Abstract
Cardiac troponins have proved to be reliable blood biomarkers for identifying a variety of myocardial alterations in humans and animals. Recently, an ultrasensitive cTnI assay (Erenna IA) has been used to demonstrate increases in baseline cTnI resulting from drug-induced myocardial injury in rats, dogs, and monkeys, as well as to document baseline cTnI ranges in Sprague-Dawley (SD) rats. The present study was initiated to use the Erenna cTnI assay to further document baseline cTnI concentrations in normal control animals from multiple strains, including SD, Spontaneous Hypertensive (SHR), Wistar, Wistar-Kyoto (WKY), and Fisher strains. Baseline cTnI concentrations were quantified in all rats tested, and males had higher mean cTnI concentrations than females of the same strain. SHR males had the highest mean cTnI concentrations and the largest cTnI variability. Interestingly, cTnI concentrations increased in castrated SHR compared with unaltered male SHR, whereas cTnI concentrations decreased in ovariectomized SHR compared with unaltered female SHR. These results show significant differences in cTnI concentrations between strains, sexes, and noncardiac surgical alterations in control animals, and identify these as potential contributing factors to cTnI baseline variability that should be taken into account when using ultrasensitive cTnI as a biomarker to assess preclinical cardiotoxicity.
Introduction
A major advance in the detection of myocardial injury occurred with the development of immunoassays that specifically measure cardiac troponin T (cTnT) and cardiac troponin I (cTnI) isoforms in plasma or serum. Since the cardiac myocyte is essentially the sole source of cardiac troponin T or I, increased serum concentration of either of these two proteins serves as a strong indication of myocardial injury. First-generation clinical assays capable of measuring cTnI and cTnT in serum or plasma were first reported by Cummins, Auckland, and Cummins (1987) and Katus et al. (1989). Cardiac troponins have become the gold standard biochemical means for diagnosis of acute coronary syndromes (Alpert et al. 2000), and further evidence has suggested that these biomarkers can also be used to detect non-ischemic myocardial alterations (Gaze and Collinson 2005). Increased specificity and sensitivity of troponin immunoassays have expanded the utility of troponins from diagnoses of acute myocardial infarction to detecting subtle cardiac injury resulting from non-ischemic alterations (Mair 1997).
A key factor in troponin assay utility is the ability to quantify the release of very small amounts of cTn that occur early in the onset of myocyte injury or when only a minimal number of myocytes are affected. Thus, there has been increasing interest in quantifying baseline concentrations of cTn in healthy populations to develop reference ranges from which to measure these subtle increases. For example, the Joint ESC/ACC Committee for the Redefinition of Myocardial Infarction set a low threshold for evidence of an ongoing myocardial alteration by indicating that a cTn value was significant if it exceeded the 99th percentile of cTn concentrations detected in a reference population that was measured with an assay having an impression of less than 10% (CV) (Alpert et al. 2000). However, this undertaking had not been possible previously because existing commercial assays did not possess the requisite sensitivity or precision to reliably detect the very low concentrations of cardiac troponin thought to be present in serum or plasma from a population of healthy individuals. A limitation noted with many current assays is that analytical imprecision is not uniform, particularly when cTn concentrations are low (Panteghini 2002; Panteghini et al. 2004). This situation has stimulated interest in developing more sensitive assays that can detect low concentrations of cTn with improved precision. A new generation of high-sensitivity assays offer analytical improvements (Todd et al. 2007; Wu et al. 2006) and are capable of detecting cardiac troponin concentrations significantly less than those in the lower range of most currently utilized commercial assays (Melanson, Morrow, and Jarolim 2007; Wu, Lu, et al. 2009).
Commercial cardiac troponin T and I assays have been used in nonclinical situations, and both cTnT and cTnI have been used as biomarkers to detect drug-induced cardiac injury in humans (Herman and Ferrans 2001) and animals (O’Brien 2008). The ability of a human assay to detect the release of cTnT in a nonhuman species was first reported by Seino et al. (1993), who administered doxorubicin to spontaneously hypertensive rats. The utility of measuring cardiac troponins in animals was confirmed in studies reported by O’Brien et al. (O’Brien et al. 1998; O’Brien et al. 1997; O’Brien, Landt, and Ladenson 1997). These studies demonstrated that the capture antibodies used in the first- and second-generation clinical cTnT immunoassays also recognized cTnT epitopes from the hearts of a variety of animals. In addition, studies examining the affects of potentially cardiotoxic compounds such as isoproterenol and doxorubicin have detected increased serum concentrations of cTnT using the second- and third-generation Roche Elecsys cTnT immunoassays (Herman et al. 2006, 1999). In all cases, the amounts of cTn released into the circulation correlated with the type and extent of myocyte injury.
Though cardiotoxin-induced increases in cardiac troponins have been well documented in rats, data related to baseline concentrations of these proteins in rats has been limited or unavailable. For example, in recent studies using second- and third-generation cTnT immunoassays (Elecsys, Roche Diagnostics, Indianapolis, IN), concentrations of cTnT in control and low-dose doxorubicin-treated SHR were below the limit of detection (concentration < 0.01 ng/ml) in almost all instances (Herman et al. 2006, 1999). Another comprehensive study, supported by the International Life Sciences Institute/Health and Environmental Sciences Institute (ILSI/HESI), found that various clinical assays can detect cardiac troponins (I and T) in the serum of common laboratory animals (Apple et al. 2008); however, some assays were less capable than others for quantifying serum cTn concentrations in certain species. Thus, the value of monitoring cardiac troponins as biomarkers of myocardial injury in animals could be enhanced by the development of more sensitive immunoassays (Herman et al. 2006, 1999) and further characterization of baseline concentrations in multiple species and strains.
Recently, an ultrasensitive cTnI assay (Erenna immunoassay system) has been described (Todd et al. 2007) with an approximate fifty-fold improvement in sensitivity over many currently utilized troponin immunoassays. This assay uses a single-molecule counting system that significantly reduces background noise and improves sensitivity of detection (Todd et al. 2007) and is one of only a few available assays that have the sensitivity and precision to detect baseline cardiac troponin concentrations in a healthy population (Wu, Lu, et al. 2009) or in stress-test-induced transient myocardial ischemia (Sabatine et al. 2009). The Erenna ultra sensitive cTnI assay has been utilized in studies as a means to monitor isoproterenol-induced increases in cTnI above baseline in healthy rats, dogs, and monkeys (Schultze et al. 2008) and to establish the preliminary reference range of cTnI in healthy rats under standard laboratory handling conditions (Schultze et al. 2009).
In the present study, we utilized this ultrasensitive assay to further document baseline concentrations of cTnI in healthy normal control animals from multiple strains commonly used in cardiotoxicity studies, including SHR, SD, Wistar, WKY, and Fisher strains. These studies investigated the degree of variability for ultrasensitive cTnI baseline measurements of control animals between strains, sexes, and surgical alterations (i.e., castration or ovariectomization) that are common to control animals in an effort to identify potential contributing factors to cTnI baseline variability. By identifying these critical factors and taking into account expected cTnI variability in control animals being utilized in each study, researchers will be able to improve upon preclinical study design when using ultrasensitive cTnI as a biomarker to assess cardiotoxicity.
Materials and Methods
Animal Care and Use
All procedures involving animals were approved by the Institutional Animal Care and Use Committee, Center for Drug Evaluation and Research, Food and Drug Administration. We examined the serum from 127 rats (33 male and 21 female spontaneously hypertensive [SHR], 26 male and 15 female Sprague-Dawley [SD], 5 male and 5 female Wistar, 6 male Wistar-Kyoto [WKY], 7 male Fisher, 4 castrated male SHR and 5 ovariectomized female SHR). Rats were obtained from Taconic (Hudson, NY) or Harlan Industries, Inc. (Indianapolis, IN) and housed individually. Each rat was given standard rodent chow and water ad libitum. The samples selected for analysis came from nontreated control animals that were part of several in-house studies at the Center for Drug Evaluation and Research, Food and Drug Administration. Animals used in the studies were aged eight to ten weeks (SD, Wistar, and Fisher) and twelve weeks (SHR and WKY) prior to study commencement. SHR rats were aged to twelve weeks to allow for systemic arterial blood pressure to reach maximum values, and WKY were similarly aged to serve as a control. Each of the studies commenced after a one-week acclimation period. These control rats received a vehicle solution for varying periods according to the particular experimental protocol (Table 1 ). Although the duration of some studies in certain strains varied and the vehicles may have been different, cTnI values from these animals were in the same range, and thus all animals of a particular strain and sex were grouped together.
Cardiac TnI concentration (Mean ± SD) and significance (p > .05) of various rat strains and sexes.
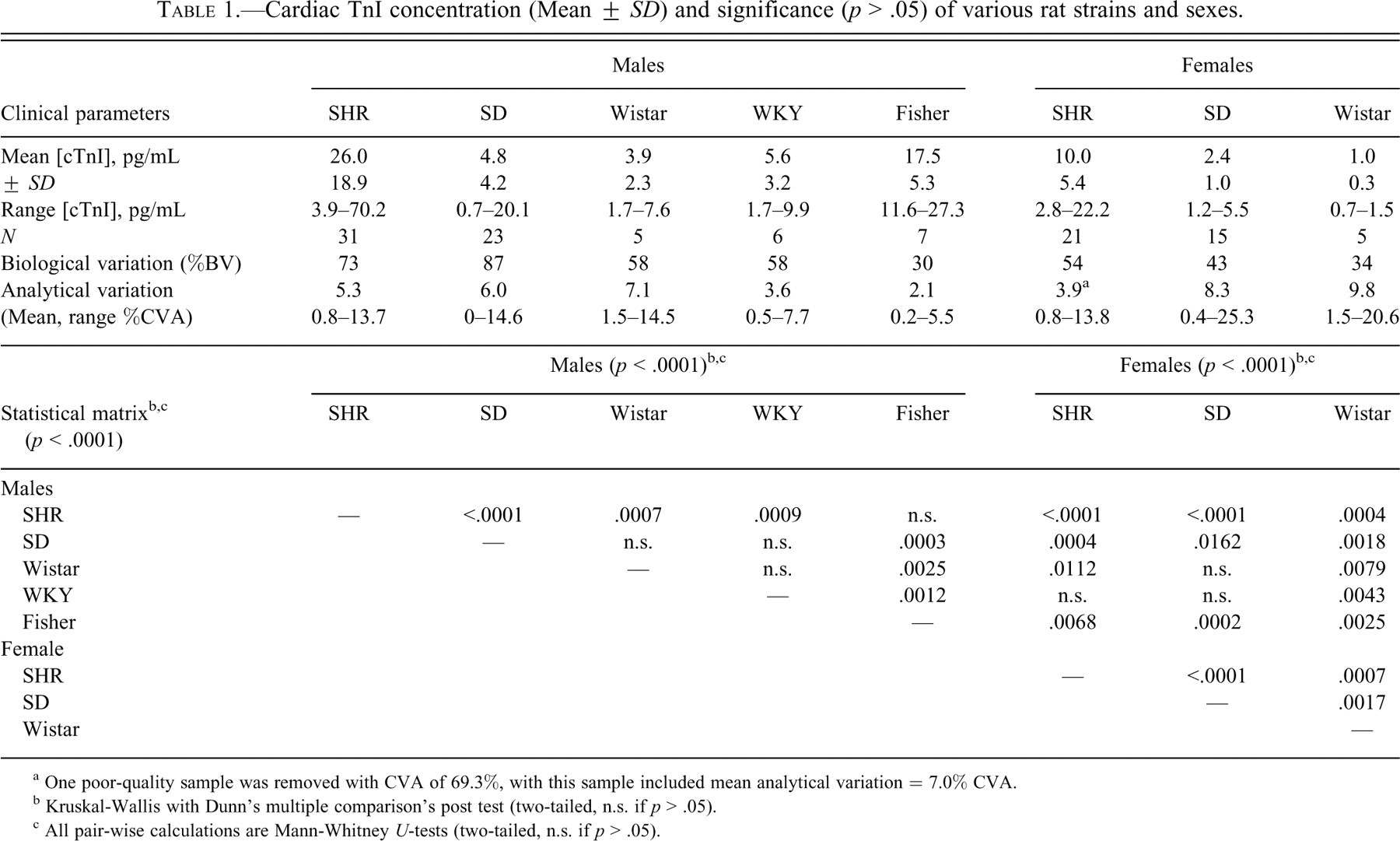
a One poor-quality sample was removed with CVA of 69.3%, with this sample included mean analytical variation = 7.0% CVA.
b Kruskal-Wallis with Dunn’s multiple comparison’s post test (two-tailed, n.s. if p > .05).
c All pair-wise calculations are Mann-Whitney U-tests (two-tailed, n.s. if p > .05).
Surgical Castration and Ovariectomization of Male and Female SHR
In a separate study, the baseline cTnI concentrations from groups of castrated male SHR (n = 4) and ovariectomized female SHR (n = 5) were compared with groups of normal male SHR (n = 5) and female SHR (n = 6). Surgical procedures were performed when the animals were approximately eight weeks of age. Dosed animals (twelve weeks of age at onset of dosing) received saline intravenously (IV) weekly for nine weeks.
Collection of Blood Samples
At the designated times (twenty-four hours or one week after vehicle administration) animals were anesthetized with isoflurane. The inferior vena cava was exposed via a midline abdominal incision. Terminal blood samples were collected via a 19-G needle-tipped Venocath catheter inserted into the vena cava. Blood samples were immediately centrifuged and the sera stored at –80°C until analyzed.
Collection of Hearts
At necropsy the entire heart was removed and fixed in 10% neutral buffered formalin. After fixation, the heart was embedded either in glycol methacrylate resin (sectioned at a thickness of 1 μm and stained with tolouidine blue) or paraffin (sectioned at 5 μm and stained with hematoxylin and eosin). Multiple sections of each heart, obtained from either longitudinal cutting or transversal cutting in different experiments, were used for histopathological evaluation. Prepared heart sections were examined by light microscopy.
Serum Cardiac Troponin I Measurement
Cardiac troponin I was measured with the ultrasensitive Erenna immunoassay system (Singulex Inc., Alameda, CA), which uses a microparticle immunoassay and single-molecule counting in a capillary flow system. The assay procedure has been described in detail elsewhere (Todd et al. 2007), and analyte stability in serum and EDTA-plasma has been demonstrated (Wu, Shea, et al. 2009). To summarize: 50 μl of serum or calibrator were combined with 150 μl of assay buffer containing biotinylated capture antibody-coated microparticles (MP). This mixture was incubated for sixty minutes, after which the MPs were magnetically separated and washed. The MPs were then incubated a second time with 20 μl of fluorescent dye-labeled detection antibody. Twenty μl of elution buffer was added after five additional magnetic separation washes. The eluted detection antibody was removed from the MPs by means of a 384 well filter plate, and the resulting eluate was measured in the Erenna instrument system. All samples were analyzed in duplicate.
Serum Cardiac Troponin T Measurement
Serum samples from 55 of the 127 rats were available to be analyzed for cTnT in addition to cTnI. The concentrations of cTnT were measured by the third-generation immunoassay (Elecsys, STAT; Roche Diagnostics) in the Clinical Chemistry Laboratory of Children’s Hospital Boston, Harvard Medical School (Boston, MA).
Statistical Methods
Outliers were identified as being greater than four standard deviations from the mean for each cohort included in the study (> 4 stdev) and were excluded from further statistical analysis. For each cohort, the mean, standard deviation, and range of values was determined. Biological variation (%BV) was calculated for each cohort independently as the percentage of the mean for each cohort divided by the standard deviation of measurements of all individuals within the corresponding cohort. Analytical variation (CVA) of the Erenna cTnI assay (Singulex) was determined by calculating the percentage of the mean of three replicates divided by the standard deviation between replicates for each sample tested. Because data were not always normally distributed and because sample sizes in some groups were small, nonparametric Kruskal-Wallace (with Dunn’s multiple comparison) and Mann-Whitney U-tests were used to evaluate significance of differences amongst various groups in rat serum concentrations of cTnI and cTnT. Alpha was set at .05, and all tests were two-tailed. The InStat and Prism (GraphPad Software, Inc., San Diego, CA) statistical software packages were used for all analyses.
Results
Cardiac Pathology
Light microscopic evaluation of 128 hearts from all groups was performed by a pathologist (J.Z.). Myocardial alterations were found in three of twenty-six hearts from male SD rats given distilled water orally for fourteen days. These rats had serum levels of cTnI that were significantly higher than other male SD rats. Myocardial inflammation was observed in the left ventricular endocardium and papillary muscle (Figure 1A ) and epicardium (Figure 1B) from one SD rat with a cTnI concentration of 202 pg/ml. Accumulation of edema fluid, numerous mononuclear cells, congested capillaries, and scattered fragments of myocytes were present in the affected inflammatory areas (Figures 1A and 1B). In another rat with an increased cTnI level (142 pg/ml), minimal myofibrillar loss was observed in the left ventricular myocardium (Figure 1C). These alterations were more prominent on 1-micron-thick sections of plastic-embedded tissue, alkaline toluidine blue stain than on H&E-stained sections. Minimal left ventricular cytoplasmic vacuolization was noted in the heart from a third SD rat with an increased concentration of cTnI level (93 pg/ml) (Figure 1D). In contrast, rats with lower baseline cTnI levels showed evenly stained cytoplasm with regular cross-striations in longitudinal cut-sections (Figure 1E) and no observable cytoplasmic vacuoles in transversal cut-sections (Figure 1F). Two of thirty-three male SHR had high serum levels of cTnI (102 and 143.6 pg/ml). Focal inflammation and fibrosis were observed in the left ventricular myocardium from one of these animals (figure not shown).
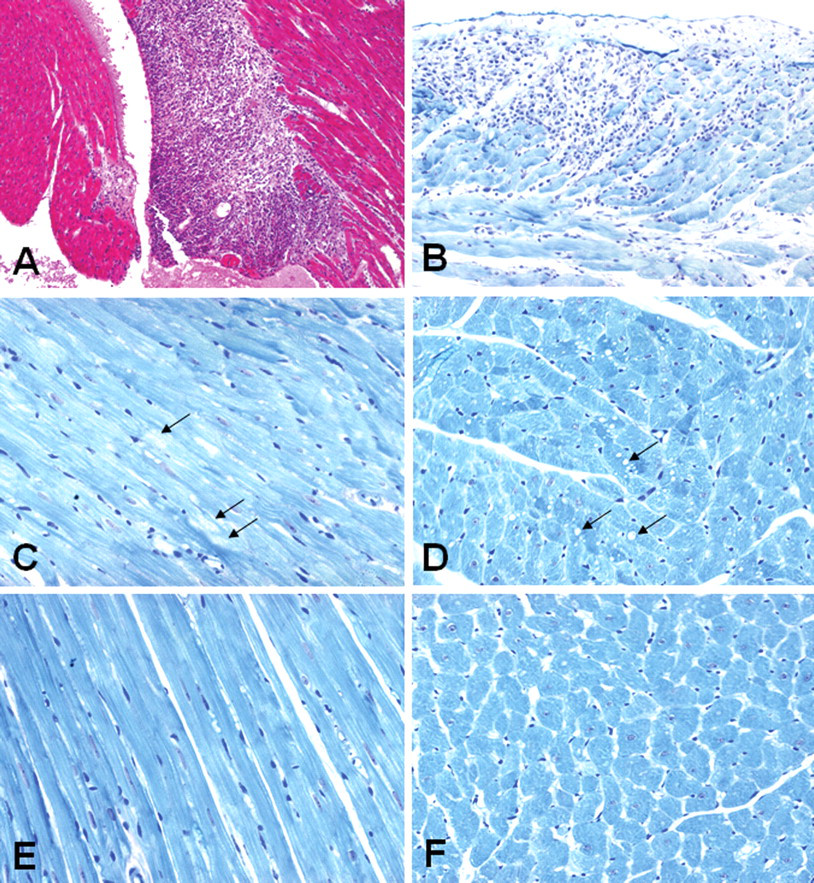
Light micrographs showing myocardial alterations in the hearts from Sprague-Dawley (SD) male rats given distilled water orally for fourteen days. (A) Focal inflammation is observed in the left ventricular endocardium and a papillary muscle from a rat with an elevated level of serum cTnI (202 pg/ml). H&E stain, X100. (B) Focal inflammation is noted in the left ventricular epicardium from a rat with an increased serum level of cTnI (202 pg/ml). (C) Minimal myofibrillar loss in the left ventricular myocardium (arrows) from a rat with an elevated concentration of cTnI (142 pg/ml). (D) Minimal cytoplasmic vacuolization in the left ventricular myocardium (arrows) from a rat with an increased serum concentration of cTnI (93 pg/ml). (E and F) Normal left ventricular myocardial morphology from a rat with a baseline (nonelevated) cTnI level. (B–F) One-micron-thick sections of plastic-embedded tissue, alkaline toluidine blue stain, X400.
Cardiac Troponin I Levels
Cardiac troponin I was measured by means of an ultrasensitive assay system in various strains of male and female rats, and results are summarized in Table 1. Cardiac troponin I was detected in all rats regardless of strain or sex, and the distribution (median ± interquartile range) of cTnI concentrations are shown for both male (Figure 2A ) and female (Figure 2B) rats, respectively. Baseline serum concentration of cTnI exceeded four standard deviations of the mean in two male SHR and three male SD rats, and evidence of cardiopathology is shown in Figure 1. Cardiac TnI values were excluded in cases where cTnI > 101.7 pg/ml for male SHR (two rats with 102 and 143.6 pg/mL excluded) and > 21.7 pg/mL in male SD (three rats with 93.4, 144, and 202.3 pg/mL) from all statistical baseline calculations (Table 1) and baseline graphical distributions (Figures 2–4).
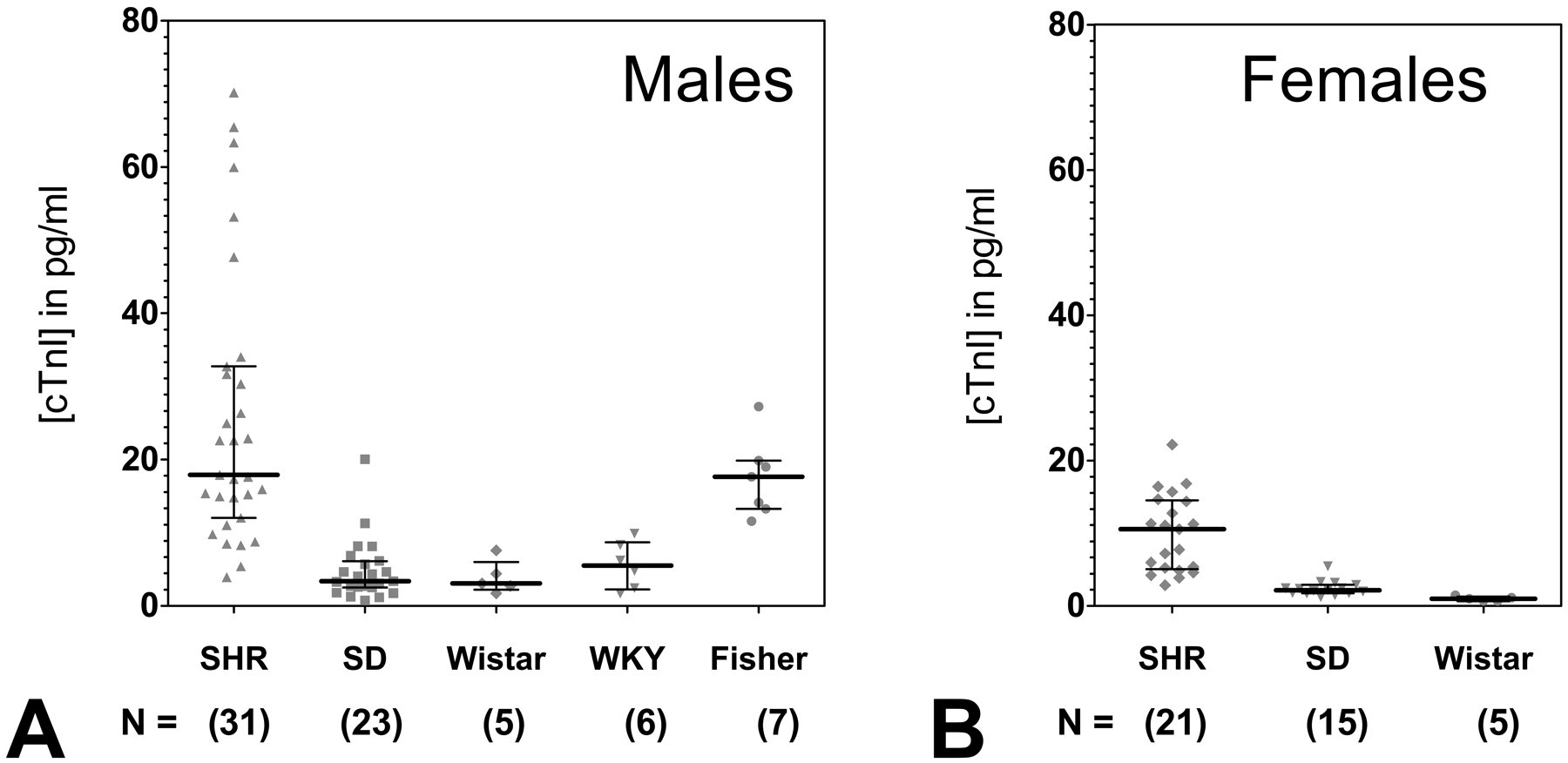
Distribution of cardiac troponin I concentrations detected by the ultrasensitive Erenna cTnI immunoassay in various strains of (A) male and (B) female control Sprague-Dawley (SD), Spontaneous Hypertensive (SHR), Wistar, Wistar-Kyoto (WKY), and Fisher rats. Lines indicate the median and interquartile range, with cTnI concentration in pg/mL. Each data point represents the mean of duplicate measurements from an individual rat. The number of rats per cohort is indicated.
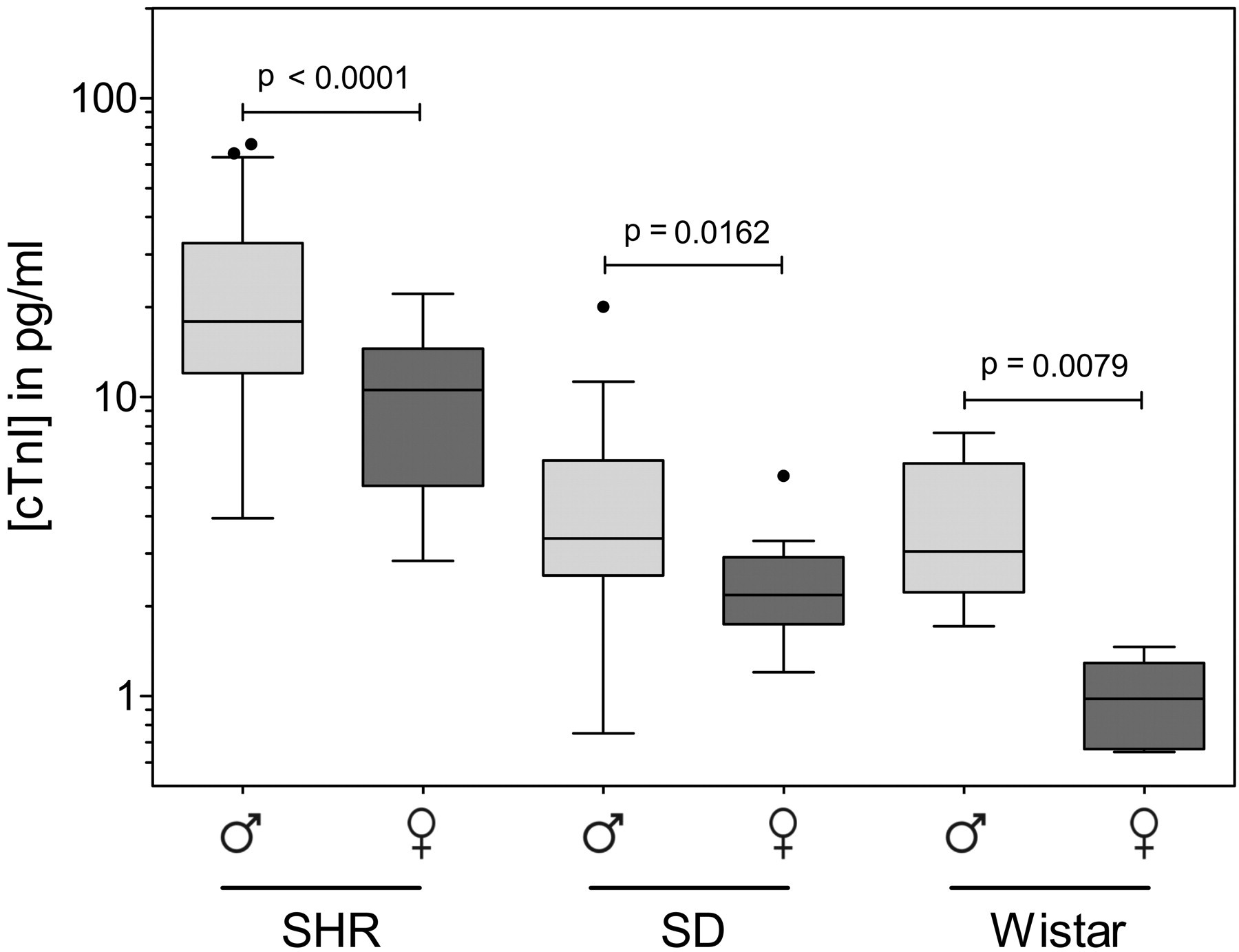
Gender comparison of serum cTnI concentrations in Sprague-Dawley (SD), Spontaneous Hypertensive (SHR), and Wistar rat strains. Concentrations of cTnI are shown for male (light gray) compared with female (dark gray) SHR, SD, and Wistar rat strains. Significance was determined using a Mann-Whitney t-test, with alpha set at p < .05. Results are shown as median and interquartile range, with cTnI concentration in pg/mL.
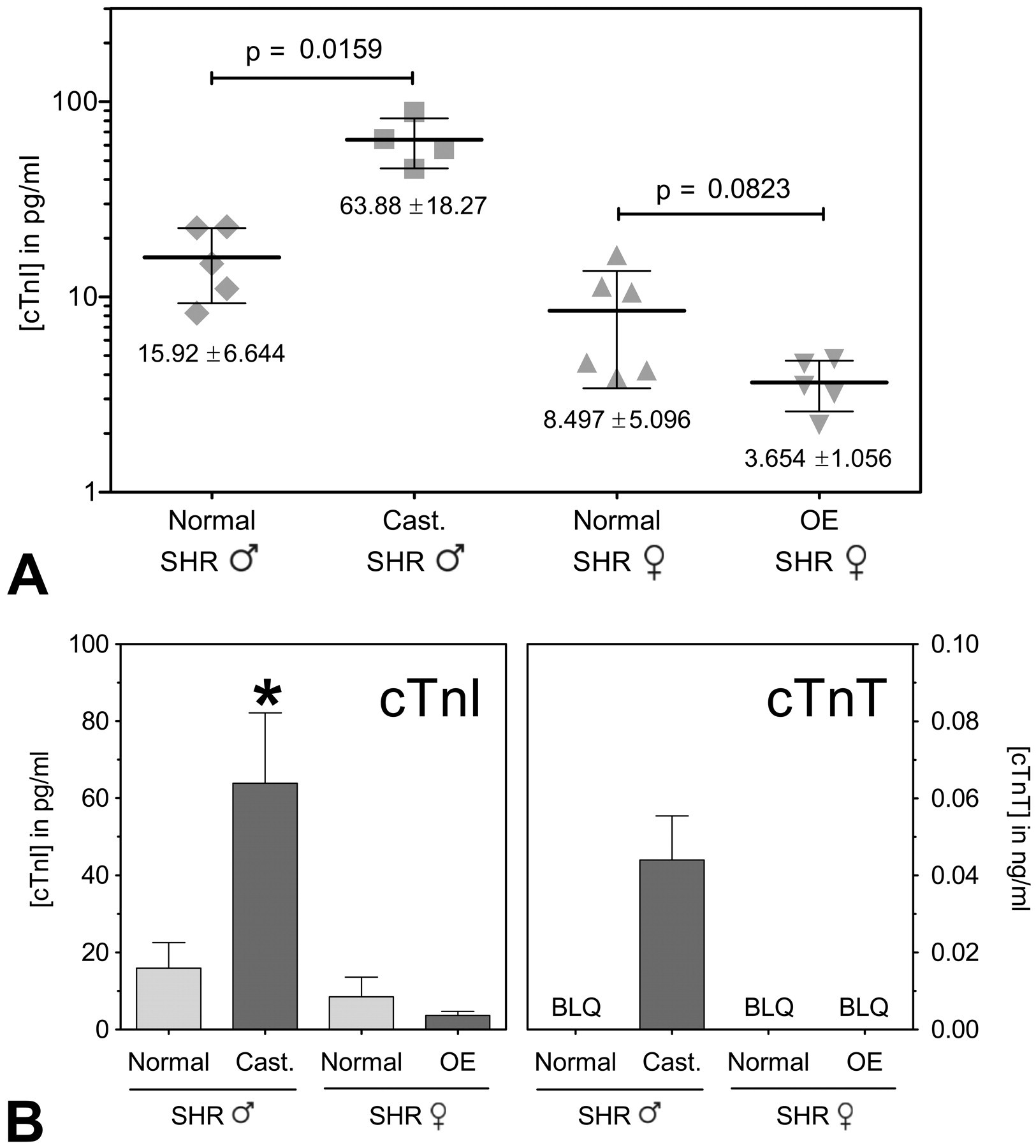
Effect of castration (Cast.) or ovariectomization (OE) on cTnI concentration in male and female Spontaneous Hypertensive rats (SHR). (A) Distribution of cTnI concentrations between normal and castrated male SHR and normal and ovariectomized female SHR. (B) Comparison of baseline results utilizing cTnI (Erenna immunoassay) and cTnT (Roche Elecsys Stat immunoassay) in normal and castrated male SHR and normal and ovariectomized female SHR. Lines in (A) and bars in (B) indicate mean ± standard deviation. BQL = below quantifiable limit. *Mean value significantly greater than that of normal male SHR, normal and ovariectomized female SHR (p < .05).
The highest strain-specific baseline concentrations of cTnI were found in male SHR (mean = 26.0 ± 18.9 pg/ml) and male Fisher rats (mean = 17.5 ± 5.3 pg/ml). Mean concentrations of cTnI detected in both male SHR and male Fisher rats were significantly higher than those found in the other strains of rats (all p < .05; Table 1, Figure 2A). Male SHR had more values of cTnI greater than 20 pg/ml than any other group of rats (Figure 2A). Male SHR also had the greatest variability in baseline serum cTnI concentrations, with values ranging from 3.9 to 70.2 pg/ml. The lowest concentrations of cTnI were detected in female Wistar rats (1.0 ± 0.3 pg/ml; Table 1, Figure 2B).
Biological variability was significantly higher than analytical variability in each group, and thus does not represent error due to the analytical method used (Table 1). The mean coefficient of analytical variation (CVA) for all cTnI analyses by the ultrasensitive assay system used in this study are shown in Table 1. Overall, the mean CVA was below 10% in all cases, and the actual CVA for individual specimens exceeded 10% in only 8.6% of the samples.
Significant differences between male and female rats are shown in Figure 2. In all strains tested (SHR, SD, and Wistar), male rats had significantly higher serum concentrations of cTnI than female rats of the same strain (all p < .05; Figure 3). The sex-related difference in cTnI concentration between male and female rats was most significant for the SHR (p < .0001) and less significant for Wistar (p < .01) and SD (p < .05) rats (Figure 3).
Cardiac Troponin T Levels
Serum concentrations of cTnT were measured in fifty-five rats (nineteen male SHR, twelve female SHR, eighteen male SD, and six female SD). Serum cTnT was detected in samples from seven of nineteen male SHR (including five castrated males) and two of eighteen male SD. The nine detectable concentrations varied between 0.01 and 0.18 ng/ml and were all from male and not female rats, regardless of strain being SHR or SD.
Effects of Noncardiac Surgery on Troponin Concentrations
Serum samples from four groups of five SHR each (male normal, male castrated, female normal, and female ovariectomized) were included in this study. The mean concentrations of cTnI and cTnT detected in this study are summarized in Figure 4. A significant increase in the mean concentrations of cTnI were found in the castrated male over that observed in the normal male SHR (63.88 ± 18.27 vs. 15.92 ± 6.64 ng/ml cTnI, p < .05). The mean cTnI concentrations were lower in ovariectomized SHR females compared with normal SHR females (3.654 ± 1.056 vs. 8.497 ± 5.096 pg/ml). This difference was not found to be statistically significant (p < .05). Hearts from all four groups of animals appeared normal by light microscopic evaluation.
Discussion
In the present study, an ultrasensitive assay (Erenna immunoassay system) was used to determine baseline concentrations of cTnI in several strains of control male and female rats. Baseline concentrations of cTnI were observed to vary from a low of 1.0 ± 0.3 (Wistar female) to a high of 26.0 ± 18.9 pg/ml (male SHR). The ability of this assay to detect low concentrations of serum cTnI in rats has previously been reported by Schultze et al. (2009, 2008). These investigators found mean cTnI concentrations of 9–20 pg/ml in male Fisher rats and 4.1 pg/ml in male SD rats. The mean baseline concentrations of cTnI observed for male Fisher rats (17.5 ± 5.3 pg/ml) and male SD rats (4.8 ± 4.2 pg/ml) in the present study are consistent with the range of the cTnI values reported by Schultze et al., indicating that an ultrasensitive assay is capable of quantifying serum concentrations of cTnI in healthy, normal rats that were previously undetectable using earlier generation commercial assays.
A significant observation of the present study is that baseline cTnI was quantified by an ultrasensitive assay in the serum of all rats that were tested with a high degree of precision. Thus, the important question of whether certain factors such as strain and gender might affect the baseline blood concentration of cTnI was able to be evaluated in the present study. This important characteristic of the Erenna assay has been examined in human patients and animals (Schultze et al. 2009, 2008; Todd et al. 2007; Wu et al. 2006; Wu, Lu, et al. 2009). These studies found that the assay has a limit of detection (LoD) of 0.2 pg/ml and a lower limit of quantification (LLoQ) at a ≤ 10% CV of 0.8 pg/ml. In the present study, none of the observed values were below the LoD. Mean analytical variation (Mean CVA) was below 10% for all cohorts tested. The CVA exceeded 10% in only 8.6% of the samples, suggesting that the majority of assay values quantified by this method represent an accurate sampling of baseline cTnI concentrations in the various types of rats.
In the present study, the ultrasensitive assay detected the presence of cTnI in all analyzed rat serum samples. The concentrations of cTnI reported by Sabatine et al. (2009) in human subjects were in the range found in four of the eight groups of rats evaluated in the present study. Low concentrations of baseline cTnI have also been noted in blood samples taken from a small number of dogs and monkeys (Schultze et al. 2009, 2008). These observations tend to suggest that low concentrations of cTnI are normally present in the blood of all species of animals, including humans, and that an ultrasensitive assay such as Erenna can detect these low baseline concentrations of the protein. The significance of detectable low blood cTnI concentrations in normal healthy human and animal populations by means of the recently developed ultrasensitive assays has not been completely determined. Clerico et al. (2008) suggest that cardiac troponins in small amounts might be released from myocytes present in healthy hearts as part of a process associated with physiological remodeling.
Until recently, the presence of cardiac troponins in the blood was considered an indication of myocardial injury such as necrosis (Morrow et al. 2007; Prontera et al. 2007). However, there are experimental studies that show that concentrations of cardiac troponins can increase in the absence of apparent alterations in myocyte morphology (Chen et al. 2000). In the present study, five untreated rats (two SHR and three SD) had elevated cTnI concentrations, and correlating evidence of myocardial alterations detected by light microscopic evaluation were observed in four of five of these animals (Figure 1). Thus, by monitoring cTnI, it was possible to identify animals with spontaneously occurring heart lesions in baseline, control animals.
Significant strain differences in baseline cTnI concentrations were noted among the four types of rats examined (SHR, SD, Wistar, and Fisher). The highest baseline concentrations of cTnI were found in the male SHR (mean of 26.0 ± 18.9 pg/ml). These animals have been bred to develop spontaneous hypertension. Systolic blood pressure begins to increase around five to six weeks of age and steadily increases to a maximum systolic level (180–200 mm Hg) at twelve to sixteen weeks of age (Okamoto and Aoki 1963). As a result of elevated blood pressure, SHR are prone to develop hypertension-induced end-organ alterations such as myocardial hypertrophy and late cardiac failure (Pinto, Paul, and Ganten 1998). The SHR assayed in the present study were twelve weeks of age at study initiation in order to have reached the peak level of hypertension, as recommended for this strain (Charles River Labs, data not shown). It should be noted that Zhu et al. (2009) reported that cardiac hypertrophy was observed in SHR at four weeks of age prior to significant increase in systolic pressure; however, the hearts from the SHR in this study were not evaluated to determine the extent of myocyte hypertrophy. Consequently, it is not known whether the higher serum baseline cTnI concentrations observed in the SHR indicates that the protein leaks from hypertrophied cardiac cells at a greater rate than from normal myocytes. A study that includes SHR over a wider range of ages might help answer this question.
Among the other strains, male Fisher rats had significantly higher cTnI concentrations than did the male SD, Wistar, and WKY rats. Control male Fisher rats, in an earlier study (Schultze et al. 2008), were found to have cTnI concentrations of 9–20 pg/ml. This concentration is in the range of the mean serum cTnI concentration detected in the present study (17.5 ± 5.3 pg/ml). It is interesting to note that the lowest baseline cTnI concentrations detected in the present study were observed in Wistar rats (Table 1, Figure 2), the strain from which the SHR were originally derived. The results of the present study indicate that rat strain is an important determinant of baseline cTnI concentrations.
Gender was also found to influence the baseline concentrations of cTnI (Figure 3). As with the male SHR rats, the baseline cTnI concentrations in the female SHR were significantly higher than those detected in either the SD or Wistar rats. However, when cTnI concentrations in all three strains of male and female rats (SHR, SD, and Wistar) were compared, mean serum concentrations were higher in male animals. The difference reached a p < .0001 level of significance in SHR and p < .05 in SD and Wistar rats (Figure 3). In clinical studies a similar significant difference has been noted between the baseline cTnI concentrations in men and women. It should be noted that the assay utilized by Clerico et al. (2008) was not able to detect the presence of cTnI in 24% of the samples analyzed. Most of these undetectable cTnI samples were from female subjects. Eggers et al. (2009) have also reported higher cTnI concentrations in men than in women. Likewise, cardiac troponin T reference concentrations, detected by a highly sensitive fourth-generation cTnT assay, are also reported higher in men than women (Mingels et al. 2009).
Subtle gender-related myocardial differences have been cited as possible factors that could possibly influence blood concentrations of cTnI. For example, in humans, the size of the male heart is reported to be larger than that of the female heart (Olivetti et al. 1995; Salton et al. 2002). In the present study, male rats of the same age and strain weighed approximately 30% more than female rats from a similar strain. Although heart weight was not determined, it is possible that the ultimate size of the heart could contribute to the observed gender difference in cTnI concentration. Other factors such as the quantity of cardiac troponin proteins (Schwarzenberger et al. 2003) and the concentration of myocyte apoptotic activity (Guerra et al. 1999; Olivetti et al. 1995) have been reported to differ in male and female hearts. It is also possible that male and female rats do not clear cTnI at the same rate. At present it is not known which of these or other factors are responsible for the observed gender-related differences in baseline cTnI concentrations.
Detection of increasing cardiac troponin concentrations is taken as an indication of myocyte injury. In the present study a relatively simple noncardiac surgical procedure was performed. Groups of female SHR were ovariectomized and male SHR castrated when the animals were eight weeks old. At approximately twenty weeks of age, cTnI concentrations were measured in these animals and compared with groups of normal male and female SHR of the same age. In this instance, the mean serum cTnI concentration was found to be four to five times higher in the castrated males compared to normal male and female SHR (p < .05; Figure 4). The group of castrated male SHR was the only group of the four in which a third-generation immunoassay was able to detect the presence of cTnT, and thus statistical significance was not able to be determined. This illustrates the importance of choosing an appropriate method for obtaining quantifiable data in order to yield significant results, and further demonstrates the superiority of ultrasensitive cTnI over cTnT for this purpose.
The changes in cTnI concentrations detected in castrated SHR occurred in the absence of any apparent alterations in myocyte morphology. No sham operated control animals were included in the present study, so it is not possible to rule out the role of surgery per se in the observed changes in cTnI concentrations. However, surgery may not be a contributing factor since cTnI concentrations did not change in the same direction (increased in castrated male SHR and decreased in ovariectomized female SHR). Both estrogen and androgen receptors have been identified in rodent hearts (Lizotte et al. 2009). Studies in rats have sought to explore the influence of gonadotropic hormones on cardiovascular toxicity. Treatment with testosterone and/or ovariectomy has been found to increase doxorubicin-induced nephrotoxicity (Sakemi et al. 1997). Ovariectomy with the resultant loss of ovarian hormones has been reported to alter markers of oxidative stress and enhance the cardiotoxic effects of doxorubicin (Munoz-Castaneda et al. 2006). Castration has been found to cause multiple changes in the myocardium. For example, surgical loss of testosterone in the rat has led to decreases both in antioxidant status of the left ventricle (Klapcinska et al. 2008) and mRNA levels for calcium regulatory proteins (Golden, Marsh, and Jiang 2002; Jiang 2002) and alterations in myosin heavy chain composition (Golden et al. 2004). Testosterone withdrawal has been associated with atrophy of androgen sensitive tissue (Boissonneault 2001). These observations tend to indicate that alterations in gonadotrophic hormone levels can influence cardiovascular activity. Whether any of these factors would help explain how the absence of estrogen or testosterone induces changes in baseline concentrations of cTnI as detected in the present study remains to be determined.
The present study had several limitations that could be addressed in future studies. Clinically, baseline cardiac troponin concentrations have been found to vary with age (Clerico et al. 2008). The rats used in the present study were adults whose collective age range (including all tested strains) was limited to eight to twelve weeks prior to study initiation. Thus, it is not known how the baseline concentrations detected compare with the cTnI concentrations in very young or old animals, and studies into the effect of age on various strains of rats may be warranted. The number of animals evaluated in certain groups was small. In this instance, an increase in the number of rats evaluated would provide a more accurate assessment of both the baseline cTnI levels and the distribution of cTnI concentrations in normal rats, and similar cross-strain and cross-gender validation studies should be conducted in the future using larger numbers of animals for validation. The number of rat strains evaluated in the present study was limited. Since other types of rats are utilized for research purposes, baseline cTnI concentrations should also determined in additional appropriate strains. Schultze et al. (2009, 2008) have reported some preliminary baseline cTnI findings in the dog and monkey. Additional studies to define baseline cardiac troponin concentrations in these and other species utilized for experimental or veterinary purposes should be undertaken.
In summary, measuring changes in blood concentrations of cTnI with ultrasensitive immunoassays with improved analytical sensitivity has proved to be a reliable means for identifying a variety of myocardial alterations in humans and animals (Apple et al. 2008; Herman and Ferrans 2001; O’Brien 2008). The enhanced analytical sensitivity of ultrasensitive cardiac troponin assays has already begun to have an impact on the clinical ability to detect various types of subtle cardiac injury (Sabatine et al. 2009; Wu, Lu, et al. 2009). The applicability of the ultrasensitive cardiac troponin assays in animal studies has only recently begun to be addressed. The studies of Schultze et al. (2009, 2008) demonstrated that an ultrasensitive assay was able to detect low concentrations of cTnI in a variety of animals and that increases in baseline concentrations occur as a result of drug-induced myocardial injury.
In conclusion, the present study demonstrates that significant variability exists in ultrasensitive cTnI baseline measurements of control animals between strains, sexes, and surgical alterations (i.e., castration or ovariectomization) that are common to control animals utilized in cardiotoxicological studies. We show that the sensitivity of the Erenna assay enabled cTnI to be quantified in the blood of all rats tested and that baseline concentrations in these animals varied significantly between strains, being highest in SHR and lowest in Wistar. Gender influenced baseline cTnI concentrations, as males had significantly higher concentrations than did female rats of the same strain. Unexpectedly, baseline cTnI concentrations were also found to be affected by noncardiac surgery, in this case castration and ovariectomy, which are common laboratory procedures not previously thought to contribute to baseline cTnI measurements for control animal cohorts. Thus, when using ultrasensitive cTnI as a biomarker to assess cardiotoxicity, researchers need to take into account the exact nature of baseline cTnI expected for the control animals being utilized in each study with regards to the factors identified in this study. Additional factors beyond those described in this study remain to be tested, and our results demonstrate a continuing need to expand the base of control information in order to realize the maximal potential of monitoring small changes in cardiac troponin as part of preclinical cardiotoxicity testing programs.
