Abstract
A single, solid, yellow-white thymic mass was found at necropsy of a two-year-old female cynomolgus macaque from a four-week, repeat-dose toxicity and immunogenicity study. Microscopically, the mass was multilobular and well encapsulated, surrounded by a thick connective tissue capsule, and composed of dense sheets of elongate or spindle-shaped cells and large cystic cavities separated by thick connective tissue stroma. Normal thymus was adjacent to the mass, but it was compressed. Within the mass were abundant interspersed Hassall’s corpuscles; individual and small clusters of mature, small lymphocytes; scattered eosinophils; large areas of necrosis; focal mineralization; and cholesterol clefts. An interesting feature was the presence of large multinucleated giant cells, which varied widely in size and nuclear number. Immunohistochemical staining for two lymphocyte markers and two structural proteins confirmed the identity of the neoplastic spindle cells and other cellular components. There was no evidence of vascular invasion or metastasis. Features of the thymoma indicated it was a pre-existing condition and not treatment related.
Thymomas are uncommon neoplasms in laboratory animals and are derived from the epithelial components of the thymus in which there are variable numbers of normal infiltrating T- and B-lymphocytes. Thymomas in veterinary medicine have been classified into lymphoid or lymphoepithelial varieties on the basis of the proportion of infiltrating lymphocytes (Valli 2007). Human medicine has used several different classification schemes based on histologic and immunophenotypic markers to distinguish resemblance to medullary or cortical regions, and the relative numbers of epithelial cells and lymphocytes (Levine and Rosai 1978; Marino and Muller-Hermelink 1985; Masaoka et al. 1981). The World Health Organization (WHO) classification system uses the shape and characteristics of the epithelial cells and the proportion of lymphocytes within the tumor to categorize them into types A, AB, B1, B2, or B3. This classification does not extend to thymic carcinoma, which is designated separately (Strobel et al. 2005). However, a recent publication employing meta-analysis of published data, which included survival times of patients with specific tumor types, found significant survival differences in only three types of tumors, A, AB, and B1 combined; B2; and B3. Their analysis also showed that different pathologists were not applying the diagnostic criteria of the WHO classification scheme consistently, even within a single ethnic group (Marchevsky et al. 2008). Moran and Suster (2008) proposed that this classification be further collapsed into just two diagnoses for benign tumors: “thymoma” for types A through B2, and “atypical thymoma” for type B3. Most authors agree that primary thymic epithelial neoplasms form part of a continuous spectrum of lesions that range from well-differentiated to poorly differentiated tumors.
Thymomas have been reported in many animal species, but they are considered to be rare in most, except for cats, dairy goats, the small rodent Praomys (Mastomys) natalensis, and cattle, in which the incidence in a survey conducted at 100 abattoirs in the UK during a one-year period found it accounted for 5% of all neoplasms detected at slaughter (Ecco et al. 2006). Other species in which it has been reported include laboratory rats and mice, ferrets, dogs, pigs, sheep, rabbits, fish, and a polar bear (Akiyama et al. 2009; Florizoone 2005; Hart et al. 2009; Kenny et al. 2004; López 2007; NTP 2010; Romano and Marozzi 2004; Taylor and Carpenter 1995; Wagner et al. 2005). As in humans, dogs with thymoma are prone to development of myasthenia gravis (MG) and autoimmune paraneoplastic syndrome as a result of deposition of circulating autoantibodies to acetylcholine receptors at the neuromuscular junction (Van Vleet and Valentine 2007). Except for a single case reported in a polar bear, acquired MG has not been reported in the other species. In B6C3F1 mice and F344/N rats (N > 1,100 animals for each species) used in two-year carcinogenicity studies during the past ten years, the National Toxicology Program (NTP) has reported incidences of total thymomas (benign, malignant, or not specified) of 0.09% in male mice, 0.16% in female mice, 0.16% in male rats, and 0.5% in female rats. In a proprietary rat strain derived from Spague-Dawley rats (Tif:RAI), thymic tumors were found in 4.5% of 4,281 rats examined (Naylor et al. 1988). In a fifteen-year period at one laboratory, spotaneous thymic neoplasms were found in 1.7% of the male and 5.7% of the female control Wistar rats (WI)BR (Poteraki and Walsh 1998; Walsh and Poteracki 1993). The inbred BUF/Mna rat strain is an animal model of human thymoma and has been in use for over forty years (Inoue 2010). These animals develop spontaneous thymomas with a significant lymphocytic component and most closely resemble the WHO type B1 tumor.
Reports of neoplasia in nonhuman primates are less common, and only one thymoma has been reported to date (Kotani et al 2010). A report from the Wisconsin National Primate Research Center documenting neoplasia in 2,660 Rhesus macaques (Macaca mulatta), many of which were aged animals with a range of 4.9 to 40.5 years, shows no thymic neoplasms (Simmons and Mattison 2011). Chamanza et al. (2010) reported spontaneous findings from 570 control cynomolgus monkeys, primarily of Mauritius origin, from twelve to thirty-six months of age. No neoplasms, including thymomas, were found in any of these animals. Kaspareit et al. (2007) reported thirty-three neoplasms in this species in control and treated animals (approximately 3,300 animals) greater than two and less than fourteen years of age, but no thymomas were found.
This monkey was a two-year-old female cynomolgus macaque from a routine four-week, repeat-dose toxicity and immunogenicity study with a potential therapeutic protein. The animal was purpose bred for laboratory use and was of Chinese origin. The study was conducted at Charles River Laboratories (CRL), Reno, Nevada, a facility fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care and registered with the United States Department of Agriculture (USDA). This facility has an Institutional Animal Care and Use Committee (IACUC) responsible for CRL compliance with applicable laws and regulations concerning the humane care and use of laboratory animals. The CRL IACUC reviewed and approved the animal use in this study. The monkey was individually housed in a stainless steel cage and commingled with other monkeys to provide psychological enrichment. Primary enclosures were as specified in the USDA Animal Welfare Act (9 CFR, Parts 1, 2 and 3) and as described in the Guide for the Care and Use of Laboratory Animals (ILAR publication, 1996, National Academy Press). Temperature and humidity were automatically controlled at 64° to 84°F (approximately 18°–29°C) and 30 to 70%, respectively, with a minimum of ten fresh air changes per hour. A twelve-hour light/twelve-hour dark cycle was maintained. Fresh drinking water was provided ad libitum, and animals were fed Purina Certified Primate Diet, No. 5048, daily. Analysis of the diet was performed by the manufacturer; no contaminants were known to be present in the diet at levels that would interfere with the study results. This diet was supplemented with fruit and/or vegetables two or three times a week.
The monkey underwent repeat physical exams and was serologically tested. While in quarantine, it was confirmed to be clinically healthy and serologically negative for the following viral infections: simian immunodeficiency virus (SIV), Cercopithecine herpesvirus 1 (B virus), simian retrovirus (SRV), and simian T-cell leukemia virus (STLV). Tuberculin tests, other bacteriology screening tests for Shigella and Salmonella, and parasitological screening were carried out upon arrival at the testing facility, and all were negative. The animal was prophylactically treated for parasites and vaccinated for measles virus.
Clinical and physical examinations, body weight assessments, clinical hematology/biochemistry, immunophenotyping, and urinalysis were performed periodically throughout the study. Clinical observations were limited to minor abrasions, bruises, and vaginal discharge (presumed menses). At the termination of the study, the animal was euthanized by exsanguination via the femoral veins while under deep and unrecoverable anesthesia induced by intravenous administration of ketamine and Beuthanasia-D solution. A detailed necropsy was performed under the supervision of a veterinary pathologist. A single, solid, yellow-white thymic mass was identified. There were no other significant gross findings. Multiple organ tissue samples including those of the thymic mass were collected, fixed in 10% neutral buffered formalin, embedded in paraffin wax, sectioned to 4–5 µm thickness, and processed to hematoxylin and eosin (HE)–stained slides. In addition, several formalin-fixed, paraffin-embedded samples of the mass and unaffected thymic lobe were evaluated by immunohistochemistry.
The additional sections were stained with the following mouse anti-human monoclonal antibodies: anti–CD3 (clone F7.2.38, Dako), anti-CD20 (clone L26, Dako), anti-pancytokeratin (clone AE1/AE3, Dako Carpinteria, CA), and anti-vimentin (clone V9, Dako, Carpinteria, CA). Tissue sections were incubated with primary antibody. Bound primary antibody was detected using the Dako EnVision visualization system with diaminobenzidine chromogen substrate and counterstained with hematoxylin. Staining intensity was graded from 1 to 4.
Microscopically, the mass was multilobular, surrounded by a thick connective tissue capsule, and composed of dense sheets of elongate or spindle-shaped cells and large cystic cavities separated by thick connective tissue stroma, and compressed adjacent normal thymic tissue (Figures 1A, 1C ). Additional features included abundant interspersed Hassall’s corpuscles, individual and small clusters of small lymphocytes [CD3+(pan T-cell) or CD20+(B-cell)] throughout the mass (Table 1 , Figures 1B, 1C, 2C, 2D), scattered eosinophils, large areas of necrosis, focal mineralization, and cholesterol clefts. The neoplastic spindle cells had a relatively low mitotic index and were strongly and consistently positive for vimentin (3–4+ intensity), a marker of intermediate filaments in mesenchymal cells (Table 1, Figure 2A ). A small percentage of the neoplastic spindle cells (<10%) was weakly positive for cytokeratins (AE1/AE3). Interspersed Hassall’s corpuscles were normal in size and morphology, had rare cytokeratin-positive staining (vimentin negative), and were surrounded by vimentin-positive neoplastic cells. Cytokeratin-positive neoplastic cells had 2+ to 3+ cytoplasmic staining. Large multinucleated giant cells (MGC), which varied widely in size and nuclear number (2–50), were identified throughout the mass, often associated with keratin whirls and/or bordering areas of cavitation. The multinucleated cells were vimentin positive, cytokeratin negative, and negative for lymphocyte markers (Table 1, Figures 2A, 2B). Cystic cavities were filled with necrotic, cellular debris, hemorrhage, and exfoliated vimentin-positive neoplastic cells. The dense connective tissue capsule was composed of mature collagen and interspersed with vimentin-positive mature fibroblasts. Surrounding thymic lobules had normal architecture. There was no evidence of vascular invasion or metastasis. Overall, the tumor appeared to be composed primarily of cells/tissue structures similar in immunohistochemical staining characteristics to those of the normal thymic medulla (Table 1). There were no test article–related findings identified in the thymus in this study and no other proliferative lesions identified in any tissues in any of the study animals. Because of the advanced anatomic features (mature fibrous capsule and large areas of necrosis) and the lack of test article–related findings in the thymus, the thymoma was considered to be a pre-existing condition and not treatment related.
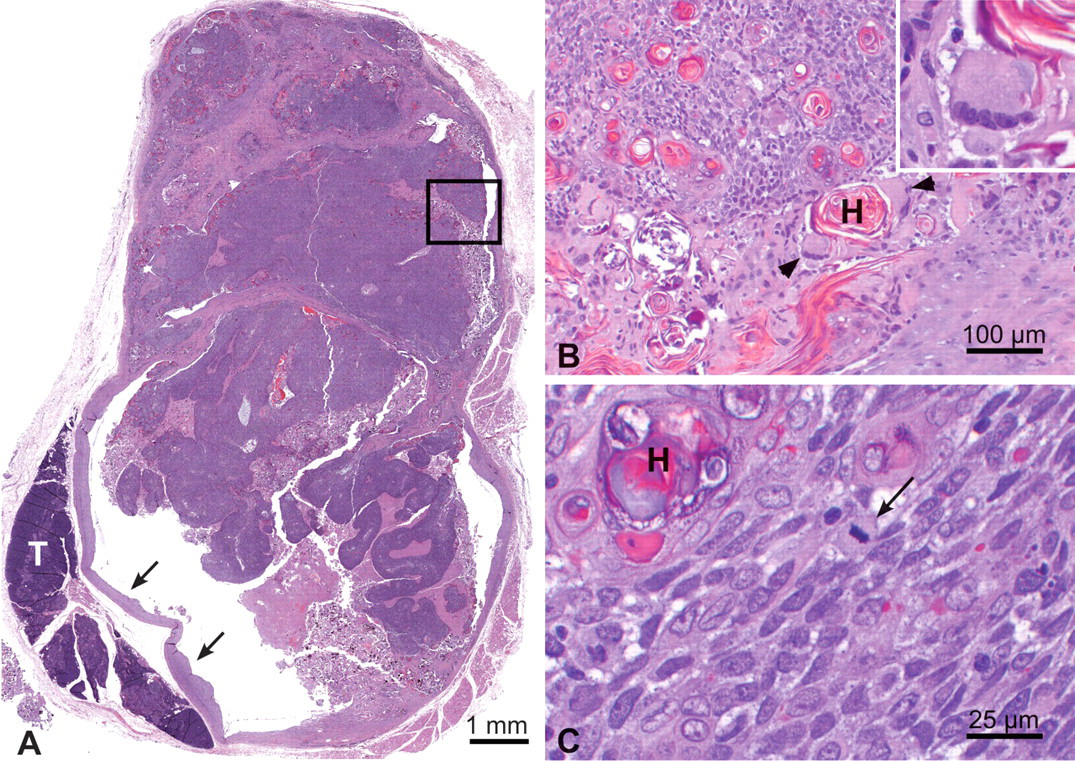
Thymus. (A) Multilobulated, well-encapsulated, expansile mass composed of sheets of neoplastic spindle cells with large cystic cavities containing abundant necrotic cellular debris and hemorrhage, and compressing surrounding normal thymic lobules (capsule shown with arrows), hematoxylin and eosin stain. (B and C) Higher magnification of area inside the box illustrating the neoplastic spindle cells, interspersed Hassall’s corpuscles, the presence of multinucleated giant cells (arrowheads), and rare mitotic figures (arrow). Higher magnification of multinucleated giant cells in inset. T, normal thymus; H, Hassall’s corpuscles.
Immunohistochemical staining of neoplastic spindle cells, multinucleate giant cells and lymphocytes.

Abbreviations: +, positive; ±, weakly positive; −, negative.
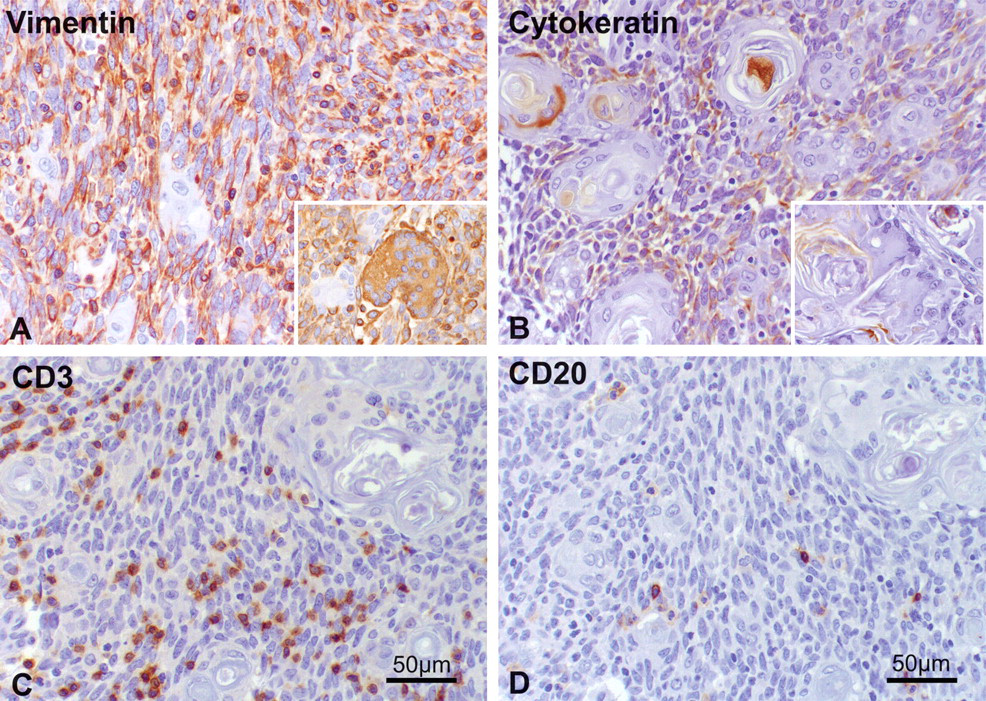
Immunohistochemistry with diaminobenzidine as chromogen and hematoxylin counterstain. The majority of neoplastic spindle cells were strongly vimentin positive (A), whereas only occasional cells had light cytokeratin staining (B). Insets (A and B) demonstrate vimentin-positive, cytokeratin-negative staining of multinucleate giant cells. Individual and small clusters of CD3+ T lymphocytes and CD20+ B lymphocytes scattered throughout the mass (C and D, respectively).
This neoplasm had, primarily, elongated, spindle-shaped nuclei most compatible with the original WHO Type A morphology (Alexiev et al 2007; Okumura et al 2008). Individual and small clusters of lymphocytes were also present within the tumor; however, they did not constitute a proliferative component. This neoplasm also had a number of other features that are not considered common in these tumors in humans, including scattered eosinophils, abundant Hassall’s corpuscles (Laeng et al 2006), and large areas of necrosis, with nearby MGC, focal mineralization, and cholesterol clefts. The MGC were usually associated with keratin whorls or bordering areas of necrosis and were of both the Langhans (Figure 1) and foreign body type. As in other conditions, formation of MGC is caused by expression of multiple specific proteins and activation of receptors on the cell membrane that leads to adhesion and fusion of macrophages to form giant cells (Brodbeck and Anderson 2009).
There were many striking similarities between the thymoma described in this report and the thymoma recently reported in a cynomolgus monkey by Kotani et al., such as the relatively young age at diagnosis, subclinical nature, benign features (well-encapsulated, low mitotic rate, and no evidence of metastasis), and prominent medullary differentiation including the presence of Hassall’s corpuscles, an unusual occurrence in human tumors. Although the behavioral characteristic of the two tumors were quite similar, there were distinct morphological differences. One of the main differences was the lack of a lymphoproliferative component in our tumor. The thymoma reported by Kotani et al. was diagnosed as a mixed Type AB thymoma (also using human thymoma WHO classification criteria) because of the presence of a dense population of proliferating thymic cortex-like lymphocytes as well as the neoplastic spindle cell population. The proliferating spindle cells were cytokeratin positive and vimentin negative. In our case, the neoplastic spindle cell population was consistently vimentin positive, only weakly cytokeratin positive with only less than 10% of the cells staining, and the cells had scattered mature CD3+ or CD20+ positive lymphocytes among them. Other prominent histological features reported in this case, but not reported by Kotani et al., include the presence of MGC, large areas of necrosis, cholesterol clefts, and mineralization. Regardless of the differences, both tumors had immunohistochemical staining characteristics similar to those of the normal thymic medulla, benign morphological features, and they were found incidentally at the time of scheduled necropsy in a toxicology study. This report should aid in the differentiation of spontaneous proliferative versus test article–induced lesions in the thymus in the laboratory nonhuman primates used in preclinical safety studies.
Footnotes
Acknowledgments
The authors wish to thank the necropsy and histopathology laboratory staff at Charles River, Preclinical Services, NV. This work was sponsored and funded by MedImmune, LLC.
