Abstract
An eosinophilic substance (ES) is usually observed in the mouse nasal septum. In contrast to textbooks and one report describing ES as amyloid, a previous study by the authors revealed that ES is not amyloid but consists of collagen and an amorphous material. Furthermore, it was suggested that the amorphous material was produced by clear HE-stained nasal gland epithelial cells present at the dorsal portion directly above the vomeronasal organ. In this histological examination, ES deposition showed sex difference (more intense in males than in females). ES increased with age but not in seniles, suggesting that the increase has a limit. In the detailed examination using subserial HE-stained nasal sections, it was revealed that the clear HE-stained nasal glands continued to the vomeronasal glands, which communicated with the lumen of the vomeronasal organ, and the vomeronasal gland epithelial cells contained strongly periodic acid-Schiff (PAS) positive granules, similar to the clear HE-stained nasal gland epithelial cells. ES also deposited in the interstitium of the vomeronasal glands. The results suggested a possibility that ES deposition may be related to vomeronasal organ.
Keywords
Introduction
An eosinophilic substance (ES) is usually observed in the mouse nasal septum. ES has been described as amyloid in textbooks (Herbert and Leininger 1999; Leininger, Herbert, and Morgan 1996) and one report (Engelhardt, Gries, and Long 1993), but a few reports have indicated that ES is not amyloid (Haines, Chattopadhyay, and Ward 2001; Monticello, Morgan, and Uraih 1990). Our previous report revealed that ES is not amyloid, because it reacted negatively to Congo red and there were no nonbranching fibrils in the electron microscope examination, and that ES consists of collagen and an amorphous material (Doi et al. 2007).
In our previous report, two kinds of glands were observed in the nasal septum in mice (Doi et al. 2007). One of them stained clear with hematoxylin and eosin (HE), the other stained dark with HE. ES deposited at the interstitium of the clear HE-stained nasal glands only. Additionally, electron microscopic findings suggested that the amorphous material, which is a major component of ES, was produced by the clear HE-stained nasal gland epithelial cells and migrated to the interstitium through the partial opening of the basement membrane (Doi et al. 2007, 2009).
The amorphous material is probably produced physiologically, because ES was found in all mice (Doi et al. 2007; Engelhardt, Gries, and Long 1993; Leininger, Herbert, and Morgan 1996), and neither nasal gland degeneration nor inflammation occurred even when the grade of ES deposition was enhanced (Doi et al. 2007), but the significance of ES is not clear. The clear HE-stained nasal gland and ES were present at the ventral nasal septum only, at the dorsal portion directly above the vomeronasal organ (Doi et al. 2007). Because of their position, we speculated that ES may be related to the vomeronasal organ. As the vomeronasal organ is known for chemoreception of pheromone-mediated behavior, we investigated the relation between ES deposition and sex and between ES deposition and age. In addition, a detailed examination using subserial HE-stained nasal sections was done to verify continuity between the clear HE-stained nasal glands and the vomeronasal organ.
Materials and Methods
The animals were 5-, 34-, 58-, 84-, and 110-week-old B6C3F1/Crlj mice and were orally administered with methylcellulose solution in a study to collect house data. They were purchased at 5 weeks of age from Charles River Laboratories Japan Inc. (Kanagawa, Japan). Their microbial level was SPF (specific pathogen free). They were housed individually in cages in an animal room maintained at a temperature of 22 ± 3°C, humidity of 55 ± 20%, with 6 to 20 air changes per hour, and a 12-hour light/dark cycle. They were given a commercial diet (CE-2, CLEA Japan Inc.) and tap water ad libitum. No abnormalities were observed in the clinical signs during the observation period. The antibody test results for Clostridium piliforme, Ectromelia virus, LCM virus, Mouse hepatitis virus, Mycoplasma pulmonis, and Sendai virus were negative in monitor mice, which were housed in the same room as the investigated mice. Antibody tests for pathogens including Pneumonia virus of mice, Minute virus of mice, Mouse parvovirus, and Helicobacter spp. were not done.
The mice were sacrificed at 5, 34, 58, 84, or 110 weeks of age by exsanguination under anesthesia with intraperitoneal injection of sodium thiopental (5 males and 5 females of each age). Their nasal tissues were removed, fixed in 10% neutral phosphate-buffered formalin, decalcified with 10% formic acid formalin for 10 days, embedded in paraffin, sectioned, and stained with HE for microscopic examination. We used the level II specimens (taken through the level of the incisive papilla, where ES and two kinds of nasal glands are abundant) in this investigation. For detailed examination, subserial nasal specimens of 110-week-old mice (1 male and 1 female) were prepared at 20-μm intervals in the same manner, from the anterior part of the upper incisor teeth (in front of the level I line) to the level III line. The sections were subjected to HE staining and periodic acid-Schiff (PAS) reaction with prior diastase treatment.
The animals were cared for according to the principles outlined in the guides for care and use of laboratory animals prepared by the Japanese Association for Laboratory Animal Science and our laboratory.
Results
In the histological examination conducted in 5-, 34-, 58-, 84-, and 110-week-old B6C3F1/Crlj mice, ES was observed in all mice, and the grades of ES deposition were comparable among mice of the same sex and same age. The grades of ES deposition were more enhanced in males than in females when they were the same age, except that both sexes of 5-week-old mice showed very little deposition (Figures 1A-1J). The grade of ES deposition increased with age, but plateaued by 58 weeks of age in males and by 34 weeks of age in females (Figures 1A-1J). Age- or sex-related difference concerning the distribution and amount of the nasal glands was not evident. Neither nasal gland degeneration nor inflammation was observed at any age, although a separation of the nasal glands and expansion of the septum width were observed, especially in 58-, 84-, and 110-week-old male mice.
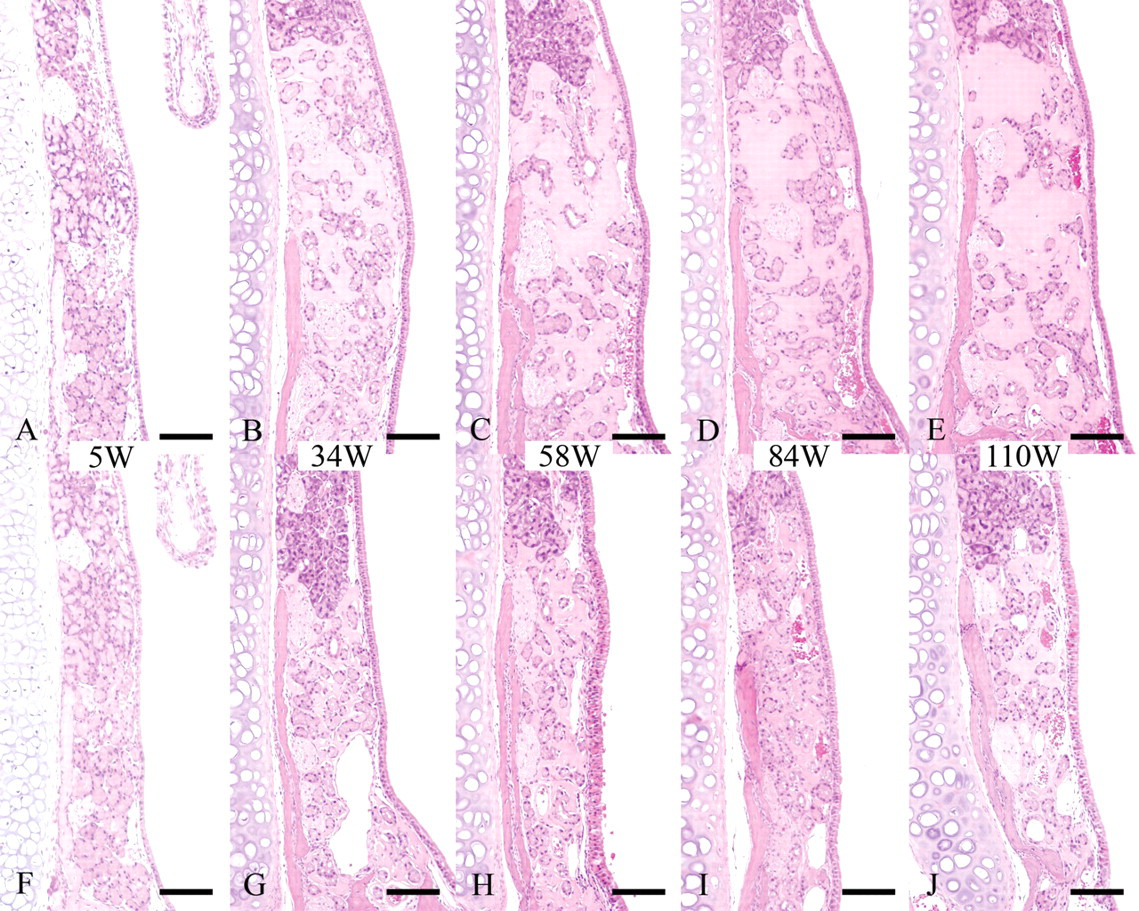
Nasal cavity (level II); B6C3F1/Crlj mice. HE.
In the histological examination using the subserial specimens, clear HE-stained nasal gland area with ES deposition was present only at the dorsal portion directly above the vomeronasal organ of level I and II nasal septum (Figures 2A, 2B). Neither clear HE-stained nasal gland nor ES was observed in level III nasal septum where vomeronasal organ was not observed in the nasal septum (Figure 2C). These findings suggested a relation between the clear HE-stained nasal gland and the vomeronasal organ. The vomeronasal glands were observed around the vomeronasal organ (Figures 3A-3D) as well as directly behind the organ (Figure 3E) and communicated with the lumen of the vomeronasal organ (Figure 3C). Although the vomeronasal glands in front of the level I line were separated from the clear HE-stained nasal glands by the bone (Figure 3A), the border bone disappeared between the glands in the sections behind the level I line (Figures 3B, 3D, 3E). The histological figures of the clear HE-stained vomeronasal glands were very similar to those of the clear HE-stained nasal glands (Figures 3A-3D). Additionally, ES also deposited in the interstitium of the vomeronasal glands (Figures 3A-3E). The vomeronasal gland epithelial cells contained strongly PAS positive granules, and ES in the interstitium was stained positively with PAS (Figure 4), similarly to the clear HE-stained nasal glands observed in our previous study (Doi et al. 2007).
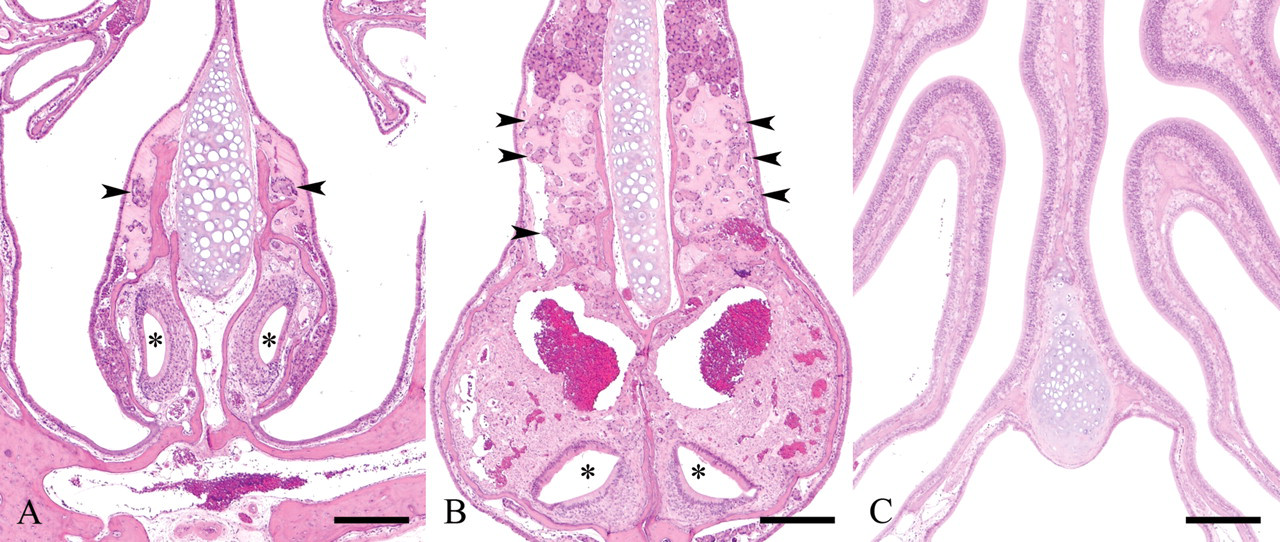
Nasal cavity; 110-week-old B6C3F1/Crlj mice. HE.
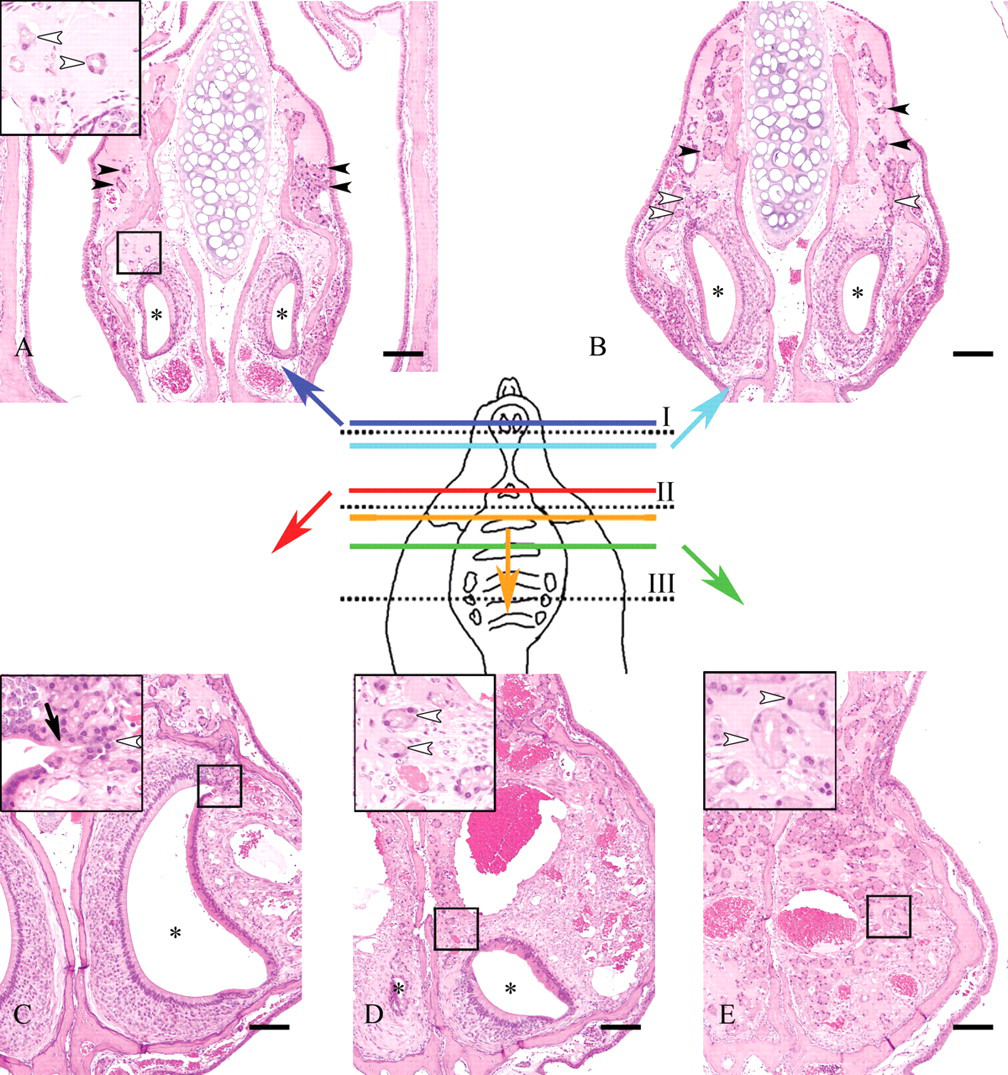
Nasal cavity; 110-week-old B6C3F1/Crlj mice. Subserial nasal specimens. HE. Bar = 50 μm.
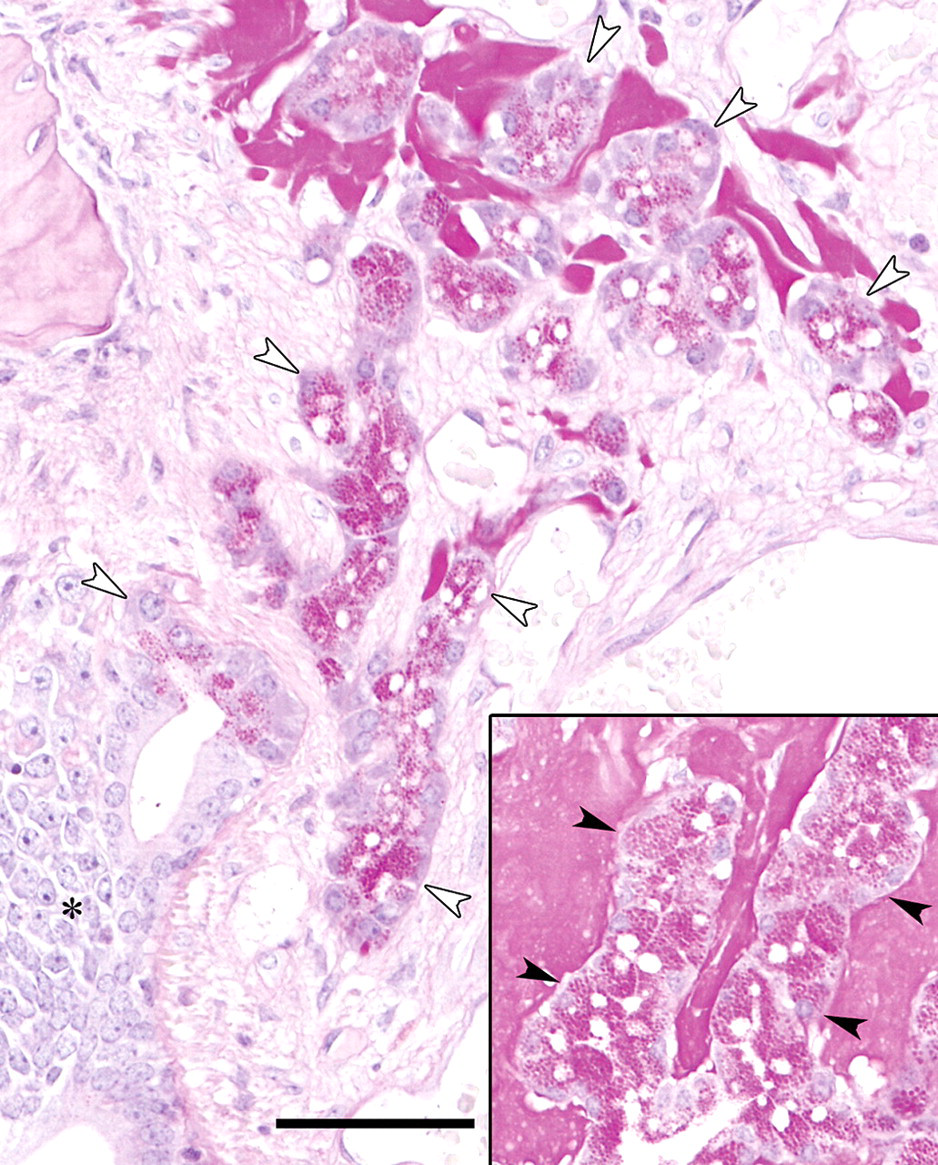
Nasal cavity; 110-week-old B6C3F1/Crlj mice. PAS. Bar = 100 μm. Vomeronasal gland epithelial cells (white arrowheads) contain strongly PAS-positive granules, similarly to the clear HE-stained nasal gland epithelial cells (black arrowheads in inset). Eosinophilic substance stains positively with PAS reaction. The asterisk shows the vomeronasal organ.
Discussion
Concerning ES, two new findings were revealed in this study: (1) ES deposited more intensely in males than in females when the age was the same, (2) the grade of ES deposition plateaued by 58 weeks of age in males and by 34 weeks of age in females. These did not occur by chance, because the grades of ES deposition were comparable in mice of the same sex and age. According to textbooks (Leininger, Herbert, and Morgan 1996) and our previous report (Doi et al. 2007), the grade of ES deposition increases with age. In this study, however, ES did not increase in seniles, suggesting that its increase has a limit. Additionally, ES deposition has been reported in mice only (Herbert and Leininger 1999; Leininger, Herbert, and Morgan 1996; Engelhardt, Gries, and Long 1993; Haines, Chattopadhyay, and Ward 2001; Monticello, Morgan, and Uraih 1990; Doi et al. 2007). We have examined the nasal septum of rats, guinea pigs, dogs, marmosets, and cynomolgus monkeys, but no ES has been observed in these animals (data not shown).
The vomeronasal glands are synonymous with Jacobson’s glands (Taniguchi and Mochizuki 1983) and communicate with the lumen of the vomeronasal organ. In this study, the vomeronasal glands had several similarities to the clear HE-stained nasal glands such as staining properties (stained clearly with HE) and ES (stained positively with PAS reaction) deposition in their interstitium. Moreover, the clear HE-stained nasal glands were continuous with the vomeronasal glands. Additionally, strongly PAS positive granules in the vomeronasal gland epithelial cells were very similar to those in the clear HE-stained nasal gland epithelial cells found in our previous study (Doi et al. 2007). Cuschieri and Bannister (1974) reported that “the posterior glands of the nasal septum,” which reacted strongly positive to PAS reaction, situated in the posterior half of the septum ventral to the junction between the respiratory and olfactory epithelia, and most of the ducts of these glands opened into the cavity of the vomeronasal organ. It was thought that the clear HE-stained nasal glands and the vomeronasal glands found in this investigation were “the posterior glands of the nasal septum” reported by Cuschieri and Bannister. Hence, it was strongly suggested that most of the clear HE-stained nasal glands were the vomeronasal glands.
The physiological significance of ES is not clear. However, it was suggested that ES would deposit in association with the vomeronasal glands because ES deposited in the interstitium of the vomeronasal glands only and a major component of ES was produced by the vomeronasal gland epithelial cells (Doi et al. 2007, 2009). Although descriptions concerning the vomeronasal glands are few, it was reported that the glands empty their secretions into the vomeronasal organ’s lumen for dissolving nonvolatile chemical stimuli to facilitate reception of chemical stimuli and then purge secretions (Roslinski et al. 2000). However, as ES was not secreted into the vomeronasal gland’s lumen, it was not suggested that ES has any effects on the vomeronasal organ’s function.
As another possibility, it was thought that ES may deposit to fill the interspaces of the nasal glands occurring with the body growth, because age- or sex-related difference concerning the distribution and amount of the nasal glands was not obvious. Based on house data of B6C3F1/Crlj mice in our laboratory, the males were heavier than the females when the age was the same, corresponding with the result that ES deposited more intensely in males than in females. However, the body weight of the males plateaued more quickly than that of the females (approximately at 48 weeks in males and 66 weeks in females), which was the same content as described in a textbook (Rao 1999). This did not correspond with the result that the grade of ES deposition of the females plateaued more quickly than that of the males. Additionally, the following findings also suggested that ES deposition would occur with not only body growth: (1) ES deposited in the interstitium of the vomeronasal glands only, (2) ES deposited with expansion of the septum width, and (3) ES deposition has been reported in mice only (Herbert and Leininger 1999; Leininger, Herbert, and Morgan 1996; Engelhardt, Gries, and Long 1993; Haines, Chattopadhyay, and Ward 2001; Monticello, Morgan, and Uraih 1990; Doi et al. 2007).
In conclusion, two pieces of evidence concerning ES deposition were revealed in this study: sex difference and no increase in seniles. Additionally, it was reaffirmed that most of the clear HE-stained nasal glands are the vomeronasal glands. Based on these findings and previous reports, we suggest the possibility of a relation between ES in the mouse nasal septum and the vomeronasal organ. Further investigation is necessary to clarify the significance of ES and its relationship to pheromones because the vomeronasal organ is known for chemoreception of pheromone-mediated behavior.
Footnotes
Acknowledgments
We thank Mr. Takayoshi Ito and Mr. Yuki Ikeda for their excellent technical assistance. We also thank Mr. Steve Yamakami and Ms. Ai Ishii for language editing.
