Abstract
The global practice of drug development is expanding into many different continents and countries. India, in particular, is rapidly emerging as an economic force in this arena by offering ever-expanding opportunities for pharmaceutical market expansion as well as productive drug development partnerships. The key to the country's current socioeconomic success appears to be education, particularly the development of higher and professional education. Also, recent modifications to India's patent laws offer greater protections and incentives for international investment. Increasing numbers of competent contract research organizations create attractive opportunities for large Western pharmaceutical companies with a desire to gain access to burgeoning markets as well as mitigate the rising cost of drug development with less costly services. Well-trained veterinary pathologists are available, appropriate facilities are being constructed, and laboratory capabilities are expanding. Developing a productive partnership with a credible laboratory service in India, as with any new provider, requires due diligence and knowledgeable scrutiny of key elements of the work stream, such as facilities, education and training of laboratory personnel, Good Laboratory Practices, animal care, timelines, and data management. Ultimately and with appropriate management, mutually beneficial drug development partnerships are available in India.
Keywords
Introduction
The professional climate in biopharmaceutical companies continues to evolve with business demands and changes in the world economy. These changes create new challenges and opportunities for pathologists. Historically, many intermediate- and larger-sized biopharmaceutical companies claimed “global” or international business practices. Major emphasis was placed on operations in North America, Western Europe, Japan, and Australia. With time and economic stimulation, working definitions have evolved. Today the global practice of biopharmaceutical development has expanded into many different continents and countries with large populations. India, China, and Korea are business markets viewed as prime for development. Opportunities in these developing geographic venues span the spectrum of novel patient populations for clinical trials, expanding markets in which to sell drugs, and human and financial resources to contribute to drug discovery and development.
The Career Development and Outreach Committee and the Education Committee of the Society of Toxicologic Pathologists (STP) co-sponsored the Second Career Development Workshop entitled “Toxicologic Pathology in a Multicultural World” as part of the activities at the 2009 STP annual symposium. The workshop provided interactive sessions to discuss the challenges of doing business and communicating effectively in two geographical areas of developing business growth, China and India. Presenters representing India included V. R. Reddy, BVSc, MS, PhD, a toxicologist and native of India; and K. B. Donnelly, DVM, PhD, a veterinary pathologist with experience interacting in a professional capacity within India. Responding to the challenge by STP leadership to share career development information and training with the general membership at times other than the annual STP symposium, the workshop facilitators have summarized the materials and discussions of toxicologic pathology in India that were covered in the workshop (SCOPE Newsletter, summer 2010; STP Education Task Force Report 2010). The article that follows provides a synopsis of the workshop materials on toxicologic pathology in India and draws heavily from the personal experiences of V. R. Reddy and K. B. Donnelly. Reference literature has been added for late-breaking actions and activities of interest that have occurred in the interim between the workshop and publication of this article.
India's Drug Development Infrastructure
India, one of the oldest civilizations in the world, is multicultural. The official language of the Republic of India is Hindi, but English is the commonly spoken and written language throughout most of the country. India has a sixty-three-year-old democracy, a solid legal framework, and strong financial markets. There is an established international industry and business community. India is committed to a free market economy and globalization, and its middle-class market of approximately seventy million continues to grow. India also has a pool of personnel with high managerial and technical competence and a large skilled work force. The Indian road, railway, and airline networks are developing. Most larger cities have a good water supply twenty-four hours a day, whereas some smaller cities face water shortages in the summer season. Almost all of the electricity in India is produced by the public sector. Since power outages are common, many industry owners buy their own power generators to ensure a continuous electricity supply.
In recent years, the Indian economy has grown remarkably, and it has become a world center for information technology (IT) and IT services (The Economist, October 2, 2010). India's economic success is mainly a result of investment in its workforce education, especially higher-level and professional education. With impending patent expirations of existing drugs, a lack of new blockbuster drugs in the market, weak pipelines, and increased costs of drug development, large Western pharmaceutical companies are rushing to set up collaborations with local companies in Asia, especially in India. There are numerous Indian pharmaceutical companies and drug-development contract research organizations (CROs) involved in both the toxicology and the pathology components of drug development. Recent sweeping regulatory improvements and restructuring of both a 1970 patent law and the Indian Drugs and Cosmetics Act of 1940 have enhanced global confidence in the drug research environment in India. For the past twenty-five years, lack of patent protection allowed India to become a leading producer of generic drugs. Under new laws, Indian companies may continue to focus on the generic market, become innovator companies, or both (Gulati 2008). Some of the generic companies are also investing in providing CRO services (clinical and/or preclinical). A decade ago, the preclinical CRO sector was virtually nonexistent in India, but today, Indian CROs offer the global pharmaceutical industry an array of opportunities in preclinical contract research. In the areas of drug discovery—which include lead optimization, medicinal chemistry research, process research and development, preclinical pharmacokinetics, and toxicology—Indian companies possess enough skills in rational drug design and optimization. However, there are only a few CROs that can conduct longer-duration studies, including six-month or one-year rat and dog studies and carcinogenicity studies under Good Laboratory Practices (GLP). Western sponsors have conducted preclinical packages on Investigational New Drug–registration compounds that have progressed into human clinical trials. There are no reports that a registration has been denied, or a clinical hold enforced on the basis of preclinical work performed in India. Overall, Indian CROs have become attractive to global pharmaceutical companies because of cost, efficiency, and time.
Regulatory Challenges
The office of Drug Controller General of India, a regulatory body, is working to streamline its processes and upgrade standards by acquiring the technical know-how and best practices of the United States and Europe. Product patents for pharmaceuticals were abolished in India with the Patents Act of 1970, and they were reintroduced in January 2005 as part of India's commitment to trade-related aspects of intellectual property rights under the World Trade Organization. Between 1970 and 2005, India granted “process patents,” which protected chemical processes for production but not for the “drug” itself. Indian pharmaceutical companies used “reverse engineering” to produce generic versions of branded drugs. Under the new law, India allows the granting of “product patents,” which protect the final drug product and are recognized worldwide (Singh 2007). Thus, Indian companies are forced to have a pipeline of innovative drugs of their own. In addition, there are sweeping regulatory improvements, such as amended rules to define “new drugs,” “clinical trials,” investigational new drug applications with a Schedule Y (requirements for clinical trials) amended, regulatory inspections, and the publication of Indian GLP guidelines, which have enhanced global confidence in the drug research environment in India. Previously, development of Schedule Y–restricted “first-in-human use” molecules that were not discovered in India was discouraged. Furthermore, restrictions were imposed on phase I clinical trials for new drug substances discovered outside India. Recently, a proposal was made to remove this restriction. Also, the term “new drugs discovered in India” has been clearly defined. It covers molecules for which the first patent is filed in India, or at least part of the preclinical testing is conducted in India, or intellectual property that has been assigned to an Indian company, and/or an Indian company has development and co-development rights.
In a recent Organisation for Economic Cooperation and Development brochure (OECD 2011), India has been recognized as a “full-adherent” country for mutual acceptance of data (GLP) from safety studies of pesticides, biocides, manufactured nanomaterials, chemicals, chemical products, and products of modern biotechnology. With new patent law, changes in regulations, and now a recognition by OECD, Indian companies are developing rapidly with many integrating full drug development capability (e.g., GCP processes, GLP/quality assurance [QA]/animal welfare compliance with regulations, toxicology/absorption and drug metabolism [ADME]/safety pharmacology studies that meet global regulatory submission, bioanalytical development, and efficacy pharmacology and biology). However, gaps remain in adapting global International Conference on Harmonisation (ICH) of Technical Requirements for Registration of Pharmaceuticals for Human Use guidance documents. Although Indian pharmaceutical companies have strengths and experience in product chemistry and custom synthesis, custom manufacturing, and bioavailability and bioequivalence studies, alliances between large, established pharmaceutical companies from the United States and Europe are critical to continue the trend in India to learn the process and eventually, fully independently develop new drugs.
Veterinary Pathology and Toxicologic Pathology
Qualifications/Training of Pathologists (Anatomic and Clinical)
There are approximately 1,500 veterinary pathologists working in academe, state and national government, and the pharmaceutical industry in India. Most have arrived at these positions from training programs within India, but many others have training from outside India. There are thirty-seven veterinary colleges in India, most of which offer both graduate and postgraduate programs. Training in veterinary medicine begins at matriculation to university from high school, and the veterinary degree is the bachelor of veterinary science (BVSc), which is modeled on the British qualification and is analogous to the doctor of veterinary medicine (DVM) in the United States. Admission to veterinary school is competitive and highly regarded. Top qualifications from high school are required, and the caliber of students seeking a five-year BVSc is high. Each year, Indian veterinary schools graduate an average of 1,000 veterinary students with the BVSc degree. A small number opt for pathology specialization and enter the master of science (MS) degree program at the same or another veterinary school. There are no official residency-style pathology programs in India. Rather, an MS degree is based on pathology course work and some research activities that usually include studies on pathology aspects of naturally occurring disease or animal models of disease. These programs are rigorous and competitive for admission. Following completion of the MS degree, most new pathologists enter the work place in academe, government, or industry and work in supervised apprenticeship-style positions for several years. Mentors are usually mature, experienced pathologists who determine the time and experience required to complete the apprenticeship period. The qualifications and experience of the mentors vary widely, and there are no regulations for continuing education. Indeed, there is no board certification examination for qualification to a society of veterinary pathology in India. Some pathologists serve apprenticeships or engage in residency training in Europe or the United States and qualify with American College of Veterinary Pathologists or European College of Veterinary Pathologists certifications. Some may return to India. Overall, there is an impressive level of dedication, enthusiasm, and capability of veterinary pathologists in India despite the lack of a cohesive certifying process.
In general, clinical pathology is underemphasized compared to anatomic pathology in veterinary pathology training, and clinical pathologists are underrepresented in the cadre of veterinary pathologists. Clinical pathology interpretation in drug discovery and toxicology studies is usually performed and reported by the study director/toxicologist, who may or may not be a veterinarian and who may lack training and depth of experience. This is an area of concern and should be addressed as an issue in the standards of practice for toxicologic pathology in India.
Professional organizations for veterinary pathology consist of an active Society of Toxicologic Pathologists of India (http://www.toxpathindia.com) and an Indian Association of Veterinary Pathologists (http://www.iavp.org). These organizations admit members by subscription, publish journals, and hold annual meetings for continuing education. More broadly, and more recently, an intermediary meeting was convened to establish an Asian Board of Veterinary Specialists (AiBVS) by the end of 2011. The AiBVS has founding organizations that include Asian societies or colleges representing veterinary internal medicine, surgery, dermatology, pathology, and ophthalmology (Newsletter #2, spring 2011, European Board of Veterinary Specialists). The organization would provide an umbrella, globally recognized group for the promotion of standards of residency training programs, certifications and qualifying examinations for diplomate status, practice standards, and regulatory issues.
Institutional Facilities Dedicated to Toxicologic Pathology
There are a number of new and old commercial facilities with a very wide range of history, infrastructure, and overall quality. Very few institutions are USA GLP certified, although standards are high at most facilities. Many facilities are renovated research and development sites and some, more recently, are purpose built to custom specifications. Overall aptitude to technology is high in India, and although technical infrastructure and scientific equipment in laboratories may not be extensive, many facilities are well equipped and there is usually very good technical support. When considering partnership or contract work in India, it is essential to visit and inspect facilities in person, since Web site and printed promotional materials may not be entirely representative.
Animal Species in Drug Research and Development
In 1960, the government of India passed a prevention of cruelty to animals act and established a Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA). The CPCSEA issues guidelines for laboratory animal facilities (CPCSEA 2003). The goal of these guidelines was to promote the humane care of animals used in biomedical and behavioral research and testing; the guidelines are similar to United States and European guidelines. Indian pharmaceutical companies and CROs have extensive experience in handling and managing rodents and only limited experience in handling dogs because of Indian governmental restrictions in conducting studies. However, within the past three years, the government has begun allowing pharmaceutical companies and CROs to conduct research using dogs. In contrast, Indian pharmaceutical companies or CROs have little or no experience in conducting nonhuman primate studies. Because of widely held cultural and religious beliefs, the Indian government has not yet allowed research studies with nonhuman primates. However, there is reason to believe that this situation may change in the near future. The National Institutes of Health (United States) and the Indian Council for Medical Research have entered into a collaboration to establish a large primate colony in the city of Hyderabad. However, the colony will take at least three years to develop from the time of writing. The source stock of research animals varies widely in India, and it is always important to establish where an institution is obtaining research animals. European and American stocks are often used in breeding facilities in India, but breeding strategies and general husbandry practices vary widely and should be scrutinized. It is possible to import rodent source animals from other countries in the region, and some institutions use quality facilities in Singapore or Australia.
Pathology/Toxicology Services
There are about fifteen CRO and thirty pharmaceutical companies offering and performing toxicology and pathology services at the time of this writing. Many institutions consist of a combination pharmaceutical company with original or in-license research and development, but they also offer CRO services as part of a business plan. When considering partnering or outsourcing in India, key issues to consider include facilities and facilities management, personnel issues, program quality (GLP and non-GLP studies, GRP), standard operating procedures (SOPs), metrics and reporting, veterinary and animal welfare management programs, IT systems validation, quality control (QC)/QA practices, and issues of peer reviews.
Drug Development Partnerships/CRO Outsourcing in India
Indian pharmaceutical companies are learning the drug development process and still have relatively limited experience in conducting preclinical toxicology/ADME studies. Prior to selecting a CRO or drug development partner, potential sponsors need to understand what Indian companies can offer in preclinical development and understand their experience in conducting these studies under GLP regulations. Sponsors need to recognize strengths and weaknesses early and build a partnership with realistic expectations. Prior to placing studies, it is most beneficial to conduct the evaluation of a potential partner in two steps. During the first step, the sponsor should visit and thoroughly inspect the facility and its vendors and assess all of the criteria in Table 1. The second step involves qualifying the company by placing pilot studies and examining the process and outcome.
Initial evaluation criteria for potential contract research organizations in India.
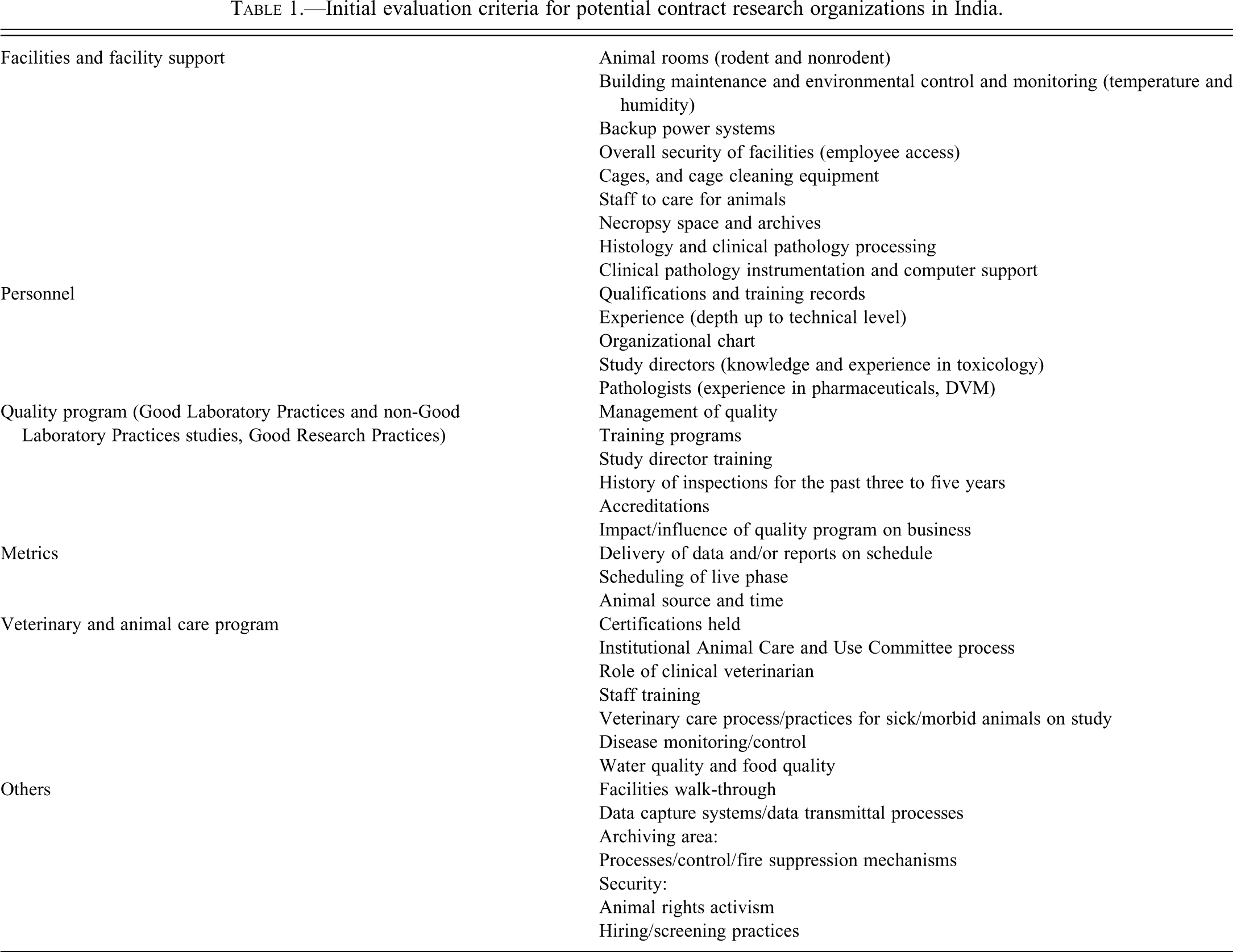
Guidelines for Inspecting a Development Partner or CRO in India
Facilities and Facilities Management
The following areas should be considered carefully in the evaluation of drug development facilities in India. Standards may be very high or surprising gaps may be discovered. In either scenario, the personnel and management are usually highly motivated, teachable, and cooperative. Inspections should include animal rooms (rodent and nonrodents), building maintenance and environmental control, caging and cage washing and storage, necropsy space and specimen handling facilities, and histology and clinical pathology laboratories, with emphasis on equipment and computer support.
Personnel
It is very important to examine personnel qualifications and training records, discuss the level of experience and cross-training, examine an organizational chart, meet study directors to assess knowledge and experience in toxicology, differentiate technical service personnel from scientific staff, and meet with pathologists to assess experience. Enthusiasm and conscientiousness are commonly found among staff and senior scientists in India, coupled with a very strong work ethic and pride in performance, even in the midst of modest facilities.
Quality Program (GLP and non-GLP Studies, GRP)
It is critical to establish who is responsible for the management of quality control at the facility. It is necessary to examine training programs for staff and directors, request a history of inspections for the past three or more years, discuss facility accreditations, and evaluate the impact/influence of the in-house quality program on the business model.
Standard Operating Procedures
It is important to read and evaluate SOPs. Constructive review and suggestions are usually very welcome, and once changes are made and instituted, they can be relied upon. In some cases it may be necessary to create SOPs and/or share a list and possibly content of SOPs from an established source in Europe or the United States. Since English is the primary language in science communities, it is not difficult to communicate and share extensively from institutional materials and also to share published peer-reviewed literature.
Metrics
Since the pace and urgency of timelines may not be clear, it is necessary to establish milestone dates for delivery of data and reports. Scheduling of live-phase and all post–live-phase activities from necropsy through reporting is worthy of particular attention. Once a pattern is set for expectations around study execution and data/reporting timelines, it will generally be adhered to with great diligence.
Veterinary and Animal Care Program
Veterinary and animal welfare programs in India suffer only slightly from lack of experience but balance the gap with diligent and conscientious personnel. For any Indian institution, it should be possible to establish what certifications are held; many are accredited by the American College of Laboratory Animal Medicine and the Association for Assessment and Accreditation of Laboratory Animal Care, International. Notwithstanding, it is important that every facility use an Institutional Animal Care and Use Committee process. Establishing the role of clinical laboratory animal veterinarian is important, as is ascertaining a perspective on intervention strategies and decision making with respect to animal welfare issues in the conduct of a study. It is important at the onset to establish expectations for veterinary care processes and practices for sick or morbid animals in a study. Also, an institutional disease monitoring and control program that includes details such as biosecurity; sentinel testing; and water, air, and food quality must be in place.
Information TechnologySystems Validation
Most institutions in India will have IT systems typical of worldwide commercial facilities and excellent technical support. However, there will likely not be dedicated study and pathology data entry systems, and hand-recorded data are often entered into a computer-program spreadsheet such as Excel. In either scenario, a robust QC/QA program will need to be in place to ensure validity and compliance. Auditing and establishing IT system validation early in a partnership or CRO relationship will save time and cost in the future.
Peer Reviews
We recommend that pathology peer reviews be performed on all study data. Receptiveness to peer reviews by Indian pathologists is very high, and the process is mutually beneficial, especially when conducted on-site.
Specialty Pathology/Toxicology Services
In India, specialty pathology services are less common. Most laboratories provide standard anatomic and clinical pathology services but have not validated special assays such as cardiac troponin. Many laboratories can perform basic or histological special stains, but more complex tissue procedures are rare and immunohistochemistry capacity is uncommon. Electron microscopy is also very rare, and other techniques such as laser capture microdissection are not available. However, the ability to quickly adopt and to perfect new procedures is remarkable, and when given the opportunity, pathologists and support staff will work tirelessly and with innovation to do so.
Guidelines for Qualifying a Development Partner or CRO in India
Based on initial inspection, a sponsor would select one or two CRO for conduct of preclinical toxicology studies. If the CRO has previously established credibility in conducting and submitting GLP studies to global regulatory authorities, then the sponsor would place a study starting with a non-GLP or shorter-duration study. However, if a CRO has not done GLP studies or worked with global pharmaceutical companies, then there is a need to take an alternate approach to establish reasonable credibility. In these cases, the decision should be made to validate these facilities using a compound with which some toxicology data has been generated.
A sponsor should send scientists and QA personnel to the selected CRO laboratory to evaluate the CRO personnel, SOPs, and documentation, and to identify the gaps and work on resolution. It is important to work on the CRO's standard protocols and bring them up to international standards following ICH and/or Organisation for Economic Co-operation and Development guidelines, as appropriate. It is also important to make sure these modified protocols fit into the facility SOPs and that there are the technical capabilities to conduct these studies. Once agreement is reached on the new protocols, strain of animals (rodents), and animal source, a sponsor would have the CRO begin a short duration (four to fourteen days) toxicology study in rodents. The sponsor would select and provide a compound for which there has already been generated short-duration toxicology/ADME data in rodents, perhaps a compound from a well-characterized class, or a terminated chemical series. The CRO conducts a rat study with limited histopathology, clinical pathology, and live-phase parameters and determines exposure levels on the first study day and on the final day. Once the CRO generates a summary report, the sponsor can compare data to the previous data. It is possible to assess overall the quality of the study, time to completion, costs, and most importantly, evaluate the quality of animals available to these facilities and identify any issues with the source of these animals. If there are any issues, such as parasitic infestation or lung lesions because of the type of bedding (rice husk), then the opportunity exists to discuss and resolve the issues with the facilities. It is also important to check whether the CRO satisfied any QA-identified gaps. If the sponsor finds that the outcome is satisfactory, then it would be best to proceed in conducting a nonrodent study, or if that is not available, a second validation study could be performed at the CRO with another four- or fourteen-day study using a different molecule or the same molecule.
If the sponsor is satisfied with all of the above studies, then it is recommended to conduct a repeat of a four-week study in the rat and the dog for a first human dose (FHD)–enabling package. Among the components of the complete FHD–enabling package would be genetic toxicology, safety pharmacology, and one-month rodent and dog toxicology studies with toxicokineticanalysis. Also, the CRO would conduct a dose form assay, develop a bioanalytical method, and complete final reports. In addition to conducting the study, the CRO should demonstrate project planning and should follow timelines. The sponsor representatives should visit the CRO during the live phase and monitor the study. If special techniques need to be developed (e.g., brain trimming), that work should be established early, and the sponsor should plan on an independent peer review by the sponsor's pathologist. Also, the sponsor should conduct a QA inspection at the end of the study (during the draft report phase), and a QA audit report with action itemsshould be submitted to the CRO. Overall, the sponsor should be able to fully evaluate the quality of the FHD–enabling package, time of generation of the FHD package, and the total cost.
Case Studies
It has been the authors' experience, when working and collaborating with institutions and scientists in India in the field of toxicology and toxicologic pathology, to expect great things and uncover some surprises. Although some of the cultural, historical, and logistical issues described above are present and challenging, the common ground is vast. No institution, facility, researcher, or study is the same, and the generalities described in this article are likely much better supported by three brief case studies illustrating some key learning points.
Summary
India is rapidly emerging as a major market for medical goods and services. An improving infrastructure, a vast population, and a rising standard of living coupled with a culture of education, collegiality, and work ethic has created great opportunity for biopharmaceutical companies that are historically active in more prosperous Western countries. Recent changes to the legal and regulatory landscapes have advanced patent protection for novel products “invented” outside India and improved care of laboratory animals. Accordingly, major pharmaceutical companies are actively pursuing ways to leverage these opportunities.
Information technology services likely represent India's flagship introduction to the global dissemination of services. Availability of clinical and preclinical, laboratory-based CRO services is also quickly developing. Historically recognized as a source of generic medications, Indian pharmaceutical companies are expanding their novel drug discovery capabilities as well as their ability to conduct clinic-enabling preclinical safety assessment studies. There are quality laboratories with relevant capabilities staffed by educated scientists available in India today with which to partner for safety assessment services. There are also laboratories that have not yet achieved the level of proficiency necessary to conduct studies supporting regulatory filings. Relevant due diligence is necessary on the part of the serviceseeker to identify a capable partner.
This article broadly outlined the opportunities and challenges offered by India's addition to the global team of drug developers striving to meet the medical needs of an expanding world population. Specific actions and guidelines were offered to assist in the development of mutually beneficial scientific partnerships.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
