Abstract
Mature rat testes and liver were fixed with Bouin’s fluid (BF) or modified Davidson’s fixative (mDF) at room temperature (23°C) or 4°C, and DNA integrity was examined by the TUNEL assay. When testes were fixed in BF, TUNEL-stained cells were more prevalent than when fixation occurred in mDF. Independent of fixative, TUNEL-staining was higher when testes were fixed at room temperature relative to 4°C. Significant effects were present for fixative and temperature of fixation, but not their interaction. Relative to BF, mDF also provided for lower TUNEL-staining in liver, but staining was not affected by fixation temperature. Since the TUNEL assay depends on the detection of fragmented DNA strands, harsh fixatives that induce breaks in the DNA can introduce substantial artifacts. Such potential artifacts are especially prevalent in a tissue such as testes with its ongoing division and differentiation activities. Therefore, the current findings lead the authors to conclude that fixation of mature testes in mDF at 4°C minimizes generation of false TUNEL-positive cells.
Introduction
Environmental chemical exposure to heavy metals (cadmium and mercury) and endocrine disrupting chemicals (dioxins, phthalates, and bisphenol A) have the potential to affect male reproduction and may result in male sterility (Diamanti-Kandarakis et al. 2009). Cellular apoptosis assays are frequently employed to evaluate environmental agents for potential toxicity and cell damage such as DNA strand breakage that is part of the apoptotic process. However, to effectively assess toxic effects in the reproductive tissue, it is important to evaluate all cell types. The terminal deoxynucleotidyl transferase dUTP nick end-labeling (TUNEL) is a preferred assay to detect and quantify apoptotic cells with fragmented DNA in fixed, paraffin embedded tissues. However, since DNA is often damaged due to fixation and processing protocols (Tornusciolo, Schmidt, and Roth 1995; Garrity et al., 2003), toxicity of environmental chemicals upon specific germ cell populations may be nonassessable. Hence, for toxico-pathological evaluation of testicular tissue with the TUNEL assay, it was imperative to minimize fixation-induced changes, especially DNA damage.
Compared to many organs, fixation of testicular tissue for histological and immunohistochemical analysis is difficult due to low protein content and minimal connective tissue in the seminiferous epithelium (Mills, Mills, and Means 1977). The reduced protein and connective tissue content causes increased demand on fixatives to effectively stabilize the cells of the seminiferous epithelium in their proper location. Since testicular cell associations are specific and progressive from periphery to lumen of seminiferous tubules and the cycle of the seminiferous epithelium is continuous longitudinally along tubular axes, maintenance of cellular positions are essential for identification of different stages of spermatogenesis. Although rodents are excellent experimental models, rodent testes are more challenging to fix and maintain localization of germ cells as compared to many other mammalian testes that have internal testicular septa to provide stability for the seminiferous tubules (Hedger and Hales 2006). Therefore, for fixation of rodent testes, it is important to use a stronger fixative that maintains cellular location. Organizations and societies, such as the Organization for Economic Cooperation and Development (OECD), Office of Prevention, Pesticides and Toxic Substances (OPPTS), and the International Conference on Harmonization (ICH), have recommended the use of either Bouin’s fluid (BF) or a comparable fixative to study testicular toxicity (Latendresse et al. 2002). In contrast, the Society of Toxicologic Pathology (STP) has recommended modified Davidson’s fixative (mDF) for toxicological studies involving testicular and epididymal tissues (Lanning et al. 2002). However, none of these societies or organizations has addressed the problem of increased DNA strand breakage with these harsher fixatives and there have been no comparisons of fixation temperatures for tissue processing.
During the executionary phase of apoptosis open 3′–OH ends are generated due to DNA fragmentation (Willingham 1999; Taatjes, Sobel, and Budd 2008), and this feature of apoptosis is exploited in the TUNEL assay for apoptotic cell detection (Gavrieli, Sherman, and Ben-Sasson 1992). Although the TUNEL assay is a very useful method for both qualitative and quantifiable assessment of apoptotic cells, false TUNEL-positive reactions are a matter of concern (Negoescu et al. 1996; Labat-Moleur et al. 1998; Garrity et al. 2003; Taatjes, Sobel, and Budd 2008). False TUNEL-positive cells are generated due to (1) the harshness of the fixatives (Tornusciolo, Schmidt, and Roth 1995; Garrity et al. 2003) and (2) the concentration and duration of proteinase K treatments prior to the terminal deoxynucleotidyl transferase (TdT) reaction for the TUNEL assay (Tornusciolo, Schmidt, and Roth 1995; Labat-Moleur et al. 1998; Stahelin et al. 1998). Since a variety of fixation methods have been employed in toxicological research, results from different studies using the same drug and model organism are often variable. For example, using ethylene dimethane sulfonate (EDS), a Leydig cell specific toxicant in rats, it was reported that, at 3-day post-EDS (75 mg/kg body weight) treatment, the apoptotic index of germ cells was approximately 100 (Bakalska et al., 2004). In contrast, another study reported the same drug, at a higher dosage (100 mg/kg body weight), to have a much lower germ cell apoptotic index (29.1) at 5-day post-EDS treatment (Woolveridge et al. 1999). Since the rate of germ cell apoptosis increases significantly with time after EDS treatment (Henriksen, Hakovirta, and Parvinen 1995), these two reports appear to offer contradictory information regarding the rate of germ cell apoptosis. In each case, BF was used for fixation of testes; however, information in regard to temperature of fixation was not provided. Mouse embryos fixed in BF at room temperature had a substantial increase of false TUNEL-positive nuclei compared to embryos fixed in BF at 4°C (Tornusciolo, Schmidt, and Roth 1995). Although reduced in number, embryos fixed in BF at 4°C had significantly more false TUNEL-positive cells than embryos fixed in 4% paraformaldehyde solution (pH 7.4) (Tornusciolo, Schmidt, and Roth 1995). Thus, variability in reported apoptotic indices may arise as a result of temperature of fixation and chemical components of fixatives. Therefore, if the TUNEL assay is to be used to assess reproductive toxicity, optimization of fixatives and fixation procedures is crucial for reliability.
In the following report, we have compared TUNEL assay results following fixation of testes in the two recommended fixatives (BF or mDF) while controlling temperature of processing for each fixative (room temperature or 4°C). Liver was used as a control tissue for this study because, unlike testes, liver has a relatively stable cell population with few mitotic cells and significant amounts of connective tissue.
Materials and Methods
Fixatives
To prepare Bouin’s fluid (BF), 1.0 g hydrated picric acid crystals, 24 ml of 37% formaldehyde solution (9% formaldehyde) and 4.76 ml of glacial acetic acid (0.833 M) was added to 70 ml of de-ionized water for a total volume of 100 ml (Kiernan 1990). To prepare modified Davidson’s fixative (mDF), 30 ml of 37% formalin (11% formaldehyde), 15 ml of absolute ethanol (15%), and 5 ml glacial acetic acid (0.9 M) were added to 50 ml of de-ionized water and mixed (Latendresse et al. 2002). The pH of BF and mDF was 1.46 and 3.4, respectively. Methylene hydrate, the reactive form of formaldehyde in water, is slow to form at a lower pH (Kiernan 1990); therefore, both fixatives were prepared several days in advance of use.
Preparation of Slides
Sectioned testicular tissue is difficult to maintain on glass slides for extended immunohistochemical procedures and with proteinase K digestion. Therefore, slides were cleaned and coated (subbed) with chromate-collagen mixture to facilitate section adhesion. The chromate-gelatin mixture was prepared by following previously described method and was applied on the frosted side of each slide with a cotton swab (Shuttlesworth and Mills 1995). Subbed slides were air dried overnight at room temperature and stored in slide boxes under refrigeration until use. To reduce accumulation of moisture on the subbed surface, the stored slides were warmed to room temperature inside slide boxes before mounting of sections.
Animals
Ninety-day-old adult male Sprague-Dawley rats (Charles River Laboratories) weighing >400 g were housed in the Texas Woman’s University Vivarium and acclimatized with food and water ad libitum. For tissue collection, animals were anesthesized with Isoflurane® before sacrifice by decapitation. All procedures for animal care and use were in accordance with the National Institutes of Health guide for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee at Texas Woman’s University.
Processing of Testes and Liver Tissues
After anesthesia and exsanguination, tissues were quickly removed through an abdominal incision and immediately rinsed in chilled phosphate buffered saline (PBS). Intact testes were pierced superficially at 5 to 6 locations on both ends with a 23-gauge needle to facilitate fixative permeation (Latendresse et al. 2002) and liver was cut into 3- to 5-mm chunks. One testis was then immersed in either BF and the contralateral testis from the same rat was immersed in mDF with a tissue to fixative ratio of 1:10 (weight: volume) and alternately placed at either room temperature (23°C) or under refrigeration (4°C). Liver tissues from the same rats were fixed in BF or mDF at either room temperature or 4°C. All steps from tissue harvest through initiation of fixation of tissues were consistently completed in less than 10 min per animal. After immersion for 2 to 3 hr, tissues were trimmed into 3- to 5-mm pieces and fixation was continued for 24 hr either at room temperature or in the refrigerator. After fixation, tissues were dehydrated with ascending concentrations of ethanol (70%, 90%, and 100%) for 2 hr each, either at room temperature for those samples fixed at 23°C or in the refrigerator for those samples fixed at 4°C. At room temperature, all tissues were cleared by immersion in a 50:50 mix of ethanol:xylene for 2 hr followed by two changes in 100% xylene for 2 hr each. For paraffin embedding, tissues were transferred to a mix of 50:50 molten paraffin wax:xylene and incubated at 60°C for 1 hr followed by three changes of pure molten paraffin wax for 1 hr each at 60°C. Tissues were then transferred to warmed stainless steel molds for paraffin wax embedding. From each tissue block, 7 µm sections were cut with a rotary microtome and the wax ribbons were floated onto a 37°C water bath to permit section spreading. Using subbed slides, three serial sections were collected from the water bath and set by drying on using a slide warmer at 40°C for 15 to 20 min to promote adhesion. Slides were then placed in a slide box and stored under refrigeration until use.
TUNEL Assay
The TUNEL assay was performed using the ApopTag® Plus Peroxidase In Situ Apoptosis Kit (Cat # S7101; Millipore, Danvers, Massachusetts, USA) following the manufacturer’s instructions with modifications. Sections were de-waxed in xylene (three changes at 10-min intervals with air-drying in between each change for better section adherence) followed by rehydration in descending concentration of ethanol (100%, 90%, and 70%) for 15 min each at room temperature followed by a 5 min PBS wash. After rehydration, sections were treated with proteinase K (3.0 µg/ml) (Amresco, Solon, Ohio, USA ) for 3 min at room temperature to lightly digest the proteins around nuclei or DNA for better accessibility of the open 3′–OH ends of DNA in apoptotic nuclei. Following proteinase K digestion, endogenous peroxidase was quenched with 3% H2O2 (30% H2O2 diluted in PBS) for 10 min at room temperature. After washing H2O2 off with PBS (10 min at room temperature), the free 3′–OH DNA termini were enzymatically labeled with digoxigenin-conjugated and unconjugated nucleotide triphosphates (NTPs) added to the fragmented DNA by the TdT in a 37°C, humidified chamber for 1 hr. The TdT enzyme reaction was terminated after 1 hr by treating sections with the Stop buffer provided in the kit. Sections were washed thrice in PBS for 10 min each. Peroxidase-conjugated anti-digoxigenin antibody was added to sections and incubated in a humidified chamber for 30 min at room temperature. Excess unbound antibodies were removed by three PBS washes (5 min each). Labeled apoptotic cells were identified by treating the sections with the peroxidase chromogenic substrate 3,3′-diaminobenzidine (DAB) as recommended in the kit. Color development was monitored by placing slides on to a white paper and the reaction was terminated by placing slides in de-ionized water as soon as sections developed a brownish tinge. Counterstaining of nuclei was performed with Immunomaster’s Hematoxylin (American MasterTech, Lodi, California, USA) (diluted 1:10 with distilled water) by incubating the sections at room temperature for 10 min. For negative controls, TdT enzyme was replaced with PBS on one section on each slide and was processed in parallel.
Image Capture and Cell Counting
All images were captured using the Nikon’s eclipse 90i digital microscope at 9.2 volts and an exposure time of 4 mSec. TUNEL-positive cells were counted using Nikon’s NIS-Elements Basic Research version 3.1 software’s object count tool from photos in raw format. From each field, at least one TUNEL-positive cell was selected to specify the intensity of the objects to be counted with the 6 point circle tool of the software. The 6 point circle tool picks the threshold of the color intensity of the selected TUNEL-positive nucleus with the circle radius defined by 6 pixels. The threshold limits were from 30 to 40 (lower limit) and 120 to 140 (upper limit). The binary image was cleaned by performing 6 iterations to remove any nonspecific object(s) and the binary image contours were smoothed by performing 16 iterations. Data from 10 randomly selected fields were collected from each tissue cross-section for analysis. To prepare images for print, Adobe Photoshop CS 4 was used to format, crop, balance the brightness and contrast, and label each panel. The merged Photoshop image was imported into Adobe Illustrator CS 4 for applying arrows from the Illustrator Palette.
Statistical Analysis
The number of TUNEL-positive cells generated by the selected fixatives and fixation temperatures were compared by 2-way analysis of variance (ANOVA) with fixatives and temperatures being independent factors using SPSS version 15 software. Post hoc comparisons were performed with Newman-Keuls test and p-value ≤ .05 were considered to be significant (Zar 1999). All data are reported as mean ± standard error of mean (SEM) and graphs were prepared by SigmaPlot version 11 software.
Results
Prior to initiating the experiments, we optimized the proteinase K concentration for a minimal concentration (3.0 µg/ml) and duration (3 min) of incubation for better accessibility of fragmented DNA without compromising the tissue architecture. It has been reported that higher concentration (>5 µg/ml) (Tornusciolo, Schmidt, and Roth 1995) and longer duration (Stahelin et al. 1998) of proteinase K treatment caused (1) increased nonspecific TUNEL staining, (2) destruction of tissue architecture, and (3) loss of antigenic sites. The protocol of the kit used in this study suggests using 20 µg/ml of proteinase K for 15 min incubation prior to the TdT reaction. However, we observed that at these recommended conditions, it was difficult to maintain tissue architecture and staining was inconsistent irrespective of the type of fixative. Although proteinase K concentrations and period of the enzyme application to the tissue may be compromising factors in apoptosis studies, all samples analyzed for this report were treated exactly the same way and should not be a contributing factor observed in the current study.
Comparison of TUNEL Assay in the Testis
Testes fixed at room temperature in BF had significantly more TUNEL-positive cells (p ≤ .05) compared to either BF fixation at 4°C or mDF fixation at room temperature (Figure 1). When testes were fixed at room temperature in mDF, the number of TUNEL-positive cells was similar to that for testes fixed at 4°C in BF. However, testes fixed in mDF at room temperature had significantly more (p ≤ .05) TUNEL-positive cells than testes fixed in mDF at 4°C. The numbers of TUNEL-positive cells in testes that were fixed at 4°C with mDF are likely to more closely reflect the actual apoptotic cell numbers expected to be seen in normal testes (Figure 1; and Kerr et al. 2006). Therefore, higher values seen at room temperature or in BF are likely to be the result of false-positives due to the fixation-induced DNA damage. The more susceptible cell types for false-positive TUNEL staining were the type B spermatogonia at stages V–VI and early primary spermatocytes at stages VII–XIII (Figures 2B and 2D).
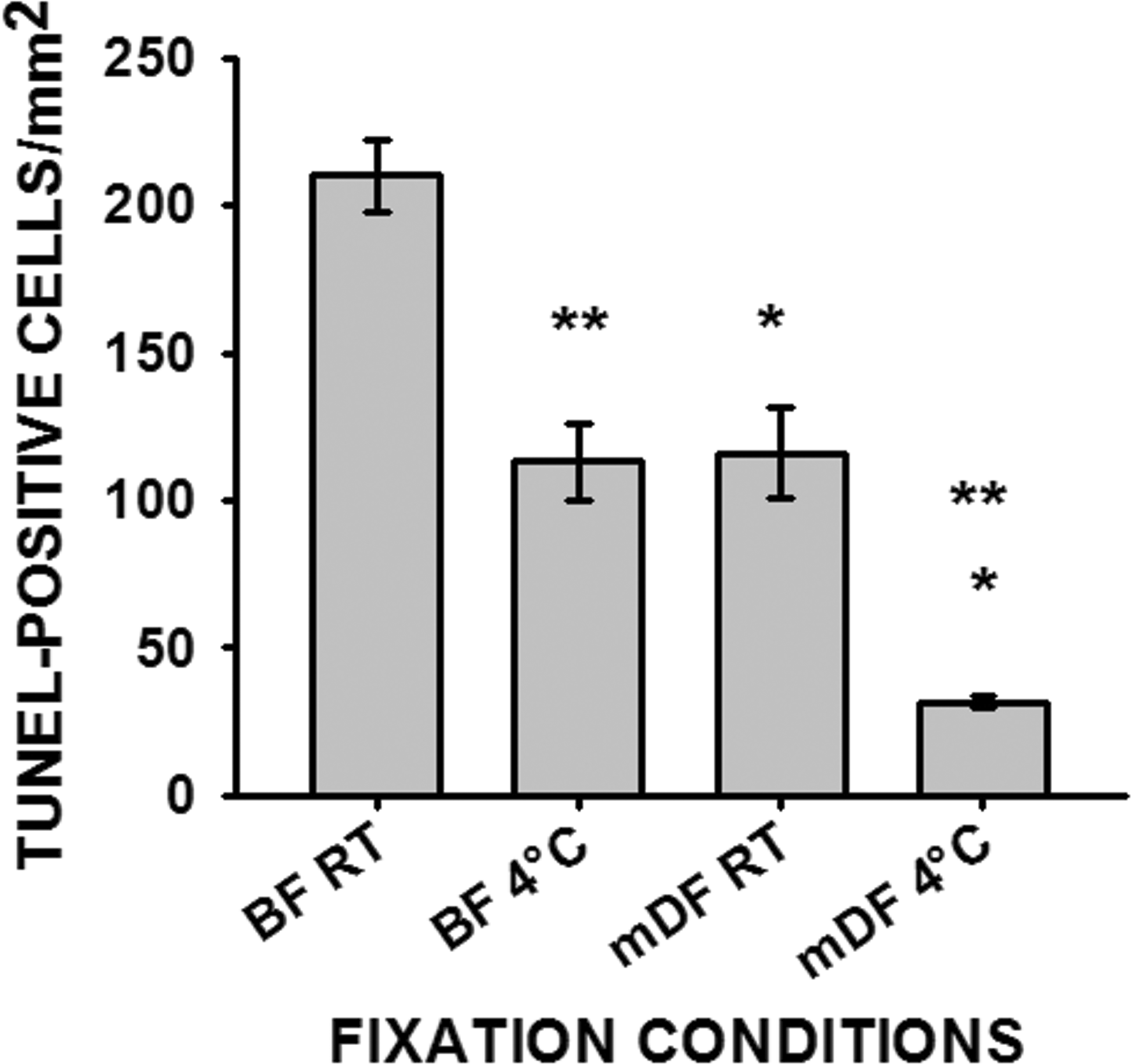
The number of TUNEL-positive cells counted from 10 random fields from each testicular cross-section and normalized to a 1.0 mm2 area. Data represented as average ± SEM (n = 3). Single asterisk (*) indicates significant difference between fixatives within same fixation temperature and double asterisks (**) indicate significant difference between fixation temperatures within same fixative. Room temperature (RT), Bouin’s fluid (BF), modified Davidson’s fixative (mDF).
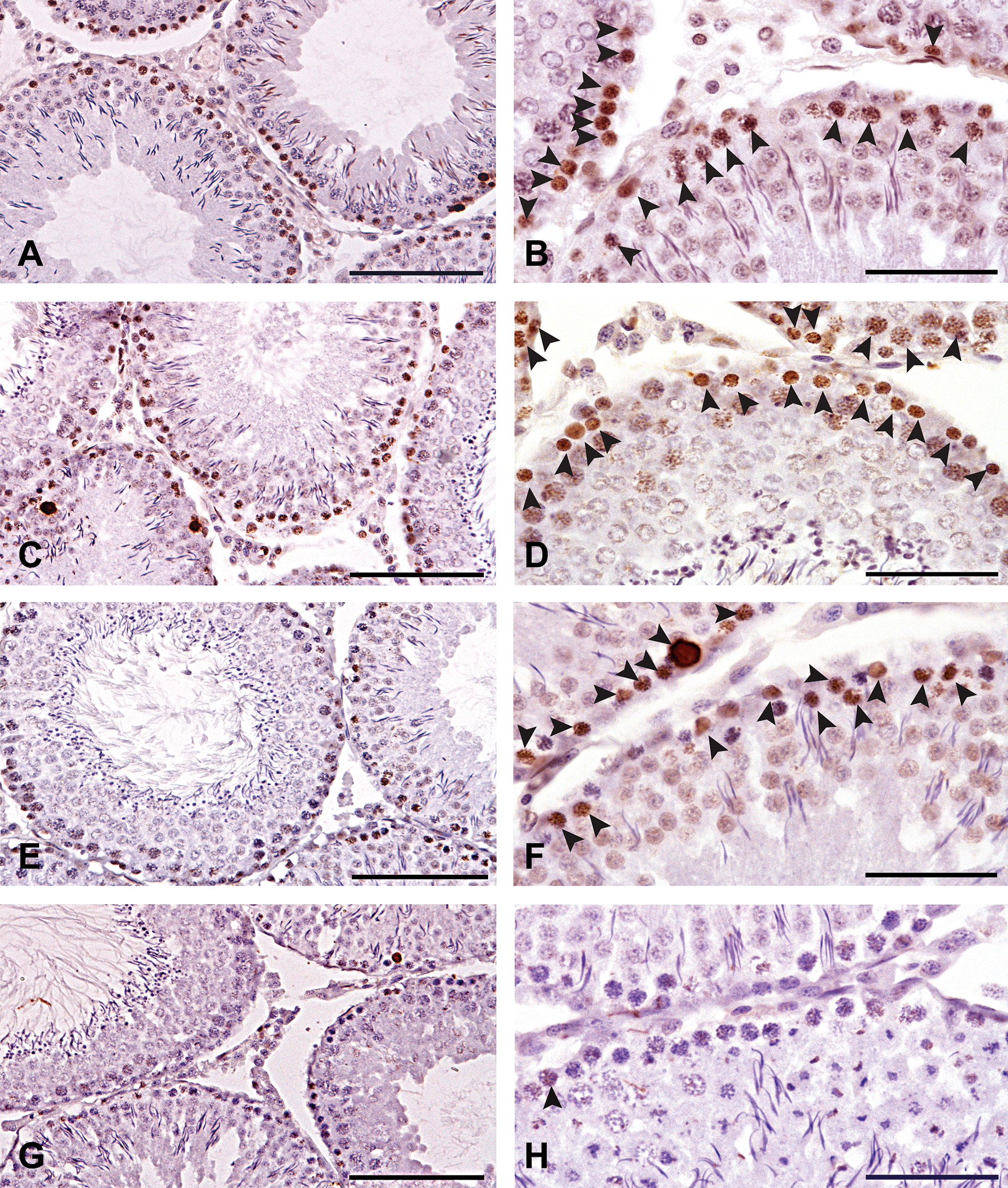
TUNEL-stained rat testes cross-sections counterstained with hematoxylin depicting TUNEL-positive cells inside the seminiferous tubules. Arrowheads indicate TUNEL-positive cells. Panels (A, B) BF RT; (C, D) BF 4°C; (E, F) mDF RT; (G, H) mDF 4°C. Panels A, C, E, and G scale bars are 100 µm and panels B, D, F, and H scale bars are 50 µm. Room temperature (RT), Bouin’s fluid (BF), modified Davidson’s fixative (mDF).
Comparison of TUNEL Assay in Liver
In liver, the type of fixative had a measurable effect on DNA integrity as determined by TUNEL staining, but no difference was found for the temperature of fixation (Figure 3). Livers fixed in BF either at room temperature or at 4°C had significantly higher numbers of TUNEL-positive cells compared to livers fixed in mDF at either of the fixation temperatures (Figure 4). The cells positive for TUNEL staining revealed a few hepatocytes, but most of the TUNEL-positive cells had smaller nuclei and were around the portal system (Figures 4A and 4B) (probably Kupffer and/or stellate cells). However, even when fixation occurred in BF, the number of false-positive TUNEL-stained cells/mm2 in liver was smaller compared to the number of false-positive TUNEL-stained cells/mm2 of testes fixed in BF.
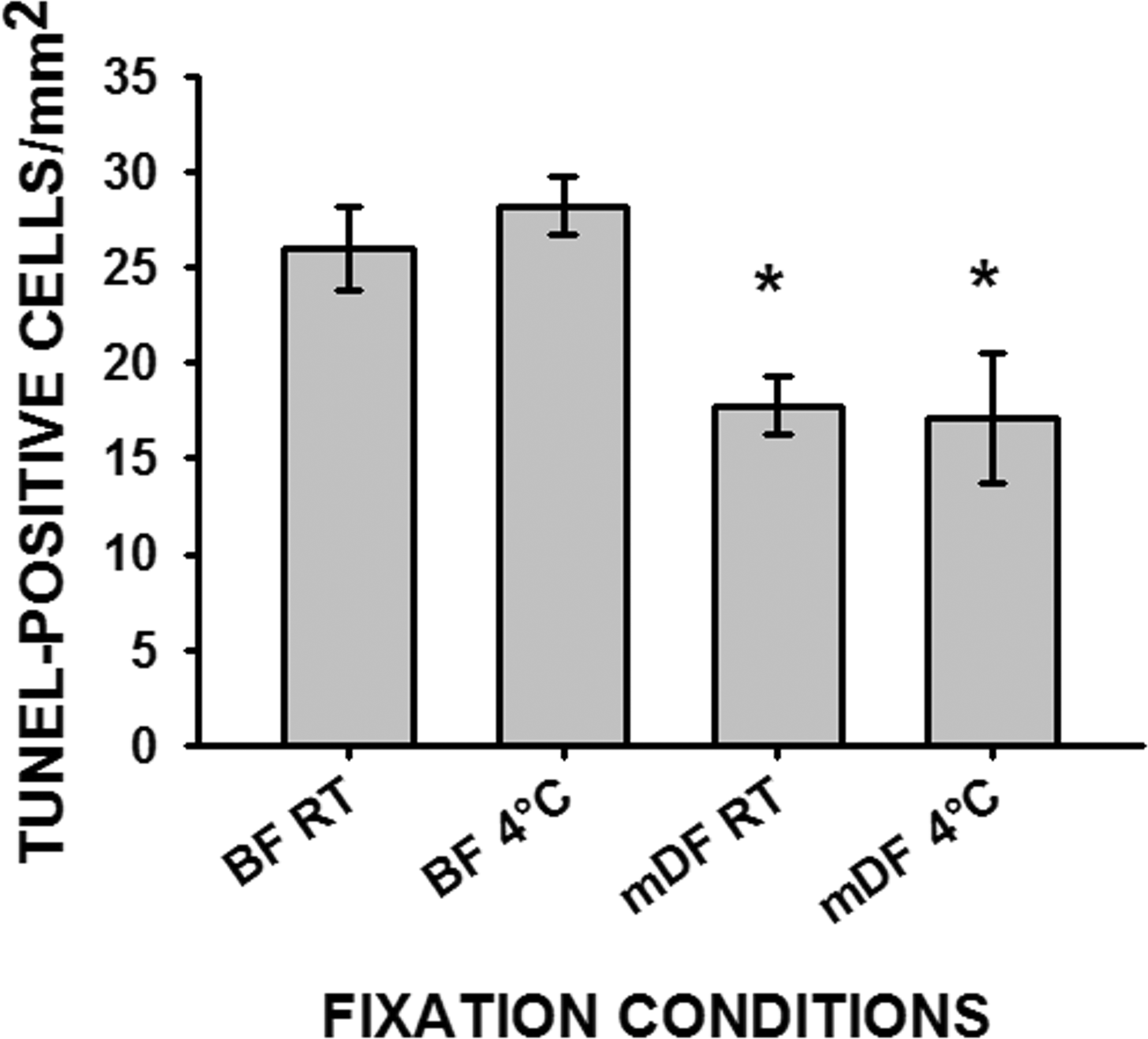
The number of TUNEL-positive cells counted from 10 random fields from each liver cross-section and normalized to 1.0 mm2 area. Data represented as average ± SEM (n = 3). Asterisk (*) indicates significant difference between fixatives within same fixation temperature. Room temperature (RT), Bouin’s fluid (BF), modified Davidson’s fixative (mDF).
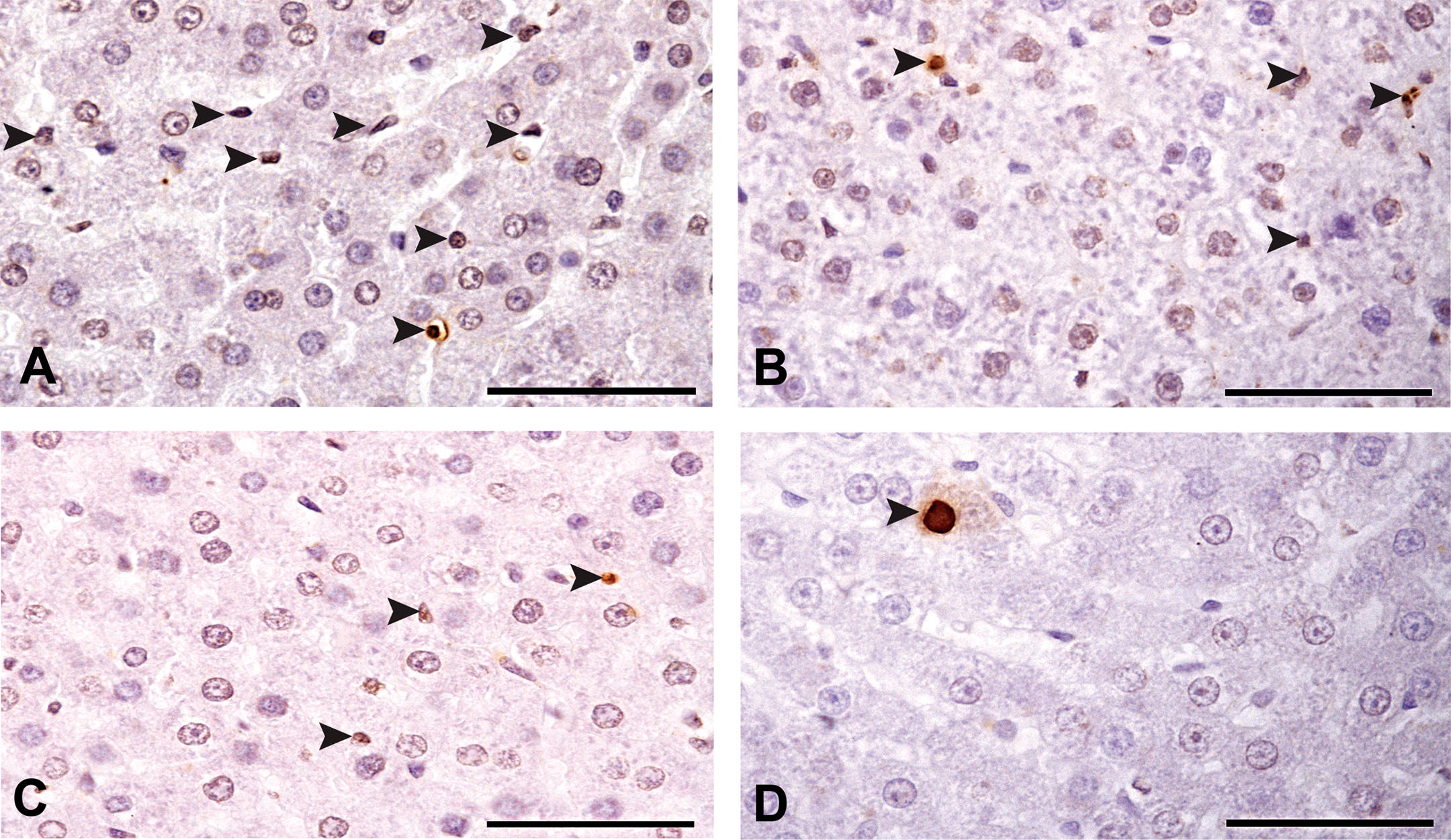
TUNEL-stained rat liver cross-sections counterstained with hematoxylin. Arrow heads indicate TUNEL-positive cells. Panels A–D are representative cross-sections depicting TUNEL-stained cells—scale bars 50µm (A) BF RT; (B) BF 4°C; (C) mDF RT; (D) mDF 4°C. Room temperature (RT), Bouin’s fluid (BF), modified Davidson’s fixative (mDF).
Discussion
Although the TUNEL assay is an accepted technique to detect DNA fragmentation for quantification of apoptotic cells, it does not differentiate cellular DNA fragmentation due to apoptosis versus chemically produced DNA fragmentation derived from conditions of tissue processing. Garrity et al. (2003) reported that improper, incomplete, or delayed fixation can generate false TUNEL-positive cells; and many reports exist of various efforts to optimize TUNEL assay protocols to overcome background problems (Tornusciolo, Schmidt, and Roth 1995; Labat-Moleur et al. 1998; Stahelin et al. 1998; Garrity et al. 2003; Taatjes, Sobel, and Budd 2008). The present study offers evidence that both the choice of fixative and temperature of fixation are important and this is essentially true for evaluation of apoptosis especially in testes.
Modified Davidson’s fixative reduced DNA strand breakage in testicular tissue when compared to Bouin’s fluid fixation at either room temperature or at 4°C. Lowering the temperature during fixation and dehydration was effective in reducing the number of false TUNEL-positive cells in normal testicular tissue using either BF or mDF. However, BF always had significantly greater numbers of false TUNEL-positive cells compared to mDF when evaluated at the same temperature. This may be due to the higher acidity of picric acid in BF, since stronger acids are reported to hydrolyze DNA (Kiernan 1990). However, both fixatives had substantial numbers of false TUNEL-positive cells in the testis when fixed at room temperature.
The DNA damage in testicular tissue fixed at room temperature in mDF is most likely due to the effects of formaldehyde since the pH was 3.4 or about 100 times less acidic than BF containing picric acid. Bouin’s fluid and modified Davidson’s fixative contain significant amounts of formaldehyde (9% and 11%, respectively) and formaldehyde is reported to slowly hydrolyze phosphodiester bonds, leading to fragments of polydeoxyribose with intact pyrimidines (Douglas and Rogers 1998; Srinivasan, Sedmak, and Jewell 2002). In addition, prolonged procedures for formaldehyde fixation at room temperature may result in DNA fragmentation due to formic acid generation and up to 30% of nucleic acids may be lost during such fixation, whereas tissues fixed in formalin at 4°C exhibited reduced degradation of nucleic acids (Tokuda et al. 1990; Srinivasan, Sedmak, and Jewell 2002). Here we have found that testes fixed in mDF at 4°C has little, if any, detectable DNA strand breakage from chemically or physically induced processes. Furthermore, the number of TUNEL-positive cells/mm2 was similar to numbers of apoptotic cells found in liver with mDF at 4°C, and this suggests that mDF used for fixation at 4°C is close to the apoptosis levels found in normal testicular tissues. A second observation made in these studies was that cells in different stages of maturation were differentially susceptible to fixation-induced elevation in number of TUNEL-positive cells. The cells that stained-positive for TUNEL were mostly from the earlier stages of spermatogenesis in either BF or mDF fixed testes at room temperature or BF fixed testes at 4°C. It is clear that the high numbers of TUNEL-positive cells observed in testes fixed at room temperature is a result of the fixation procedure, since the number of TUNEL positive cells in the contralateral testes fixed at 4°C was much lower. In addition, if apoptosis were to occur during normal “in-life” spermatogenesis at the rate demonstrated in the room temperature-fixed testes, it would rapidly result in testicular atrophy. In healthy adult rat testes, large numbers of cells do not undergo apoptosis, only a few spermatogonia and primary spermatocytes become apoptotic to purge potentially defective cells from the maturing germ cells (Kerr et al. 2006).
The increased numbers of TUNEL-positive nuclei in testes fixed with the harsher fixative conditions were primarily type B spermatogonia at stages V–VI and preleptotene, leptotene, zygotene, and early pachytene spermatocytes at stages VII–XIII of the cycle of the seminiferous epithelium. These cells, located on the inside perimeter of the seminiferous tubule, have DNA that may be especially vulnerable to chemical and physical strand breakage since they are replicating DNA and rearranging proteins in the first stages of meiosis. At stages V and VI of the seminiferous epithelium, type B spermatogonia are replicating DNA for the last mitotic division that gives rise to cells committed to meiosis. Cells at stages VII–XIII, mainly preleptotene, leptotene, zygotene, and early pachytene spermatocytes, are replicating DNA for meiosis and the newly synthesized DNA of meiotic cells is being rearranged with exchange of mitotic histones for meiotic histones leading to differentially organized DNA for meiotic chromosomal condensation (stages VII–XII) (Mills and Means 1977; Mills, Van, and Means 1977; Meistrich et al. 2003). Following chromosomal condensation, cells at stages IX–XII undergo crossing over (Perey, Clermont, and Leblond 1961). Thus, germ cells active in DNA replication and reorganization appear to have DNA that is more vulnerable to exhibiting false TUNEL-positive cells when fixed in harsher conditions.
Apoptosis in normal liver facilitates replacement of old or damaged cells (mainly hepatocytes) to maintain tissue homeostasis (Schulte-Hermann et al. 1993). The extent of DNA damage with BF and mDF was significantly less in liver than in testes, suggesting that cells of liver are less susceptible to the chemical and physical conditions of fixation when compared to testes. BF fixed liver still exhibited DNA damage in hepatocytes, Kupffer, and/or stellate cells at both room temperature and 4°C as compared to mDF fixed liver. Without specific markers, it is difficult to differentiate between Kupffer cells and stellate cells with only hematoxylin staining (Baratta et al. 2009). However, the localization and variable shape of these TUNEL-positive cells suggests them to be mostly Kupffer cells (Bloom and Fawcett 1975). Since Kupffer cells are phagocytic in nature and are responsible to keep liver free from foreign particles, they frequently undergo mitosis to maintain a substantial number (18%) (Baratta et al. 2009). Therefore, mitotic Kupffer cells may also be susceptible to fixation-induced DNA damage. However, unlike germ cells and Kupffer cells, cell division in hepatocytes is infrequent. This may explain the relative absence of false positive TUNEL staining in liver hepatocytes.
In summary, while the chosen fixation methods may vary with the type of tissue to be examined, it is important to consider the pH, fixation temperature, and fixative components to reduce fixation artifacts. Here, we have shown that if the TUNEL assay is to be used for studies of apoptosis in testes and liver, or in other tissues with high mitotic indices (e.g., tumors or embryos), it is preferable to choose mDF or a fixative (devoid of picric acid) with a moderate pH (3.0 to 4.0) or higher if the tissue integrity permits. In addition, fixation with mDF and dehydration of tissues should be carried out at lower temperatures (refrigerator, 4°C) if the TUNEL assay is to be performed.
Footnotes
Acknowledgments
We thank Dr. Lynda Uphouse for her help in statistical analysis and editing of the article and Dr. Mary Anderson for giving access to SigmaPlot software.
The Texas Woman’s University’s 2008–2009 Research Enhancement Program and Department of Biology provided funds for this research.
