Abstract
Toxicity studies of intranasally administered, live attenuated influenza virus vaccine candidates conducted in male and female ferrets led to the microscopic observation of individual differences in the size of nasal turbinates, especially in the dorsal aspect of the nasal cavity. The association of these enlarged turbinates with acute to subacute inflammation, which is sometimes common in ferrets given live attenuated influenza virus vaccine candidates, led to this detailed microscopic evaluation of turbinate enlargement (cartilaginous and osseous thickening, or COT) in control animals dosed intranasally with saline. Results of this evaluation led to the conclusion that COT is a normal developmental feature of growing ferrets, irrespective of inflammation in nasal tissues or inflammatory exudate in the nasal cavity.
Keywords
Introduction
Histopathologic evaluations of 9- to 17-week-old ferrets used for nasal vaccine inoculation studies showed cartilaginous and osseous thickening (COT) in some animals at Level 1 of the nasal turbinates. Overall, there was no apparent relationship between the occurrence of COT and vaccine treatment. In a few of these studies, COT was associated with significant intranasal inflammation, which in some instances was thought to be causative. In other experiments, however, ferret nasal turbinates were thickened without microscopic evidence of underlying inflammation or other obvious etiologies. Because ferret nasal COT has not been previously reported, and because it is possible that COT could be misinterpreted as a treatment effect, a retrospective analysis of nasal turbinates from control ferrets was undertaken to more fully characterize the prevalence and extent of this finding.
Materials and Methods
Two hundred male and female vehicle control ferrets from seven vaccine studies were reviewed for the incidence and severity of COT over time. Ferrets received from one to three doses of 0.1–0.5 mL of saline (PBS, Gibco) intranasally. Dosing was done on Days 0, 14, and 28; therefore, ferrets sacrificed at Day 0/3 received one dose, those sacrificed on Day 31 received two doses, and those sacrificed on Day 56 received three doses.
Ferrets were supplied by Triple F Farms, Inc. (Sayre, PA) at seven to ten weeks of age. The ferret breeding colony is maintained from five different genetic lines; DNA testing has indicated as much variation within the lines as between the five genetic lines. At the vendor facility, serum was collected from each ferret at five to eight weeks of age and analyzed for the presence of antibodies to A/New Caledonia/20/99 (H1N1), A/California/7/04 (H3N2), A/VN/1203/04 (H5N1), B/Jiangsu/10/03 (type B), A/Solomon Island/3/06 (H1N1), H9N2 A/Chicken/Hong Kong/G9/97, H2N2 A/Ann Arbor/6/60, H6N1 A/Teal/Hong Kong/W312/97, A/South Dakota/6/07, A/Uruguay/716/07, A/Solomon Island//06 (H1N1), A/Wisconsin/67/05 (H3N2), B/Florida/4/06 (B/Yamagata lineage, 08-09 vaccine), and/or B/Malaysia/2506/04 (B/Victoria lineage) influenza strains using hemagglutination inhibition (HAI). Only animals negative for antibodies to influenza strains as defined by an HAI titer <8.0 were used in these studies. No animals with positive titers were found from this supplier. For one study, ferrets had been vaccinated against canine distemper virus at 7.5 weeks of age.
Upon receipt at the test facility, animals were examined physically for general health and viability. Each ferret was identified by an implanted, programmable microchip (IPT 300, BioMedic Data Systems, Seaford, DE) that was cross-referenced with the vendor’s ear tag. For three of the studies, feces from each shipping container were analyzed for the presence of coccidian ova. Ferrets from these same three studies received a maintenance dose of Banax Oral Solution (9.6%) (amprolium, Vedco, Saint Joseph, MO) in drinking water during the first three days of acclimation as a prophylactic treatment for coccidia. At the end of the acclimation period, each ferret received a physical examination by a veterinarian, including evaluation of appearance, behavior, mobility, posture, assessment of food and water intake and urine/feces output, palpation of body surfaces and internal organs, confirmation of sex, and determination of body weight. Acceptance of each ferret into a trial occurred only upon the veterinarian’s approval.
Ferrets were acclimated for at least ten days prior to study initiation. They were housed individually in polycarbonate shoebox cages (Udel Large Rat Cage, Model # 5, Thoren Caging Systems, Hazleton, PA) with stainless steel wire lids (Model #6B, Thoren) and microisolator tops (Udel Filter Cover Model #6, Thoren) during acclimation and throughout the in-life stage of the study. Certified Omega Dry Bedding (Harlan, Madison, WI) or equivalent, analyzed by the supplier for environmental contaminants (heavy metals, chlorinated hydrocarbons, total aflatoxin, organophosphates, bacteria, yeast, and mold; limits set by the manufacturer), was changed two or three times weekly or more often as needed. Environmental conditions in the animal room were set to 62°F–82°F and 30%–70% relative humidity. Deviations from these set points occurred on several of the studies, and actual conditions ranged from 61°F–81°F and 17%–92% relative humidity. Excursions from set points were of limited duration and were not expected to have any impact on animal health. The air flow in the animal room exchanged ten to fifteen times per hour, and illumination was on an automatic twelve-hour day/night cycle.
Ferrets were fed certified Harlan Teklab Ferret Diet #2072C (Harlan, Madison, WI) offered ad libitum, except during fasting periods prior to dosing and necropsy, and during urine collection. Feed was certified by the manufacturer as to nutritional components and contaminant levels (heavy metals, pesticides, polychlorinated biphenyls, and total aflatoxin). Except during urine collection, ferrets had ad libitum access to individual water that met U.S. Environmental Protection Agency potable water standards supplied in water bottles (Udel, sixteen-ounce bottles, Thoren).
Blood was collected for determination of clinical pathology values at nine to eleven weeks and seventeen to nineteen weeks. Animals were anesthetized with either isoflurane (nine to eleven weeks) or ketamine/xylazine (seventeen to nineteen weeks), and blood was collected from either the vena cava or via cardiac puncture.
Ferrets were sacrificed humanely using ketamine/xylazine anesthesia followed by exsanguination via the femoral and brachial arteries. At necropsy, thirty-seven tissue types, including nasal turbinates, femoral and sternal bone, kidney, and parathyroid, were collected and placed in 10% neutral buffered formalin (NBF). Neutral buffered formalin was also injected into the nasal cavity via the nasopharynx, after which the entire head was placed in NBF. After a minimum of four days in NBF, extraneous tissue was trimmed from the head, and transverse segments of nasal turbinates, each containing one of the evaluated levels, were cut on a band saw (model #80, Marmed, Inc., Cleveland, OH). These segments were then decalcified in a formic acid/EDTA solution (Formical-2000, Decal Chemical Corporation, Tallman, NY) for up to six days. Bone was considered decalcified when the tissue was flexible by manual manipulation.
Following decalcification, nasal turbinate segments were further trimmed to approximately 3 mm thick for placement in standard tissue cassettes. Evaluated sites included: (1) the nares (site of the saline inoculation); (2) Level 1, which included the hard palate (roof of the mouth); and (3) Level 2, which included the soft palate and pharynx. Level 1 was obtained immediately caudal to the incisors, and Level 2 was acquired at the anterior portion of the olfactory bulbs and the ventral orbital rim (the eyes and brain had been previously removed); each of these levels was placed in the cassette so that the sectioning would begin with the anterior side. Guidelines for nasal turbinate sectioning were based on those for the rat (Young 1981).
Tissues were processed to paraffin blocks using a Tissue-Tek VIP (Sakura Finetek U.S.A., Inc., Torrance, CA) according to standard, accepted procedures. Sections were prepared (HM 315, HM 320, and HM 325 Manual Microtomes, MICROM International GmbH, Waldorf, Germany) at 4–6 μm thickness, mounted on glass slides using an appropriate mounting medium (Permount Solution, Fisher Scientific Co., Pittsburgh, PA), stained with hematoxylin and eosin (Hematoxylin 2 and Eosin-Y, Richard-Allan Scientific, Kalamazoo, MI), and coverslipped using automated staining and coverslipping machinery (H/I Linear Stainer and HCM6000 Automated Coverslipping Machine, respectively, Hacker Instruments and Industries, Inc., Winnsboro, SC).
The slides were evaluated by brightfield microscopy by the initial study pathologist (KAF). The following severity scoring system was used: Grade 1 = minimal, Grade 2 = slight/mild, Grade 3 = moderate, Grade 4 = moderately severe, and Grade 5 = severe/high (throughout this retrospective study, the highest severity rating assigned for COT was moderate). Grade 1 changes were generally unilateral, and Grade 2 and 3 changes were bilateral. All turbinate slides were reviewed by a second pathologist (JRL).
Results
Cartilaginous and osseous thickening was diagnosed if there appeared to be active bone or cartilage proliferation in the nasal turbinates. Cartilaginous and osseous thickening was observed most often at Level 1 of the nasal turbinates, and it occurred most commonly in the dorsal aspect of the nasal cavity (Figure 1 ). In both sexes, the severity of this finding among individual ferrets varied from minimal to moderate. The incidence and mean severity scores of nasal turbinate COT among multiple studies over time are reported in Table 1 .
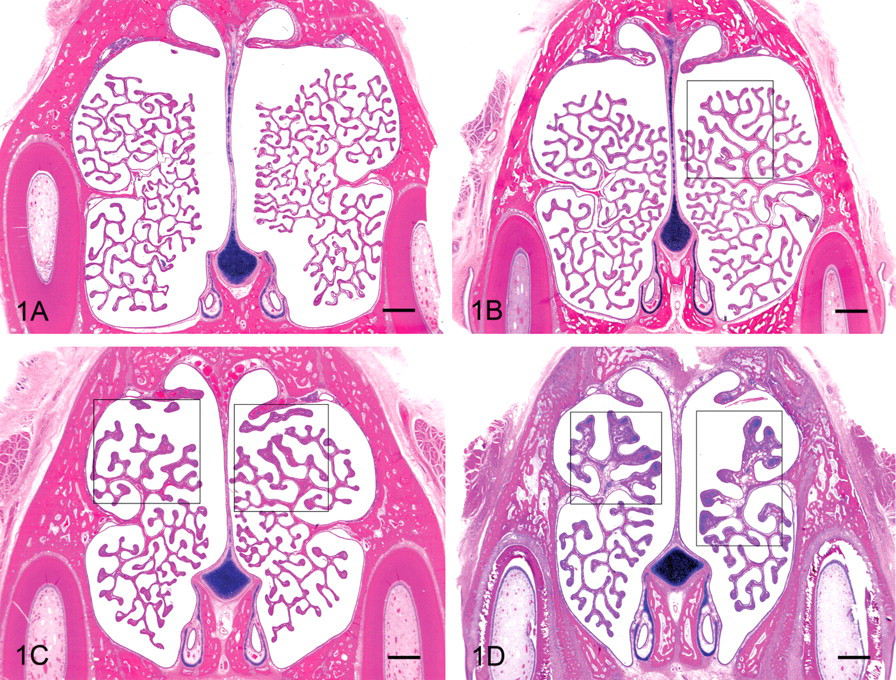
Nasal tubinates at Level 1. Turbinates of a Day 56 male (A) are within normal limits, a Day 31 female (B) has Grade 1 cartilaginous and osseous thickening (COT), a Day 31 male has Grade 2 (C), and a Day 3 male has Grade 3 (D). In all instances, COT was restricted to the dorsal turbinate scrolls. Note the assymmetry in the example of Grade 1 COT (boxed area) and the increased number of thin and fairly uniform scrolls in the older ferrets (A, B, and C) compared with the Day 3 ferret (D). Evidence that the sections represent the same level includes the uniform appearance of the vomeronasal organ (small, oval lumen surrounded by j-shaped cartilage), the lateral presence of teeth, and the single point at which the turbinate scrolls originate from the lateral wall of the nasal cavity. All figures hematoxylin and eosin, bar = 1 mm.
Incidence and mean severity of cartilaginous and osseous thickening and inflammation at Level 1 of the nasal turbinates over time among seven studies.
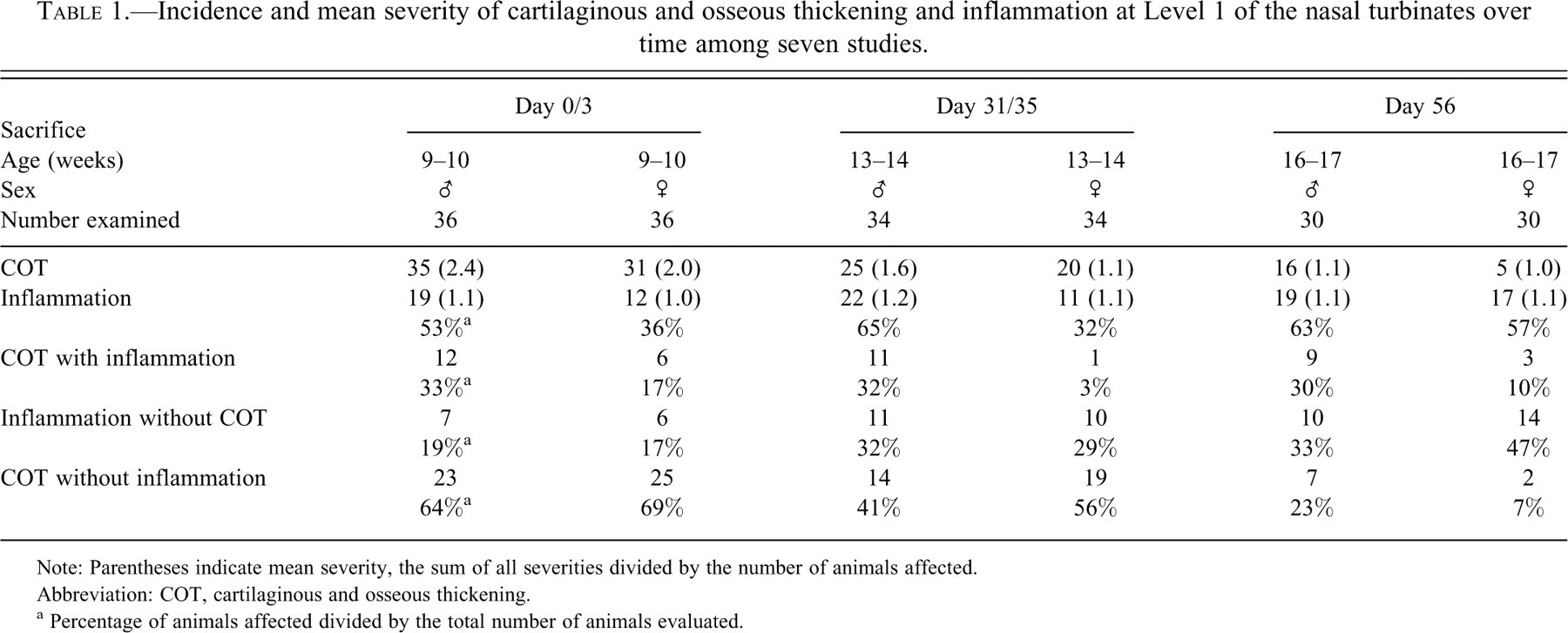
Note: Parentheses indicate mean severity, the sum of all severities divided by the number of animals affected.
Abbreviation: COT, cartilaginous and osseous thickening.
a Percentage of animals affected divided by the total number of animals evaluated.
By far, the highest prevalence and severity scores of COT for both males and females occurred at Day 0/3, and the lowest occurred at Day 56. At Day 56, females exhibited a greater decrease in COT prevalence as compared with males. Cartilaginous and osseous thickening presented as thickening of the turbinate scroll and was unilateral (minimal) in 42% of the evaluated animals, mostly at Day 56 (Table 2 ). An equal percentage of COT was at a slight/mild severity level and bilateral, mostly at the Day 0/3 sacrifice interval. The few moderate severity grades were primarily in males at Day 0/3. Focal knobby proliferation was primarily composed of cartilage; bone was present when the length of a turbinate scroll was more uniformly thickened. In a very few cases the proliferation appeared to form a bud, as if a new scroll were developing (Figure 2 ).
Severity grade occurrence of cartilaginous and osseous thickening at Level 1 of the nasal turbinates over time among seven studies.

Abbreviation: COT, cartilaginous and osseous thickening.
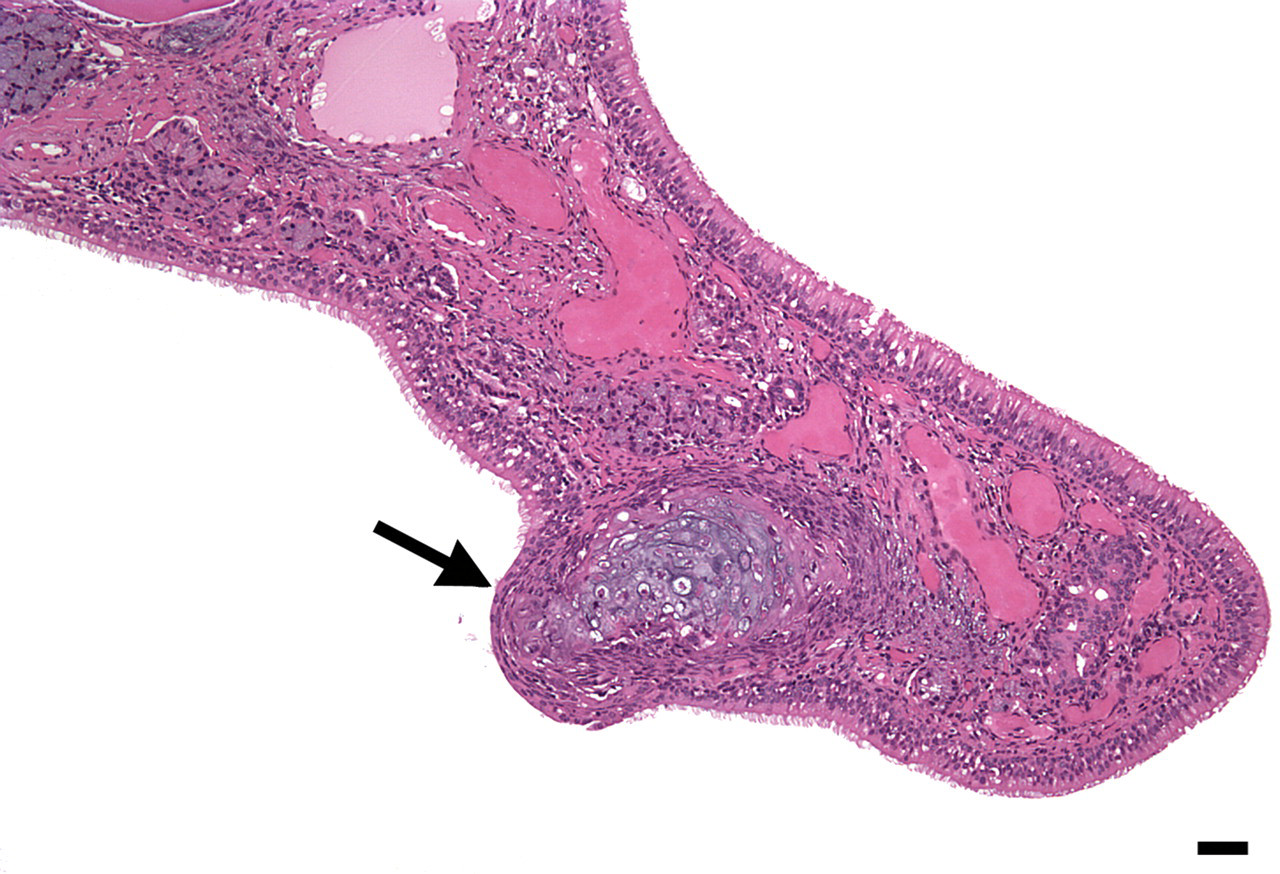
Day 3 female with cartilage and osseous bud (arrow), which may be the beginning of a new turbinate scroll. Hematoxylin and eosin, bar = 20 μm.
Roughly a third to over half of male and female ferrets at each sacrifice interval had generally minimal inflammation within the nasal cavity (Table 1). Inflammation at Day 0/3 consisted of neutrophils located within the lamina propria and less commonly within the epithelium of the turbinate scroll, lateral and/or ventral walls of the nasal cavity. Neutrophils occasionally composed an exudate in the meatus. At Days 31 and 56, neutrophils still predominated, but a few lymphocytes and macrophages were also occasionally observed in the lamina propria of the turbinate scrolls. Fibroplasia and edema were not associated with the inflammatory cell infiltration. Inflammatory cell distribution was not consistent, being dorsal or ventral, unilateral or bilateral, and occasionally in all locations. Inflammation occurred without concomitant COT at every sacrifice interval (19%–47%). Cartilaginous and osseous thickening and inflammation were diagnosed together in roughly a third of males at all sacrifice intervals and in 3%–17% of females. Females at Day 31 had the lowest concordance of COT with inflammation; only 3% had both diagnoses. Cartilaginous and osseous thickening without inflammation occurred in roughly half to two-thirds of evaluated ferret nasal cavities from most sacrifice intervals, although females at Day 56 had only 7% of COT incidences occurring without inflammation.
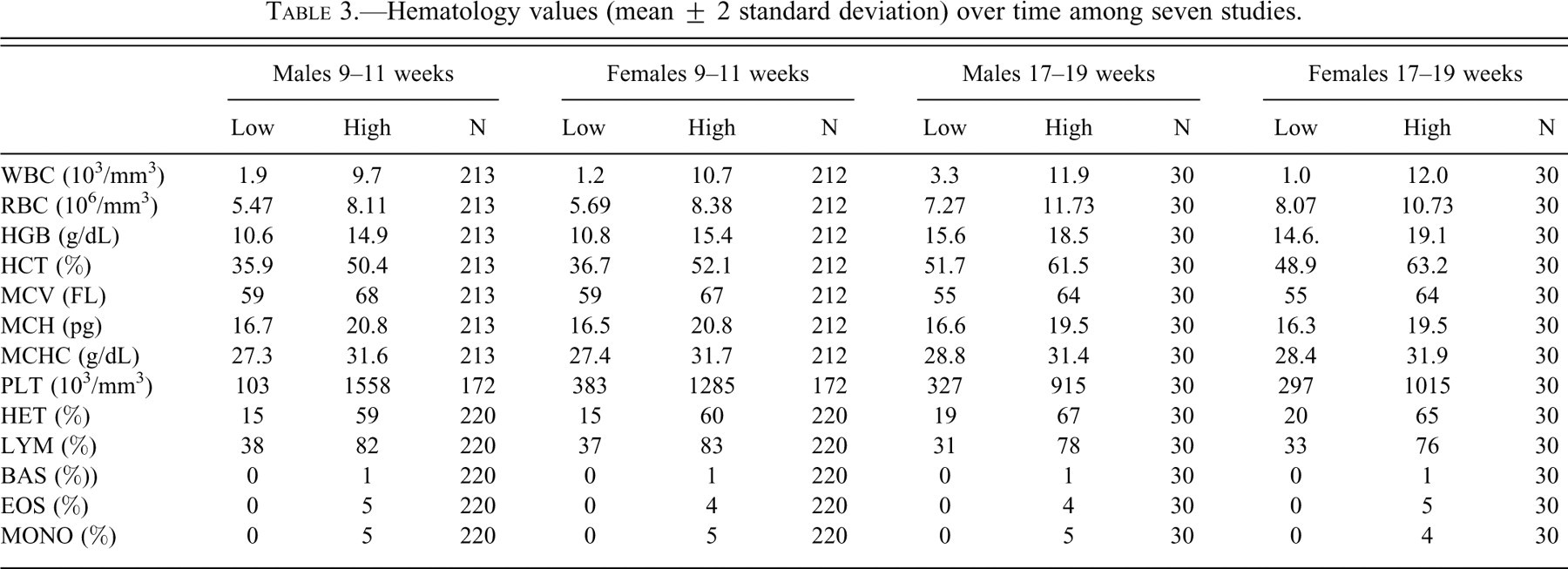
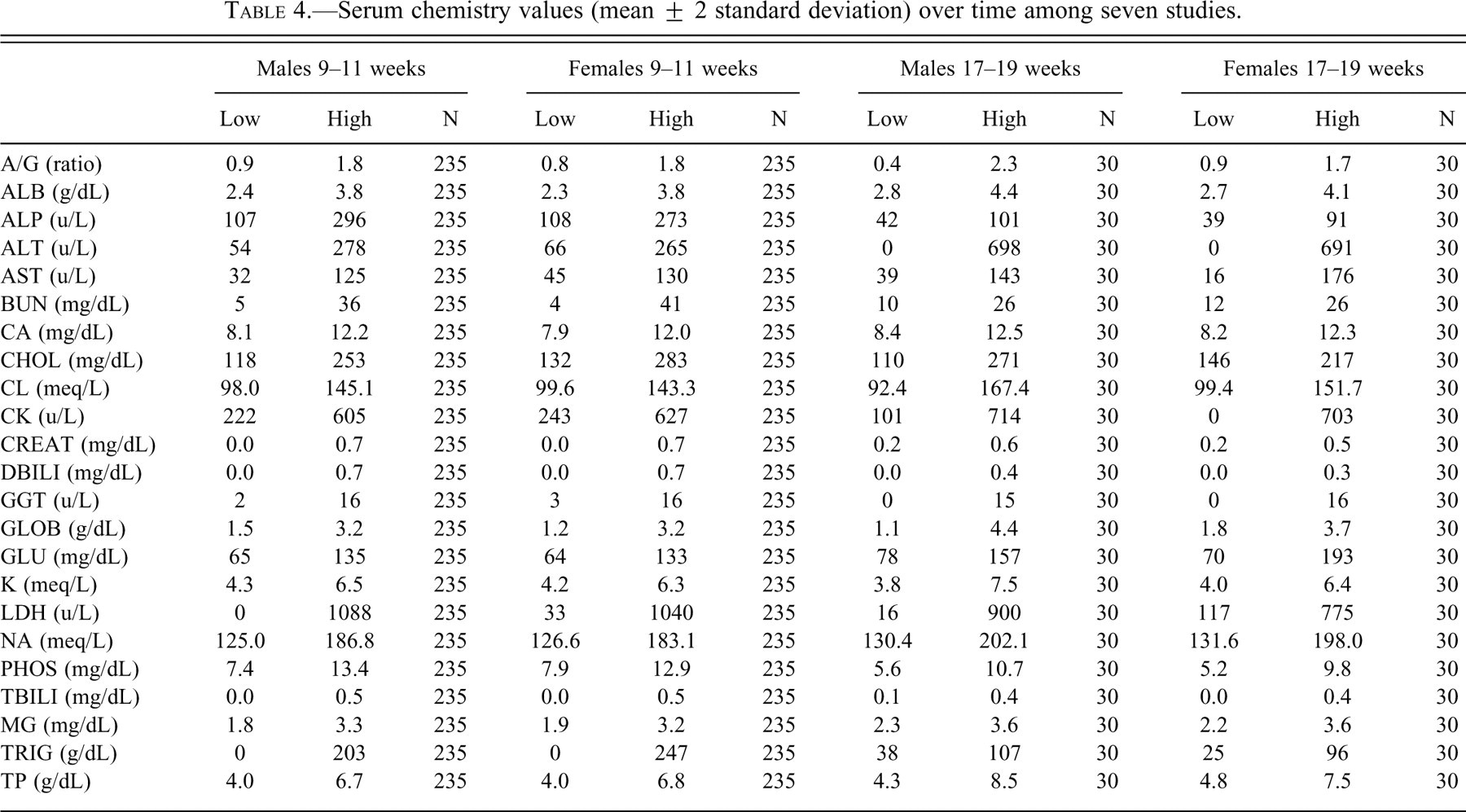
Hematologic and serum chemistry values were considered to be within expected limits for laboratory ferrets (Tables 3 and 4 ) (Ohwada and Katahira 1993).
Hematology values (mean ± 2 standard deviation) over time among seven studies.
Serum chemistry values (mean ± 2 standard deviation) over time among seven studies.
Discussion
Intranasal instillation of live attenuated influenza vaccine candidates, depending on the concentration and virulence of the virus, often resulted in acute to subacute inflammation in the nasal mucosa, characterized by infiltration of neutrophils and lymphocytes in the lamina propria. More severe responses had the addition of a predominantly neutrophil-rich exudate in the nasal passages. The association of these inflammatory responses with proliferation of cartilage and osseous tissue in the turbinates led to the consideration that inflammation may be the cause of COT. However, as shown in this study, the occurrence of COT was often independent of inflammation.
We did not examine nasal tissues from ferrets that had not been injected with PBS as controls. Since the PBS-inoculated ferrets had little or no inflammation in the lamina propria, and rare instances of inflammatory exudate, we believe PBS-inoculated ferrets have nasal tissues comparable to those of noninjected ferrets.
Additionally, the PBS-inoculated ferrets were determined by antibody titer serology to be free of vaccine antigens prior to the start of the studies and at necropsy. To prevent inadvertent virus antigen exposure, the control ferrets were handled prior to treated animals during dosing procedures and returned to the microisolator housing.
Although the scientific literature contains a number of articles pertaining to early craniofacial growth in ferrets and other laboratory animals (He 2004; He and Kiliardis 2003; He and Kiliardis 2004; Losken et al. 1994), information that specifically concerns nasal turbinate development appears to be limited. Because the incidence and severity of COT tended to wane with age in our study, it is presumed that this finding represents a previously unrecognized, age-related, regional pattern of “normal” nasal turbinate growth and expansion. To some degree, this idea is supported by the observation that in Day 8–11 week (Day 0/3) ferrets, areas of cartilage and bone budding were occasionally evident in the dorsal nasal turbinates. Additional support is provided by evidence that COT appeared to resolve more rapidly with age in females as compared with males. This finding is consistent with an earlier report, which indicated that craniofacial growth in ferrets ceases earlier in females than males (He and Kiliaridis 2004).
Also, craniofacial growth in ferrets and rodents is at least partially dependent on masticatory function and diet (Abed et al. 2007; He 2004). Although it is possible that dietary differences (e.g., hard pellets versus soft feed) may have resulted in a pattern of nasal turbinate development that differed from previous studies, this possibility seems unlikely, because ferrets in the studies described here were fed a standard diet of certified Harlan Teklab Ferret Diet #2072C.
Congenital abnormalities and hereditary chondro-osseus dysplasia were considered to be improbable explanations based on the percentage of ferrets affected, the lack of functional consequence, and the dissipation of COT with increasing age. Metabolic conditions that may result in cartilaginous and/or osseous thickening include nutritional, renal, endocrine, or neoplastic conditions. None of these potential etiologies were supported by additional tissue changes in affected ferrets (e.g., sternal and femoral bones were normal), and serum chemistry results did not indicate evidence of endocrine or renal disease.
The most likely explanation is that COT is a function of normal growth and expansion of ferret nasal turbinates. As such, the recording of COT as a diagnosis in toxicity bioassays is discouraged. If this finding is diagnosed, we recommend that terminology implying a pathologic condition or process (e.g., hyperostosis or turbinate hypertrophy) not be used.
Footnotes
Acknowledgment
The authors wish to thank Dr. Jeff Wolf for his invaluable editorial and pictorial help.
