Abstract
Histological examination of the heart of a clinically normal, 10-week-old female Han Wistar rat revealed a 600 × 400-μm mass of ectopic thyroid tissue within the subendothelial connective tissue of the aortic valve. The mass protruded into the left ventricular lumen and was composed of single layers of cuboidal to low-columnar epithelium organized into follicles often containing colloid. Parafollicular cells were not evident. To the authors' knowledge, this is the first report of intracardiac ectopic thyroid gland in the rat and the first report of ectopic thyroid within a heart valve in a mammal.
Ectopic thyroid tissue is a frequent, congenital abnormality in domestic mammals, reported in approximately 50% adult dogs, 10% adult cats, 10% adult humans, and 0.3% BALB/c mice (Hunt 1963; Naan et al. 2006; Capen 2007). It is reported in the rat, although there are no data available on the frequency with which it occurs (Anderson and Capen 1978).
Thyroid ectopia may occur at any location between the tongue and diaphragm, particularly within the mediastinum at the heart base, the ventral wall of the pharynx, the base of the tongue, and along remnants of the thyroglossal duct. Prevalent locations are species-specific (Hunt 1963; Sauk 1970; Naan et al. 2006; Capen 2007). In the dog, neoplastic transformation of ectopic thyroid tissue is seen relatively commonly, with ectopic thyroid neoplasms accounting for 1–10% of all heart base tumors (Capen 1978; Bracha et al. 2009).
Intracardiac thyroid tissue is rare in all mammalian species. Only 21 cases are reported in humans, with the majority of cases involving middle-aged women (Casanova et al. 2000; Wu, Zhou, and Wang 2009). In all of the cases described, the ectopic tissue was located within the interventricular septum, often protruding into the right ventricle and affecting the right ventricular outflow tract (Casanova et al. 2000). In the dog, despite the prevalence of mediastinal and intrapericardial (heart base) ectopic thyroid tissue, nonneoplastic ectopic thyroid has not been reported within the heart itself. Three cases of intracardiac, thyroid neoplasia, thought to have arisen from ectopic thyroid tissue, have been described in the dog (Richmond et al. 1990; Almes, Heaney, and Andrews 2008; Bracha et al. 2009).
To the best of our knowledge, intracardiac ectopic thyroid tissue has not been reported in the rat or mouse, and ectopic thyroid tissue within the heart valves has not been reported in any mammalian species.
Case Report
A ten-week-old, clinically normal, female Han Wistar rat was sacrificed at the conclusion of a seven-day, repeat-dose oral toxicity study, carried out in accordance with U.K. Home Office legislation (Animals [Scientific Procedures] Act 1986) and AstraZeneca’s institutional policies.
No gross abnormalities were detected at necropsy examination. Tissues were fixed in 10% buffered formalin, processed and paraffin embedded before staining with hematoxylin and eosin (HE). The heart was fixed whole and longitudinal; 5 μm step sections were cut every 100 μm to identify all four heart valves.
Histologically, within one section, the aortic valve protruded into the left ventricular lumen. The valve was expanded by a discrete, 600 × 400-μm, subendocardial, unencapsulated accumulation of well-differentiated thyroid tissue, embedded in a fine fibrous stroma and overlain by normal endothelium (Figure 1 ). Centrally, cuboidal to low columnar, multifocally attenuated epithelium was arranged in single layers, forming follicles filled with eosinophilic homogeneous material (colloid—Figure 2 ), whilst more peripherally cells were arranged in solid, radial nests. Epithelial cells had indistinct cell borders with moderate amounts of pale eosinophilic, finely granular cytoplasm and round, central to basally located nuclei. Parafollicular cells were not evident.
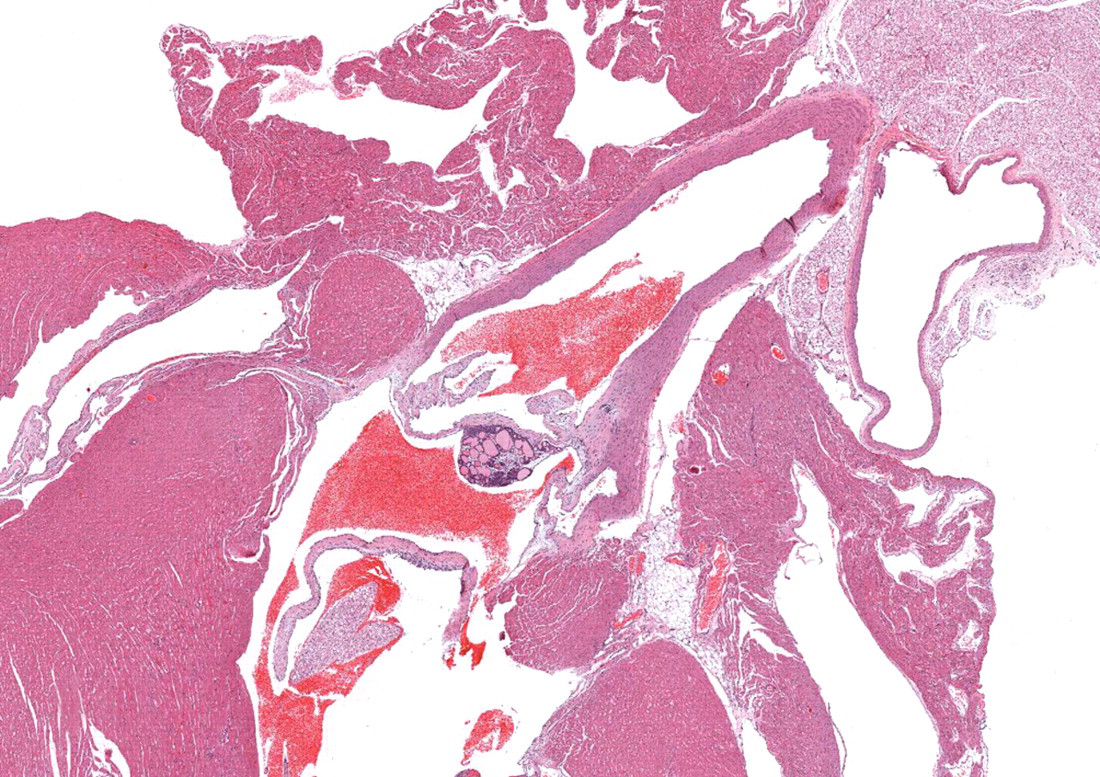
Left ventricular outflow tract. The aortic valve is expanded by a 600 × 400-μm mass of ectopic thyroid tissue. 20× (HE).
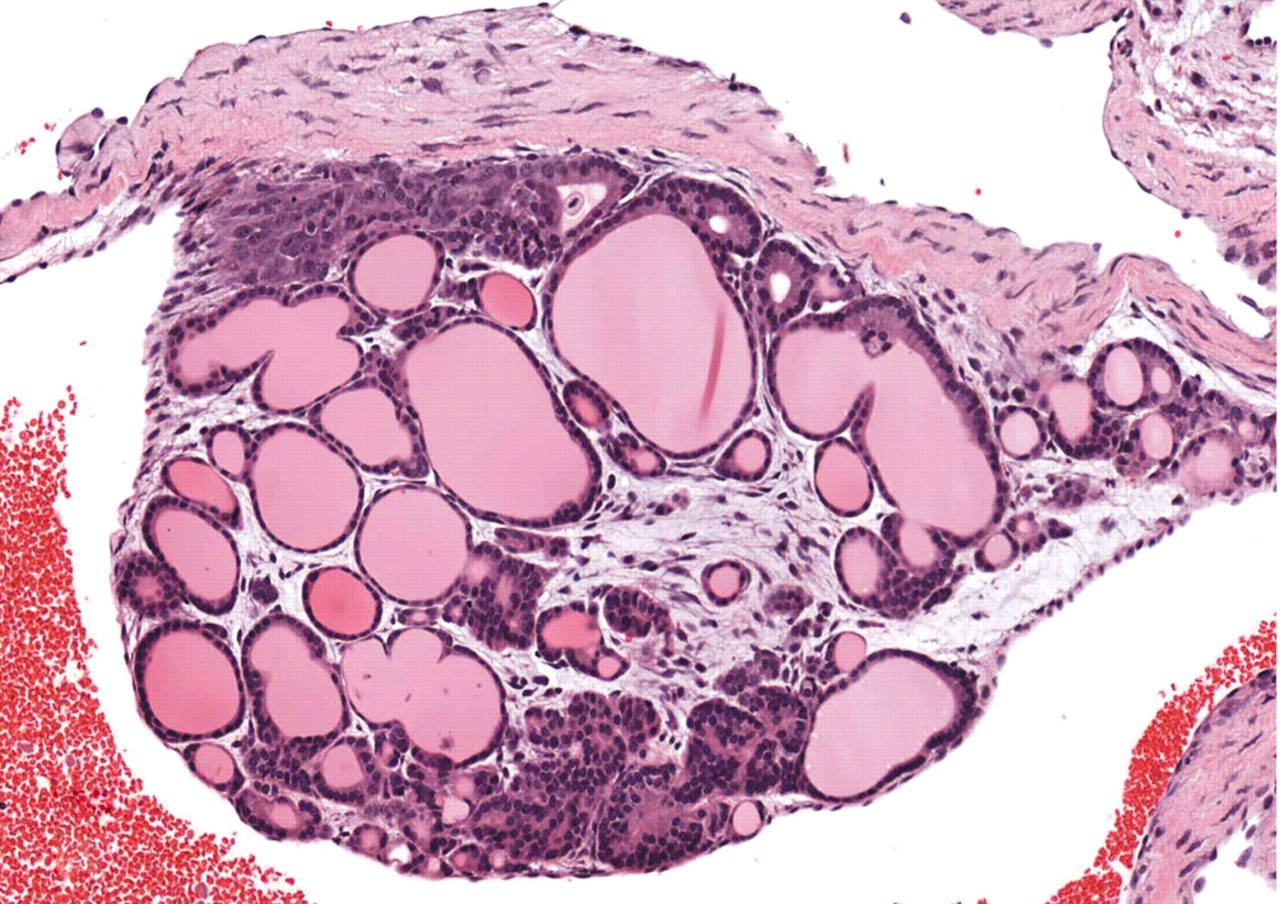
The aortic valve is expanded by colloid-filled follicles that are overlain by normal endothelium. 200× (HE).
The thyroid gland was normally located at necropsy and histologically unremarkable.
Discussion
Whilst ectopic thyroid tissue, located anywhere between the base of the tongue and the diaphragm, is a common occurrence in humans and animals, intracardiac ectopic thyroid tissue is rare in all species (Hunt 1963; Naan et al. 2006; Capen 2007; Wu, Zhou, and Wang 2009).
In humans, ectopic thyroid tissue is present in approximately 10% of individuals, and 90% of ectopic thyroid tissue is sublingual (Sauk 1970). Twenty-one cases of intracardiac thyroid tissue have been reported, of which nineteen involved the right interventricular septum, one affected the left interventricular septum, and one affected the left ventricular outflow tract (Wu, Zhou, and Wang 2009).
In dogs, ectopic thyroid is present in approximately 50% of animals, predominantly intrapericardially, around the heart base (Capen 2007). All reported cases of intracardiac thyroid tissue in the dog have been neoplastic, involving the interventricular septum with protrusion into the right ventricle (Almes, Heaney, and Andrews 2008; Bracha et al. 2009).
In nonhuman primates (cynomologous monkeys and marmosets), subepicardial, heart base, and pericardial ectopic thyroid tissue is identified in approximately 0.2% of individuals (Chamanza et al. 2006). Ectopic thyroid tissue is similarly localized in the mouse, predominantly in the adipose tissue around the heart base. In the mouse, occurrence is variable, present in 0.3% BALB/c mice but not identified in the Strong A mouse (Hunt 1963). Although the actual incidence of ectopic thyroid tissue is not reported in the rat, it has been identified along the midline of the neck and thoracic cavity, particularly within the thymus and around the intrathoracic aorta (Anderson and Capen 1978). In rats and mice, intracardiac ectopic thyroid is not reported.
During embryofetal development, the thyroid gland relocates from its site of induction to a species-specific position in the cervical mesenchyme.
The early development and descent of the thyroid is intimately anatomically related to the heart. In vertebrates, the thyroid gland forms from the thyroid diverticulum—an evagination of endodermal epithelium on the ventral floor of the foregut between the first two pharyngeal pouches. The developing thyroid remains attached to the tongue at the base of the pharyngeal floor by the thyroglossal duct, descending to the dorsal aspect of the aortic sac, the bulbis cordis and truncus arteriosis, which later develop into the interventricular septum, the aorta, and the pulmonary artery. The thyroid then relocates cranially, to a species-specific location in the neck, and there is regression of the thyroglossal duct. Parafollicular (C cells) arise from migrating neural crest cells from the ultimobranchial body, which fuses with, and disseminates into, the thyroid gland (Noden and De Lahunta 1985; Alt et al. 2006).
The occurrence of intracardiac ectopic thyroid tissue is likely to be the result of multiple developmental events in the embryo. The intimate and prolonged association of the thyroid diverticulum with the aortic sac may lead to incorporation of thyroid remnants into the mediastinum and associated tissues, with seeding of the thyroid epithelium into what will later become the interventricular septum. In contrast, failure to associate fully with the aortic sac leads to abnormal descent of the thyroid and the development of lingual ectopic thyroid tissue (Lewis and Walley 1998). Parafollicular cells are generally not identified in ectopic thyroid tissue as fusion of the ultimobranchial body and the developing thyroid gland occurs after seeding of ectopic thyroid tissue (Noden and De Lahunta 1985).
Normal development of the thyroid and the interventricular septum is also dependent on the normal migration of neural crest cells; therefore, defects in this process may be responsible, in part, for the occurrence of thyroid tissue within the septum. This is exemplified by abnormalities of neural crest development (such as DiGeorge syndrome in humans), in which there are defects of cardiac septation, thyroid, parathyroid, and thymic dysgenesis (Ruberg et al. 2006).
In addition, the development of mature thyroid tissue requires environmental support in the form of local expression of thyroid transcription factors, such as PAX-8 and TTF-1 (Christophe 2004).
Whilst ectopic thyroid tissue is a frequent occurrence in humans and animals, ectopic thyroid tissue within the heart is rare in all species. Benign intracardiac ectopic thyroid tissue is often only identified at postmortem examination. However, rests of ectopic thyroid tissue is susceptible to the same disease processes as the thyroid gland–hyperplastic, and neoplastic ectopic thyroid tissue within the heart may not only cause clinical signs of endocrine disruption and malignant disease but may also impact cardiac function by obstruction of ventricular outflow and the induction of ventricular arrhythmias (Richmond et al. 1990; Olson, Bulmer, and Heaney 2007; Almes, Heaney, and Andrews 2008; Wu, Zhou, and Wang 2009).
This is the first report of intracardiac, ectopic thyroid tissue in the rat and the first report of ectopic thyroid tissue within a heart valve in any species. The occurrence of remnants of mature thyroid tissue at ectopic locations are also of interest, particularly in laboratory animals, because of possible interference with thyroid function investigation post-thyroidectomy, and the confounding effects of xenobiotics on thyroid tissue in short- or long-term toxicity studies.
Footnotes
All authors are employed by AstraZeneca.
