Abstract
Wistar rats are frequently selected for use in carcinogenicity studies because of their advantageous survival rate, which is more favorable than other strains such as the Sprague-Dawley (SD) strain. Uterine and mammary tumors are relatively common spontaneous neoplasms of both strains. We examined the incidence and coincidence of uterine tumors and mammary tumors in control animals of both strains within the RITA database. There was a strong inverse relationship between these tumor types in Wistar rats (p < .001). A less strong relationship was present in SD rats (p = .057). This association is likely to be related to prolactin. A short review of the role of prolactin in rats is given. These results are also discussed in the background of nonspecific toxicity at high dose levels in carcinogenicity studies above MTD levels resulting in reduction in body weights of >10%.
Introduction
The interpretation of carcinogenicity studies in rodents is focused on dose-related increased incidences of tumors. However, hormonal changes, as a consequence of normal aging, frequently dictate the spontaneous incidence of several tumor types. Many of the common spontaneous tumors in rats are dependent on hormonal stimulus in their pathogenesis (e.g., adrenal, pituitary, mammary, thyroid; Greaves 2007). Disturbances in hormonal control will lead to changes in tumor patterns; for example, simple food restriction leading to reduced body weight gain is known to dramatically affect the incidence of pituitary tumors and mammary tumors. The reduced incidence of mammary tumors is due to lower sustained levels of prolactin observed in the animals on a restricted diet. In dietary restriction studies, both Sprague-Dawley (SD) and Wistar rats showed reduced pituitary and mammary tumors. Studies in Wistar rats, but not in SD rats, also showed an increase in uterine tumors (Roe et al. 1995; Keenan, Soper, Smith, et al. 1995). The trophic drive of prolactin for mammary tumor formation in the rat is well established (Hargreaves and Harleman 2011). Food restriction leads to increased life span and cycling due to the positive effect on longevity and lower luteotrophic action on the ovary by prolactin. The mechanism of action proposed for the uterine tumor formation in Wistar rats is that the increased cycling and relatively longer estrogen stimulation leads to this effect (Neumann 1991). If the pathogenesis of these tumors is linked to a certain hormonal stimulation, one would expect that the incidence of these tumors is linked as well and not randomly distributed. We therefore examined, in the RITA database of tumors, the interdependency of mammary and uterine tumor incidences for SD- and Wistar-derived strains (Mohr et al. 1990). The RITA database is a company-sponsored database of peer-reviewed historical tumor data in control animals. Companies participating in this activity are Abbott, AstraZeneca, BASF AG, Bayer AG (BHC, BCS), Boehringer Ingelheim Pharma GmbH & Co. KG, F. Hoffmann-LaRoche AG, Merck-Serono, Novartis Pharma AG, Novo Nordisk, Nycomed GmbH, Pfizer, Sanofi-Aventis, and Syngenta CTL. This database is used in the interpretation of carcinogenicity studies (Keenan et al. 2009).
Materials and Methods
The incidence of mammary, pituitary, and uterine tumors was examined in the RITA database for SD and Wistar rat strains. The number of female animals in the database was 5419 Wistar rats and 2158 SD rats, respectively. Also examined was the coexistence of mammary tumors and uterine tumors on an individual animal basis. The association between mammary and uterine tumors was tested statistically using a chi-squared test.
Results
Tables 1 and 2 give the incidence of mammary and uterine tumors in the RITA database for, respectively, the Wistar and the SD rat strains. Mammary tumors were combined and include animals with adenocarcinoma and/or adenocarcinoma arising in fibroadenoma, adenomas, and/or fibroadenoma. Uterine epithelial tumors combined include animals with adenocarcinoma and/or adenoma.
Incidence of mammary tumors and uterine tumors in Wistar rats.

Incidence of mammary tumors and uterine tumors in Sprague-Dawley rats.

From Tables 1 and 2, it is clear that there are differences in background incidence data between the two strains. The Wistar rat has a markedly lower incidence of mammary tumors compared with SD rats (24% compared with 58%, respectively). On the other hand, the Wistar rat has a markedly higher spontaneous incidence of uterine tumors compared with the SD rats (5% compared with 0.9%, respectively). In both strains, the occurrence of uterine tumors arises predominantly in animals without a mammary tumor. This effect is more pronounced in Wistar rats than in SD rats. For animals with a uterine tumor, the odds of a mammary tumor are 0.17 and 0.58 for Wistar and SD rats, respectively. Similarly for animals without a uterine tumor, the odds of a mammary tumor are 0.33 for Wistar rats and 1.41 for SD rats. Taking the odds ratios, Wistar rats are twice as likely to have a mammary tumor when a rat does not have a uterine tumor compared with a rat that does have a uterine tumor. Equivalently, Wistar rats with a uterine tumor have half the odds of having a mammary tumor compared with rats that do not have a uterine tumor. In comparison, SD rats are 2.4 times as likely to have a mammary tumor when a rat does not have a uterine tumor. Equivalently, SD rats with a uterine tumor have 0.4 times the odds of having a mammary tumor compared with rats that do not have a uterine tumor. Chi-squared tests (on 1 degree of freedom) were used to test the association between mammary and uterine tumors, separately for Wistar and SD rats. For the Wistar rat strain, there was strong evidence of an inverse relationship between mammary and uterine tumors (χ2 = 14.364, p < .001). However, there was insufficient evidence at the 5% significance level to reject the null hypothesis of no association in the SD rat strain (χ2 = 3.612, p = .0574).
Discussion
The results of these analyses show that, especially in Wistar rats, there is an inverse relationship between the incidence of mammary tumors and uterine tumors. This is less obvious for SD rats, although a relationship does seem to exist. The p value would have been significant if the same number of animals as in Wistar rats would have been available assuming the ratio remained the same. The factor driving this response is likely to be prolactin levels. Plasma prolactin levels are known to increase with age in rats due to reduced hypothalamic dopamine drive and correlate well with the age-dependent increase in pituitary hyperplasia and tumors observed in this species (Greaves 2007; Tucker 1997).
Literature data clearly demonstrate that most rat pituitary hyperplasias and adenomas are prolactin positive (Kovacs et al. 1977). They are functional prolactin-producing tumors and appear chromophobic because of the rapid discharge of the hormones they produce (Greaves and Faccini 1984). Studies in rats show that many animals with pituitary adenomas have high levels of circulating prolactin and that this correlates with the size of the tumor (Greaves 2007). The increase in circulating prolactin is directly associated with an increase in the incidence of mammary tumors, as prolactin is the major promotional driver of mammary tumors in this species (Welsch, Nagasawa, and Meites 1970; Tucker 1997). Compounds such as neuroleptics, which increase the levels of prolactin, are associated with an increased incidence of pituitary and mammary tumors (Gopinath, Prentice, and Lewis 1987; Greaves 2007). In contrast, compounds that reduce prolactin secretion such as bromocriptine are associated with a reduction in the incidence of pituitary hyperplasia and adenomas as well as a striking reduction in the incidence of mammary tumors (Griffith 1977; O’Connor et al. 2000). Prolactin has been demonstrated to influence the incidence of mammary tumors via a direct trophic effect. In studies with diet restriction, a reduction in pituitary hyperplasia together with a reduced incidence of mammary adenomas and mammary tumors was also observed and linked to the decreased levels of circulating prolactin (Roe et al. 1995).
However, in studies on changes in tumor patterns following either a direct reduction of prolactin with bromocriptine or indirectly by reduced body weight gain, the correlation between the presence of, for example, mammary tumors and pituitary tumors is variable. This may reflect the multifactorial mechanism and genetic predisposition involved in tumorgenesis and the fact that not all pituitary tumors are prolactin positive. Despite this, it has been established that prolactin levels in rat models are the major driving force influencing the incidence of mammary tumors through prolactin’s major trophic action for this organ (O’Connor et al. 2000; Blankenstein et al. 1984; Hargreaves and Harleman 2011). Hence, we investigated the coincidence of mammary tumors and uterine tumors, assuming that if the animal had a mammary tumor, this was likely a sign of prolactin stimulation.
Prolactin is luteotropic in rats in contrast to primates; that is, it promotes progesterone production of the corpus luteum after ovulation and maintains gestation. During the luteal phase of the reproductive cycle, the corpus luteum produces large quantities of progesterone, which antagonizes estrogenic stimulation of uterine growth (Gambrell, Bagnell, and Greenblatt 1983). Therefore, seen over a series of cycles, inhibition of prolactin results in an increased estradiol:progesterone ratio in rats. Unopposed estrogen is trophic in the uterine endometrium, promoting an increased incidence of uterine tumors. In addition, low prolactin counteracts age-related reproductive changes in rats, maintaining reproductive viability and cyclicity for longer into old age, and therefore animals are exposed to high levels of unopposed estradiol for longer than normal. This effect is reported to be rat specific as prolactin is not luteotrophic in humans (Neumann 1991; Ben-Jonathan, LaPensee, and LaPensee 2008).
Exogenous agents or treatments, which lower prolactin drive and pituitary responses in rodents, do not just reduce pituitary and mammary tumor rates but also (indirectly) increase the incidence of uterine tumors. Studies with bromocriptine, which inhibits prolactin secretion, also showed a marked increase of uterine tumors (Griffith 1977; Richardson, Turkali, and Flueckiger 1984). Studies in Wistar rats, such as the Biosure study with 1200 animals on diet restriction, clearly showed that a reduction of body weight development by diet restriction of approximately 20% was also associated with a reduction of pituitary hyperplasia and adenomas, reduction of mammary tumors, and an increase (approximately three to four times over background) of uterine tumors, albeit not all dietary manipulations with a reduced prolactin level resulted in the same effect (Roe et al. 1995). The mechanism proposed for this increased incidence of uterine tumors in rats with a lifetime of relative hypoprolactinemia is the reduced luteotrophic effect of prolactin in the rat.
Because of the markedly different mechanisms involved in female reproductive hormonal regulation between man and rat, these rat tumors are generally considered to have no significance to humans (Alison, Capen, and Prentice 1994; Burek, Patrick, and Gerson 1988; Ettlin and Prentice 2002; Cohen et al. 2004).
Apart from its role in the development and function of the mammary gland, pituitary prolactin has a fundamental role in the ovarian function of rats. Figure 1 depicts the regulation of prolactin and its effect on the female genital system (modified from Neumann 1991).
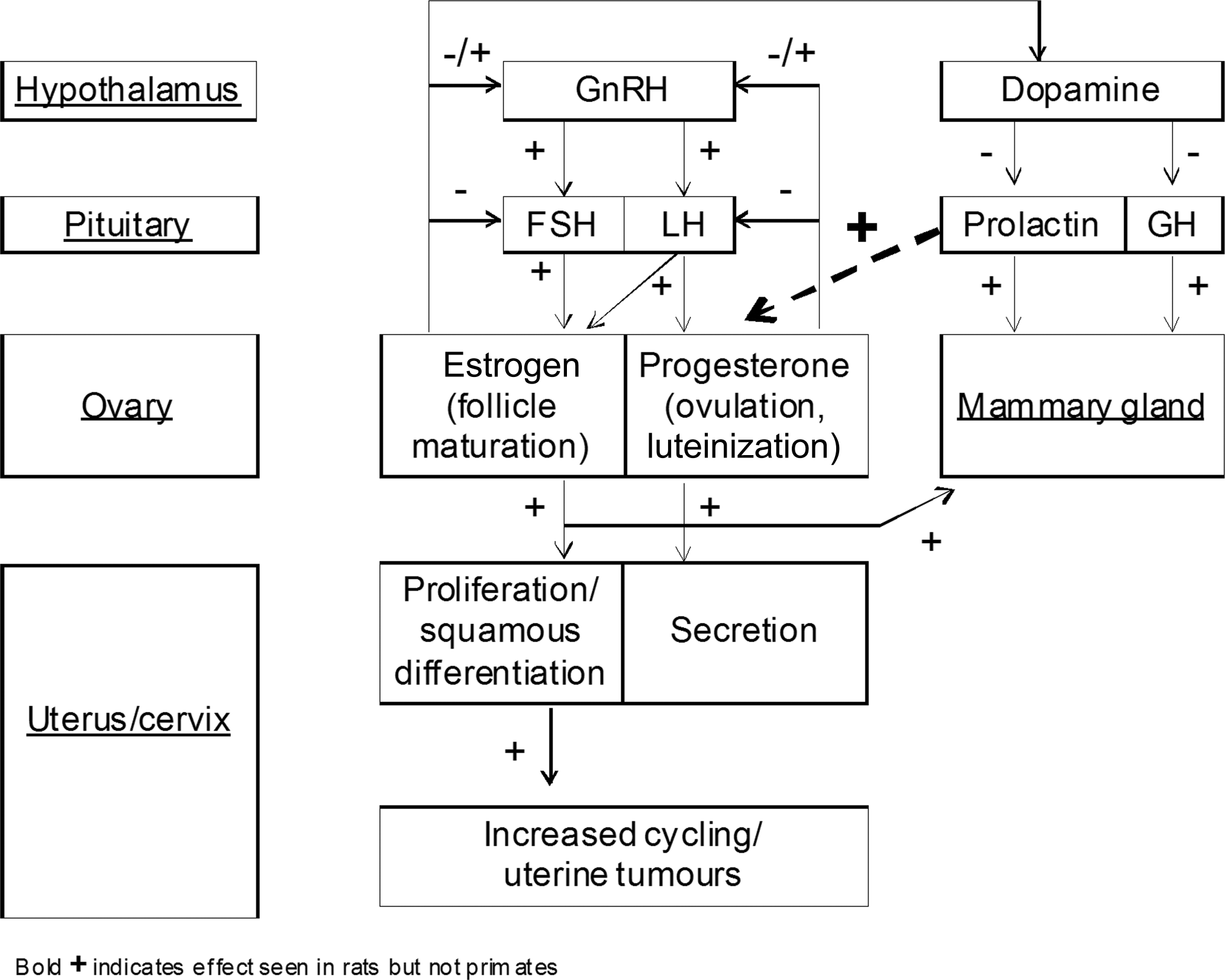
Hormonal regulation of the female rat and effect on uterus.
In rats (but not humans), prolactin is essential for corpora lutea function and progesterone production (by inhibition of the progesterone-catabolizing enzyme, 20α-hydroxysteriod dehydrogenase), and prolactin surges rescue the corpora lutea from luteolysis in the mated or pregnant rat (Ben-Jonathan, LaPensee, and LaPensee 2008; Grattan et al. 2008; Freeman et al. 2000), as summarized in Table 3.
Regulation of ovarian steroids in humans and rats in relation to cycle phase.
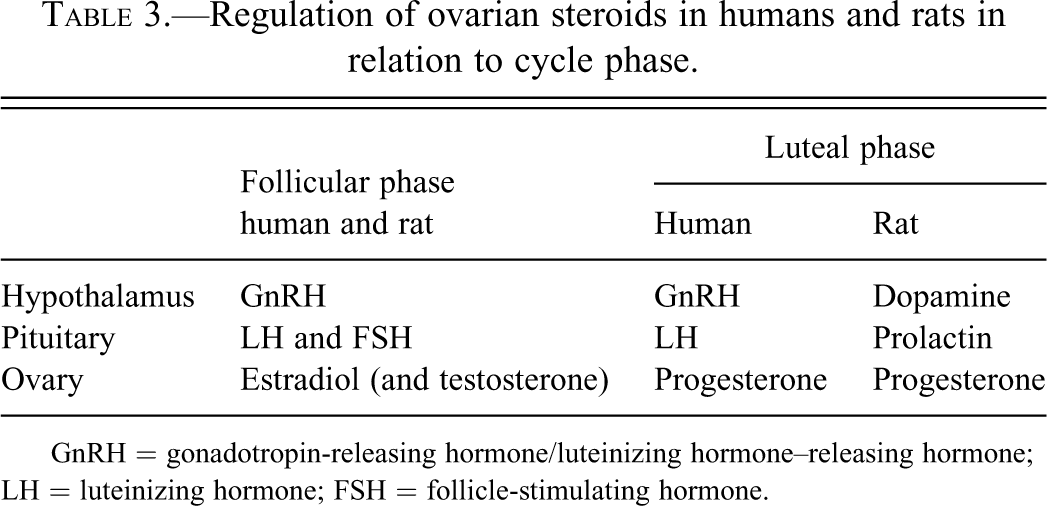
GnRH = gonadotropin-releasing hormone/luteinizing hormone–releasing hormone; LH = luteinizing hormone; FSH = follicle-stimulating hormone.
Consequently, prolactin is secreted in a typical triphasic (a peak, a plateau, and a termination phase) pattern during the estrous cycle in rats. In the unmated rat, prolactin peaks in proestrus and declines during estrus (Ben-Jonathan, LaPensee, and LaPensee 2008). The prolactin peak is driven by rising estrogen levels interacting with all three dopamine systems (tuberoinfundibular, tuberohypophyseal, and periventricular hypophyseal dopaminegric neurons) that regulate prolactin release in this species (Freeman et al. 2000), and inhibition of dopamine transporters have been shown to decrease estrogen-triggered prolactin release in rats (Demaria et al. 2000). Other sex steroids, such as progesterone in late pregnancy and exogenous testosterone, negatively influence pituitary prolactin release in rodents (Grattan et al. 2008; Sinha, Gilligan, and Barkley 1984), and any disruption, or reduction, of pituitary prolactin secretion negatively affects the luteal phases in rats and thereby the balance between the estrogen and progestin drive. In rats, prolactin also plays a role in correct luteolysis, and since estradiol is the major stimulus for both the preovulatory LH surge and the prolactin peak, reduced estradiol secretion during the follicular phase can delay the progressive positive priming effect on the hypothalamus and thus cause alterations of ovarian cycle phases, such as extended estrous (Freeman et al. 2000).
In the aging female rat, reproductive irregularities, such as irregular estrous cycles and spontaneous pseudopregancy, leading up to reproductive senescence are common (Dudley 1982). In contrast to humans, these reproductive aging effects in rats do not result from follicle depletion in the ovary but rather from age-related changes in the hypothalamus (e.g., a progressive loss of tuberoinfundibular and periventricular dopaminergic neurons; Everitt 1980; Reymond, Donda, and Lemarchand-Béraud 1989; Sánchez et al. 2003). Pituitary prolactin is critical for the transformation of a regular estrous cycle into pseudopregancy in the rat (Smith, McLean, and Neill 1976), and the age-related increase in prolactin due to reduced dopamine activity is considered a major contributing factor for the increased incidence of pseudopregnancy (Lu et al. 1979). Consequently, but perhaps rather paradoxically, in the aging female rat, a reduction of pituitary prolactin release (e.g., by restricted feeding) will favor continued cyclicity (and thereby continued periodic estrogenic drive) and delay reproductive senescence (Keenan, Soper, Smith, et al. 1995; Keenan, Soper, Hertzog, et al. 1995). Although the age-related effect of prolactin and estrogenic drive on the uterus is well established (Griffith 1977; Alison, Capen, and Prentice 1994), it is clear that not all studies with reduced prolactin show an increase in uterine tumors, possibly because of different background strain susceptibilities. Reports of increased uterine tumors associated with reduced levels of prolactin are found in studies conducted in Wistar rat strains (Roe et al. 1995; Griffith 1977) but not in diet restriction studies in SD rats, showing the apparent specific sensitivity for this effect in the Wistar strain (Keenan et al. 1994; Keenan, Soper, Smith, et al. 1995; Keenan, Soper, Hertzog, et al. 1995).
Our analysis showed that the inverse relationship of mammary and uterine tumors was stronger in Wistar than in SD rats. This possibly explains why increased levels of uterine tumors are generally seen only in food restriction studies in Wistar rats and not in SD rats. In the analysis of tumor incidences, several observations such as a reduced incidence of mammary tumors and pituitary tumor lend support that the prolactin mechanism is responsible for the increased incidence of uterine tumors in treated animals. An increased incidence of uterine tumors associated with reduced mammary and pituitary tumors is more likely to occur in Wistar rats than in SD rats because of the differences in the background incidence in pituitary tumors and mammary tumors. This is important as the Wistar strain is more frequently used in carcinogenicity studies because of the better overall survival of this strain compared with SD rats. In the interpretation of carcinogenicity studies, the increased levels of uterine tumors may also be an indirect effect of reduced body weight gain due to nonspecific toxicity induced by dosing at supra MTD exposures. Effects of 10% to 20% body weight gain reductions may induce the pattern of hormonal changes, and subsequent changes in tumor patterns, as described. In these cases, direct effects on the prolactin system, such as effects on dopamine or estrogen, need to be excluded.
Footnotes
Acknowledgment
The authors thank Rupert Kellner from the Fraunhofer Institute for Toxicology and Experimental Medicine for his excellent support in extracting the combined tumor data out of the RITA database.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
