Abstract
Diseases that commonly cause dementia in the general population are Alzheimer’s disease, microvascular brain injury, and Lewy body disease. Each of these is a chronic disease with latent, prodromal, and dementia stages. Here I present outcomes from validated neuropathologic assessments and cerebrospinal fluid biomarkers to estimate the prevalence of the latent stage of diseases that commonly contribute to the dementia syndrome. The latent stage of diseases that commonly cause dementia is highly prevalent in the adult population, highlighting the need for prevention strategies and longitudinal studies.
Stages of Chronic Disease
A chronic disease model is widely applied in medicine. Common examples are malignancies like carcinoma of the cervix or prostate, and some forms of infectious disease like HIV infection and AIDS. In theory, chronic diseases exist in four stages: two that can be defined exclusively by functional (clinical) data and two requiring both functional and laboratory data for accurate definition (Table 1 ). The most advanced stage of chronic disease (Stage 3) occurs with overt and severe functional manifestations. Although not necessary to establish Stage 3 disease, laboratory tests would be expected to show a heavy burden of disease. Individuals with less severe functional impairment who are also not functionally normal constitute a prodromal stage of illness (Stage 2). Again, although defined exclusively by functional data, laboratory data from individuals in the prodromal stage of disease would be expected to show substantial disease burden, albeit less than in Stage 3. The earliest two stages of chronic disease are both characterized by normal function. Although the definition of normal function is fluid because the methods used to assess function can change, we are assuming no indication of disease upon the “usual” clinical or functional evaluations. Reliance exclusively on functional data for this level of disease leads some to describe this stage as “preclinical.” It is our opinion that this is a true but ambiguous designation since the “preclinical” stage in theory can be composed of two different disease stages that are distinguished by laboratory data. Some individuals with normal function will show no evidence of disease by all manner of laboratory testing (Stage 0). However, others who are functionally normal will have initiated disease that is detectable by laboratory testing; this is called latency (Stage 1).
Chronic disease model for diseases that can cause dementia.
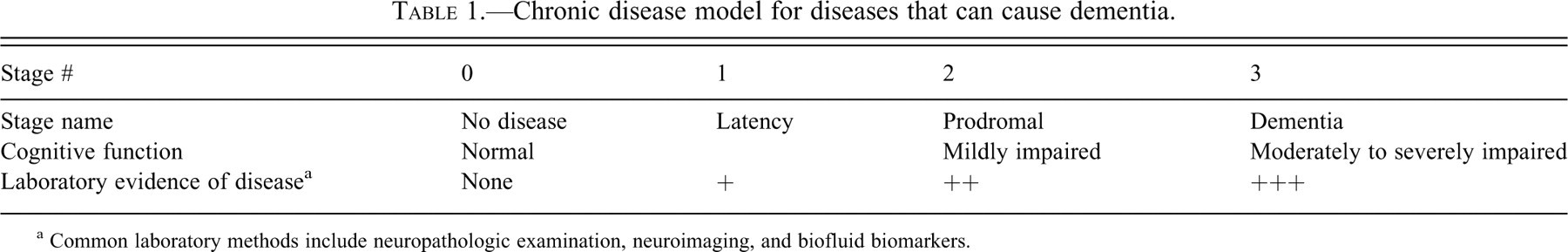
a Common laboratory methods include neuropathologic examination, neuroimaging, and biofluid biomarkers.
Chronic Disease Model of Illnesses That Can Culminate in Dementia
Some confusion may arise from the common usage of the term dementia. Dementia is a functional (clinical) assessment of severe impairments in cognition and behavior, that is, dementia is Stage 3 of chronic disease that impairs cognition. Moreover, dementia is a syndrome, not a single disease. Operationalized criteria for dementia (Stage 3) are highly codified in the Diagnostic and Statistical Manual of Mental Disorders (DSM) (American Psychiatric Association 2000). Prodromal or Stage 2 of diseases that can cause dementia have received intense attention over the past decade, and some operationalized criteria are in common use for this stage as well. One example is criteria that define amnestic mild cognitive impairment (aMCI), which appears to approximate closely prodromal Alzheimer’s disease (AD) (Petersen and Negash 2008). Most recently, attention has turned to identifying latent (Stage 1) disease among individuals who are functionally normal. Since Stages 0 and 1 can be distinguished only by laboratory data, many investigators are now focused on developing biofluid and neuroimaging biomarkers of diseases that can culminate in dementia. Some success has been achieved, especially for some facets of AD cerebrospinal fluid (CSF) biomarkers in mild cognitive impairment and dementia (Sonnen et al. 2010); however, there are as yet no validated biomarkers for all three of the diseases that commonly cause dementia, namely, AD, Lewy body disease (LBD), and microvascular brain injury (µVBI). As we await progress in this area, several groups have made use of laboratory data from neuropathologic evaluations of cognitively normal individuals to estimate the prevalence of latent neurodegenerative disease. Alzheimer’s disease is characterized by the accumulation of large extracellular structures partially composed of amyloid β peptides called neuritic plaques as well as by neuronal inclusions composed largely of modified tau protein called neurofibrillary tangles; LBD is characterized by neuronal inclusions (Lewy bodies) composed largely of α-synuclein; and µVBI is characterized by the formation of partial or complete ischemic lesions that are too small to be observed by eye but rather are identified by microscopic examination.
Estimate of the Population-attributable Risk for Dementia
The Adult Changes in Thought (ACT) study is a prospective population-based study of brain aging and incident dementia in women and men aged sixty-five years or older who live in the Seattle, Washington, greater metropolitan area (Larson et al. 2004). Since autopsy remains the only means to assess a wide array of potential contributors to dementia, we analyzed over sixteen structural changes (Sonnen et al. 2007) associated with neurodegeneration in the ACT autopsy cohort and determined that three were significantly and independently associated with dementia: high Braak score as a structural indicator of advanced AD, high LBD score, and more than two cerebral microinfarcts as a structural indicator of advanced µVBI (Sonnen et al. 2007). Because ACT is a population-based study, from these data we were able to compute the population-attributable risk (PAR) for dementia in this cohort (Figure 1 ). It is important to stress that although AD is the largest contributor to the PAR for dementia, in this cohort that does not derive from a tertiary medical center, the contribution from AD is relatively less than estimates from AD research center cohorts. Moreover, approximately 12% of dementia remains unexplained by these estimates. It should be noted that the error in our estimates of the PAR for each disease is sufficiently large to account for 100% of dementia, so it is possible that these three diseases represent all common contributors to the risk of dementia. Alternatively, another contributor, or contributors, that is not detected by neuropathologic examination may account for some small but measureable amount of PAR for dementia, for example, normal pressure hydrocephalus or chronic alcoholism. However, less common diseases that can be observed by neuropathologic examination, such as frontal temporal lobar degeneration, Huntington’s disease, or prion disease, were not present as contributors to dementia in this sampling of the ACT cohort.
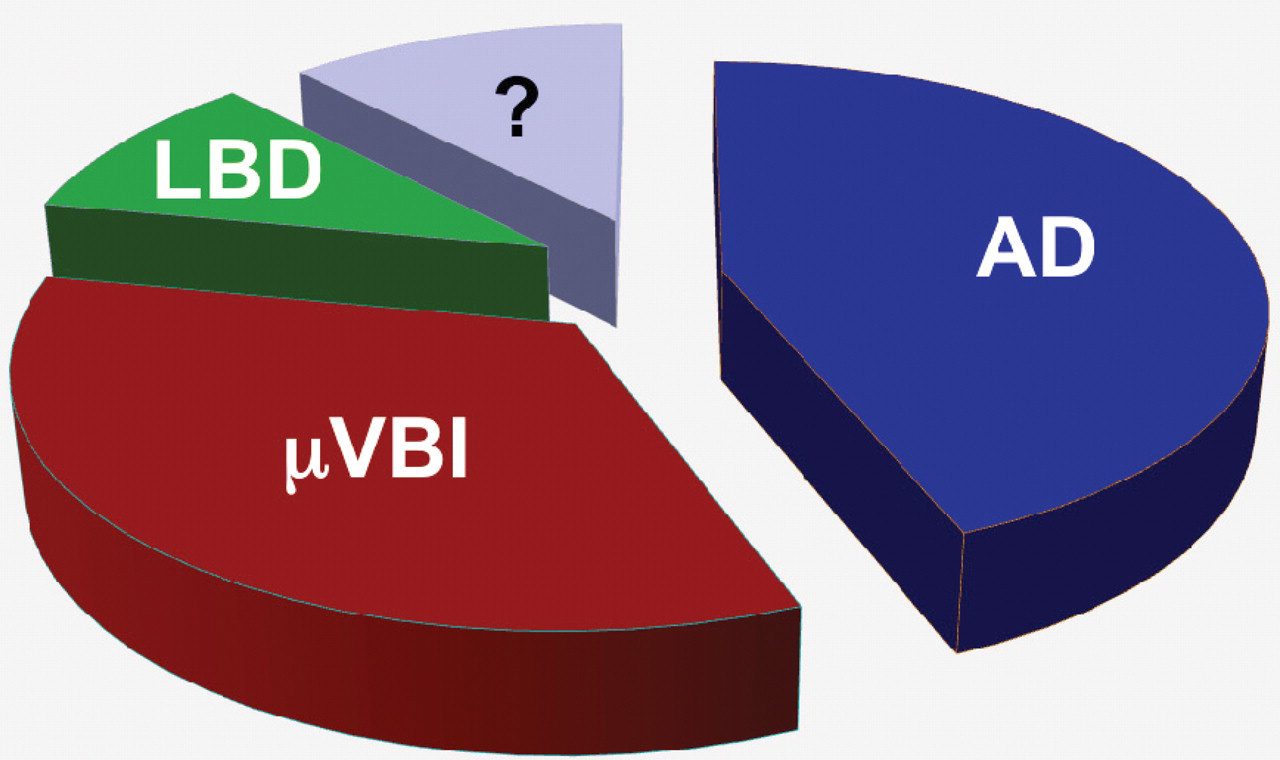
Population-attributable risk of neuropathologically identified diseases that commonly contribute to dementia. Alzheimer’s disease is 44%, microvascular brain injury is 34%, Lewy body disease is 10%, and unknown (?) is 12%.
Estimate of the Prevalence of the Latent Stage of Diseases That Can Cause Dementia in ACT
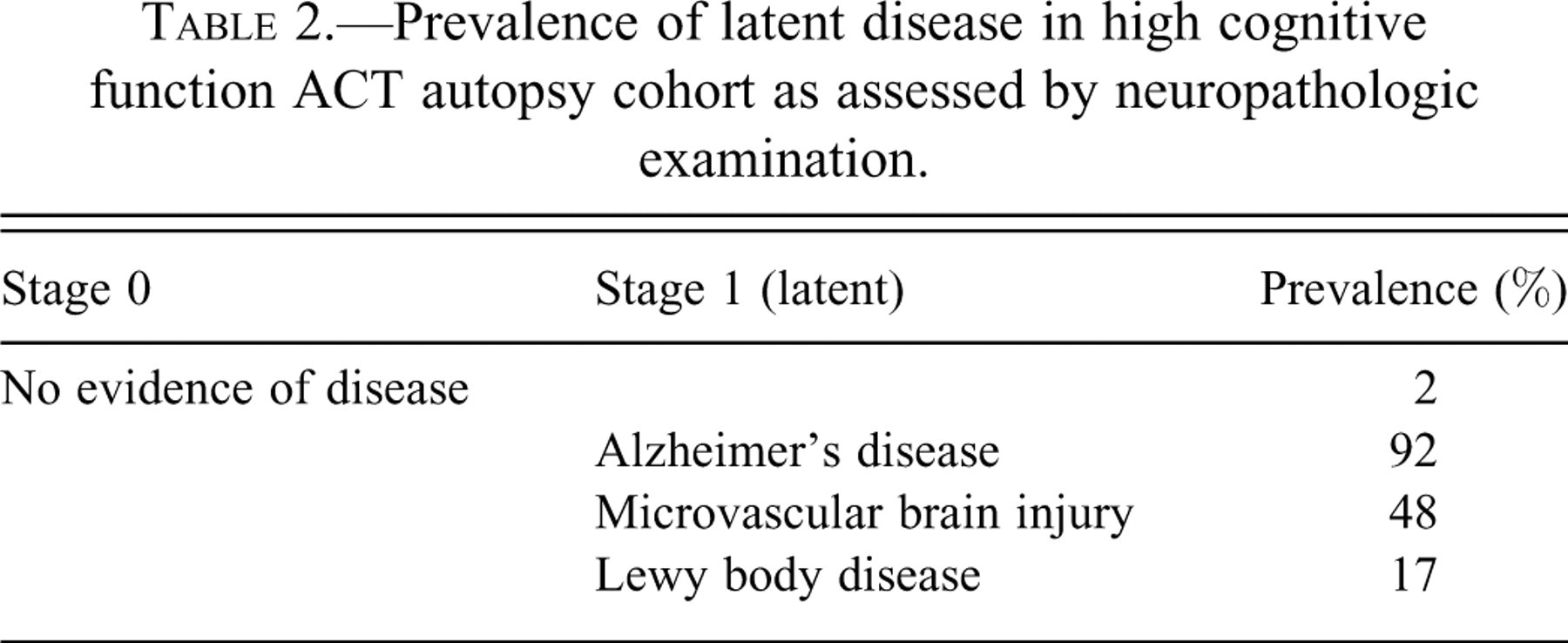
We next estimated the prevalence of latency for AD, µVBI, and LBD in the ACT autopsy cohort by performing similar postmortem analysis on individuals with normal cognitive function close to the time of death. The normal cognition group was defined as those ACT participants who had their last structured evaluation within two years of death at which time they did not meet DSM-IV criteria for dementia, and whose cognitive function screening test results were within the upper four quintiles for the entire ACT cohort at baseline. More concretely, inclusion in this analysis required that ACT participants who donated their brains for research were last functionally evaluated within two years of death, at which time they achieved 91 to 100 on the Cognitive Assessment Screening Battery (maximum score = 100). Characteristics of the 116 ACT autopsy cases that met these criteria for high cognitive function are: average ± SD age = 84 ± 6 years, male:female ratio of 52:64. Using the same extensive neuropathologic examination described above (Sonnen et al. 2007), we estimated the prevalence of latent AD, µVBI, and LBD in this group of 116 ACT cases with high cognitive function (Table 2 ). These data suggest that only a very small subset of older adults is free from a latent form of at least one of these three diseases. Indeed, the vast majority of high cognitive functioning older adults appear to have latent AD, µVBI, or LBD, and about one-quarter appear to have the latent stage of two or more of these diseases.
Prevalence of latent disease in high cognitive function ACT autopsy cohort as assessed by neuropathologic examination.
Estimate of the Prevalence of the Latent Stage of AD With Biomarkers
An advantage of the above autopsy-based study is that we can simultaneously assess the presence of a wide array of diseases by neuropathologic examination. The disadvantage is that there is asynchrony between collection of functional data and laboratory data. This asynchrony is especially important when investigating latent stages. For this reason, as well as to meet the goal of generating a useful laboratory test to aid in clinical management, several groups are investigating the use of neuroimaging and biofluid biomarkers for diseases that commonly cause dementia. Since these are intra vitam laboratory tests, they can be performed simultaneously with functional assessments of cognition. Our focus has been CSF biomarkers. By far the greatest progress has been made with AD, for which several groups have repeatedly validated the utility of quantifying CSF amyloid (A) β42 peptide and tau protein, commonly expressed as the tau/Aβ42 ratio, to characterize the dementia and prodromal stages of AD (Sonnen et al. 2010). Indeed, both prodromal and dementia stages of AD display a significantly increased CSF tau/Aβ42 ratio that derives from increased CSF tau, thought to be a nonspecific marker of neuronal injury, and decreased CSF Aβ42, a process associated with increased deposition of Aβ peptides in brain (Fagan et al. 2006). Four groups have now used elevation of the CSF tau/Aβ42 ratio to estimate the prevalence and functional significance of latent AD (De Meyer et al. 2010; Fagan et al. 2007; Li et al. 2007; Stomrud et al. 2010). Figure 2 is an example from our earlier study (Li et al. 2007). Here we began with the assumption that individuals less than fifty years old who performed within the normal range on a battery of neuropsychological tests at the time of lumbar tap were very unlikely to harbor latent AD, and thus that their tau/Aβ42 ratios represented values for Stage 0. This assumption of normality was substantiated by the Gaussian distribution of CSF tau/Aβ42 in these thirty-four younger individuals. Using these data, we then determined an upper limit of CSF tau/Aβ42 ratio (the value of which will vary with the reagents) for Stage 0, namely, a cutoff value between normal and abnormally increased CSF tau/Aβ42 ratio. We applied this criterion to a group of 102 volunteers fifty years or older who also performed in the normal range on a battery of neuropsychological tests at the time of lumbar tap. The distribution of CSF tau/Aβ42 ratios for those with values below the cutoff mirrored the younger group (Stage 0). It is important to note that a normal CSF tau/Aβ42 ratio was not a function of age, since our oldest participant (over 102 years old) had a normal CSF tau/Aβ42 ratio. However, CSF tau/Aβ42 ratio was skewed to higher values in this older cohort, with 21% above the cutoff. This result can be interpreted as laboratory evidence of AD in this group of cognitively normal individuals, or the latent stage (Stage 1) of AD. Proof that these individuals have latent AD requires clinical follow-up and demonstration of subsequent functional impairment. Indeed, our group demonstrated that the individuals with elevated CSF tau/Aβ42 were at significantly increased risk for progressing to prodromal or dementia stage disease within four years, a finding that has been observed in other studies (Fagan et al. 2007).
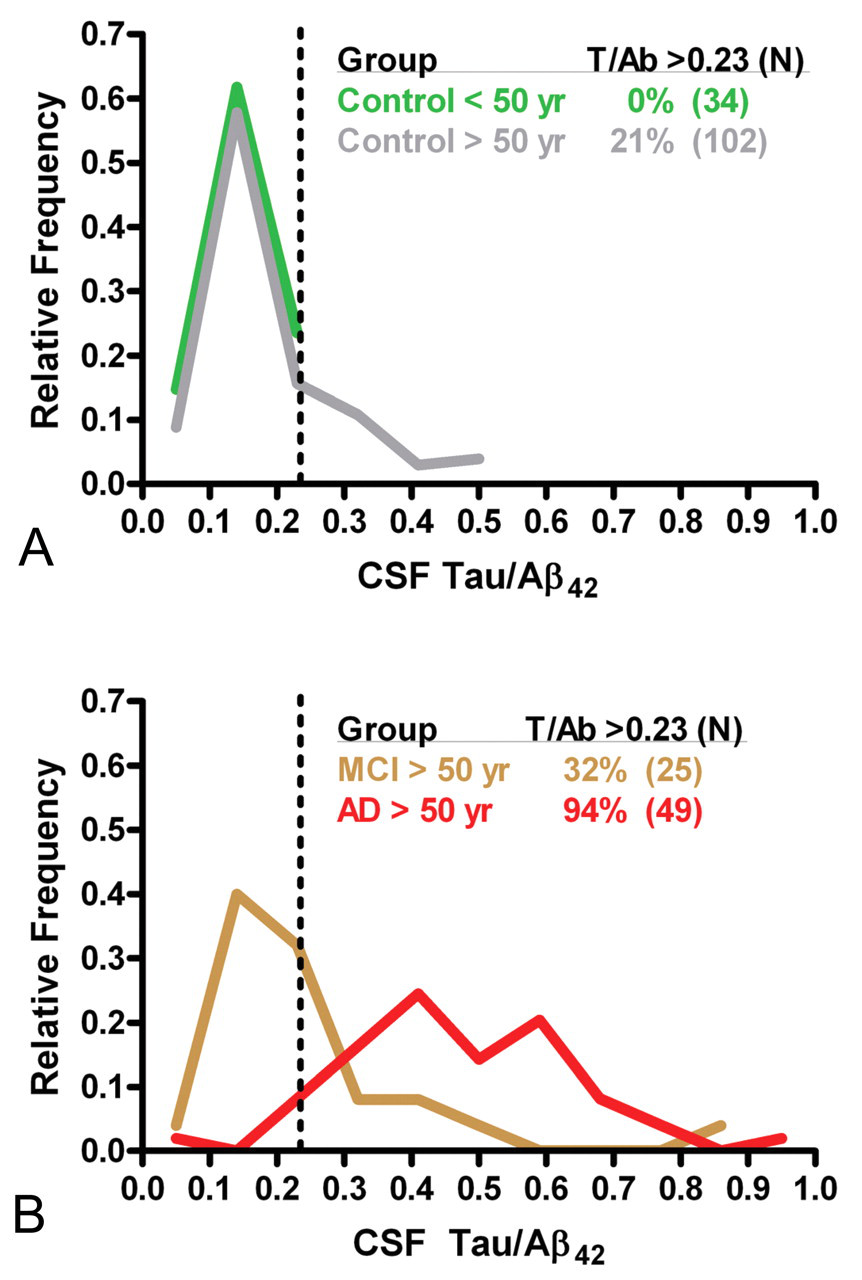
CSF tau/Aβ42 ratio as a laboratory method to detect latent Alzheimer’s disease in cognitively normal individuals (Panel A). Examples of data from patients with amnestic mild cognitive impairment (Stage 2) or Alzheimer’s disease (Stage 3) are included for comparison (Panel B).
Summary
At some point in the hopefully not-too-distant future, there will be disease-modifying therapies for AD and LBD; such therapies likely already exist for µVBI. History teaches us to expect that these new disease-modifying therapies likely will be most effective when applied early in the course of disease and that these therapies will likely carry some risk or untoward side effect(s) that need to be balanced against the benefits. For both of these reasons, identification of latent-stage disease will greatly enhance such developments.
Using our data from a population-based study of brain aging and incident dementia with autopsy end point to estimate the prevalence of latent AD, µVBI, and LBD in a group of women and men from the Seattle area who were on average in their mid-eighties, we estimate the prevalence of latent AD as approximately 60%. Using validated CSF biomarkers for dementia and prodromal-stage AD in a group of cognitively normal volunteers fifty years and older, we estimate the prevalence of latent AD in this group as 21%. Estimates from most other biomarker and neuroimaging studies fall somewhere within this wide range. Likely one major reason for this wide range in prevalence estimates is the difference in age between the two groups investigated. Indeed, there are substantial existing data to suggest that the prevalence of latent AD increases with advancing age. However, this may not be the full explanation. It is also possible that the sensitivity of the two laboratory methods (autopsy vs. CSF protein measurement) is very different; we suspect that autopsy might be substantially more sensitive.
Finally, although our data as well as that of others indicate that individuals with latent AD are at high risk for near-term clinical display, it is important to stress that latent-stage disease does not carry the requirement for advancement to prodromal or dementia stages. Indeed, it is entirely possible that latent disease may be static in some individuals and perhaps even reversible in others. More extensive longitudinal biomarker and neuroimaging studies are needed to fully understand the natural history of these chronic diseases and their response to therapeutic interventions.
Footnotes
Acknowledgments
This work was supported by AG05136, NS62684, and the Nancy and Buster Alvord Endowment. Thanks to Dr. Kathleen Montine for editorial assistance.
