increase of neoplasms that may be chemically related. No evidence is defined as no chemically related increase in tumors. All assays with results classified as clear evidence, some evidence, and equivocal evidence and with both male and female rats were included. A subset of 477 bioassays included both genders, and 86% of these employed Fisher 344 rats. Bioassays performed in one gender, classified by NTP as an “inadequate experiment,” or with no evidence were excluded. Also excluded were results without statistically significant effects at a specific site. In older studies, some or equivocal evidence does not appear, but these assays were few and do not affect the analysis.
Tumors in the following male and female reproductive organs were not included in the analysis: penis, testis, epididymis, vas deferens, seminal vesicles, ejaculatory ducts, prostate and bulbourethral glands, vulva, clitoral glands, clitoris, vagina, uterus, ovary, fallopian tubes, and mammary glands. Tumors in the tunica vaginalis in scrotum were included as mesotheliomas originating in this tissue may spread to abdominal mesothelium and site of origin was not reported.
Statistical Analysis
We compared the tumor rates of males and females by each organ separately using standard analysis of variance. Since there were several potential differences among various studies, such as year of the study, number of doses used in the study, route of exposure, feed, and so forth, we compared the two sexes after adjusting for these study differences. This was done by nesting sex within study (i.e., chemical). Note that a large number of comparisons are being made when investigating the differences between the sexes with respect to each chemical (i.e., each NTP study) for a given tumor. To deal with multiple comparison issues, we performed the pairwise comparisons only in those cases in which the overall “sex within study” effect was significant at p ≤ .05. Consistent with the NTP’s standard protocol, all dose groups were compared with the untreated vehicle control group, and we did not perform multiple testing adjustments for different tumor types. We did not use historical control data since there is no convenient historical database that would match data with each study used in this investigation.
Results
To identify gender differences in chemical carcinogenesis, 2-year rat NTP bioassay data were analyzed. This was done using the evidence level as an indicator of carcinogenic potential for the chemicals. A subset of 477 bioassays included both genders. Among these, 278 showed clear, some, or equivocal evidence for carcinogenicity and were identified as carcinogens. Of these 278 carcinogens, 13 exhibited tumors only in mammary or reproductive organs and were excluded from further analysis. Two hundred and one exhibited some type of gender difference in nonreproductive organs, as defined in the Methods section (Table 1). Eighty-seven (31%) gave tumors in either males or females (68 in males only and 19 in females only). One hundred and two gave tumors in at least one organ in one sex but showed no evidence in the same organ in the other sex (62 in males and 40 in females). A third category was 12 chemicals that gave tumors in both sexes but significantly more in one sex (8 in males and 4 in females; Table 1). Thus, of 278 agents, 87 were gender specific, and the rest, 191 agents, induced tumors in both sexes. It is also apparent from Table 1 that there was a male dominance with, for example, 68 assays with tumors in males only and 19 in females only. In further analysis, we excluded equivocal test results and found even stronger dominance for males. Thus, 40 male-specific and 7 female-specific chemicals were identified. In other words, male-specific chemicals were 5.7 times more common than female-specific chemicals when these more stringent criteria for carcinogenic activity (see the Methods section) were used. All these data indicate that male rats were more susceptible to chemical carcinogenesis in nonreproductive organs.
Gender differences in National Toxicology Program rat cancer bioassays.a

a Four hundred and seventy-seven rat bioassays were analyzed (http://ntp.niehs.nih.gov/).
b Gender differences based on clear, some, and equivocal evidence.
c Five carcinogens were included in both of these categories.
d Gender differences were based on clear and some evidence.
* Significantly greater than the opposite gender, p ≤ .05.
We also performed an overall analysis of data including all nonreproductive and reproductive organs. It revealed that male rats were 1.69 times (p < .001) more often classified to a higher level of evidence for carcinogenic activity than female rats.
Most bioassays (86%) employed Fischer 344 rats (King-Herbert, Sills, and Bucher 2010), and we did not observe any bias for other strains among bioassays showing gender differences. There was a change in diet 1995, which has been reported to affect tumor incidences of, for example, pituitary and testis tumors (Haseman et al. 2003). However, most bioassays used in these studies were performed prior to the food change, and we did not observe trend changes in later studies. The publication years for bioassays used in this study are given in supplementary Table 1. As can be seen, assays exhibiting gender dimorphisms were evenly spread over the years, and we did not observe trends that can be explained by, for example, genetic drift among test animals. Likewise, we did not observe any bias for route of exposure.
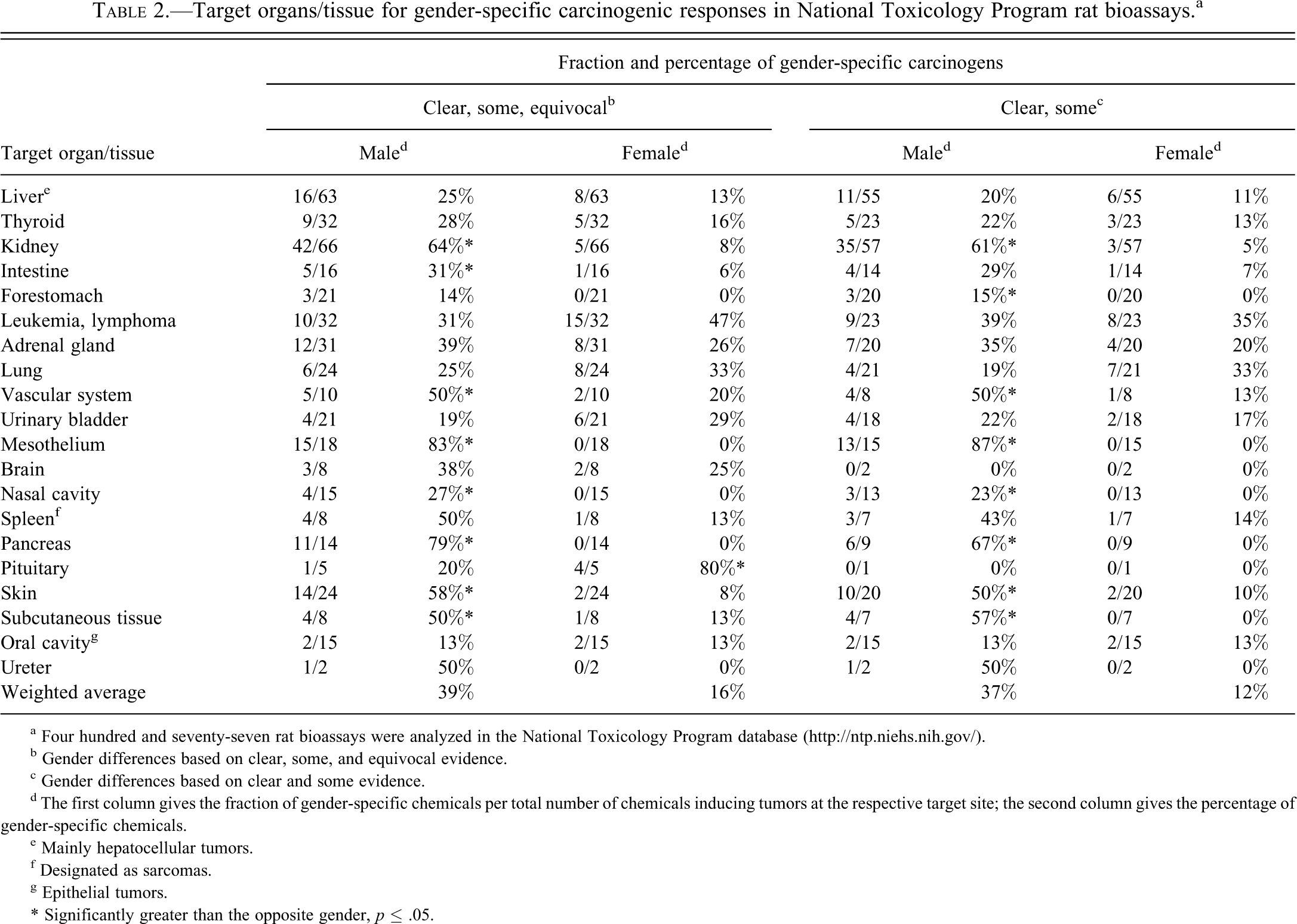
Table 2 shows the fraction of rat carcinogens inducing tumors in an organ or tissue in a gender-specific way (i.e., there were tumors in one organ/site in one sex but no tumors in the same organ/site in the other sex). Table 2 is based on results from 189 (87 + 102) bioassays (or carcinogens) shown in Table 1 and lists 171 positive male organs and tissue sites and 70 positive female sites. It should be noted that some chemicals were multisite carcinogens. The results are presented as fractions and percentages of the total number of chemicals inducing tumors at the same site. Overall, males were more affected, and more often tumors were induced in male rats than in female rats. The four sites that exhibited the highest fractions of male-specific tumors were mesothelium (mesotheliomas; 83% of all mesothelioma-inducing carcinogens), pancreas (79%), kidney (64%), and skin (58%). The four sites that exhibited the highest fractions of female-specific tumors were pituitary (80%), bone marrow and lymphoid tissues (leukemias and lymphomas; 47%), lung (33%), and urinary bladder (29%). Fifteen of 18 chemicals inducing mesotheliomas induced this tumor in males only. Of importance is that the majority, if not all, of mesotheliomas likely derived from the male-specific tunica vaginals in scrotum (Haber et al. 2009) and may thus be regarded as a male-specific reproductive organ tumor.
Target organs/tissue for gender-specific carcinogenic responses in National Toxicology Program rat bioassays.a
a Four hundred and seventy-seven rat bioassays were analyzed in the National Toxicology Program database (http://ntp.niehs.nih.gov/).
b Gender differences based on clear, some, and equivocal evidence.
c Gender differences based on clear and some evidence.
d The first column gives the fraction of gender-specific chemicals per total number of chemicals inducing tumors at the respective target site; the second column gives the percentage of gender-specific chemicals.
e Mainly hepatocellular tumors.
f Designated as sarcomas.
g Epithelial tumors.
* Significantly greater than the opposite gender, p ≤ .05.
Another conspicuous finding was that 11 chemicals of 14 pancreatic carcinogens induced tumors in males only and none in females. Eight chemicals induced exocrine pancreatic cancer in males only but none in females only. In other organs, such as lung, both female- and male-specific carcinogens were identified. Female-specific carcinogens dominated among chemicals inducing leukemia and lymphoma. Interestingly, for some organ/tissues, a high proportion of carcinogens gave gender-specific responses. For example, 78% (31% in males and 47% in females) of chemicals induced leukemia/lymphoma in one gender only. Also for lung carcinogens, the majority (58%) induced tumors in one gender only (25% and 33%, respectively). All gender-specific carcinogens (and their technical report numbers) are listed in supplementary Table 1. As expected, male-specific carcinogens causing tumors in the kidney were common (64%), whereas female-specific carcinogens causing tumors in the kidney were uncommon (8%). When results assigned equivocal evidence were excluded, a male dominance was even stronger. However, the isolated female dominance regarding pituitary tumors was lost. The difference in weighted averages was strengthened (Table 2).
Weighted averages for all listed organs affected by gender-specific carcinogens were 39% for males and 16% for females (Table 2). If mesotheliomas were excluded, the weighted averages were 38% for males and 18% for females. Many kidney tumors and splenic leukemia have previously been discussed as rat specific, and if these two tumor types were excluded, the weighted averages were 35% for males and 18% for females. If all these three tumors types (mesothelioma, kidney, and splenic leukemia) were excluded, the weighted averages were 32% for males and 19% for females. If chemicals with equivocal evidence were excluded as well, weighted averages were 28% for males and 14% for females.
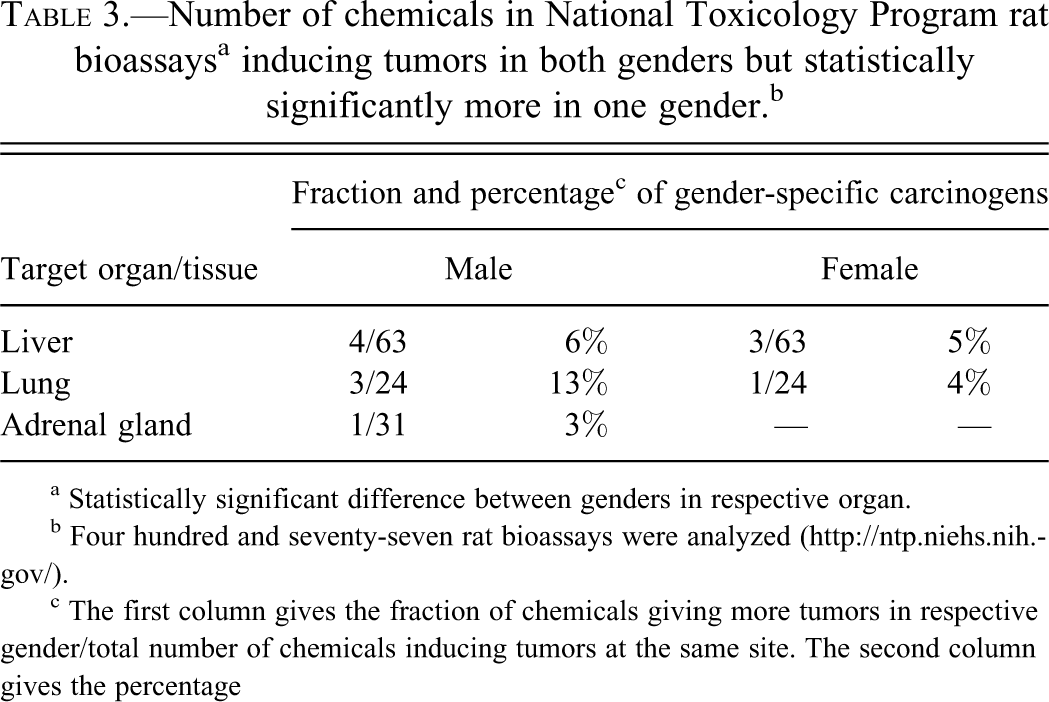
Table 3 shows the number of chemicals in the NTP database inducing tumors in both genders but statistically significantly more in one gender. Table 3 is based on results from 12 bioassays, but 5 of these were also included in Table 2 as they also exhibited gender-specific tumor sites. Only three organs were affected in this way, and again males were more affected. The list of individual chemicals and technical report numbers are shown in supplementary Table 2.
Number of chemicals in National Toxicology Program rat bioassaysa inducing tumors in both genders but statistically significantly more in one gender.b
a Statistically significant difference between genders in respective organ.
b Four hundred and seventy-seven rat bioassays were analyzed (http://ntp.niehs.nih.gov/).
c The first column gives the fraction of chemicals giving more tumors in respective gender/total number of chemicals inducing tumors at the same site. The second column gives the percentage
Next, we studied how often the 189 gender-specific carcinogens, analyzed in Table 2, induced tumors at other sites in the same gender. Table 4 shows that weighted average for both male-specific and female-specific carcinogens inducing additional tumors in the same gender was high, 55% and 59%, respectively. We also analyzed whether the gender-specific carcinogens induced tumors in other nonreproductive organs in the opposite gender. As shown in Table 5, both gender/organ-specific carcinogens in males and females commonly induced tumors in other organs in the opposite gender. For skin-inducing carcinogens, 11 of 14 induced additional tumors in females. The weighted average of male organ–specific carcinogens inducing other tumors in females was 46%, and the weighted average of female organ–specific carcinogens inducing other tumors in males was 66% (Table 5).
Gender-specific rat carcinogens inducing tumors in other organs in the same gender.a
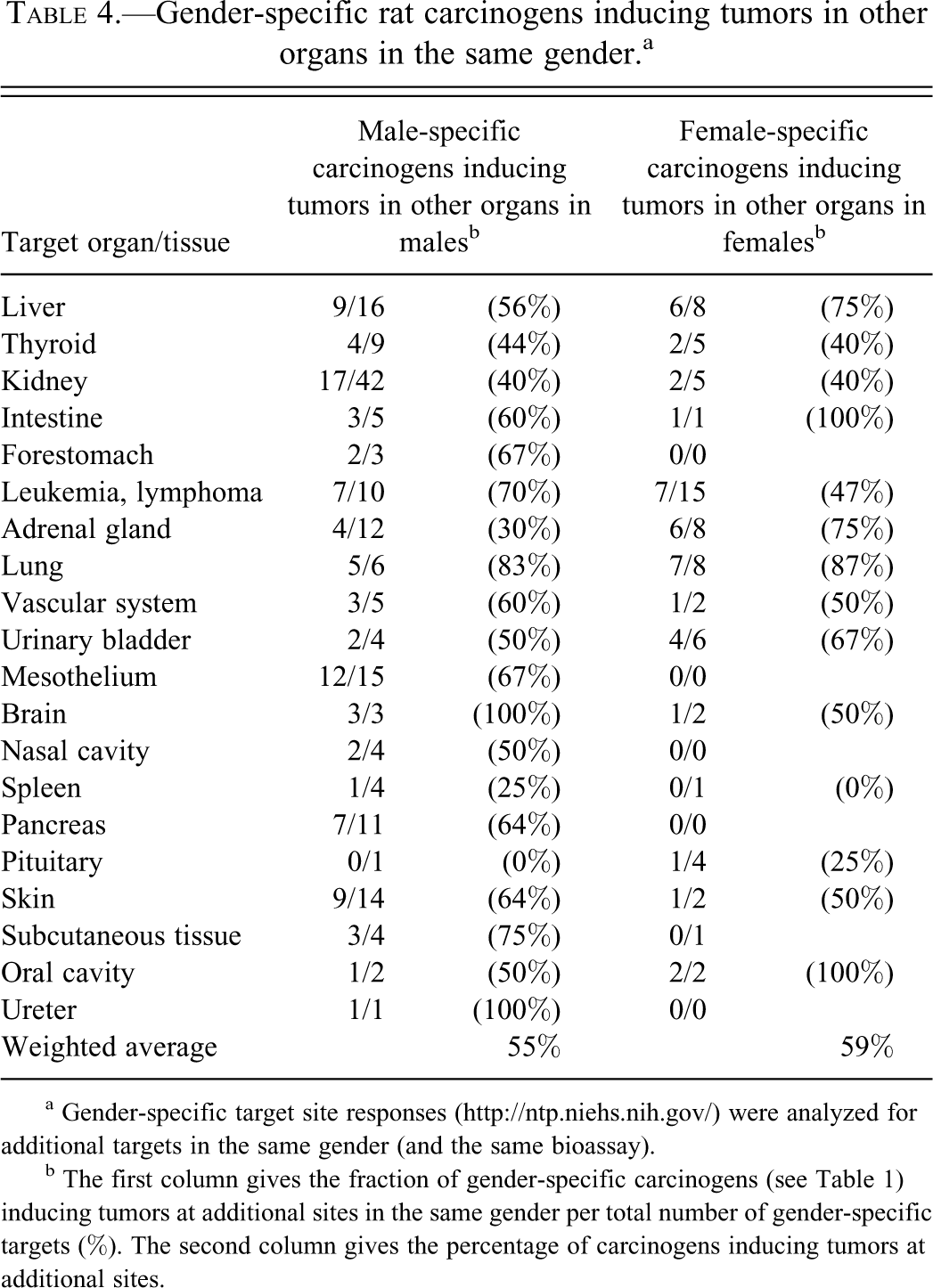
a Gender-specific target site responses (http://ntp.niehs.nih.gov/) were analyzed for additional targets in the same gender (and the same bioassay).
b The first column gives the fraction of gender-specific carcinogens (see Table 1) inducing tumors at additional sites in the same gender per total number of gender-specific targets (%). The second column gives the percentage of carcinogens inducing tumors at additional sites.
Gender-specific rat carcinogens inducing tumors in other organs in the other gender.a
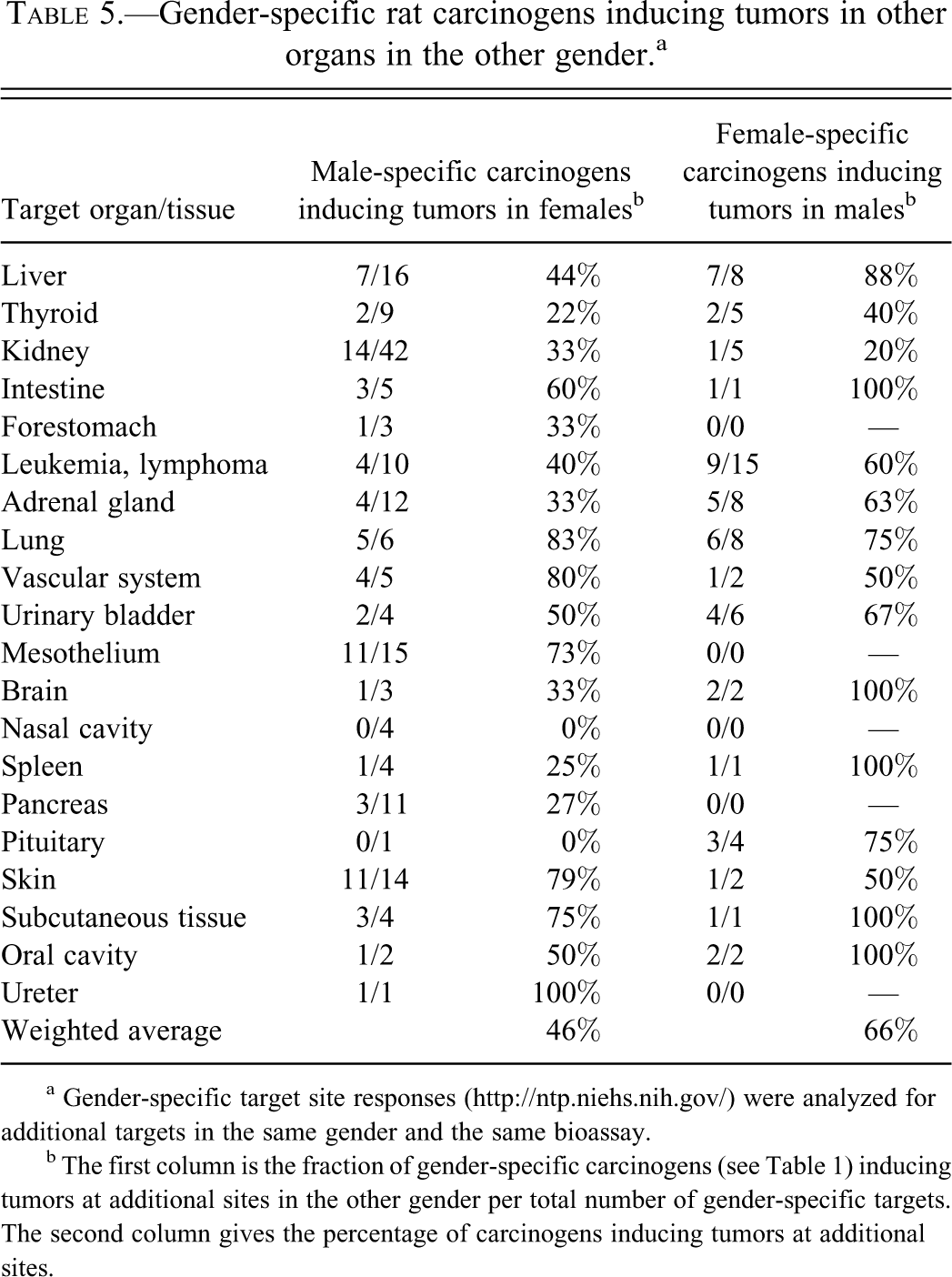
a Gender-specific target site responses (http://ntp.niehs.nih.gov/) were analyzed for additional targets in the same gender and the same bioassay.
b The first column is the fraction of gender-specific carcinogens (see Table 1) inducing tumors at additional sites in the other gender per total number of gender-specific targets. The second column gives the percentage of carcinogens inducing tumors at additional sites.
Male- and female-specific carcinogens may have different modes of action (MOA), and we investigated whether gender-specific chemicals (analyzed in Tables 2, 4, and 5) could be classified as genotoxic or nongenotoxic. For this classification, we used test results given by NTP and classified chemicals as genotoxic if the Salmonella assay was positive in at least one test (for more comprehensive results, see supplementary Table 1). Weakly positive results according to NTP were classified as negative. Overall, genotoxicity was indicated for 38% (105) of 278 carcinogenic chemicals in the NTP database. Table 6 shows the fraction of gender-specific carcinogens that were classified as genotoxic. The weighted average for male-specific carcinogens was 43%, and for female-specific carcinogens, the same value was 31%. These data indicate an overall difference in genotoxicity between male- and female-specific carcinogens. Ten (71%) male-specific skin carcinogens were genotoxic, and five (45%) of pancreatic carcinogens were genotoxic. Also among leukemia- and lymphoma-inducing male-specific carcinogens, genotoxic chemicals dominated. Thus, 60% of male-specific chemicals were genotoxic, while the corresponding value for female-specific carcinogens was 13%. However, the male dominance among genotoxic chemicals was not true for all tumor sites. For example, 63% (5/8) of female-specific lung carcinogens were genotoxic, while 33% (2/6) of male-specific lung carcinogens were genotoxic (Table 6). Together, these data indicate differences in genotoxicity between male- and female-specific carcinogens and that genotoxicity was more common among male- than among female-specific carcinogens. An exception was lung carcinogens.
Genotoxicitya among rat carcinogens inducing gender-specific tumors.b
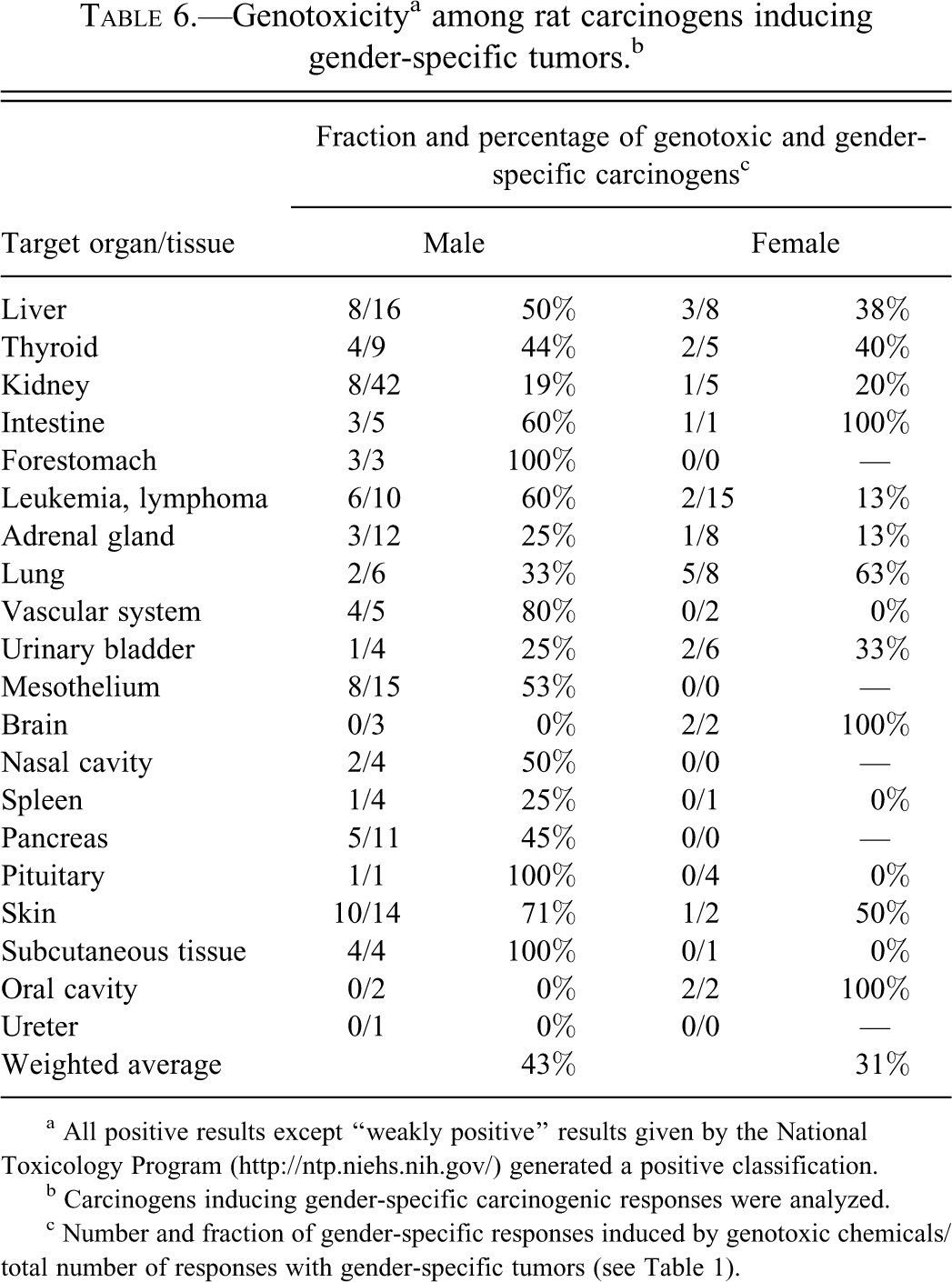
a All positive results except “weakly positive” results given by the National Toxicology Program (http://ntp.niehs.nih.gov/) generated a positive classification.
b Carcinogens inducing gender-specific carcinogenic responses were analyzed.
c Number and fraction of gender-specific responses induced by genotoxic chemicals/total number of responses with gender-specific tumors (see Table 1).
Discussion
There are numerous publications showing gender differences in carcinogenicity, but the contribution of sex-specific susceptibility for carcinogens is to a large extent unknown. We found that of 278 rat carcinogens in the NTP database, 201 exhibit gender differences in at least one tumor site. Sixty-eight of these chemicals induced tumors only in males, and 19 induced tumor in females only. The male sensitivity is further supported by the fact that male rats were 1.69 (p < .001) times more often classified to a higher level of evidence for carcinogenicity than female rats. We thus conclude that gender-specific responses to carcinogens are common and that males are more often affected. In the site-specific analysis, we found that mesothelium, pancreas, kidney, and skin exhibited the highest frequencies of male-specific tumors. The male dominance is quite strong, and to our knowledge, this observation has not previously been discussed.
We do not have an overarching explanation for the male dominance. However, there are two recent human studies showing a similar pattern. These studies, comparing incidence rates for men and women, concluded that there might be “universal mechanisms that increase male susceptibility to cancer” (Cook et al. 2009) or might be “intrinsic sex specific biological factors . . . modify the susceptibility to extrinsic oncogenic factors” (Edgren et al. 2012). Our data tend to support these conclusions, although human incidence rates are affected by infections, food habits, obesity, and other factors presumably not influencing NTP assays. It is interesting to note that Cook et al. (2009) found that the overall male-to-female incidence rate ratio (IRR) was 1.77 for cancers in nonsex organs. This IRR is not directly comparable but strikingly similar to our overall male-to-female ratio of 1.69, which is based on NTP evidence levels. Future studies may clarify if this similarity is coincident or if it reflects a common biology in human and rat.
The intention with our study was to perform a statistical analysis of the NTP database to document gender differences in response to chemical carcinogens and to get support for future mechanistic studies. Such future studies may also include a more detailed analysis of different tumor types at a single site. Here we will discuss our site-specific findings in connection to already published data.
Male-specific mesotheliomas originating in the tunica vaginalis might be regarded as a reproductive organ–derived tumor and will not be comprehensively discussed here. However, it is of interest that mesotheliomas in pleura or peritoneum in humans are far more common among men than among women (Wolf et al. 2010), and it has been suggested that estrogen receptor (ER) β expression may confer resistance among women (Pinton et al. 2010).
Male rats express a small protein, α2u-globulin, in liver and plasma, and its expression is androgen dependent (Chatterjee et al. 1989). It is excreted in primary urine and is absorbed in kidney tubular cells. Xenobiotics, including many NTP test chemicals, may bind α2u-globulin and inhibit its degradation so that it accumulates in tubular cells and becomes toxic. The cytotoxicity may promote renal tubular tumor development (Hard, Johnson, and Cohen 2009; Leavens and Borghoff 2009), which may work in concert with a chronic progressive nephropathy (CPN) in male rats (Hard, Johnson, and Cohen 2009). Both of these modes of action have been discussed as nonrelevant for humans (Hard, Johnson, and Cohen 2009). Challenging is, however, that kidney cancer is more common among men (Cook et al. 2009; Knox and Colli 2011; Edgren et al. 2012), and a significant proportion of renal cell carcinomas in humans express androgen receptor (AR; Brown et al. 1998; Langner et al. 2004). Further MOA analysis that can ascertain that rat male kidney carcinogens do not cause cancer via other mechanisms than α2u-globulin toxicity or CPN is thus warranted.
The male dominance regarding exocrine pancreatic cancer was discussed 20 years ago (Longnecker 1990; Longnecker and Sumi 1990), and our data confirm and extend those earlier observations. Rats (and mice) develop an acinar type of exocrine tumor, whereas humans usually develop a ductal type, and this phenotypic difference has raised questions about the relevance for humans of pancreatic carcinogenesis in rodents (Maronpot, Flake, and Huff 2004). However, pancreatic cells exhibit great plasticity (Puri and Hebrok 2010), and uncertainty still remains regarding cells of origin for ductal cancer (Kong et al. 2011). Early work shows that caerulin promotes cancer development in rats by inducing growth of azaserine-induced acinar cell nodules (Lhoste and Longnecker 1987), whereas caerulin-induced chronic pancreatitis leads to ductal carcinoma in mice harboring mutated K-ras in acinar or centroacinar cells (Guerra et al. 2007). These data suggest that long-term treatments resulting in pancreatitis may promote ductal cancer development provided key oncogenic mutations are expressed in progenitor cells. Xenobiotic-induced ductal carcinomas in rats have also been reported (Rivera et al. 1997), and it can be suggested that the induction of acinar carcinomas in the NTP tests reflects critical carcinogenic stimuli that may result in either acinar or ductal carcinomas depending on whether cells of origin harbor, for example, K-ras mutations. Further studies are thus needed to understand the relevance, if any, of acinar rat pancreatic tumors for humans. It should also be noted that pancreatic cancer is more common among males (Cook et al. 2009; Edgren et al. 2012) and that young men (<60 years of age) are 1.6 times more likely to develop this neoplasm than young women (Zhou et al. 2010). This difference might be explained by, for example, differences in alcohol consumption, but AR signaling has been reported in pancreatic cancer (Okitsu et al. 2010). Furthermore, there are epidemiological studies supporting a male-specific susceptibility to, for example, crystalline silica dust (Birk et al. 2009) and to red meat (Larsson and Wolk 2012).
We also evaluated data on genotoxicity in the NTP database. Genotoxicity was 1.4 times more common among the male-specific carcinogens than among the female-specific carcinogens. Considering, for example, the high numbers of nongenotoxic kidney carcinogens in male rats, this figure is surprisingly high. A site-specific analysis is hampered by low numbers in many cases, but it is interesting to note that 63% of female-specific lung carcinogens were genotoxic, whereas only 33% of male-specific lung carcinogens were genotoxic. This suggests a greater susceptibility among female rats for genotoxic lung carcinogens and is in line with epidemiology suggesting that women are more susceptible to tobacco smoking (Kiyohara and Ohno 2010). Further support is provided by biomonitoring data on susceptibility to tobacco mutagens among women (Kirsch-Volders et al. 2010) and by lung epithelial cell studies suggesting that xenobiotic metabolism in lung cells is tuned to give more DNA adducts in women (Mollerup et al. 2006; Uppstad et al. 2011). It has also been observed that expression of high levels of ERα receptors is associated with poor prognosis in men or women with lung cancer (Olivo-Marston et al. 2010). This observation may reflect estrogen influences on lung carcinogenesis but may also reflect influences on cancer therapy.
Site-specific carcinogens in both males and females commonly induced tumors at other sites in both sexes. This suggests that the majority of site-specific effects were not a consequence of well-studied gender dimorphisms in xenobiotic metabolism (Oertelt-Prigione and Regitz-Zagrosek 2009; Kirsch-Volders et al. 2010), altering, for example, blood levels or elimination rates. A possible exception is urinary bladder tumors, which might be heavily influenced by excreted metabolites (Zenser, Lakshmi, and Davis 1998). This tumor type did not exhibit marked differences between females and males in the NTP assays, so toxicokinetic differences may have concealed an influence of AR expression, as reported by Miyamoto et al. (2007).
There is ample literature support for an influence of hormones in rats and humans. It may, for example, involve oncogenes and suppressor genes and their signaling pathways. Thus, hormonal- and age-related decline in p53 function, occurring later in male than in female mice, has been reported (Feng et al. 2007). If this also occurs in rats, it may explain high incidences in 3-year-old female rats (Anisimov, Ukraintseva, and Yashin 2005), and if it occurs in humans, it may explain the above-discussed susceptibility to tobacco smoke among women (Kirsch-Volders et al. 2010). Other influences may include liver cancer–promoting effects of AR (Yu et al. 2000; Yoon et al. 2006; Ma et al. 2008; Zender and Kubicka 2008) or upstream regulators of AR (Tsuei et al. 2011). Inhibiting effects of estrogen, decreasing IL-6 production, and inhibiting hepatocellular carcinoma development have also been shown (Naugler et al. 2007). Of further interest is that shorter CAG repeats in the AR gene associate with hepatocellular carcinoma (Yu et al. 2000) and with pulmonary, esophageal, or bladder cancer (Teng et al. 2010) and that the expression of a transporter of hormones, including testosterone and estrogens, correlated to the differentiation of liver, colon, and other cancer types (Pressler et al. 2011). Yet another example is that estrogen may have a protective effect in human stomach cancer (Chandanos and Lagergren 2008). These studies support the notion that sex hormones may have influenced not only human cancers in nonreproductive organs but also our NTP data.
In summary, we show that gender-specific tumor responses in rats are common. We also show that male rats are more often affected than female rats by chemical carcinogens. The male dominance in rats is in line with human incidence rates being higher in males than in females. Thus, although some affected rat tumor sites are regarded as nonrelevant to humans, and, for example, male rat kidney tumors have been explained in molecular terms, the male dominance should be further studied with the aim to understand this susceptibility among male rats and men. Literature data on sex hormone receptor expression in nonreproductive organs might provide a base for one of several possible explanations. Further experimental and epidemiological studies may disclose interactions between chemical carcinogen and hormones overlooked in the past.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swedish Research Council for the Environment, Agricultural Sciences and Spatial Planning (FORMAS) and by the Intramural Research Program of the National Institutes of Health, National Institute of Environmental Health Sciences, project ES045005-14.
