Abstract
Although gastrointestinal (GI) toxicity is a significant dose-limiting safety concern noted in multiple therapeutic areas, there are no GI biomarkers that can accurately track, precede, or reliably correlate with histologic evidence of injury. While significant efforts have been made within the pharmaceutical industry, academia, and consortia to address the biomarker gaps in other target organs such as liver, kidney, and muscle (cardiac and skeletal), there have been no concerted efforts in the area of GI biomarkers. Using PAK4 inhibitor as a preclinical rat model of gastric toxicity, selected candidate biomarkers from literature were evaluated to test their usefulness as gastric injury biomarkers in this study. Biomarkers selected in this study include plasma diamino oxidase and citrulline, fecal calprotectin, bile acids, and miRNA. Based on the results, L-citrulline and miR-194 results appear to correlate well with histopathology findings. Although these biomarkers will need additional assay validation and qualification to test if they truly predict the injury prior to histopathology, the results provide promise for further testing using additional GI toxicants. In addition, this article highlights important gaps in GI biomarkers and provides substrate and rationale for additional investments either for further testing of already available biomarkers or to pursue extensive biomarker discovery approaches.
Introduction
Gastrointestinal (GI) toxicity remains a significant dose- limiting safety concern, especially in the oncology therapeutic area, as the majority of anticancer drugs target hyperproliferative cells and tissues such as the epithelial lining of the GI tract. Similarly, one of the dose limiting effects of non-steroidal inflammatory drugs (NSAIDs) is they can adversely affect the entire gastrointestinal system through local and systemic effects (Green 2001) causing epigastric pain, abdominal pain, constipation, and even ulceration (Higuchi et al. 2009).
Although several noninvasive biomarkers for GI toxicity exist, the available GI biomarkers are not technically validated, lack sensitivity and specificity, and have questionable translatability. However, early (preclinical) detection of GI toxicity is imperative to establishing GI safety profiles of compounds, guiding drug structure design and synthesis, establishing therapeutic indices, supporting decision making, and ultimately, reducing drug development attrition. Thus, identification of sensitive, convenient, and reliable GI toxicity biomarkers is rapidly increasing in importance.
At early stages of drug discovery and development, exploratory in vivo toxicity studies are performed for lead identification and clinical candidate validation. During exploratory toxicology studies, rodents (mouse/rat) and nonrodent species (dog, rabbit, guinea pig, and/or monkey) are used in conjunction with histopathology to identify morphologic and structural changes and characterize GI tract damage from compound treatment. While histopathology can profile GI toxicity in a temporal fashion and quantify the severity of damage, it has limited sensitivity and sometimes only identifies lesions at a late, irreversible stage. Thus, there are significant efforts under way within pharmaceutical companies to identify the most sensitive, specific, and reliable GI biomarker(s) that can be applied into routine exploratory toxicology and screening studies with the hope that such biomarkers translate into clinical GI toxicity prediction.
The goal of this article is threefold: first, to review reported GI toxicity biomarkers in the literature; second, to present recent preliminary findings on the potential GI biomarkers that can be applied preclinically; finally, to provide substrate for academic and pharmaceutical research efforts to develop validated and translatable GI biomarkers.
Literature Review
A number of GI toxicity biomarkers have been tested to investigate their usefulness to track adverse events. During this process, multiple analytical methodologies have been described for in vivo applications to track GI function in a number of species and in accessible biologic fluids such as blood and urine. Several gastrointestinal biomarkers that are investigated in scientific literature are discussed in the following sections.
Blood Biomarkers
Diamine oxidase (DAO)
DAO, a highly active degradative enzyme of the polyamine metabolic pathways, catabolizes a variety of substrates including histamine and diamines and is localized to the mature villus epithelial cells of rodent intestinal mucosa (Wolvekamp and de Bruin 1994). It has been reported that blood DAO activity level correlates with both DAO expression in the villi of the small intestinal mucosa and the severity of small intestinal mucosal lesions induced by anticancer drugs (Luk, Bayles, and Baylin 1980; Luk et al. 1981). Nevertheless, the measurement of blood DAO has been analytically difficult because of the extremely low level in the blood. Wolvekamp and de Bruin (1994) and Tsunooka et al. (2004) report that DAO measurement is confounded by the fact that plasma levels rise markedly upon heparin stimulation prior to blood draws, with peak elevations between 30 and 60 minutes. This renders blood DAO levels both impractical and inconvenient to incorporate into routine GI toxicity screening in a preclinical setting.
Citrulline
Citrulline is an intermediary metabolic amino acid produced mainly by enterocytes of the small bowel. Levels have been found to correlate with chemotherapeutically reduced enterocyte mass, independent of nutritional and inflammatory status (Crenn, Messing, and Cynober 2008). Similar to DAO, analysis of blood citrulline is also challenging, and routine analysis has been limited to enzymatic assays, which are often less sensitive. However, as Crenn, Messing, and Cynober (2008) postulate with the application of High Performance Liquid Chromatography (HPLC) and mass spectrometry, it is becoming a promising GI biomarker. It is important to point out that alteration in diet and gut microflora may potentially alter biochemical and metabolic pathways, leading to changes in metabolite measurements such as citrulline. However, the impact of these variables in this study is minimal or less likely considering the design of these experiments with uniform diet, age, and sex. Also, because of the inherent specificity of the LC/MS/MS method employed in this study to measure citrulline, the likelihood of an interfering metabonomic sample component that would bias citrulline measurements would be insignificant.
CD64
Tillinger et al. (2009) proposed flow cytometric measurement of neutrophilic CD64 as a biomarker for gastroenterologic diagnostics in patients with inflammatory and functional diseases of the intestine. CD64, a high-affinity FcRI receptor for IgG, serves as a ligand for acute phase reactants c-reactive protein and amyloid P (Tillinger et al. 2009). Because CD64 responded to therapeutic interventions and was significantly higher in patients with inflammatory bowel disease (IBD), it could serve as a biomarker to discriminate between gastroenterologic disorders. However, only mouse antibodies are available at this time, limiting our ability to evaluate CD64 in a rat preclinical in vivo toxicology study.
Gastrin
Gastrins are a family of sequence-related carboxyamidated peptides produced by the endocrine G cells of the gastric antrum and duodenum in response to a number of digestion-associated stimuli. The mechanisms for the release of gastrin from G cells are not entirely understood, although antral distension, partially digested proteins, amino acids, and vagal stimulation seem to contribute to its release. Increased serum gastrin levels are associated with duodenal ulcers, bacterial infections, tumors and a variety of other causative factors associated with GI damage. Fattaha and Abdel-Rahman (2000) measured serum gastrin levels in rats treated with omeprazole and studied the effects of omeprazole on both normal and ethanol-damaged gastric mucosa. Serum gastrin levels increased significantly in omeprazole treated animals versus control or ethanol-treated rats. Also, it is known that chronic use of antisecretory drugs may induce hypergastrinemia by inflammatory mechanisms and the release of gastrin from antral G cells is closely regulated by neuroendocrine mechanisms that may be sensitive to local pH. For example, gastric lumen acidity decreases gastrin section through the release of somatostatin from D cells (Alvarez et al. 2007). Sun et al. (2002) observed increases in plasma gastrin levels in rats treated with acetic acid, which is known to induce gastric ulcers. Similarly, Garcia-Sancho et al. (2005) have also demonstrated increased serum gastrin concentrations in dogs with chronic lymphocytic-plasmacytic enteritis compared with dogs without GI disease.
Orally Administered Probes as GI Functional Biomarkers
Urinary excretion of orally administered probes has been used to assess mucosal permeability as a marker of GI tract dysfunction and drug-induced intestinal barrier damage. Yanez et al. (2003) reported that urine sucrose and 51Cr-EDTA measured by radioimmunoassay correlated with gross toxicological and pathological changes in a rat model of gastrointestinal damage induced by various anti-neoplastic agents could be used as gastroduodenal and intestinal permeability biomarkers. Owing to radioactivity, 51Cr-EDTA is confined to animal studies and is often not practical (Butler 2008). Bjarnason, MacPherson, and Hollander (1995) have discussed D-xylose and 3-oxy-methyl-D-glucose (3-OMG) as additional permeability functional biomarkers to reflect intestinal epithelial absorptive capacity and rhamnose/3-OMG ratio to assess intestinal transport function. However, Butler (2008) claims these tests only measure small intestinal leakiness and additionally require HPLC. The 13C sucrose breath test (SBT) is another assay used to evaluate the absorptive capacity of the small intestine after ingestion of an isotope substrate and has been applied in several animal models of chemotherapy-induced intestinal damage (Butler 2008). Pelton et al. (2004) investigated changes due to chemotherapy-induced small bowel damage in the rat utilizing SBT and demonstrated significant impairment in 13CO2 exhalation, indicating decreased sucrose activity in the small intestine after 4 days. Butler, Tivey, and Davidson (2008) postulate that SBT is appropriate if hydrogen-producing bacteria are present in the colon. They report a significant proportion of human and animal populations does not have hydrogen-producing bacteria in the large intestine, which increases the likelihood for false negative results. According to the authors, because the SBT is qualitative, it does not give an estimate of the degree of damage or dysfunction.
Fecal Biomarkers
Fecal calprotectin
Fecal calprotectin is reported as a sensitive biomarker of intestinal inflammation (Poullis et al. 2004; Konikoff and Denson 2006). Calprotectin, a 36.5 kD non-glycosylated protein, constitutes over half of the soluble cytosol proteins in neutrophil granulocytes and plays a central role in neutrophil defense, and its fecal level correlates well with the numbers of neutrophils infiltrating the intestinal mucosa and the overall severity of GI inflammation. It is known to be stable in feces for several days after excretion (El Saadany 2008). However, the presence of calprotectin in feces appears to be directly related to neutrophil infiltration rather than GI damage.
Fecal bile acids
The cathartic effect of excessive fecal bile acids may be an unappreciated cause of chronic diarrhea in several clinical settings (Porter et al. 2003). Bile acids are minimally absorbed in the proximal small intestine, and the bile acid pool flows to the distal ileum, where the acids are reabsorbed by the enterhepatic transport system and then returned to the liver by the portal vein (Westergaard 2007). Remaining bile acids are excreted in feces. The bile acids expelled in feces are a complex mixture of metabolites of biliary bile acids produced by intestinal microorganisms (Hirofuji 1965). As a result, increases in fecal bile acids are indicative of malabsorption, which can cause diarrhea. However, the ability of fecal bile acids to accurately detect GI damage and/or decreased GI functionality is questionable based on assay reliability.
Miscellaneous fecal biomarkers
Fecal occult blood tests, fecal proteolytic activity, and fecal fat excretion measurement is possible, although their value for gastric injury assessment is questionable based on the assay, difficulty in the interpretation of the results, and logistical challenges. For example, the Guaiac fecal occult blood test has been available for many years but lacks sensitivity. Fecal protease activity varies in different portions of the fecal specimen and significant day to day as well as diet-related alterations in fecal protease activity occur. This variability necessitates collection of feces at different days with pooling of fecal collection. Furthermore, it is known that the protease activities in the feces are labile resulting in logistical challenges.
MicroRNAs (miRNAs)
miRNAs are small, endogenous noncoding RNAs that act as post-transcriptional regulators of gene expression (Boyd 2008). As emerging biomarkers of several organ toxicities, miRNAs are being evaluated for their potential to track damage to the gastric and intestinal mucosa. Upon cell death, miRNAs are released into the surrounding environment and are stably present in the circulation (Mitchell et al. 2008; Gilad et al. 2008). Therefore, detection of tissue-specific or tissue-enriched miRNA in biofluids might be used as a biomarker of damage of that tissue. As such, serum miRNAs have been identified as biomarkers for liver toxicity (Wang et al. 2009) and myocardial injury (Adachi et al. 2010; Cheng et al. 2010), as well as for lung, colorectal, ovarian, and prostate cancers (Chen et al. 2008; Lodes et al. 2009; Mitchell et al. 2008). Urine miRNAs that are kidney-specific have been found to indicate renal tubular damage (manuscript in preparation). Finally, a fecal miRNA assay is being developed as a screening tool for sporadic human colon cancer and active ulcerative colitis (Ahmed et al. 2009). miR-194 was chosen for initial experiments in this study since it is highly expressed in small intestine and colon, although it is not entirely specific (Hino et al. 2008; Godwin et al. 2010; Lee et al. 2008). Therefore, the presence of GI-specific or GI-enriched miRNA in biofluids or feces could be indicative of GI toxicity.
Taken together, identifying, investigating, and validating sensitive, reliable, and predictable noninvasive biomarkers are likely to be very beneficial for GI toxicity profiling, not only in preclinical drug development but also to enable selection of treatment regimen and dose. In addition, application of these biomarker assays will allow longitudinal, noninvasive monitoring of GI tract damage and repair following compound/drug treatment.
Materials and Methods
To explore the utility of available biomarkers from literature to monitor GI toxicity, an experimental design using p21-activated kinase 4 (PAK4) inhibitor (a known GI toxicant) was utilized. Blood and fecal samples from this study were employed to assess selected GI biomarkers and to correlate them with histopathologic findings. The analytes for measurement from this study were selected based on literature findings and assay availability.
Animals and Treatment
Wistar Han rats (n = 8/group) from Charles River Laboratories (Hollister, California) were administered PAK4 inhibitor orally once daily over a 5-day period at two doses (5 or 15 mg/kg) to induce GI toxicity. The control groups were administered vehicle alone (50 mM sodium acetate at pH 4.5). The dose and time duration of treatment were based on previous experience of GI toxicity with this compound and treatment regimen. Clinical signs were observed daily and body weights were measured on days 1, 3, 5, and 6 prior to termination. Approximately 1.5 mL of blood was collected in EDTA anticoagulant via lateral tail vein or inferior vena cava (at termination) for biomarker analysis. Blood samples were collected from all rats at predose, day 3 (∼48 hours post–1st dose), and on day 6 (at termination). Fecal samples were collected daily. Following the final treatment, animals were fasted for 20 hours and euthanized at 24 hours postdosing. Representative samples of the GI tract were collected, fixed in 10% (v/v) buffered formalin, and embedded in paraffin
Stomachs and ceca were removed from all rats. Stomachs were opened along the greater curvature to expose the mucosal surface, and ceca were flushed with 0.9% saline. Tissues were then incubated in 16 mmol/L HEPES buffer containing 125 mmol/L NaCl, 3.5 mmol/L KCl, 10 mmol/L glucose, and 1 mmol/L nitroblue tetrazolium for 20 minutes. Stomach and cecum were then examined for positive (blue) staining and for general evaluation of lesions. Microscopic examination of tissues was performed on all animals by a board certified veterinary pathologist. Animal care and maintenance were in accordance with the principles described in the guide for care and use of laboratory animals in NIH Publication No. 85-23 (1985). Animals were single housed in a controlled environment with constant temperature (21°C) and a 12-hour light/dark cycle. Food and water were available ad libitum.
Plasma L-Citrulline Assay
Citrulline was quantified by liquid chromatography tandem mass spectrometry (LC/MS/MS) in the selected reaction monitoring (SRM) mode on a Waters Micromass (Milford, MA) Quattro Premier tandem quadrupole mass spectrometer operated in the positive ionization mode. A deuterium-labeled isotope of citrulline (d4-L-citrulline, CDN Isotopes, Pointe-Claire, Quebec, Canada) was used as an internal standard. Samples were diluted 20-fold in deionized water, and then protein precipitation was performed with acetonitrile (J.T. Baker, Phillipsburg, NJ) and injection of 10 μL of supernatant into the LC/MS/MS system. For the analytes of interest, the following precursor-to-product ion transitions were obtained and optimized through direct infusion of each compound into the mass spectrometer: citrulline (Sigma, St Louis, MO) mass-to-charge ratio value (m/z) 176→70 and d4-citrulline m/z 180→74. Chromatographic conditions for the method consisted of a Shimadzu (Columbia, MD) HPLC system and a Waters Atlantis HILIC silica HPLC column. The mobile phase consisted of 0.1% formic acid (Fluka, Steinheim, Germany) and acetonitrile with the HPLC pumps programmed to deliver a linear gradient at a flow rate of 0.3 mL/minute.
Blood Diamine Oxidase Assay
A commercially available ELISA-based method (DAO Rodent ELISA Kit, Wuhan USCN Sciences, Ltd Cat. #E0656r) containing microplate wells precoated with a biotin-conjugated polyclonal antibody specific to DAO was employed. Standards or samples were added to the microplate wells. Avidin conjugated to horseradish peroxidase (HRP) was then added to each microplate well and incubated. Finally, a TMB (3, 3′5, 5′ tetramethyl-benzidine) substrate solution was added wherein those wells that contain DAO, biotin-conjugated antibody and enzyme-conjugated avidin exhibited a change in color. The enzyme-substrate reaction was terminated by the addition of a sulfuric acid solution and the color change was measured spectrophotometrically at a wavelength of 450 nm ± 2 nm. The concentration of DAO in the samples was then determined by comparing the O.D. of the samples to the standard curve.
Measurement of Fecal Bile Acids
Bile acids include a large class of sterols which differ widely in their physiochemical and biological properties. Because of these differences, analytical methods for their measurement must account for both the type of bile acids present in a biological fluid as well as the complexity of the biological matrix. Bile acids in fecal matter are comprised primarily of 3α-hydroxy-bile acids that need to be extracted from feces using an organic solvent (DeWael, Raaymakers, and Endeman 1977; van der Meer, deVries, and Glatz 1985). The extracted fecal bile acid can then be subjected to enzymatic cleavage for quantification.
Animals were single housed in metabonomic caging designed to separate the solid feces from urine, through a funnel-like system. In instances where liquid/soft feces were observed, samples were collected in the funnel. Thus both solid and liquid feces were obtained. Samples were weighed while frozen, then diluted with water. Water was added based upon stool consistency which averaged to approximately 3 mL of water per gram of feces. With soft stools, less water was added to the sample. The fecal/H2O mixture was allowed to thaw overnight at 4°C and then homogenized until a thick smooth texture was achieved. 1.4 ± 0.05 grams of homogenate were removed and approximately 2.6 mL of t-ButOH/H2O was added to the homogenate before vortexing thoroughly to mix the solution. The solution was incubated at 37 ± 2°C for approximately 1 hour and vortexed again until thoroughly mixed. Samples were then centrifuged at 1,800–2,500 rpm for 15–20 minutes. The resultant supernatant was then assayed for bile acid content using a 3α-HSD (hydroxysteroid dehydrogenase) enzyme assay on the Hitachi 911 automated chemistry analyzer. 3α-HSD oxidizes 3α-hydroxy-bile acids, which make up the majority of bile acids present in feces. During this oxidative reaction, an equimolar quantity of nicotinamide adenine dinucleotide (NAD) is reduced to NADH. The NADH is subsequently oxidized to NAD with a concomitant reduction of nitroblue tetrazolium salt (NBT) to formazan by the catalytic action of diaphorase. The absorbance is read spectrophotometrically, and the intensity of the color produced is directly proportional to the bile acid concentration in the sample. The results are then calculated in reference to a standard curve generated using taurocholic acid.
Fecal Calprotectin Assay
Fecal pellets from rats were obtained in overnight collection and homogenized in a buffer solution. Analysis was performed by the commercially available Hbt human Calprotectin ELISA kit (Hycult Biotechnology, Cat.# HK325), which is based on the sandwich principle, where standards and unknowns are captured by a solid bound heterodimer-specific antibody. Biotinylated tracer antibody binds to captured calprotectin and streptavidin. Strepavidin conjugates then binds to the biotinylated antibody, which in turn conjugates with TMB. The substrate reaction was terminated by the addition of oxalic acid and color change was measured spectrophotometrically at a wavelength of 450 nm. Two standard curve points (0.8 and 0.4 ng/mL) were added in an attempt to capture any potential sensitivity issues using a human kit for rat feces.
Fecal miRNA
One gram of feces was homogenized in 3 mL water, and total RNA was extracted using Trizol LS (Invitrogen, Carlsbad, CA) and mirVana RNA isolation kit (Applied Biosystems, Foster City, CA). qPCR was performed on the purified RNA using TaqMan microRNA assays (Applied Biosystems) according to the manufacturer’s instructions. Following homogenization of 1 gram feces in 3 mL water, 1 gram was aliquoted for RNA extraction, with the assumption the aliquot contained 1/3 g of feces.
Briefly, 5 ul of RNA was used in a 15 ul RT reaction. Triplicate qPCR reactions were run using 1.33 ul of the RT on a StepOne Plus Real-Time PCR System (Applied Biosystems). Copies per reaction were determined using a standard curve of synthetic microRNA (Integrated DNA Technologies, Coralville, IA). The data are expressed as copies of miRNA/mg feces.
Results
Clinical Signs and Histopathology Findings
Clinical signs were observed on day 4 post-treatment in the 15 mg/kg group and included reduced activity, hunched posture, decreased skin turgor (suggestive of dehydration), and soft and watery feces. Treatment of rats with PAK4 inhibitor for 5 days resulted in grossly evident reddening and edema of the gastrointestinal tract (data not shown). Histologically, this correlated with neutrophilic inflammation, degeneration/atrophy, and necrosis affecting the nonglandular gastric mucosa, as well as multifocal gastric erosions and/or ulcerations (Figure 1B). Marked small intestinal lesions were also noted, characterized by crypt necrosis and accompanying villus atrophy and fusion, as well as attenuation of remaining epithelium (Figure 1D). There were no gastric or intestinal lesions noted in the vehicle-treated group (Figures 1A and 1C).
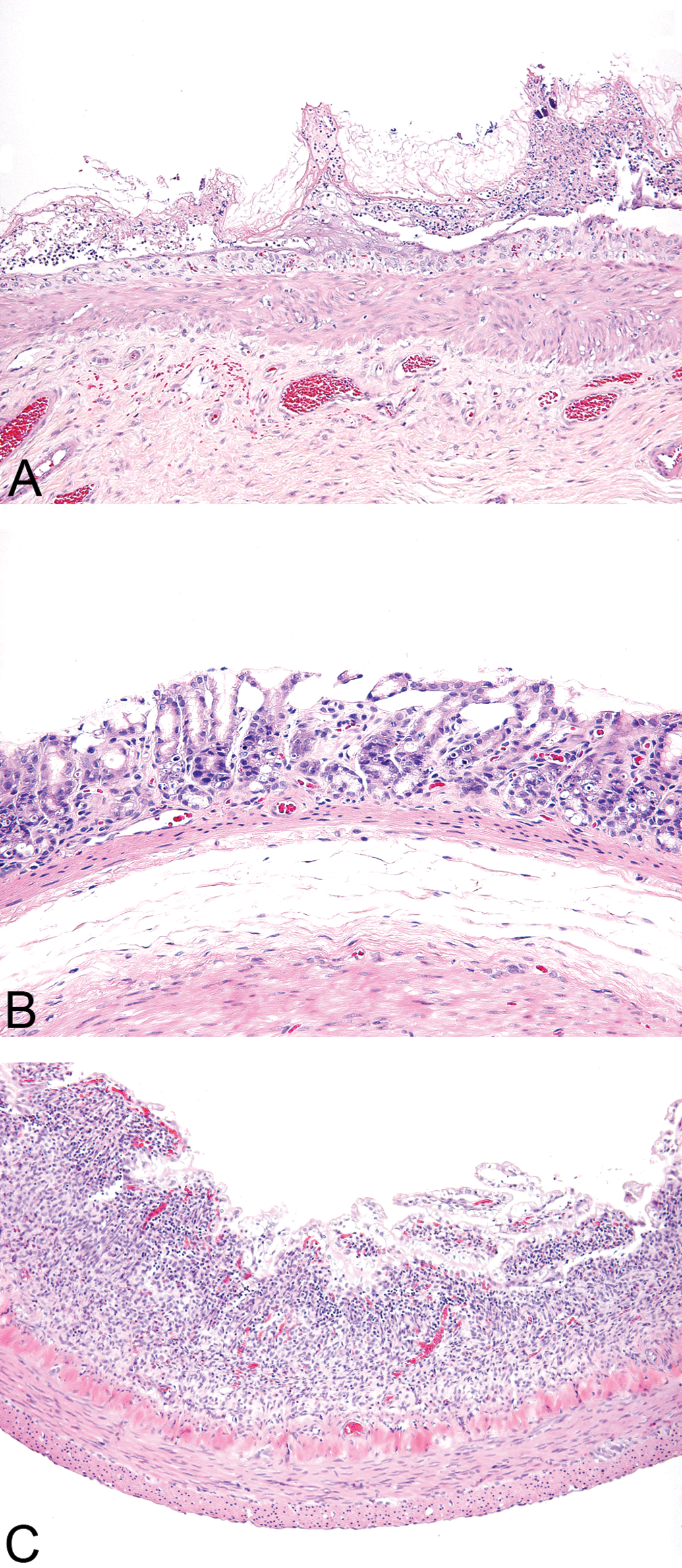
Histologic sections of nonglandular stomach and jejunum showing mucosal detail. (A) Nonglandular stomach from a rat treated with 15 mg/kg PAK4 inhibitor for 5 days. The mucosal epithelium is edematous, infiltrated by neutrophils, and exhibits necrosis and sloughing. 200x magnification. (B) Antral gastric mucosa from a rat treated with 15 mg/kg PAK4 inhibitor for 5 days. The epithelium is atrophied, and mild individual cell necrosis is present. 200x magnification. (C) Jejunum from a rat treated with
Plasma L-Citrulline Assay
Chromatographic retention times as evaluated by liquid chromatography for citrulline and d4-citrulline (internal standard) were 2.6 minutes (Figure 2A, 2B).
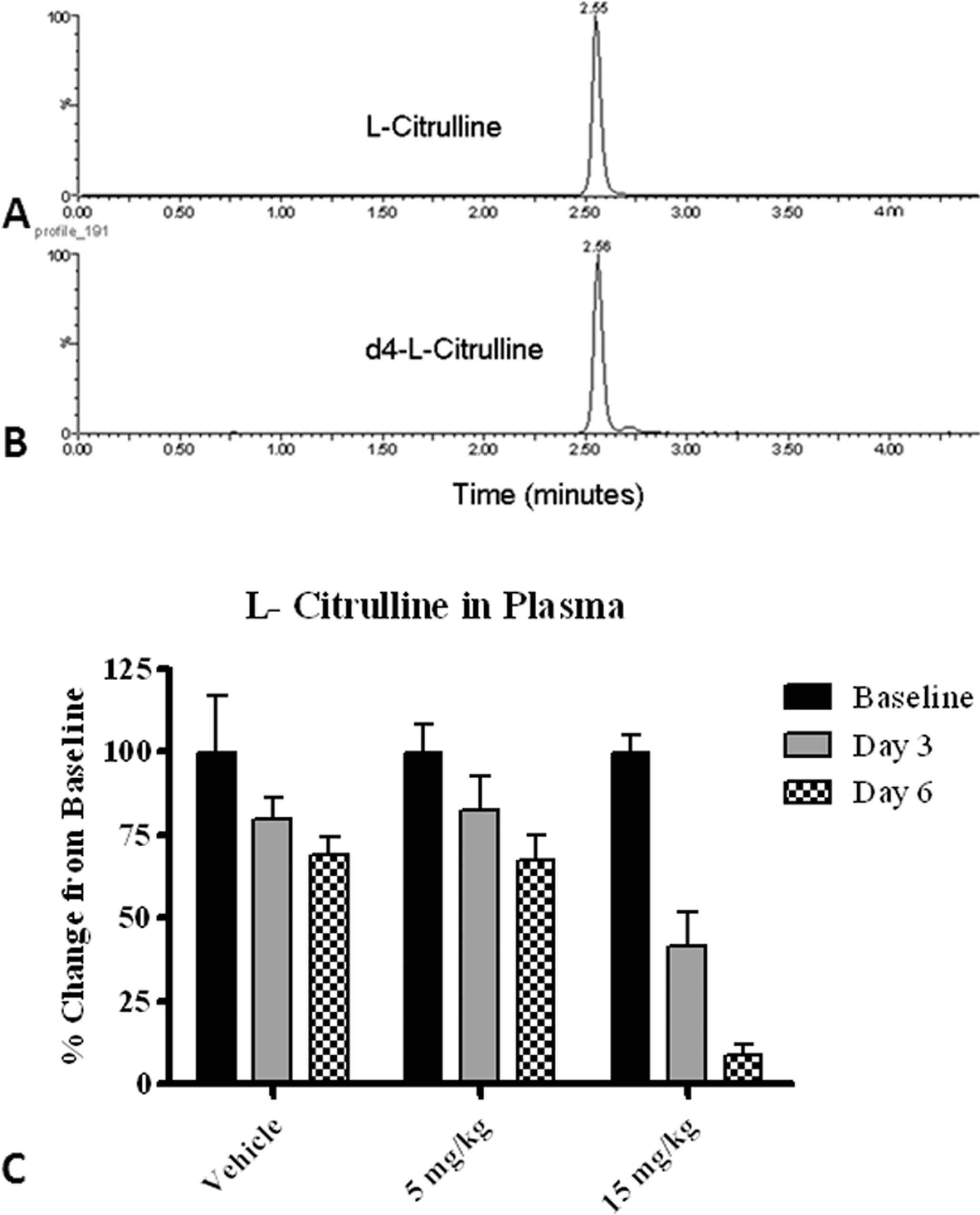
Plasma citrulline as a potential GI toxicity biomarker. (A–B) Chromatogram of citrulline (A) and d4-L-citrulline (internal standard, B) from a rat plasma extract using the LC/MS/MS method. The retention time of the analytes is approximately 2.6 minutes at a flow rate of 0.3 mL/minute. (C) Plasma L-Citrulline demonstrated a dose- and time-dependent decrease of ∼60% on day 3 and ∼90% on day 5 in the 15mg/kg treatment group compared with the baseline. Data are expressed as mean ± SD. Refer to experimental methods for additional details.
With a linear calibration range from 0.5 to 50 μg/mL for citrulline using aqueous based standards, quality control samples prepared in rat plasma demonstrated the assay to have an accuracy and precision error within 15%. Robustness of the assay was also confirmed through assessments of analyte recovery from different lots of plasma, freeze-thaw and sample storage stability, as well as selectivity from other potentially interfering components.
The results for L-citrulline levels in rat plasma detected by mass spectrometry (Figure 2C) showed a significant dose- and time-dependent decrease with a ∼60% decrease on day 3 and ∼90% decrease on day 5 in the 15mg/kg treatment group compared with baseline. The decrease on day 5 in the 15 mg/kg treatment group compared with baseline was consistent with an overall reduction in enterocyte mass, which manifested histologically as crypt necrosis and villus atrophy and fusion.
Blood Diamine Oxidase Assay
Using the commercially available rodent DAO ELISA kit, plasma DAO levels were less than 0.312 U/mL demonstrating no biological meaningful correlation with dose or findings.
Fecal Bile Acids
Fecal bile acid (FBA) levels determined by an automated clinical chemistry analyzer showed high variability but revealed peaks on days 1 and 4 in the 5 mg/kg group and a gradual increase with a peak on day 4 in the 15 mg/kg group (Figure 3). There was a lack of clear correlation between the histopathology findings and fecal bile acid values with the PAK4 inhibitor.
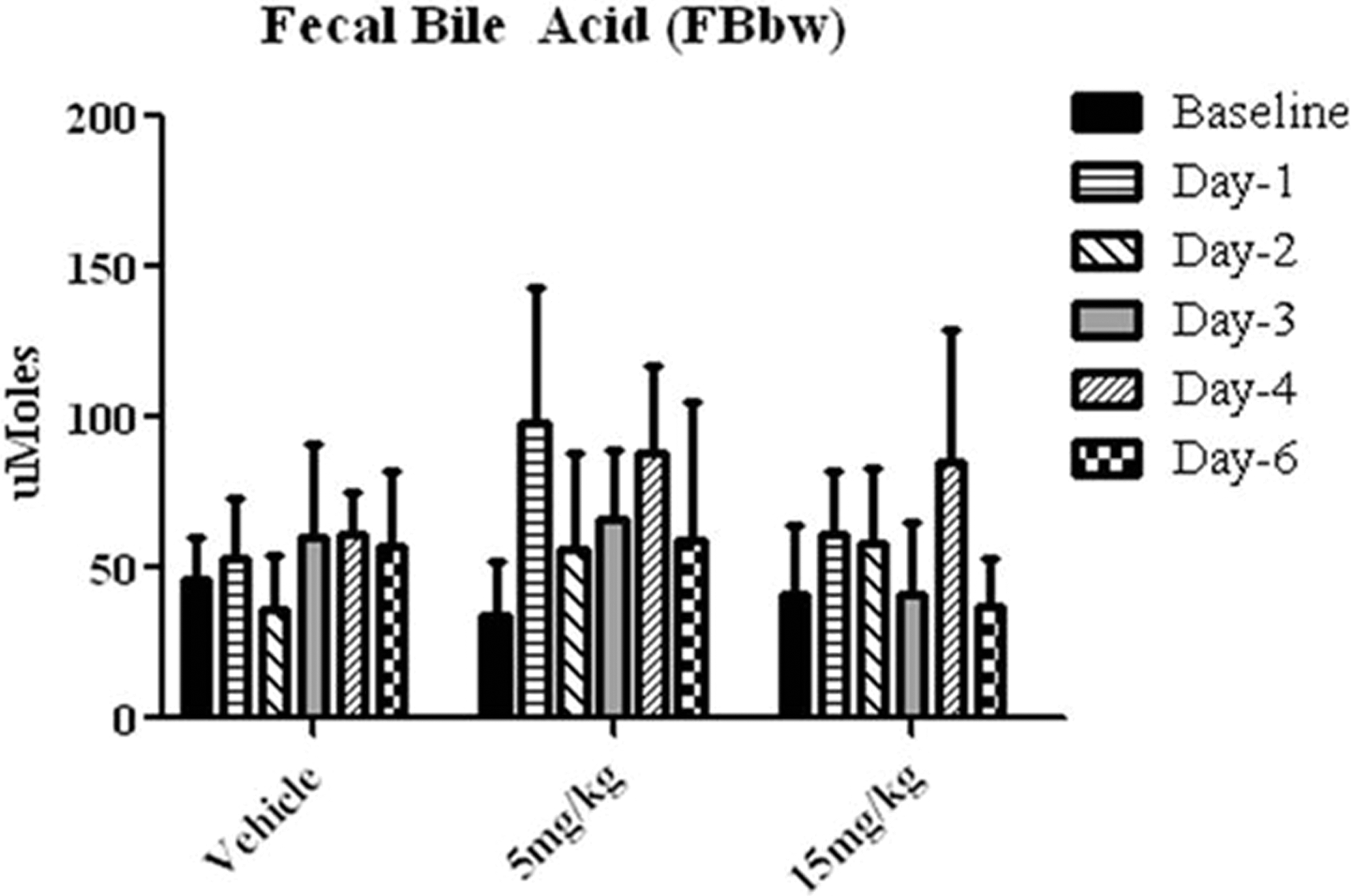
Fecal bile acid levels as measured by automated clinical chemistry analyzer at various time points following compound administration. Considerable variability was noted with fecal bile acid levels and no clear lack of correlation with histopathology findings. Data are expressed as mean ± SD.
Fecal Calprotectin Assay
Using the commercially available human fecal calprotectin ELISA kit, calprotectin was detectable at all time points. However, all values were <0.400 ng/mL (below the recommended minimum detection level of 1.6 ng/ml).
Fecal miRNA
In the predosing group and following days 1 and 2 of treatment, there was minimal or no miR-194 in the feces in any animal. However, on day 3, there was a spike in miR-194 in all animals PAK4-treated animals. In the low-dose (5mg/kg) group, after this initial spike, fecal miR-194 decreased to near baseline by day 5. On day 4, all animals in the high-dose (15 mg/kg) group showed a large decrease in fecal miR-194 with demonstrated increases again on day 5, correlating with histologic lesions in the GI tract. Although a spike in miR-194 was observed in the feces of low-dose animals, no significant clinical histological findings were observed in GI tissues at necropsy suggesting fecal miRNA might detect transient GI toxicity that would otherwise be missed. Interestingly, some vehicle control animals also showed transient increases in fecal miR-194 on day 3, 4, or 5 (Figure 4), again suggesting transient GI damage, possibly due to irritation from the vehicle or mechanical damage from gavage.
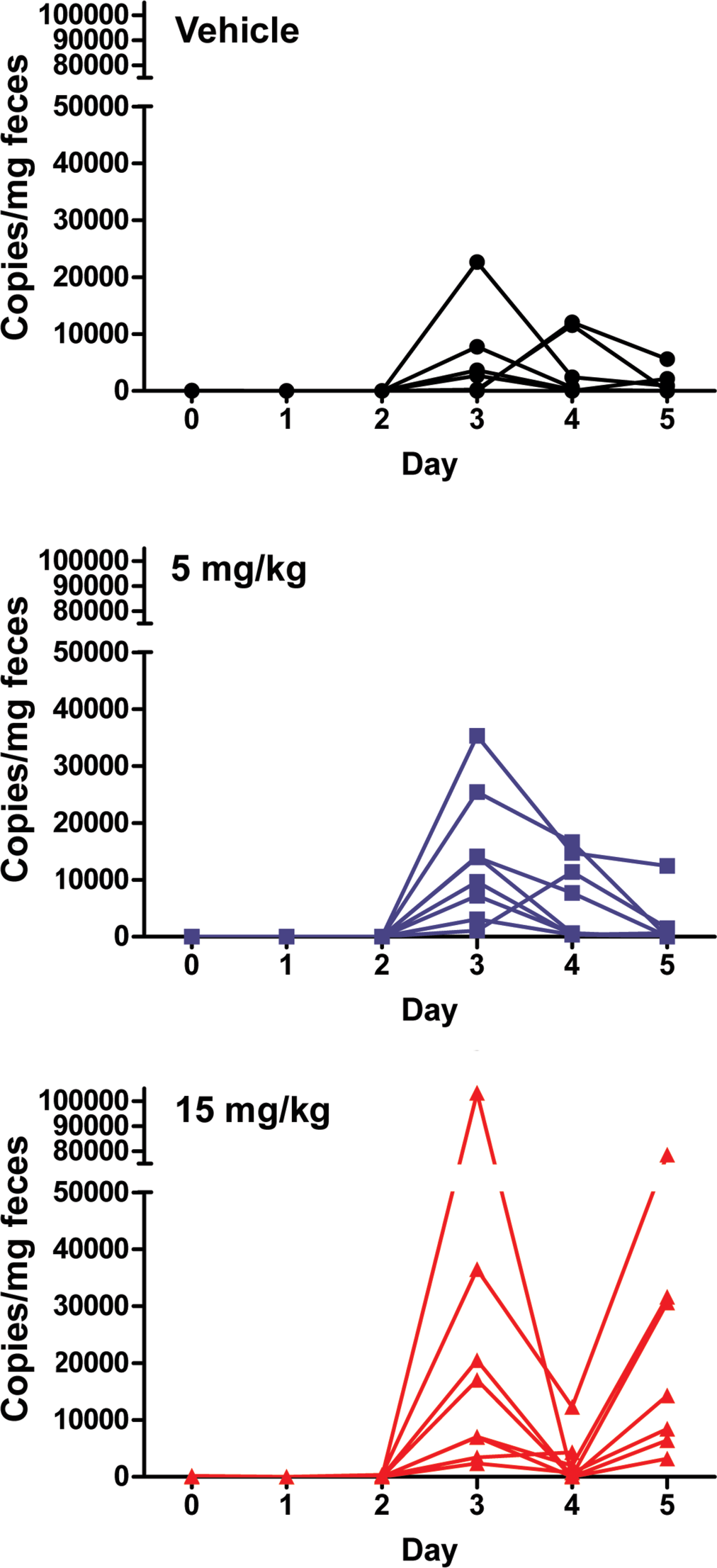
GI-enriched miR-194 is elevated in the feces of animals with GI toxicity. miR-194 was measured by qRT-PCR in the feces of control and treated animals. The compound was administered once daily for 5 days. Data are expressed as copies of miR-194 per milligram of feces. Each line represents miR-194 copies from individual rats within vehicle and treated groups. Two-way ANOVA revealed statistically significant (p < .01) effect of time, treatment, and their interaction on miR-194 levels. Bonferroni posttest identified statistically significant (p < .01) differences in miR-194 levels between 15 mg/kg and vehicle groups on days 3 and 5.
Discussion
Gastrointestinal toxicity is a common side effect during the drug development process, especially with kinase inhibitors. To generate samples to evaluate gastrointestinal biomarkers, we induced GI toxicity with a PAK4 inhibitor in rats, then confirmed this toxicity with clinical signs and histopathology. For this study, five analytes were selected from literature to test their usefulness as a GI biomarker mainly based on the assay reagents, platforms, and test utility. Of the five analytes, plasma L-citrulline assay using mass spectrometry appeared to be the most promising in predicting and correlating with GI injury; the plasma L-citrulline level changes were dose- and time-dependent and correlated well with histopathological findings. Also, preliminary fecal miR-194 data appeared to be promising based on the correlation with histopathology. As expected, fecal bile acid levels appeared variable over time but demonstrated peaks at certain treatment days although they were not dose-related. Plasma DAO and fecal calprotectin levels were detectable but below assay sensitivity despite adding two additional concentration levels on the low end of the standard curve. Although serum gastrin levels were not tested in this study, recent analysis of gastrin levels from a study with GI lesions in both rats and monkeys did not generate reliable data. Additional analytes that appear to have some potential as GI biomarkers that are not discussed in this article include plasma ghrelin, trefoil factor 3, and vitamin B12.
Based on these preliminary exploratory findings, our results provide some promise for identification of reliable GI biomarkers such as blood citrulline and fecal miR-194 levels. Caution should be exercised while interpreting this data, since these biomarkers are validated as “fit for purpose,” and results are generated without extensive assay validation and lack of adequate data to anchor these biomarkers to disease process.
Before these biomarkers are accepted, additional method validation such as determination of sensitivity, specificity, accuracy, and precision of the individual assays would be required. The assay validation should be clearly distinguished from “biomarker qualification,” which is an evidentiary rolling process of linking a specific biomarker to a phenotypic endpoint such as histologic correlate (e.g., GI damage). Clearly, additional prospective studies with multiple compounds, incorporation of additional time points (including recovery), and evaluation of assay performance characteristics are needed to accurately test the validity of these biomarkers to track GI damage. The level of assay validation will depend on the stage of drug development and thus would be “fit for purpose” as employed in this study (Lee et al. 2006). Relatively speaking, less time and investment are allocated to assay validation during biomarker discovery and early preclinical drug development as compared with during a regulatory toxicology study, clinical trial, or prior to regulatory acceptance. In fact, regulatory agencies provide guidance to effectively and proactively apply predictive and clinically translatable biomarkers during drug development (Goodsaid, Frueh, and Mattes 2008).
Several challenges exist for GI biomarkers in terms of measuring them in noninvasive biologic fluids such as blood, urine, or feces. First, if gastrointestinal damage does exist, it appears that based on the path of least resistance for entry of biomarkers into accessible biologic fluids, the intestinal biomarker is most likely to appear in the feces. However, it should be noted that measurement of biomarkers within feces is challenging based on the presence of microflora, ingesta, and metabolites, which necessitates extensive processing before analytical measurements. This can ultimately result in either loss of the biomarker or lack of assay sensitivity. As noted in this study, fecal calprotectin and DAO were below the detection limit for the assay. Also, fecal microbes are likely to further process protein biomarkers, resulting in the need for the measurement of both intact and processed protein analytes. Clearly, refinement in sample processing and/or sensitive platforms are needed when measuring analytes from feces. Furthermore, the mechanism of biomarker movement from the GI lumen to the blood is not always known. Therefore, unless there is clear compromise in the GI epithelial integrity, there may be little benefit to the measurements of these biomarkers within the blood. However, based on the preliminary data with PAK4, it appears that the measurements of blood biomarkers may be reliable and additional studies are needed to confirm this hypothesis.
Additional considerations when assessing GI biomarkers include the value of these biomarkers to address gastric versus intestinal lesions and whether the nature of GI lesions impacts the changes of these biomarkers (e.g., gastric/intestinal ulcerations and necrosis versus gastric inflammation). In addition, several biomarkers, such as bile acids and vitamin B12, change as a result of secondary changes, such as alteration of bacterial microflora and their metabolites. Thus, caution has to be exercised when interpreting such GI biomarker parameters. Also, it is not clear if there are correlations between biomarker changes and altered GI function or motility; this also needs to be tested.
For a gastrointestinal biomarker to be sensitive and of maximal utility during preclinical development and phase I clinical trials, it is imperative that biomarker changes occur prior to tissue damage and correlate with lesion severity. Additionally, a reliable GI biomarker needs to be specific, meaning such biomarkers have to originate from within the affected region, and studies are needed to demonstrate specificity by evaluating multiple organs as well as testing with non-GI toxicants. Future studies will further test these biomarkers by including additional time points, experimental designs, refined assays, and specificity analysis. To conclude, this article has reviewed and tested already-reported GI biomarkers in accessible biologic fluids using an in vivo study design with a known GI toxicant. Blood citrulline and fecal miR-194 appear to be promising to track GI toxicity, although additional assay validation is required. Significant limitations in assaying GI biomarkers is also discussed. Hopefully, this review will provide impetus and the needed substrate to academic institutions and pharmaceutical companies to recognize the significant biomarker gaps in this area so that collaborations are initiated for successful biomarker development.
Footnotes
Acknowledgments
The authors acknowledge the contributions of Drug Safety Research and Development La Jolla General Toxicology, Histology and Biomarker colleagues for successful conduct of the study and assay support.
