Abstract
Two-year 1-bromopropane (1-BP) inhalation studies were conducted because of the potential for widespread exposure, the lack of chronic toxicity and carcinogenicity data, and the known carcinogenicity of structurally related compounds. Male and female F344/N rats and B6C3F1/N mice were exposed by inhalation to 0, 62.5 (mice only), 125, 250, or 500 (rats only) ppm 1-BP for 6 hr/day, 5 days/week for 105 weeks. Exposure of male and female rats to 1-BP resulted in significantly increased incidences of adenomas of the large intestine and skin neoplasms. In male rats, the incidence of malignant mesothelioma of the epididymis was statistically significantly increased at 500 ppm, but the biological significance of this common lesion is unclear. Incidences of pancreatic islet adenoma in male rats were significantly increased at all concentrations relative to concurrent controls but were within the historical control range for inhalation studies. There was no evidence of carcinogenic activity of 1-BP in male B6C3F1 mice; however, significantly increased incidences of alveolar/bronchiolar neoplasms of the lung were present in female mice. Exposure to 1-BP also resulted in increased incidences of nonneoplastic lesions in the nose of rats and mice, the larynx of rats and male mice, the trachea of female rats and male and female mice, and the lungs of mice. Inflammatory lesions with Splendore Hoeppli (S-H) material were present primarily in the nose and skin of exposed male and female rats, indicating that 1-BP caused immunosuppression.
Introduction
1-Bromopropane (1-BP; n-propyl bromide) is a highly versatile chemical that is being used for an increasing number of applications in the United States. In the early to mid-1990s, 1-BP was used primarily as an intermediate in the production of pesticides, quaternary ammonium compounds, flavors and fragrances, pharmaceuticals, and other chemicals in well-controlled, closed processes (Hanley et al. 2006). However, in the mid- to late 1990s, 1-BP was introduced as a less toxic replacement for methylene chloride in emissive applications such as vapor and immersion degreasing operations and critical cleaning of electronics and metals, resulting in an increase in worker exposure. Because 1-BP has a relatively high vapor pressure, it is well suited for use in cleaning equipment where it is repeatedly vaporized. 1-BP is also relatively nonflammable and can be used safely in metal-cleaning processes where heating is required. These properties led to a new application for 1-BP as a nonflammable, nontoxic, fast-drying, and inexpensive solvent for adhesive resins. Aerosol-applied adhesives containing 1-BP are used extensively by foam fabricating companies (Hanley et al. 2006). The highly emissive application of spray adhesives containing 1-BP was found to significantly increase worker exposure to 1-BP. The National Institute of Occupational Safety and Health (NIOSH) measured 1-BP concentrations ranging from 18 to 381 ppm in the breathing zone of workers in three plants where 1-BP–containing spray adhesives were used (Reh and Mortimer 1999; Reh 2000, 2001). Because 1-BP has a relatively short atmospheric half-life (16 days), it is considered to have a relatively low ozone depletion potential and has been marketed as a replacement for ozone-depleting refrigerants (e.g., chlorofluorohydrocarbons and hydrochlorofluorocarbons; U.S. Environmental Protection Agency 2000). In 2006, worldwide annual production capacity of 1-BP was estimated at greater than 20,000 metric tons (44 million pounds/year), of which 5,000 metric tons were thought to be used as a pharmaceutical intermediate or process agent. U.S. production was estimated at approximately 5,000 metric tons (11 million pounds/year) and growing at a rate of 15% to 20% per year (United Nations Environmental Programme 2001; Sclar 1999).
A number of short-term studies have demonstrated that inhalation exposure to 1-BP can cause neurotoxicity and reproductive toxicity in humans (Sclar 1999; NIOSH 2003; National Toxicology Program [NTP] 2003) and laboratory animals (Yu et al. 2008; Ichihara, Kitoh, et al. 2000; Ichihara, Yu, et al. 2000; Yamada et al. 2003), yet little is known about its potential long-term toxicity and carcinogenicity. The current studies were conducted because of the potential for widespread human exposure to 1-BP, a lack of chronic toxicity and carcinogenicity data, and the known carcinogenicity of structurally related chemicals. The results of this study demonstrate that 1-BP is a multisite carcinogen in rats and mice and causes unusual Splendore-Hoeppli (S-H) lesions in rats.
Materials and Methods
Chemical
1-BP was obtained from Albemarle PCC (Thann, France). The purity of the chemical was determined by gas chromatography to be approximately 99%. Periodic reanalyses of the bulk chemical were performed during the 2-year studies using gas chromatography, and no degradation of the chemical was detected.
Vapor Generation and Monitoring
1-BP was pumped into a heated glass column filled with glass beads that increased the surface area for vaporization. Heated nitrogen entered the column from below and assisted in vaporizing the chemical while conveying it into a short distribution manifold. Metering valves at the manifold controlled flow to each chamber inlet duct, where it was further diluted with filtered, conditioned air to achieve the desired exposure concentration. Chamber concentrations of 1-BP were monitored by an online gas chromatograph. Samples were drawn from each exposure chamber approximately every 30 min during each 6-hr exposure period. Evaluations of chamber uniformity and monitoring for 1-BP degradation impurities were conducted periodically throughout the studies. Chamber uniformity was maintained, and no degradation was detected.
Animals
Male and female F344/N rats and B6C3F1/N mice (Taconic Farms, Inc., Germantown, NY) were housed individually and were quarantined for 10 to 13 days. Animals were approximately 5 to 6 weeks old at the beginning of the exposures. Feed (NTP 2000) was available ad libitum except during exposure periods; water was always available. Cages, racks, and chambers were changed weekly, and cages were rotated weekly in chambers. Groups of 50 male and 50 female rats and mice were exposed to 1-BP vapor at concentrations of 0, 62.5 (mice only), 125, 250, or 500 (rats only) ppm, 6 hr per day, 5 days per week for up to 105 weeks in Hazelton 2000 inhalation chambers. These exposure concentrations were selected based on results of 14- and 90-day subchronic inhalation studies (data not shown). All animals were observed twice daily, and clinical findings were recorded for all animals every 4 weeks through week 93, every 2 weeks thereafter, and at the end of the studies. Rats and mice were weighed weekly for the first 13 weeks, then every 4 weeks through week 93, and every 2 weeks thereafter. These studies were conducted in the Association for Assessment and Accreditation of Laboratory Animal Care–accredited facility of Battelle Toxicology Northwest (BTNW; Richland, WA) and approved by the BTNW Institute Animal Care and Use Committee. Animal use was in accordance with the U.S. Public Health Service policy on humane care and use of laboratory animals and the Guide for the Care and Use of Laboratory Animals. Animal handling and husbandry met all National Institutes of Health (NIH) guidelines (Grossblatt 1996). These studies were conducted in compliance with the Food and Drug Administration Good Laboratory Practice Regulations (Food and Drug Adminstration 1987).
Necropsy and Histopathology
Complete necropsies and microscopic examinations were performed on all rats and mice. At necropsy, all organs and tissues were examined for grossly visible lesions, and all major tissues were fixed and preserved in 10% neutral buffered formalin (NBF), processed and trimmed, embedded in paraffin, sectioned to a thickness of 4 to 6 µm, and stained with hematoxylin and eosin for microscopic examination. The nasal cavities were fixed by retrograde flushing of NBF through the nasopharynx and the skull decalcified. Three transverse sections through the nose, designated levels I to III, were trimmed at specific anatomical landmarks for all animals: Level I immediately posterior to the upper incisor teeth, Level II through the level of the incisive papilla anterior to the first palatial ridge, and Level III through the middle of the second molar teeth. Levels I and II contain the naso- and maxillo-turbinates, the nasal passages (meatuses), and the septum, which are lined almost entirely by ciliated respiratory-type epithelium. Level III contains the olfactory region of the nose and ethmoid turbinates, which are lined entirely by specialized olfactory neuroepithelium.
Statistical Methods
The probability of survival was estimated by the product-limit procedure (Kaplan and Meier 1958). Statistical analyses for possible exposure-related effects on survival used Cox’s method (Cox 1972) for testing two groups for equality and Tarone’s life table test (Tarone 1975) to identify exposure-related trends. All reported p values for the survival analyses are two-sided. For neoplasm and nonneoplastic lesion incidences, continuity-corrected Poly-3 tests were used to test for exposure-related trends and pairwise comparisons of each exposure group to the control group, and reported p values are one-sided (Bieler and Williams 1993; Portier and Bailer 1989; Bailer and Portier 1988).
Results
F344/N Rats
Survival, Body Weight, and Clinical Findings
Estimates of 2-yr survival probabilities for male and female rats are shown in Table 1 . Survival of male rats exposed to 500 ppm 1-BP was significantly less than that of the controls. Most of the early deaths in the 500-ppm males were attributed to various types of neoplasia, none of which were treatment related. However, in nine of these males, the cause of death was attributed to inflammation in various organs, all of which contained S-H material. Survival of exposed female rats decreased with increasing 1-BP exposure concentration, but survival in each exposed group was not significantly different from that of controls. Mean body weights of exposed male and female rats were within 8% of the controls throughout the study (data not shown). No clinical findings related to exposure to 1-BP were observed in rats.
Survival of Rats in the 2-Year Inhalation Study of 1-Bromopropane.
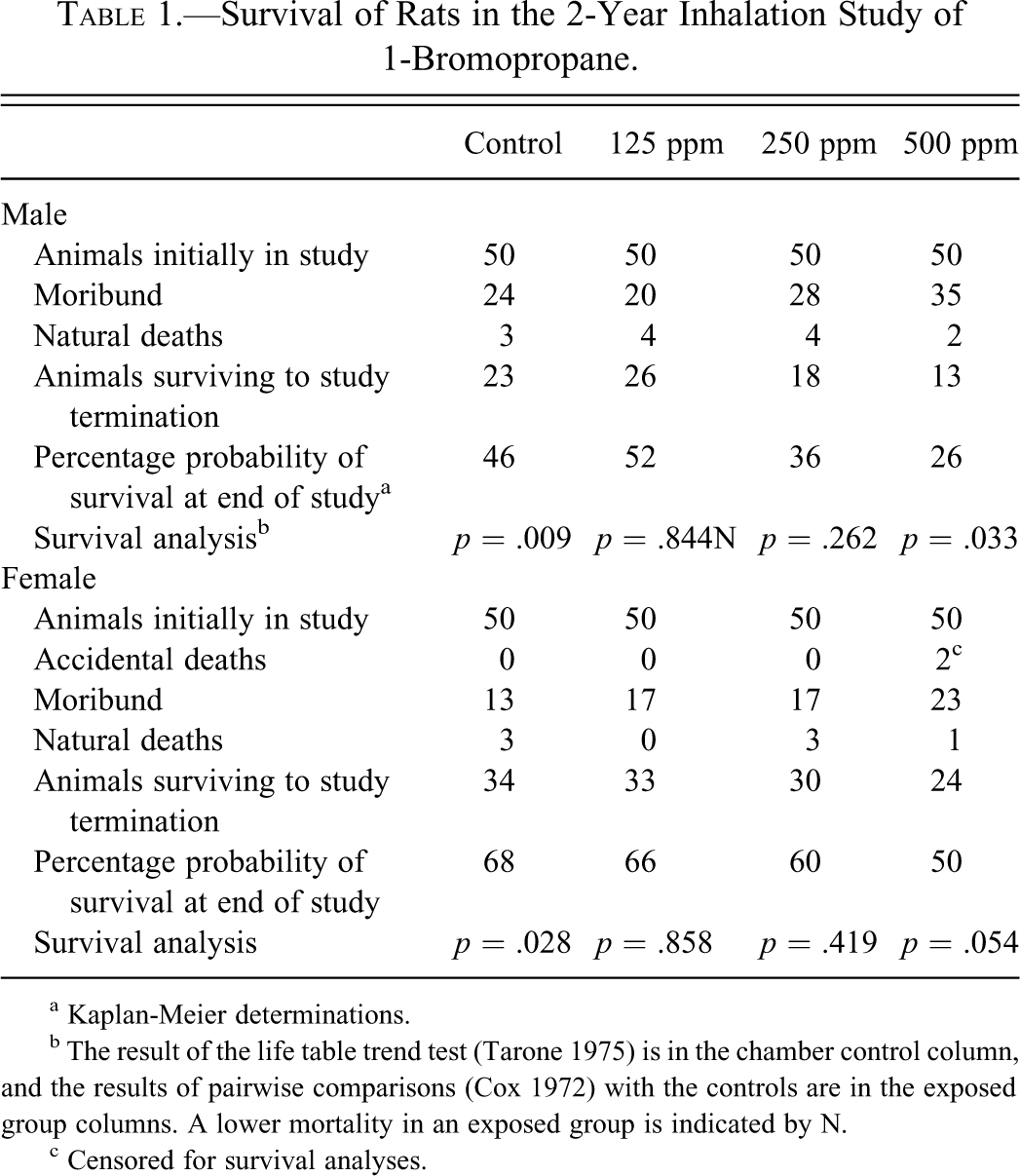
a Kaplan-Meier determinations.
b The result of the life table trend test (Tarone 1975) is in the chamber control column, and the results of pairwise comparisons (Cox 1972) with the controls are in the exposed group columns. A lower mortality in an exposed group is indicated by N.
c Censored for survival analyses.
Histopathology
Large Intestine
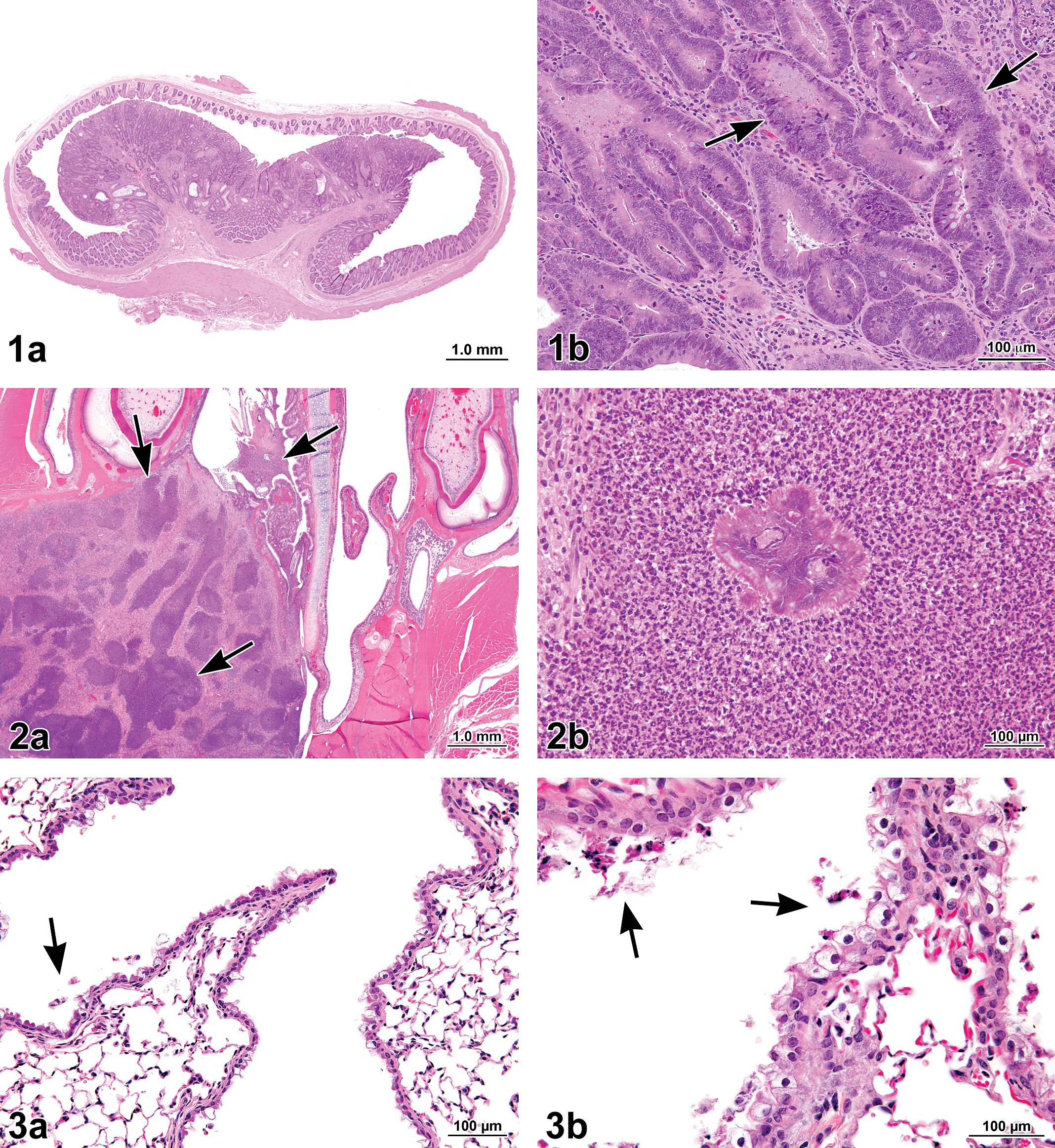
The incidence of adenoma of the large intestine (colon or rectum) in 500-ppm females was significantly greater than that in the controls (Table 2 ). Incidences of adenoma of the large intestine in 125- and 250-ppm females and in 250- and 500-ppm males were slightly increased compared with that of the controls and exceeded historical control rates. Large intestine adenomas were polypoid masses that protruded into the intestinal lumen (Figure 1a, b). The epithelium lining the glands had fewer goblet cells than the epithelium from normal glands and was occasionally thickened by multiple layers of slightly enlarged neoplastic epithelial cells with basophilic cytoplasm and enlarged nuclei. There were variable numbers of mixed inflammatory cells in the stroma around the glands and in the stalk. No invasion of the submucosa of the large intestine was observed.
Figure 1. (a) Polypoid adenoma in the large intestine (rectum) of a female F344/N rat exposed to 500 ppm 1-bromopropane (1-BP) by inhalation for 2 years. The adenoma occupies the majority of the intestinal lumen. (b) Higher magnification of (a). Note the multiple layers of basophilic neoplastic epithelial cells lining the glands (arrows). Hematoxylin and eosin.
Figure 2. (a) Suppurative chronic inflammation (arrows) in Level II of the nasal cavity of a male F344/N rat exposed to 500 pp 1-bromopropane (1-BP) for 2 years. (b) Higher magnification of (a). The suppurative chronic inflammation is composed of abundant eosinophilic necrotic cellular debris, fibrin, and neutrophils and contains Splendore-Hoeppli material (center). These lesions were often bordered by a dense fibrous capsule. Hematoxylin and eosin.
Figure 3. (a) Bronchiolar epithelial regeneration and vacuolization in the lung of a male B6C3F1 mouse exposed to 250 ppm 1-bromopropane (1-BP) by inhalation for 2 years. There are sloughed epithelial cells in the lumen of the bronchiole (arrows) indicative of epithelial cell loss. (b) Bronchiolar epithelial necrosis and cytoplasmic vacuolization in the lung of a male B6C3F1 mouse exposed to 250 ppm 1-BP by inhalation for 2 years. Note the abundant necrotic cells sloughing into the bronchiolar lumen (arrows). Hematoxylin and eosin.
Increased Incidences of Neoplasms in Rats Exposed to 1-Bromopropane.
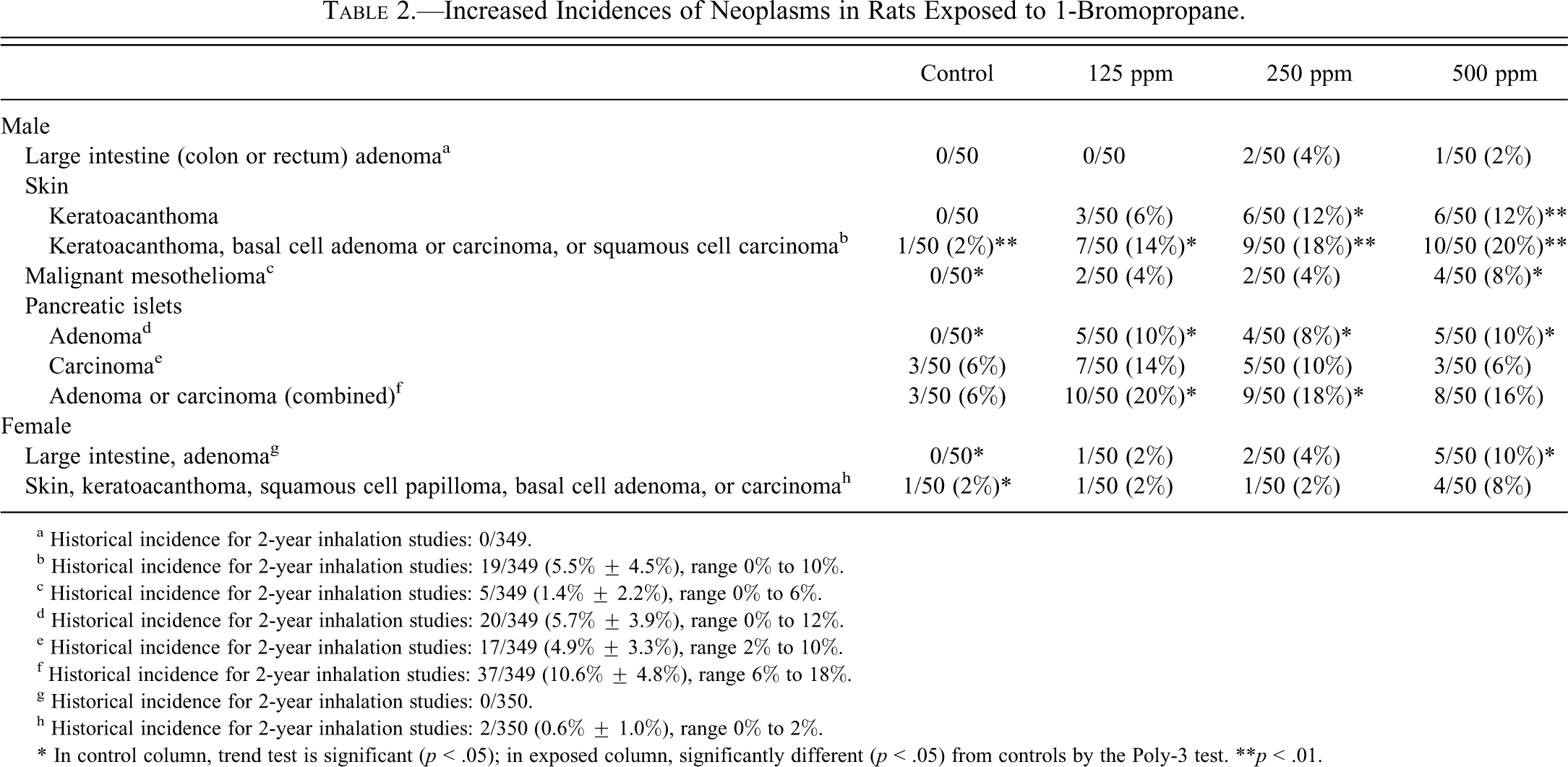
a Historical incidence for 2-year inhalation studies: 0/349.
b Historical incidence for 2-year inhalation studies: 19/349 (5.5% ± 4.5%), range 0% to 10%.
c Historical incidence for 2-year inhalation studies: 5/349 (1.4% ± 2.2%), range 0% to 6%.
d Historical incidence for 2-year inhalation studies: 20/349 (5.7% ± 3.9%), range 0% to 12%.
e Historical incidence for 2-year inhalation studies: 17/349 (4.9% ± 3.3%), range 2% to 10%.
f Historical incidence for 2-year inhalation studies: 37/349 (10.6% ± 4.8%), range 6% to 18%.
g Historical incidence for 2-year inhalation studies: 0/350.
h Historical incidence for 2-year inhalation studies: 2/350 (0.6% ± 1.0%), range 0% to 2%.
* In control column, trend test is significant (p < .05); in exposed column, significantly different (p < .05) from controls by the Poly-3 test.
**p < .01.
Skin
The incidences of keratoacanthoma in 250- and 500-ppm males were significantly increased (Table 2). The incidences of combined keratoacanthoma, basal cell adenoma, basal cell carcinoma, or squamous cell carcinoma were significantly increased in all exposed groups of males. In female rats, there was a significant exposure-related trend in the combined incidences of squamous cell papilloma, keratoacanthoma, basal cell adenoma, or basal cell carcinoma; however, the increased incidences in the 500-ppm group, while exceeding the historical control range, were not statistically significant (Table 2).
Keratoacanthomas were well-demarcated, variably sized, crateriform masses in the dermis composed of squamous epithelium that formed thick folds. The center of the mass was filled with abundant keratin. Squamous cell carcinomas were characterized as masses composed of cords of pleomorphic squamous cells infiltrating the dermis and/or subcutis. There were varying amounts of keratin and fibrous connective tissue, which separated the cords of squamous cells within the neoplasms. Basal cell adenomas were well-demarcated masses composed of cords and lobules of basal cells with areas of sebaceous or squamous differentiation. Basal cell carcinomas had a similar appearance, but the cells in the carcinomas were more pleomorphic. The carcinomas were locally invasive and often contained areas of necrosis.
Malignant Mesothelioma
The incidence of malignant mesothelioma in 500-ppm male rats was significantly greater than that in the controls (Table 2). This neoplasm was found in the epididymis in all affected animals with other tissues variably affected, particularly the testis, which was affected in all but one animal. This suggested that the tunica vaginalis, the most common site of origin for mesotheliomas in F344/N rats (Hall 1990), was the site of origin for all mesotheliomas in this study. These mesotheliomas were characterized by numerous, complex, papillary structures composed of pedunculated, fibrovascular stalks covered by one to several layers of cuboidal to flattened mesothelial cells. The stroma was prominent and often contained clusters of pleomorphic mesothelial cells that sometimes formed disorganized tubular structures. There was often extensive invasion of skeletal muscle and adipose tissue but minimal invasion of underlying tissues at other sites.
Pancreatic Islets
In male rats, the incidences of pancreatic islet adenoma were significantly increased in all exposed groups compared with the controls (Table 2). The incidences of pancreatic islet carcinoma were increased in the 125- and 250-ppm groups and met or exceeded the upper range of the historical controls, but these increases were not statistically significant. The incidences of pancreatic islet adenoma or carcinoma (combined) were significantly increased in the 125- and 250-ppm groups. Adenomas were usually discrete, well-circumscribed masses of islet cells that were 1 mm in diameter or larger and compressed the surrounding acinar tissue. Occasionally, adenomas were encapsulated by a thin band of fibrous connective tissue. In some adenomas, groups of exocrine pancreatic acini were present within the adenomas at their periphery. Carcinomas, which tended to be larger than adenomas, were characterized by varying degrees of atypia and pleomorphism of the neoplastic cells, and they typically invaded the surrounding fibrous capsule or pancreatic tissue.
Nonneoplastic Lesions
Systemic Chronic Suppurative Inflammation with S-H Material
The number of animals with multiple masses was increased in the 500-ppm groups. These lesions were located predominantly in the nose (Figure 2a, b) and/or skin but also in the bone, ear, Harderian gland, larynx, lung, muscle, peritoneum, preputial gland, and prostate. In most cases, these lesions were microscopically shown to be suppurative inflammation, many with S-H material.
Both male and female rats had concentration-related increases in the incidences of chronic suppurative inflammation that contained S-H material. These lesions were not seen in controls (Table 3 ). Most of these lesions were observed macroscopically as soft, pale yellow to green, variably sized masses or foci. Microscopically, these lesions were characterized by variably sized accumulations of mostly degenerate neutrophils (abscesses) that surrounded a central nodule of S-H material. S-H material was composed of homogenous, eosinophilic material that formed a radiating corona of club-shaped bodies and often surrounded a bacterial colony. The abscesses were separated and surrounded by fibrous connective tissue with numerous inflammatory cells including macrophages, neutrophils, lymphocytes, and plasma cells.
Organ Distribution and Incidence of Suppurative Inflammation with Splendore-Hoeppli Material in Rats Exposed to 1-Bromopropane.

a Number of animals with a lesion.
b Total number of lesions for all animals in the treatment group.
Nose
Increased incidences of chronic suppurative inflammation, chronic active inflammation respiratory epithelial hyperplasia, and respiratory metaplasia of the olfactory epithelium were also diagnosed in female rats; chronic suppurative inflammation was also increased in male rats (Table 4 ). Chronic active inflammation was present in many animals of both sexes, including the chamber controls, but in females, the incidences in all exposed groups were significantly increased compared with that in the chamber controls (Table 4). Chronic active inflammation was characterized by the presence of lymphocytes and macrophages, with fewer neutrophils within or around the nasolacrimal duct, or within the lumina of submucosal glands or the lamina propria of the septum and turbinates. The incidences of respiratory epithelial hyperplasia were significantly increased in 125- and 500-ppm females, and the incidence of respiratory metaplasia of the olfactory epithelium was significantly increased in 500-ppm females (Table 4). Respiratory epithelial hyperplasia consisted of thickening of the respiratory epithelium by increased numbers of tall, columnar, respiratory epithelial cells, and respiratory metaplasia of the olfactory epithelium was characterized by replacement of the olfactory epithelium by ciliated respiratory epithelium. The incidences of glandular hyperplasia were significantly increased in all exposed groups of male and female rats (Table 4). This lesion was characterized by mild to moderate proliferation of submucosal glands and was sometimes associated with inflammation. These lesions were most commonly observed in Level II of nasal sections.
Increased Incidences of Nonneoplastic Lesions of the Respiratory Tract in Rats Exposed to 1-Bromopropane.
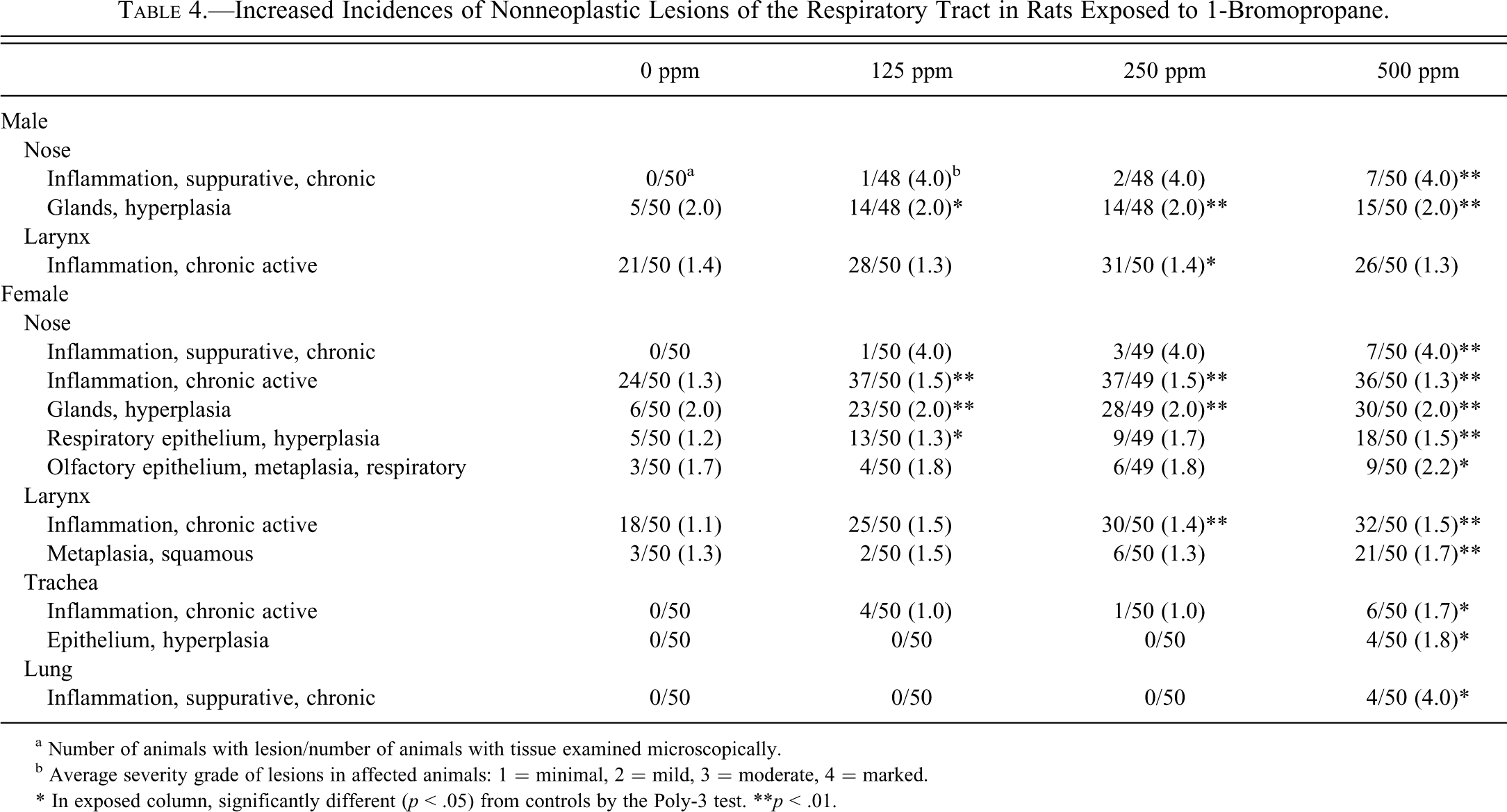
a Number of animals with lesion/number of animals with tissue examined microscopically.
b Average severity grade of lesions in affected animals: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
* In exposed column, significantly different (p < .05) from controls by the Poly-3 test.
**p < .01.
Larynx
The incidences of chronic active inflammation in 250-ppm males and females and 500-ppm females were significantly increased (Table 4). This lesion consisted of minimal to moderate numbers of lymphocytes, macrophages, and neutrophils within the lamina propria and glandular lumena. In females, the incidence of squamous metaplasia in the 500-ppm group was significantly greater than that in the chamber controls. Squamous metaplasia was characterized by the replacement of normal respiratory epithelium by squamous epithelial cells.
Trachea
In females, the incidences of chronic active inflammation and epithelial hyperplasia were significantly increased in the 500-ppm group (Table 4). Both lesions also occurred in males, but the incidences were not significantly increased. Chronic active inflammation was characterized by the presence of minimal to moderate infiltrates of lymphocytes, and macrophages, and fewer neutrophils in the lamina propria. Hyperplasia of the epithelium consisted of increased numbers of respiratory epithelial cells with densely packed nuclei.
Lung
The incidence of suppurative inflammatory lesions with S-H material was significantly increased in the 500-ppm female group (4/50) compared with controls (Table 4). There were also increases in the incidences of these lesions in the 250- and 500-ppm male dose groups (1/50 and 3/50, respectively), but these increases were not statistically significant. These lesions were morphologically similar to those seen in other organs, as described above.
B6C3F1/N Mice
Survival, Body Weights, and Clinical Findings
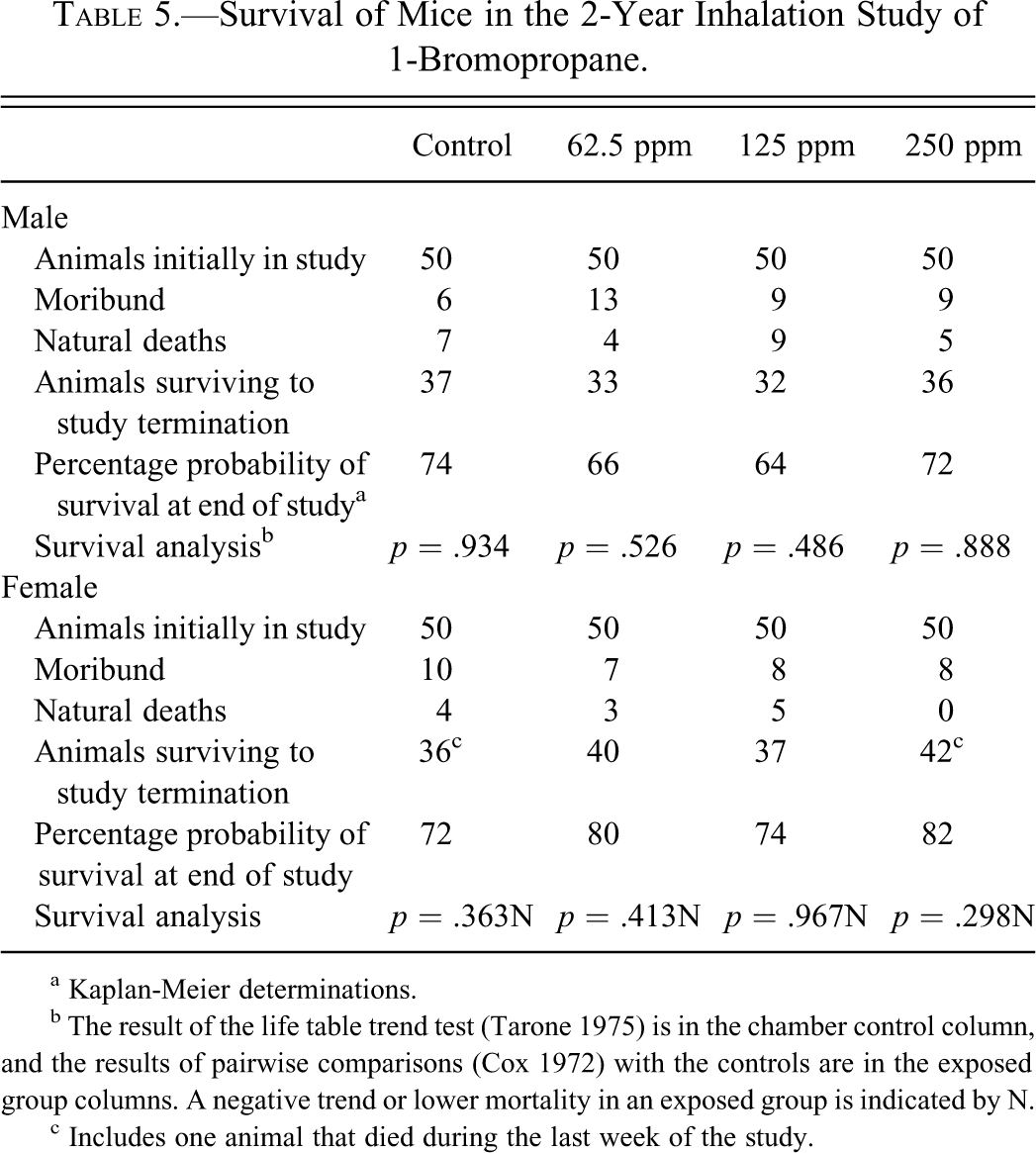
Survival of exposed groups of mice was similar to that of the control groups (Table 5 ). Mean body weights of exposed groups of male and female mice were within 8% of the controls throughout the study (data not shown). No clinical findings related to exposure to 1-BP were observed in mice.
Survival of Mice in the 2-Year Inhalation Study of 1-Bromopropane.
a Kaplan-Meier determinations.
b The result of the life table trend test (Tarone 1975) is in the chamber control column, and the results of pairwise comparisons (Cox 1972) with the controls are in the exposed group columns. A negative trend or lower mortality in an exposed group is indicated by N.
c Includes one animal that died during the last week of the study.
Histopathology
Statistically significant or biologically noteworthy changes were observed in the incidences of neoplasms of the lungs of female mice exposed to 1-BP. In female mice, there were treatment-related increased incidences of alveolar/bronchiolar (A/B) adenoma, A/B carcinoma, and A/B adenoma or carcinoma (combined). The incidences of A/B adenoma or carcinoma (combined) were significantly increased in all exposed groups (Table 6 ). The incidence of A/B adenoma in 250-ppm females and the incidences of A/B carcinoma in 62.5- and 125-ppm females were significantly increased. A/B carcinomas varied from moderately differentiated, circumscribed lesions to anaplastic, poorly circumscribed, infiltrative lesions. They were variable in size and shape, and their growth patterns were papillary or solid with regions of squamous metaplasia, spindle cell differentiation, and necrosis. A/B adenomas were usually smaller, well-differentiated, circumscribed lesions that often compressed the adjacent parenchyma, and most were papillary.
Increased Neoplasms of the Lung in Female Mice Exposed to 1-Bromopropane.
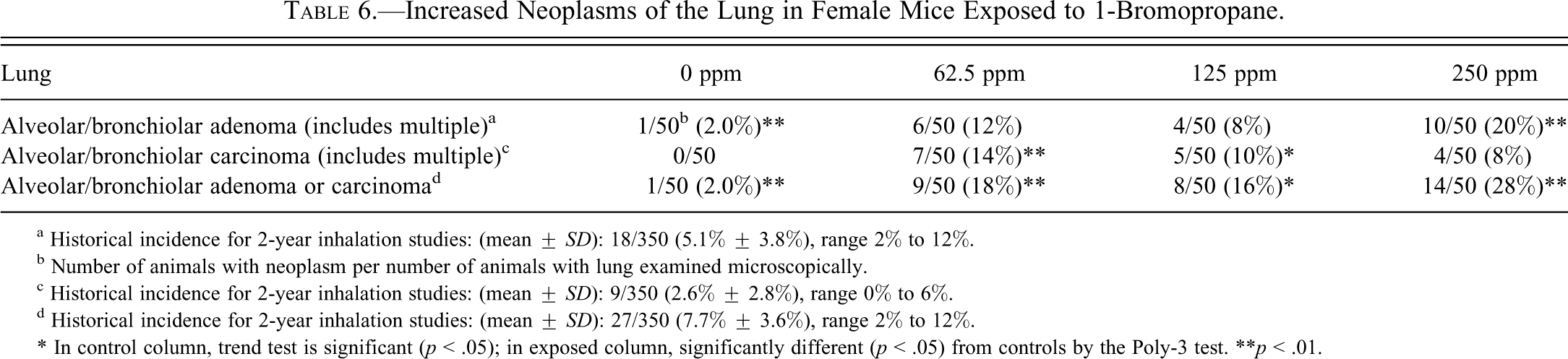
a Historical incidence for 2-year inhalation studies: (mean ± SD): 18/350 (5.1% ± 3.8%), range 2% to 12%.
b Number of animals with neoplasm per number of animals with lung examined microscopically.
c Historical incidence for 2-year inhalation studies: (mean ± SD): 9/350 (2.6% ± 2.8%), range 0% to 6%.
d Historical incidence for 2-year inhalation studies: (mean ± SD): 27/350 (7.7% ± 3.6%), range 2% to 12%.
* In control column, trend test is significant (p < .05); in exposed column, significantly different (p < .05) from controls by the Poly-3 test.
**p < .01.
Nonneoplastic Lesions of the Respiratory Tract
Lungs
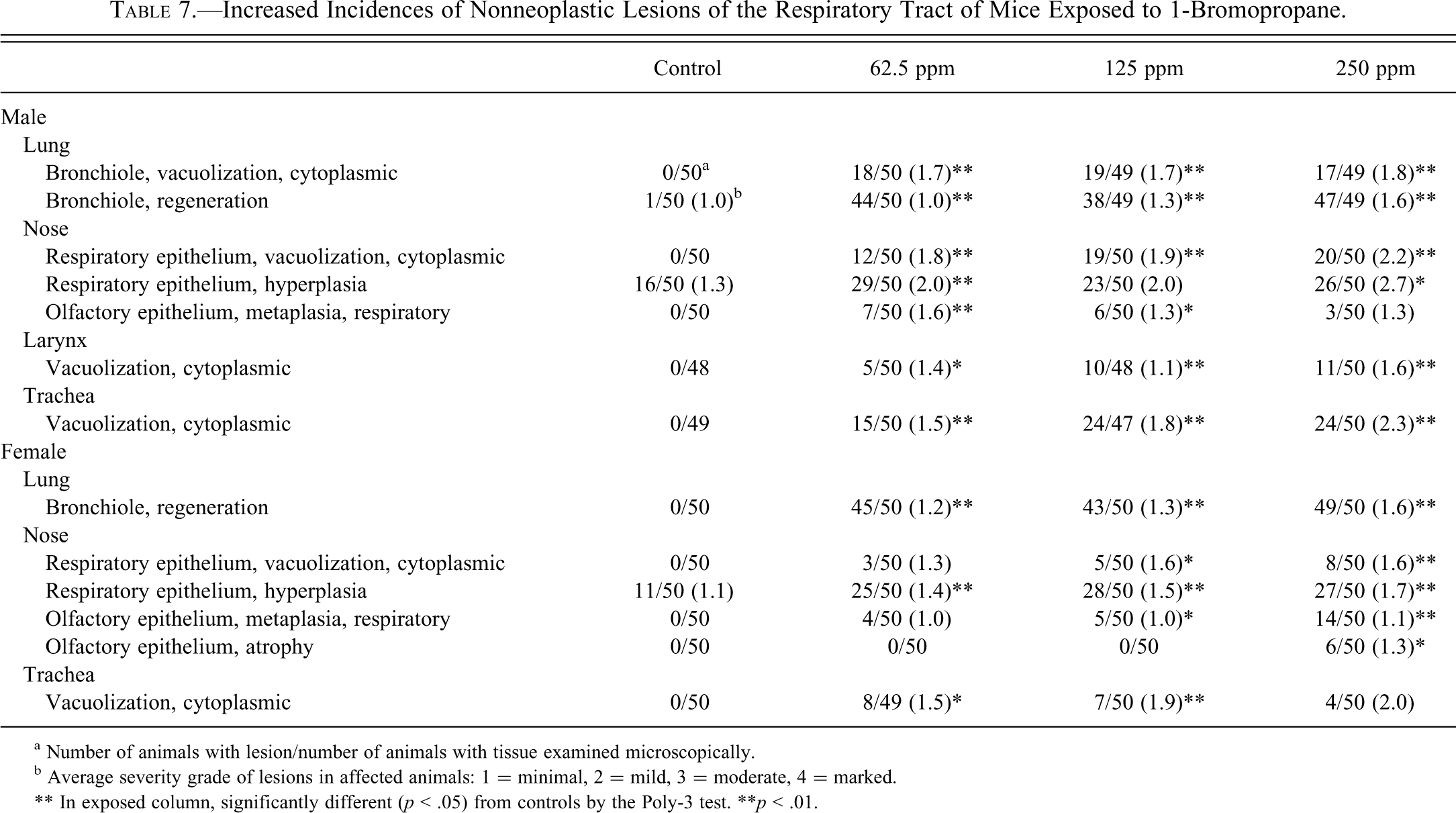
Cytoplasmic vacuolization of bronchiolar epithelium occurred in all exposed groups of male mice, and the incidences in all exposed groups of male mice were significantly increased (Table 7 ). This lesion did not occur in control mice. Histologically, this lesion was characterized by large, solitary, clear vacuoles, expanding the cytoplasm of bronchiolar epithelial cells. Affected epithelial cells often lacked cilia. Cytoplasmic vacuolization of bronchiolar epithelium often accompanied bronchiolar regeneration (Table 7). Bronchiolar regeneration occurred in most exposed male and female mice but not in controls (except for a single control male with minimal regeneration). The incidences were similar among exposed groups, although slight exposure concentration–related increases in severities were noted. Histologically, bronchiolar regeneration was characterized by disorganized, elongated, and flattened epithelial cells lining terminal bronchioles (Figure 3a, b). Regenerative epithelial cells often had increased basophilia, lacked cilia, had variable amounts of cytoplasm, and had increased nuclear to cytoplasmic ratios. Nuclei of regenerative cells tended to be apical rather than basilar and often varied in size and shape. In the affected bronchioles, there was evidence of cell loss. Many of the affected bronchioles contained sloughed epithelial cells and occasional necrotic epithelial cells characterized by small, pyknotic nuclei and homogenous, hypereosinophilic cytoplasm.
Increased Incidences of Nonneoplastic Lesions of the Respiratory Tract of Mice Exposed to 1-Bromopropane.
a Number of animals with lesion/number of animals with tissue examined microscopically.
b Average severity grade of lesions in affected animals: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
** In exposed column, significantly different (p < .05) from controls by the Poly-3 test. **p < .01.
Nose
There were exposure concentration–related increased incidences of cytoplasmic vacuolization of respiratory epithelium in male and female mice; the incidences in all exposed groups of males and in 125- and 250-ppm females were significantly increased (Table 7). This lesion did not occur in control mice. Cytoplasmic vacuolization involved the respiratory epithelium on the lateral surfaces of nasoturbinates in Levels I and II. Histologically, this lesion was characterized by large, solitary, clear vacuoles expanding the cytoplasm of respiratory epithelial cells. Oftentimes, affected epithelial cells lacked cilia. Occasionally, nuclei of affected epithelial cells were pyknotic.
There were exposure-related increased incidences of respiratory epithelial hyperplasia in the dorsal meatus(es) in Level I in mice of both sexes (Table 7); the incidences in all exposed female groups and in 62.5- and 250-ppm males were significantly increased. Although present in controls, the incidence and severity of respiratory epithelial hyperplasia increased with increasing exposure concentration, and lesions in male mice were more severe than those in females. Histological criteria for respiratory epithelial hyperplasia included thickened epithelium due to increased cellularity, submucosal epithelial invaginations with intraepithelial cryptlike structures (pseudo-glands), and/or fingerlike projections of epithelium that extended into the nasal cavity.
There were exposure-related increased incidences of respiratory metaplasia of olfactory epithelium in male mice and exposure concentration-related increases in female mice; incidences of this lesion were significantly increased in 62.5- and 125-ppm males and 125- and 250-ppm females (Table 7). This lesion was not observed in chamber control mice. Located at Levels II and/or III of the nasal sections, the lesion was similar to that described in the rats and was often associated with submucosal inflammation and/or glandular hyperplasia.
The incidence of olfactory epithelial atrophy was significantly increased in 250-ppm females (Table 7). The incidences of this lesion were slightly increased in all exposed groups of males, but the increases were not statistically significant. This lesion consisted of segmental thinning of the olfactory epithelium at Level II and/or III of the nasal cavity due to loss of neuronal cells with primarily sustentacular and basal cells remaining. It was often associated with submucosal inflammation and/or glandular hyperplasia and usually was present within the dorsal meatuses.
Larynx and Trachea
There were increased incidences of cytoplasmic vacuolization of respiratory epithelium in the larynx and the trachea of exposed males and trachea of females; the incidences were significantly increased in both tissues in all exposed groups of males and in the trachea of 62.5- and 125-ppm females (Table 7). This lesion did not occur in control mice. Histologically, this lesion was characterized by large, solitary, clear vacuoles expanding the cytoplasm of respiratory epithelial cells. Affected epithelial cells often lacked cilia. Occasionally, nuclei of affected epithelial cells were pyknotic.
Discussion
The results of these studies provide evidence that 1-BP is a multisite carcinogen in rodents. In female rats, 1-BP caused a significant increase in the incidence of adenoma of the large intestine (colon and rectum). The incidences of adenoma increased with exposure concentration, and the incidence in the 500-ppm group was significantly greater than that in the controls. In addition, adenomas were found in the large intestine of two 250-ppm male rats (4%) and in one 500-ppm male rat (2%). Although the increased incidences in males were not statistically significant, the presence of these neoplasms in females and the low historical occurrence of these neoplasms in NTP studies (0% in inhalation controls and less than 0.2% in control male and female rats by any route of exposure) suggest that they were exposure related. No potentially preneoplastic changes (i.e., hyperplasia, dysplasia) were noted in the intestinal epithelium in these 2-year studies.
There was some evidence that chronic exposure to 1-BP caused an increase in the incidences of neoplasms of the skin in male rats. The combined incidences of all neoplasms of epithelial origin increased in a significant exposure concentration–dependent manner. The incidences were significantly increased in all exposed groups of males relative to controls. The most numerous neoplasms were identified as keratoacanthoma, a benign neoplasm of squamous epithelium. The incidences of keratoacanthoma increased with a positive trend, and the incidences in the 250- and 500-ppm groups were significantly increased compared with controls. The increased incidence of keratoacanthoma is a concern because there is evidence that this neoplasm can progress to squamous cell carcinoma, a highly malignant tumor (Elwell, Stedham, and Kovatch 1990). Squamous cell carcinoma is a rare neoplasm occurring in only 0.6% of historical controls from all NTP studies. Although not statistically significant, the incidence of squamous cell carcinoma in the 500-ppm male rats was greater than that in the concurrent controls and exceeded the historical control range for NTP studies using all routes of administration.
In female rats, the incidences of squamous cell papilloma, keratoacanthoma, basal cell adenoma, or basal cell carcinoma (combined) in the 500-ppm group exceeded the NTP historical control ranges for inhalation studies. Keratoacanthomas occurred in females, but unlike in the males, the incidences were not increased compared with the concurrent controls or historical controls. No squamous cell carcinomas were seen in female rats.
Although an exposure-related increased incidence of malignant mesothelioma was seen in the 500-ppm exposed male rats, it was unclear whether these lesions were exposure related. In this study, the incidence in the 500-ppm dose group was significantly increased relative to controls, but the incidence in this group exceeded the historical control range by only a single neoplasm. Mesothelioma is a relatively common neoplasm of F344/N male rats, and the tunica vaginalis is the most common site of origin, whether occurring spontaneously or as an exposure-related lesion (Maronpot et al. 2009). In addition, there is an apparent association with the high background incidence of testicular Leydig-cell tumors of these rats. It has been suggested that hormone imbalance brought about by perturbations of the endocrine system is a key factor leading to both spontaneous and exposure-associated mesotheliomas in the tunica vaginalis (Maronpot et al. 2009).
Increased incidences of pancreatic islet adenoma in male rats were statistically significant at all exposure concentrations relative to the concurrent controls. However, because the incidence in the controls (0/50) was less than the mean in historical controls from inhalation studies (6%), and the incidences in the exposed groups did not exceed the historical ranges of all studies, we considered these effects to be equivocal.
Many straight chain brominated hydrocarbons have been shown to be potent carcinogens in rodents. Chronic inhalation exposure of F344/N rats to 1,2-dibromoethane (ethylene dibromide) caused carcinomas and adenocarcinomas of the nasal cavity and hemangiosarcomas of the circulatory system in males and females, mesotheliomas of the tunica vaginalis and adenomatous polyps of the nasal cavity in males, and fibroadenomas of the mammary gland and alveolar/bronchiolar adenomas and carcinomas in females (NTP 1982a). 1,2-Dibromoethane was also carcinogenic in B6C3F1 mice, causing alveolar/bronchiolar adenomas and carcinomas in males and females and hemangiosarcomas of the circulatory system, fibrosarcomas in subcutaneous tissue, carcinomas of the nasal cavity, and adenocarcinomas of the mammary gland in females. Inhalation exposure to 1,2-dibromo-3-chloropropane caused carcinomas, squamous-cell carcinomas, and adenocarcinomas of the nasal cavity and squamous cell papillomas of the tongue in male and female F344/N rats (NTP 1982b). In B6C3F1/N mice, 1,2-dibromo-3-chloropropane caused adenocarcinomas and papillary carcinomas of the nasal cavity in females and squamous cell carcinomas of the nasal cavity and alveolar/bronchiolar adenomas and carcinomas in males and females. In addition to these multisite carcinogens, other brominated hydrocarbons have been shown to be carcinogenic in rodents, although with fewer target sites (Dunnick, Eustis, and Lilja 1987).
Although the mechanism(s) by which brominated hydrocarbons cause tumors in rodents is not clear, it likely involves an initial metabolic activation of the chemical to a reactive intermediate. Metabolism of many halogenated hydrocarbons results in the formation of strong alkylating intermediates. In mice, 1-BP is oxidatively metabolized in the liver by cytochrome P450 2E1 (CYP2E1) to reactive metabolites that may be responsible for the toxicity of 1-BP (Garner et al. 2006; Liu et al. 2009). 2-Oxo-1-bromopropane, a highly reactive oxidative metabolite of 1-BP (Robinson et al. 1989), has been shown to alkylate proteins and inactivate enzymes (Beeley and Neurath 1968; Cohen et al. 1982; Mitchell et al. 1998). The carcinogenic mechanism of 1-BP may involve alkylation of essential proteins or nucleic acids by reactive metabolites. Alternatively, 1-BP toxicity and carcinogenicity may result from oxidative stress due to depletion of cellular glutathione (GSH) by 1-BP metabolites. Glutathione is important in protecting tissues from alkylating metabolites, and marked reduction or depletion of liver GSH results in enhanced toxicity (Ketterer, Coles, and Meyer 1983). Detoxification of 1-BP metabolites occurs primarily via glutathione-S-transferase (GST)–mediated conjugation with GSH (Liu et al. 2009). Chronic exposure to 1-BP may have produced levels of metabolites that exceeded the amount of GSH available for conjugation. Oxidative metabolites of 1-BP may also deplete GSH by inhibiting enzymes required for GSH synthesis. Alternatively, 1-BP metabolites may inhibit hepatocellular GST, thereby preventing GSH conjugation (Liu et al. 2009). Additional studies are needed to determine the mechanism of 1-BP toxicity and carcinogenicity.
The exposure-related increase in the incidences of chronic active inflammation in the nasal cavity, larynx, and trachea in the rats, and the increased incidence of cytoplasmic vacuolization of the respiratory epithelium in the nasal cavity, larynx, trachea, and bronchiolar epithelium in the mice, may reflect the local irritant activity of 1-BP. Degeneration of the upper and lower respiratory tract epithelium is a frequently observed effect of inhaled materials of low toxicity or of lower concentration of potent toxicants (Miller and Renne 1996). The progression to inflammation of the same sites, as seen in the rats, is suggestive of greater irritant potential of 1-BP in rats compared with the mice. Although 1-BP was a lung carcinogen in female mice, the association of the lung tumors with the irritant effects of 1-BP are unclear due to the lack of a lung tumor response in male mice.
An unusual nonneoplastic finding in rats exposed to 1-BP for 2 years was the presence of S-H reaction material associated with chronic suppurative inflammation. Lesions with S-H material were present primarily in the nose and skin of exposed male and female rats, although other sites were affected. Skin lesions were not associated with a specific location on the body. The exact composition and mechanism of formation of S-H material is unknown but it is thought to represent deposition of antigen-antibody complexes and debris from host inflammatory cells (Hussein 2008). Alternatively, it may represent glycoproteins, lipid, and calcium derived from host leukocytes (Bhagavan, Ruffier, and Shinn 1982). S-H material is typically found surrounding or adjacent to the causative agent, typically fungi, helminthes, or bacteria (Hussein 2008). In the United States, S-H bodies are often seen in association with botryomycotic infections caused by Proteus, Escherichia coli, Pseudomonas aeruginosa, or Staphylococcus (Percy and Barthold 2001; Weinig, Smirniotopoulos, and Heffner 1996). In this study, cultures were obtained from abscessed lesions of the skin, Harderian gland, tail, salivary gland, and head of 1-BP–exposed rats. These cultures were positive for P. aeruginosa in four of five 1-BP–exposed rats with abscessed lesions. Abscesses, other than those of the preputial and clitoral glands, are uncommon in rats in chronic studies. A culture from a representative clitoral gland abscess in a 1-BP–exposed rat was negative for P. aeruginosa.
The presence of lesions with S-H bodies in rats was considered to be exposure related. The incidence of lesions with S-H bodies increased with 1-BP exposure concentration and was considerably higher in males (34%) and females (28%) exposed to 500 ppm. Lesions with S-H bodies were not present in control rats. Although the mechanism is not clear, immunosuppression in 1-BP–exposed rats may have contributed to the development of S-H bodies. The development of botryomycosis and formation of S-H bodies in humans may be facilitated by immunosuppression, as it has been associated with diabetes, immunoglobulin deficiency, cutaneous anergy, corticosteroid treatment, and AIDS (Schlossberg et al. 1980; Brunken, Lichon-Chao, and van den Broek 1983; Patterson, Kitces, and Neafie 1987). It is not clear why lesions with S-H bodies were present only in rats, since immunotoxicity tests on subgroups of animals demonstrated that both rats and mice were immunosuppressed after 1-BP exposure (Anderson et al. 2010). Species differences in the presence of opportunistic bacteria, or differences in innate resistance to infection, may have played a role in this difference. Despite the reported immunosupression for 1-BP (Anderson et al. 2010), in this study we observed no morphologic changes in the thymus, spleen, lymph nodes, and so forth that may also be associated with immunosuppression.
Footnotes
Acknowledgments
This research was carried out under the auspices of the National Toxicology Program and supported by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences (NIEHS). The authors wish to thank Drs. Cunny and Dixon for critical review of this article. The statements, opinions, and conclusions contained herein do not necessarily represent the statements, opinions, or conclusions of the NIEHS, NIH, or U.S. government.
