Abstract
Spontaneous salivary gland tumors in rats are rare. The authors report a poorly differentiated carcinoma of a submandibular gland in a ten-week-old rat that was positive for vimentin. Microscopically, the neoplastic cells showed a diffuse growth pattern in most areas of the tumor mass and a nestlike structure in a part of the peripheral area. Immunohistochemically, the cells were positive for keratin and vimentin but not for α-smooth muscle actin. Ultrastructurally, desmosome-like structures were observed. Based on these findings, the tumor was diagnosed as a poorly differentiated carcinoma. The origin of the neoplastic cells would be either acinar or ductal cells. This suggests that acinar or ductal cells have the potential to transform into vimentin-expressing cells.
Introduction
Spontaneous epithelial tumors of salivary glands in rats are rare (Neuenschwander and Elewell 1990; Elwell et al. 1990). So far, several cases in rodents have been reported: epithelial neoplasm in a Wistar rat (Sasaki 1982); adenocarcinoma in a rnu/rnu nude rat (Halter, Wetherall, and Holscher 1983); pleomorphic adenomas in a Sprague-Dawley rat (Okada et al. 1985), a B6C3F1 mouse (Hagiwara et al. 1993), and prairie dogs (Ozaki and Narama 2003); poorly differentiated carcinoma in a Sprague-Dawley rat (Tsunenari et al. 1997); and papillary cystadenocarcinoma in a Fischer 344 rat (Hosokawa et al. 2000). In these reports, the tumor cells had obvious epithelial features (epithelial structures, ductal structures, cysts with luminal papillae, squamous cell differentiation, or positive reaction for pankeratin). Some of these tumors were examined immunohistochemically and were negative for vimentin, except the tumors derived from myoepithelial cells.
In the present case, although the neoplastic cells had epithelial characteristics, they were positive for vimentin and negative for α-smooth muscle actin (SMA). We describe the histopathological characteristics of this tumor in this report.
Case Report
All procedures were performed according to the Rules for Feeding and Storage of Experimental Animals and Animal Experiments and were approved by the Institutional Animal Care and Use Committee of the testing facility. The animal was a ten-week-old male Sprague-Dawley rat (Crl:CD(SD); Charles River Japan Inc., Kanagawa, Japan). It was used as a control animal for a repeated-dose toxicity study and was submitted for sacrifice at the end of the study. The animal was housed in a wire mesh cage in an animal room under controlled conditions (temperature of 23 ± 3°C, relative humidity of 50 ± 20%) and fed CRF-1 (Oriental Yeast Co., Ltd., Tokyo, Japan) with sterilized tap water provided ad libitum. At the end of the study, the animal was euthanized by exsanguination via the abdominal artery under thiopental anesthesia.
The animal showed no abnormalities with regard to clinical signs, body weight, hematology, or blood chemistry.
At necropsy, a 15 × 10 mm–diameter mass was found in the cervical area in which normal submandibular and sublingual glands were not identified. The normal parotid gland was present. The cut surface of the mass was whitish, and one large cyst was observed at its center. The mass and other organs (including submandibular gland, sublingual gland, parotid glands, and submandibular lymph node) were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 4-μm thickness, stained with hematoxylin and eosin, and examined microscopically. Sections for the mass were immunohistochemically stained using antibodies against keratin (undiluted; DakoCytomation, Glostrup, Denmark), CK19 (1:20 dilution; Novus Biologicals, Littleton, CO, USA), vimentin (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA), S-100 (1:200; DakoCytomation), SMA (undiluted; DakoCytomation), proliferating cell nuclear antigen (PCNA; 1:300; DakoCytomation), glial fibrillary acidic protein (GFAP; 1:500; DakoCytomation), and Ki-67 (1:200; DakoCytomation).
Microscopically, the mass was relatively well demarcated from the adjacent areas (Figure 1A, B ), but partial invasion into the adjacent tissue was observed (Figure 1C). Compressions of the residual submandibular gland (normal part) or metastases to other organs, including the submandibular lymph node, were not observed. The majority of the mass consisted of a diffuse sheet of epithelial cells without any acinar or ductal structure (Figure 1D). However, some of the peripheral cells were arranged in a nestlike structure (Figure 1E). In both areas, the neoplastic cells had eosinophilic cytoplasm, spherical nuclei, and prominent nucleoli (Figure 1D). Mitotic figures were frequently observed. Cell debris, slight infiltration of neutrophils, and vascular development were present throughout the mass.
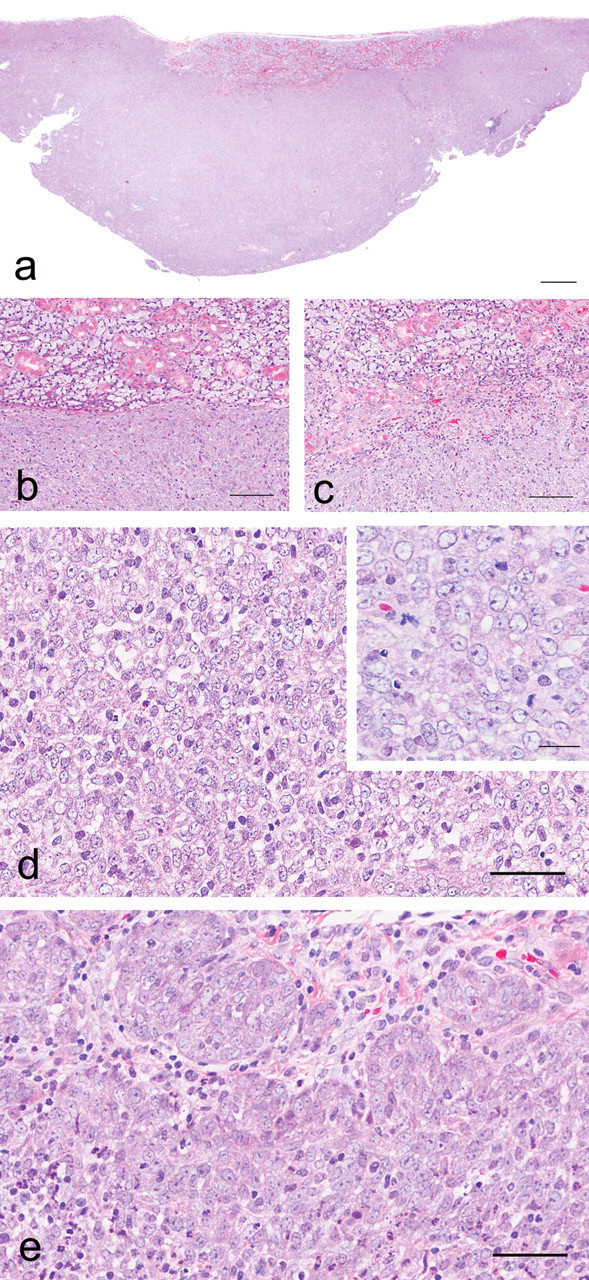
Histological appearance of the tumor. Hematoxylin and eosin staining (A–E). The mass was relatively well demarcated from the adjacent area by a thin fibrous capsule (A, B). However, a slight invasion was observed (C). The majority of the tumor cells had a diffuse sheet structure (D). Some tumor cells were arranged in a nestlike structure (E). The neoplastic cells had eosinophilic cytoplasm, spherical nuclei, and prominent nucleoli (insert in d). Bars: (A) 500 μm, (B, C) 100 μm, (D, E) 50 μm, (insert in D) 20 μm.
Immunoreactivities for the mass and the normal part are summarized in Table 1 . The neoplastic cells in the diffuse sheet area and nestlike area were stained positive for vimentin (Figure 2A, B ). In the normal part, mesenchymal and myoepithelial cells were positive for vimentin. The neoplastic cells in both the diffuse sheet and nestlike areas (Figure 2C, D) as well as myoepithelial cells, intercalated ductal cells, and striated ductal cells in the normal part were positive for keratin. Most of the neoplastic cells were positive for PCNA and Ki-67. The neoplastic cells in the mass were negative for CK19, S-100, SMA, and GFAP.
Results of immunohistochemical staining.
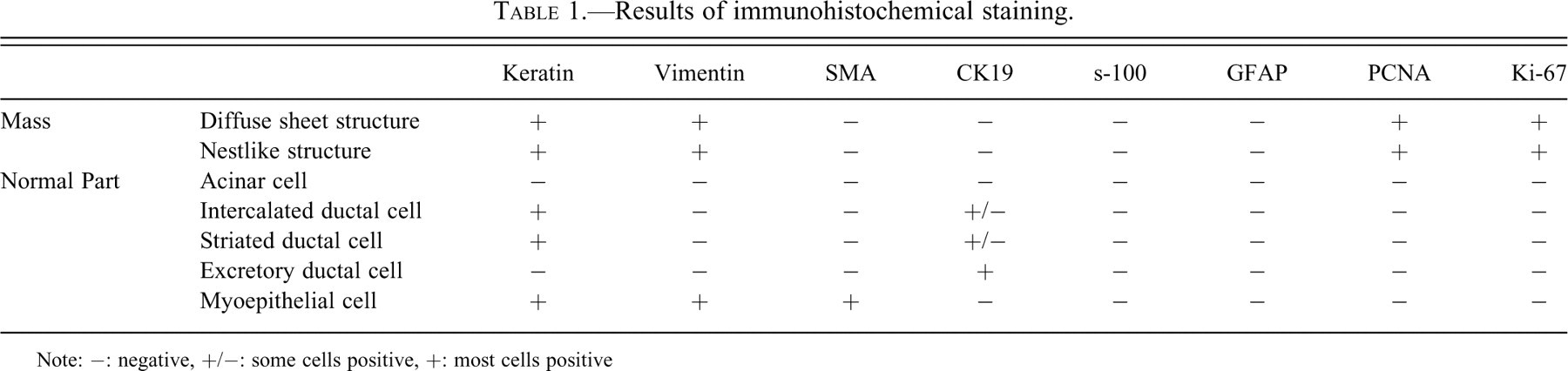
Note: −: negative, +/−: some cells positive, +: most cells positive
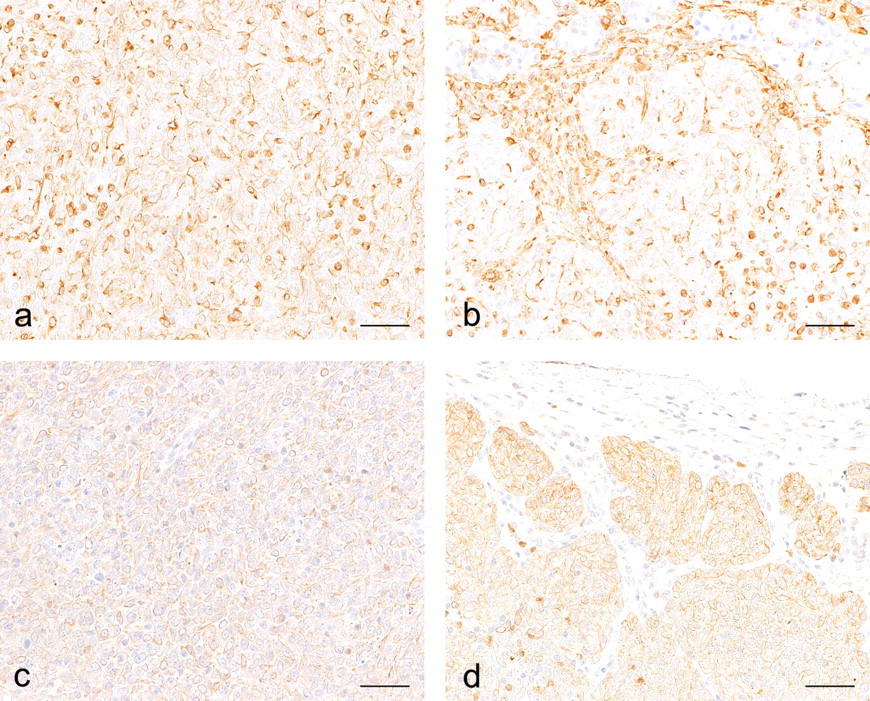
Immunostaining for vimentin (A, B) and keratin (C, D). Neoplastic cells having a diffuse sheet structure were positive for vimentin (A) and keratin (C). Cells in a nestlike structure were also positive for vimentin (B) and keratin (D). Bar: 50 μm (A–D).
Ultrastructurally, desmosome-like structures were observed in the mass. However, microfilaments with dense bodies, which are often seen in myoepithelial cells, were not observed (Figure 3 ).
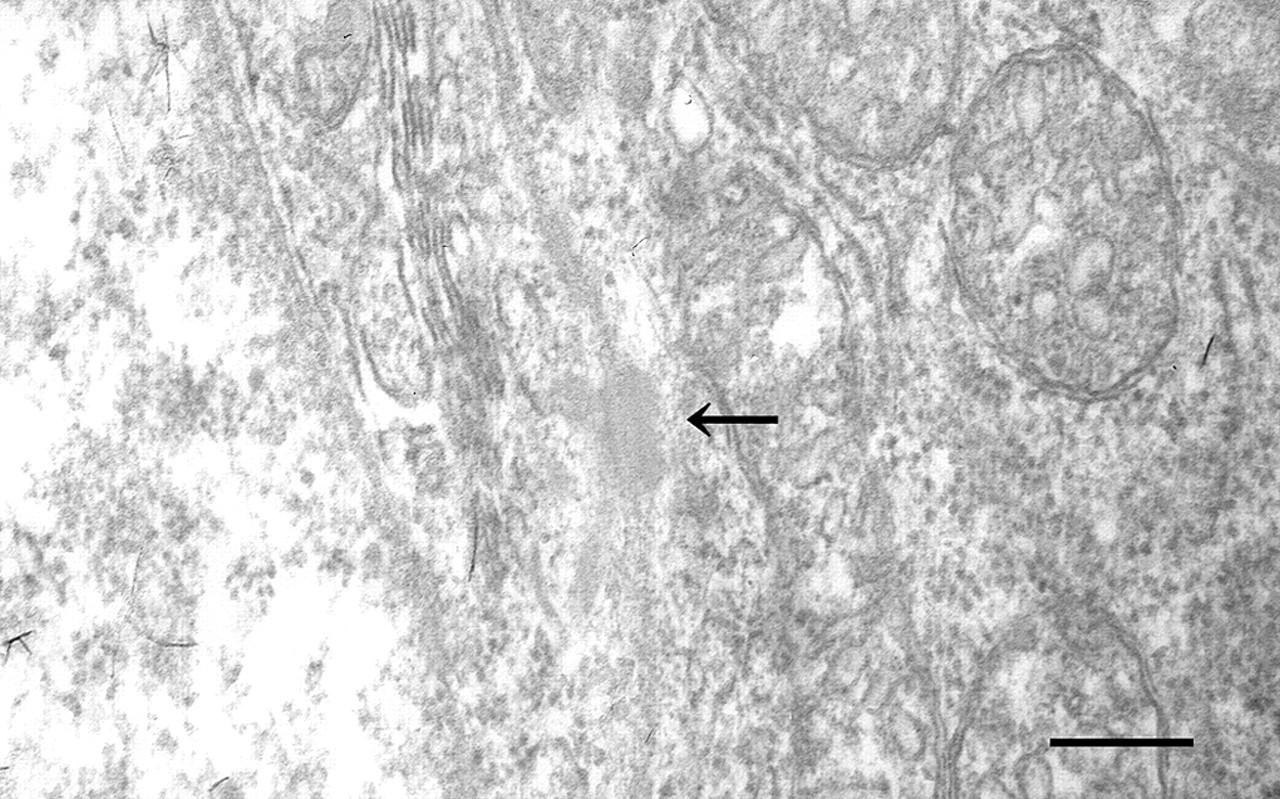
Electron micrograph of the mass. A desmosome-like structure (arrow) was found between the neoplastic cells. Bar: 250 Nm.
The lack of any clear acinar or ductal structure indicated that the tumor was undifferentiated. The keratin expression, nestlike structure and presence of a desmosome-like structure are characteristics of an epithelial tumor. Therefore, we diagnosed this case as a poorly differentiated carcinoma of a submandibular gland.
In this case, the neoplastic cells were positive for keratin and negative for SMA, suggesting that the origins for the cells of this tumor would be acinar or ductal cells but not myoepithelial cells. In humans, some tumors derived from nonmyoepithelial cells are positive for both keratin and vimentin but not for SMA. Examples include plasmacytoid cells in pleomorphic adenoma and myoepithelioma, basal cell adenoma, and polymorphous low-grade adenocarcinoma (PLGA; de Araujo et al. 2000). De Araujo, Sousa, and Jaeger (1999) reported that PLGA originated from cells located at the acinar-intercalated duct junction. Ogawa et al. (2003) reported that the plasmacytoid cells in a pleomorphic adenoma originated from luminal cells.
Based on these considerations, acinar and ductal cells may have the potential to express vimentin, although normal acinar and ductal cells are negative for vimentin (Ogawa et al. 1999). Until now, there has been no report of a spontaneous tumor derived from acinar or ductal cells of a salivary gland of a rodent that was positive for both vimentin and keratin. This case raises the possibility that acinar or ductal cells of the salivary glands could be transformed into vimentin-expressing cells during canceration.
Footnotes
Acknowledgments
The authors would like to thank Ms. Hiroko Hirano, Mr. Katsuya Fujiki, and Mr. Kennichi Ohta for skillful technical assistance.
