Abstract
This study was designed to investigate the toxico-pathological effects of in ovo inoculation of ochratoxin A (OTA) in chicken embryos and subsequently in the hatching chicks. Nine hundred fertile white leghorn (WL) layer breeder eggs were divided into eight groups (A–H). Group A was maintained as untreated control, whereas group B was kept as sham control (10 µL of 0.1 M NaHCO3 solution). Before incubation, groups C, D, E, F, G, and H were injected with 0.01, 0.03, 0.05, 0.10, 0.50, and 1.00 µg OTA/egg, respectively. At 53 hrs of incubation, crown to rump length, optic cups, and eye lens diameters were significantly (p ≤ .05) lower, whereas neural tube closure defects were higher in the OTA-treated embryos. Teratogenic defects (studied at day 9 of incubation) and embryonic mortalities were higher in the groups administered high doses of OTA. A significant increase was noted in the serum concentration of ALT, urea, and creatinine, along with higher weights of liver and kidney, in chicks hatched from OTA-contaminated eggs. These findings suggested that there are teratogenic and substantive toxicological risks in the developing chicken embryos and hatched chicks that could be exposed to OTA in ovo.
Introduction
The widespread occurrence of mycotoxins producing fungi and their ability to grow on the variety of economical important cereal crops have rendered them as unavoidable contaminants of animal and human food and feeds. In the long list of more than 300 known mycotoxins, the most important for human and animal health significance are ochratoxins, aflatoxins, T-2 toxin, zearalenone, fumonisins, and deoxynivalenol (Binder et al. 2007; Devegowda, Raju, and Swamy 1998).
The presence of ochratoxin A (OTA), a mycotoxin well known for its nephrotoxic activities, has been reported in finished poultry feed and its ingredients throughout the world, including Pakistan (Saleemi et al. 2009; Hanif et al. 2006; Rehman, Bhatti, and Hanif 2003). Besides its hepatotoxic, immunosuppreseant, teratogenic, neurotoxic, and mutagenic effects (Zahoor-ul-Hassan et al. 2010; Wangikar et al. 2007; Elaroussi et al. 2006; Sava et al. 2006; Kumar et al. 2004; Gilani, Bancroft, and Reily 1978), it has also been classified by the International Agency for Research on Cancer (IARC; 1993) as a possible human carcinogen (2B). In experimental studies, residues of OTA have been detected in the blood and tissues (Denli et al. 2008; Biro et al. 2002; Niemiec et al. 1994) and also in the eggs (Frye and Chu 1978; Fuchs et al. 1988; Niemiec et al. 1994) of birds kept on OTA-contaminated ration. Teratogenic and embryotoxic effects of OTA alone and in combination with other mycotoxins have been reported in rat, rabbit, and chicken (Wangikar et al. 2007, 2005; Wangikar, Dwivedi, and Sinha 2004; Wei and Sulik 1996; Vesala, Vesely, and Jelinek 1983; Gilani, Bancroft, and Reily 1978). A significant decrease in the number of immunoglobulin bearing cells in spleen and bursa of Fabricius and also poor production performance of the progeny of breeder hens maintained on OTA-contaminated ration have been reported by Zahoor-ul-Hassan et al. (2011) and Choudhury, Carlson, and Semeniuk et al. (1971).
Information is available about the contamination of poultry feed with a high concentration of 16 (Hamilton et al. 1982) and 80 mg OTA/kg (Visconti and Bottalico 1983) in turkey and broiler feed, respectively. Also feeding OTA-contaminated diet to breeder hens at 5 and 10 mg/kg, resulted in its residues at ∼0.44 and 1.053 µg/egg, respectively (unpublished data—authors’ study conducted at University of Agriculture Faisalabad, Pakistan). These findings and a study conducted by Frye and Chu (1978), in which they recovered 0.168 µg OTA/egg laid by the hens fed OTA-contaminated ration at 5 mg/kg for 2 weeks, warrants what might be the safe level for potential exposure of embryos to this mycotoxin. For this reason, the calculated amount of OTA (0.01 to 1.0 µg/egg) was used to evaluate its effects in developing embryos at different stages, that is, 53 hrs and 9 days of incubation, and then in the hatched chicks.
Materials and Methods
Experimental Design
A total of 900 fertile white leghorn (WL) layer breeder eggs were procured from a local breeder farm whose birds were maintained under standard environmental conditions. Total mycotoxin (aflatoxin B1 [AFB1] and OTA) contents of eggs was determined by HPLC (Sizoo and van Egmond 2005). For this purpose, a total of ninety eggs were selected and twenty pooled samples, each composed of five blended eggs, were prepared. None of the egg samples was positive for AFB1 and/or OTA beyond our limit of detection, that is, ≥ 0.02 ng/g. Remaining eggs were divided into eight groups (A–H). Group A was maintained as untreated control, and group B was kept as sham control (10 µL of 0.1 M NaHCO3). All the eggs in groups C, D, E, F, G, and H were injected with 0.01, 0.03, 0.05, 0.10, 0.50, and 1.00 µg OTA/egg (CAT #001108, Biopure, Technopark I, Tulln, Austria), respectively, which was dissolved in 10 µL of 0.1 M NaHCO3, using standard window opening technique described by Vesala, Vesely, and Jelinek (1983). Required amount of OTA was placed onto the air cell membrane, and the hole was sealed with melted wax. After dosing eggs were incubated at 37.5°C and 65% relative humidity.
Embryonic Studies at 53 Hours of Incubation
All of the animal experiments were conducted according to the rules and regulations of the Animal Ethics Committee, Faculty of Veterinary Science, at the University of Agriculture Faisalabad (Pakistan). At 53 hrs of incubation, fifteen fertile eggs were collected from each group and opened to collect the embryos. Collected embryos were washed in normal saline solution, preserved in Bouin’s fixative for 2 hrs, and then stained with Borax Carmine (Asmatullah and Shakoori 1998). Embryos were mounted on microscopic slides using DPX and studied under light microscope. Measurements of optic cup, eye lens diameter, and crown to rump length were recorded using ocular micrometer. Neural tube of developing embryos was studied for any closure defect.
At Day 9 of Incubation
At day 9 of incubation, eggs were candled, and twenty viable embryos were collected from each group by opening the eggs at the wider end. After washing in normal saline solution, embryos were observed under stereomicroscope to study any gross abnormalities.
Embryonic Mortalities and Hatchabilities
All eggs were candled at alternate days starting from day 4 of incubation (i.e., day 4, 6, 8, 10, 12, 14, 16, and 18), and dead embryos (if any) were examined under stereomicroscope to determine the stage of death according to HH scale (Hamburger and Hamilton 1951). Embryonic mortalities were divided into early (HH scale 4–15), mid (HH scale 16–30), and late (HH scale 31–45). Hatchability of each group, on the fertile eggs basis, was calculated at day 22 of incubation.
Parameters Studied on Hatched Chicks
Hatched chicks were weighed, and ten chicks from each group were killed and their relative organ weights of liver, kidneys, bursa of Fabricius, and spleen were determined. Remaining chicks were reared under standard brooding conditions. Water and chick starter ration (verified not to contain OTA or AFB1> 1 µg/kg) containing 21% crude protein was offered ad libitum. At day 7, blood (2 ml) from chicks was collected and serum was separated to determine concentrations of total proteins, albumin, ALT, urea, and creatinine using commercially available kits (DDS).
Statistical Analysis
The data were subjected to analysis of variance tests. Means of the different groups were compared using a Duncan’s multiple range test within a MSTATC statistical package (Department of Crop and Soil Sciences, Michigan State University, East Lansing, MI). Data were considered significantly different from one another at a p value ≤ .05. Data regarding gross teratogenic effects, embryonic mortalities, and hatchabilities were compared by calculating percentage differences in each group.
Results
Pathological Effects on Embryos
Morphometric studies of embryos collected at 53 hrs of incubation are shown in Table 1 . A dose-dependent decrease was observed in the diameter of optic cups and eye lens, with a significant decrease (p ≤ .05) in all the embryos collected from the eggs contaminated with OTA (Figure 1 ), compared with the embryos recovered from untreated eggs (group A). The values of crown to rump length of embryos from groups D, E, F, and G were significantly lower than control. No defects were observed on the closure of neural tubes in the embryos from group A, B, C, and D; while embryos in group E (5%), F (8%), G (8%), and H (10%) showed defective closure (Figure 2 and 3).
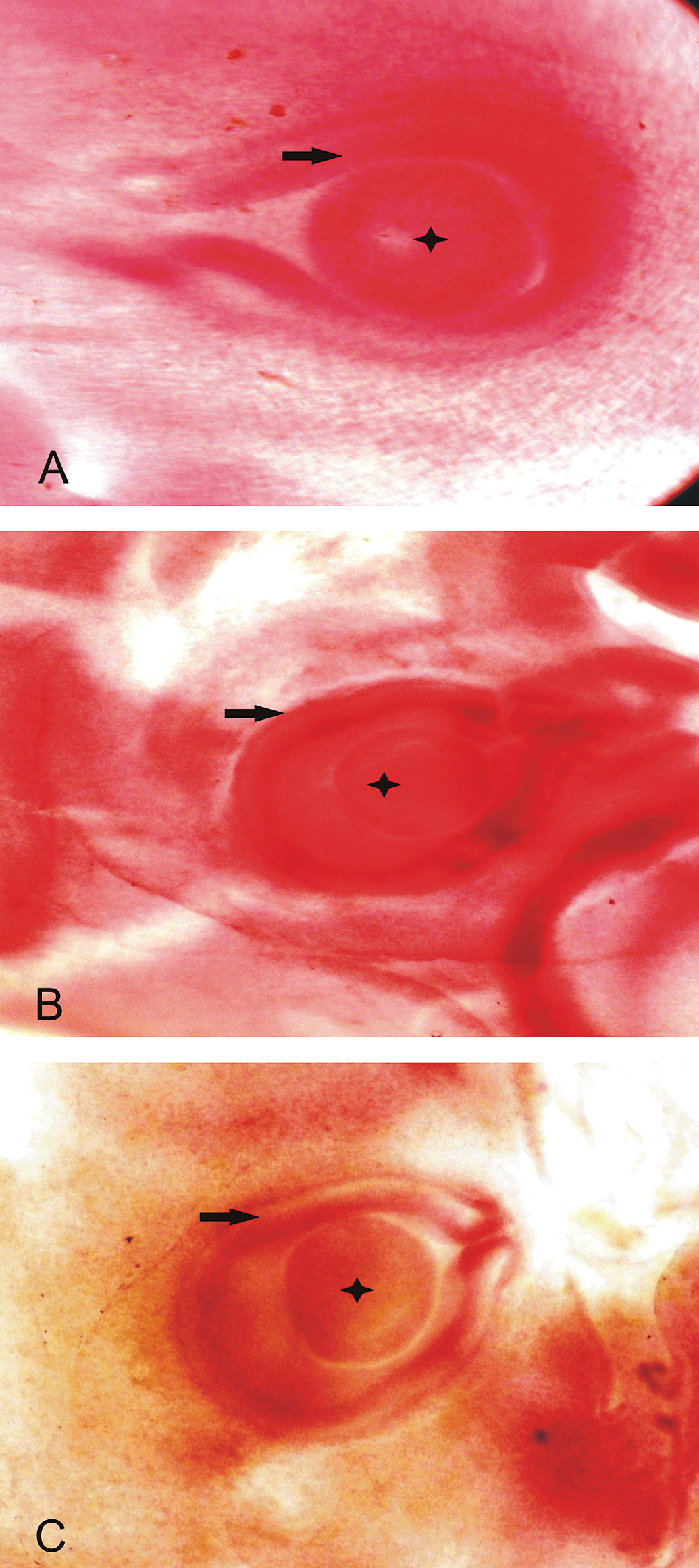
Photomicrograph of chick embryos at 53 hrs of incubation. A. Control group showing normal development of eye lens (star) and optic cup (arrow). B. 1.0 µg OTA-inoculated group. C. 0.5 µg OTA group. B & C are showing defective development of eye lens (Borax Carmine staining).
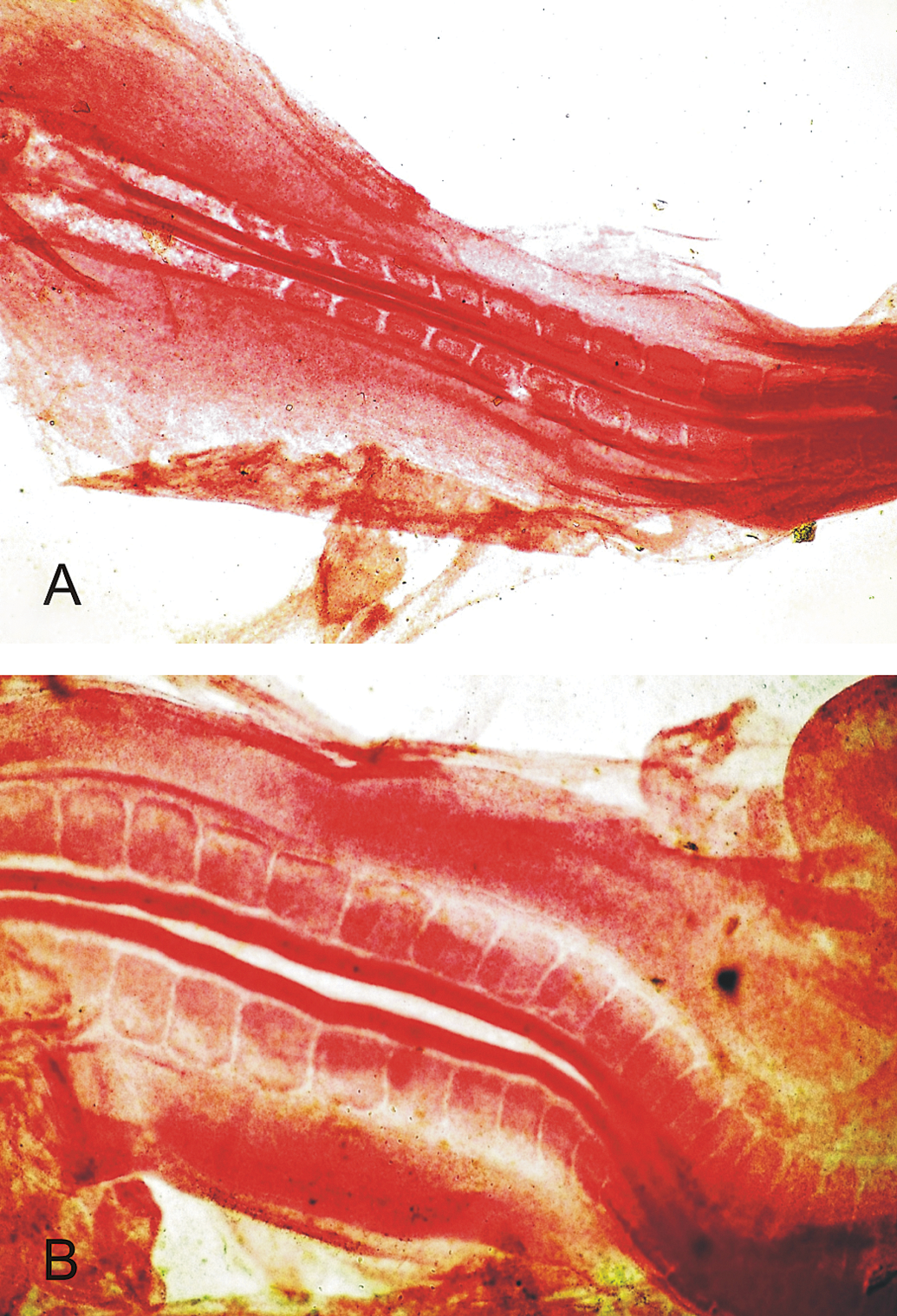
Photomicrograph of the chick embryo at 53 hrs of incubation. A. Embryo from control group showing normal closure of neural tube. B. Embryo from group H (1.0 µg OTA), showing defective closure of neural tube (Borax Carmine staining).
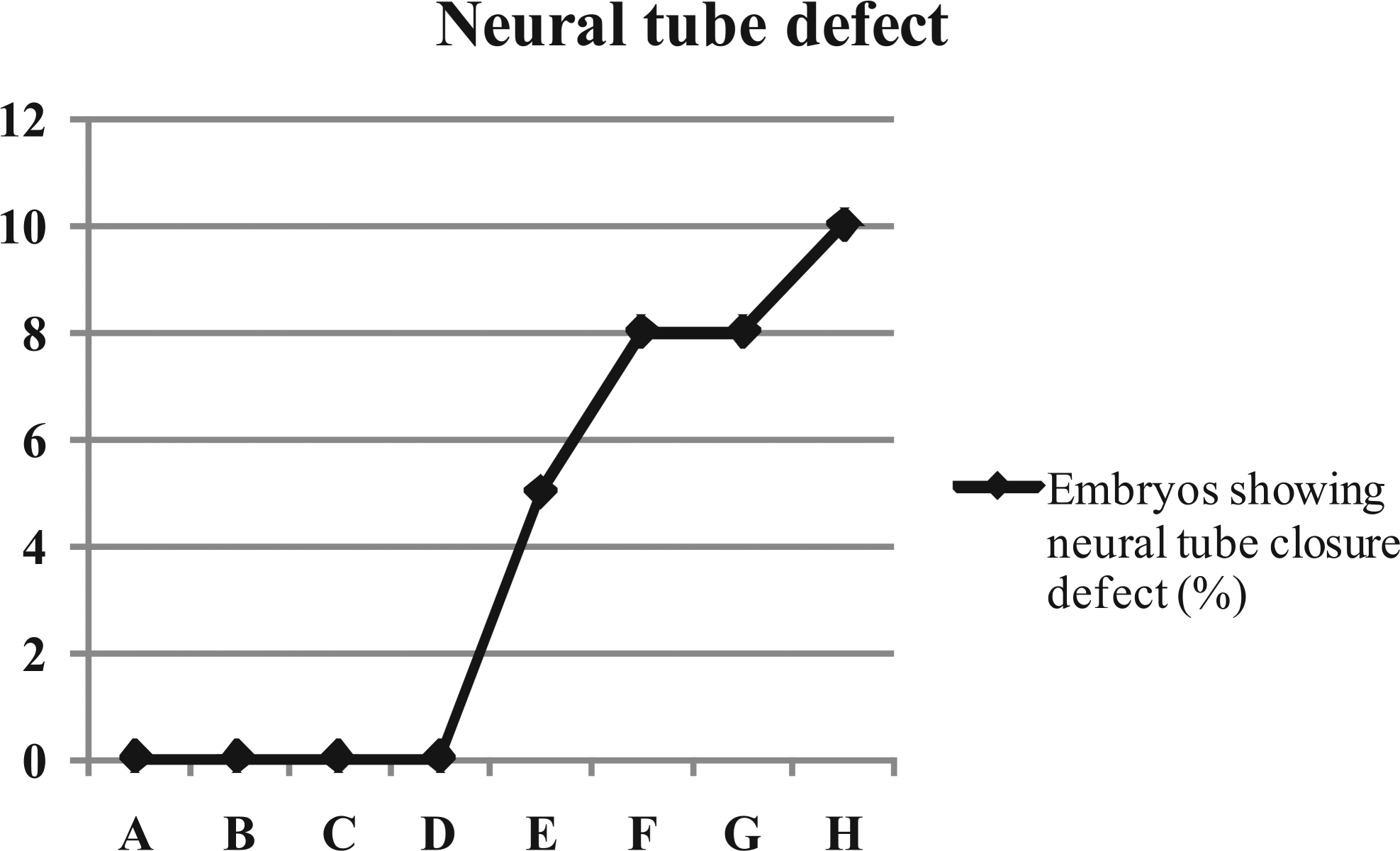
Neural tube closure defects in embryos (%) studied at 53 hrs of incubation from the eggs inoculated with OTA. A (Control), B (0.1 M NaHCO3), C (0.01 µg/egg), D (0.03 µg/egg), E (0.05 µg/egg), F (0.10 µg/egg), G (0.50 µg/egg), and H (1.00 µg/egg).
Morphometric studies of embryos (µm) at 53 hours of incubation inoculated with OTA.
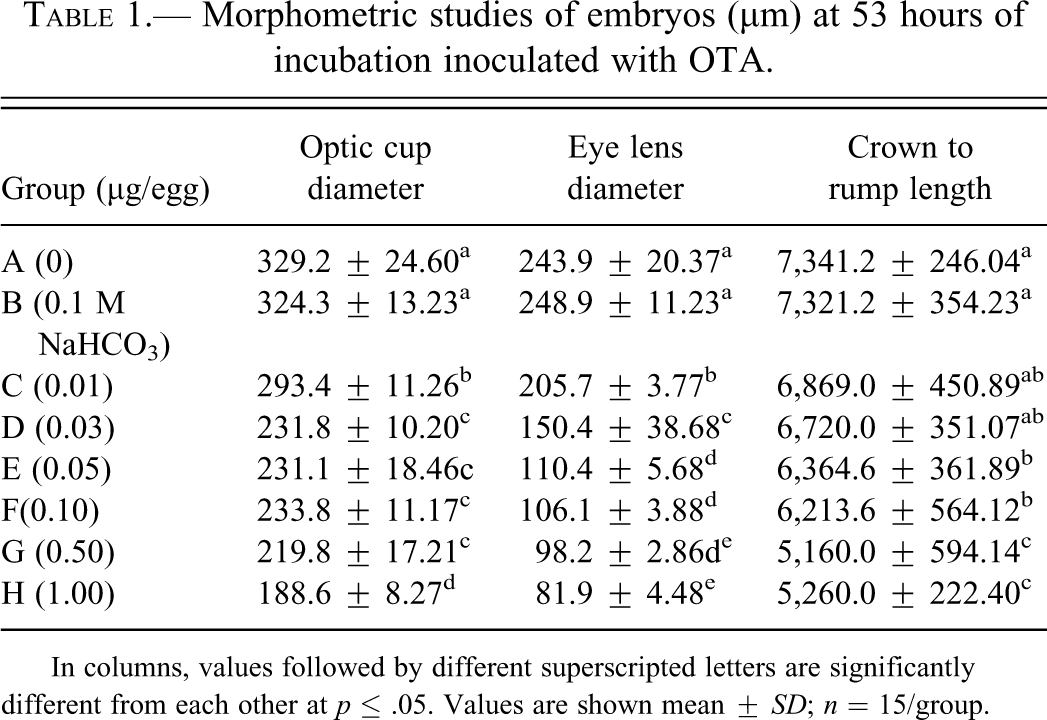
In columns, values followed by different superscripted letters are significantly different from each other at p ≤ .05. Values are shown mean ± SD; n = 15/group.
Teratogenic effects of OTA (in percentage) in different groups have been presented in Figure 4 . Anophthalmia (Figure 5 ) was the most frequent (49% of the embryos) defect, followed by mandibular hypoplasia (45%), micropthalmia (35%), maxillary retrognathism (12%), reduced body size (15%), everted visera (8%), spina bifida (10%), and exencephaly (4%), in the highest dosed group (group H). All the teratogenic effects showed increasing trend with the increase in dose of OTA in the eggs (Figure 5). No developmental defects were observed in the embryos belonging to group A and B.
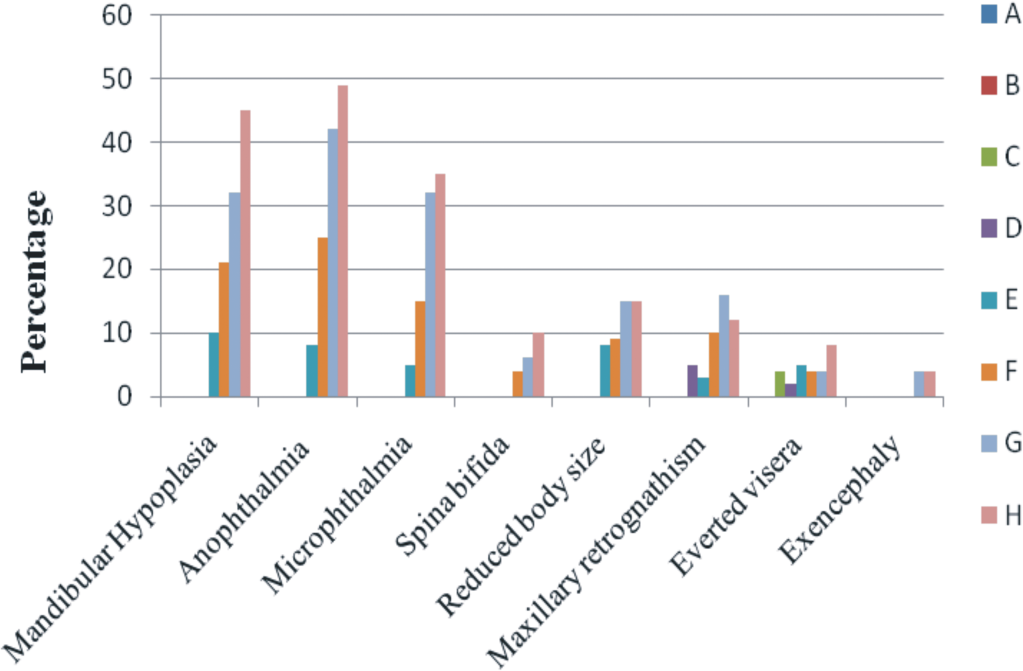
Teratogenic effects (percentage) of in ovo inoculation of OTA studied at day 9 of incubation. A (Control), B (0.1 M NaHCO3), C (0.01 µg/egg), D (0.03 µg/egg), E (0.05 µg/egg), F (0.10 µg/egg), G (0.50 µg/egg), and H (1.00 µg/egg).
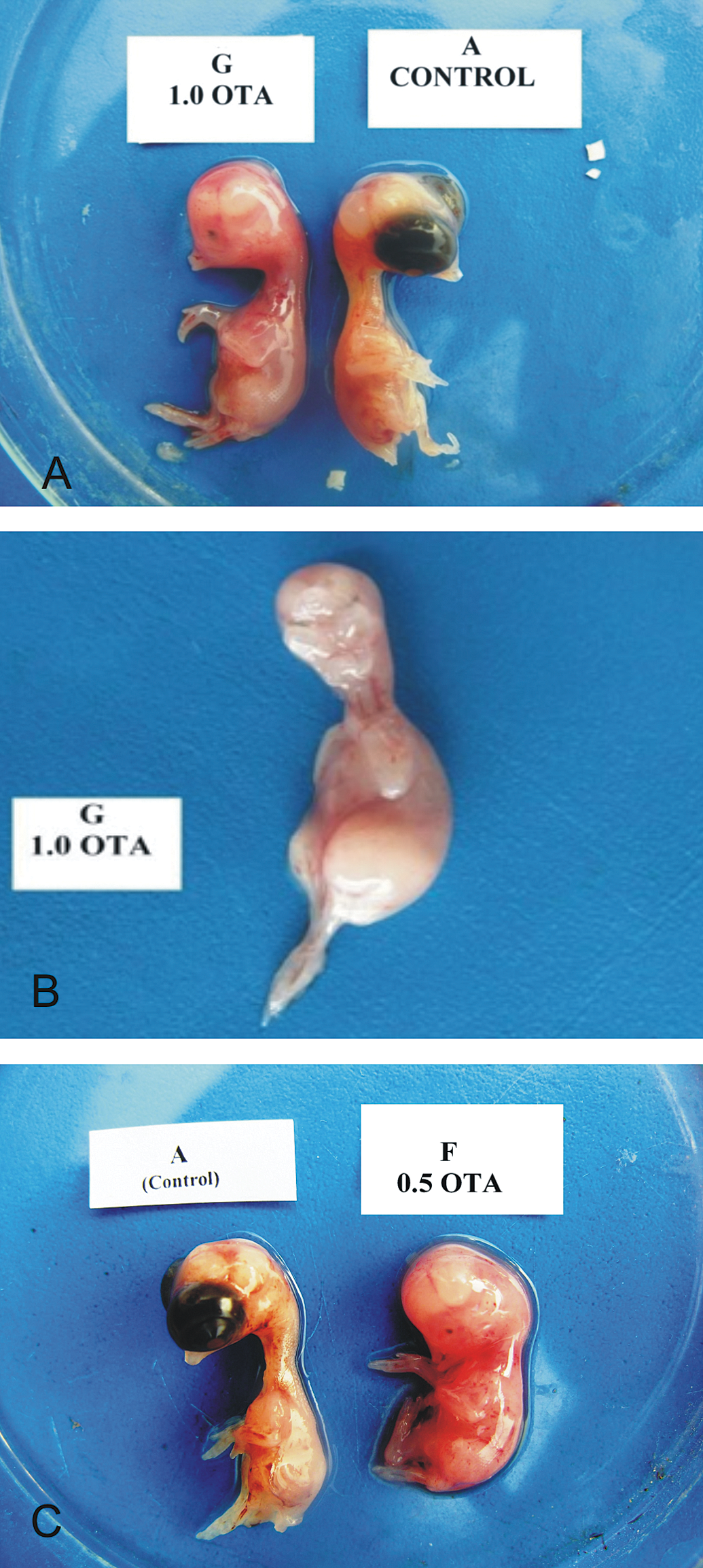
Photograph of chick embryos at day 9 of incubation. A. Embryo from group H, inoculated with OTA at 1.0 µg/egg, is showing anophthalmia. B. Embryo showing maxillary retrognathism, mandibular hypoplasia, and anophthalmia (bilateral). C. Embryo from group G, inoculated with OTA at 0.5 µg/egg, is showing anophthalmia.
Embryonic deaths (Figure 6 ) were higher in the early stages of development, that is, HH scale 4–15, while the lowest were found in the late stages (HH scale 31–45). This trend in mortalities was found in the embryos from group E, F, G, and H (Figure 6). Increasing trend in the embryonic deaths was found with increases in the dose of toxin injected.
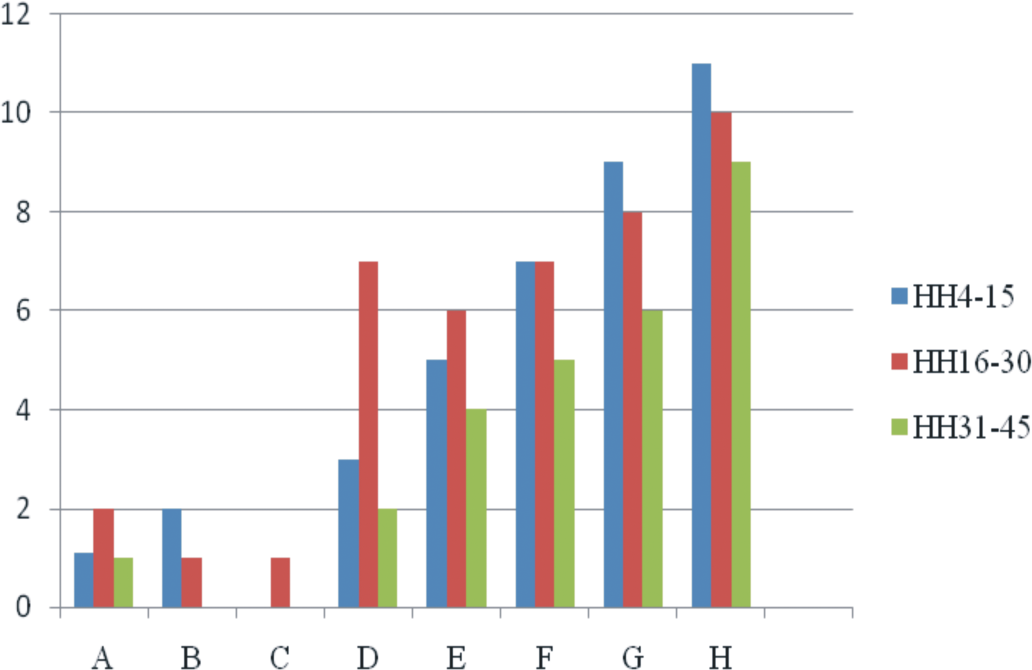
Embryonic mortalities (percentage) at different developmental stages due to in ovo OTA inoculation. A (Control), B (0.1 M NaHCO3), C (0.01 µg/egg), D (0.03 µg/egg), E (0.05 µg/egg), F (0.10 µg/egg), G (0.50 µg/egg), and H (1.00 µg/egg).
Hatchabilities, on a fertile egg basis, in the different groups showed inverse trend to the dose of OTA injected in the eggs (Figure 7 ). In groups A and B, hatchability was 79% and 80%, while it was 73%, 43%, 35%, 38%, 31%, and 32% in groups C, D, E, F, G, and H, respectively.
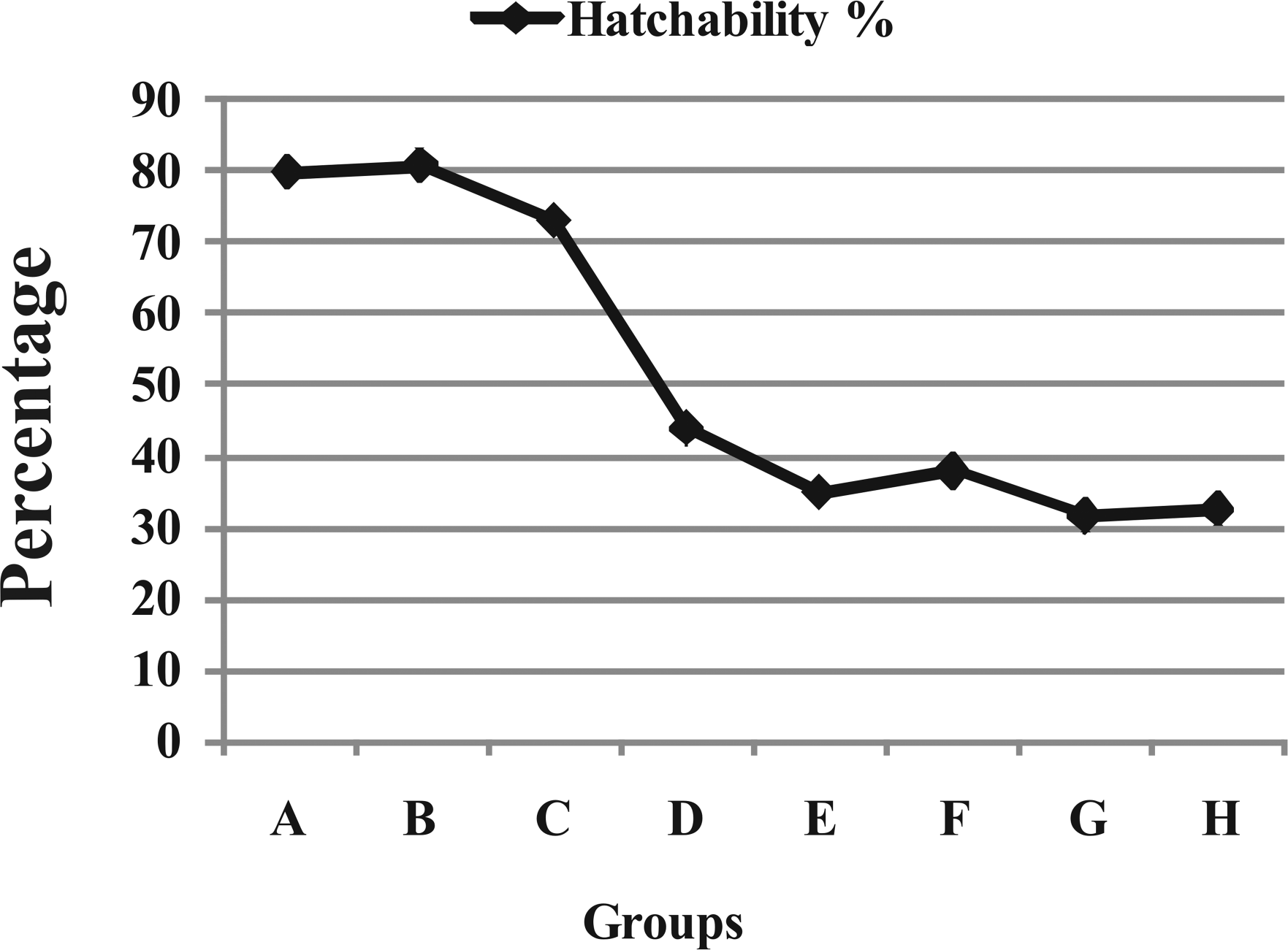
Hatchability (percentage) in different groups treated with different doses of OTA. A (Control), B (0.1 M NaHCO3), C (0.01 µg/egg), D (0.03 µg/egg), E (0.05 µg/egg), F (0.10 µg/egg), G (0.50 µg/egg), and H (1.00 µg/egg).
Pathological Effects in Hatched Chicks
Relative organ weights of 1-day-old hatched chicks (from OTA-contaminated eggs) are presented in Table 2 . Weight of liver of all the groups showed dose-dependent increase, except in B and C, which showed nonsignificantly higher values than their counterpart chicks in group A. Weight of kidney of all groups belonging to OTA-contaminated eggs was significantly higher than its corresponding values in group A chicks. The weight of bursa of Fabricius showed significantly lower values in the chicks hatched from the eggs contaminated with OTA than their counterpart chicks in group A. This trend was inversely related with dietary OTA levels.
Body weight (g) and relative organ weights (% of body weight) of 1-day-old chicks, hatched from the eggs inoculated with OTA.
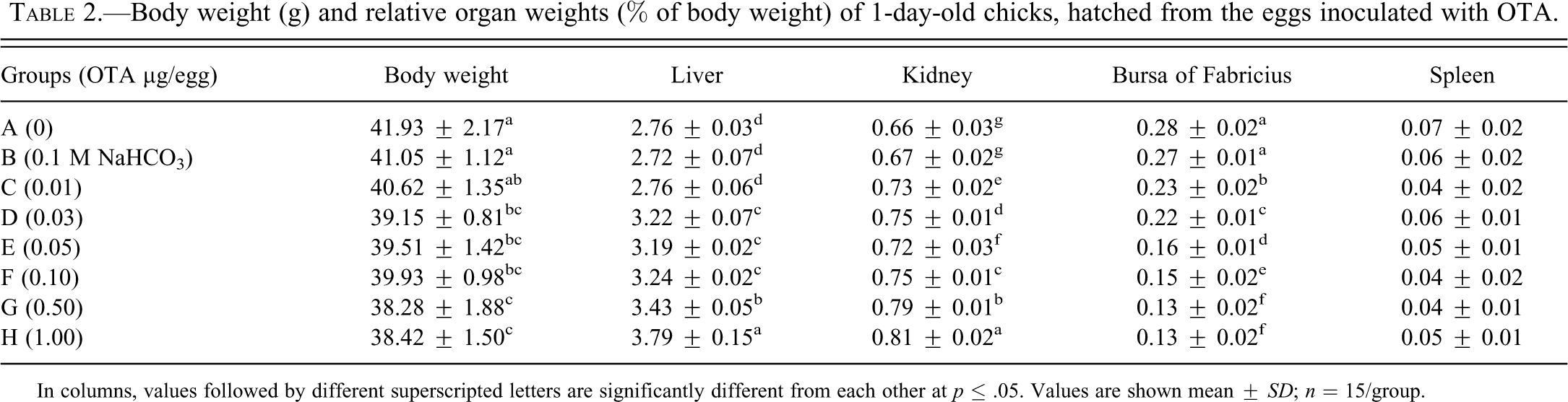
In columns, values followed by different superscripted letters are significantly different from each other at p ≤ .05. Values are shown mean ± SD; n = 15/group.
Serum ALT and creatinine values of all the groups, except B and C, were significantly higher than the values obtained from the chicks belonging to group A (Table 3 ). Chicks hatched from the eggs contaminated with higher doses of OTA, that is, groups E, F, G, and H, showed significantly higher concentration of urea in their sera as compared with the values in the sera of group A chicks. The values of serum total protein and albumin were lower in the chicks hatched from OTA-intoxicated eggs; however, a significantly lower (p ≤ .05) difference was noted in the chicks from the highest dosed group (group H), compared with values shown in chicks from the control group.
Serum biochemical profile of chicks hatched from eggs inoculated with OTA.
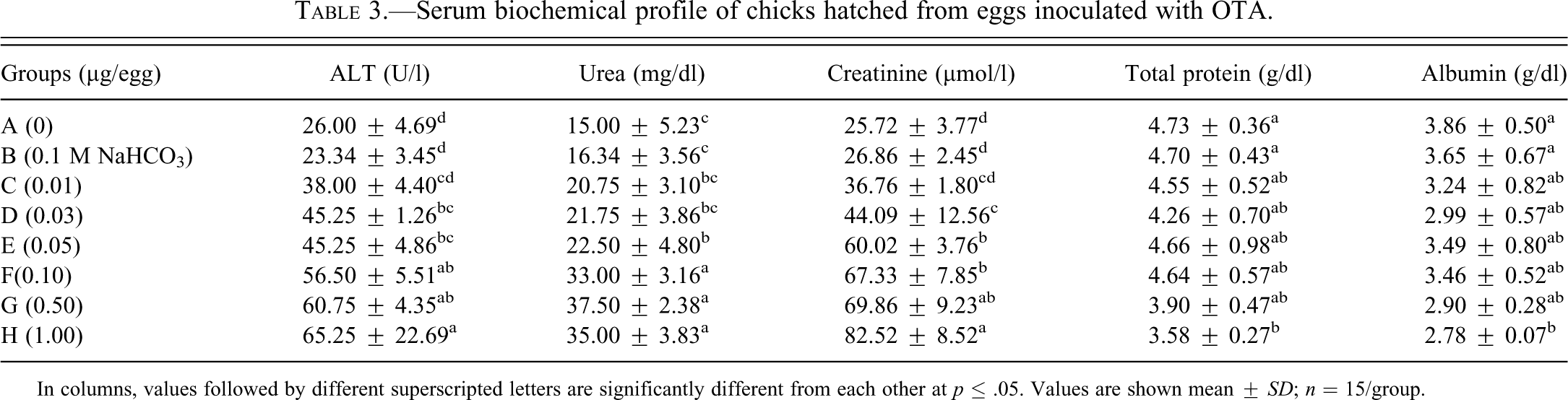
In columns, values followed by different superscripted letters are significantly different from each other at p ≤ .05. Values are shown mean ± SD; n = 15/group.
Discussion
At 53 hrs of incubation, defective closure of neural tube was observed in the embryos belonging to the high-dosed groups (≥ 0.05 µg OTA/egg). This defective or incomplete closure may be due to cell death in the ventral part of the neural tube, as reported by Wei and Sulik (1996) in OTA-treated chicken embryos at 1 µg/egg at the age of 48 hrs of incubation. The diameter of optic cup and eye lens were significantly decreased in all the OTA-treated embryos. This decrease in the diameter of optical structures may be due to necrotic/cytotoxic effects of OTA in the developing optic cells, which subsequently resulted in the uni- or bi lateral anopthalmia, observed at day 9 of incubation (in this study) in OTA-treated embryos. Unfortunately, there is no assessable report in the literature describing the effect of OTA in the developing chick embryo at so early stages. Lastly, OTA treatment at higher doses (≥ 0.05 µg/egg) also resulted in significant reduction in the crown to rump length of the developing embryos, studied at 53 hrs of incubation, which may also be attributed to cytotoxic effects of OTA.
Teratogenic effects of OTA in the embryos observed at day 9 of incubation included anopthalmia, mandibular hypoplasia, maxillary retrognathism, everted visera, microphthalmia, spina bifida, exencephaly, and reduced body size. Gilani, Bancroft, and Reily (1978) and Vesala, Vesely, and Jelinek (1983) also reported similar teratogenic effects, except spina bifida, in the chicken embryos inoculated with different doses of OTA. These effects of OTA may be due to DNA adduct formation and subsequently inhibition of protein synthesis, as has previously been reported by Petkova-Bocharova et al. (2003).
The mortalities in the chicken embryos showed age- and dose-dependent relationship in the OTA-intoxicated groups. Early embryonic mortalities (HH scale 5–15) were most frequent, followed by mid (HH scale 16–30) and late (HH scale 31–45) deaths. In an experimental inoculation of OTA in the amniotic cavity of developing chicken embryos at days 2, 3, and 4 of incubation, Vesala, Vesely, and Jelinek (1983) reported a range of embryotoxic dose to be in between 0.01 to 0.05 µg/egg. Embryonic mortalities in the intoxicated groups may be attributed to cytotoxic effects of OTA, as has previously been reported in chicken (Wei and Sulik 1996; Choudhury and Carlson 1973) and mice embryos (Wei and Sulik 1993), intoxicated with different doses. Although no relationship is reported in the published literature about the embryonic mortality induced by OTA in chicken embryos to the stage of development, Celik et al. (2000) and Neldon-Ortiz and Qureshi (1992) reported that the maximum mortality occurs at early developmental stages in developing chick embryos within AFB1-inoculated eggs. Hatchabilities calculated on fertile eggs basis were lower in all groups inoculated with different doses of OTA. This decrease in the hatchabilities was mainly due to embryonic mortalities and also due to teratogenic effects of OTA, particularly mandibular hypoplasia, maxillary retrognathism, and everted visera, which caused hindrance in the hatching processes in fully developed chick embryos.
Body weights of chicks hatched from OTA-contaminated eggs were significantly lower as compared with the values shown by their control counterpart chicks. This decrease in the body weight is attributed to the adverse effects of OTA, particularly a decrease in protein synthesis vital for the development of embryos. Relative weights of liver and kidneys of the chicks hatched from OTA-inoculated eggs were significantly higher, while the relative weights of bursa of Fabricius was significantly lower than the chicks in the control group. Unfortunately, the literature is lacking information about the effect of in ovo OTA on the body and relative organ weights of developing chicks. However, studies made on OTA-fed chicks reported an increase in the weights of liver and kidneys (Hanif et al. 2008; Stoev et al. 2002) and a decrease in weights of bursa of Fabricius (Singh et al. 1990; Gibson et al. 1989). This increase in the weights of liver and kidneys is correlated with nephro- and hepatotoxic effects of OTA, resulting in the accumulation of inflammatory cells (Zahoor-ul-Hassan et al. 2010). However, decrease in the relative weight of bursa of Fabricius may be due to degenerative/necrotic effects of OTA in the follicles of immunological organs, as observed in our previous studies (Zahoor-ul-Hassan et al. 2011) in the progeny of hens kept on OTA-contaminated feed.
Hepatotoxic and nephrotoxic activity of OTA in various animal species, as indicated by altered serum biochemical profile, is well established. In the present study, increased serum ALT concentration and decrease in total protein and albumin levels in the hatched chicks are indicative of liver damage. In the same way, increased urea and creatinine levels may be due to nephrotoxic effects of in ovo inoculation of OTA. To the best of our knowledge, we are the first to report the hepatoxic and nephrotoxic effects of OTA in the chicks obtained from OTA-contaminated eggs; however, a lot of information is available in the literature regarding altered serum biochemical profiles of chicks kept on OTA-contaminated ration (Wang et al. 2009; Koynarski et al. 2007; Agawane and Lonkar 2004).
From the results of the present study, it can be concluded that inoculation of OTA in the fertile eggs not only resulted in developmental defects in embryos, increased embryonic mortalities, and decreased hatchabilities, but also ultimately gave rise to a decrease in production performance of the hatched chicks due to its pathological effects.
