Abstract
Two-year toxicity studies were conducted on the widely used herbal products, goldenseal and milk thistle, in male and female F344/N rats and B6C3F1 mice. With goldenseal root powder, the primary finding was an increase in liver tumors in rats and mice, and with milk thistle extract, a decrease in spontaneous background tumors including mammary gland tumors in female rats and liver tumors in male mice. Increased tumorigenicity in rodents exposed to goldenseal root powder may be due in part to the topoisomerase inhibition properties of berberine, a major alkaloid constituent in goldenseal, or its metabolite, berberrubine. In the clinic, use of topoisomerase-inhibiting agents has been associated with secondary tumor formation and inhibition in DNA repair processes. In contrast, the radical-scavenging and antioxidant properties of silibinin and other flavonolignans in milk thistle extract may have contributed to the decrease in background tumors in rodents in the present studies. The fate of the active constituents of goldenseal and milk thistle is similar in humans and rodents; therefore, the modes of action may translate across species. Further studies are needed to extrapolate the findings to humans.
Introduction
The Dietary Supplement Health and Education Act (DSHEA) of 1994 places dietary supplements, including herbs or other botanicals, in a special category under the general umbrella of “foods” (U.S. Food and Drug Administration 1994). More than half the people in the United States use herbal products, including goldenseal and milk thistle (Barnes, Bloom, and Nahin 2008; Cohen 2009), and up to 80% of the world population may use herbal products to treat or prevent disease (World Health Organization 2010). While herbal products are reported to have beneficial effects, none have been approved as drugs by the U.S. Food and Drug Administration. The National Institutes of Health (NIH) is currently supporting a number of clinical trials to obtain more information on clinical efficacy of these herbal products (U.S. National Institutes of Health 2009).
Goldenseal (Hydrastis canadensis), a plant native to North America, is a commonly used herbal product. The Cherokees used this plant to treat wounds, ulcers, and digestive disorders as well as skin and eye ailments and a wide variety of other ailments, and they passed this knowledge on to the early European settlers in North America (Hamon 1990). Alkaloids, specifically hydrastine and berberine, are reported to be the active components of goldenseal (Abourashed and Khan 2001). The concentration of berberine was shown to be higher than that of hydrastine in 10 of 11 commercial goldenseal products (neither alkaloid was detected in a 12th product), and berberine was the major alkaloid detected in the goldenseal root powder used in the present study (Table 1 ). Berberine has apparent antimicrobial activity in humans and has demonstrated anti-tumor and anti-inflammatory activity, neuroprotective and immunomodulary properties, and the ability to decrease plasma cholesterol in animals and/or in vitro models (Abidi et al. 2006; Grycova, Dostal, and Marek 2007; Kulkarni and Dhir 2009). The mechanism of actions for berberine, its metabolites, and/or other alkaloids in goldenseal may involve inhibition of enzymes, including CYPs and topoisomerases, and/or effects on cell signaling, receptors, and transporters (Abidi et al. 2006; Etheridge et al. 2007; Kulkarni and Dhir 2009; Makhey et al. 1994). The metabolism of berberine is similar in rodents and humans (Figure 1 ) (Pan et al. 2002; Qiu et al. 2009; Zuo et al. 2006). The major berberine metabolites shown in Figure 1 are generally excreted as sulfate and/or glucuronide conjugates in urine.
Composition content of goldenseal and milk thistle.
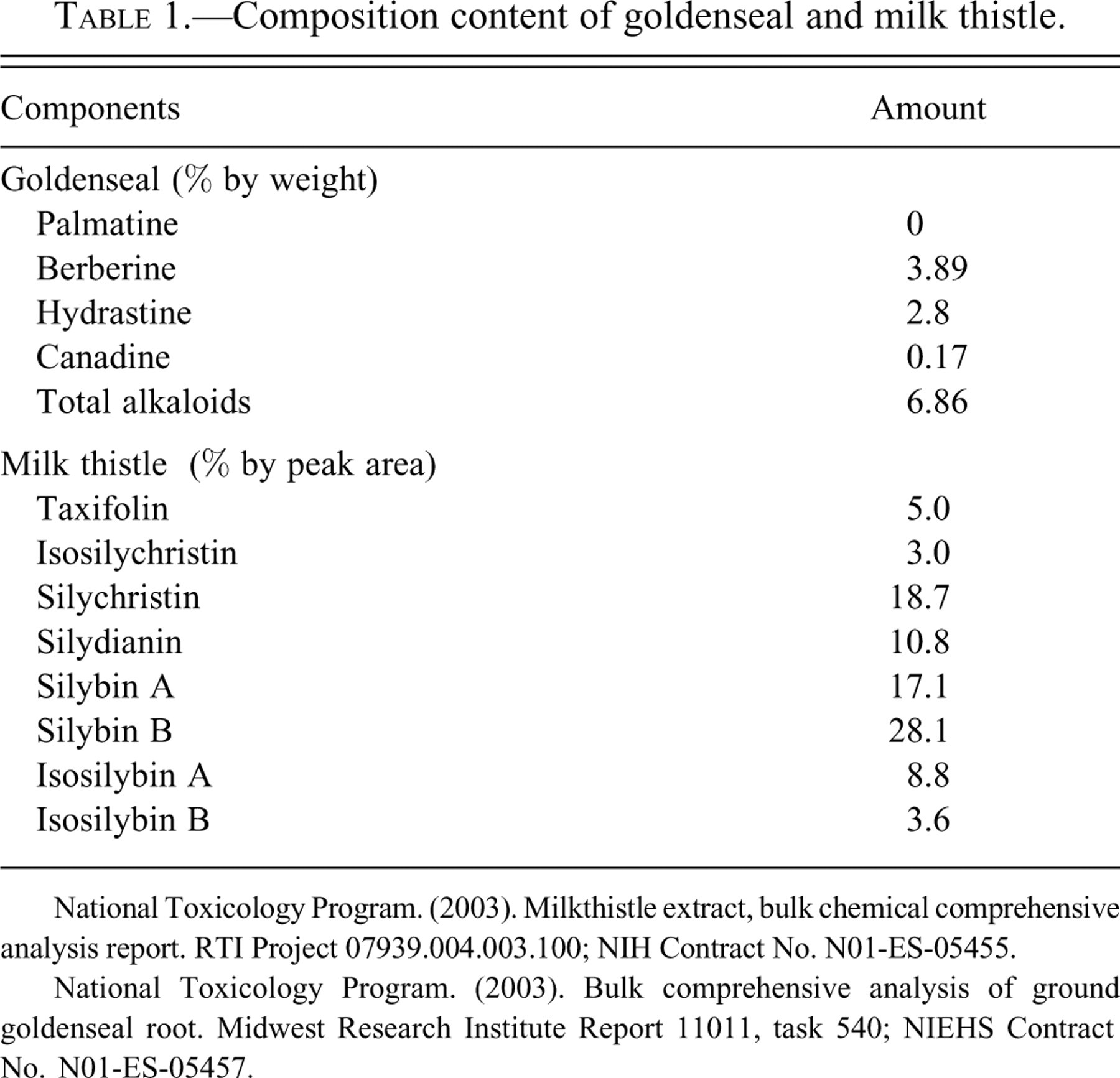
National Toxicology Program. (2003). Milkthistle extract, bulk chemical comprehensive analysis report. RTI Project 07939.004.003.100; NIH Contract No. N01-ES-05455.
National Toxicology Program. (2003). Bulk comprehensive analysis of ground goldenseal root. Midwest Research Institute Report 11011, task 540; NIEHS Contract No. N01-ES-05457.
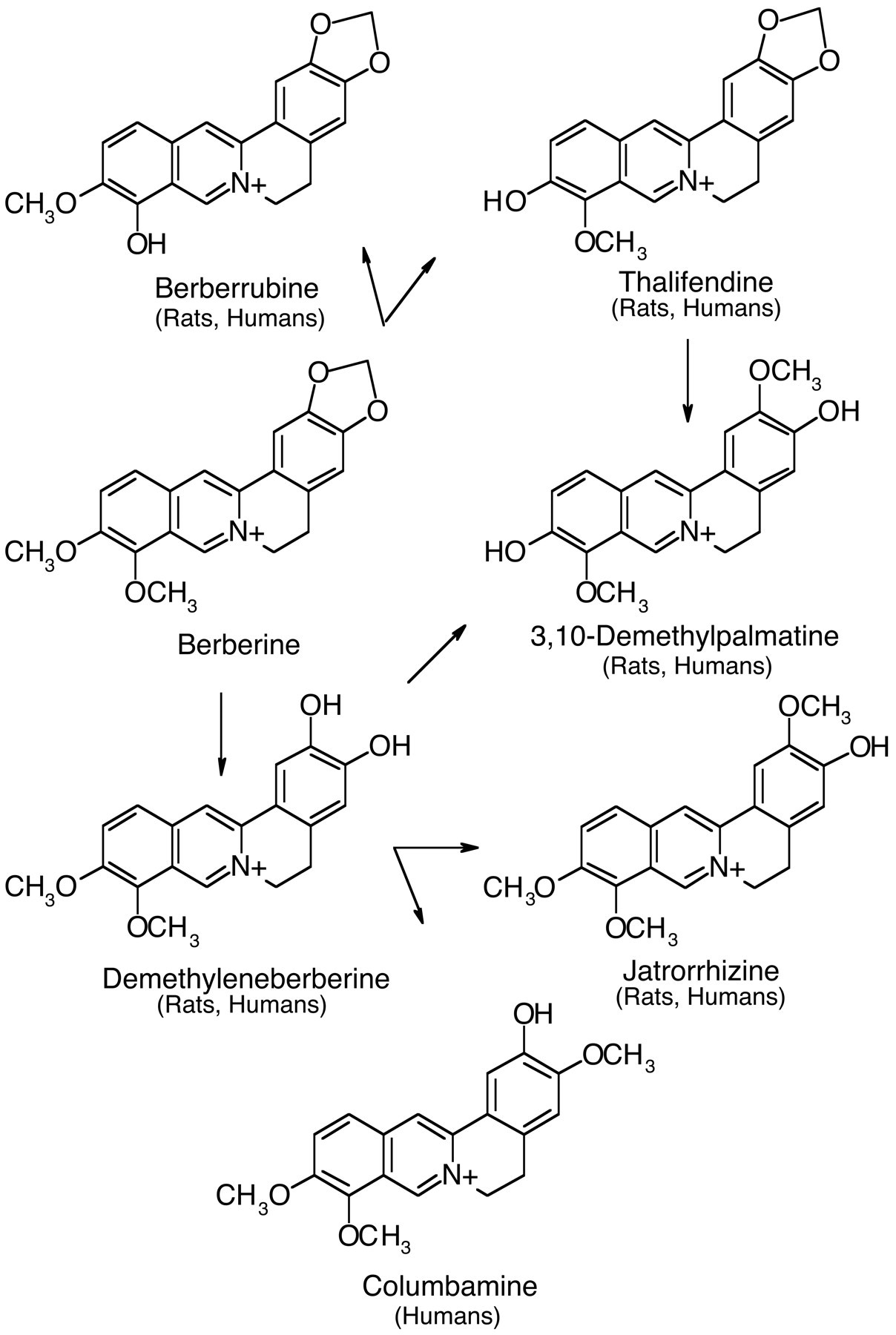
Berberine metabolites.
Milk thistle, Silybum marianum, a member of the Aster family, is a tall edible plant used in herbal medicine treatment of liver disease of alcoholic, viral, toxic, cholestatic, and primary malignant etiologies (Greenlee et al. 2007). The fruits of this plant contain the relatively water-insoluble flavonolignans silybin (A and B), isosilybin (A and B), silydianin, silychristin, and isosilychristin, as well as the flavonoid, taxifolin (Figure 2 ) (Davis-Searles et al. 2005; Kim et al. 2003; Kroll, Shaw, and Oberlies 2007). These chemicals form a complex known as silymarin, comprising up to 80% of milk thistle extract (Kroll, Shaw, and Oberlies 2007). Silybins A and B are the most abundant flavonolignans in silymarin and together comprise the fraction of silymarin known as silibinin (Davis-Searles et al. 2005; Kim et al. 2003; Kroll, Shaw, and Oberlies 2007). The composition of the milk thistle extract used in the present study is shown in Table 1. Components of milk thistle, silibinin and silymarin, possess significant antiproliferative activity in cultured cancer cells (Davis-Searles et al. 2005). Milk thistle extracts have antioxidant and radical-scavenging activities, affect cellular glutathione content and membrane stability, promote liver regeneration, and inhibit myofibroblast formation (Fraschini, Demartini, and Esposti 2002). Silybins A and B and isosilybin B (most potent on a molar basis) inhibited growth, suppressed prostate-specific antigen secretion, and/or suppressed activity of the DNA topoisomerase IIα gene promoter in specific human prostate cell lines (Davis-Searles et al. 2005). Milk thistle extract inhibited CYP3A4 and CYP2C8 activity in human hepatic microsomes, but did not affect human P-glycoprotein expressed in baculovirus (Etheridge et al. 2007). The flavonolignans detected in plasma of humans and rodents exposed to milk thistle extract are primarily in the form of glucuronide and/or sulfate conjugates (Morazzoni et al. 1993; Wen et al. 2008).
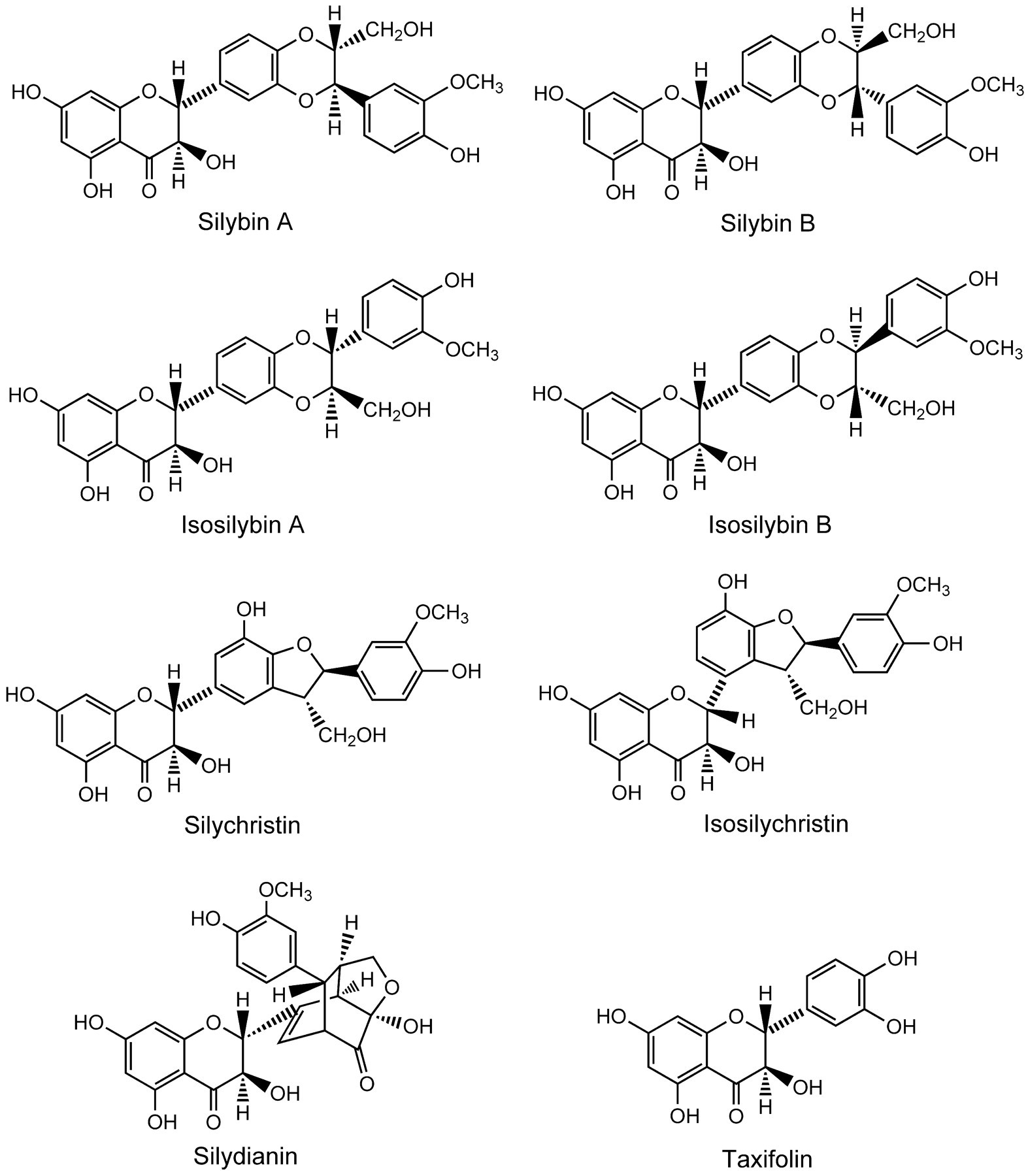
Milk thistle components.
While goldenseal and milk thistle have been used as herbal medicines to treat a variety of ailments, there are few toxicity or carcinogenesis studies reported in the literature. Therefore, the present studies were performed to provide more information on the toxicity and carcinogenicity of these widely used herbal medicines.
Materials and Methods
Herbal Products
Goldenseal root was purchased from Strategic Sourcing, Inc. (Reading, PA) (lot HYCA 10/7-10.28.01-C). The major alkaloid content for goldenseal used in the two-year study was 3.89% berberine, 2.8% hydrastine, and 0.17% canadine (Table 1). No alkaloids characteristic of common adulterants were found in either lot of the goldenseal root powder.
Milk thistle extract (alcohol/water extract) was purchased from Indena USA, Inc. (Seattle, WA) (lot 27691/N). The major components for milk thistle were silybins A and B, isosilybins A and B, silychristin, isosilychristin, silydianin, and taxifolin (Table 1). Based on chromatographic comparison to known reference standards, these compounds constituted approximately 65% by weight of the milk thistle extract used in these studies. Silybins A and B accounted for approximately 33% by weight of the milk thistle extract used these studies.
Goldenseal or milk thistle were mixed with NTP 2000 diet (Zeigler Brothers, Inc., Gardners, PA) using a Patterson Kelley V-blender with an intensifier bar.
Animals and Exposures
Male and female F344/N rats and B6C3F1 mice were obtained from Taconic Laboratory (Germantown, NY). At the start of the study the animals were five to six weeks of age. The animals were housed by species and sex, two to three male rats per cage, five female rats per cage, one male mouse per cage, and five female mice per cage. Tap water and NTP-2000 diet (Zeigler Brothers, Inc. Gardners, PA) were made available ad libitum. The care of animals in this study was according to NIH procedures as described in the “The U.S. Public Health Service Policy on Humane Care and Use of Laboratory Animals” (available from the Office of Laboratory Animal Welfare, National Institutes of Health, Department of Health and Human Services, RKLI, Suite 360, MSC 7982, 6705 Rockledge Drive, Bethesda, MD 20892-7982 or online at http://grants.nih.gov/grants/olaw/olaw.htm#pol).
For the goldenseal study, the herb was administered in the feed to male and female F344/N rats and B6C3F1 mice at 0, 3,000, 9,000, or 25,000 ppm for up to two years. For the milk thistle studies, the herb was administered in the feed to male and female F344/N rats and B6C3F1 mice at 0, 12,500, 25,000, or 50,000 ppm for up to two years. There were 50 animals/sex/species/per exposure group.
Pathology
Complete necropsies were performed on all animals after sacrifice when moribund or at the end of the two-year exposure period. At necropsy, all organs and tissues were examined for grossly visible lesions. Tissues were preserved in 10% neutral buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin. The following tissues were examined microscopically from male and/or female animals: gross lesions and tissue masses, adrenal gland, bone with marrow, brain, clitoral gland, esophagus, heart, large intestine (cecum, colon, rectum), small intestine (duodenum, jejunum, ileum), kidney, liver, lung, lymph nodes (mandibular and mesenteric), mammary gland (except male mice), nose, ovary, pancreas, pancreatic islets, parathyroid gland, pituitary gland, preputial gland, prostate gland, salivary gland, skin, spleen, stomach (forestomach and glandular), testis with epididymis and seminal vesicle, thymus, thyroid gland, trachea, urinary bladder, and uterus. Following completion of the studies, the accuracy of the histopathologic diagnosis was determined by microscopic reviews of neoplasms and target organs by quality assessment from a pathology working group (Boorman and Eustis 1986; Hardisty and Boorman 1986).
Statistical Analysis
The poly-3 test, which takes survival differences into account, was used to assess neoplasm and nonneoplastic lesion incidence (Bailer and Portier 1988; Piegorsch and Bailer 1997; Portier and Bailer 1989). When applied to all exposure groups, this test evaluates the significance of a dose-related trend in lesions; when applied to the control group and one exposure group, the test evaluates the significance of the pairwise difference of lesion incidence in the exposed group compared to the control group.
Tarone’s life table test (Tarone 1975) and Cox’s method (Cox 1972) were used to test survival for, respectively, dose-related trends and pairwise differences from the control group.
Results
Goldenseal
Survival and Body Weights
Feed consumption was similar among control and treated groups and was used to estimate compound consumption (Table 2 ). Final survival of treated groups of male and female rats and mice was similar to controls, with the exception of 9,000 ppm female rats where final survival was somewhat reduced. Final mean body weights of treated male rats and treated male and female mice were within ±5% of controls. Mean body weights of 9,000 and 25,000 female rats were 95% and 83% of controls, respectively (Table 3 ).
Estimated compound consumption—goldenseal and milk thistle consumption in rats and mice, two-year study.
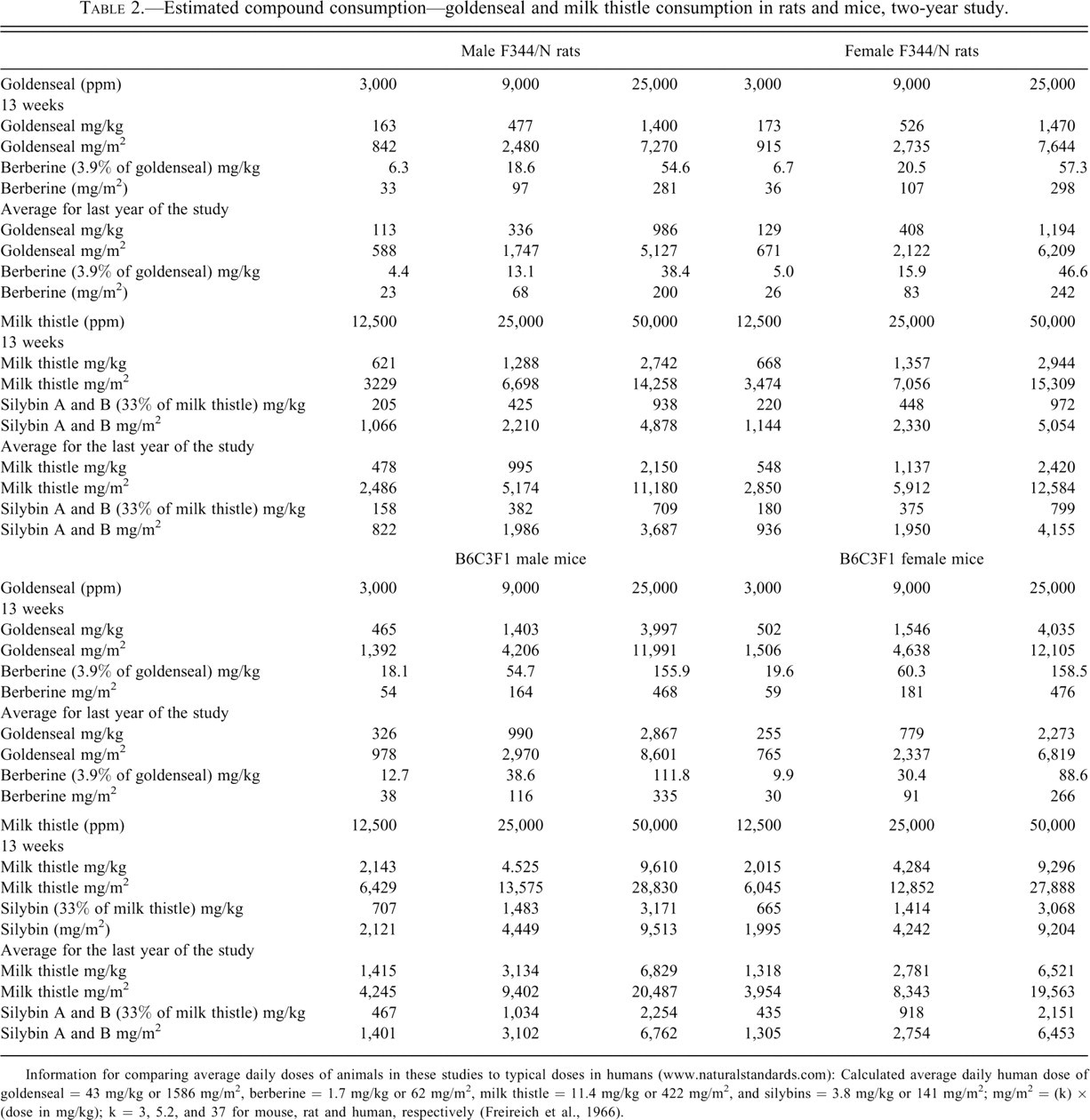
Information for comparing average daily doses of animals in these studies to typical doses in humans (www.naturalstandards.com): Calculated average daily human dose of goldenseal = 43 mg/kg or 1586 mg/m2, berberine = 1.7 mg/kg or 62 mg/m2, milk thistle = 11.4 mg/kg or 422 mg/m2, and silybins = 3.8 mg/kg or 141 mg/m2; mg/m2 = (k) × (dose in mg/kg); k = 3, 5.2, and 37 for mouse, rat and human, respectively (Freireich et al., 1966).
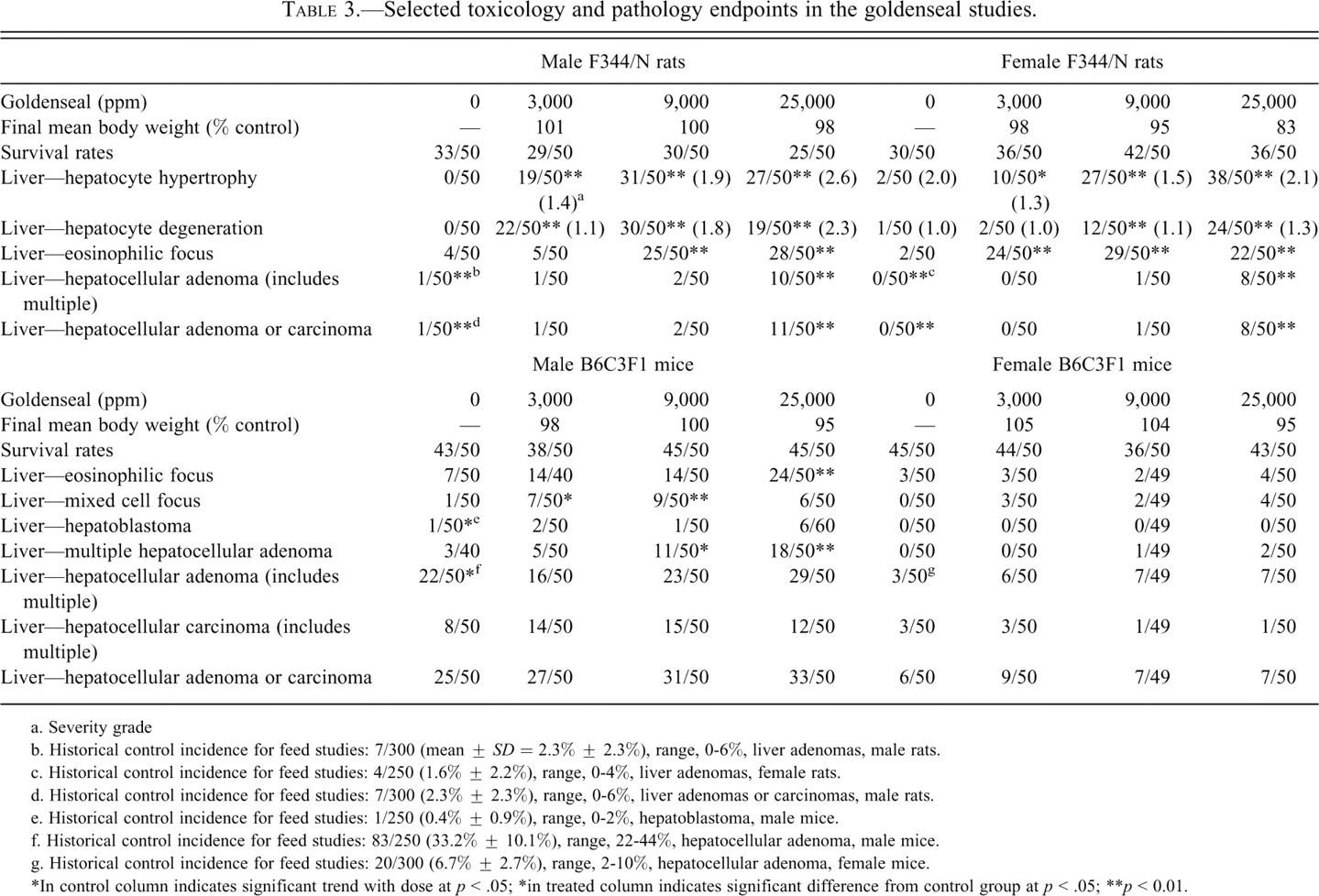
Selected toxicology and pathology endpoints in the goldenseal studies.
a. Severity grade
b. Historical control incidence for feed studies: 7/300 (mean ± SD = 2.3% ± 2.3%), range, 0-6%, liver adenomas, male rats.
c. Historical control incidence for feed studies: 4/250 (1.6% ± 2.2%), range, 0-4%, liver adenomas, female rats.
d. Historical control incidence for feed studies: 7/300 (2.3% ± 2.3%), range, 0-6%, liver adenomas or carcinomas, male rats.
e. Historical control incidence for feed studies: 1/250 (0.4% ± 0.9%), range, 0-2%, hepatoblastoma, male mice.
f. Historical control incidence for feed studies: 83/250 (33.2% ± 10.1%), range, 22-44%, hepatocellular adenoma, male mice.
g. Historical control incidence for feed studies: 20/300 (6.7% ± 2.7%), range, 2-10%, hepatocellular adenoma, female mice.
*In control column indicates significant trend with dose at p < .05; *in treated column indicates significant difference from control group at p < .05
**p < 0.01.
Histopathology—Rats
The major target organ in the goldenseal study was the liver of rats and mice. In male rats there was a significant increase in the incidence of hepatocellular adenoma and hepatocellular adenoma or carcinoma (combined) at the high dose (p < .01, Table 3). The trend statistic and the pairwise statistics showed increase in the incidences of these tumors. Male rats have a low background rate of liver neoplasms in current controls (2%) and in historical feed controls (mean, 2.3%; range, 0-6%). Thus, the increased incidence of liver tumors in 25,000 ppm male rats was considered to be related to treatment. Furthermore, there was an earlier time to first appearance of hepatocellular adenomas in treated groups (study day 612) than in controls (study day 729).
In 25,000 ppm female rats, there was an increased incidence of hepatocellular adenoma and the incidences exceeded the rate in the current controls and historical controls (Table 3; mean, 1.6%; range, 0-4%). Thus, the occurrence of liver tumors in treated female rats was considered to be evidence of carcinogenic activity.
The hepatocellular adenomas were typically composed of solid sheets of large eosinophilic, often vacuolated cells with no normal hepatic lobular architecture. There was compression of the surrounding nonneoplastic liver parenchyma. Multiple hepatocellular adenomas were seen in two 25,000 ppm males. The hepatocellular carcinoma in one 25,000 ppm male was a large mass composed of large, pleomorphic, markedly vacuolated cells that formed trabeculae often more than five hepatocytes thick.
Increased incidences of nonneoplastic hepatic lesions in exposed groups of males and females included hepatocyte hypertrophy, hepatocyte degeneration, eosinophilic focus, and mixed cell focus. The incidences generally increased with increasing exposure concentration.
The incidences of eosinophilic focus were significantly increased in 9,000 and 25,000 ppm males and all exposed groups of females. Eosinophilic foci were discrete areas composed of enlarged hepatocytes containing homogenous or finely granular, eosinophilic cytoplasm. Hepatocytes within eosinophilic foci tended to be larger than surrounding hepatocytes, often resulting in some compression of adjacent parenchyma.
All exposed groups of male and female rats had significantly increased incidences of hepatocyte hypertrophy, and the severity increased with increasing exposure concentration. The incidences of hepatocyte degeneration were significantly increased in all exposed groups of males and in 9,000 and 25,000 ppm females, and the severity increased with increasing exposure concentration. Hepatocyte hypertrophy consisted of lobular areas of minimally to moderately enlarged hepatocytes with increased amounts of eosinophilic cytoplasm accompanied by various degrees of vacuolization. Centrilobular and midzonal regions of the liver were typically affected with relative sparing of the periportal areas. In more severely affected livers, however, the altered hepatocytes comprised virtually the entire liver parenchyma. Hepatocyte degeneration was characterized by large hepatocytes distended with either finely granular, pale, slightly eosinophilic cytoplasm or numerous small, discrete, clear vacuoles. These microvesicular cytoplasmic changes did not progress to larger clear vacuoles seen with fatty changes. The degeneration was accompanied by occasional individual cell necrosis.
The incidences of hematopoietic cell proliferation in the spleen were significantly increased in 3,000 and 25,000 ppm males (0 ppm, 2/49; 3,000 ppm, 8/50; 9,000 ppm, 4/49; 25,000 ppm, 11/50). Hematopoietic cell proliferation was characterized by an increase in small, deeply basophilic erythroid cells and less often by large immature myeloid cells within the red pulp.
The incidences of cardiomyopathy were significantly decreased in all exposed groups of males and in 25,000 ppm females (males: 0 ppm, 47/50; 3,000 ppm, 38/50; 9,000 ppm, 39/50; 25,000 ppm, 36/50, females: 24/50, 24/50, 21/50, 15/50). The severities of this lesion were generally similar across exposure groups. Cardiomyopathy consisted of several microscopic changes in localized, often multiple areas of the myocardium, including the loss of myocytes, coagulative necrosis and mineralization of small numbers of individual myocytes, limited inflammatory cell infiltrates composed of a few macrophages and lymphocytes with occasional neutrophils, and various degrees of fibrosis.
Other changes noted in rats included decreases in chronic lung inflammation, which were significantly decreased in 25,000 ppm males (0 ppm, 16/50; 3,000 ppm, 10/50; 9,000 ppm, 17/50; 25,000 ppm, 3/50), and the lungs of all exposed groups of males had significant decreases in the incidences of histiocytic cellular infiltrates (34/50, 24/50, 25/50, 24/50). Infiltrates in the alveolar, peribronchiolar, or peribronchial tissues of the lungs consisting of mixed cell types such as neutrophils, macrophages, and lymphocytes were termed chronic inflammation.
The incidences of chronic nasal inflammation were significantly decreased in all exposed groups of males (12/50, 4/50, 3/50, 3/50). When the nasal tissues had infiltrates composed of a variety of cell types, including neutrophils, macrophages, and lymphocytes, accompanied by some degree of fibrosis in the submucosal tissues, the lesion was termed chronic inflammation.
Goldenseal administration was associated with a significant decrease in the incidence of mammary gland fibroadenoma in all exposed groups of females (0 ppm, 30/50; 3,000 ppm, 20/50; 9,000 ppm, 11/50; 25,000 ppm, 17/50). The incidence of mammary gland hyperplasia was also reduced in the high dose females (0 ppm, 49/50; 3,000 ppm, 46/50; 9,000 ppm, 45/50; 25,000 ppm, 34/50; pairwise comparison with control group, p ≤ 0.01). Because mammary gland tumor rates in female rats are correlated with body weight and the mean body weights of high-dose female rats were reduced up to 20% that of controls, we analyzed the relationship between body weight and predicted mammary gland tumor rates using a logistic regression model developed with data from female NTP control F344/N rats (Haseman et al. 1997). The model predicts 18.8, 17.5, 15.4, and 13.0 mammary gland tumors in the four dose groups of 50 animals, respectively, based on body weight and assuming no effect of goldenseal, whereas 31, 22, 12, and 19 mammary gland tumors were observed in the four groups, respectively. It is difficult to draw a definitive conclusion about the effects of goldenseal on mammary gland tumor development in this study because mammary gland tumor rates in controls and the low-dose group were higher than predicted by the model. However, it should be noted that the model predicts a 31% reduction in mammary gland tumors in the high dose based on body weight effects alone (13 HD, 18.8, C), and the actual reduction was 39% (19 HD, 31 C), suggesting that body weight may be the primary factor in the reduction of the incidence of this tumor.
Histopathology—Mice
The incidences of hepatocellular adenoma occurred with a positive trend in males with a slight but not significant increase in the 25,000 ppm group (Table 3). The incidences of multiple hepatocellular adenoma were significantly increased in 9,000 and 25,000 ppm males. The incidences of hepatocellular carcinoma were increased, but not significantly, in all exposed groups of males. Small and not significant increases in the incidences of hepatocellular adenoma occurred in all exposed groups of females.
Many of the hepatocellular adenomas had the typical appearance of those reported in B6C3F1 mice. These were discrete masses with distinct borders that caused compression of the surrounding normal hepatic parenchyma, sometimes projecting above the surface of the liver. They were composed of mildly to moderately pleomorphic hepatocytes that were of normal size or slightly larger than normal. Neoplastic hepatocytes within the adenomas frequently resembled those seen in eosinophilic foci, but basophilic, clear, and vacuolated hepatocytes were also present. The normal lobular architecture was disrupted when plates of neoplastic hepatocytes intersected the surrounding normal hepatocytes at oblique or right angles, making the adenoma stand out distinctly from the rest of the liver. Also, the normal arrangement of central veins and portal triads was absent. Sometimes a few portal triads or bile ducts were found at the periphery of the lesion that appeared to be entrapped structures. Some of the adenomas in this study were atypical in appearance and size. These did not have well-demarcated borders and were composed of variable numbers of eosinophilic, basophilic, clear, or vacuolated hepatocytes and resembled large mixed cell foci involving multiple hepatic lobules. They had well-defined hepatic lobules with central veins and portal triads. These portal triads usually contained bile ductules accompanied by variable oval cell proliferation. Central veins and portal triads within these lesions were more frequent than in typical adenomas. However, because of other histologic features (absence of normal lobular architecture, cellular atypia, and increase mitotic activity) characteristic of benign neoplasia, they were identified as adenoma.
In the male mice, increased incidences of hepatoblastoma occurred with a positive trend and a marginal, but not statistically significant, increase in the 25,000 ppm group. Multiple hepatoblastomas were seen in two 25,000 ppm males. Hepatoblastomas are malignant neoplasms that are presumed to be a primitive form of hepatocellular carcinoma. Microscopically, the hepatoblastomas were usually well demarcated from the surrounding tissue. They consisted of nests, clusters, or sheets of small-to medium-sized, generally spindle-shaped cells that had scant amounts of deeply basophilic cytoplasm and round to oval hyperchromatic nuclei. Hepatoblastomas sometimes occurred within a hepatocellular carcinoma and at other times appeared to arise directly from the liver parenchyma.
Significantly increased incidences of eosinophilic focus or mixed cell focus in the liver occurred in all exposed groups of males. The eosinophilic foci were small to moderately large lesions composed of hepatocytes that were somewhat enlarged and had homogenous or finely granular, eosinophilic cytoplasm. The hepatocytes were arranged in normal lobular patterns in which hepatic cords merged with the surrounding normal hepatocytes. Blood vessels and portal areas were sometimes present within these foci. There was little or no compression of the surrounding normal hepatocytes, although some degree of compression was present in some larger foci. The mixed cell foci were composed of two or more kinds of cell types, with no cell type comprising 80% or more of the focus. Mixed cell foci were often larger than other types of foci and consisted of a mixture of basophilic or eosinophilic cells and clear cells. They had normal lobular hepatocyte patterns that merged into the adjacent parenchyma with variable numbers of blood vessels and portal areas.
In summary, in male mice, hepatoblastomas were statistically significant by the trend statistic, and there was an increased incidence of multiple hepatocellular adenomas in both the 9,000 and 25,000 ppm groups. The incidence of hepatocellular carcinomas or combined incidence of hepatocellular adenoma, carcinoma, or hepatoblastoma in male mice was not statistically increased, although the rate in the high dose was outside of the historical range for this tumor. There was evidence for a carcinogenic response in male mice based on the increase in hepatoblastomas and adenomas, particularly multiples.
In contrast, to the findings in male mice, the incidence of liver neoplasms in treated female mice was not statistically increased. However, the incidence of hepatocellular adenoma in all treated groups exceeded the range in the historical feed female mouse controls (Table 3).
Milk Thistle
Survival and Body Weight
Feed consumption was similar among control and treated rat groups and among control and treated mouse groups and was used to estimate compound consumption (Table 2). Mean body weights of treated groups of male and female rats were generally similar to controls (±10% of controls), and survival of treated rats was also similar to controls. Exposure to milk thistle had no effects on the survival of treated mice. However, treated groups of male and female mice had substantial reductions in body weights (up to 28%, see Table 4 ).
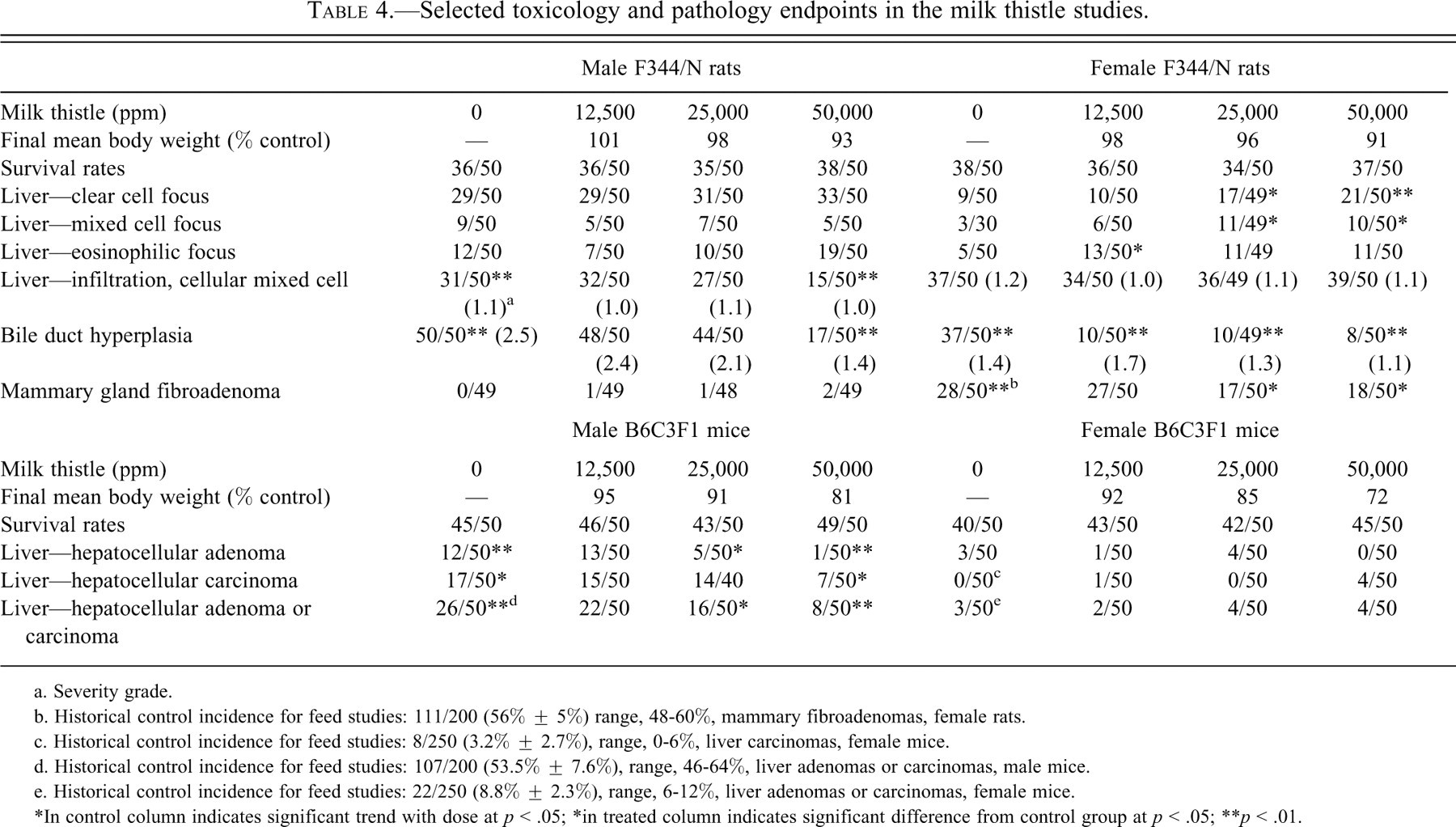
Selected toxicology and pathology endpoints in the milk thistle studies.
a. Severity grade.
b. Historical control incidence for feed studies: 111/200 (56% ± 5%) range, 48-60%, mammary fibroadenomas, female rats.
c. Historical control incidence for feed studies: 8/250 (3.2% ± 2.7%), range, 0-6%, liver carcinomas, female mice.
d. Historical control incidence for feed studies: 107/200 (53.5% ± 7.6%), range, 46-64%, liver adenomas or carcinomas, male mice.
e. Historical control incidence for feed studies: 22/250 (8.8% ± 2.3%), range, 6-12%, liver adenomas or carcinomas, female mice.
*In control column indicates significant trend with dose at p < .05; *in treated column indicates significant difference from control group at p < .05
**p < .01.
Histopathology—Rats
Incidences of mammary gland fibroadenoma (single or multiple) occurred with a negative trend in females and were significantly decreased in groups exposed to 25,000 or 50,000 ppm (Table 4). Mammary gland fibroadenoma consisted of both ductular and/or alveolar epithelium and fibrous connective tissue, and its texture and consistency are related to the amount of collagen present. Mammary gland fibroadenoma is a common finding in aged F344/N rats, with a mean historical incidence of 56% (range, 48-60%) for untreated female rats in feed studies. Furthermore, the incidences of mammary gland fibroadenoma in the 25,000 and 50,000 ppm groups were below the historical control range for feed studies (Table 4). Because body weights of female rats did not differ across the four groups, the reduction in mammary tumors in the 25,000 and 50,000 ppm female rat groups in the current study cannot be attributed to body weight differences.
In the liver of female rats, there were increases in the incidence of clear cell, eosinophilic, and mixed cell foci, but there was no evidence for an increase in liver tumors. A significantly decreased incidence of mixed inflammatory cell infiltration was noted in the liver of 50,000 ppm males; the severity of this lesion was similar in control and treated males. The lesion consisted of randomly distributed foci of inflammation consisting primarily of mixed mononuclear cells that varied from foci of macrophages to a mixture of lymphocytes and the rare plasma cell. Significantly decreased incidences of bile duct hyperplasia (with lowered average severity grade) were noted in 50,000 ppm males and in all exposed groups of females. Bile duct hyperplasia was characterized by the proliferation of biliary epithelial cells within portal areas with occasional minimal extension into the adjacent hepatic lobules.
Decreased incidences of pigmentation were noted in the mesenteric lymph node of all exposed groups of males and females [males: 0 ppm, 45/50 (1.2); 12,500 ppm, 27/50 (1.0); 25,000 ppm, 17/50 (1.0); 50,000 ppm, 9/50 (1.0); females: 47/49 (1.5), 39/48 (1.2), 29/50 (1.3), 18/50 (1.2), respectively, as in males]. The scoring criteria for pigmentation were as follows: normal = no detectable pigment, minimal = pigment in one to three macrophages is detected, mild = pigment is fairly easily detected in four to six macrophages, and moderate = pigment is easily detected in seven or more macrophages. Histologically this pigment was brown in color, finely granular, and found distinctly throughout the cytoplasm of macrophages. In the more severe cases, the pigment appeared more densely packed in the cytoplasm of macrophages, whereas in less severe cases, macrophages appeared to have a lower density of the pigment throughout the cytoplasm. Special staining was used to determine the nature of the pigment in the mesenteric lymph nodes. The staining included PAS for lipopolysaccharides, Hall’s stain for bile and bilirubin, Schmorl’s stain for lipofuscin, Perl’s Prussion blue for iron, and Acid Fast for ceroid. It was concluded that the pigment seen in H&E sections was likely a mixture of iron, lipofuscin, and other black pigment, which was present in control and treated animals, both at extracellular and intracellular locations and looked different than the brown/yellow pigments as described in the H&E staining. The significance of this black granular pigment was unknown. This decreased incidence of pigmentation may have been related to the antioxidative effect of milk thistle and decreasing the age-related subcellular membranous structure degradation (Fraschini, Demartini, and Esposti 2002).
Histopathology—Mice
In male mice, the incidences of hepatocellular adenoma, hepatocellular carcinoma, and hepatocellular adenoma or carcinoma (combined) occurred with negative trends (Table 4). Significantly decreased incidences of hepatocellular carcinoma occurred in 50,000 ppm males; decreased incidences of hepatocellular adenoma and of hepatocellular adenoma or carcinoma (combined) occurred in 25,000 and 50,000 ppm males. All of these decreased incidences were below the respective historical ranges for these tumors. Decreased incidences of clear and mixed cell foci and hepatocytic cytoplasmic vacuolization were noted in the liver of all exposed groups of males (data not shown). Altered cell foci (eosinophilic, mixed, basophilic, and clear) were characterized by a focus of hepatocytes with altered tinctorial properties.
The decreased incidences of hepatocellular adenoma or carcinoma (combined) in males may be attributed to decreases in body weights (Stout et al. 2008), but a potential direct effect of the milk thistle extract exposure cannot be excluded.
Hepatocellular adenomas were variably sized, nodular lesions composed of well-differentiated, neoplastic hepatocytes that typically compressed the adjacent hepatic parenchyma. Portal areas and central veins were typically absent. Hepatocellular carcinomas were well demarcated from the surrounding hepatic parenchyma and were composed of neoplastic hepatocytes that displayed mild to marked cellular and nuclear pleomorphism and mitoses. The predominant pattern displayed by most neoplasms was trabecular, although focal areas displayed glandular and solid patterns of growth. Necrosis was occasionally quite extensive.
Incidences of hepatocellular carcinoma occurred with a positive trend in females.
Because the incidences of hepatocellular adenoma or carcinoma (combined) were similar across groups, the increased incidence of hepatocellular carcinoma in females is considered unrelated to exposure to milk thistle extract.
Discussion
These studies demonstrate the diversity of responses that can occur from long-term use of herbal medicines, from beneficial to carcinogenic effects. For goldenseal there was evidence for a carcinogenic effect in the liver of rats and mice, while with milk thistle there was evidence for antiproliferative effects in the liver of rats and male mice and in the mammary gland in female rats.
While goldenseal contains other components, biological activity related to exposure to this herb is considered to be due to the presence of alkaloids, particularly berberine (Abourashed and Khan 2001). Berberine and its more potent metabolite, berberrubine, are topoisomerase inhibitors (Kobayashi et al. 1995; Makhey et al. 1994). Recombination, transcription, and replication of prokaryotic and eukaryotic DNA are facilitated by topoisomerases due to their ability to break and rejoin the phosphodiester backbone of DNA (Champoux 2001; Kobayashi et al. 1995; Makhey et al. 1994). Topoisomerases have been shown to be principal targets for some anticancer agents, and inhibition of the enzymes has been associated with antineoplastic properties of protoberberines and berberine (Kettmann et al. 2004; Mazzini et al. 2003; Sun et al. 2009). The mechanism involves disruption of DNA ligation leading to apoptotic cell death (Kettmann et al. 2004). In the present studies, anti-tumorigenic activity of berberine or its metabolites was not apparent in the liver of rats and mice chronically exposed to goldenseal root powder. Although topoisomerase inhibitors are used as anti-cancer agents in humans, this class of drugs can disrupt DNA repair processes and induce chromosomal aberrations leading to malignancies at secondary sites (Malik et al. 2006). In fact, the International Agency for Research on Cancer (IARC) has listed etoposide and teniposide, topoisomerase inhibitors used as anti-cancer drugs, as probably carcinogenic to humans (Group 2A) (International Agency for Cancer Research 2000). Therefore, it could be postulated that inhibition of topoisomerases by berberine and its metabolites lead to the increased incidence of liver tumors in rodents receiving goldenseal root powder. The liver of the rat is one of the tissues with the highest rates of DNA repair (Gospodinov et al. 2003), and thus, disruption of DNA repair in this tissue might make it more susceptible to exogenous oxidative stress than other tissues. In B6C3F1 mice, the liver is predisposed to neoplasm formation; therefore, disruption of DNA repair processes may enhance carcinogenic events in this organ. Because the essential components for DNA repair processes are conserved among eukaryotes (Malik et al. 2006), the findings of the present goldenseal studies in rodents impact considerations for risk assessment in humans.
In humans, hepatocellular carcinoma is the fifth most common cancer in the world (El-Serag and Rudolph 2007), with limited treatments available (Hoshida et al. 2009). Hepatitis B is a primary risk factor for liver disease and cancer, but environmental factors including alcohol, vinyl chloride, and aflatoxin also contribute to liver disease and cancer. Combination exposures to liver toxins including goldenseal could work synergistically to cause liver disease.
Decreases in liver proliferative response after exposure to milk thistle extract (silymarin) have been observed in rodents (Ramakrishnan et al. 2009; Wu et al. 2008) and in human liver disease (El-Kamary et al. 2009; Seeff et al. 2008). Milk thistle is currently a subject of study in NIH clinical trials as a treatment for chronic hepatitis C, hepatic failure after mushroom poisoning, and in other liver disease conditions (National Institutes of Health 2010).
In the present studies, male mice treated with milk thistle extract had a treatment-related decrease in the incidences of liver foci (including mixed and clear cell foci), hepatocellular adenoma, hepatocellular carcinoma, and the combined incidences of hepatocellular adenoma or carcinoma. Liver foci are considered to be composed of cells with increased proliferative activity (Goldsworthy and Fransson-Steen 2002; Ittrich et al. 2003; Takahashi et al. 2002), and thus the decreases in liver foci incidences support the finding of decreased incidences of liver neoplasms in male mice. Silymarin treatment is reported to reduce human hepatocellular carcinoma xenograft growth in nude mice by inhibiting cell proliferation (Cui, Gu, and Hu 2009); this finding supports our observation of reduced liver neoplasms in mice receiving milk thistle extract.
In female rats, there were some increases in the incidence of liver foci while in male rats there was no evidence for this increase in liver foci, but there was a decrease in liver cellular infiltrates. The background rate of liver neoplasms in rats is low (male rats: mean, 2.0%, range, 0-6%; female rats: mean, 2.0%, range, 0-4% for feed studies). Milk thistle treatment had no effect on the occurrence of liver tumors in male or female rats. We do not know the reasons for the differences in milk thistle effects on the liver foci incidence in male and female rats. It should be noted that silymarin in the presence of activation enzymes induced mutations in Salmonella strains (Kaleeswaran et al. 2009), and it is possible that this mutagenic activity may have stimulated liver foci formation in the female rats in the presence of a background of antiproliferative activity.
A decrease in mammary gland tumors in female rats was also observed in the milk thistle extract study, with no accompanying decrease in body weight. This effect may have been related to milk thistle extract exposure. However, our milk thistle extract study was not designed to determine the mechanisms for the observed decreased incidence of liver neoplasms in exposed male mice or decreases in mammary gland neoplasms in exposed female rats. Studies in the literature have reported that milk thistle exposure may have antiproliferative activity and/or radical scavenging and antioxidant activity (Fraschini, Demartini, and Esposti 2002; Fu et al. 2009). Whether any of these proposed milk thistle activities impacted the development of liver or mammary gland neoplasms would require additional mechanistic studies.
Oxidative stress accumulates with age and leads to increased levels of DNA damage (Olinski et al. 2007). As animals age, there may also be decreased p53-mediated tumor suppression (Feng et al. 2007) and decreased antioxidant activity (Shih and Yen 2007). This combination of factors may contribute to carcinogenesis particularly at sites of heightened susceptibility such as the male B6C3F1 mouse liver. The protective effects of milk thistle (or its components), including antioxidant activity, may help to prevent carcinogenicity-associated proliferative processes (Comelli et al. 2007; Gazak, Walterova, and Kren 2007). Silibinin lowers the level of mitochondrial reactive oxygen species production in perfused rat hepatocytes (Detaille et al. 2008). This finding is supported by additional findings for anti-cancer/anti-proliferative activity of milk thistle or its components, including antioxidant activity (Awang 1993); inhibition of cyclin-dependent kinases; G2/M arrest and inhibition of Cdc25c, Cdc252, and cyclin B1 protein expression (Meeran and Katiyar 2008); and/or hepatocellular cholestatic activity (Crocenzi and Roma 2006; Meeran and Katiyar 2008). Another hypothesis for milk thistle antioxidant activity is that silibinin acts as a chemopreventive agent through the up-regulation of insulin-like growth factor binding protein 3, sequestering high levels of insulin-like growth factor, a mitogenic signal and an antiapoptotic signal, from tissue milieu (Comelli et al. 2007; Singh et al. 2002). Blocking of matrix-metalloproteinase-9, which may lead to suppression of tumor growth, has also been proposed as a mechanism by which silibinin inhibits tumor growth (in vitro studies in MCF-7 cells) (Lee, Narayan, and Barrett 2007). Others propose that silybin may prevent cancer through G1 arrest (Deep et al. 2006; Meeran and Katiyar 2008). Thus, the biochemical modulation of cell cycle processes by milk thistle components, along with the observed decreases in body weight gain, may have been at least partly responsible for the reduction in normal neoplasm incidences in mice. These mechanistic studies reported in the literature help explain how exposure to milk thistle extract may have worked to prevent the carcinogenesis process in the male mouse liver in the current study.
The doses of milk thistle extract and goldenseal root powder used in these studies were generally higher than exposure in humans for the treatment of a variety of diseases (Figure 3; Table 2 footnotes). Work is currently being conducted to understand the metabolic and toxicokinetic parameters of herbal medicines in humans (National Institutes of Health 2010).
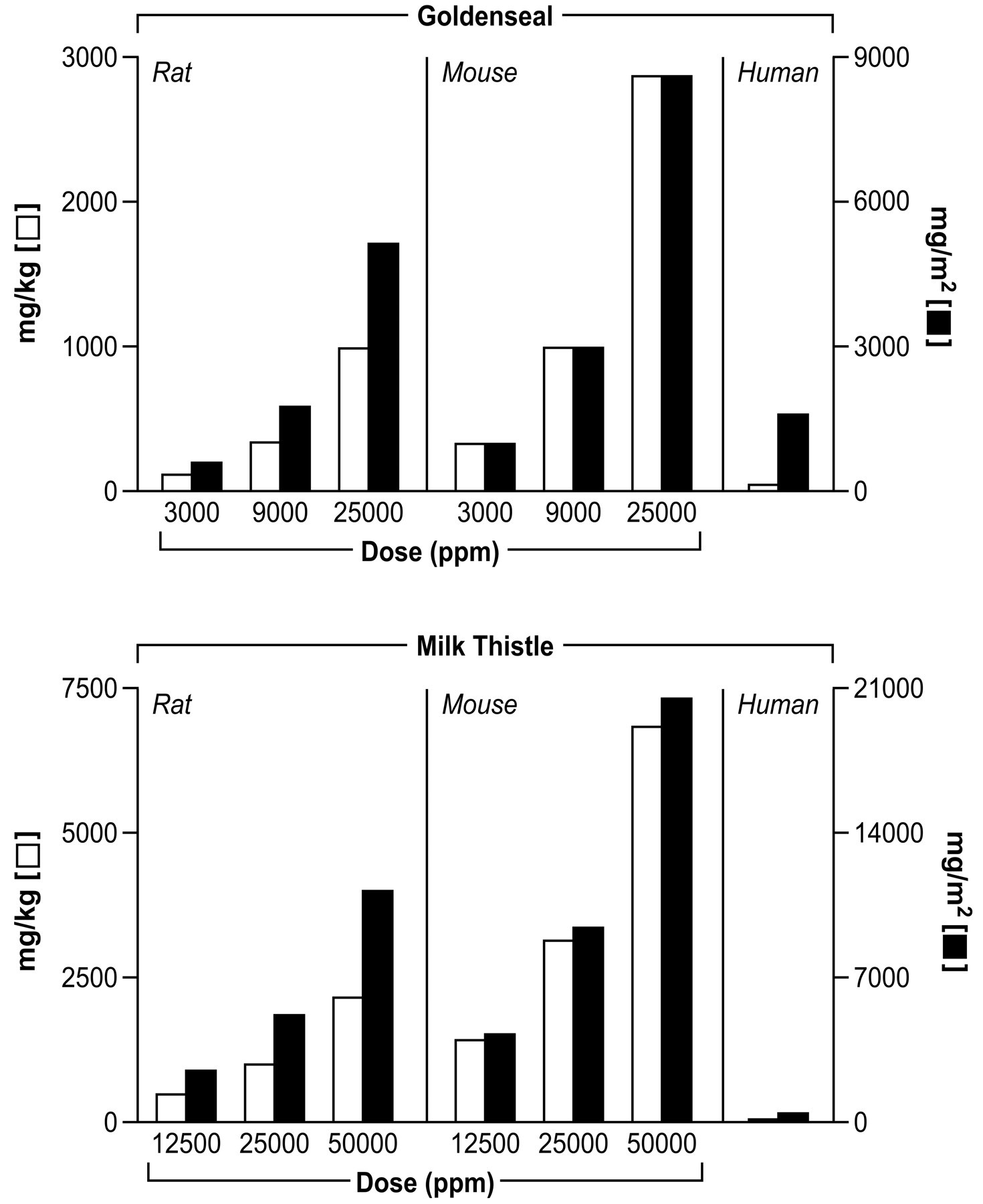
Comparison of human and rodent exposures to herbal products.
Footnotes
This research was supported in part by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences. The animals for the goldenseal and milk thistle study were dosed and housed at Southern Research Laboratory (NTP contract N01-ES-15463). We gratefully acknowledge the expert review of Dr. R. Irwin and Dr. A. Brix, NIEHS, RTP, NC. This paper is dedicated in memory of Dr. John Peckham, a great colleague with extensive knowledge and expertise in pathology.
